Multidisciplinary Clinical Approach to Cancer Patients with Immune-Related Adverse Events Induced by Checkpoint Inhibitors
Abstract
Simple Summary
Abstract
1. Introduction
2. Coordinated Management of irAEs
2.1. Skin
2.2. Gastrointestinal Tract
2.3. Lung
2.4. Rheumatic and Systemic Autoimmune Syndromes
2.5. Endocrine
2.6. Liver
2.7. Nervous System
2.8. Eye
2.9. Heart
2.10. Muscle
2.11. Kidney
2.12. Haematological
3. Critical Care of Severe irAEs
4. Nursing and Immunotherapy
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Palucka, A.K.; Coussens, L.M. The Basis of Oncoimmunology. Cell 2016, 164, 1233–1247. [Google Scholar] [CrossRef] [PubMed]
- Krummel, M.F.; Allison, J.P. CD28 and CTLA-4 have opposing effects on the response of T cells to stimulation. J. Exp. Med. 1995, 182, 459–465. [Google Scholar] [CrossRef] [PubMed]
- Okazaki, T.; Honjo, T. PD-1 and PD-1 ligands: From discovery to clinical application. Int. Immunol. 2007, 19, 813–824. [Google Scholar] [CrossRef] [PubMed]
- Wilky, B. Immune checkpoint inhibitors: The linchpins of modern immunotherapy. Immunol. Rev. 2019, 290, 6–23. [Google Scholar] [CrossRef] [PubMed]
- Qureshi, O.S.; Zheng, Y.; Nakamura, K.; Attridge, K.; Manzotti, C.; Schmidt, E.M.; Baker, J.; Jeffery, L.E.; Kaur, S.; Briggs, Z.; et al. Trans-Endocytosis of CD80 and CD86: A Molecular Basis for the Cell-Extrinsic Function of CTLA-4. Science 2011, 332, 600–603. [Google Scholar] [CrossRef]
- Topalian, S.L.; Hodi, F.S.; Brahmer, J.R.; Gettinger, S.N.; Smith, D.C.; McDermott, D.F.; Powderly, J.D.; Carvajal, R.D.; Sosman, J.A.; Atkins, M.B.; et al. Safety, Activity, and Immune Correlates of Anti–PD-1 Antibody in Cancer. N. Engl. J. Med. 2012, 366, 2443–2454. [Google Scholar] [CrossRef] [PubMed]
- Khan, S.; Gerber, D.E. Autoimmunity, checkpoint inhibitor therapy and immune-related adverse events: A review. Semin. Cancer Biol. 2020, 64, 93–101. [Google Scholar] [CrossRef]
- Martins, F.; Sofiya, L.; Sykiotis, G.P.; Lamine, F.; Maillard, M.; Fraga, M.; Shabafrouz, K.; Ribi, C.; Cairoli, A.; Guex-Crosier, Y.; et al. Adverse effects of immune-checkpoint inhibitors: Epidemiology, management and surveillance. Nat. Rev. Clin. Oncol. 2019, 16, 563–580. [Google Scholar] [CrossRef]
- Centanni, M.; Moes, D.J.A.R.; Trocóniz, I.F.; Ciccolini, J.; van Hasselt, J.G.C. Clinical Pharmacokinetics and Pharmacodynamics of Immune Checkpoint Inhibitors. Clin. Pharmacokinet. 2019, 58, 835–857. [Google Scholar] [CrossRef]
- Silbermann, M.; Pitsillides, B.; Al-Alfi, N.; Omran, S.; Al-Jabri, K.; ElShamy, K.; Ghrayeb, I.; Livneh, J.; Daher, M.; Charalambous, H.; et al. Multidisciplinary care team for cancer patients and its implementation in several Middle Eastern countries. Ann. Oncol. 2013, 24, vii41–vii47. [Google Scholar] [CrossRef]
- Selby, P.; Popescu, R.; Lawler, M.; Butcher, H.; Costa, A. The Value and Future Developments of Multidisciplinary Team Cancer Care. Am. Soc. Clin. Oncol. Educ. Book 2019, 39, 332–340. [Google Scholar] [CrossRef] [PubMed]
- Haanen, J.; Carbonnel, F.; Robert, C.; Kerr, K.; Peters, S.; Larkin, J.; Jordan, K. Management of toxicities from immunotherapy: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann. Oncol. 2017, 28, iv119–iv142. [Google Scholar] [CrossRef] [PubMed]
- Arnaud-Coffin, P.; Maillet, D.; Gan, H.K.; Stelmes, J.J.; You, B.; Dalle, S.; Péron, J. A systematic review of adverse events in randomized trials assessing immune checkpoint inhibitors. Int. J. Cancer 2019, 145, 639–648. [Google Scholar] [CrossRef] [PubMed]
- Sibaud, V. Dermatologic Reactions to Immune Checkpoint Inhibitors: Skin Toxicities and Immunotherapy. Am. J. Clin. Dermatol. 2018, 19, 345–361. [Google Scholar] [CrossRef] [PubMed]
- Postow, M.A.; Sidlow, R.; Hellmann, M.D. Immune-Related Adverse Events Associated with Immune Checkpoint Blockade. N. Engl. J. Med. 2018, 378, 158–168. [Google Scholar] [CrossRef]
- Sanlorenzo, M.; Vujic, I.; Daud, A.; Algazi, A.; Gubens, M.A.; Luna, S.A.; Lin, K.; Quaglino, P.; Rappersberger, K.; Ortiz-Urda, S. Pembrolizumab Cutaneous Adverse Events and Their Association With Disease Progression. JAMA Dermatol. 2015, 151, 1206–1212. [Google Scholar] [CrossRef]
- Teulings, H.E.; Limpens, J.; Jansen, S.N.; Zwinderman, A.H.; Reitsma, J.B.; Spuls, P.I.; Luiten, R.M. Vitiligo-Like Depigmentation in Patients With Stage III-IV Melanoma Receiving Immunotherapy and Its Association With Survival: A Systematic Review and Meta-Analysis. J. Clin. Oncol. 2015, 33, 773–781. [Google Scholar] [CrossRef]
- Boada, A.; Carrera, C.; Segura, S.; Collgros, H.; Pasquali, P.; Bodet, D.; Puig, S.; Malvehy, J. Cutaneous toxicities of new treatments for melanoma. Clin. Transl. Oncol. 2018, 20, 1373–1384. [Google Scholar] [CrossRef]
- Khoja, L.; Day, D.; Chen, T.W.W.; Siu, L.L.; Hansen, A.R. Tumour- and class-specific patterns of immune-related adverse events of immune checkpoint inhibitors: A systematic review. Ann. Oncol. 2017, 28, 2377–2385. [Google Scholar] [CrossRef]
- Wang, D.Y.; Ye, F.; Zhao, S.; Johnson, D.B. Incidence of immune checkpoint inhibitor-related colitis in solid tumor patients: A systematic review and meta-analysis. OncoImmunology 2017, 6, e1344805. [Google Scholar] [CrossRef]
- Tandon, P.; Bourassa-Blanchette, S.; Bishay, K.; Parlow, S.; Laurie, S.A.; McCurdy, J.D. The Risk of Diarrhea and Colitis in Patients With Advanced Melanoma Undergoing Immune Checkpoint Inhibitor Therapy. J. Immunother. 2018, 41, 101–108. [Google Scholar] [CrossRef] [PubMed]
- Soularue, E.; Lepage, P.; Colombel, J.F.; Coutzac, C.; Faleck, D.M.; Marthey, L.; Collins, M.; Chaput, N.; Robert, C.; Carbonnel, F. Enterocolitis due to immune checkpoint inhibitors: A systematic review. Gut 2018, 67, 2056–2067. [Google Scholar] [CrossRef] [PubMed]
- Kim, K.W.; Ramaiya, N.H.; Krajewski, K.M.; Shinagare, A.B.; Howard, S.A.; Jagannathan, J.P.; Ibrahim, N. Ipilimumab-Associated Colitis: CT Findings. Am. J. Roentgenol. 2013, 200, W468–W474. [Google Scholar] [CrossRef] [PubMed]
- De Martin, E.; Michot, J.M.; Papouin, B.; Champiat, S.; Mateus, C.; Lambotte, O.; Roche, B.; Antonini, T.M.; Coilly, A.; Laghouati, S.; et al. Characterization of liver injury induced by cancer immunotherapy using immune checkpoint inhibitors. J. Hepatol. 2018, 68, 1181–1190. [Google Scholar] [CrossRef]
- Verschuren, E.C.; Eertwegh, A.J.V.D.; Wonders, J.; Slangen, R.M.; van Delft, F.; van Bodegraven, A.; Neefjes-Borst, A.; de Boer, N.K.; Information, P.E.K.F.C. Clinical, Endoscopic, and Histologic Characteristics of Ipilimumab-Associated Colitis. Clin. Gastroenterol. Hepatol. 2016, 14, 836–842. [Google Scholar] [CrossRef]
- Abu-Sbeih, H.; Ali, F.S.; Luo, W.; Qiao, W.; Raju, G.S.; Wang, Y. Importance of endoscopic and histological evaluation in the management of immune checkpoint inhibitor-induced colitis. J. Immunother. Cancer 2018, 6, 95. [Google Scholar] [CrossRef]
- Jain, A.; Lipson, E.J.; Sharfman, W.H.; Brant, S.R.; Lazarev, M.G. Colonic ulcerations may predict steroid-refractory course in patients with ipilimumab-mediated enterocolitis. World J. Gastroenterol. 2017, 23, 2023–2028. [Google Scholar] [CrossRef]
- Johnston, R.L.; Lutzky, J.; Chodhry, A.; Barkin, J.S. Cytotoxic T-Lymphocyte-Associated Antigen 4 Antibody-Induced Colitis and Its Management with Infliximab. Dig. Dis. Sci. 2008, 54, 2538–2540. [Google Scholar] [CrossRef]
- Hillock, N.T.; Heard, S.; Kichenadasse, G.; Hill, C.L.; Andrews, J. Infliximab for ipilimumab-induced colitis: A series of 13 patients. Asia-Pacific J. Clin. Oncol. 2017, 13, e284–e290. [Google Scholar] [CrossRef]
- Collins, M.; Soularue, E.; Marthey, L.; Carbonnel, F. Management of Patients With Immune Checkpoint Inhibitor-Induced Enterocolitis: A Systematic Review. Clin. Gastroenterol. Hepatol. 2020, 18, 1393–1403. [Google Scholar] [CrossRef]
- Bergqvist, V.; Hertervig, E.; Gedeon, P.; Kopljar, M.; Griph, H.; Kinhult, S.; Carneiro, A.; Marsal, J. Vedolizumab treatment for immune checkpoint inhibitor-induced enterocolitis. Cancer Immunol. Immunother. 2017, 66, 581–592. [Google Scholar] [CrossRef]
- Abu-Sbeih, H.; Ali, F.S.; Alsaadi, D.; Jennings, J.; Luo, W.; Gong, Z.; Richards, D.M.; Charabaty, A.; Wang, Y. Outcomes of vedolizumab therapy in patients with immune checkpoint inhibitor–induced colitis: A multi-center study. J. Immunother. Cancer 2018, 6, 142. [Google Scholar] [CrossRef] [PubMed]
- Disayabutr, S.; Calfee, C.S.; Collard, H.R.; Wolters, P.J. Interstitial lung diseases in the hospitalized patient. BMC Med. 2015, 13, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Nishino, M.; Giobbie-Hurder, A.; Hatabu, H.; Ramaiya, N.H.; Hodi, F.S. Incidence of Programmed Cell Death 1 Inhibitor–Related Pneumonitis in Patients With Advanced Cancer. JAMA Oncol. 2016, 2, 1607–1616. [Google Scholar] [CrossRef] [PubMed]
- Naidoo, J.; Wang, X.; Woo, K.M.; Iyriboz, T.; Halpenny, D.; Cunningham, J.; Chaft, J.E.; Segal, N.H.; Callahan, M.K.; Lesokhin, A.M.; et al. Pneumonitis in Patients Treated With Anti–Programmed Death-1/Programmed Death Ligand 1 Therapy. J. Clin. Oncol. 2017, 35, 709–717. [Google Scholar] [CrossRef]
- Delaunay, M.; Cadranel, J.; Lusque, A.; Meyer, N.; Gounaut, V.; Moro-Sibilot, D.; Michot, J.M.; Raimbourg, J.; Girard, N.; Guisier, F.; et al. Immune-checkpoint inhibitors associated with interstitial lung disease in cancer patients. Eur. Respir. J. 2017, 50, 1700050. [Google Scholar] [CrossRef]
- Calabrese, L.H.; Calabrese, C.; Cappelli, L.C. Rheumatic immune-related adverse events from cancer immunotherapy. Nat. Rev. Rheumatol. 2018, 14, 569–579. [Google Scholar] [CrossRef]
- Weiden, J.; Tel, J.; Figdor, C.G. Synthetic immune niches for cancer immunotherapy. Nat. Rev. Immunol. 2018, 18, 212–219. [Google Scholar] [CrossRef]
- Calabrese, C.; Kirchner, E.; Kontzias, K.; Velcheti, V.; Calabrese, L.H. Rheumatic immune-related adverse events of checkpoint therapy for cancer: Case series of a new nosological entity. RMD Open 2017, 3, e000412. [Google Scholar] [CrossRef]
- Brahmer, J.R.; Lacchetti, C.; Schneider, B.J.; Atkins, M.B.; Brassil, K.J.; Caterino, J.M.; Chau, I.; Ernstoff, M.S.; Gardner, J.M.; Ginex, P.; et al. Management of immune-related adverse events in patients treated with immune checkpoint inhibitor therapy: American Society of Clinical Oncology Clinical Practice Guideline. J. Clin. Oncol. 2018, 36, 1714–1768. [Google Scholar] [CrossRef]
- Cappelli, L.C.; Dorak, M.T.; Bettinotti, M.P.; O Bingham, C.; Shah, A.A. Association of HLA-DRB1 shared epitope alleles and immune checkpoint inhibitor-induced inflammatory arthritis. Rheumatol. 2018, 58, 476–480. [Google Scholar] [CrossRef] [PubMed]
- Marin-Acevedo, J.A.; Chirila, R.M.; Dronca, R.S. Immune Checkpoint Inhibitor Toxicities. Mayo Clin. Proc. 2019, 94, 1321–1329. [Google Scholar] [CrossRef] [PubMed]
- Crout, T.M.; Lennep, D.S.; Kishore, S.; Majithia, V. Systemic Vasculitis Associated With Immune Check Point Inhibition: Analysis and Review. Curr. Rheumatol. Rep. 2019, 21, 28. [Google Scholar] [CrossRef] [PubMed]
- Richter, M.D.; Crowson, C.; Kottschade, L.A.; Finnes, H.D.; Markovic, S.N.; Thanarajasingam, U. Rheumatic Syndromes Associated With Immune Checkpoint Inhibitors: A Single-Center Cohort of Sixty-One Patients. Arthritis Rheumatol. 2019, 71, 468–475. [Google Scholar] [CrossRef]
- Villarreal-Compagny, M.; Iglesias, P.; Marco-Hernández, H.; Milisenda, J.C.; Casanova-Molla, J.; Hernández-Rodríguez, J.; Puig, S.; Carrera, C.; Prieto-González, S. ANCA-associated Vasculitic Neuropathy During Treatment With Ipilimumab. Rheumatology 2020, 59, 251–252. [Google Scholar] [CrossRef]
- Kostine, M.; Rouxel, L.; Barnetche, T.; Veillon, R.; Martin, F.; Dutriaux, C.; Dousset, L.; Pham-Ledard, A.; Prey, S.; Beylot-Barry, M.; et al. Rheumatic disorders associated with immune checkpoint inhibitors in patients with cancer—clinical aspects and relationship with tumour response: A single-centre prospective cohort study. Ann. Rheum. Dis. 2018, 77, 393–398. [Google Scholar] [CrossRef]
- Abdel-Wahab, N.; Shah, M.; Lopez-Olivo, M.A.; Suarez-Almazor, M.E. Use of Immune Checkpoint Inhibitors in the Treatment of Patients With Cancer and Preexisting Autoimmune Disease. Ann. Intern. Med. 2018, 168, 121–130. [Google Scholar] [CrossRef]
- Chang, L.S.; Barroso-Sousa, R.; Tolaney, S.M.; Hodi, F.S.; Kaiser, U.B.; Min, L. Endocrine Toxicity of Cancer Immunotherapy Targeting Immune Checkpoints. Endocr. Rev. 2019, 40, 17–65. [Google Scholar] [CrossRef]
- Castinetti, F.; Albarel, F.; Archambeaud, F.; Bertherat, J.; Bouillet, B.; Buffier, P.; Briet, C.; Cariou, B.; Caron, P.; Chabre, O.; et al. French Endocrine Society Guidance on endocrine side effects of immunotherapy. Endocr.-Related Cancer 2018, 26, G1–G18. [Google Scholar] [CrossRef]
- Faje, A. Immunotherapy and hypophysitis: Clinical presentation, treatment, and biologic insights. Pituitary 2016, 19, 82–92. [Google Scholar] [CrossRef]
- Osorio, J.C.; Ni, A.; Chaft, J.E.; Pollina, R.; Kasler, M.K.; Stephens, D.; Rodriguez, C.; Cambridge, L.; Rizvi, H.; Wolchok, J.D.; et al. Antibody-mediated Thyroid Dysfunction During T-cell Checkpoint Blockade in Patients With Non-Small-Cell Lung Cancer. Ann. Oncol. 2017, 28, 583–589. [Google Scholar] [CrossRef] [PubMed]
- González-Rodríguez, E.; Rodríguez-Abreu, D.; On behalf of the Spanish Group for Cancer Immuno-Biotherapy (GETICA). Immune Checkpoint Inhibitors: Review and Management of Endocrine Adverse Events. Oncologist 2016, 21, 804–816. [Google Scholar] [CrossRef]
- De Filette, J.; Andreescu, C.E.; Cools, F.; Bravenboer, B.; Velkeniers, B. A Systematic Review and Meta-Analysis of Endocrine-Related Adverse Events Associated with Immune Checkpoint Inhibitors. Horm. Metab. Res. 2019, 51, 145–156. [Google Scholar] [CrossRef] [PubMed]
- Di Dalmazi, G.; Ippolito, S.; Lupi, I.; Caturegli, P. Hypophysitis induced by immune checkpoint inhibitors: A 10-year assessment. Expert Rev. Endocrinol. Metab. 2019, 14, 381–398. [Google Scholar] [CrossRef]
- Nishida, N.; Kudo, M. Liver damage related to immune checkpoint inhibitors. Hepatol. Int. 2019, 13, 248–252. [Google Scholar] [CrossRef]
- Wenjun, W.; Lie, P.; Guo, M.; He, J. Risk of hepatotoxicity in cancer patients treated with immune checkpoint inhibitors: A systematic review and meta-analysis of published data. Int. J. Cancer 2017, 141, 1018–1028. [Google Scholar] [CrossRef]
- Zen, Y.; Yeh, M.M. Checkpoint inhibitor-induced liver injury: A novel form of liver disease emerging in the era of cancer immunotherapy. Semin. Diagn. Pathol. 2019, 36, 434–440. [Google Scholar] [CrossRef]
- Cunningham, M.; Iafolla, M.; Kanjanapan, Y.; Cerocchi, O.; Butler, M.; Siu, L.; Bedard, P.; Ross, K.; Spreafico, A.; Feld, J. PS-139-Liver enzyme elevations and hepatotoxicity in patients treated with checkpoint inhibitor immunotherapy. J. Hepatol. 2019, 70, e89. [Google Scholar] [CrossRef]
- Andrade, R.J.; Aithal, G.P.; Björnsson, E.S.; Kaplowitz, N.; Kullak-Ublick, G.A.; Larrey, D.; Karlsen, T.H. EASL Clinical Practice Guidelines: Drug-induced liver injury. J. Hepatol. 2019, 70, 1222–1261. [Google Scholar] [CrossRef]
- Kawakami, H.; Tanizaki, J.; Tanaka, K.; Haratani, K.; Hayashi, H.; Takeda, M.; Kamata, K.; Takenaka, M.; Kimura, M.; Chikugo, T.; et al. Imaging and clinicopathological features of nivolumab-related cholangitis in patients with non-small cell lung cancer. Investig. New Drugs 2017, 35, 529–536. [Google Scholar] [CrossRef]
- Sangro, B.; Chan, S.L.; Meyer, T.; Reig, M.; El-Khoueiry, A.; Galle, P.R. Diagnosis and management of toxicities of immune checkpoint inhibitors in hepatocellular carcinoma. J. Hepatol. 2020, 72, 320–341. [Google Scholar] [CrossRef] [PubMed]
- Riveiro-Barciela, M.; Mínguez, B.; LLarch, N.; Rimola, J.; Sala, M.; Reig, M.; Londoño, M.C. Efectes Adversos Hepàtics de la Immunoteràpia i el seu Impacte en el Maneig de Pacients amb Carcinoma Hepatocel·Lular. Available online: http://www.scdigestologia.org/docs/docs_posicionament/23_Efectes_adversos_hepatics_immunoterapia.pdf (accessed on 6 February 2020).
- Cuzzubbo, S.; Javeri, F.; Tissier, M.; Roumi, A.; Barlog, C.; Doridam, J.; Lebbe, C.; Belin, C.; Ursu, R.; Carpentier, A. Neurological adverse events associated with immune checkpoint inhibitors: Review of the literature. Eur. J. Cancer 2017, 73, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Spain, L.; Walls, G.; Julve, M.; O’Meara, K.; Schmid, T.; Kalaitzaki, E.; Turajlic, S.; Gore, M.; Rees, J.; Larkin, J. Neurotoxicity from immune-checkpoint inhibition in the treatment of melanoma: A single centre experience and review of the literature. Ann. Oncol. 2017, 28, 377–385. [Google Scholar] [CrossRef] [PubMed]
- Yshii, L.M.; Hohlfeld, R.; Liblau, R.S. Inflammatory CNS disease caused by immune checkpoint inhibitors: Status and perspectives. Nat. Rev. Neurol. 2017, 13, 755–763. [Google Scholar] [CrossRef] [PubMed]
- Johnson, D.B.; Manouchehri, A.; Haugh, A.M.; Quach, H.T.; Balko, J.M.; Lebrun-Vignes, B.; Mammen, A.; Moslehi, J.J.; Salem, J.E. Neurologic toxicity associated with immune checkpoint inhibitors: A pharmacovigilance study. J. Immunother. Cancer 2019, 7, 134. [Google Scholar] [CrossRef]
- Graus, F.; Dalmau, J. Paraneoplastic neurological syndromes in the era of immune-checkpoint inhibitors. Nat. Rev. Clin. Oncol. 2019, 16, 535–548. [Google Scholar] [CrossRef]
- Manson, G.; Maria, A.T.J.; Poizeau, F.; Danlos, F.X.; Kostine, M.; Brosseau, S.; Aspeslagh, S.; du Rusquec, P.; Roger, M.; Pallix-Guyot, M.; et al. Worsening and newly diagnosed paraneoplastic syndromes following anti-PD-1 or anti-PD-L1 immunotherapies, a descriptive study. J. Immunother. Cancer 2019, 7, 337. [Google Scholar] [CrossRef]
- Larkin, J.; Chmielowski, B.; Lao, C.D.; Hodi, F.S.; Sharfman, W.; Weber, J.; Suijkerbuijk, K.P.M.; Azevedo, S.; Li, H.; Reshef, D.; et al. Neurologic Serious Adverse Events Associated with Nivolumab Plus Ipilimumab or Nivolumab Alone in Advanced Melanoma, Including a Case Series of Encephalitis. The Oncologist 2017, 22, 709–718. [Google Scholar] [CrossRef]
- Taylor, A.W. Ocular immune privilege. Eye 2009, 23, 1885–1889. [Google Scholar] [CrossRef]
- Dalvin, L.A.; Shields, C.L.; Orloff, M.; Sato, T.; Shields, J.A. Checkpoint inhibitor immune therapy: systemic indications and ophthalmic side effects. Retina 2018, 38, 1063–1078. [Google Scholar] [CrossRef]
- Moorthy, R.S.; Moorthy, M.S.; Cunningham, E.T. Drug-induced uveitis. Curr. Opin. Ophthalmol. 2018, 29, 588–603. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.M.; Materin, M.A.; Sznol, M.; Kluger, H.M.; Weiss, S.; Chow, J.; Stoessel, K.; Kombo, N.; Del Priore, L.; Pointdujour-Lim, R. Ophthalmic Immune-Related Adverse Events of Immunotherapy: A Single-Site Case Series. Ophthalmology 2019, 126, 1058–1062. [Google Scholar] [CrossRef] [PubMed]
- Davies, M.J.; Duffield, E.A. Safety of checkpoint inhibitors for cancer treatment: Strategies for patient monitoring and management of immune-mediated adverse events. Immuno Targets Ther. 2017, 6, 51–71. [Google Scholar] [CrossRef] [PubMed]
- Roberts, K.; Culleton, V.; Lwin, Z.; O’Byrne, K.; Hughes, B.G. Immune checkpoint inhibitors: Navigating a new paradigm of treatment toxicities. Asia-Pacific J. Clin. Oncol. 2017, 13, 277–288. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, A.T.; Elia, M.; Materin, M.A.; Sznol, M.; Chow, J. Cyclosporine for Dry Eye Associated With Nivolumab. Cornea 2016, 35, 399–401. [Google Scholar] [CrossRef] [PubMed]
- Cappelli, L.C.; Gutierrez, A.K.; Baer, A.N.; Albayda, J.; Manno, R.L.; Haque, U.; Lipson, E.J.; Bleich, K.B.; Shah, A.A.; Naidoo, J.; et al. Inflammatory arthritis and sicca syndrome induced by nivolumab and ipilimumab. Ann. Rheum. Dis. 2017, 76, 43–50. [Google Scholar] [CrossRef]
- Acaba-Berrocal, L.A.; Lucio-Alvarez, J.A.; Mashayekhi, A.; Ho, A.C.; Dunn, J.P.; Shields, C.L. Birdshot-like Chorioretinopathy Associated With Pembrolizumab Treatment. JAMA Ophthalmol. 2018, 136, 1205–1207. [Google Scholar] [CrossRef]
- Wang, W.; Lam, W.C.; Chen, L. Recurrent grade 4 panuveitis with serous retinal detachment related to nivolumab treatment in a patient with metastatic renal cell carcinoma. Cancer Immunol. Immunother. 2018, 68, 85–95. [Google Scholar] [CrossRef]
- Conrady, C.D.; LaRochelle, M.; Pecen, P.; Palestine, A.; Shakoor, A.; Singh, A. Checkpoint inhibitor-induced uveitis: A case series. Graefe’s Arch. Clin. Exp. Ophthalmol. 2017, 256, 187–191. [Google Scholar] [CrossRef]
- Makarious, D.; Horwood, K.; Coward, J. Myasthenia gravis: An emerging toxicity of immune checkpoint inhibitors. Eur. J. Cancer 2017, 82, 128–136. [Google Scholar] [CrossRef]
- Chang, E.; Sabichi, A.L.; Sada, Y.H. Myasthenia Gravis after Nivolumab Therapy for Squamous Cell Carcinoma of the Bladder. J. Immunother. 2017, 40, 114–116. [Google Scholar] [CrossRef] [PubMed]
- Hu, Y.B.; AME Academic Lung Cancer Cooperation Group; Zhang, Q.; Li, H.J.; Michot, J.M.; Liu, H.B.; Zhan, P.; Lv, T.F.; Song, Y. Evaluation of rare but severe immune related adverse effects in PD-1 and PD-L1 inhibitors in non-small cell lung cancer: A meta-analysis. Transl. Lung Cancer Res. 2017, 6, S8–S20. [Google Scholar] [CrossRef] [PubMed]
- Escudier, M.; Cautela, J.; Malissen, N.; Ancedy, Y.; Orabona, M.; Pinto, J.; Monestier, S.; Grob, J.J.; Scemama, U.; Jacquier, A.; et al. Clinical Features, Management, and Outcomes of Immune Checkpoint Inhibitor–Related Cardiotoxicity. Circulation 2017, 136, 2085–2087. [Google Scholar] [CrossRef] [PubMed]
- Lyon, A.R.; Yousaf, N.; Battisti, N.M.L.; Moslehi, J.; Larkin, J. Immune checkpoint inhibitors and cardiovascular toxicity. Lancet Oncol. 2018, 19, e447–e458. [Google Scholar] [CrossRef]
- Mahmood, S.S.; Fradley, M.G.; Cohen, J.V.; Nohria, A.; Reynolds, K.L.; Heinzerling, L.M.; Sullivan, R.J.; Damrongwatanasuk, R.; Chen, C.L.; Gupta, D.; et al. Myocarditis in Patients Treated With Immune Checkpoint Inhibitors. J. Am. Coll. Cardiol. 2018, 71, 1755–1764. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, S.; Ishikawa, N.; Konoeda, F.; Seki, N.; Fukushima, S.; Takahashi, K.; Uhara, H.; Hasegawa, Y.; Inomata, S.; Otani, Y.; et al. Nivolumab-related myasthenia gravis with myositis and myocarditis in Japan. Neurology 2017, 89, 1127–1134. [Google Scholar] [CrossRef] [PubMed]
- Montes, V.; Sousa, S.; Pita, F.; Guerreiro, R.; Carmona, C. Myasthenia Gravis Induced by Ipilimumab in a Patient With Metastatic Melanoma. Front. Neurol. 2018, 9, 150. [Google Scholar] [CrossRef] [PubMed]
- Anquetil, C.; Salem, J.E.; Lebrun-Vignes, B.; Johnson, D.B.; Mammen, A.L.; Stenzel, W.; Léonard-Louis, S.; Benveniste, O.; Moslehi, J.J.; Allenbach, Y. Immune Checkpoint Inhibitor–Associated Myositis. Circulation 2018, 138, 743–745. [Google Scholar] [CrossRef]
- Moreira, A.; Loquai, C.; Pföhler, C.; Kähler, K.C.; Knauss, S.; Heppt, M.V.; Gutzmer, R.; Dimitriou, F.; Meier, F.; Mitzel-Rink, H.; et al. Myositis and neuromuscular side-effects induced by immune checkpoint inhibitors. Eur. J. Cancer 2019, 106, 12–23. [Google Scholar] [CrossRef] [PubMed]
- Liewluck, T.; Kao, J.C.; Mauermann, M.L. PD-1 Inhibitor-associated Myopathies. J. Immunother. 2018, 41, 208–211. [Google Scholar] [CrossRef]
- Puzanov, I.; Society for Immunotherapy of Cancer Toxicity Management Working Group; Diab, A.; Abdallah, K.; Bingham, C.O.; Brogdon, C.; Dadu, R.; Hamad, L.; Kim, S.; Lacouture, M.E.; et al. Managing toxicities associated with immune checkpoint inhibitors: Consensus recommendations from the Society for Immunotherapy of Cancer (SITC) Toxicity Management Working Group. J. Immunother. Cancer 2017, 5, 95. [Google Scholar] [CrossRef] [PubMed]
- Varricchi, G.; Galdiero, M.R.; Marone, G.; Criscuolo, G.; Triassi, M.; Bonaduce, D.; Marone, G.; Tocchetti, C.G. Cardiotoxicity of immune checkpoint inhibitors. ESMO Open 2017, 2, e000247. [Google Scholar] [CrossRef] [PubMed]
- Zimmer, L.; Goldinger, S.M.; Hofmann, L.; Loquai, C.; Ugurel, S.; Thomas, I.; Schmidgen, M.I.; Gutzmer, R.; Utikal, J.S.; Göppner, D.; et al. Neurological, respiratory, musculoskeletal, cardiac and ocular side-effects of anti-PD-1 therapy. Eur. J. Cancer 2016, 210–225. [Google Scholar] [CrossRef] [PubMed]
- Ganatra, S.; Neilan, T.G. Immune Checkpoint Inhibitor-Associated Myocarditis. Oncol. 2018, 23, 879–886. [Google Scholar] [CrossRef]
- Kao, J.C.; Liao, B.; Markovic, S.N.; Klein, C.J.; Naddaf, E.; Staff, N.P.; Liewluck, T.; Hammack, J.E.; Sandroni, P.; Finnes, H.; et al. Neurological Complications Associated With Anti–Programmed Death 1 (PD-1) Antibodies. JAMA Neurol. 2017, 74, 1216–1222. [Google Scholar] [CrossRef]
- Touat, M.; Maisonobe, T.; Knauss, S.; Salem, O.B.H.; Hervier, B.; Auré, K.; Szwebel, T.A.; Kramkimel, N.; Lethrosne, C.; Bruch, J.F.; et al. Immune checkpoint inhibitor-related myositis and myocarditis in patients with cancer. Neurology 2018, 91, e985–e994. [Google Scholar] [CrossRef]
- Wanchoo, R.; Karam, S.; Uppal, N.N.; Barta, V.; Deray, G.; DeVoe, C.; Launay-Vacher, V.; Jhaveri, K.D.; on behalf of Cancer and Kidney International Network Workgroup on Immune Checkpoint Inhibitors. Adverse Renal Effects of Immune Checkpoint Inhibitors: A Narrative Review. Am. J. Nephrol. 2017, 45, 160–169. [Google Scholar] [CrossRef]
- Xipell, M.; Victoria, I.; Hoffmann, V.; Villarreal, J.; García-Herrera, A.; Reig, O.; Rodas, L.; Blasco, M.; Poch, E.; Mellado, B.; et al. Acute tubulointerstitial nephritis associated with atezolizumab, an anti-programmed death-ligand 1 (pd-l1) antibody therapy. OncoImmunology 2018, 7, e1445952. [Google Scholar] [CrossRef]
- Perazella, M.A.; Shirali, A.C. Nephrotoxicity of Cancer Immunotherapies: Past, Present and Future. J. Am. Soc. Nephrol. 2018, 29, 2039–2052. [Google Scholar] [CrossRef]
- Cortazar, F.B.; Marrone, K.A.; Troxell, M.L.; Ralto, K.M.; Hoenig, M.P.; Brahmer, J.R.; Le, D.T.; Lipson, E.J.; Glezerman, I.G.; Wolchok, J.; et al. Clinicopathological features of acute kidney injury associated with immune checkpoint inhibitors. Kidney Int. 2016, 90, 638–647. [Google Scholar] [CrossRef]
- Barnett, R.; Barta, V.; Jhaveri, K.D. Preserved Renal-Allograft Function and the PD-1 Pathway Inhibitor Nivolumab. N. Engl. J. Med. 2017, 376, 191–192. [Google Scholar] [CrossRef] [PubMed]
- Tanios, G.E.; Doley, P.B.; Munker, R. Autoimmune hemolytic anemia associated with the use of immune checkpoint inhibitors for cancer: 68 cases from the Food and Drug Administration database and review. Eur. J. Haematol. 2019, 102, 157–162. [Google Scholar] [CrossRef] [PubMed]
- Merryman, R.W.; Kim, H.T.; Zinzani, P.L.; Carlo-Stella, C.; Ansell, S.M.; Perales, M.A.; Avigdor, A.; Halwani, A.S.; Houot, R.; Marchand, T.; et al. Safety and efficacy of allogeneic hematopoietic stem cell transplant after PD-1 blockade in relapsed/refractory lymphoma. Blood 2017, 129, 1380–1388. [Google Scholar] [CrossRef] [PubMed]
- Kroschinsky, F.; Intensive Care in Hematological and Oncological Patients (iCHOP) Collaborative, Group; Stölzel, F.; von Bonin, S.; Beutel, G.; Kochanek, M.; Kiehl, M.; Schellongowski, P. New drugs, new toxicities: Severe side effects of modern targeted and immunotherapy of cancer and their management. Crit. Care 2017, 21, 1–11. [Google Scholar] [CrossRef]
- Lemiale, V.; Grrr-Oh, G.D.R.E.R.R.D.P.D.H.; Meert, A.P.; Vincent, F.; Darmon, M.; Bauer, P.R.; van de Louw, A.; Azoulay, E. Severe toxicity from checkpoint protein inhibitors: What intensive care physicians need to know? Ann. Intensive Care 2019, 9, 1–16. [Google Scholar] [CrossRef]
- Assoun, S.; Lemiale, V.; Azoulay, É. Molecular targeted therapy-related life-threatening toxicity in patients with malignancies. A systematic review of published cases. Intensive Care Med. 2019, 45, 988–997. [Google Scholar] [CrossRef]
- Gutierrez, C.; McEvoy, C.; Munshi, L.; Stephens, R.S.; Detsky, M.E.; Nates, J.L.; Pastores, S.M. Critical Care Management of Toxicities Associated With Targeted Agents and Immunotherapies for Cancer. Crit. Care Med. 2020, 48, 10–21. [Google Scholar] [CrossRef]
- Lisberg, A.; Tucker, D.A.; Goldman, J.W.; Wolf, B.; Carroll, J.; Hardy, A.; Morris, K.; Linares, P.; Adame, C.; Spiegel, M.L.; et al. Treatment-Related Adverse Events Predict Improved Clinical Outcome in NSCLC Patients on KEYNOTE-001 at a Single Center. Cancer Immunol. Res. 2018, 6, 288–294. [Google Scholar] [CrossRef]
- Valverde, M.; Vidal, M.; Jansa, M. Therapeutic Education Didactic Techniques. Rev. Enferm. 2012, 35, 42–51. Available online: https://pubmed.ncbi.nlm.nih.gov/23157069/?from_single_result=Valverde+M%2C+Vidal+M%2C+Jansa+M.+Técnicas+didácticas+en+educacíon+terapéutic (accessed on 2 June 2020).
- Suter, P.M.; Suter, W.N. Patient Education. Timeless Principles of Learning: A Solid Foundation for Enhancing Chronic Disease Self-Management. Home Healthc. Nurse. 2008, 26, 82–88. Available online: https://pubmed.ncbi.nlm.nih.gov/18301109/ (accessed on 2 June 2020). [CrossRef]
- Jørgensen, C.R.; Thomsen, T.G.; Ross, L.; Dietz, S.M.; Therkildsen, S.; Groenvold, M.; Rasmussen, C.L.; Johnsen, A.T. What Facilitates “Patient Empowerment” in Cancer Patients During Follow-Up: A Qualitative Systematic Review of the Literature. Qual. Heal. Res. 2017, 28, 292–304. [Google Scholar] [CrossRef] [PubMed]

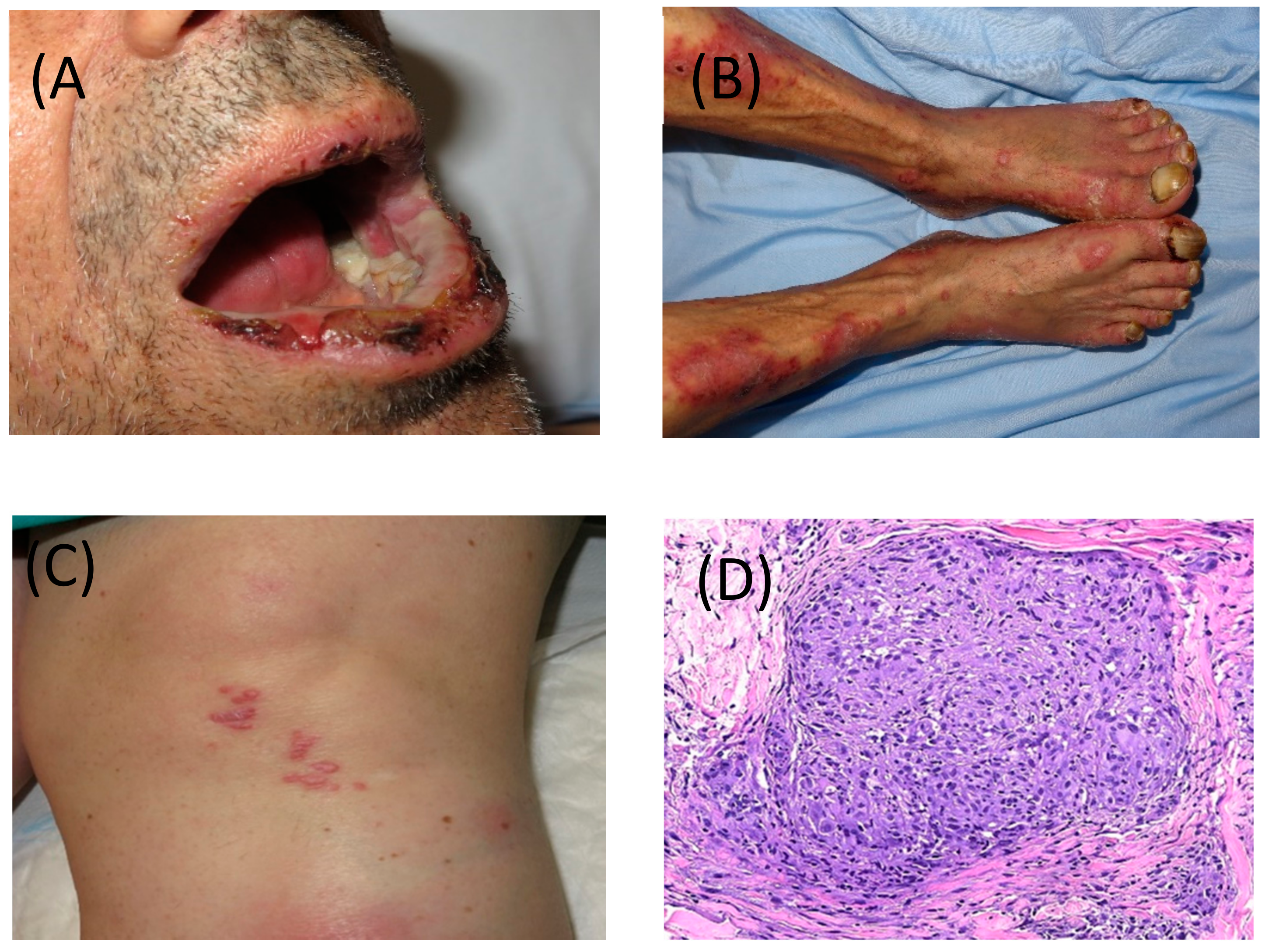
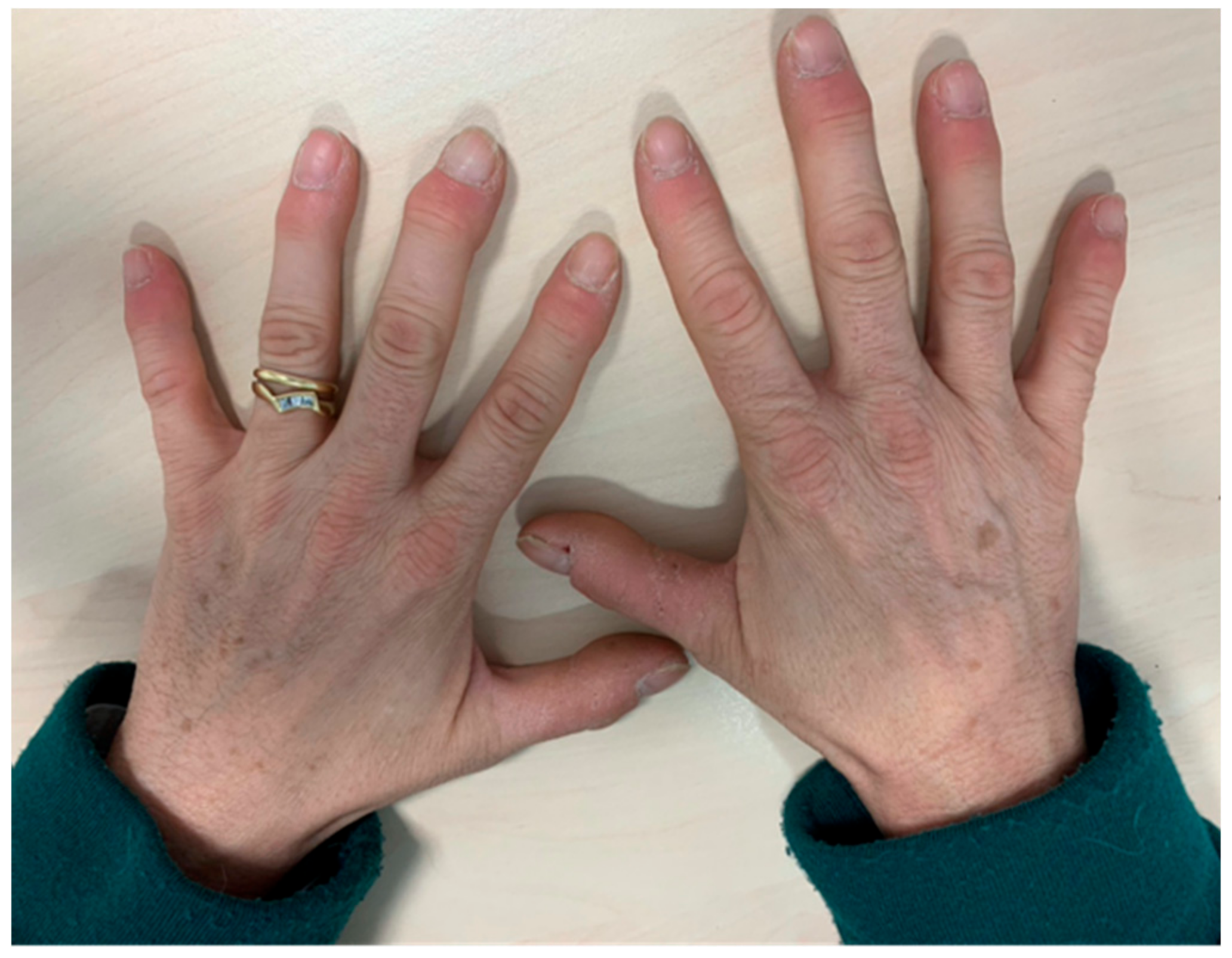
| Organ | Grade 1 | Grade 2 | Grade 3 | Grade 4 |
|---|---|---|---|---|
| Skin rash | -Physical examination and exclude other causes (viral rash, infection, other drug-induced rash); -Avoid skin irritants and sunlight; -Topical emollients and corticosteroids (mild strength) ± oral antihistamines for itching; -Continue ICPI treatment | -Consider dermatology consultation and skin biopsy; -Supportive management, as grade 1 + topical corticosteroides (moderate strength); | -Withhold ICPI treatment; -Management as grade 2 + oral prednisone (0.5–1 mg/kg); -For severe rash IV methylprednisolone (0.5–1 mg/kg) and convert to oral prednisone depending on response, wean over 2–4 weeks; -Resume ICPI at grade1/mild grade 2 after discussion with dermatologist; | -IV methylprednisolone (1–2 mg/kg); -Urgent dermatology consultation; -Discontinue ICPI treatment; |
| Colitis | -Rule out infections (see Section 2.2); -Symptomatic treatment with low-fibre diet, anti-diarrhoeals (loperamide), oral fluids; -Continue ICPI treatment; | -Rule out infections; -Consider colonoscopy (with biopsies); -Oral prednisone (1 mg/kg), tapered in 8–12 weeks; -If steroid-dependency consider IFX or VDZ; -GI consultation;-Withhold ICPI treatment; | -Rule out infections; -GI consultation; -Colonoscopy (with biopsies); -CT scan; -Hospital admission; -IV methylprednisolone (1 mg/kg); -Supportive measures; -If steroid-refractoriness (within 3–5 days) treat with IFX (vs. VDZ); -Discontinue ICPI treatment; | |
| Pneumonitis | -Symptomatic treatment; -Delay immunotherapy; - Monitoring; | -Withhold ICPI treatment; -Oral prednisone (1 mg/kg); | -Discontinue ICPI treatment; -IV methylprednisolone (2–4 mg/kg); | |
| Arthritis | -NSAID and acetaminophen; -Continue ICPI treatment; | -NSAID plus low doses of oral prednisone (< than 10 mg/day in tapering doses); -Continue ICPI treatment; | -Oral prednisone (0.5 mg/kg) in tapering doses; -Withhold ICPI treatment and resume upon symptom control; -If arthritis persists, consider MTX or LEF as maintenance treatment and steroid sparing agent; | -Consider IV methylprednisolone pulses (125 mg) followed by oral prednisone (0.5–1 mg/kg) in tapering doses; -Add MTX or LEF for persistent arthritis; -Discontinue ICPI treatment; |
| Renal | -Monitor renal function; -Check urine: sterile pyuria, WBC cast, proteinuria; | -Nephrology consultation; -Discontinue ICPI treatment; -Rule out other causes; -Monitoring; | -Nephrology consultation; -Renal biopsy; -IV methylprednisolone (1 mg/kg) if ATIN in the biopsy; -MMF if no response; | |
| Hepatitis | Monitoring | -Hepatology consultation; -Withhold ICPI; -Rule out other causes; -Monitoring; | -Hepatology consultation; -Check INR/albumin; -Liver biopsy; -Oral prednisone or IV methylprednisolone (0.5–1 mg/kg) if liver dysfunction *, severe inflammation in the liver biopsy or worsening (72 h) ;-AZA, MMF, or Tac if no response; | -Hepatology consultation; -Check INR/albumin; -Liver biopsy (should not delay treatment in hepatic encephalopathy or liver dysfunction *); -IV methylprednisolone (1 mg/kg/d); -AZA, MMF, or Tac if no response; |
| Neurological | -Delay immunotherapy; -Monitor symptoms; | -Central involvement: brain/spine MRI, CSF and antibody studies. Empiric antibacterial and antivirals; -Peripheral involvement: EMG, CK and antibody study; -Withhold ICPI treatment; -Neurology consultation; -Symptomatic treatment; -Oral prednisone (0.5–1 mg/kg); | -Central involvement: EEG, brain/ spine MRI, CSF and antibody studies. Empiric antibacterial and antivirals; -Peripheral involvement: EMG, CK and antibody study, vital capacity, cardiac monitoring to detect overlap syndromes; -Hospital admission, ICU monitoring or support; -Discontinue ICPI treatment; -Avoid medications that may worsen myasthenia; -IV methylprednisolone (1 gr × 3–5 days); -If partial or no improvement within one week IVIG (2 g/kg over 5 days) or plasma exchange; -Consider rituximab/ cyclophosphamide in refractory cases, paraneoplastic syndromes or proven antibody-mediated disease; | |
| Myopathy | -Monitor symptoms and muscle enzymes (aldolase and CK); | -Delay ICPI treatment; -Aldolase and CK. EMG, antibody study (specific and associated for myositis); -Whole body MRI and muscle biopsy; -Oral prednisone (0.5 mg/kg); | -Withhold ICPI treatment; -Aldolase and CK. EMG, antibody study (specific and associated for myositis); -Whole body MRI and muscle biopsy; -IV methylprednisolone(250 mg/day for 3 days) and then oral prednisone (0.5 mg/kg); | -Withhold ICPI treatment; -Aldolase and CK, EMG, antibody study (specific and associated for myositis); -Whole body MRI and muscle biopsy; -IV methylprednisolone (250 mg/day for 3 days) and then oral prednisone (0.5 mg/kg) plus IVIG or plasma exchange; |
| Blood disorders (Cytopoenia) | -Laboratory monitoring; -Continue ICPI treatment; | -Haematology consultation -Extended laboratory study ± bone marrow study; -Continue ICPI treatment; | -Withhold ICPI treatment; -Consider oral prednisone (1 mg/kg), haematopoietic growth factors, IVIG, rituximab or immunosuppressive drugs according to disorder type and severity; | -Blood disorders (Cytopoenia) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Londoño, M.-C.; Reig, M.; on behalf of the RETOINMUNO Multidisciplinary Group. Multidisciplinary Clinical Approach to Cancer Patients with Immune-Related Adverse Events Induced by Checkpoint Inhibitors. Cancers 2020, 12, 3446. https://doi.org/10.3390/cancers12113446
Londoño M-C, Reig M, on behalf of the RETOINMUNO Multidisciplinary Group. Multidisciplinary Clinical Approach to Cancer Patients with Immune-Related Adverse Events Induced by Checkpoint Inhibitors. Cancers. 2020; 12(11):3446. https://doi.org/10.3390/cancers12113446
Chicago/Turabian StyleLondoño, Maria-Carlota, Maria Reig, and on behalf of the RETOINMUNO Multidisciplinary Group. 2020. "Multidisciplinary Clinical Approach to Cancer Patients with Immune-Related Adverse Events Induced by Checkpoint Inhibitors" Cancers 12, no. 11: 3446. https://doi.org/10.3390/cancers12113446
APA StyleLondoño, M.-C., Reig, M., & on behalf of the RETOINMUNO Multidisciplinary Group. (2020). Multidisciplinary Clinical Approach to Cancer Patients with Immune-Related Adverse Events Induced by Checkpoint Inhibitors. Cancers, 12(11), 3446. https://doi.org/10.3390/cancers12113446





