Early Predictors of Objective Response in Patients with Hepatocellular Carcinoma Undergoing Lenvatinib Treatment
Abstract
1. Introduction
2. Results
2.1. Patient Characteristics
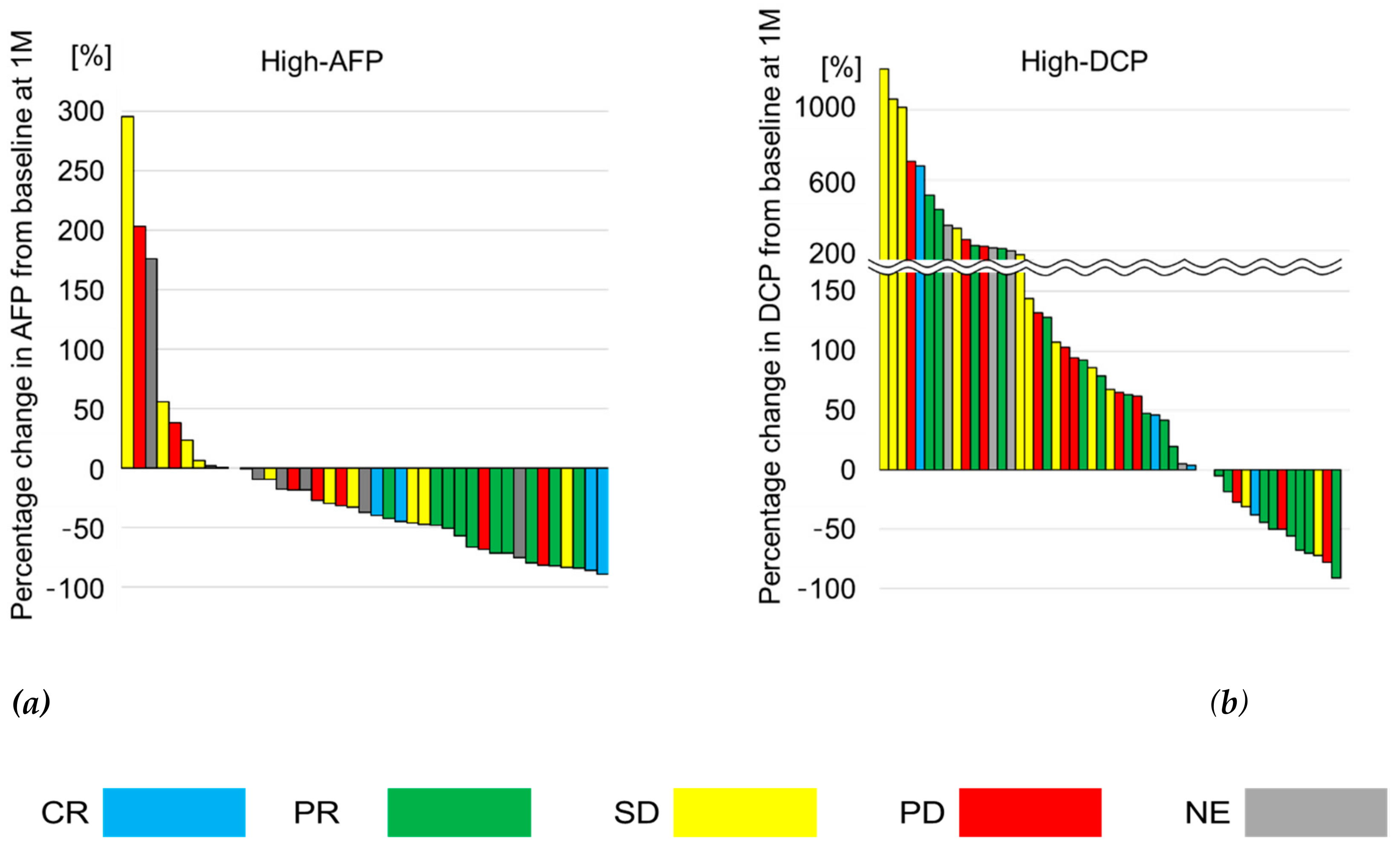
2.2. Relationship between Treatment Response and Response of Tumor Markers
2.3. Predictors of Objective Response
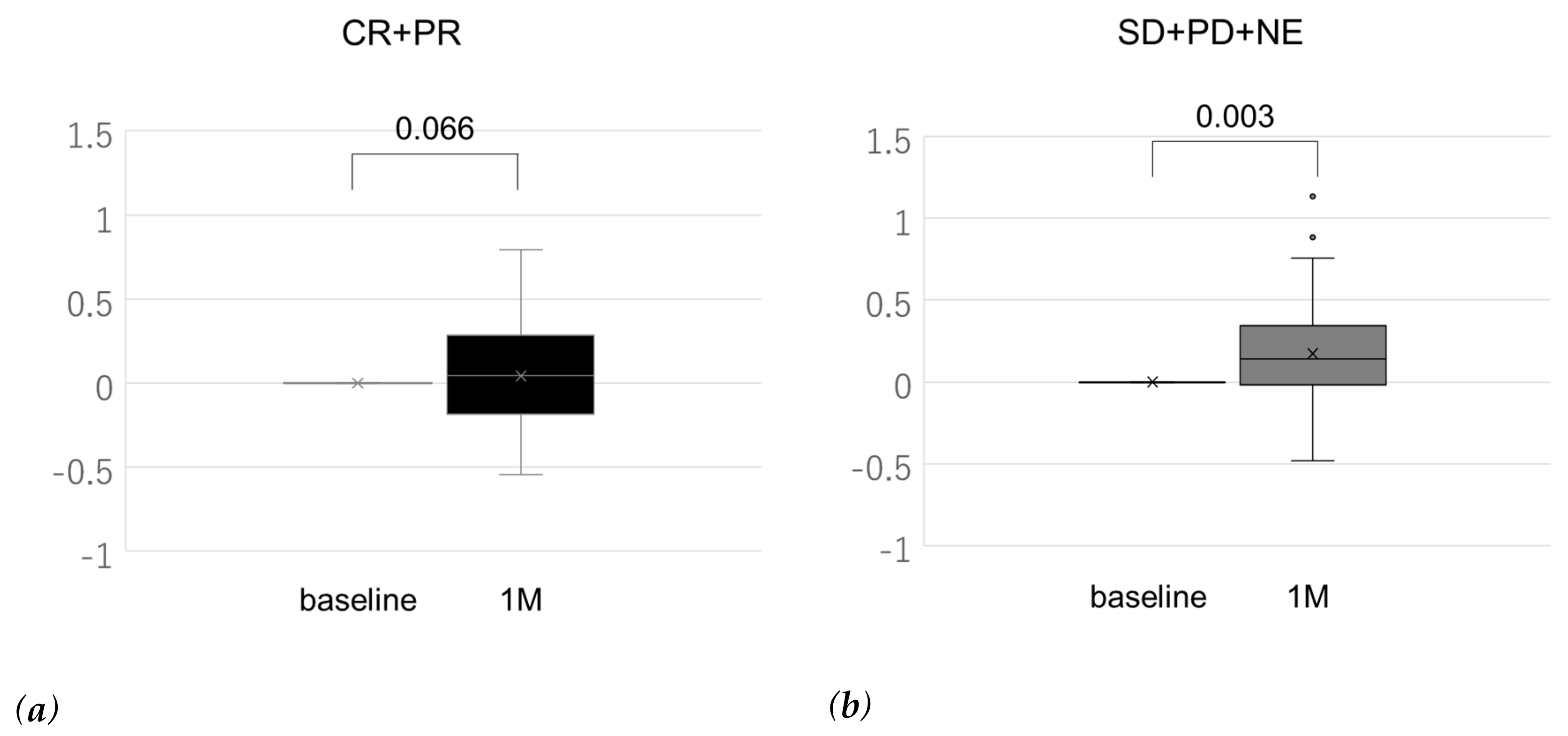
2.4. Change in ALBI Score
3. Discussion
4. Materials and Methods
4.1. Patients
4.2. Treatment Protocol
4.3. Evaluation of Treatment Response, Tumor Markers, and Liver Function
4.4. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Kudo, M. Lenvatinib May Drastically Change the Treatment Landscape of Hepatocellular Carcinoma. Liver Cancer 2018, 7, 1–19. [Google Scholar] [CrossRef]
- Kudo, M.; Finn, R.S.; Qin, S.; Han, K.H.; Ikeda, K.; Piscaglia, F.; Baron, A.; Park, J.W.; Han, G.; Jassem, J.; et al. Lenvatinib versus sorafenib in first-line treatment of patients with unresectable hepatocellular carcinoma: A randomised phase 3 non-inferiority trial. Lancet 2018, 391, 1163–1173. [Google Scholar] [CrossRef]
- Marrero, J.A.; Kulik, L.M.; Sirlin, C.B.; Zhu, A.X.; Finn, R.S.; Abecassis, M.M.; Roberts, L.R.; Heimbach, J.K. Diagnosis, Staging, and Management of Hepatocellular Carcinoma: 2018 Practice Guidance by the American Association for the Study of Liver Diseases. Hepatology 2018, 68, 723–750. [Google Scholar] [CrossRef] [PubMed]
- European Association for the Study of the Liver. Clinical Practice Guidelines: Management of hepatocellular carcinoma. J. Hepatol. 2018, 69, 182–236. [Google Scholar] [CrossRef] [PubMed]
- Japan Society of Hepatology. Clinical Practice Guidelines for Hepatocellular Carcinoma, 2017 version; Kanehara: Tokyo, Japan, 2017. [Google Scholar]
- Korean Liver Cancer Association (KLCA), National Cancer Center (NCC). 2018 Korean Liver Cancer Association-National Cancer Center Korea Practice Guidelines for the Management of Hepatocellular Carcinoma. Korean J. Radiol. 2019, 20, 1042–1113. [Google Scholar] [CrossRef]
- Finn, R.S.; Merle, P.; Granito, A.; Huang, Y.H.; Bodoky, G.; Pracht, M.; Yokosuka, O.; Rosmorduc, O.; Gerolami, R.; Caparello, C.; et al. Outcomes of sequential treatment with sorafenib followed by regorafenib for HCC: Additional analyses from the phase III RESORCE trial. J. Hepatol. 2018, 69, 353–358. [Google Scholar] [CrossRef]
- Alsina, A.; Kudo, M.; Vogel, A.; Cheng, A.L.; Tak, W.Y.; Ryoo, B.-Y.; Evans, T.R.J.; López López, C.; Daniele, B.; Misir, S.; et al. Effects of Subsequent Systemic Anticancer Medication Following First-Line Lenvatinib: A Post Hoc Responder Analysis from the Phase 3 REFLECT Study in Unresectable Hepatocellular Carcinoma. Liver Cancer 2020, 9, 93–104. [Google Scholar] [CrossRef]
- Takahashi, A.; Moriguchi, M.; Seko, Y.; Ishikawa, H.; Yo, T.; Kimura, H.; Fujii, H.; Shima, T.; Mitsumoto, Y.; Ishiba, H.; et al. Impact of Relative Dose Intensity of Early-phase Lenvatinib Treatment on Therapeutic Response in Hepatocellular Carcinoma. Anticancer Res. 2019, 39, 5149–5156. [Google Scholar] [CrossRef]
- Kodama, K.; Kawaoka, T.; Namba, M.; Uchikawa, S.; Ohya, K.; Morio, K.; Nakahara, T.; Murakami, E.; Yamauchi, M.; Hiramatsu, A.; et al. Correlation between Early Tumor Marker Response and Imaging Response in Patients with Advanced Hepatocellular Carcinoma Treated with Lenvatinib. Oncology 2019, 97, 75–81. [Google Scholar] [CrossRef]
- Park, H.; Park, J.Y. Clinical significance of AFP and PIVKA-II responses for monitoring treatment outcomes and predicting prognosis in patients with hepatocellular carcinoma. Biomed. Res. Int. 2013, 2013, 310427. [Google Scholar] [CrossRef]
- Vora, S.R.; Zheng, H.; Stadler, Z.K.; Fuchs, C.S.; Zhu, A.X. Serum alpha-fetoprotein response as a surrogate for clinical outcome in patients receiving systemic therapy for advanced hepatocellular carcinoma. Oncologist 2009, 14, 717–725. [Google Scholar] [CrossRef]
- Lee, M.H.; Kim, S.U.; Kim, D.Y.; Ahn, S.H.; Choi, E.H.; Lee, K.H.; Lee, D.Y.; Seong, J.; Han, K.H.; Chon, C.Y.; et al. Early on-treatment predictions of clinical outcomes using alpha-fetoprotein and des-gamma-carboxy prothrombin responses in patients with advanced hepatocellular carcinoma. J. Gastroenterol. Hepatol. 2012, 27, 313–322. [Google Scholar] [CrossRef] [PubMed]
- Saeki, I.; Yamasaki, T.; Tanabe, N.; Iwamoto, T.; Matsumoto, T.; Urata, Y.; Hidaka, I.; Ishikawa, T.; Takami, T.; Yamamoto, N.; et al. A new therapeutic assessment score for advanced hepatocellular carcinoma patients receiving hepatic arterial infusion chemotherapy. PLoS ONE 2015, 10, e0126649. [Google Scholar] [CrossRef] [PubMed]
- Johnson, P.J.; Berhane, S.; Kagebayashi, C.; Satomura, S.; Teng, M.; Reeves, H.L.; O’Beirne, J.; Fox, R.; Skowronska, A.; Palmer, D.; et al. Assessment of liver function in patients with hepatocellular carcinoma: A new evidence-based approach-the ALBI grade. J. Clin. Oncol. 2015, 33, 550–558. [Google Scholar] [CrossRef] [PubMed]
- Forner, A.; Reig, M.; Bruix, J. Hepatocellular carcinoma. Lancet 2018, 391, 1301–1314. [Google Scholar] [CrossRef]
- Kudo, M. A New Treatment Option for Intermediate-Stage Hepatocellular Carcinoma with High Tumor Burden: Initial Lenvatinib Therapy with Subsequent Selective TACE. Liver Cancer 2019, 8, 299–311. [Google Scholar] [CrossRef]
- Omata, M.; Cheng, A.L.; Kokudo, N.; Kudo, M.; Lee, J.M.; Jia, J.; Tateishi, R.; Han, K.H.; Chawla, Y.K.; Shiina, S.; et al. Asia-Pacific clinical practice guidelines on the management of hepatocellular carcinoma: A 2017 update. Hepatol. Int. 2017, 11, 317–370. [Google Scholar] [CrossRef]
- Murata, K.; Suzuki, H.; Okano, H.; Oyamada, T.; Yasuda, Y.; Sakamoto, A. Hypoxia-induced des-gamma-carboxy prothrombin production in hepatocellular carcinoma. Int. J. Oncol. 2010, 36, 161–170. [Google Scholar]
- Ueshima, K.; Kudo, M.; Takita, M.; Nagai, T.; Tatsumi, C.; Ueda, T.; Kitai, S.; Ishikawa, E.; Yada, N.; Inoue, T.; et al. Des-γ-carboxyprothrombin may be a promising biomarker to determine the therapeutic efficacy of sorafenib for hepatocellular carcinoma. Dig. Dis. 2011, 29, 321–325. [Google Scholar] [CrossRef]
- Kuzuya, T.; Asahina, Y.; Tsuchiya, K.; Tanaka, K.; Suzuki, Y.; Hoshioka, T.; Tamaki, S.; Kato, T.; Yasui, Y.; Hosokawa, T.; et al. Early decrease in α-fetoprotein, but not des-γ-carboxy prothrombin, predicts sorafenib efficacy in patients with advanced hepatocellular carcinoma. Oncology 2011, 81, 251–258. [Google Scholar] [CrossRef]
- Hiraoka, A.; Michitaka, K.; Kumada, T.; Izumi, N.; Kadoya, M.; Kokudo, N.; Kubo, S.; Matsuyama, Y.; Nakashima, O.; Sakamoto, M.; et al. Validation and Potential of Albumin-Bilirubin Grade and Prognostication in a Nationwide Survey of 46,681 Hepatocellular Carcinoma Patients in Japan: The Need for a More Detailed Evaluation of Hepatic Function. Liver Cancer 2017, 6, 325–336. [Google Scholar] [CrossRef] [PubMed]
- Ueshima, K.; Nishida, N.; Hagiwara, S.; Aoki, T.; Minami, T.; Chishina, H.; Takita, M.; Minami, Y.; Ida, H.; Takenaka, M.; et al. Impact of Baseline ALBI Grade on the Outcomes of Hepatocellular Carcinoma Patients Treated with Lenvatinib: A Multicenter Study. Cancers 2019, 11, 952. [Google Scholar] [CrossRef] [PubMed]
- Hiraoka, A.; Kumada, T.; Atsukawa, M.; Hirooka, M.; Tsuji, K.; Ishikawa, T.; Takaguchi, K.; Kariyama, K.; Itobayashi, E.; Tajiri, K.; et al. Early Relative Change in Hepatic Function with Lenvatinib for Unresectable Hepatocellular Carcinoma. Oncology 2019, 97, 334–340. [Google Scholar] [CrossRef] [PubMed]
- Hiraoka, A.; Kumada, T.; Atsukawa, M.; Hirooka, M.; Tsuji, K.; Ishikawa, T.; Takaguchi, K.; Kariyama, K.; Itobayashi, E.; Tajiri, K.; et al. Important Clinical Factors in Sequential Therapy Including Lenvatinib against Unresectable Hepatocellular Carcinoma. Oncology 2019, 97, 277–285. [Google Scholar] [CrossRef]
- Kudo, M. Objective Response by mRECIST is an Independent Prognostic Factor of Overall Survival in Systemic Therapy for Hepatocellular Carcinoma. Liver Cancer 2019, 8, 73–77. [Google Scholar] [CrossRef]
- Lencioni, R.; Llovet, J.M. Modified RECIST (mRECIST) assessment for hepatocellular carcinoma. Semin. Liver Dis. 2010, 30, 52–60. [Google Scholar] [CrossRef]

| Factor | Total |
|---|---|
| Age | 72.8 ± 9.1 |
| Sex male/female | 50/20 |
| BW (kg) | 57.8 ± 12.1 |
| Etiology C/B/Alc/N | 31/14/7/18 |
| Child–Pugh class A/B | 57/13 |
| ALBI score | −2.27 ± 0.53 |
| ALBI grade 1/2/3 | 18/48/4 |
| PS 0/1/2 | 58/11/1 |
| BCLC a A/B/C | 3/29/38 |
| MVI absence/presence | 55/15 |
| EHS absence/presence | 51/19 |
| Tumor number | 4 (1–8) |
| Tumor size (mm) | 29.0 (20.0–45.3) |
| AFP (ng/mL) | 14.5 (3.9–603.6) |
| DCP (mAU/mL) | 271.0 (35.6–2158.5) |
| Prior treatment (+/−) | 61/9 |
| Prior treatment of MTA (+/−): SORA/REGO | 6/64:6/3 |
| Response a | CR | PR | SD | PD | NE | ORR | DCR |
|---|---|---|---|---|---|---|---|
| Total (n = 70) | 6 | 23 | 19 | 15 | 7 | 41.4 % | 68.6 % |
| High-AFP group (AFP ≥ 10ng/mL) | |||||||
| Total (n = 41) | 3 | 11 | 11 | 10 | 6 | 34.2% | 61.0% |
| AFP responder b (n = 19) | 2 | 11 | 3 | 2 | 1 | 68.4%]* | 84.2%]+ |
| AFP non-responder (n = 22) | 1 | 0 | 8 | 8 | 5 | 7.1% | 36.0% |
| High-DCP group (DCP ≥ 40ng/mL) | |||||||
| Total (n = 51) | 4 | 20 | 11 | 12 | 4 | 47.1% | 68.6% |
| DCP responder b (n = 9) | 0 | 6 | 1 | 2 | 0 | 66.7% | 78.8% |
| DCP non-responder (n = 42) | 4 | 14 | 10 | 10 | 4 | 42.9% | 66.7% |
| Factors | Univaritate Analysis | Multivariate Analysis | ||||
|---|---|---|---|---|---|---|
| Odds Ratio | 95% CI | p-Value | Odds ratio | 95% CI | p-Value | |
| Sex (M/F) | 4.125 | 0.767–22.184 | 0.099 | 4.825 | 0.560–41.587 | 0.152 |
| Age (<65 years/≥65 years) | 2.857 | 0.736–11.086 | 0.129 | |||
| BW (<60 kg/≥60 kg) | 3.000 | 0.549–16.379 | 0.245 | |||
| PS (0/1–) | 2.100 | 0.374–11.807 | 0.400 | |||
| ALBI (<−2.44/≥−2.44) | 3.167 | 0.827–12.126 | 0.093 | 2.714 | 0.371–19.854 | 0.325 |
| BCLC a A,B/C | 1.700 | 0.460–6.280 | 0.426 | |||
| Tumor size (mm) (<30/≥30) | 1.938 | 0.513–7.319 | 0.329 | |||
| Tumor number (<4/≥4) | 1.091 | 0.295–4.033 | 0.896 | |||
| EHS absence/presence | 2.521 | 0.568–11.181 | 0.224 | |||
| MVI absence/presence | 1.053 | 0.254–4.371 | 0.944 | |||
| AFP (ng/mL) (<200/≥200) | 0.808 | 0.220–2.964 | 0.748 | |||
| DCP (mAU/mL) (<40/≥40) | 0.556 | 0.096–3.207 | 0.511 | |||
| AFP response b | 45.500 | 4.907–421.933 | <0.001 | 51.389 | 4.888–540.281 | 0.001 |
| DCP response b | 0.917 | 0.146–5.757 | 0.926 | |||
| Factors | Univaritate Analysis | Multivariate Analysis | |||||
|---|---|---|---|---|---|---|---|
| Odds Ratio | 95% CI | p-Value | Odds Ratio | 95% CI | p-Value | ||
| Sex (M/F) | 0.750 | 0.135–4.165 | 0.742 | ||||
| Age (<65 years/≥65 years) | 0.655 | 0.134–3.187 | 0.600 | ||||
| BW (<60 kg/≥60 kg) | 0.857 | 0.198–3.713 | 0.837 | ||||
| PS (0/1–) | 0.500 | 0.040–6.128 | 0.590 | ||||
| ALBI (<−2.44/≥−2.44) | 5.500 | 1.065–28.416 | 0.042 | 6.866 | 1.098–42.923 | 0.039 | |
| BCLC a A,B/C | 0.656 | −0.151–2.843 | 0.573 | ||||
| Tumor size (mm) (<30/≥30) | 0.667 | 0.153–2.903 | 0.589 | ||||
| Tumor number (<4/≥4) | 3.667 | 0.771–17.429 | 0.102 | ||||
| EHS absence/presence | 0.667 | 0.094–4.373 | 0.685 | ||||
| MVI absence/presence | 0.500 | 0.040–6.218 | 0.590 | ||||
| DCP (mAU/mL) (<40/≥40) | 0.256 | 0.048–1.282 | 0.098 | 0.191 | 0.029–1.266 | 0.086 | |
| AFP response b | 1.333 | 0.240–7.405 | 0.742 | ||||
| DCP response b | - | - | - | ||||
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Saeki, I.; Yamasaki, T.; Yamashita, S.; Hanazono, T.; Urata, Y.; Furutani, T.; Yokoyama, Y.; Oishi, T.; Maeda, M.; Kimura, T.; et al. Early Predictors of Objective Response in Patients with Hepatocellular Carcinoma Undergoing Lenvatinib Treatment. Cancers 2020, 12, 779. https://doi.org/10.3390/cancers12040779
Saeki I, Yamasaki T, Yamashita S, Hanazono T, Urata Y, Furutani T, Yokoyama Y, Oishi T, Maeda M, Kimura T, et al. Early Predictors of Objective Response in Patients with Hepatocellular Carcinoma Undergoing Lenvatinib Treatment. Cancers. 2020; 12(4):779. https://doi.org/10.3390/cancers12040779
Chicago/Turabian StyleSaeki, Issei, Takahiro Yamasaki, Satoyoshi Yamashita, Tadasuke Hanazono, Yohei Urata, Takakazu Furutani, Yuichiro Yokoyama, Toshiyuki Oishi, Masaki Maeda, Teruaki Kimura, and et al. 2020. "Early Predictors of Objective Response in Patients with Hepatocellular Carcinoma Undergoing Lenvatinib Treatment" Cancers 12, no. 4: 779. https://doi.org/10.3390/cancers12040779
APA StyleSaeki, I., Yamasaki, T., Yamashita, S., Hanazono, T., Urata, Y., Furutani, T., Yokoyama, Y., Oishi, T., Maeda, M., Kimura, T., Kotoh, Y., Sasaki, R., Miyaji, T., Oono, T., Aibe, Y., Hisanaga, T., Iwamoto, T., Matsumoto, T., Hidaka, I., ... Sakaida, I. (2020). Early Predictors of Objective Response in Patients with Hepatocellular Carcinoma Undergoing Lenvatinib Treatment. Cancers, 12(4), 779. https://doi.org/10.3390/cancers12040779






