FET PET Radiomics for Differentiating Pseudoprogression from Early Tumor Progression in Glioma Patients Post-Chemoradiation
Abstract
:Simple Summary
Abstract
1. Introduction
2. Patients and Methods
2.1. Patients
2.2. Ethics Statement
2.3. Determination of the IDH Genotype and MGMT Promoter Methylation Status
2.4. Diagnosis of Pseudoprogression and Early Tumor Progression
2.5. FET PET Imaging
2.6. Evaluation of Static and Dynamic FET PET Parameters
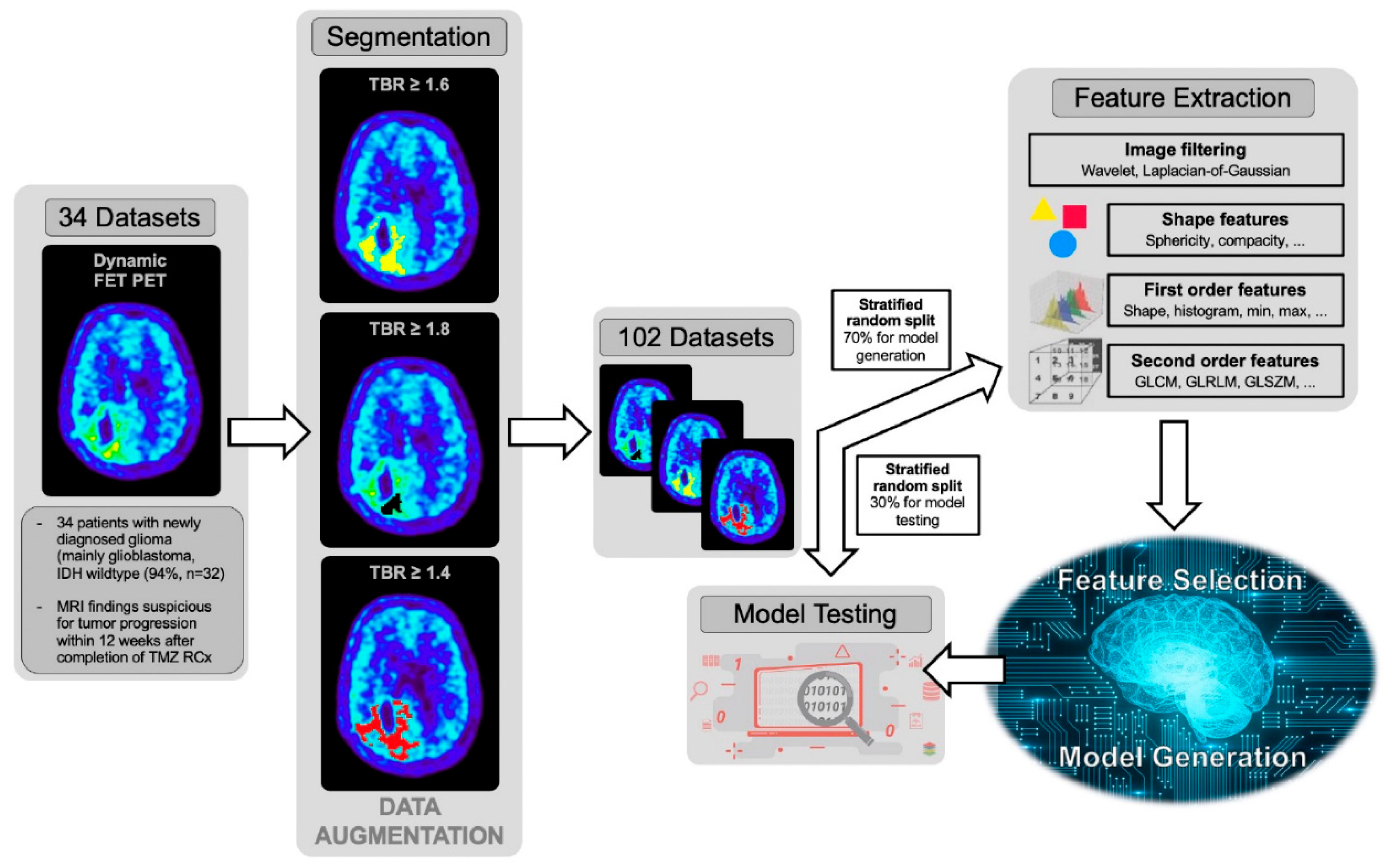
2.7. Image Pre-Processing and Radiomics Feature Extraction
2.8. Feature Selection
2.9. Model Generation and Validation
2.10. Model Testing
2.11. Statistical Evaluation
3. Results
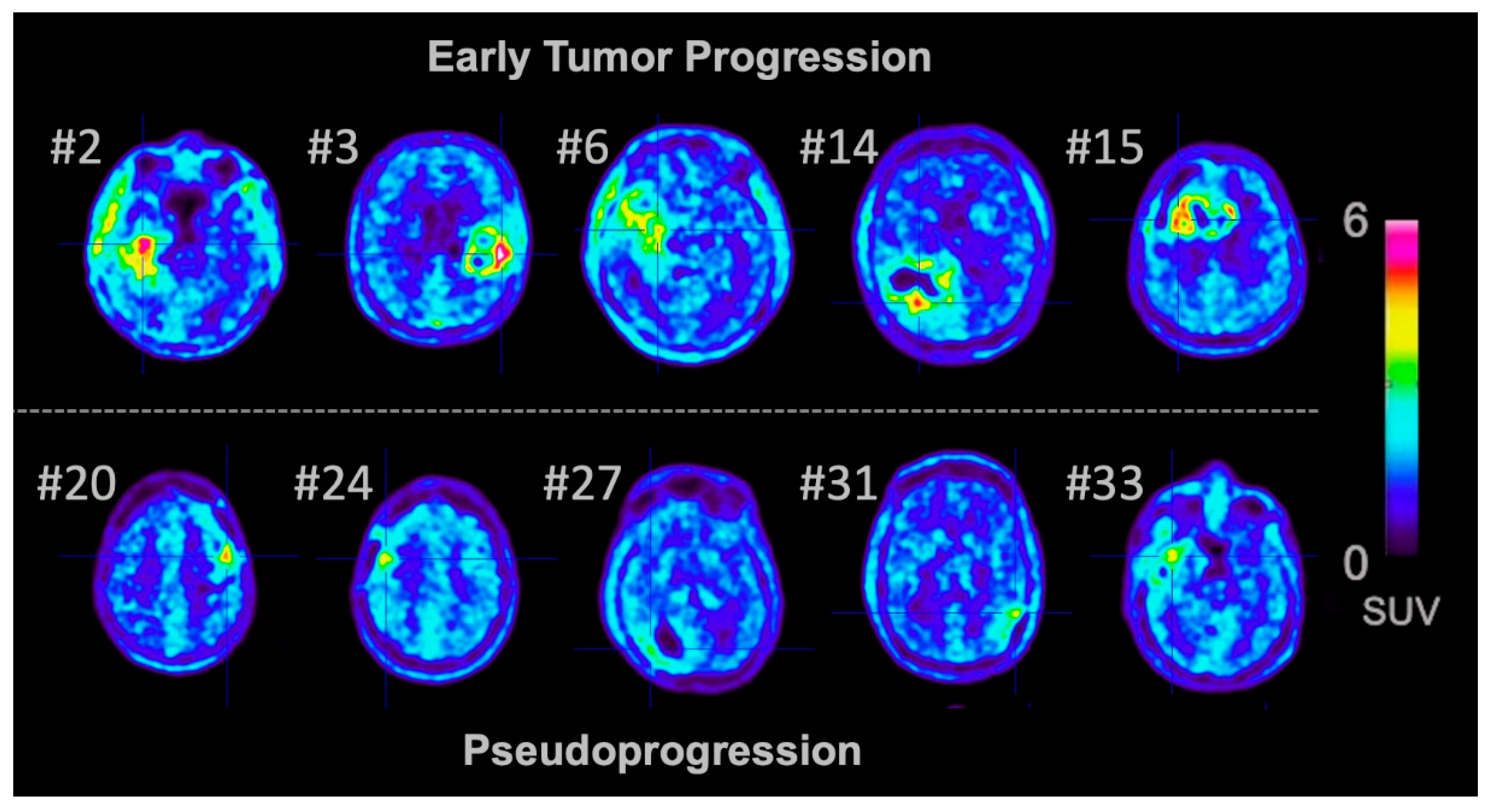
3.1. Pseudoprogression and Early Tumor Progression
3.2. Group Comparison of Static and Dynamic FET PET Parameters
3.3. ROC Analysis of Static and Dynamic FET PET Parameters
3.4. Performance of Machine Learning Models in the Training Dataset
3.5. Performance of the Machine Learning Model in the Test Dataset
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Taal, W.; Brandsma, D.; de Bruin, H.G.; Bromberg, J.E.; Swaak-Kragten, A.T.; Smitt, P.A.; van Es, C.A.; van den Bent, M.J. Incidence of early pseudo-progression in a cohort of malignant glioma patients treated with chemoirradiation with temozolomide. Cancer 2008, 113, 405–410. [Google Scholar] [CrossRef] [PubMed]
- Wen, P.Y.; Macdonald, D.R.; Reardon, D.A.; Cloughesy, T.F.; Sorensen, A.G.; Galanis, E.; Degroot, J.; Wick, W.; Gilbert, M.R.; Lassman, A.B.; et al. Updated response assessment criteria for high-grade gliomas: Response assessment in neuro-oncology working group. J. Clin. Oncol. 2010, 28, 1963–1972. [Google Scholar] [CrossRef] [PubMed]
- Brandsma, D.; Stalpers, L.; Taal, W.; Sminia, P.; van den Bent, M.J. Clinical features, mechanisms, and management of pseudoprogression in malignant gliomas. Lancet Oncol. 2008, 9, 453–461. [Google Scholar] [CrossRef]
- Brandsma, D.; van den Bent, M.J. Pseudoprogression and pseudoresponse in the treatment of gliomas. Curr. Opin. Neurol. 2009, 22, 633–638. [Google Scholar] [CrossRef]
- Galldiks, N.; Kocher, M.; Langen, K.J. Pseudoprogression after glioma therapy: An update. Expert Rev. Neurother. 2017, 17, 1109–1115. [Google Scholar] [CrossRef] [PubMed]
- Langen, K.J.; Galldiks, N.; Hattingen, E.; Shah, N.J. Advances in neuro-oncology imaging. Nat. Rev. Neurol. 2017, 13, 279–289. [Google Scholar] [CrossRef] [PubMed]
- Kumar, A.J.; Leeds, N.E.; Fuller, G.N.; Van Tassel, P.; Maor, M.H.; Sawaya, R.E.; Levin, V.A. Malignant gliomas: MR imaging spectrum of radiation therapy- and chemotherapy-induced necrosis of the brain after treatment. Radiology 2000, 217, 377–384. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Thust, S.C.; van den Bent, M.J.; Smits, M. Pseudoprogression of brain tumors. J. Magn. Reson. Imaging 2018, 48, 571–589. [Google Scholar] [CrossRef] [Green Version]
- van Dijken, B.R.J.; van Laar, P.J.; Holtman, G.A.; van der Hoorn, A. Diagnostic accuracy of magnetic resonance imaging techniques for treatment response evaluation in patients with high-grade glioma, a systematic review and meta-analysis. Eur. Radiol. 2017, 27, 4129–4144. [Google Scholar] [CrossRef] [Green Version]
- Lerche, C.; Radomski, T.; Lohmann, P.; Regio-Brambilla, C.; Tellmann, L.; Scheins, J.; Rota Kops, E.; Herzog, H.; Langen, K.-J.; Shah, N.J. Clinical applicability of a mathematical model for FET PET uptake kinetics in brain tumor patients. Eur. J. Nucl. Med. Mol. Imaging 2019, 46, S231–S232. [Google Scholar]
- Werner, J.M.; Stoffels, G.; Lichtenstein, T.; Borggrefe, J.; Lohmann, P.; Ceccon, G.; Shah, N.J.; Fink, G.R.; Langen, K.J.; Kabbasch, C.; et al. Differentiation of treatment-related changes from tumour progression: A direct comparison between dynamic FET PET and ADC values obtained from DWI MRI. Eur. J. Nucl. Med. Mol. Imaging 2019, 46, 1889–1901. [Google Scholar] [CrossRef] [PubMed]
- Galldiks, N.; Dunkl, V.; Stoffels, G.; Hutterer, M.; Rapp, M.; Sabel, M.; Reifenberger, G.; Kebir, S.; Dorn, F.; Blau, T.; et al. Diagnosis of pseudoprogression in patients with glioblastoma using O-(2-[18F]fluoroethyl)-L-tyrosine PET. Eur. J. Nucl. Med. Mol. Imaging 2015, 42, 685–695. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kebir, S.; Fimmers, R.; Galldiks, N.; Schafer, N.; Mack, F.; Schaub, C.; Stuplich, M.; Niessen, M.; Tzaridis, T.; Simon, M.; et al. Late Pseudoprogression in Glioblastoma: Diagnostic Value of Dynamic O-(2-[18F]fluoroethyl)-L-Tyrosine PET. Clin. Cancer Res. 2016, 22, 2190–2196. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gillies, R.J.; Kinahan, P.E.; Hricak, H. Radiomics: Images Are More than Pictures, They Are Data. Radiology 2016, 278, 563–577. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Shofty, B.; Artzi, M.; Ben Bashat, D.; Liberman, G.; Haim, O.; Kashanian, A.; Bokstein, F.; Blumenthal, D.T.; Ram, Z.; Shahar, T. MRI radiomics analysis of molecular alterations in low-grade gliomas. Int J. Comput Assist. Radiol Surg. 2018, 13, 563–571. [Google Scholar] [CrossRef] [PubMed]
- Zhou, H.; Chang, K.; Bai, H.X.; Xiao, B.; Su, C.; Bi, W.L.; Zhang, P.J.; Senders, J.T.; Vallieres, M.; Kavouridis, V.K.; et al. Machine learning reveals multimodal MRI patterns predictive of isocitrate dehydrogenase and 1p/19q status in diffuse low- and high-grade gliomas. J. Neurooncol. 2019, 142, 299–307. [Google Scholar] [CrossRef]
- Lohmann, P.; Lerche, C.; Bauer, E.K.; Steger, J.; Stoffels, G.; Blau, T.; Dunkl, V.; Kocher, M.; Viswanathan, S.; Filss, C.P.; et al. Predicting IDH genotype in gliomas using FET PET radiomics. Sci. Rep. 2018, 8, 13328. [Google Scholar] [CrossRef]
- Li, Z.C.; Bai, H.; Sun, Q.; Li, Q.; Liu, L.; Zou, Y.; Chen, Y.; Liang, C.; Zheng, H. Multiregional radiomics features from multiparametric MRI for prediction of MGMT methylation status in glioblastoma multiforme: A multicentre study. Eur. Radiol. 2018, 28, 3640–3650. [Google Scholar] [CrossRef]
- Xi, Y.B.; Guo, F.; Xu, Z.L.; Li, C.; Wei, W.; Tian, P.; Liu, T.T.; Liu, L.; Chen, G.; Ye, J.; et al. Radiomics signature: A potential biomarker for the prediction of MGMT promoter methylation in glioblastoma. J. Magn. Reson. Imaging 2018, 47, 1380–1387. [Google Scholar] [CrossRef]
- Kong, Z.; Lin, Y.; Jiang, C.; Li, L.; Liu, Z.; Wang, Y.; Dai, C.; Liu, D.; Qin, X.; Wang, Y.; et al. (18)F-FDG-PET-based Radiomics signature predicts MGMT promoter methylation status in primary diffuse glioma. Cancer Imaging 2019, 19, 58. [Google Scholar] [CrossRef] [Green Version]
- Kickingereder, P.; Burth, S.; Wick, A.; Gotz, M.; Eidel, O.; Schlemmer, H.P.; Maier-Hein, K.H.; Wick, W.; Bendszus, M.; Radbruch, A.; et al. Radiomic Profiling of Glioblastoma: Identifying an Imaging Predictor of Patient Survival with Improved Performance over Established Clinical and Radiologic Risk Models. Radiology 2016, 280, 880–889. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Yang, J.; Ho, A.; Jiang, W.; Logan, J.; Wang, X.; Brown, P.D.; McGovern, S.L.; Guha-Thakurta, N.; Ferguson, S.D.; et al. A predictive model for distinguishing radiation necrosis from tumour progression after gamma knife radiosurgery based on radiomic features from MR images. Eur. Radiol. 2018, 28, 2255–2263. [Google Scholar] [CrossRef] [PubMed]
- Peng, L.; Parekh, V.; Huang, P.; Lin, D.D.; Sheikh, K.; Baker, B.; Kirschbaum, T.; Silvestri, F.; Son, J.; Robinson, A.; et al. Distinguishing True Progression From Radionecrosis After Stereotactic Radiation Therapy for Brain Metastases With Machine Learning and Radiomics. Int. J. Radiat. Oncol. Biol. Phys. 2018, 102, 1236–1243. [Google Scholar] [CrossRef] [PubMed]
- Lohmann, P.; Kocher, M.; Ceccon, G.; Bauer, E.K.; Stoffels, G.; Viswanathan, S.; Ruge, M.I.; Neumaier, B.; Shah, N.J.; Fink, G.R.; et al. Combined FET PET/MRI radiomics differentiates radiation injury from recurrent brain metastasis. Neuroimage Clin. 2018, 20, 537–542. [Google Scholar] [CrossRef]
- Kim, J.Y.; Park, J.E.; Jo, Y.; Shim, W.H.; Nam, S.J.; Kim, J.H.; Yoo, R.E.; Choi, S.H.; Kim, H.S. Incorporating diffusion- and perfusion-weighted MRI into a radiomics model improves diagnostic performance for pseudoprogression in glioblastoma patients. Neuro Oncol. 2019, 21, 404–414. [Google Scholar] [CrossRef]
- Elshafeey, N.; Kotrotsou, A.; Hassan, A.; Elshafei, N.; Hassan, I.; Ahmed, S.; Abrol, S.; Agarwal, A.; El Salek, K.; Bergamaschi, S.; et al. Multicenter study demonstrates radiomic features derived from magnetic resonance perfusion images identify pseudoprogression in glioblastoma. Nat. Commun. 2019, 10, 3170. [Google Scholar] [CrossRef] [Green Version]
- Najjar, A.M.; Johnson, J.M.; Schellingerhout, D. The Emerging Role of Amino Acid PET in Neuro-Oncology. Bioengineering (Basel) 2018, 5, 104. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Galldiks, N.; Lohmann, P.; Albert, N.L.; Tonn, J.C.; Langen, K.J. Current status of PET imaging in neuro-oncology. Neurooncol. Adv. 2019, 1, vdz010. [Google Scholar] [CrossRef] [Green Version]
- Youland, R.S.; Kitange, G.J.; Peterson, T.E.; Pafundi, D.H.; Ramiscal, J.A.; Pokorny, J.L.; Giannini, C.; Laack, N.N.; Parney, I.F.; Lowe, V.J.; et al. The role of LAT1 in (18)F-DOPA uptake in malignant gliomas. J. Neurooncol. 2013, 111, 11–18. [Google Scholar] [CrossRef]
- Habermeier, A.; Graf, J.; Sandhofer, B.F.; Boissel, J.P.; Roesch, F.; Closs, E.I. System L amino acid transporter LAT1 accumulates O-(2-fluoroethyl)-L-tyrosine (FET). Amino Acids 2015, 47, 335–344. [Google Scholar] [CrossRef]
- Langen, K.J.; Hamacher, K.; Weckesser, M.; Floeth, F.; Stoffels, G.; Bauer, D.; Coenen, H.H.; Pauleit, D. O-(2-[18F]fluoroethyl)-L-tyrosine: Uptake mechanisms and clinical applications. Nucl. Med. Biol. 2006, 33, 287–294. [Google Scholar] [CrossRef] [PubMed]
- Okubo, S.; Zhen, H.N.; Kawai, N.; Nishiyama, Y.; Haba, R.; Tamiya, T. Correlation of L-methyl-11C-methionine (MET) uptake with L-type amino acid transporter 1 in human gliomas. J. Neurooncol. 2010, 99, 217–225. [Google Scholar] [CrossRef] [PubMed]
- Papin-Michault, C.; Bonnetaud, C.; Dufour, M.; Almairac, F.; Coutts, M.; Patouraux, S.; Virolle, T.; Darcourt, J.; Burel-Vandenbos, F. Study of LAT1 Expression in Brain Metastases: Towards a Better Understanding of the Results of Positron Emission Tomography Using Amino Acid Tracers. PLoS ONE 2016, 11, e0157139. [Google Scholar] [CrossRef] [Green Version]
- Galldiks, N.; Unterrainer, M.; Judov, N.; Stoffels, G.; Rapp, M.; Lohmann, P.; Vettermann, F.; Dunkl, V.; Suchorska, B.; Tonn, J.C.; et al. Photopenic defects on O-(2-[18F]-fluoroethyl)-L-tyrosine PET: Clinical relevance in glioma patients. Neuro Oncol. 2019, 21, 1331–1338. [Google Scholar] [CrossRef] [PubMed]
- Pirotte, B.; Goldman, S.; Massager, N.; David, P.; Wikler, D.; Vandesteene, A.; Salmon, I.; Brotchi, J.; Levivier, M. Comparison of 18F-FDG and 11C-methionine for PET-guided stereotactic brain biopsy of gliomas. J. Nucl. Med. 2004, 45, 1293–1298. [Google Scholar]
- Lohmann, P.; Stavrinou, P.; Lipke, K.; Bauer, E.K.; Ceccon, G.; Werner, J.M.; Neumaier, B.; Fink, G.R.; Shah, N.J.; Langen, K.J.; et al. FET PET reveals considerable spatial differences in tumour burden compared to conventional MRI in newly diagnosed glioblastoma. Eur. J. Nucl. Med. Mol. Imaging 2019, 46, 591–602. [Google Scholar] [CrossRef] [Green Version]
- Navarria, P.; Reggiori, G.; Pessina, F.; Ascolese, A.M.; Tomatis, S.; Mancosu, P.; Lobefalo, F.; Clerici, E.; Lopci, E.; Bizzi, A.; et al. Investigation on the role of integrated PET/MRI for target volume definition and radiotherapy planning in patients with high grade glioma. Radiother. Oncol. 2014, 112, 425–429. [Google Scholar] [CrossRef]
- Weber, D.C.; Zilli, T.; Buchegger, F.; Casanova, N.; Haller, G.; Rouzaud, M.; Nouet, P.; Dipasquale, G.; Ratib, O.; Zaidi, H.; et al. [(18)F]Fluoroethyltyrosine- positron emission tomography-guided radiotherapy for high-grade glioma. Radiat. Oncol. 2008, 3, 44. [Google Scholar] [CrossRef]
- Ceccon, G.; Lohmann, P.; Werner, J.M.; Tscherpel, C.; Dunkl, V.; Stoffels, G.; Rosen, J.; Rapp, M.; Sabel, M.; Herrlinger, U.; et al. Early treatment response assessment using (18)F-FET PET compared to contrast-enhanced MRI in glioma patients following adjuvant temozolomide chemotherapy. J. Nucl. Med. 2020. [Google Scholar] [CrossRef]
- Galldiks, N.; Dunkl, V.; Ceccon, G.; Tscherpel, C.; Stoffels, G.; Law, I.; Henriksen, O.M.; Muhic, A.; Poulsen, H.S.; Steger, J.; et al. Early treatment response evaluation using FET PET compared to MRI in glioblastoma patients at first progression treated with bevacizumab plus lomustine. Eur J. Nucl. Med. Mol. Imaging 2018, 45, 2377–2386. [Google Scholar] [CrossRef]
- Suchorska, B.; Jansen, N.L.; Linn, J.; Kretzschmar, H.; Janssen, H.; Eigenbrod, S.; Simon, M.; Popperl, G.; Kreth, F.W.; la Fougere, C.; et al. Biological tumor volume in 18FET-PET before radiochemotherapy correlates with survival in GBM. Neurology 2015, 84, 710–719. [Google Scholar] [CrossRef] [Green Version]
- Galldiks, N.; Ullrich, R.; Schroeter, M.; Fink, G.R.; Jacobs, A.H.; Kracht, L.W. Volumetry of [(11)C]-methionine PET uptake and MRI contrast enhancement in patients with recurrent glioblastoma multiforme. Eur J. Nucl. Med. Mol. Imaging 2010, 37, 84–92. [Google Scholar] [CrossRef] [Green Version]
- Albert, N.L.; Weller, M.; Suchorska, B.; Galldiks, N.; Soffietti, R.; Kim, M.M.; la Fougere, C.; Pope, W.; Law, I.; Arbizu, J.; et al. Response Assessment in Neuro-Oncology working group and European Association for Neuro-Oncology recommendations for the clinical use of PET imaging in gliomas. Neuro Oncol. 2016, 18, 1199–1208. [Google Scholar] [CrossRef] [PubMed]
- Galldiks, N.; Langen, K.J.; Albert, N.L.; Chamberlain, M.; Soffietti, R.; Kim, M.M.; Law, I.; Le Rhun, E.; Chang, S.; Schwarting, J.; et al. PET imaging in patients with brain metastasis-report of the RANO/PET group. Neuro Oncol. 2019, 21, 585–595. [Google Scholar] [CrossRef] [PubMed]
- Pyka, T.; Gempt, J.; Hiob, D.; Ringel, F.; Schlegel, J.; Bette, S.; Wester, H.J.; Meyer, B.; Forster, S. Textural analysis of pre-therapeutic [18F]-FET-PET and its correlation with tumor grade and patient survival in high-grade gliomas. Eur. J. Nucl. Med. Mol. Imaging 2016, 43, 133–141. [Google Scholar] [CrossRef] [PubMed]
- Louis, D.N.; Perry, A.; Reifenberger, G.; von Deimling, A.; Figarella-Branger, D.; Cavenee, W.K.; Ohgaki, H.; Wiestler, O.D.; Kleihues, P.; Ellison, D.W. The 2016 World Health Organization Classification of Tumors of the Central Nervous System: A summary. Acta Neuropathol. 2016, 131, 803–820. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Stupp, R.; Mason, W.P.; van den Bent, M.J.; Weller, M.; Fisher, B.; Taphoorn, M.J.; Belanger, K.; Brandes, A.A.; Marosi, C.; Bogdahn, U.; et al. Radiotherapy plus concomitant and adjuvant temozolomide for glioblastoma. N. Engl. J. Med. 2005, 352, 987–996. [Google Scholar] [CrossRef]
- Capper, D.; Zentgraf, H.; Balss, J.; Hartmann, C.; von Deimling, A. Monoclonal antibody specific for IDH1 R132H mutation. Acta Neuropathol. 2009, 118, 599–601. [Google Scholar] [CrossRef]
- Capper, D.; Weissert, S.; Balss, J.; Habel, A.; Meyer, J.; Jager, D.; Ackermann, U.; Tessmer, C.; Korshunov, A.; Zentgraf, H.; et al. Characterization of R132H mutation-specific IDH1 antibody binding in brain tumors. Brain Pathol. 2010, 20, 245–254. [Google Scholar] [CrossRef]
- Woehrer, A.; Sander, P.; Haberler, C.; Kern, S.; Maier, H.; Preusser, M.; Hartmann, C.; Kros, J.M.; Hainfellner, J.A.; Research Committee of the European Confederation of Neuropathological Societies. FISH-based detection of 1p 19q codeletion in oligodendroglial tumors: Procedures and protocols for neuropathological practice - a publication under the auspices of the Research Committee of the European Confederation of Neuropathological Societies (Euro-CNS). Clin. Neuropathol. 2011, 30, 47–55. [Google Scholar]
- Felsberg, J.; Rapp, M.; Loeser, S.; Fimmers, R.; Stummer, W.; Goeppert, M.; Steiger, H.J.; Friedensdorf, B.; Reifenberger, G.; Sabel, M.C. Prognostic significance of molecular markers and extent of resection in primary glioblastoma patients. Clin. Cancer Res. 2009, 15, 6683–6693. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Young, R.J.; Gupta, A.; Shah, A.D.; Graber, J.J.; Zhang, Z.; Shi, W.; Holodny, A.I.; Omuro, A.M. Potential utility of conventional MRI signs in diagnosing pseudoprogression in glioblastoma. Neurology 2011, 76, 1918–1924. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hamacher, K.; Coenen, H.H. Efficient routine production of the 18F-labelled amino acid O-2-18F fluoroethyl-L-tyrosine. Appl. Radiat. Isot. 2002, 57, 853–856. [Google Scholar] [CrossRef]
- Langen, K.J.; Bartenstein, P.; Boecker, H.; Brust, P.; Coenen, H.H.; Drzezga, A.; Grunwald, F.; Krause, B.J.; Kuwert, T.; Sabri, O.; et al. [German guidelines for brain tumour imaging by PET and SPECT using labelled amino acids]. Nuklearmedizin 2011, 50, 167–173. [Google Scholar]
- Caldeira, L.; Kops, E.R.; Yun, S.D.; Da Silva, N.; Mauler, J.; Weirich, C.; Scheins, J.; Herzog, H.; Tellmann, L.; Lohmann, P.; et al. The Julich Experience With Simultaneous 3T MR-BrainPET: Methods and Technology. IEEE Trans. Radiat. Plasma Med. Sci. 2019, 3, 352–362. [Google Scholar] [CrossRef]
- Pauleit, D.; Floeth, F.; Hamacher, K.; Riemenschneider, M.J.; Reifenberger, G.; Muller, H.W.; Zilles, K.; Coenen, H.H.; Langen, K.J. O-(2-[18F]fluoroethyl)-L-tyrosine PET combined with MRI improves the diagnostic assessment of cerebral gliomas. Brain 2005, 128, 678–687. [Google Scholar] [CrossRef] [Green Version]
- Galldiks, N.; Stoffels, G.; Filss, C.; Rapp, M.; Blau, T.; Tscherpel, C.; Ceccon, G.; Dunkl, V.; Weinzierl, M.; Stoffel, M.; et al. The use of dynamic O-(2-18F-fluoroethyl)-l-tyrosine PET in the diagnosis of patients with progressive and recurrent glioma. Neuro Oncol. 2015, 17, 1293–1300. [Google Scholar] [CrossRef] [PubMed]
- Lambin, P.; Leijenaar, R.T.H.; Deist, T.M.; Peerlings, J.; de Jong, E.E.C.; van Timmeren, J.; Sanduleanu, S.; Larue, R.; Even, A.J.G.; Jochems, A.; et al. Radiomics: The bridge between medical imaging and personalized medicine. Nat. Rev. Clin. Oncol. 2017, 14, 749–762. [Google Scholar] [CrossRef]
- van Griethuysen, J.J.M.; Fedorov, A.; Parmar, C.; Hosny, A.; Aucoin, N.; Narayan, V.; Beets-Tan, R.G.H.; Fillion-Robin, J.C.; Pieper, S.; Aerts, H. Computational Radiomics System to Decode the Radiographic Phenotype. Cancer Res. 2017, 77, e104–e107. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Orlhac, F.; Nioche, C.; Soussan, M.; Buvat, I. Understanding Changes in Tumor Texture Indices in PET: A Comparison Between Visual Assessment and Index Values in Simulated and Patient Data. J. Nucl. Med. 2017, 58, 387–392. [Google Scholar] [CrossRef]
- Orlhac, F.; Soussan, M.; Maisonobe, J.A.; Garcia, C.A.; Vanderlinden, B.; Buvat, I. Tumor texture analysis in 18F-FDG PET: Relationships between texture parameters, histogram indices, standardized uptake values, metabolic volumes, and total lesion glycolysis. J. Nucl. Med. 2014, 55, 414–422. [Google Scholar] [CrossRef] [Green Version]
- Kickingereder, P.; Gotz, M.; Muschelli, J.; Wick, A.; Neuberger, U.; Shinohara, R.T.; Sill, M.; Nowosielski, M.; Schlemmer, H.P.; Radbruch, A.; et al. Large-scale Radiomic Profiling of Recurrent Glioblastoma Identifies an Imaging Predictor for Stratifying Anti-Angiogenic Treatment Response. Clin. Cancer Res. 2016, 22, 5765–5771. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yasaka, K.; Akai, H.; Mackin, D.; Court, L.; Moros, E.; Ohtomo, K.; Kiryu, S. Precision of quantitative computed tomography texture analysis using image filtering: A phantom study for scanner variability. Medicine (Baltimore) 2017, 96, e6993. [Google Scholar] [CrossRef] [PubMed]
- Harrell, F.E., Jr.; Lee, K.L.; Mark, D.B. Multivariable prognostic models: Issues in developing models, evaluating assumptions and adequacy, and measuring and reducing errors. Stat. Med. 1996, 15, 361–387. [Google Scholar] [CrossRef]
- Vittinghoff, E.; McCulloch, C.E. Relaxing the rule of ten events per variable in logistic and Cox regression. Am. J. Epidemiol. 2007, 165, 710–718. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Le, T.T.; Fu, W.; Moore, J.H. Scaling tree-based automated machine learning to biomedical big data with a feature set selector. Bioinformatics 2020, 36, 250–256. [Google Scholar] [CrossRef] [Green Version]
- Virtanen, P.; Gommers, R.; Oliphant, T.E.; Haberland, M.; Reddy, T.; Cournapeau, D.; Burovski, E.; Peterson, P.; Weckesser, W.; Bright, J.; et al. SciPy 1.0: Fundamental algorithms for scientific computing in Python. Nat. Methods 2020, 17, 261–272. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bashir, A.; Brennum, J.; Broholm, H.; Law, I. The diagnostic accuracy of detecting malignant transformation of low-grade glioma using O-(2-[18F]fluoroethyl)-l-tyrosine positron emission tomography: A retrospective study. J. Neurosurg. 2018, 130, 451–464. [Google Scholar] [CrossRef]
- Balagurunathan, Y.; Gu, Y.; Wang, H.; Kumar, V.; Grove, O.; Hawkins, S.; Kim, J.; Goldgof, D.B.; Hall, L.O.; Gatenby, R.A.; et al. Reproducibility and Prognosis of Quantitative Features Extracted from CT Images. Transl. Oncol. 2014, 7, 72–87. [Google Scholar] [CrossRef] [Green Version]
- Grootjans, W.; Tixier, F.; van der Vos, C.S.; Vriens, D.; Le Rest, C.C.; Bussink, J.; Oyen, W.J.; de Geus-Oei, L.F.; Visvikis, D.; Visser, E.P. The Impact of Optimal Respiratory Gating and Image Noise on Evaluation of Intratumor Heterogeneity on 18F-FDG PET Imaging of Lung Cancer. J. Nucl. Med. 2016, 57, 1692–1698. [Google Scholar] [CrossRef] [Green Version]
- Larue, R.T.; Defraene, G.; De Ruysscher, D.; Lambin, P.; van Elmpt, W. Quantitative radiomics studies for tissue characterization: A review of technology and methodological procedures. Br. J. Radiol. 2017, 90, 20160665. [Google Scholar] [CrossRef] [PubMed]
- Wang, K.; Qiao, Z.; Zhao, X.; Li, X.; Wang, X.; Wu, T.; Chen, Z.; Fan, D.; Chen, Q.; Ai, L. Individualized discrimination of tumor recurrence from radiation necrosis in glioma patients using an integrated radiomics-based model. Eur. J. Nucl. Med. Mol. Imaging 2020, 47, 1400–1411. [Google Scholar] [CrossRef] [PubMed] [Green Version]
| # | Gender | Age | Initial Diagnosis | IDH Genotype | MGMT Promoter Methylation | EoR | Weeks Since Last Radiation | FU Diagnosis | Confirmation of FU Diagnosis | PFS [Months] | OS [Months] | TBRmean | TBRmax | TTP [min] | Slope [SUV/h] |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | m | 58 | GBM | wt | unmeth | PR | 6 | EP | Histomolecular | 4 | 9 | 1.8 | 1.9 | 12.5 | −0.4 |
| 2 | m | 61 | GBM | wt | unmeth | CR | 4 | EP | Histomolecular | 5 | 5 * | 2.1 | 2.4 | 37.5 | 0.4 |
| 3 | m | 52 | GBM | wt | unmeth | PR | 4 | EP | Histomolecular | 5 | 15 | 2.2 | 2.8 | 37.5 | 1.1 |
| 4 | m | 69 | GBM | wt | unmeth | PR | 8 | EP | Histomolecular | 6 | 12 | 1.9 | 1.9 | 22.5 | 0.3 |
| 5 | f | 43 | GBM | wt | unmeth | CR | 8 | EP | Follow-up | 5 | 24 * | 2.0 | 2.5 | 32.5 | 0.3 |
| 6 | m | 52 | GBM | wt | unmeth | CR | 7 | EP | Follow-up | 5 | 16 | 1.9 | 2.3 | 32.5 | 0.6 |
| 7 | m | 72 | GBM | wt | unmeth | B | 5 | EP | Follow-up | 4 | 7 | 2.2 | 3.7 | 18.5 | −0.8 |
| 8 | m | 51 | GBM | wt | meth | PR | 6 | EP | Histomolecular | 5 | 22 * | 2.5 | 3.9 | 12.5 | −1.9 |
| 9 | m | 44 | GBM | wt | unmeth | CR | 3 | EP | Follow-up | 3 | 9 | 1.9 | 2.6 | 37.5 | 1.3 |
| 10 | m | 61 | GBM | wt | unmeth | B | 8 | EP | Follow-up | 5 | 6 * | 2.2 | 2.8 | 27.5 | −0.2 |
| 11 | m | 57 | GBM | wt | unmeth | B | 5 | EP | Follow-up | 2 | 2 * | 2.1 | 2.8 | 18.5 | −1.5 |
| 12 | m | 42 | GBM | wt | meth | CR | 12 | EP | Follow-up | 5 | 34 | 2.1 | 2.5 | 15.5 | 0.1 |
| 13 | m | 79 | GBM | wt | n.a. | CR | 12 | EP | Histomolecular | n.a. | 6 * | 1.9 | 1.9 | 22.5 | 0.1 |
| 14 | m | 76 | GBM | wt | meth | B | 4 | EP | Follow-up | 4 | 6 | 2.0 | 2.6 | 18.5 | −0.2 |
| 15 | f | 52 | GBM | wt | meth | PR | 8 | EP | Follow-up | 5 | 15 * | 2.0 | 2.0 | 27.5 | 0.2 |
| 16 | m | 54 | GBM | wt | unmeth | PR | 8 | EP | Follow-up | 8 | 10 | 1.8 | 1.8 | 32.5 | 0.9 |
| 17 | m | 69 | GBM | wt | meth | B | 1 | EP | Follow-up | 4 | 4 | 1.9 | 2.1 | 27.5 | 0.9 |
| 18 | f | 52 | GBM | wt | unmeth | PR | 4 | EP | Follow-up | 8 | 11 * | 2.6 | 3.5 | 37.5 | 1.0 |
| 19 | f | 71 | GBM | wt | meth | PR | 3 | PSP | Histomolecular | 12 | 21 | 1.8 | 1.8 | 37.5 | 0.4 |
| 20 | f | 76 | GBM | wt | meth | CR | 8 | PSP | Histomolecular | 20 | 20 * | 1.9 | 2.2 | 22.5 | −0.3 |
| 21 | f | 58 | GBM | wt | unmeth | PR | 8 | PSP | Follow-up | 24 | 38 | 2.0 | 2.1 | 22.5 | −0.1 |
| 22 | m | 50 | GBM | wt | unmeth | CR | 4 | PSP | Follow-up | 16 | 23 | 1.9 | 2.0 | 37.5 | 1.0 |
| 23 | m | 34 | GBM | wt | meth | PR | 8 | PSP | Follow-up | 60 | 65 * | 1.8 | 1.8 | 37.5 | 0.4 |
| 24 | f | 48 | GBM | wt | n.a. | CR | 8 | PSP | Histomolecular | n.a. | 14 * | 1.9 | 1.9 | 27.5 | −0.5 |
| 25 | f | 64 | GBM | wt | meth | CR | 4 | PSP | Follow-up | n.a. | 50 | 1.7 | 1.7 | 37.5 | 1.2 |
| 26 | f | 66 | GBM | wt | unmeth | B | 12 | PSP | Follow-up | 10 | 12 | 2.3 | 3.0 | 22.5 | −0.7 |
| 27 | m | 66 | GBM | wt | meth | B | 12 | PSP | Follow-up | 16 | 23 * | 2.2 | 2.5 | 27.5 | 0.4 |
| 28 | f | 49 | GBM | wt | unmeth | PR | 6 | PSP | Follow-up | 10 | 11 * | 1.9 | 2.2 | 27.5 | 0.0 |
| 29 | f | 24 | AA | wt | unmeth | B | 11 | PSP | Follow-up | 8 | 12 * | 2.1 | 2.4 | 18.5 | −0.5 |
| 30 | f | 51 | GBM | wt | unmeth | CR | 8 | PSP | Follow-up | 8 | 18 | 1.8 | 1.8 | 27.5 | 0.6 |
| 31 | m | 44 | GBM | wt | unmeth | PR | 10 | PSP | Follow-up | 6 | 12 | 1.9 | 1.9 | 32.5 | 0.2 |
| 32 | m | 65 | GBM | wt | meth | CR | 6 | PSP | Follow-up | 11 | 13 * | 1.8 | 1.8 | 37.5 | 0.9 |
| 33 | f | 68 | GBM | wt | unmeth | PR | 9 | PSP | Follow-up | 10 | 10 * | 1.8 | 1.8 | 37.5 | 0.9 |
| 34 | m | 48 | GBM | mut | meth | CR | 6 | PSP | Follow-up | 24 * | 24 * | 1.8 | 1.8 | 37.5 | 0.5 |
| Parameter | TBRmean | TBRmax | TTP | Slope |
|---|---|---|---|---|
| Sensitivity | 75% | 81% | 75% | 56% |
| Specificity | 61% | 67% | 44% | 61% |
| PPV | 63% | 68% | 55% | 56% |
| NPV | 73% | 80% | 67% | 61% |
| FNR | 25% | 19% | 25% | 44% |
| FPR | 39% | 33% | 56% | 39% |
| Accuracy | 68% | 74% | 59% | 59% |
| F1 Score | 0.69 | 0.74 | 0.63 | 0.56 |
| MCC | 0.36 | 0.48 | 0.20 | 0.17 |
| AUC | 0.73 | 0.79 | 0.61 | 0.55 |
| Cut-off | ≤ 1.95 | ≤ 2.25 | ≥ 25 min | ≥ 0.3 SUV/h |
| p-value # | 0.045 | 0.007 | 0.297 | 0.492 |
| Parameter Combinations § | TBRmean + TBRmax | TBRmean + TTP | TBRmean + Slope | TBRmax + TTP | TBRmax + Slope | TTP + Slope | TBRmean + TBRmax + TTP |
|---|---|---|---|---|---|---|---|
| Sensitivity | 75% | 69% | 50% | 69% | 50% | 56% | 69% |
| Specificity | 72% | 78% | 78% | 83% | 89% | 61% | 89% |
| PPV | 71% | 73% | 67% | 79% | 80% | 56% | 85% |
| NPV | 76% | 74% | 64% | 75% | 67% | 61% | 76% |
| FNR | 25% | 31% | 50% | 31% | 50% | 44% | 31% |
| FPR | 28% | 22% | 22% | 17% | 11% | 39% | 11% |
| Accuracy | 74% | 74% | 65% | 76% | 71% | 59% | 79% |
| F1 Score | 0.73 | 0.71 | 0.57 | 0.73 | 0.62 | 0.56 | 0.76 |
| MCC | 0.47 | 0.47 | 0.29 | 0.53 | 0.43 | 0.17 | 0.59 |
| p-value # | 0.015 | 0.014 | 0.151 | 0.005 | 0.023 | 0.492 | 0.001 |
| Training Data | Test Data | |
|---|---|---|
| Number of datasets | 72 | 30 |
| Feature selection method | RFE (random forest) | |
| Number of features | 4 | |
| Classifier | Random forest | |
| Sensitivity | 82% | 100% |
| Specificity | 90% | 40% |
| PPV | 87% | 63% |
| NPV | 85% | 100% |
| FNR | 18% | 0% |
| FPR | 10% | 60% |
| Accuracy | 86% | 70% |
| F1 Score | 0.84 | 0.77 |
| MCC | 0.72 | 0.50 |
| AUC | 0.74 | 0.74 |
| p-value # | < 0.001 | 0.017 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lohmann, P.; Elahmadawy, M.A.; Gutsche, R.; Werner, J.-M.; Bauer, E.K.; Ceccon, G.; Kocher, M.; Lerche, C.W.; Rapp, M.; Fink, G.R.; et al. FET PET Radiomics for Differentiating Pseudoprogression from Early Tumor Progression in Glioma Patients Post-Chemoradiation. Cancers 2020, 12, 3835. https://doi.org/10.3390/cancers12123835
Lohmann P, Elahmadawy MA, Gutsche R, Werner J-M, Bauer EK, Ceccon G, Kocher M, Lerche CW, Rapp M, Fink GR, et al. FET PET Radiomics for Differentiating Pseudoprogression from Early Tumor Progression in Glioma Patients Post-Chemoradiation. Cancers. 2020; 12(12):3835. https://doi.org/10.3390/cancers12123835
Chicago/Turabian StyleLohmann, Philipp, Mai A. Elahmadawy, Robin Gutsche, Jan-Michael Werner, Elena K. Bauer, Garry Ceccon, Martin Kocher, Christoph W. Lerche, Marion Rapp, Gereon R. Fink, and et al. 2020. "FET PET Radiomics for Differentiating Pseudoprogression from Early Tumor Progression in Glioma Patients Post-Chemoradiation" Cancers 12, no. 12: 3835. https://doi.org/10.3390/cancers12123835
APA StyleLohmann, P., Elahmadawy, M. A., Gutsche, R., Werner, J.-M., Bauer, E. K., Ceccon, G., Kocher, M., Lerche, C. W., Rapp, M., Fink, G. R., Shah, N. J., Langen, K.-J., & Galldiks, N. (2020). FET PET Radiomics for Differentiating Pseudoprogression from Early Tumor Progression in Glioma Patients Post-Chemoradiation. Cancers, 12(12), 3835. https://doi.org/10.3390/cancers12123835







