Chronic Plasma Exposure to Kinase Inhibitors in Patients with Oncogene-Addicted Non-Small Cell Lung Cancer
Abstract
:Simple Summary
Abstract
1. Introduction
2. Patients and Methods
2.1. Patients
2.2. Assessment of Plasma Exposure
- Optimal: The optimal concentration corresponds to the true residual plasma concentration at a steady state, in the blood collection performed immediately before the next administration.
- Evaluable: The residual plasma concentration is estimated by an extrapolation method from known pharmacokinetic parameters (distribution volume, half-life, clearance) and from data obtained in population pharmacokinetic models [14]. This estimate of standard trough concentration (Cmin, std) is only possible when blood samples were collected at steady state or during the terminal elimination phase of the drug, since in this phase, the elimination rate is linear [14,15].
- -
- C (min, std) = C(t) * 0.5 ^ (Delta (t)/t1/2)
- -
- C (min, std) = C(t) * exp (k(e) x Delta (t))
- 3.
- Not interpretable: Extrapolation is not feasible for samples taken during the plasma peak period.
2.3. Somatic Molecular Analysis
- -
- CHP2 (Ion AmpliSeq Cancer Hotspot Panel v2 (CHP2)) designed to amplify 207 amplicons covering 50 genes (Thermo Fisher Scientific)).
- -
- Oncomine lung (Oncomine Lung ctDNA Assay contains 35 amplicons covering 11 genes (Thermo Fisher Scientific)). A unique molecular identifier is combined with each single DNA molecule. For calling the detected variants, the following parameters were applied: Allele Read count > 10; Fusion read count > 1; Variant type: Single nucleotide variant, insertion-deletion, multi-nucleotide variant, copy number variant, long deletion, fusion; Variant effect: Unknown, missense, none frameshift Insertion, none frameshift Deletion, non-sense, stop loss, frameshift insertion, frameshift deletion.
- -
- MOSC4 (Ion AmpliSeq MOSC4 designed to cover 82 genes, combining two other panels (CHP2 + Safir02)).
- -
- OCAV3 (Ion AmpliSeq Oncomine Comprehensive Assay V3 enables the detection of mutations across 161 genes, gene fusions, and copy number variations (Thermo Fisher Scientific)).
- -
- Sentosa SQ NSCLC (Sentosa SQ Non-Small Cell Lung Cancer panel targets 11 genes with 28 amplicons (Vela Diagnostics)). For calling the detected variants, following parameters were applied: 5000 Exomes Global MAF < 0.01; Allele frequency > 0.02; Allele ratio > 0.02; Allele read counts > 50; Alternate read counts > 30; Fusion read counts > 50; Variant effect: Unknown, missense, none frameshift insertion, none frameshift deletion, stop loss, non-sense, frameshift insertion, frameshift deletion; Variant type: Single nucleotide variant, insertion-deletion, multi-nucleotide variant, copy number variant, long deletion, fusion.
2.4. Statistical Analysis
3. Results
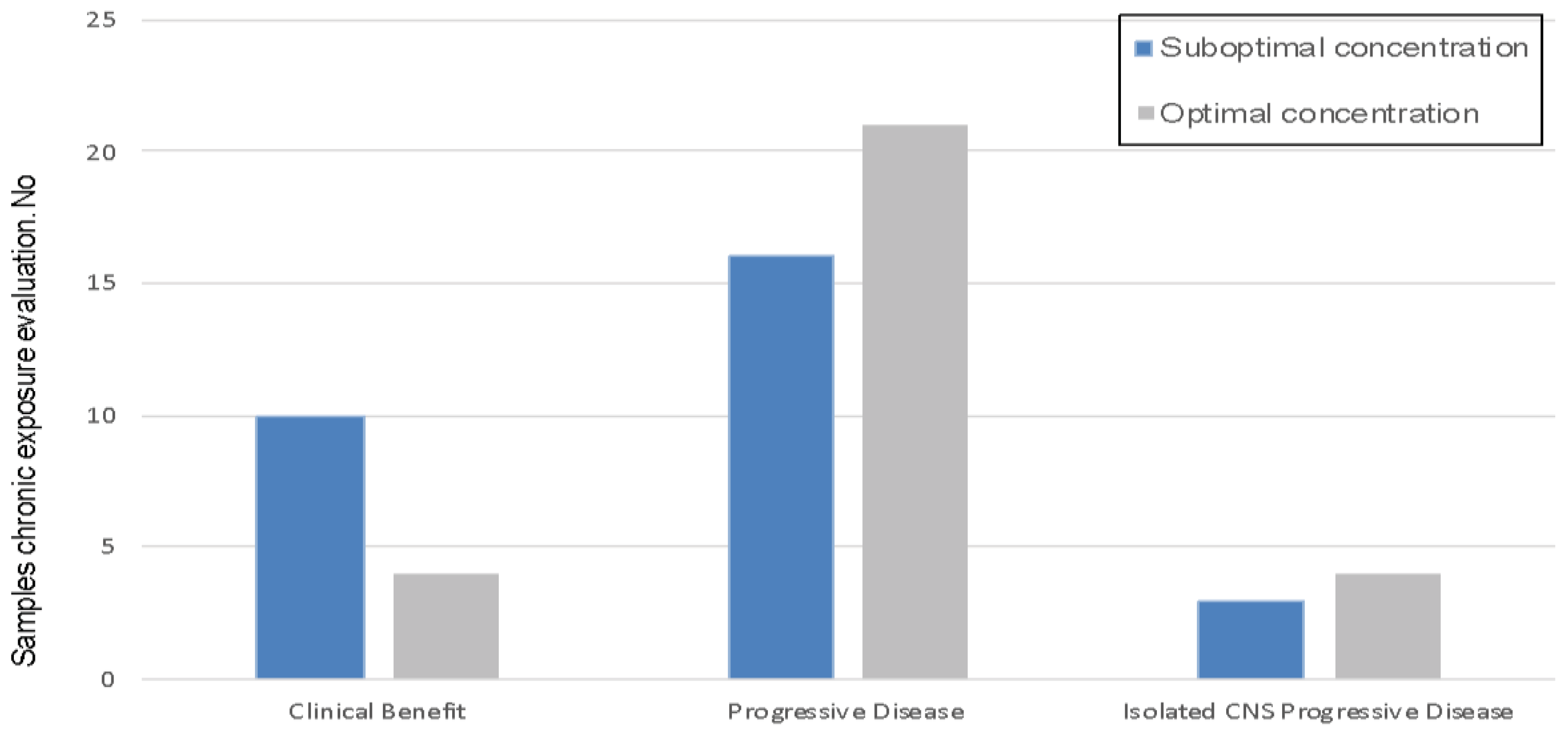
3.1. Suboptimal Concentration and KI Response
3.2. Clinical Relevance of Suboptimal Concentration
3.3. KI Exposure and Resistance Mechanisms
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Kalemkerian, G.P.; Narula, N.; Kennedy, E.B.; Biermann, W.A.; Donington, J.; Leighl, N.B.; Lew, M.; Pantelas, J.; Ramalingam, S.S.; Reck, M.; et al. Molecular Testing Guideline for the Selection of Patients With Lung Cancer for Treatment With Targeted Tyrosine Kinase Inhibitors: American Society of Clinical Oncology Endorsement of the College of American Pathologists/International Association for the Study of Lung Cancer/Association for Molecular Pathology Clinical Practice Guideline Update. J. Clin. Oncol. 2018, 36, 911–919. [Google Scholar] [CrossRef] [PubMed]
- Soria, J.-C.; Ohe, Y.; Vansteenkiste, J.; Reungwetwattana, T.; Chewaskulyong, B.; Lee, K.H.; Dechaphunkul, A.; Imamura, F.; Nogami, N.; Kurata, T.; et al. Osimertinib in UntreatedEGFR-Mutated Advanced Non–Small-Cell Lung Cancer. N. Engl. J. Med. 2018, 378, 113–125. [Google Scholar] [CrossRef] [PubMed]
- Verheijen, R.B.; Yu, H.; Schellens, J.H.; Beijnen, J.H.; Steeghs, N.; Huitema, A.D. Practical Recommendations for Therapeutic Drug Monitoring of Kinase Inhibitors in Oncology. Clin. Pharmacol. Ther. 2017, 102, 765–776. [Google Scholar] [CrossRef] [PubMed]
- Yu, H.; Steeghs, N.; Nijenhuis, C.M.; Schellens, J.H.M.; Beijnen, J.H.; Huitema, A.D.R. Practical Guidelines for Therapeutic Drug Monitoring of Anticancer Tyrosine Kinase Inhibitors: Focus on the Pharmacokinetic Targets. Clin. Pharmacokinet. 2014, 53, 305–325. [Google Scholar] [CrossRef]
- Groenland, S.L.; Mathijssen, R.H.J.; Beijnen, J.H.; Huitema, A.D.R.; Steeghs, N. Individualized dosing of oral targeted therapies in oncology is crucial in the era of precision medicine. Eur. J. Clin. Pharmacol. 2019, 75, 1309–1318. [Google Scholar] [CrossRef]
- Hamilton, M.; Wolf, J.L.; Rusk, J.; Beard, S.E.; Clark, G.M.; Witt, K.; Cagnoni, P.J. Effects of Smoking on the Pharmacokinetics of Erlotinib. Clin. Cancer Res. 2006, 12, 2166–2171. [Google Scholar] [CrossRef] [Green Version]
- Van Leeuwen, R.W.F.; Van Gelder, T.; Mathijssen, R.H.J.; A Jansman, F.G. Drug–drug interactions with tyrosine-kinase inhibitors: A clinical perspective. Lancet Oncol. 2014, 15, e315–e326. [Google Scholar] [CrossRef]
- Mir, O.; Touati, N.; Lia, M.; Litière, S.; Le Cesne, A.; Sleijfer, S.; Blay, J.-Y.; Leahy, M.; Young, R.; Mathijssen, R.H.; et al. Impact of Concomitant Administration of Gastric Acid–Suppressive Agents and Pazopanib on Outcomes in Soft-Tissue Sarcoma Patients Treated within the EORTC 62043/62072 Trials. Clin. Cancer Res. 2019, 25, 1479–1485. [Google Scholar] [CrossRef] [Green Version]
- Marin, D.; Bazeos, A.; Mahon, F.-X.; Eliasson, L.; Milojkovic, D.; Bua, M.; Apperley, J.F.; Szydlo, R.; Desai, R.; Kozlowski, K.; et al. Adherence Is the Critical Factor for Achieving Molecular Responses in Patients With Chronic Myeloid Leukemia Who Achieve Complete Cytogenetic Responses on Imatinib. J. Clin. Oncol. 2010, 28, 2381–2388. [Google Scholar] [CrossRef]
- Arrondeau, J.; Mir, O.; Boudou-Rouquette, P.; Coriat, R.; Ropert, S.; Dumas, G.; Rodrigues, M.J.; Rousseau, B.; Blanchet, B.; Goldwasser, F. Sorafenib exposure decreases over time in patients with hepatocellular carcinoma. Investig. New Drugs 2011, 30, 2046–2049. [Google Scholar] [CrossRef]
- Hiemke, C.; Bergemann, N.; Clement, H.W.; Conca, A.; Deckert, J.; Domschke, K.; Eckermann, G.; Egberts, K.; Gerlach, M.; Greiner, C.; et al. Consensus Guidelines for Therapeutic Drug Monitoring in Neuropsychopharmacology: Update 2017. Pharmacopsychiatry 2018, 51, 9–62. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rousselot, P.; Johnson-Ansah, H.; Huguet, F.; Legros, L.; Escoffre-Barbe, M.; Gardembas, M.; Cony-Makhoul, P.; Coiteux, V.; Sutton, L.; Abarah, W.; et al. Personalized Daily Doses of Imatinib By Therapeutic Drug Monitoring Increase the Rates of Molecular Responses in Patients with Chronic Myeloid Leukemia. Final Results of the Randomized OPTIM Imatinib Study. Blood 2015, 126, 133. [Google Scholar] [CrossRef]
- Widmer, N.; Bardin, C.; Chatelut, E.; Paci, A.; Beijnen, J.; Levêque, D.; Veal, G.; Astier, A. Review of therapeutic drug monitoring of anticancer drugs part two–Targeted therapies. Eur. J. Cancer 2014, 50, 2020–2036. [Google Scholar] [CrossRef] [PubMed]
- Reis, R.; Labat, L.; Allard, M.; Boudou-Rouquette, P.; Chapron, J.; Bellesoeur, A.; Thomas-Schoemann, A.; Arrondeau, J.; Giraud, F.; Alexandre, J.; et al. Liquid chromatography-tandem mass spectrometric assay for therapeutic drug monitoring of the EGFR inhibitors afatinib, erlotinib and osimertinib, the ALK inhibitor crizotinib and the VEGFR inhibitor nintedanib in human plasma from non-small cell lung cancer patients. J. Pharm. Biomed. Anal. 2018, 158, 174–183. [Google Scholar] [CrossRef]
- Wang, Y.; Chia, Y.L.; Nedelman, J.; Schran, H.; Mahon, F.-X.; Molimard, M. A Therapeutic Drug Monitoring Algorithm for Refining the Imatinib Trough Level Obtained at Different Sampling Times. Ther. Drug Monit. 2009, 31, 579–584. [Google Scholar] [CrossRef]
- Lankheet, N.A.G.; Knapen, L.M.; Schellens, J.H.M.; Beijnen, J.H.; Steeghs, N.; Huitema, A.D.R. Plasma Concentrations of Tyrosine Kinase Inhibitors Imatinib, Erlotinib, and Sunitinib in Routine Clinical Outpatient Cancer Care. Ther. Drug Monit. 2014, 36, 326–334. [Google Scholar] [CrossRef]
- Van Leeuwen, R.W.F.; Jansman, F.G.A.; Hunfeld, N.G.; Peric, R.; Reyners, A.K.L.; Imholz, A.L.T.; Brouwers, J.R.B.J.; Aerts, J.G.; Van Gelder, T.; Mathijssen, R.H.J. Tyrosine Kinase Inhibitors and Proton Pump Inhibitors: An Evaluation of Treatment Options. Clin. Pharmacokinet. 2017, 56, 683–688. [Google Scholar] [CrossRef]
- Pistilli, B.; Paci, A.; Ferreira, A.R.; Di Meglio, A.; Poinsignon, V.; Bardet, A.; Menvielle, G.; Dumas, A.; Pinto, S.; Dauchy, S.; et al. Serum Detection of Nonadherence to Adjuvant Tamoxifen and Breast Cancer Recurrence Risk. J. Clin. Oncol. 2020, 38, 2762–2772. [Google Scholar] [CrossRef]
- Gougis, P.; Palmieri, L.-J.; Funck-Brentano, C.; Paci, A.; Flippot, R.; Mir, O.; Coriat, R. Major pitfalls of protein kinase inhibitors prescription: A review of their clinical pharmacology for daily use. Crit. Rev. Oncol. 2019, 141, 112–124. [Google Scholar] [CrossRef]
- Yu, H.A.; Arcila, M.E.; Rekhtman, N.; Sima, C.S.; Zakowski, M.F.; Pao, W.; Kris, M.G.; Miller, V.A.; Ladanyi, M.; Riely, G.J. Analysis of Tumor Specimens at the Time of Acquired Resistance to EGFR-TKI Therapy in 155 Patients with EGFR-Mutant Lung Cancers. Clin. Cancer Res. 2013, 19, 2240–2247. [Google Scholar] [CrossRef] [Green Version]
- Huillard, O.; Blanchet, B.; Boudou-Rouquette, P.; Thomas-Schoemann, A.; Wassermann, J.; Goldwasser, F. Sorafenib in Thyroid Cancer Patients: Learning From Toxicity. Oncol. 2014, 19, e3. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Doherty, G.J.; Lynskey, D.; Matakidou, A.; Fife, K.; Eisen, T. Dose escalation of axitinib on disease progression as a strategy in the treatment of metastatic renal cell carcinoma. ESMO Open 2018, 3, e000445. [Google Scholar] [CrossRef] [PubMed] [Green Version]


| N# | Driver Alteration | Other Alteration | Kinase Inhibitor | NGS Panel (Tissue) | NGS Panel (Blood) | EGFR T790M ddPCR Stilla® (Blood) |
|---|---|---|---|---|---|---|
| 1 | EGFR exon 21 p. L858R | No | Gefitinib | - | CHP2 *: Not detected | Not detected |
| 2 | EGFR exon 21 p. L858R | No | Gefitinib | - | - | T790M detected |
| 3 | EGFR exon 21 p. L858R | No | Gefitinib | Oncomine® Lung: T790M detected | CHP2 *: Not detected | - |
| 4 | EGFR exon 21 p. L858R | No | Gefitinib | - | CHP2 *: Not detected | Not detected |
| 5 | EGFR exon 19 deletion | TP53 | Gefitinib | MOSC4: T790M not detected | - | - |
| 6 | EGFR exon 19 deletion | No | Gefitinib | - | - | Not detected |
| 7 | EGFR exon 19 deletion | No | Gefitinib | MOSC4: T790M not detected | - | T790M detected |
| 8 | EGFR exon 21 p.L833F | TP53, CKDN2A | Erlotinib | - | - | ddPCR Not detected |
| 9 | EGFR exon 19 deletion | No | Erlotinib | - | - | Not detected |
| 10 | EGFR exon 19 deletion | No | Erlotinib | - | - | Not detected |
| 11 | EGFR exon 19 deletion | No | Erlotinib | - | - | Not detected |
| 12 | EGFR exon 19 deletion | No | Erlotinib | MOSC4: T790M detected | - | T790M detected |
| 13 | EGFR exon 21 p. L858R | No | Erlotinib | Unknown: not detected | - | - |
| 14 | EGFR exon 19 deletion | No | Erlotinib | MOSC4: T790M not detected | - | Not detected |
| 15 | EGFR exon 19 deletion | No | Erlotinib | MOSC4: T790M not detected | - | - |
| 16 | EGFR exon 19 deletion | No | Erlotinib | MOSC4: T790M not detected | CHP2 *: Not detected | - |
| 17 | EGFR exon 21 p. L858R | No | Erlotinib | OCAV3: T790M not detected | Oncomine® Lung: Not detected | - |
| 18 | EGFR exon 19 deletion | No | Erlotinib | NSCLC: T790M not detected | - | Not detected |
| Characteristics, No. (%) | All Samples (n = 51) | Optimal Concentration (n = 25) | Suboptimal Concentration (n = 26) | p-Value |
|---|---|---|---|---|
| Molecular alteration:ALK-rearranged, BRAFV600E mutation, EGFR deletion exon 19, EGFR mutation exon 21 (L858R), EGFR mutation exon 21 (L833F), ROS-1 rearranged, MET mutation exon 14, No molecular alteration (ALK, BRAF, EGFR, KRAS, MET, ROS-1) | 3 (7%), 10 (19%), 23 (45%), 9 (17%), 1 (2%), 2 (4%), 2 (4%), 1 (2%) | 0 (0%), 4 (16%), 12 (48%), 6 (24%), 1 (4%), 1 (4%), 1 (4%), 0 | 3 (12%), 6 (22%), 11 (42%), 3 (12%), 0, 1 (4%), 1 (4%), 1 (4%) | p = 0.50 |
| Kinase inhibitor: Erlotinib, Gefitinib, Osimertinib, Crizotinib, Dabrafenib, Trametinib | 13 (26%), 11 (21%), 10 (19%), 7 (14%), 5 (10%), 5 (10%) | 7 (28%), 8 (32%), 4 (16%), 2 (8%), 2 (8%), 2 (8%) | 6 (23%), 3 (11.5%), 6 (23%), 5 (19.5%), 3 (11.5%), 3 (11.5%) | p = 0.52 |
| Stage at diagnosis: I-II, III, IV | 1 (2%), 2 (4%), 48 (94%) | 0, 2 (8%), 23 (92%) | 1 (4%), 0, 25 (96%) | p = 0.24 |
| Lines of previous kinase inhibitors: ≤2, >2 | 44 (86%), 7 (14%) | 20 (80%), 5 (20%) | 24 (92%), 2 (8%) | p = 0.25 |
| Current smoker: Yes, No | 5 (10%), 46 (90%) | 1 (4%), 24 (96%) | 4 (15%), 22 (85%) | p = 0.35 |
| Concomitant proton pump inhibitor: No, Yes | 35 (69%), 16 (31%) | 15 (60%), 10 (40%) | 20 (77%), 6 (23%) | p = 0.19 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Geraud, A.; Mezquita, L.; Auclin, E.; Combarel, D.; Delahousse, J.; Gougis, P.; Massard, C.; Jovelet, C.; Caramella, C.; Adam, J.; et al. Chronic Plasma Exposure to Kinase Inhibitors in Patients with Oncogene-Addicted Non-Small Cell Lung Cancer. Cancers 2020, 12, 3758. https://doi.org/10.3390/cancers12123758
Geraud A, Mezquita L, Auclin E, Combarel D, Delahousse J, Gougis P, Massard C, Jovelet C, Caramella C, Adam J, et al. Chronic Plasma Exposure to Kinase Inhibitors in Patients with Oncogene-Addicted Non-Small Cell Lung Cancer. Cancers. 2020; 12(12):3758. https://doi.org/10.3390/cancers12123758
Chicago/Turabian StyleGeraud, Arthur, Laura Mezquita, Edouard Auclin, David Combarel, Julia Delahousse, Paul Gougis, Christophe Massard, Cécile Jovelet, Caroline Caramella, Julien Adam, and et al. 2020. "Chronic Plasma Exposure to Kinase Inhibitors in Patients with Oncogene-Addicted Non-Small Cell Lung Cancer" Cancers 12, no. 12: 3758. https://doi.org/10.3390/cancers12123758
APA StyleGeraud, A., Mezquita, L., Auclin, E., Combarel, D., Delahousse, J., Gougis, P., Massard, C., Jovelet, C., Caramella, C., Adam, J., Naltet, C., Lavaud, P., Gazzah, A., Lacroix, L., Rouleau, E., Vasseur, D., Mir, O., Planchard, D., Paci, A., & Besse, B. (2020). Chronic Plasma Exposure to Kinase Inhibitors in Patients with Oncogene-Addicted Non-Small Cell Lung Cancer. Cancers, 12(12), 3758. https://doi.org/10.3390/cancers12123758





