Animal Safety, Toxicology, and Pharmacokinetic Studies According to the ICH S9 Guideline for a Novel Fusion Protein tTF-NGR Targeting Procoagulatory Activity into Tumor Vasculature: Are Results Predictive for Humans?
Abstract
:Simple Summary
Abstract
1. Introduction
- procoagulatory activity of tTF-NGR similar to TF in a factor X/factor Xa assay,
- specific binding to the respective target molecules (CD13) on stimulated EC,
- in vivo intratumoral accumulation shown with imaging techniques,
- in vivo induced intratumoral activation of coagulation with consecutive tumor vascular occlusion and inhibition of tumor vessel blood flow,
- in vivo therapeutic antitumor activity in xenotransplants and mouse tumor models independent of tumor histology,
- combinatorial activity with cytotoxic drugs and radiotherapy when applied in a specific sequence.
2. Materials and Methods
2.1. Academic Toxicology Studies for Intravenous Application of tTF-NGR in Mice
2.2. Safety Pharmacology and Toxicology Studies According to ICH S9 and S6 Guidelines
2.3. Pharmacokinetic and Toxicokinetic Studies
2.4. Clinical Study
3. Results
3.1. Academic Toxicology Studies for Intravenous Application of tTF-NGR in Mice
3.2. External Safety Pharmacology and Toxicology Studies According to ICH S9 and S6 Guidelines
3.2.1. Mouse Studies
3.2.2. Rat Studies
3.2.3. Guinea Pig Studies
3.2.4. Beagle Dog Studies
- upon one single application starting at 4 mg tTF-NGR/kg b.w., equivalent to 80 mg/m2 b.s.a. (changes in behavior).
- upon daily applications for 5 consecutive days starting at 1 mg tTF-NGR/kg b.w., equivalent to 20 mg/m2 b.s.a. (bile acids and bilirubin serum levels increased in both of 2 animals, ALAT serum activity increased and behavioral changes were observed in 1 out of 2 animals).No immunological effects consistent with decreasing or ameliorating the tolerance of secondary or follow-up applications were noted.
3.3. Pharmacokinetic (PK) and Toxicokinetic Studies in Beagle Dogs
4. Discussion
4.1. Quality of the Non-Clinical Studies
4.2. Predictability of the Non-Clinical Studies for the Clinical Phase I Study in Cancer Patients
4.3. Predictive Value of Non-Clinical Safety and Toxicology Studies
4.3.1. Mouse Data
4.3.2. Other Species
4.3.3. Pharmacokinetics (PK)
4.4. Central Statement and Consequences to Be Discussed
5. Conclusions and Alternative Options for a Translational Procedure in Oncology
Author Contributions
Funding
Conflicts of Interest
References and Notes
- Kessler, T.; Bieker, R.; Padró, T.; Schwöppe, C.; Persigehl, T.; Bremer, C.; Kreuter, M.; Berdel, W.E.; Mesters, R.M. Inhibition of Tumor Growth by RGD Peptide-Directed Delivery of Truncated Tissue Factor to the Tumor Vasculature. Clin. Cancer Res. 2005, 11, 6317–6324. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Persigehl, T.; Bieker, R.; Matuszewski, L.; Wall, A.; Kessler, T.; Kooijman, H.; Meier, N.; Ebert, W.; Berdel, W.E.; Heindel, W.; et al. Antiangiogenic Tumor Treatment: Early Noninvasive Monitoring with USPIO-enhanced MR Imaging in Mice. Radiology 2007, 244, 449–456. [Google Scholar] [CrossRef] [PubMed]
- Persigehl, T.; Matuszewski, L.; Kessler, T.; Wall, A.; Meier, N.; Ebert, W.; Berdel, W.E.; Heindel, W.; Mesters, R.; Bremer, C. Prediction of Antiangiogenic Treatment Efficacy by Iron Oxide Enhanced Parametric Magnetic Resonance Imaging. Investig. Radiol. 2007, 42, 791–796. [Google Scholar] [CrossRef] [PubMed]
- Kessler, T.; Schwöppe, C.; Liersch, R.; Schliemann, C.; Hintelmann, H.; Bieker, R.; Berdel, W.E.; Mesters, R.M. Generation of Fusion Proteins for Selective Occlusion of Tumor Vessels. Curr. Drug Discov. Technol. 2008, 5, 1–8. [Google Scholar] [CrossRef]
- Bieker, R.; Kessler, T.; Schwöppe, C.; Padro, T.; Persigehl, T.; Bremer, C.; Dreischalück, J.; Kolkmeyer, A.; Heindel, W.; Mesters, R.M.; et al. Infarction of tumor vessels by NGR-peptide directed targeting of tissue factor. Experimental results and first-in-man experience. Blood 2009, 113, 5019–5027. [Google Scholar] [CrossRef] [Green Version]
- Schwöppe, C.; Kessler, T.; Persigehl, T.; Liersch, R.; Hintelmann, H.; Dreischalück, J.; Ring, J.; Bremer, C.; Heindel, W.; Mesters, R.M.; et al. Tissue-factor fusion proteins induce occlusion of tumor vessels. Thromb. Res. 2010, 125, S143–S150. [Google Scholar] [CrossRef]
- Dreischalück, J.; Schwöppe, C.; Spieker, T.; Kessler, T.; Tiemann, K.; Liersch, R.; Schliemann, C.; Kreuter, M.; Kolkmeyer, A.; Hintelmann, H.; et al. Vascular infarction by subcutaneous application of tissue factor targeted to tumor vessels with NGR-peptides: Activity and toxicity profile. Int. J. Oncol. 2010, 37, 1389–1397. [Google Scholar] [CrossRef] [Green Version]
- Von Maltzahn, G.; Park, J.-H.; Lin, K.Y.-M.; Singh, N.; Schwöppe, C.; Mesters, R.M.; Berdel, W.E.; Ruoslahti, E.; Sailor, M.J.; Bhatia, S.N. Nanoparticles that communicate in vivo to amplify tumour targeting. Nat. Mater. 2011, 10, 545–552. [Google Scholar] [CrossRef] [Green Version]
- Brand, C.; Dencks, S.; Schmitz, G.; Mühlmeister, M.; Stypmann, J.; Ross, R.; Hintelmann, H.; Schliemann, C.; Müller-Tidow, C.; Mesters, R.M.; et al. Low-Energy Ultrasound Treatment Improves Regional Tumor Vessel Infarction by Retargeted Tissue Factor. J. Ultrasound Med. 2015, 34, 1227–1236. [Google Scholar] [CrossRef]
- Persigehl, T.; Ring, J.; Bremer, C.; Heindel, W.; Holtmeier, R.; Stypmann, J.; Claesener, M.; Hermann, S.; Schäfers, M.; Zerbst, C.; et al. Non-invasive monitoring of tumor-vessel infarction by retargeted truncated tissue factor tTF-NGR using multi-modal imaging. Angiogenesis 2014, 17, 235–246. [Google Scholar] [CrossRef]
- Schwöppe, C.; Zerbst, C.; Fröhlich, M.; Schliemann, C.; Kessler, T.; Liersch, R.; Overkamp, L.; Holtmeier, R.; Stypmann, J.; Dreiling, A.; et al. Anticancer Therapy by Tumor Vessel Infarction with Polyethylene Glycol Conjugated Retargeted Tissue Factor. J. Med. Chem. 2013, 56, 2337–2347. [Google Scholar] [CrossRef] [PubMed]
- Brand, C.; Fröhlich, M.; Ring, J.; Schliemann, C.; Kessler, T.; Mantke, V.; König, S.; Lücke, M.; Mesters, R.M.; Berdel, W.E.; et al. Tumor Growth Inhibition via Occlusion of Tumor Vasculature Induced by N-Terminally PEGylated Retargeted Tissue Factor tTF-NGR. Mol. Pharm. 2015, 12, 3749–3758. [Google Scholar] [CrossRef] [PubMed]
- Schwöppe, C.; Hintelmann, H.; Mesters, R.M.; Berdel, W.E.; König, S. Using native gel electrophoresis or isoelectric focusing as experimental “clock” for the (iso)aspartate formation of tTF-NGR fusion proteins. Biomacromol. Mass Spectrom. 2012, 2, 285–293. [Google Scholar]
- Brand, C.; Schliemann, C.; Ring, J.; Kessler, T.; Bäumer, S.; Angenendt, L.; Mantke, V.; Ross, R.; Hintelmann, H.; Spieker, T.; et al. NG2 proteoglycan as a pericyte target for anticancer therapy by tumor vessel infarction with retargeted tissue factor. Oncotarget 2016, 7, 6774–6789. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Stucke-Ring, J.; Ronnacker, J.; Brand, C.; Höltke, C.; Schliemann, C.; Kessler, T.; Schmidt, L.H.; Harrach, S.; Mantke, V.; Hintelmann, H.; et al. Combinatorial effects of doxorubicin and retargeted tissue factor by intratumoral entrapment of doxorubicin and proapoptotic increase of tumor vascular infarction. Oncotarget 2016, 7, 82458–82472. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Schmidt, L.H.; Stucke-Ring, J.; Brand, C.; Schliemann, C.; Harrach, S.; Muley, T.; Herpel, E.; Kessler, T.; Mohr, M.; Görlich, D.; et al. CD13 as target for tissue factor induced tumor vascular infarction in small cell lung cancer. Lung Cancer 2017, 113, 121–127. [Google Scholar] [CrossRef]
- Schmidt, L.H.; Brand, C.; Stucke-Ring, J.; Schliemann, C.; Kessler, T.; Harrach, S.; Mohr, M.; Görlich, D.; Marra, A.; Hillejan, L.; et al. Potential therapeutic impact of CD13 expression in non-small cell lung cancer. PLoS ONE 2017, 12, e0177146. [Google Scholar] [CrossRef] [Green Version]
- Kessler, T.; Baumeier, A.; Brand, C.; Grau, M.; Angenendt, L.; Harrach, S.; Stalmann, U.; Schmidt, L.H.; Gosheger, G.; Hardes, J.; et al. Aminopeptidase N (CD13): Expression, Prognostic Impact, and Use as Therapeutic Target for Tissue Factor Induced Tumor Vascular Infarction in Soft Tissue Sarcoma. Transl. Oncol. 2018, 11, 1271–1282. [Google Scholar] [CrossRef]
- Höink, A.; Persigehl, T.; Kwiecien, R.; Balthasar, M.; Mesters, R.; Berdel, W.E.; Heindel, W.; Bremer, C.; Schwöppe, C. Gadofosveset-enhanced MRI as simple surrogate parameter for real-time evaluation of the initial tumour vessel infarction by retargeted tissue factor tTF-NGR. Oncol. Lett. 2019, 17, 270–280. [Google Scholar] [CrossRef] [Green Version]
- Gerwing, M.; Herrmann, K.; Helfen, A.; Schliemann, C.; Berdel, W.E.; Eisenblätter, M.; Wildgruber, M. The beginning of the end for conventional RECIST—novel therapies require novel imaging approaches. Nat. Rev. Clin. Oncol. 2019, 16, 442–458. [Google Scholar] [CrossRef]
- Brand, C.; Greve, B.; Bölling, T.; Eich, H.T.; Willich, N.; Harrach, S.; Hintelmann, H.; Lenz, G.; Mesters, R.M.; Kessler, T.; et al. Radiation synergizes with antitumor activity of CD13-targeted tissue factor in a HT1080 xenograft model of human soft tissue sarcoma. PLoS ONE 2020, 15, e0229271. [Google Scholar] [CrossRef] [PubMed]
- The Human Protein Atlas. Available online: http://www.proteinatlas.org/ENSG00000166825-ANPEP/tissue (accessed on 26 November 2020).
- World Medical Association. WMA Declaration of Helsinki-Ethical Principles for Medical Research involving Human Subjects. In Proceedings of the 64th WMA General Assembly, Fortaleza, Brazil, October 2013. [Google Scholar]
- ICH TOPIC M3 (R2). Non-Clinical Safety Studies for the Conduct of Human Clinical Trials and Marketing Authorization for Pharmaceuticals. EMEA/CHMP/ICH 286/95. Available online: https://www.ema.europa.eu/en/ich-m3-r2-non-clinical-safety-studies-conduct-human-clinical-trials-pharmaceuticals (accessed on 26 November 2020).
- ICH Topic S9. Non-Clinical Evaluation for Anticancer Pharmaceuticals. EMEA/CHMP/ICH/646107/2008. Available online: https://www.ema.europa.eu/en/ich-s9-non-clinical-evaluation-anticancer-pharmaceuticals (accessed on 26 November 2020).
- Schliemann, C.; Gerwing, M.; Heinzow, H.S.; Harrach, S.; Schwöppe, C.; Wildgruber, M.; Hansmeier, A.A.; Angenendt, L.; Berdel, A.F.; Stalmann, U.; et al. First-In-Class CD13-Targeted Tissue Factor tTF-NGR in Patients with Recurrent or Refractory Malignant Tumors: Results of a Phase I Dose-Escalation Study. Cancers 2020, 12, 1488. [Google Scholar] [CrossRef]
- Beyerle, A.; Nolte, M.W.; Solomon, C.; Herzog, E.; Dickneite, G. Analysis of the safety and pharmacodynamics of human fibrinogen concentrate in animals. Toxicol. Appl. Pharmacol. 2014, 280, 70–77. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chabbat, J.; Hampikian-Lenin, S.; Toully, V.; Gaillandre, A.; Pejaudier, L.; Steinbuch, M. A human factor VIIa concentrate and its effects in the hemophilic A dog. Thromb. Res. 1989, 54, 603–612. [Google Scholar] [CrossRef]
- Du, L.M.; Nurden, P.; Nurden, A.T.; Nichols, T.C.; Bellinger, D.A.; Jensen, E.S.; Haberichter, S.L.; Merricks, E.; Raymer, R.A.; Fang, J.; et al. Platelet-targeted gene therapy with human factor VIII establishes haemostasis in dogs with haemophilia A. Nat. Commun. 2013, 4, 2773. [Google Scholar] [CrossRef] [Green Version]
- Knudsen, T.; Kristensen, A.T.; Sørensen, B.B.; Olsen, O.H.; Stennicke, H.R.; Petersen, L.C. Characterization of canine coagulation factor VII and its complex formation with tissue factor: canine-human cross-species compatibility. J. Thromb. Haemost. 2010, 8, 1763–1772. [Google Scholar] [CrossRef] [PubMed]
- Knudsen, T.; Ohlsen, O.H.; Petersen, L.C. Tissue factor and factor VIIa cross-species compatibility. Front. Biosci. 2011, 16, 3196–3215. [Google Scholar] [CrossRef] [PubMed]
- Kristensen, A.T.; Wiinberg, B.; Jessen, L.R.; Andreasen, E.; Jensen, A.L. Evaluation of human recombinant tissue factor-activated thrombelastography in 49 dogs with neoplasia. J. Vet. Intern. Med. 2008, 22, 140–147. [Google Scholar] [CrossRef] [PubMed]
- Mahmood, I. Designing first-in-human dose of coagulation factors: application of pharmacokinetic allometric scaling. Haemophilia 2013, 20, 32–38. [Google Scholar] [CrossRef] [PubMed]
- Røttingen, J.-A.; Enden, T.; Camerer, E.; Iversen, J.-G.; Prydz, H. Binding of Human Factor VIIa to Tissue Factor Induces Cytosolic Ca2+Signals in J82 Cells, Transfected COS-1 Cells, Madin-Darby Canine Kidney Cells and in Human Endothelial Cells Induced to Synthesize Tissue Factor. J. Biol. Chem. 1995, 270, 4650–4660. [Google Scholar] [CrossRef] [Green Version]
- Wiinberg, B.; Jensen, A.L.; Rojkjaer, R.; Johansson, P.; Kjelgaard-Hansen, M.; Kristensen, A.T. Validation of human recombinant tissue factor-activated thrombelastography on citrated whole blood from clinically healthy dogs. Vet. Clin. Pathol. 2005, 34, 389–393. [Google Scholar] [CrossRef]
- Ruf, W.; Miles, D.J.; Rehemtulla, A.; Edgington, T.S. Cofactor residues lysine 165 and 166 are critical for protein substrate recognition by the tissue factor-factor VIIa protease complex. J. Biol. Chem. 1992, 267, 6375–6381. [Google Scholar] [PubMed]
- Roy, S.; Hass, P.E.; Bourell, J.H.; Henzel, W.J.; Vehar, G.A. Lysine residues 165 and 166 are essential for the cofactor function of tissue factor. J. Biol. Chem. 1991, 266, 22063–22066. [Google Scholar]
- Kelley, R.F.; Refino, C.J.; O’Connell, M.P.; Modi, N.B.; Sehl, P.; Lowe, D.; Pater, C.; Bunting, S. A Soluble Tissue Factor Mutant Is a Selective Anticoagulant and Antithrombotic Agent. Blood 1997, 89, 3219–3227. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Investigator’s Brochure tTF-NGR, v.15, 2020, on file.
- Brennan, F.R.; Baumann, A.; Blaich, G.; De Haan, L.; Fagg, R.; Kiessling, A.; Kronenberg, S.; Locher, M.; Milton, M.; Tibbitts, J.; et al. Nonclinical safety testing of biopharmaceuticals—Addressing current challenges of these novel and emerging therapies. Regul. Toxicol. Pharmacol. 2015, 73, 265–275. [Google Scholar] [CrossRef]
- Baldrick, P. Getting a molecule into the clinic: Nonclinical testing and starting dose considerations. Regul. Toxicol. Pharmacol. 2017, 89, 95–100. [Google Scholar] [CrossRef]
- Baldrick, P.; Cosenza, M.E.; Alapatt, T.; Bolon, B.; Rhodes, M.; Waterson, I. Toxicology Paradise: Sorting Out Adverse and Non-adverse Findings in Animal Toxicity Studies. Int. J. Toxicol. 2020, 39, 365–378. [Google Scholar] [CrossRef]
- Lima, B.S.; Videira, M.A. Toxicology and Biodistribution: The Clinical Value of Animal Biodistribution Studies. Mol. Ther-Methods Clin. Dev. 2018, 8, 183–197. [Google Scholar] [CrossRef] [Green Version]
- Hartung, T. Opinion versus evidence for the need to move away from animal testing. Altex 2017, 34, 193–200. [Google Scholar] [CrossRef] [Green Version]
- Sandoe, P.; Svendsen, O. Animal burdens versus human benefits--how should the ethical limits be drawn for use of animals as models in toxicology? Arch. Toxicol. 1998, 20, 31–40. [Google Scholar] [CrossRef]
- Guidance for Industry, Investigators, and Reviewers: Exploratory IND Studies; US Department of Health and Human Services, Food and Drug Administration, Center for Drug Evaluation and Research, Pharmacology/Toxicology: Rockville, MD, USA, 2006. Available online: https://www.fda.gov/regulatory-information/search-fda-guidance-documents/exploratory-ind-studies (accessed on 26 November 2020).
- Burt, T.; Young, G.; Lee, W.; Kusuhara, H.; Langer, O.; Rowland, M.; Sugiyama, Y. Phase 0/microdosing approaches: time for mainstream application in drug development? Nat. Rev. Drug Discov. 2020, 19, 801–818. [Google Scholar] [CrossRef] [PubMed]

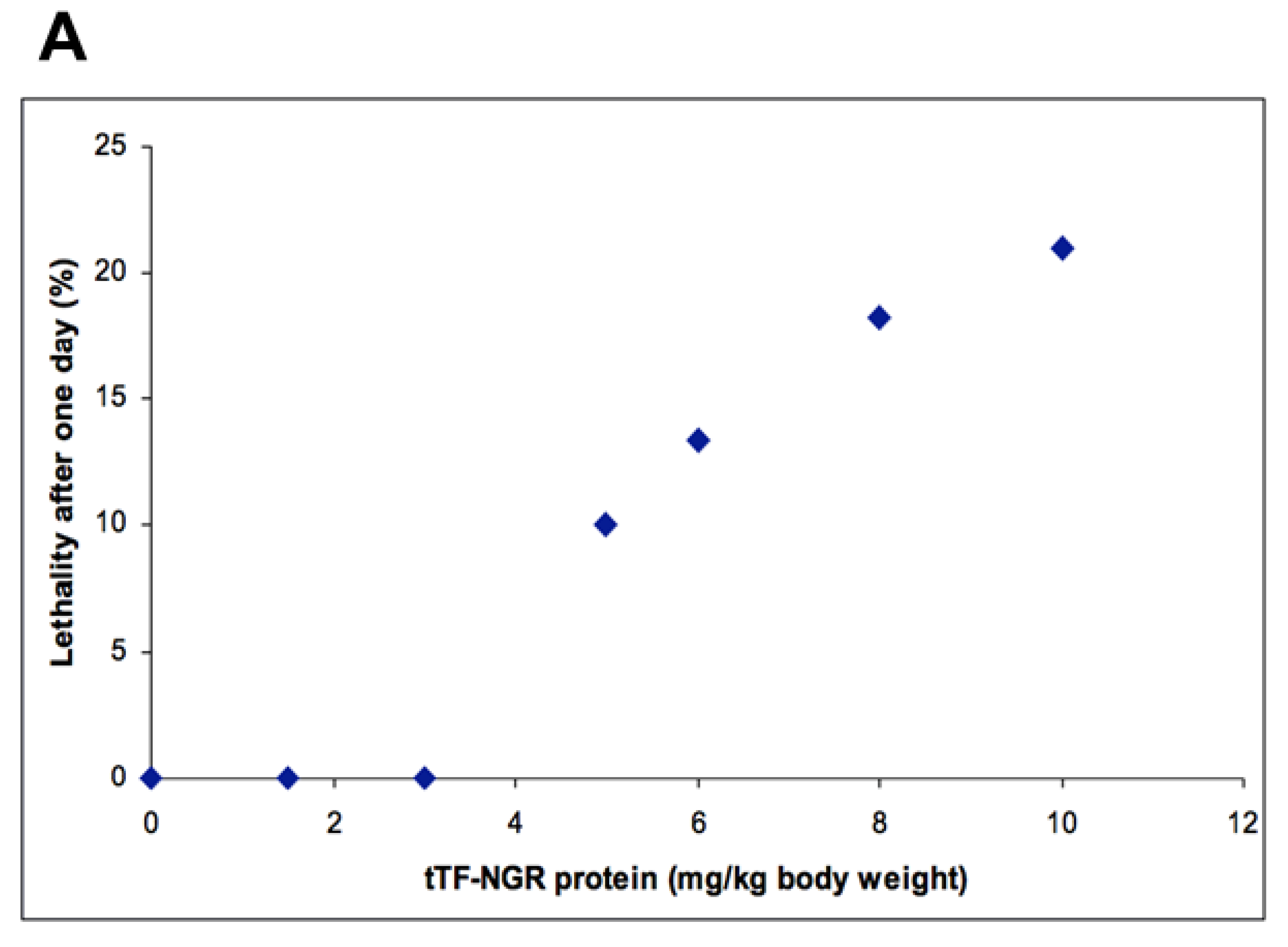
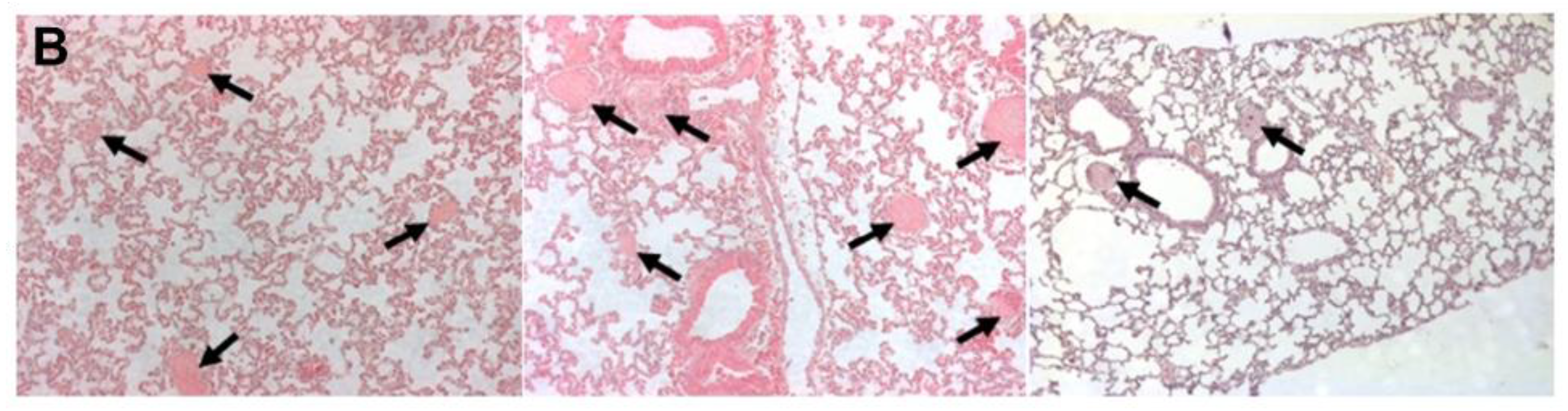
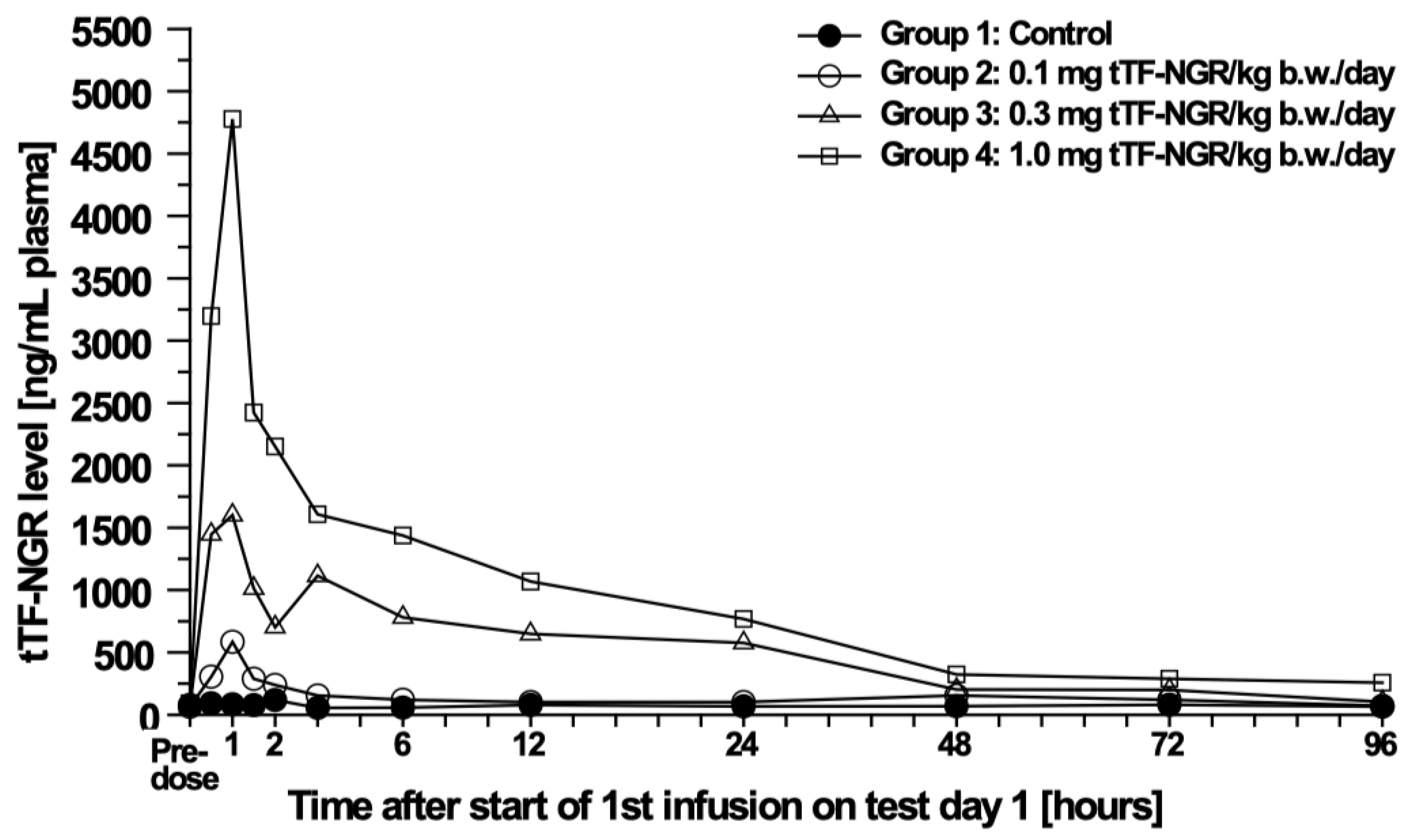
| Type of Study/Dose Duration | Administration Route | Species (tTF-NGR Dosage) | Animals n/ Sex/Group |
|---|---|---|---|
| Single Dose | |||
| In-house UKM | i.v. | mice (DRF, e.g., Figure 2) | 101/F+M |
| Study 1 | i.v. | mice (DRF 0; 1.8; 6.0; 18.0 mg/m2) | 40/F |
| Study 2 | i.v. | mice (DRF 0; 1.8; 6.0; 18.0 mg/m2) | 32/F |
| Study 3 | i.v. | mice (DRF 6; 20; 60 mg/m2) | 12/F |
| Study 4 | i.v. | rats (DRF 12; 36; 60; 120 mg/m2) | 10/F |
| Study 5 | i.v. | beagle dogs (DRF 20; 40; 80 mg/m2) | 6/F+M |
| Study 6 | i.v. | beagle dogs (DRF 0; 2; 6; 20 mg/m2) | 4/F+M |
| Repeat Dose | |||
| In-house UKM | i.v. | mice (1.0; 1.5 mg/kg b.w.) | in all therapeutic exp./F+M |
| Study 7 | i.v. | beagle dogs (0; 2; 6; 20 mg/m2) | 32/F+M |
| Study 8 | i.c. | guinea pigs (stock solution) | 30/M |
| Study 9 | i.p., i.v. | guinea pigs (stock solution) | 5/M |
| Genotoxicity NA | |||
| Reproductive Toxicity NA | |||
| Local Tolerance | |||
| In-house UKM | i.v. | mice (1.0; 1.5 mg/kg b.w.) | in all therapeutic |
| exp./F+M | |||
| Studies 5, 7 | i.v. | beagle dogs (DRF) | see above |
| Other Toxicity Studies 8, 9: Non-antigenicity + sensitization | Guinea pigs (stock solution) | see above |
| Mouse | Rat | Dog | ||||
|---|---|---|---|---|---|---|
| Findings | Effect Dose (mg/m2) | No Effect Dose (mg/m2) | Effect Dose (mg/m2) | No Effect Dose (mg/m2) | Effect Dose (mg/m2) | No Effect Dose (mg/m2) |
| Xenograft therapy trials | >3 (x6) | |||||
| Academic | >/=15 * LD10 | 9 * | ||||
| toxicology | ||||||
| local at all doses; | ||||||
| Study 3 | systemic | systemic 6 * | ||||
| 20 * | ||||||
| Study 1 | >18 * | |||||
| Study 2 | >18 * | |||||
| Study 4 | >120 * | |||||
| Study 5 | 20 (x5) | |||||
| Study 7 | >20 (x5) | |||||
| Non-Compartment Analysis of to tTF-NGR Test Day 1 (up to 24 h after Start of 1st Infusion) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Dose [mg/m2] | Animal no. | Cmax#1 [ng/mL] | t max#1 [h] | t1/2 alpha [h] | t1/2 terminal [h] | Kel [1/h] | AUC0-t last [ng*h/mL] | AUC0-t last/dose [h*kg*ng/mL/mg] | DPF |
| Males | |||||||||
| 2.0 | 11 | 723.6 | 1.0 | 1.17 | 40.23 | 0.02 | 3712.05 | 37,120.50 | |
| 12 | 531.2 | 1.0 | 1.12 | 36.87 | 0.02 | 2867.16 | 28,671.60 | ||
| 13 | 498.2 | 1.0 | 1.14 | 50.80 | 0.01 | 2922.71 | 29,227.10 | ||
| mean | 584.33 | 1.00 | 1.14 | 42.63 | 0.02 | 3167.31 | 31,673.07 | n.a. | |
| 6.0 | 17 | 1613.9 | 1.0 | 0.88 | 20.85 | 0.03 | 19,250.05 | 64,166.83 | |
| 18 | 1567.5 | 1.0 | 0.88 | 24.83 | 0.03 | 17,272.91 | 57,576.37 | ||
| 19 | 1715.8 | 0.5 | 0.77 | 37.11 | 0.02 | 16,311.34 | 54,371.13 | ||
| mean | 1632.40 | 0.83 | 0.84 | 27.60 | 0.03 | 17,611.43 | 58,704.78 | 1.85 | |
| 20.0 | 23 | 5335.5 | 1.0 | 1.54 | 28.15 | 0.02 | 31,557.99 | 31,557.99 | |
| 24 | 4533.8 | 1.0 | 1.05 | 23.20 | 0.03 | 29,021.97 | 29,021.97 | ||
| 25 | 4462.5 | 1.0 | 1.94 | 13.79 | 0.05 | 31,633.30 | 31,633.60 | ||
| mean | 4777.27 | 1.00 | 1.51 | 21.71 | 0.03 | 30,737.85 | 30,737.85 | 0.97 | |
| Females | |||||||||
| 2.0 | 14 | 385.5 | 1.0 | 0.86 | 47.90 | 0.01 | 2137.61 | 21,376.10 | |
| 15 | 330.7 | 1.0 | 0.80 | - | - | 2020.55 | 20,205.50 | ||
| 16 | 500.3 | 1.0 | 0.67 | 108.75 | 0.01 | 2022.04 | 20,220.40 | ||
| mean | 405.05 | 1.00 | 0.78 | 78.33 | 0.01 | 2060.07 | 20,600.67 | n.a. | |
| 6.0 | 20 | 1624.9 | 1.0 | 1.68 | 18.27 | 0.04 | 15,059.23 | 50,197.43 | |
| 21 | 1292.5 | 1.0 | 2.49 | - | - | 12,383.58 | 41,278.60 | ||
| 22 | 1542.8 | 1.0 | 1.20 | - | - | 14,870.10 | 49,567.00 | ||
| mean | 1486.73 | 1.00 | 1.79 | 18.27 | 0.04 | 14,104.30 | 47,014.34 | 2.28 | |
| 20.0 | 28 | 3274.7 | 1.0 | 1.60 | 18.31 | 0.04 | 21,901.66 | 21,901.66 | |
| 29 | 3819.7 | 1.0 | 1.51 | 18.40 | 0.04 | 21,570.02 | 21,570.02 | ||
| 30 | 3309.1 | 1.0 | 1.19 | 28.80 | 0.02 | 22,450.51 | 22,450.51 | ||
| mean | 3467.83 | 1.00 | 1.43 | 21.84 | 0.03 | 21,974.06 | 21,974.06 | 1.07 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Berdel, W.E.; Harrach, S.; Brand, C.; Brömmel, K.; Berdel, A.F.; Hintelmann, H.; Schliemann, C.; Schwöppe, C. Animal Safety, Toxicology, and Pharmacokinetic Studies According to the ICH S9 Guideline for a Novel Fusion Protein tTF-NGR Targeting Procoagulatory Activity into Tumor Vasculature: Are Results Predictive for Humans? Cancers 2020, 12, 3536. https://doi.org/10.3390/cancers12123536
Berdel WE, Harrach S, Brand C, Brömmel K, Berdel AF, Hintelmann H, Schliemann C, Schwöppe C. Animal Safety, Toxicology, and Pharmacokinetic Studies According to the ICH S9 Guideline for a Novel Fusion Protein tTF-NGR Targeting Procoagulatory Activity into Tumor Vasculature: Are Results Predictive for Humans? Cancers. 2020; 12(12):3536. https://doi.org/10.3390/cancers12123536
Chicago/Turabian StyleBerdel, Wolfgang E., Saliha Harrach, Caroline Brand, Kathrin Brömmel, Andrew F. Berdel, Heike Hintelmann, Christoph Schliemann, and Christian Schwöppe. 2020. "Animal Safety, Toxicology, and Pharmacokinetic Studies According to the ICH S9 Guideline for a Novel Fusion Protein tTF-NGR Targeting Procoagulatory Activity into Tumor Vasculature: Are Results Predictive for Humans?" Cancers 12, no. 12: 3536. https://doi.org/10.3390/cancers12123536
APA StyleBerdel, W. E., Harrach, S., Brand, C., Brömmel, K., Berdel, A. F., Hintelmann, H., Schliemann, C., & Schwöppe, C. (2020). Animal Safety, Toxicology, and Pharmacokinetic Studies According to the ICH S9 Guideline for a Novel Fusion Protein tTF-NGR Targeting Procoagulatory Activity into Tumor Vasculature: Are Results Predictive for Humans? Cancers, 12(12), 3536. https://doi.org/10.3390/cancers12123536





