Using Hepatocellular Carcinoma Tumor Burden Score to Stratify Prognosis after Liver Transplantation
Abstract
Simple Summary
Abstract
1. Introduction
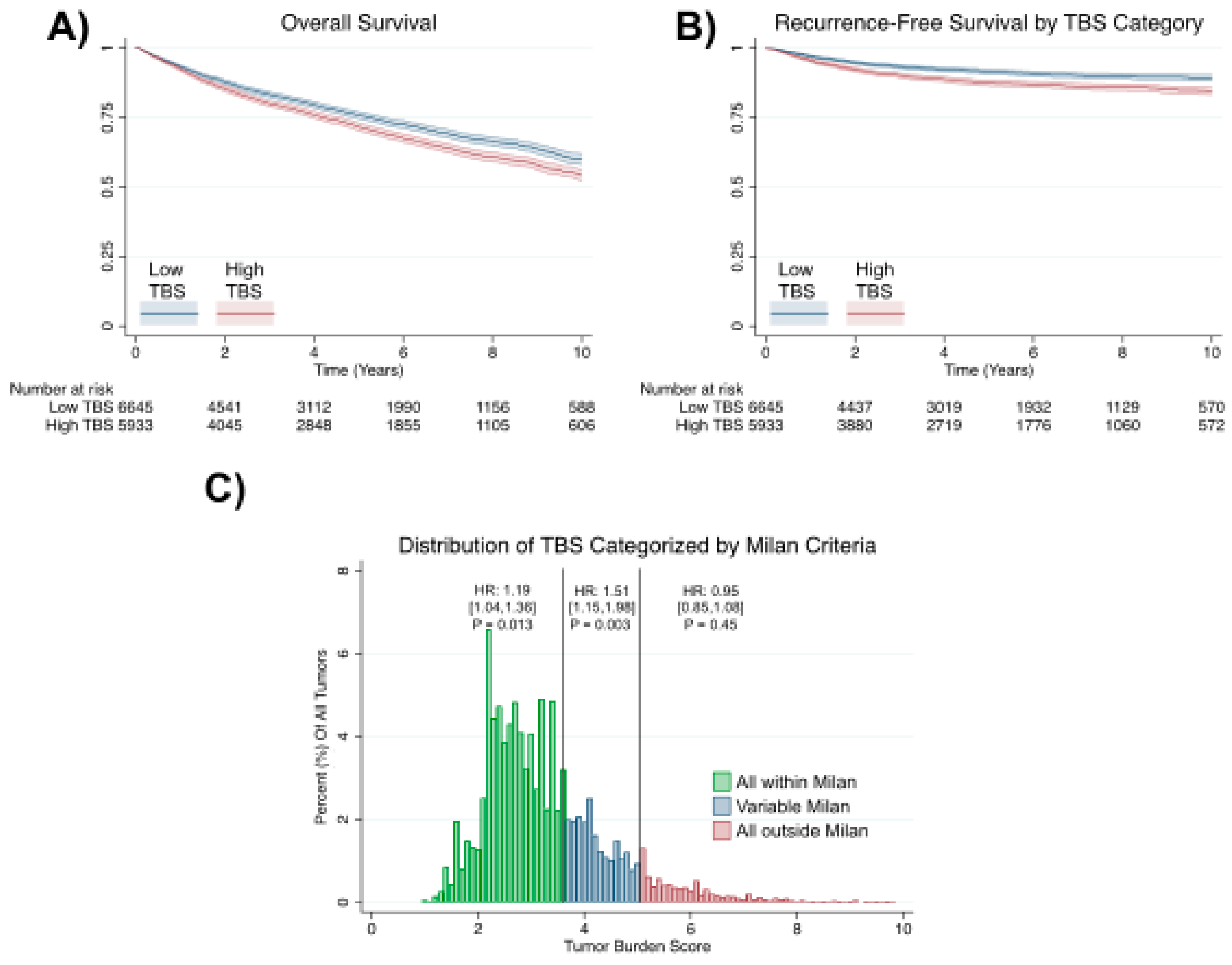
2. Results
3. Discussion
4. Materials and Methods
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Beal, E.W.; Tumin, D.; Kabir, A.; Moris, D.; Zhang, X.-F.; Chakedis, J.; Washburn, K.; Black, S.; Schmidt, C.M.; Pawlik, T.M. Trends in the Mortality of Hepatocellular Carcinoma in the United States. J. Gastrointest. Surg. 2017, 21, 2033–2038. [Google Scholar] [CrossRef] [PubMed]
- Petrick, J.L.; Kelly, S.P.; Altekruse, S.F.; McGlynn, K.A.; Rosenberg, P.S. Future of Hepatocellular Carcinoma Incidence in the United States Forecast Through 2030. J. Clin. Oncol. 2016, 34, 1787–1794. [Google Scholar] [CrossRef] [PubMed]
- Estes, C.; Anstee, Q.M.; Arias-Loste, M.T.; Bantel, H.; Bellentani, S.; Caballeria, J.; Colombo, M.; Craxì, A.; Crespo, J.; Day, C.P.; et al. Modeling NAFLD disease burden in China, France, Germany, Italy, Japan, Spain, United Kingdom, and United States for the period 2016–2030. J. Hepatol. 2018, 69, 896–904. [Google Scholar] [CrossRef] [PubMed]
- Argyrou, C.; Moris, D.; Vernadakis, S. Hepatocellular carcinoma development in non-alcoholic fatty liver disease and non-alcoholic steatohepatitis. Is it going to be the "Plague" of the 21st century? A literature review focusing on pathogenesis, prevention and treatment. J. BUON 2017, 22, 6–20. [Google Scholar]
- Beal, E.W.; Tumin, D.; Kabir, A.; Moris, D.; Zhang, X.-F.; Chakedis, J.; Washburn, K.; Black, S.; Schmidt, C.M.; Pawlik, T.M. Cohort Contributions to Race- and Gender-Specific Trends in the Incidence of Hepatocellular Carcinoma in the USA. World J. Surg. 2017, 42, 835–840. [Google Scholar] [CrossRef]
- Forner, A.; Llovet, J.M.; Bruix, J. Hepatocellular carcinoma. Lancet 2012, 379, 1245–1255. [Google Scholar] [CrossRef]
- Dimitroulis, D.; Damaskos, C.; Valsami, S.; Davakis, S.; Garmpis, N.; Spartalis, E.; Athanasiou, A.; Moris, D.; Sakellariou, S.; Kykalos, S.; et al. From diagnosis to treatment of hepatocellular carcinoma: An epidemic problem for both developed and developing world. World J. Gastroenterol. 2017, 23, 5282–5294. [Google Scholar] [CrossRef]
- Tsilimigras, D.I.; Bagante, F.; Moris, D.; Merath, K.; Paredes, A.Z.; Sahara, K.; Ratti, F.; Marques, H.P.; Soubrane, O.; Lam, V.; et al. Defining the chance of cure after resection for hepatocellular carcinoma within and beyond the Barcelona Clinic Liver Cancer guidelines: A multi-institutional analysis of 1,010 patients. Surgery 2019, 166, 967–974. [Google Scholar] [CrossRef]
- Moris, D.; Felekouras, E. Ignore reality but not the consequences of its ignorance: Broaden guidelines in surgery of hepatocellular carcinoma. Hepatology 2017, 65, 1772–1773. [Google Scholar] [CrossRef]
- Tsilimigras, D.I.; Mehta, R.; Moris, D.; Sahara, K.; Bagante, F.; Paredes, A.Z.; Farooq, A.; Ratti, F.; Marques, H.P.; Silva, S.; et al. Utilizing Machine Learning for Pre- and Postoperative Assessment of Patients Undergoing Resection for BCLC-0, A and B Hepatocellular Carcinoma: Implications for Resection Beyond the BCLC Guidelines. Ann. Surg. Oncol. 2019, 27, 866–874. [Google Scholar] [CrossRef]
- Tsilimigras, D.I.; Bagante, F.; Moris, D.; Ms, J.M.H.; Sahara, K.; Paredes, A.Z.; Mehta, R.; Ratti, F.; Marques, H.P.; Soubrane, O.; et al. Recurrence Patterns and Outcomes after Resection of Hepatocellular Carcinoma within and beyond the Barcelona Clinic Liver Cancer Criteria. Ann. Surg. Oncol. 2020, 27, 2321–2331. [Google Scholar] [CrossRef] [PubMed]
- Moris, D.; Vernadakis, S.; Papalampros, A.; Petrou, A.; Dimitroulis, D.; Spartalis, E.; Felekouras, E.; Fung, J.J. The effect of Guidelines in surgical decision making: The paradigm of hepatocellular carcinoma. J. BUON 2016, 21, 1332–1336. [Google Scholar] [PubMed]
- Gholami, S.; Perry, L.M.; Denbo, J.W.; Chavin, K.; Newell, P.; Ly, Q.; Hill, C.S.; Morris-Stiff, G.; Kessler, J.; Frankel, T.L.; et al. Management of early hepatocellular carcinoma: Results of the Delphi consensus process of the Americas Hepato-Pancreato-Biliary Association. HPB 2020. [Google Scholar] [CrossRef] [PubMed]
- Moris, D.; Tsilimigras, D.I.; Kostakis, I.D.; Ntanasis-Stathopoulos, I.; Shah, K.N.; Felekouras, E.; Pawlik, T.M. Anatomic versus non-anatomic resection for hepatocellular carcinoma: A systematic review and meta-analysis. Eur. J. Surg. Oncol. 2018, 44, 927–938. [Google Scholar] [CrossRef] [PubMed]
- Mazzaferro, V.; Regalia, E.; Doci, R.; Andreola, S.; Pulvirenti, A.; Bozzetti, F.; Montalto, F.; Ammatuna, M.; Morabito, A.; Gennari, L. Liver Transplantation for the Treatment of Small Hepatocellular Carcinomas in Patients with Cirrhosis. N. Engl. J. Med. 1996, 334, 693–700. [Google Scholar] [CrossRef] [PubMed]
- Sasaki, K.; Morioka, D.; Conci, S.; Margonis, G.A.; Sawada, Y.; Ruzzenente, A.; Kumamoto, T.; Iacono, C.; Andreatos, N.; Guglielmi, A.; et al. The Tumor Burden Score. Ann. Surg. 2018, 267, 132–141. [Google Scholar] [CrossRef]
- Tsilimigras, D.I.; Moris, D.; Hyer, J.M.; Baganate, F.; Sahara, K.; Moro, A.; Paredes, A.Z.; Mehta, R.; Ratti, F.; Marques, H.P.; et al. Hepatocellular carcinoma tumour burden score to stratify prognosis after resection. Br. J. Surg. 2020, 107, 854–864. [Google Scholar] [CrossRef]
- Tsilimigras, D.I.; Mehta, R.; Paredes, A.Z.; Moris, D.; Sahara, K.; Bagante, F.; Ratti, F.; Marques, H.P.; Silva, S.; Soubrane, O.; et al. Overall Tumor Burden Dictates Outcomes for Patients Undergoing Resection of Multinodular Hepatocellular Carcinoma Beyond the Milan Criteria. Ann. Surg. 2020, 272, 574–581. [Google Scholar] [CrossRef]
- Tsilimigras, D.I.; Mehta, R.; Guglielmi, A.; Ratti, F.; Marques, H.P.; Soubrane, O.; Lam, V.; Poultsides, G.A.; Popescu, I.; Alexandrescu, S.; et al. Recurrence beyond the Milan criteria after curative-intent resection of hepatocellular carcinoma: A novel tumor-burden based prediction model. J. Surg. Oncol. 2020. [Google Scholar] [CrossRef]
- Sasaki, K.; Firl, D.J.; Hashimoto, K.; Fujiki, M.; Diago-Uso, T.; Quintini, C.; Eghtesad, B.; Fung, J.; Aucejo, F.N.; Miller, C.M. Development and validation of the HALT-HCC score to predict mortality in liver transplant recipients with hepatocellular carcinoma: A retrospective cohort analysis. Lancet Gastroenterol. Hepatol. 2017, 2, 595–603. [Google Scholar] [CrossRef]
- Moris, D.; Kostakis, I.D.; Machairas, N.; Prodromidou, A.; Tsilimigras, D.I.; Ravindra, K.V.; Sudan, D.L.; Knechtle, S.J.; Barbas, A.S. Comparison between liver transplantation and resection for hilar cholangiocarcinoma: A systematic review and meta-analysis. PLoS ONE 2019, 14, e0220527. [Google Scholar] [CrossRef] [PubMed]
- Machairas, N.; Kostakis, I.D.; Tsilimigras, D.I.; Prodromidou, A.; Moris, D. Liver transplantation for hilar cholangiocarcinoma: A systematic review. Transplant. Rev. 2020, 34, 100516. [Google Scholar] [CrossRef] [PubMed]
- Moris, D.; Tsilimigras, D.I.; Ntanasis-Stathopoulos, I.; Beal, E.W.; Felekouras, E.; Vernadakis, S.; Fung, J.J.; Pawlik, T.M. Liver transplantation in patients with liver metastases from neuroendocrine tumors: A systematic review. Surgery 2017, 162, 525–536. [Google Scholar] [CrossRef] [PubMed]
- Moris, D.; Tsilimigras, D.I.; Chakedis, J.; Beal, E.; Felekouras, E.; Vernadakis, S.; Schizas, D.; Fung, J.J.; Pawlik, T.M. Liver transplantation for unresectable colorectal liver metastases: A systematic review. J. Surg. Oncol. 2017, 116, 288–297. [Google Scholar] [CrossRef] [PubMed]
- Dueland, S.; Syversveen, T.; Solheim, J.M.; Solberg, S.; Grut, H.; Bjørnbeth, B.A.; Hagness, M.; Line, P.-D. Survival Following Liver Transplantation for Patients With Nonresectable Liver-only Colorectal Metastases. Ann. Surg. 2020, 271, 212–218. [Google Scholar] [CrossRef]
- Mazzaferro, V.; Gorgen, A.; Roayaie, S.; Busset, M.D.D.; Sapisochin, G. Liver resection and transplantation for intrahepatic cholangiocarcinoma. J. Hepatol. 2020, 72, 364–377. [Google Scholar] [CrossRef]
- Makary, M.S.; Khandpur, U.; Cloyd, J.M.; Mumtaz, K.; Dowell, J.D. Locoregional Therapy Approaches for Hepatocellular Carcinoma: Recent Advances and Management Strategies. Cancers 2020, 12, 1914. [Google Scholar] [CrossRef]
- Gabr, A.; Kulik, L.; Mouli, S.; Riaz, A.; Ali, R.; Desai, K.; Mora, R.A.; Ganger, D.; Maddur, H.; Flamm, S.; et al. Liver Transplantation Following Yttrium-90 Radioembolization: 15-Year Experience in 207-Patient Cohort. Hepatology 2020. [Google Scholar] [CrossRef]
- Hasan, S.; Abel, S.; Uemura, T.; Verma, V.; Koay, E.J.; Herman, J.; Thai, N.; Kirichenko, A. Liver transplant mortality and morbidity following preoperative radiotherapy for hepatocellular carcinoma. HPB 2020, 22, 770–778. [Google Scholar] [CrossRef]
- Moris, D.; Rahnemai-Azar, A.A.; Zhang, X.-F.; Ntanasis-Stathopoulos, I.; Tsilimigras, D.I.; Chakedis, J.; Argyrou, C.; Fung, J.J.; Pawlik, T.M. Program death-1 immune checkpoint and tumor microenvironment in malignant liver tumors. Surg. Oncol. 2017, 26, 423–430. [Google Scholar] [CrossRef]
- Schwacha-Eipper, B.; Minciuna, I.; Banz, V.; Dufour, J.-F.F. Immunotherapy as a downstaging therapy for liver transplantation. Hepatology 2020. [Google Scholar] [CrossRef] [PubMed]
- Kole, C.; Charalampakis, N.; Tsakatikas, S.; Vailas, M.G.; Moris, D.; Gkotsis, E.; Kykalos, S.; Karamouzis, M.V.; Schizas, D. Immunotherapy for Hepatocellular Carcinoma: A 2021 Update. Cancers 2020, 12, 2859. [Google Scholar] [CrossRef] [PubMed]
- Mehta, N.; Dodge, J.L.; Grab, J.D.; Yao, F.Y. National Experience on Down-Staging of Hepatocellular Carcinoma Before Liver Transplant: Influence of Tumor Burden, Alpha-Fetoprotein, and Wait Time. Hepatology 2020, 71, 943–954. [Google Scholar] [CrossRef] [PubMed]
- Mazzaferro, V.; Citterio, D.; Bhoori, S.; Bongini, M.; Miceli, R.; De Carlis, L.; Colledan, M.; Salizzoni, M.; Romagnoli, R.; Antonelli, B.; et al. Liver transplantation in hepatocellular carcinoma after tumour downstaging (XXL): A randomised, controlled, phase 2b/3 trial. Lancet Oncol. 2020, 21, 947–956. [Google Scholar] [CrossRef]
- Firl, D.J.; Sasaki, K.; Agopian, V.G.; Gorgen, A.; Kimura, S.; Dumronggittigule, W.; McVey, J.C.; Iesari, S.; Mennini, G.; Vitale, A.; et al. Charting the Path Forward for Risk Prediction in Liver Transplant for Hepatocellular Carcinoma: International Validation of HALTHCC Among 4,089 Patients. Hepatology 2019, 71, 569–582. [Google Scholar] [CrossRef] [PubMed]
- Mehta, N.; Heimbach, J.; Harnois, D.M.; Sapisochin, G.; Dodge, J.L.; Lee, D.; Burns, J.M.; Sanchez, W.; Greig, P.D.; Grant, D.R.; et al. Validation of a Risk Estimation of Tumor Recurrence After Transplant (RETREAT) Score for Hepatocellular Carcinoma Recurrence After Liver Transplant. JAMA Oncol. 2017, 3, 493–500. [Google Scholar] [CrossRef]
- Agopian, V.G.; Harlander-Locke, M.; Zarrinpar, A.; Kaldas, F.M.; Farmer, D.G.; Yersiz, H.; Finn, R.S.; Tong, M.; Hiatt, J.R.; Busuttil, R.W. A Novel Prognostic Nomogram Accurately Predicts Hepatocellular Carcinoma Recurrence after Liver Transplantation: Analysis of 865 Consecutive Liver Transplant Recipients. J. Am. Coll. Surg. 2015, 220, 416–427. [Google Scholar] [CrossRef]
- Mazzaferro, V.; Sposito, C.; Zhou, J.; Pinna, A.D.; De Carlis, L.; Fan, J.; Cescon, M.; Di Sandro, S.; Yi-Feng, H.; Lauterio, A.; et al. Metroticket 2.0 Model for Analysis of Competing Risks of Death After Liver Transplantation for Hepatocellular Carcinoma. Gastroenterology 2018, 154, 128–139. [Google Scholar] [CrossRef]
- Moris, D.; Shaw, B.; Ong, C.; Connor, A.; Samoylova, M.; Kesseli, S.; Abraham, N.; Gloria, J.; Schmitz, R.; Fitch, Z.; et al. A simple scoring system to estimate perioperative mortality following liver resection for primary liver malignancies—The hepatectomy risk score (HeRS). Hepatobiliary Surg. Nutr. 2020. [Google Scholar] [CrossRef]
- Cucchetti, A.; Serenari, M.; Sposito, C.; Di Sandro, S.; Mosconi, C.; Vicentin, I.; Garanzini, E.; Mazzaferro, V.; De Carlis, L.; Golfieri, R.; et al. Including mRECIST in the Metroticket 2.0 criteria improves prediction of hepatocellular carcinoma-related death after liver transplant. J. Hepatol. 2020, 73, 342–348. [Google Scholar] [CrossRef]
- Kardashian, A.; Florman, S.S.; Haydel, B.; Ruiz, R.M.; Klintmalm, G.B.; Lee, D.D.; Taner, C.B.; Aucejo, F.; Tevar, A.D.; Humar, A.; et al. Liver Transplantation Outcomes in a U.S. Multicenter Cohort of 789 Patients with Hepatocellular Carcinoma Presenting Beyond Milan Criteria. Hepatology 2020. [Google Scholar] [CrossRef] [PubMed]
- Moris, D.; Lu, L.; Qian, S. Mechanisms of liver-induced tolerance. Curr. Opin. Organ Transplant. 2017, 22, 71–78. [Google Scholar] [CrossRef] [PubMed]
- Refolo, M.G.; Messa, C.; Guerra, V.; Carr, B.I.; D’Alessandro, R. Inflammatory Mechanisms of HCC Development. Cancers 2020, 12, 641. [Google Scholar] [CrossRef] [PubMed]
- Der Stroth, L.I.; Tharehalli, U.; Günes, C.; Lechel, A. Telomeres and Telomerase in the Development of Liver Cancer. Cancers 2020, 12, 2048. [Google Scholar] [CrossRef] [PubMed]
- Moris, D.; Beal, E.W.; Chakedis, J.; Burkhart, R.A.; Schmidt, C.; Dillhoff, M.; Zhang, X.; Theocharis, S.; Pawlik, T.M. Role of exosomes in treatment of hepatocellular carcinoma. Surg. Oncol. 2017, 26, 219–228. [Google Scholar] [CrossRef]
- Wang, G.; Wang, Q.; Liang, N.; Xue, H.; Yang, T.; Chen, X.; Qiu, Z.; Zeng, C.; Sun, T.; Yuan, W.; et al. Oncogenic driver genes and tumor microenvironment determine the type of liver cancer. Cell Death Dis. 2020, 11, 1–13. [Google Scholar] [CrossRef]
- Chang, Y.; Cho, Y.; Lee, J.-H.; Bin Lee, Y.; Cho, E.J.; Yu, S.J.; Sinn, D.H.; Kim, B.H.; Kim, S.H.; Yi, N.-J.; et al. Comparison of Models for Tumor Recurrence after Liver Transplantation for the Patients with Hepatocellular Carcinoma: A Multicenter Long-Term Follow-Up Study. Cancers 2019, 11, 1295. [Google Scholar] [CrossRef]
- Firl, D.J.; Kimura, S.; McVey, J.; Hashimoto, K.; Yeh, H.; Miller, C.M.; Markmann, J.F.; Sasaki, K.; Aucejo, F.N. Reframing the approach to patients with hepatocellular carcinoma: Longitudinal assessment with hazard associated with liver transplantation for HCC (HALTHCC) improves ablate and wait strategy. Hepatology 2018, 68, 1448–1458. [Google Scholar] [CrossRef]
- Lai, Q.; Vitale, A.; Halazun, K.; Iesari, S.; Viveiros, A.; Bhangui, P.; Mennini, G.; Wong, T.; Uemoto, S.; Lin, C.-C.; et al. Identification of an Upper Limit of Tumor Burden for Downstaging in Candidates with Hepatocellular Cancer Waiting for Liver Transplantation: A West–East Collaborative Effort. Cancers 2020, 12, 452. [Google Scholar] [CrossRef]
- Dutkowski, P.; Linecker, M.; DeOliveira, M.L.; Müllhaupt, B.; Clavien, P.-A. Challenges to Liver Transplantation and Strategies to Improve Outcomes. Gastroenterology 2015, 148, 307–323. [Google Scholar] [CrossRef]
- Gao, Q.; Mulvihill, M.S.; Scheuermann, U.; Davis, R.P.; Yerxa, J.; Yerokun, B.A.; Hartwig, M.G.; Sudan, D.L.; Knechtle, S.J.; Barbas, A.S. Improvement in Liver Transplant Outcomes From Older Donors. Ann. Surg. 2019, 270, 333–339. [Google Scholar] [CrossRef] [PubMed]
- Taylor, R.; Allen, E.; Richards, J.A.; Goh, M.A.; Neuberger, J.; Collett, D.; Pettigrew, G.J.; Goh, A.M.; Liver Advisory Group to NHS Blood and Transplant. Survival advantage for patients accepting the offer of a circulatory death liver transplant. J. Hepatol. 2019, 70, 855–865. [Google Scholar] [CrossRef] [PubMed]
- Moris, D.; Shaw, B.I.; Gloria, J.; Kesseli, S.J.; Samoylova, M.L.; Schmitz, R.; Manook, M.; McElroy, L.M.; Patel, Y.; Berg, C.L.; et al. Textbook Outcomes in Liver Transplantation. World J. Surg. 2020, 44, 3470–3477. [Google Scholar] [CrossRef] [PubMed]
- Muller, X.; Marcon, F.; Sapisochin, G.; Marquez, M.; Dondero, F.; Rayar, M.; Doyle, M.M.B.; Callans, L.; Li, J.; Nowak, G.; et al. Defining Benchmarks in Liver Transplantation. Ann. Surg. 2018, 267, 419–425. [Google Scholar] [CrossRef] [PubMed]

| Low TBS (<3.1) | High TBS (≥3.1) | p-Value | |
|---|---|---|---|
| n = 6645 | n = 5933 | ||
| Female gender, n (%) | 1594 (24%) | 1155 (19%) | <0.001 |
| Age, median (IQR) | 60.0 (55.0–64.0) | 60.0 (55.0–65.0) | 0.36 |
| Race, n (%) | 0.018 | ||
| Asian | 542 (8%) | 536 (9%) | |
| Black | 641 (10%) | 631 (11%) | |
| Multi | 31 (0%) | 35 (1%) | |
| Native | 36 (1%) | 50 (1%) | |
| Pacific | 23 (0%) | 15 (0%) | |
| White | 5372 (81%) | 4666 (79%) | |
| Insurance status, n (%) | 0.045 | ||
| Public | 3186 (48%) | 2714 (46%) | |
| Private | 3415 (51%) | 3175 (54%) | |
| Self | 44 (1%) | 44 (1%) | |
| Outside of Milan criteria, n (%) | 0 (0%) | 1548 (26%) | <0.001 |
| TBS, median (IQR) | 2.4 (2.2–2.8) | 3.9 (3.4–4.7) | <0.001 |
| Positive HBV status, n (%) | 2189 (35%) | 1998 (36%) | 0.19 |
| Positive HCV status, n (%) | 4162 (65%) | 3568 (64%) | 0.037 |
| Cold Ischemia Time, median h (IQR) | 6.0 (4.6–7.7) | 6.0 (4.8–8.0) | 0.017 |
| Recurrence of primary malignancy, n (%) | 460 (7%) | 608 (10%) | <0.001 |
| Death, n (%) | 1641 (25%) | 1798 (30%) | <0.001 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Moris, D.; Shaw, B.I.; McElroy, L.; Barbas, A.S. Using Hepatocellular Carcinoma Tumor Burden Score to Stratify Prognosis after Liver Transplantation. Cancers 2020, 12, 3372. https://doi.org/10.3390/cancers12113372
Moris D, Shaw BI, McElroy L, Barbas AS. Using Hepatocellular Carcinoma Tumor Burden Score to Stratify Prognosis after Liver Transplantation. Cancers. 2020; 12(11):3372. https://doi.org/10.3390/cancers12113372
Chicago/Turabian StyleMoris, Dimitrios, Brian I. Shaw, Lisa McElroy, and Andrew S. Barbas. 2020. "Using Hepatocellular Carcinoma Tumor Burden Score to Stratify Prognosis after Liver Transplantation" Cancers 12, no. 11: 3372. https://doi.org/10.3390/cancers12113372
APA StyleMoris, D., Shaw, B. I., McElroy, L., & Barbas, A. S. (2020). Using Hepatocellular Carcinoma Tumor Burden Score to Stratify Prognosis after Liver Transplantation. Cancers, 12(11), 3372. https://doi.org/10.3390/cancers12113372






