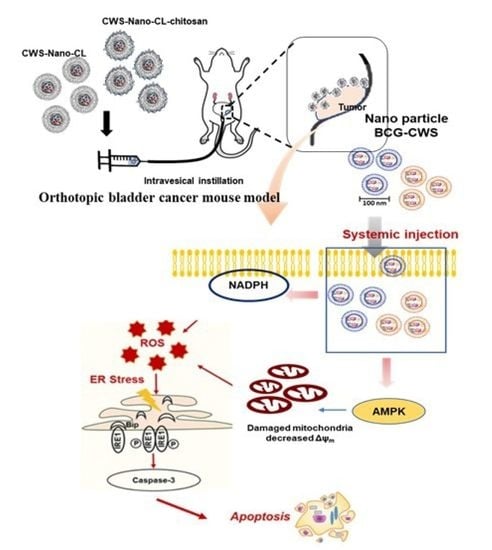
Liposome-Encapsulated Bacillus Calmette–Guérin Cell Wall Skeleton Enhances Antitumor Efficiency for Bladder Cancer In Vitro and In Vivo via Induction of AMP-Activated Protein Kinase
Abstract
Simple Summary
Abstract
1. Introduction
2. Results
2.1. Physical Characteristics of CWS-Loaded Formulations
2.2. CWS-Loaded Formulations Inhibit the Growth of Bladder Cancer Cells
2.3. CWS-Loaded Formulations Inhibit the mTOR Pathway and Induce the Initiation of Autophagy through AMPK Activation
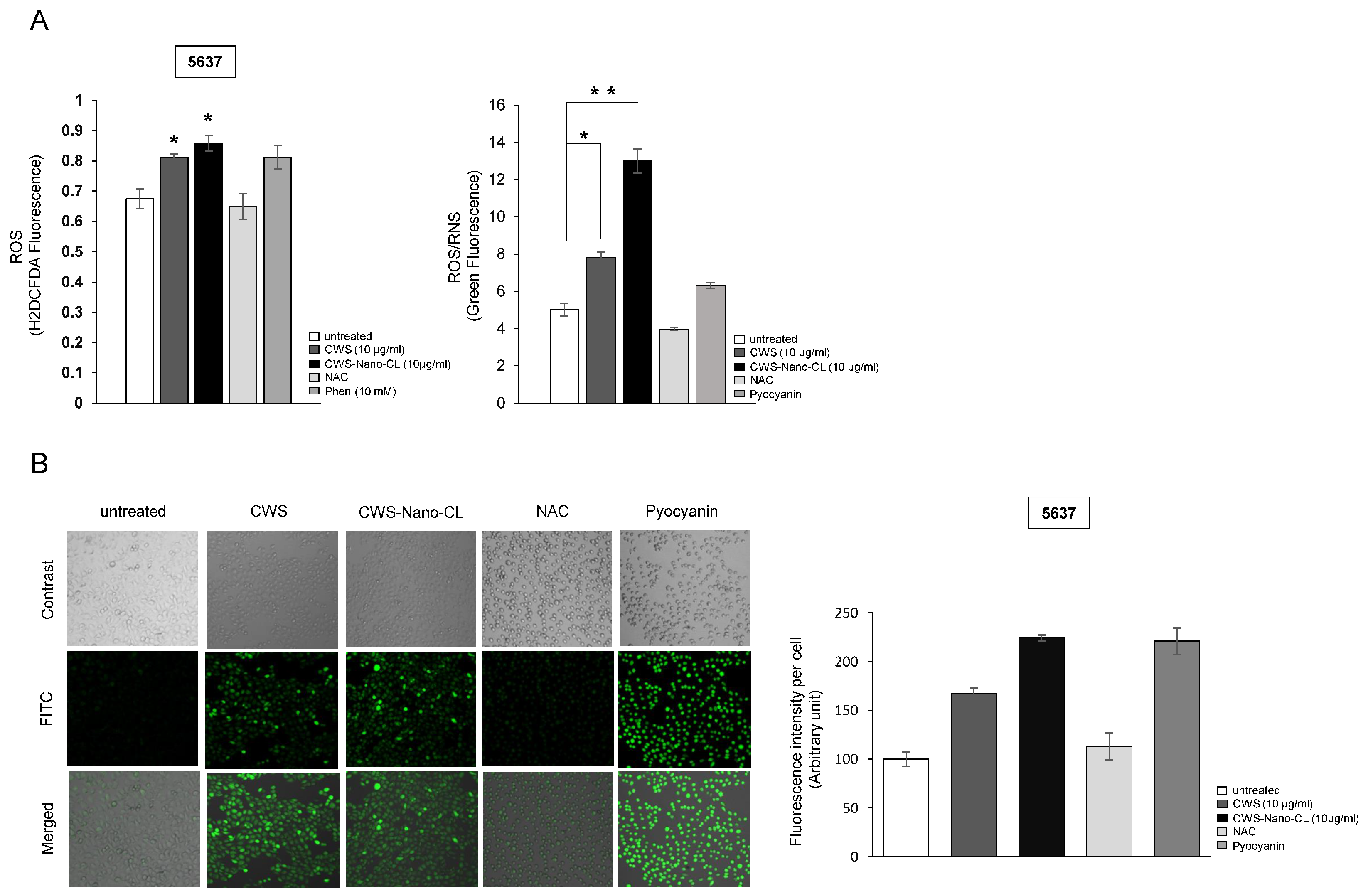
2.4. CWS-Loaded Formulations Increase ROS Accumulation and ER Stress through AMPK Activation in Bladder Cancer Cells
2.5. CWS-Loaded Formulations Induce Tumor Regression in an Orthotopic Bladder Cancer Mouse Model
3. Discussion
4. Materials and Methods
4.1. Cell Culture and Antibodies
4.2. Preparation of the CWS-Loaded Liposomal Formulations
4.2.1. Preparation of CWS-Nano-CL
4.2.2. Preparation of CWS-Nano-CL-Chitosan
4.2.3. Particle Size and Zeta Potential (ZP) Analysis
4.2.4. Entrapment Efficiency (EE) of the BCG-CWS Liposomal Formulations
4.2.5. Chitosan Coating Efficiency
4.2.6. Colloidal Stability of Liposomal Formulations
4.2.7. Transmission Electron Microscopy (TEM)
4.3. Cell Viability and Clonogenic Assays
4.4. Western Blot Analysis
4.5. ROS Accumulation and ROS/RNS Production
4.6. Animal Studies
4.7. Statistical Analyses
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Song, W.; Jeon, H.G. Incidence of kidney, bladder, and prostate cancers in Korea: An update. Korean J. Urol. 2015, 56, 422–428. [Google Scholar] [CrossRef] [PubMed]
- Han, R.F.; Pan, J.G. Can intravesical bacillus Calmette-Guérin reduce recurrence in patients with superficial bladder cancer? A meta-analysis of randomized trials. Urology 2006, 67, 1216–1223. [Google Scholar] [CrossRef] [PubMed]
- Kamat, A.M.; Bellmunt, J.; Galsky, M.D.; Konety, B.R.; Lamm, D.L.; Langham, D.; Lee, C.T.; Milowsky, M.I.; O’Donnell, M.A.; O’Donnell, P.H.; et al. Society for Immunotherapy of Cancer consensus statement on immunotherapy for the treatment of bladder carcinoma. J. Immunother. Cancer 2017, 5, 68. [Google Scholar] [CrossRef] [PubMed]
- Lamm, D.L.; van der Meijden, P.M.; Morales, A.; Brosman, S.A.; Catalona, W.J.; Herr, H.W.; Soloway, M.S.; Steg, A.; Debruyne, F.M. Incidence and treatment of complications of bacillus Calmette-Guerin intravesical therapy in superficial bladder cancer. J. Urol. 1992, 147, 596–600. [Google Scholar] [CrossRef]
- Steg, A.; Leleu, C.; Debré, B.; Boccon-Gibod, L.; Sicard, D. Systemic bacillus Calmette-Guérin infection, ‘BCGitis’, in patients treated by intravesical bacillus Calmette-Guérin therapy for bladder cancer. Eur. Urol. 1989, 16, 161–164. [Google Scholar] [CrossRef]
- Azuma, I.; Ribi, E.E.; Meyer, T.J.; Zbar, B. Biologically Active Components from Mycobacterial Cell Walls. I. Isolation and Composition of Cell Wall Skeleton and Component P3. JNCI J. Natl. Cancer Inst. 1974, 52, 95–101. [Google Scholar] [CrossRef]
- Yamamura, Y.; Azuma, I.; Taniyama, T.; Sugimura, K.; Hirao, F.; Tokuzen, R.; Okabe, M.; Nakahara, W.; Yasumoto, K.; Ohta, M. Immunotherapy of Cancer with Cell Wall Skeleton of Mycobacterium Bovis-Bacillus Calmette-Guérin: Experimental and Clinical Results. Ann. N. Y. Acad. Sci. 1976, 277, 209–227. [Google Scholar] [CrossRef]
- Kodama, K.; Higashiyama, M.; Takami, K.; Oda, K.; Okami, J.; Maeda, J.; Akazawa, T.; Matsumoto, M.; Seya, T.; Wada, M.; et al. Innate immune therapy with a Bacillus Calmette-Guérin cell wall skeleton after radical surgery for non-small cell lung cancer: A case-control study. Surg. Today 2009, 39, 194–200. [Google Scholar] [CrossRef]
- Yasumoto, K.; Manabe, H.; Yanagawa, E.; Nagano, N.; Ueda, H.; Hirota, N.; Ohta, M.; Nomoto, K.; Azuma, I.; Yamamura, Y. Nonspecific adjuvant immunotherapy of lung cancer with cell wall skeleton of Mycobacterium bovis Bacillus Calmette-Guérin. Cancer Res. 1979, 39, 3262–3267. [Google Scholar]
- Nakamura, T.; Fukiage, M.; Higuchi, M.; Nakaya, A.; Yano, I.; Miyazaki, J.; Nishiyama, H.; Akaza, H.; Ito, T.; Hosokawa, H.; et al. Nanoparticulation of BCG-CWS for application to bladder cancer therapy. J. Control. Release 2014, 176, 44–53. [Google Scholar] [CrossRef]
- Masuda, H.; Nakamura, T.; Noma, Y.; Harashima, H. Application of BCG-CWS as a Systemic Adjuvant by Using Nanoparticulation Technology. Mol. Pharm. 2018, 15, 5762–5771. [Google Scholar] [CrossRef] [PubMed]
- Yoon, Y.H.; Yang, H.M.; Kim, C.H.; Goo, Y.T.; Hwang, G.Y.; Chang, I.H.; Whang, Y.M.; Choi, Y.W. Enhanced Intracellular Delivery of BCG Cell Wall Skeleton into Bladder Cancer Cells Using Liposomes Functionalized with Folic Acid and Pep-1 Peptide. Pharmaceutics 2019, 11, 652. [Google Scholar] [CrossRef] [PubMed]
- Azuma, I.; Seya, T. Development of immunoadjuvants for immunotherapy of cancer. Int. Immunopharmacol. 2001, 1, 1249–1259. [Google Scholar] [CrossRef]
- Murata, M. Activation of Toll-like receptor 2 by a novel preparation of cell wall skeleton from Mycobacterium bovis BCG Tokyo (SMP-105) sufficiently enhances immune responses against tumors. Cancer Sci. 2008, 99, 1435–1440. [Google Scholar] [CrossRef]
- Tsuji, S.; Matsumoto, M.; Tsuji, S.; Akazawa, T.; Takeuchi, O.; Akira, S.; Kawata, T.; Azuma, I.; Toyoshima, K.; Seya, T. Maturation of human dendritic cells by cell wall skeleton of Mycobacterium bovis bacillus Calmette-Guérin: Involvement of toll-like receptors. Infect. Immun. 2000, 68, 6883–6890. [Google Scholar] [CrossRef]
- Yuk, J.M.; Shin, D.M.; Song, K.S.; Lim, K.; Kim, K.H.; Lee, S.H.; Kim, J.M.; Lee, J.S.; Paik, T.H.; Kim, J.S.; et al. Bacillus calmette-guerin cell wall cytoskeleton enhances colon cancer radiosensitivity through autophagy. Autophagy 2010, 6, 46–60. [Google Scholar] [CrossRef]
- Guma, M.; Wang, Y.; Viollet, B.; Liu-Bryan, R. AMPK Activation by A-769662 Controls IL-6 Expression in Inflammatory Arthritis. PLoS ONE 2015, 10, e0140452. [Google Scholar] [CrossRef]
- Han, J.-Y.; Lim, Y.J.; Choi, J.A.; Lee, J.H.; Jo, S.H.; Oh, S.; Song, C.H. The Role of Prostate Apoptosis Response-4 (Par-4) in Mycobacterium tuberculosis Infected Macrophages. Sci. Rep. 2016, 6, 32079. [Google Scholar] [CrossRef]
- Noda, T.; Ohsumi, Y. Tor, a Phosphatidylinositol Kinase Homologue, Controls Autophagy in Yeast. J. Biol. Chem. 1998, 273, 3963–3966. [Google Scholar] [CrossRef]
- Shaw, R.J.; Bardeesy, N.; Manning, B.D.; Lopez, L.; Kosmatka, M.; DePinho, R.A.; Cantley, L.C. The LKB1 tumor suppressor negatively regulates mTOR signaling. Cancer Cell 2004, 6, 91–99. [Google Scholar] [CrossRef]
- Whang, Y.M.; Park, S.I.; Trenary, I.A.; Egnatchik, R.A.; Fessel, J.P.; Kaufman, J.M.; Carbone, D.P.; Young, J.D. LKB1 deficiency enhances sensitivity to energetic stress induced by erlotinib treatment in non-small-cell lung cancer (NSCLC) cells. Oncogene 2016, 35, 856–866. [Google Scholar] [CrossRef]
- Whang, Y.M.; Kim, M.J.; Cho, M.J.; Yoon, H.; Choi, Y.W.; Kim, T.H.; Chang, I.H. Rapamycin enhances growth inhibition on urothelial carcinoma cells through LKB1 deficiency-mediated mitochondrial dysregulation. J. Cell. Physiol. 2019, 234, 13083–13096. [Google Scholar] [CrossRef]
- Yorimitsu, T.; Nair, U.; Yang, Z.; Klionsky, D.J. Endoplasmic Reticulum Stress Triggers Autophagy. J. Biol. Chem. 2006, 281, 30299–30304. [Google Scholar] [CrossRef]
- Sun, X.; Zhang, X.; Zhai, H.; Zhang, D.; Ma, S. Chicoric acid (CA) induces autophagy in gastric cancer through promoting endoplasmic reticulum (ER) stress regulated by AMPK. Biomed. Pharmacother. 2019, 118, 109144. [Google Scholar] [CrossRef]
- Urano, F.; Wang, X.; Bertolotti, A.; Zhang, Y.; Chung, P.; Harding, H.P.; Ron, D. Coupling of stress in the ER to activation of JNK protein kinases by transmembrane protein kinase IRE1. Science 2000, 287, 664–666. [Google Scholar] [CrossRef]
- Matsumoto, M.; Seya, T.; Kikkawa, S.; Tsuji, S.; Shida, K.; Nomura, M.; Kurita-Taniguchi, M.; Ohigashi, H.; Yokouchi, H.; Takami, K.; et al. Interferon gamma-producing ability in blood lymphocytes of patients with lung cancer through activation of the innate immune system by BCG cell wall skeleton. Int. Immunopharmacol. 2001, 1, 1559–1569. [Google Scholar] [CrossRef]
- Akazawa, T.; Masuda, H.; Saeki, Y.; Matsumoto, M.; Takeda, K.; Tsujimura, K.; Kuzushima, K.; Takahashi, T.; Azuma, I.; Akira, S.; et al. Adjuvant-Mediated Tumor Regression and Tumor-Specific Cytotoxic Response Are Impaired in MyD88-Deficient Mice. Cancer Res. 2004, 64, 757. [Google Scholar] [CrossRef]
- Ishii, K.; Kurita-Taniguchi, M.; Aoki, M.; Kimura, T.; Kashiwazaki, Y.; Matsumoto, M.; Seya, T. Gene-inducing program of human dendritic cells in response to BCG cell-wall skeleton (CWS), which reflects adjuvancy required for tumor immunotherapy. Immunol. Lett. 2005, 98, 280–290. [Google Scholar] [CrossRef]
- Lum, J.J.; Bauer, D.E.; Kong, M.; Harris, M.H.; Li, C.; Lindsten, T.; Thompson, C.B. Growth Factor Regulation of Autophagy and Cell Survival in the Absence of Apoptosis. Cell 2005, 120, 237–248. [Google Scholar] [CrossRef]
- He, C.; Klionsky, D.J. Regulation Mechanisms and Signaling Pathways of Autophagy. Annu. Rev. Genet. 2009, 43, 67–93. [Google Scholar] [CrossRef]
- Burikhanov, R.; Zhao, Y.; Goswami, A.; Qiu, S.; Schwarze, S.R.; Rangnekar, V.M. The Tumor Suppressor Par-4 Activates an Extrinsic Pathway for Apoptosis. Cell 2009, 138, 377–388. [Google Scholar] [CrossRef]
- Sui, X.; Kong, N.; Ye, L.; Han, W.; Zhou, J.; Zhang, Q.; He, C.; Pan, H. p38 and JNK MAPK pathways control the balance of apoptosis and autophagy in response to chemotherapeutic agents. Cancer Lett. 2014, 344, 174–179. [Google Scholar] [CrossRef]
- Jiang, Q.; Li, F.; Shi, K.; Wu, P.; An, J.; Yang, Y.; Xu, C. Involvement of p38 in signal switching from autophagy to apoptosis via the PERK/eIF2α/ATF4 axis in selenite-treated NB4 cells. Cell Death Dis. 2014, 5, e1270. [Google Scholar] [CrossRef]
- Thayyullathil, F.; Rahman, A.; Pallichankandy, S.; Patel, M.; Galadari, S. ROS-dependent prostate apoptosis response-4 (Par-4) up-regulation and ceramide generation are the prime signaling events associated with curcumin-induced autophagic cell death in human malignant glioma. FEBS Open Bio 2014, 4, 763–776. [Google Scholar] [CrossRef]
- Muzzarelli, R.A. Colorimetric Determination of Chitosan. Anal. Biochem. 1998, 260, 255–257. [Google Scholar] [CrossRef]
- Nakamura, T.; Fukiage, M.; Suzuki, Y.; Yano, I.; Miyazaki, J.; Nishiyama, H.; Akaza, H.; Harashima, H. Mechanism responsible for the antitumor effect of BCG-CWS using the LEEL method in a mouse bladder cancer model. J. Control. Release 2014, 196, 161–167. [Google Scholar] [CrossRef]
- Joraku, A.; Homhuan, A.; Kawai, K.; Yamamoto, T.; Miyazaki, J.; Kogure, K.; Yano, I.; Harashima, H.; Akaza, H. Immunoprotection against murine bladder carcinoma by octaarginine-modified liposomes incorporating cell wall of Mycobacterium bovis bacillus Calmette-Guérin. BJU Int. 2009, 103, 686–693. [Google Scholar] [CrossRef]
- Miyazaki, J.; Nishiyama, H.; Yano, I.; Nakaya, A.; Kohama, H.; Kawai, K.; Joraku, A.; Nakamura, T.; Harashima, H.; Akaza, H. The therapeutic effects of R8-liposome-BCG-CWS on BBN-induced rat urinary bladder carcinoma. Anticancer. Res. 2011, 31, 2065–2071. [Google Scholar]
- Miyazaki, J.; Kawai, K.; Kojima, T.; Oikawa, T.; Joraku, A.; Shimazui, T.; Nakaya, A.; Yano, I.; Nakamura, T.; Harashima, H.; et al. The liposome-incorporating cell wall skeleton of Mycobacterium bovis bacillus Calmette-Guéin can directly enhance the susceptibility of cancer cells to lymphokine-activated killer cells through up-regulation of natural-killer group 2, member D ligands. BJU Int. 2011, 108, 1520–1526. [Google Scholar] [CrossRef]
- Li, N.; Zhuang, C.; Wang, M.; Sun, X.; Nie, S.; Pan, W. Liposome coated with low molecular weight chitosan and its potential use in ocular drug delivery. Int. J. Pharm. 2009, 379, 131–138. [Google Scholar] [CrossRef]
- Seo, H.K.; Shin, S.P.; Jung, N.R.; Kwon, W.A.; Jeong, K.C.; Lee, S.J. The establishment of a growth-controllable orthotopic bladder cancer model through the down-regulation of c-myc expression. Oncotarget 2016, 8, 50500–50509. [Google Scholar] [CrossRef]
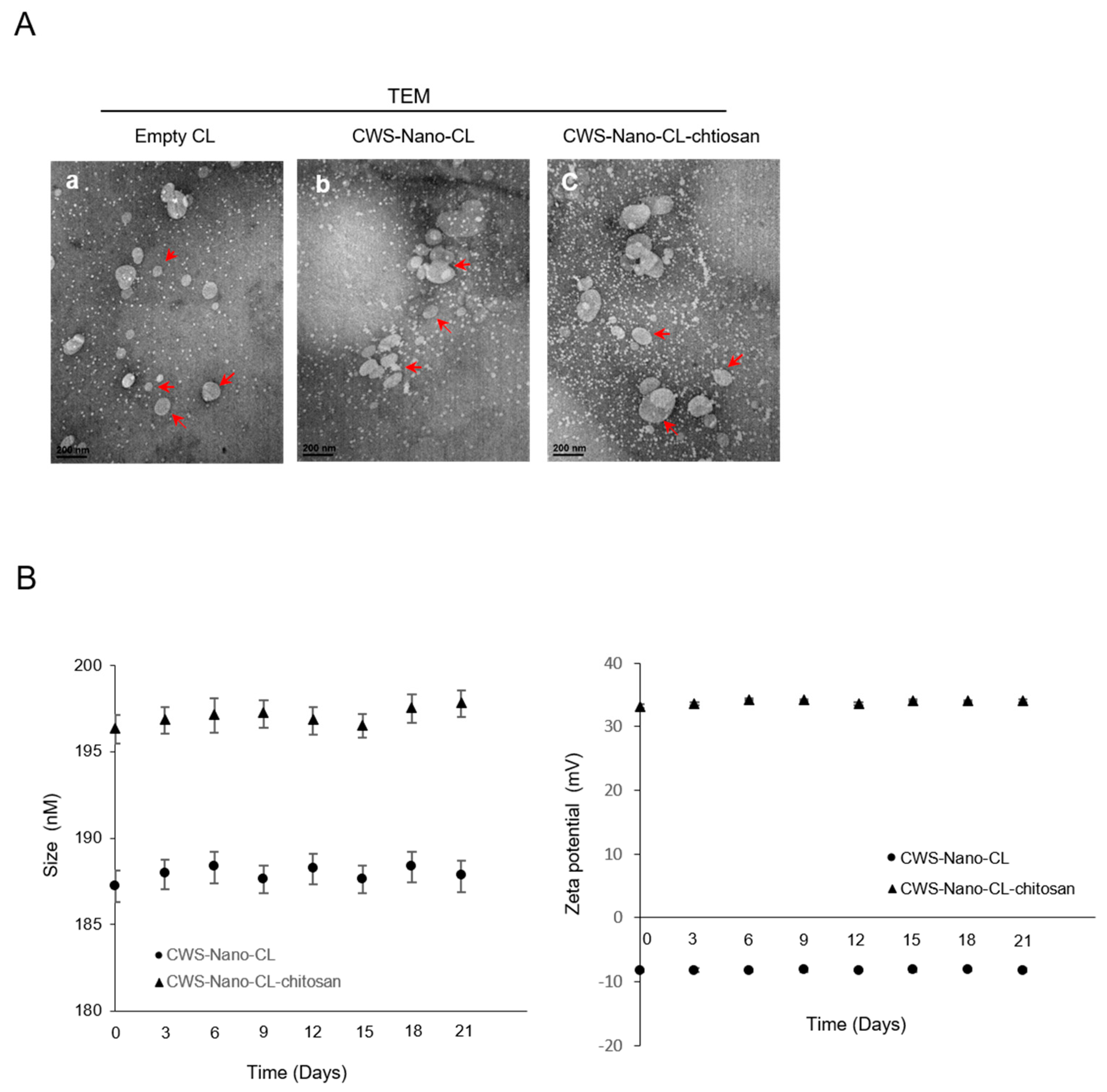




| Formulation Liposome Composition (mol ratio) | Empty-CL | CWS-Nano-CL | CWS-Nano-CL-chitosan |
|---|---|---|---|
| PC | 90 | 90 | 90 |
| CH | 10 | 10 | 10 |
| Physical properties | |||
| Size (nm) | 185.1 ± 0.19 | 187.2 ± 0.19 | 196.33 ± 0.34 |
| PDI | 0.11 ± 0.05 | 0.13 ± 0.04 | 0.16 ± 0.03 |
| ZP (mV) | −8.26 ± 0.17 | −8.31 ± 0.37 | 33.17 ± 0.24 |
| EE (%) | 60.15 ± 0.28 | 58.25 ± 0.31 | |
| DL | 219.87 ± 4.69 | 217.91 ± 3.29 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Whang, Y.M.; Yoon, D.H.; Hwang, G.Y.; Yoon, H.; Park, S.I.; Choi, Y.W.; Chang, I.H. Liposome-Encapsulated Bacillus Calmette–Guérin Cell Wall Skeleton Enhances Antitumor Efficiency for Bladder Cancer In Vitro and In Vivo via Induction of AMP-Activated Protein Kinase. Cancers 2020, 12, 3679. https://doi.org/10.3390/cancers12123679
Whang YM, Yoon DH, Hwang GY, Yoon H, Park SI, Choi YW, Chang IH. Liposome-Encapsulated Bacillus Calmette–Guérin Cell Wall Skeleton Enhances Antitumor Efficiency for Bladder Cancer In Vitro and In Vivo via Induction of AMP-Activated Protein Kinase. Cancers. 2020; 12(12):3679. https://doi.org/10.3390/cancers12123679
Chicago/Turabian StyleWhang, Young Mi, Da Hyeon Yoon, Gwang Yong Hwang, Hoyub Yoon, Serk In Park, Young Wook Choi, and In Ho Chang. 2020. "Liposome-Encapsulated Bacillus Calmette–Guérin Cell Wall Skeleton Enhances Antitumor Efficiency for Bladder Cancer In Vitro and In Vivo via Induction of AMP-Activated Protein Kinase" Cancers 12, no. 12: 3679. https://doi.org/10.3390/cancers12123679
APA StyleWhang, Y. M., Yoon, D. H., Hwang, G. Y., Yoon, H., Park, S. I., Choi, Y. W., & Chang, I. H. (2020). Liposome-Encapsulated Bacillus Calmette–Guérin Cell Wall Skeleton Enhances Antitumor Efficiency for Bladder Cancer In Vitro and In Vivo via Induction of AMP-Activated Protein Kinase. Cancers, 12(12), 3679. https://doi.org/10.3390/cancers12123679