Completion Total Thyroidectomy Is Not Necessary for Papillary Thyroid Microcarcinoma with Occult Central Lymph Node Metastasis: A Long-Term Serial Follow-Up
Simple Summary
Abstract
1. Introduction
2. Results
2.1. Characteristics of the Two Groups
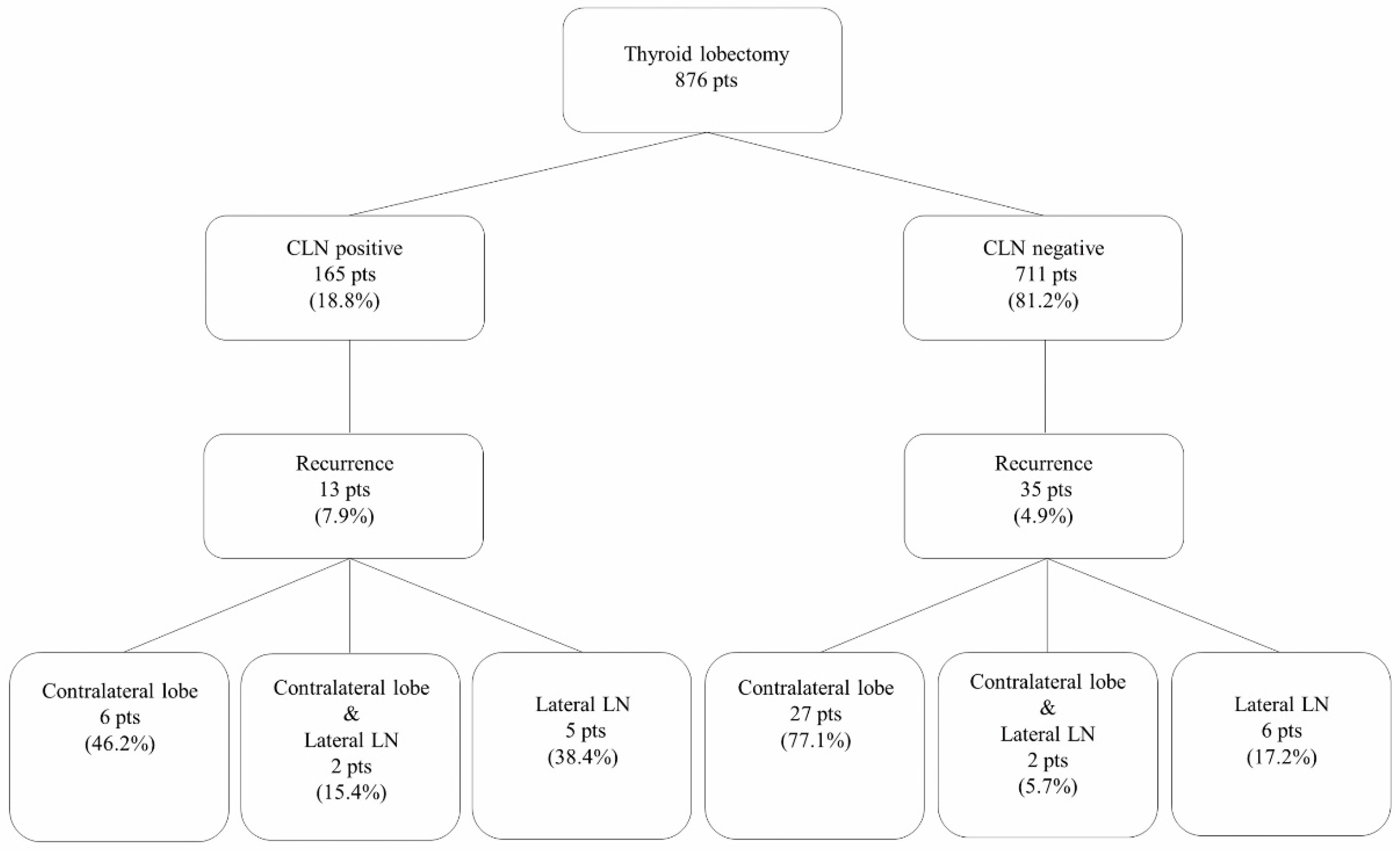
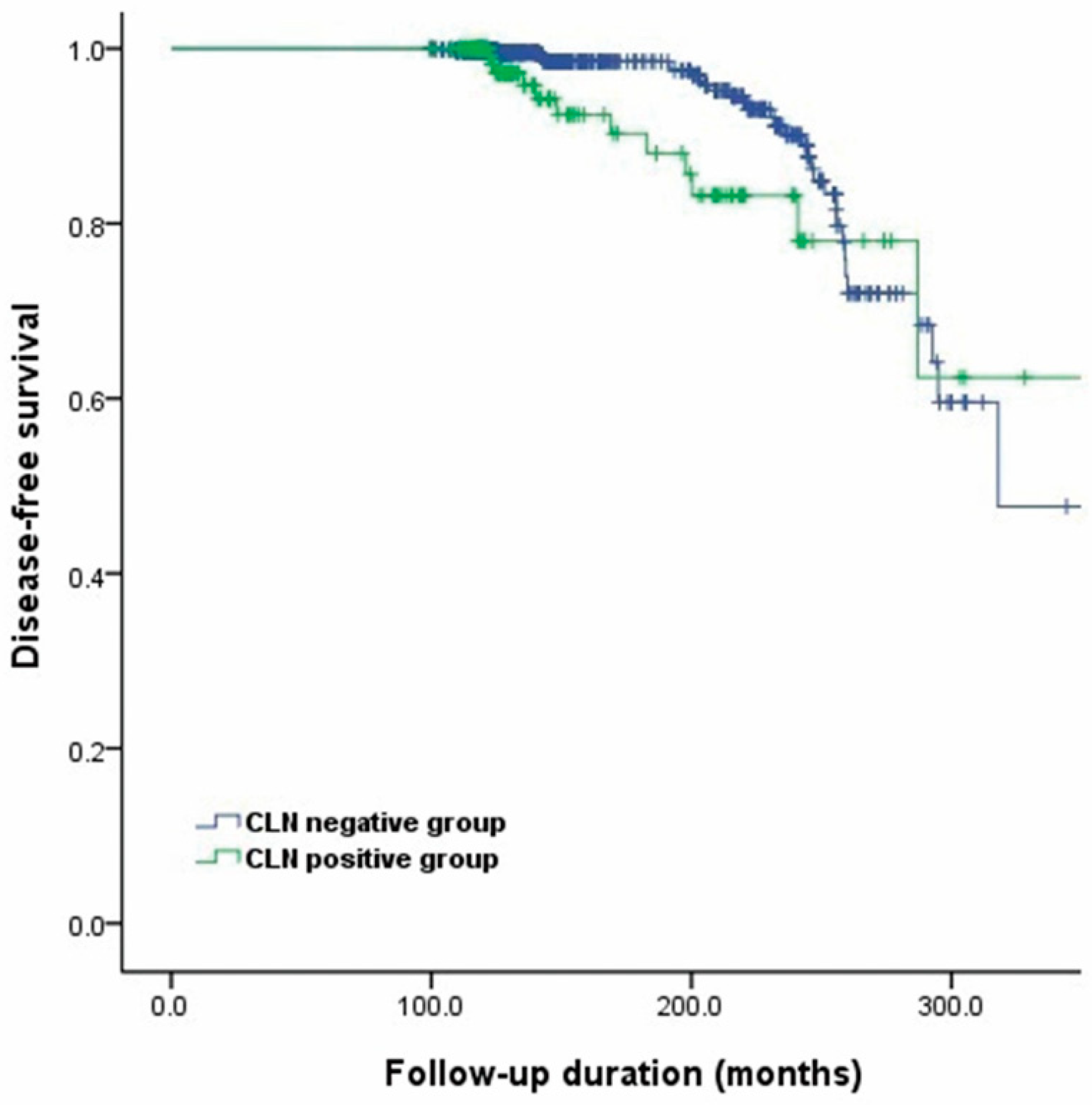
2.2. Recurrence in the Two Groups
2.3. Postoperative Complications in the Two Groups
2.4. Comparison Characteristics of the Enrolled Patients in Two Periods
3. Discussion
4. Materials and Methods
4.1. Ethics Statement
4.2. Patients
4.3. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Lundgren, C.I.; Hall, P.; Dickman, P.W.; Zedenius, J. Clinically significant prognostic factors for differentiated thyroid carcinoma: A population-based, nested case–control study. Cancer Cytopathol. 2006, 106, 524–531. [Google Scholar] [CrossRef] [PubMed]
- Chen, A.Y.; Jemal, A.; Ward, E.M. Increasing incidence of differentiated thyroid cancer in the united states, 1988–2005. Cancer 2009, 115, 3801–3807. [Google Scholar] [CrossRef] [PubMed]
- Thompson, L.D. World health organization classification of tumours: Pathology and genetics of head and neck tumours. Ear Nose Throat J. 2006, 85, 74–75. [Google Scholar] [CrossRef]
- Giordano, D.; Gradoni, P.; Oretti, G.; Molina, E.; Ferri, T. Treatment and prognostic factors of papillary thyroid microcarcinoma. Clin. Otolaryngol. 2010, 35, 118–124. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.; Chu, B.; Zhu, J.; Zhang, S.; Liu, Y.; Zhuang, M.; Yang, Y. Clinical analysis of prophylactic central neck dissection for papillary thyroid carcinoma. Clin. Transl. Oncol. 2014, 16, 44–48. [Google Scholar] [CrossRef] [PubMed]
- McHenry, C.R. Prophylactic central compartment neck dissection for papillary thyroid cancer: The search for justification continues. Surgery 2011, 150, 1058–1060. [Google Scholar] [CrossRef] [PubMed]
- Hay, I.D.; Hutchinson, M.E.; Gonzalez-Losada, T.; McIver, B.; Reinalda, M.E.; Grant, C.S.; Thompson, G.B.; Sebo, T.J.; Goellner, J.R. Papillary thyroid microcarcinoma: A study of 900 cases observed in a 60-year period. Surgery 2008, 144, 980–988. [Google Scholar] [CrossRef]
- Baudin, E.; Travagli, J.P.; Ropers, J.; Mancusi, F.; Bruno-Bossio, G.; Caillou, B.; Cailleux, A.F.; Lumbroso, J.D.; Parmentier, C.; Schlumberger, M. Microcarcinoma of the thyroid gland: The gustave-roussy institute experience. Cancer Cytopathol. 1998, 83, 553–559. [Google Scholar] [CrossRef]
- Chow, S.M.; Law, S.C.; Chan, J.K.; Au, S.K.; Yau, S.; Lau, W.H. Papillary microcarcinoma of the thyroid—prognostic significance of lymph node metastasis and multifocality. Cancer 2003, 98, 31–40. [Google Scholar] [CrossRef]
- Choi, J.B.; Lee, W.K.; Lee, S.G.; Ryu, H.; Lee, C.R.; Kang, S.W.; Jeong, J.J.; Nam, K.-H.; Lee, E.J.; Chung, W.Y. Long-term oncologic outcomes of papillary thyroid microcarcinoma according to the presence of clinically apparent lymph node metastasis: A large retrospective analysis of 5348 patients. Cancer Manag. Res. 2018, 10, 2883. [Google Scholar] [CrossRef]
- Ito, Y.; Tomoda, C.; Uruno, T.; Takamura, Y.; Miya, A.; Kobayashi, K.; Matsuzuka, F.; Kuma, K.; Miyauchi, A. Clinical significance of metastasis to the central compartment from papillary microcarcinoma of the thyroid. World J. Surg. 2006, 30, 91–99. [Google Scholar] [CrossRef]
- Wang, X.; Lei, J.; Wei, T.; Zhu, J.; Li, Z. Clinicopathological characteristics and recurrence risk of papillary thyroid microcarcinoma in the elderly. Cancer Manag. Res. 2019, 11, 2371. [Google Scholar] [CrossRef] [PubMed]
- Amin, S.N.; Shinn, J.R.; Naguib, M.M.; Netterville, J.L.; Rohde, S.L. Risk factors and outcomes of postoperative recurrent well-differentiated thyroid cancer: A single institution’s 15-year experience. Otolaryngol. Head Neck Surg. 2020, 162, 469–475. [Google Scholar] [CrossRef] [PubMed]
- Ryu, Y.J.; Cho, J.S.; Park, M.H.; Yoon, J.H. Identifying risk factors of recurrence for clinically node negative papillary thyroid carcinoma with pathologic n1a. BMC Surg. 2019, 19, 78. [Google Scholar] [CrossRef]
- Haugen, B.R.; Alexander, E.K.; Bible, K.C.; Doherty, G.M.; Mandel, S.J.; Nikiforov, Y.E.; Pacini, F.; Randolph, G.W.; Sawka, A.M.; Schlumberger, M. 2015 american thyroid association management guidelines for adult patients with thyroid nodules and differentiated thyroid cancer: The american thyroid association guidelines task force on thyroid nodules and differentiated thyroid cancer. Thyroid 2016, 26, 1–133. [Google Scholar] [CrossRef] [PubMed]
- So, Y.K.; Son, Y.-I.; Hong, S.D.; Seo, M.Y.; Baek, C.-H.; Jeong, H.-S.; Chung, M.K. Subclinical lymph node metastasis in papillary thyroid microcarcinoma: A study of 551 resections. Surgery 2010, 148, 526–531. [Google Scholar] [CrossRef]
- Shen, W.T.; Ogawa, L.; Ruan, D.; Suh, I.; Kebebew, E.; Duh, Q.-Y.; Clark, O.H. Central neck lymph node dissection for papillary thyroid cancer: Comparison of complication and recurrence rates in 295 initial dissections and reoperations. Arch. Surg. 2010, 145, 272–275. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Liu, Z.; Liu, Y.; Gao, W.; Zheng, C. The clinical prognosis of patients with cn0 papillary thyroid microcarcinoma by central neck dissection. World J. Surg. Oncol. 2015, 13, 1–5. [Google Scholar] [CrossRef]
- Hyun, S.M.; Song, H.Y.; Kim, S.Y.; Nam, S.Y.; Roh, J.-L.; Han, M.W.; Choi, S.-H. Impact of combined prophylactic unilateral central neck dissection and hemithyroidectomy in patients with papillary thyroid microcarcinoma. Ann. Surg. Oncol. 2012, 19, 591–596. [Google Scholar] [CrossRef]
- Caliskan, M.; Park, J.H.; Jeong, J.S.; Lee, C.-R.; Park, S.K.; Kang, S.-W.; Jeong, J.J.; Chung, W.Y.; Park, C.S. Role of prophylactic ipsilateral central compartment lymph node dissection in papillary thyroid microcarcinoma. Endocr. J. 2012, 59, 305–311. [Google Scholar] [CrossRef]
- Lee, C.R.; Son, H.; Lee, S.; Kang, S.-W.; Jeong, J.J.; Nam, K.-H.; Chung, W.Y.; Park, C.S. Lobectomy and prophylactic central neck dissection for papillary thyroid microcarcinoma: Do involved lymph nodes mandate completion thyroidectomy? World J. Surg. 2014, 38, 872–877. [Google Scholar] [CrossRef] [PubMed]
- La Vecchia, C.; Malvezzi, M.; Bosetti, C.; Garavello, W.; Bertuccio, P.; Levi, F.; Negri, E. Thyroid cancer mortality and incidence: A global overview. Int. J. Cancer 2015, 136, 2187–2195. [Google Scholar] [CrossRef]
- Wiltshire, J.; Drake, T.M.; Uttley, L.; Balasubramanian, S.P. Systematic review of trends in the incidence rates of thyroid cancer. Thyroid 2016, 26, 1541–1552. [Google Scholar] [CrossRef]
- Ruiz, J.; Ríos, A.; Rodríguez, J.; Paredes, M.; Soriano, V.; Oviedo, M.; Hernández, A.; Parrilla, P. Incidental versus clinical diagnosis of papillary thyroid microcarcinoma. Long-term prognosis. Endocrinol. Diabetes Nutr. 2020, 67, 317–325. [Google Scholar] [CrossRef] [PubMed]
- Jeon, Y.W.; Gwak, H.G.; Lim, S.T.; Schneider, J.; Suh, Y.J. Long-term prognosis of unilateral and multifocal papillary thyroid microcarcinoma after unilateral lobectomy versus total thyroidectomy. Ann. Surg. Oncol. 2019, 26, 2952–2958. [Google Scholar] [CrossRef]
- Ito, Y.; Miyauchi, A. Active surveillance as first-line management of papillary microcarcinoma. Annu. Rev. Med. 2019, 70, 369–379. [Google Scholar] [CrossRef] [PubMed]
- Kong, S.H.; Ryu, J.; Kim, M.J.; Cho, S.W.; Song, Y.S.; Yi, K.H.; Park, D.J.; Hwangbo, Y.; Lee, Y.J.; Lee, K.E. Longitudinal assessment of quality of life according to treatment options in low-risk papillary thyroid microcarcinoma patients: Active surveillance or immediate surgery (interim analysis of maestro). Thyroid 2019, 29, 1089–1096. [Google Scholar] [CrossRef]
- Jeon, M.J.; Lee, Y.-M.; Sung, T.-Y.; Han, M.; Shin, Y.-W.; Kim, W.G.; Kim, T.Y.; Chung, K.-W.; Shong, Y.K.; Kim, W.B. Quality of life in patients with papillary thyroid microcarcinoma managed by active surveillance or lobectomy: A cross-sectional study. Thyroid 2019, 29, 956–962. [Google Scholar] [CrossRef]
- Oda, H.; Miyauchi, A.; Ito, Y.; Yoshioka, K.; Nakayama, A.; Sasai, H.; Masuoka, H.; Yabuta, T.; Fukushima, M.; Higashiyama, T.; et al. Incidences of unfavorable events in the management of low-risk papillary microcarcinoma of the thyroid by active surveillance versus immediate surgery. Thyroid 2016, 26, 150–155. [Google Scholar] [CrossRef]
- Kwon, H.; Oh, H.-S.; Kim, M.; Park, S.; Jeon, M.J.; Kim, W.G.; Kim, W.B.; Shong, Y.K.; Song, D.E.; Baek, J.H.; et al. Active surveillance for patients with papillary thyroid microcarcinoma: A single center’s experience in korea. J. Clin. Endocrinol. Metab. 2017, 102, 1917–1925. [Google Scholar] [CrossRef]
- Miyauchi, A. Clinical trials of active surveillance of papillary microcarcinoma of the thyroid. World J. Surg. 2016, 40, 516–522. [Google Scholar] [CrossRef]
- Tuttle, R.M.; Fagin, J.A.; Minkowitz, G.; Wong, R.J.; Roman, B.; Patel, S.; Untch, B.; Ganly, I.; Shaha, A.R.; Shah, J.P. Natural history and tumor volume kinetics of papillary thyroid cancers during active surveillance. JAMA Otolaryngol. Head Neck Surg. 2017, 143, 1015–1020. [Google Scholar] [CrossRef]
- Griffin, A.; Brito, J.P.; Bahl, M.; Hoang, J.K. Applying criteria of active surveillance to low-risk papillary thyroid cancer over a decade: How many surgeries and complications can be avoided? Thyroid 2017, 27, 518–523. [Google Scholar] [CrossRef]
- Mazzaferri, E. Management of low-risk differentiated thyroid cancer. Endocr. Pract. 2007, 13, 498–512. [Google Scholar] [CrossRef]
- Hay, I. Management of patients with low-risk papillary thyroid carcinoma. Endocr. Pract. 2007, 13, 521–533. [Google Scholar] [CrossRef]
- Gui, C.Y.; Qiu, S.L.; Peng, Z.H.; Wang, M. Clinical and pathologic predictors of central lymph node metastasis in papillary thyroid microcarcinoma: A retrospective cohort study. J. Endocrinol. Investig. 2018, 41, 403–409. [Google Scholar] [CrossRef] [PubMed]
- Sheng, L.; Shi, J.; Han, B.; Lv, B.; Li, L.; Chen, B.; Liu, N.; Cao, Y.; Turner, A.G.; Zeng, Q. Predicting factors for central or lateral lymph node metastasis in conventional papillary thyroid microcarcinoma. Am. J. Surg. 2019. [Google Scholar] [CrossRef]
- Choi, Y.S.; Kim, D.W.; Lee, Y.J.; Ha, T.K.; Jung, S.J.; Baek, H.J. Appropriate neck ultrasonography surveillance during the first 10 years after hemithyroidectomy in papillary thyroid microcarcinoma patients: A single-center study. Ultrasound Q. 2019, 35, 275–280. [Google Scholar] [CrossRef] [PubMed]
- Siddiqui, S.; White, M.G.; Antic, T.; Grogan, R.H.; Angelos, P.; Kaplan, E.L.; Cipriani, N.A. Clinical and pathologic predictors of lymph node metastasis and recurrence in papillary thyroid microcarcinoma. Thyroid 2016, 26, 807–815. [Google Scholar] [CrossRef]
- So, Y.K.; Seo, M.Y.; Son, Y.-I. Prophylactic central lymph node dissection for clinically node-negative papillary thyroid microcarcinoma: Influence on serum thyroglobulin level, recurrence rate, and postoperative complications. Surgery 2012, 151, 192–198. [Google Scholar] [CrossRef]
- Kwon, H.; Jeon, M.J.; Kim, W.G.; Park, S.; Kim, M.; Song, D.E.; Sung, T.-Y.; Yoon, J.H.; Hong, S.J.; Kim, T.Y. A comparison of lobectomy and total thyroidectomy in patients with papillary thyroid microcarcinoma: A retrospective individual risk factor-matched cohort study. Eur. J. Endocrinol. 2017, 176, 371–378. [Google Scholar] [CrossRef] [PubMed]
- Pelizzo, M.R.; Boschin, I.M.; Toniato, A.; Pagetta, C.; Piotto, A.; Bernante, P.; Casara, D.; Pennelli, G.; Rubello, D. Natural history, diagnosis, treatment and outcome of papillary thyroid microcarcinoma (ptmc): A mono-institutional 12-year experience. Nucl. Med. Commun. 2004, 25, 547–552. [Google Scholar] [CrossRef] [PubMed]
- Hay, I.D.; Grant, C.S.; van Heerden, J.A.; Goellner, J.R.; Ebersold, J.R.; Bergstralh, E.J. Papillary thyroid microcarcinoma: A study of 535 eases observed in a 50-year period. Surgery 1992, 112, 1139–1147. [Google Scholar] [PubMed]
- Gulcelik, M.A.; Dogan, L.; Akgul, G.G.; Güven, E.H.; Gulcelik, N.E. Completion thyroidectomy: Safer than thought. Oncol. Res. Treat. 2018, 41, 386–390. [Google Scholar] [CrossRef]
- Ito, Y.; Kihara, M.; Kobayashi, K.; Miya, A.; Miyauchi, A. Permanent hypoparathyroidism after completion total thyroidectomy as a second surgery: How do we avoid it? Endocr. J. 2014, 61, 403–408. [Google Scholar] [CrossRef]
- Rafferty, M.A.; Goldstein, D.P.; Rotstein, L.; Asa, S.L.; Panzarella, T.; Gullane, P.; Gilbert, R.W.; Brown, D.H.; Irish, J.C. Completion thyroidectomy versus total thyroidectomy: Is there a difference in complication rates? An analysis of 350 patients. J. Am. Coll. Surg. 2007, 205, 602–607. [Google Scholar] [CrossRef]
- Erdem, E.; Gülçelik, M.; Kuru, B.; Alagöl, H. Comparison of completion thyroidectomy and primary surgery for differentiated thyroid carcinoma. Eur. J. Surg. Oncol. 2003, 29, 747–749. [Google Scholar] [CrossRef]
- Zhao, H.; Huang, T.; Li, H. Risk factors for skip metastasis and lateral lymph node metastasis of papillary thyroid cancer. Surgery 2019, 166, 55–60. [Google Scholar] [CrossRef]
- Back, K.; Kim, J.S.; Kim, J.-H.; Choe, J.-H. Superior located papillary thyroid microcarcinoma is a risk factor for lateral lymph node metastasis. Ann. Surg. Oncol. 2019, 26, 3992–4001. [Google Scholar] [CrossRef]
- Feng, J.W.; Yang, X.H.; Wu, B.Q.; Sun, D.L.; Jiang, Y.; Qu, Z. Predictive factors for central lymph node and lateral cervical lymph node metastases in papillary thyroid carcinoma. Clin. Transl. Oncol. 2019, 21, 1482–1491. [Google Scholar] [CrossRef]
- Wu, X.; Li, B.; Zheng, C.; He, X. Predicting factors of lateral neck lymph node metastases in patients with papillary thyroid microcarcinoma. Medicine 2019, 98, e16386. [Google Scholar] [CrossRef]
- Ryu, Y.J.; Kang, S.J.; Cho, J.S.; Yoon, J.H.; Park, M.H. Identifying risk factors of lateral lymph node recurrence in clinically node-negative papillary thyroid cancer. Medicine 2018, 97, e13435. [Google Scholar] [CrossRef]
- Lee, Y.S.; Shin, S.C.; Lim, Y.S.; Lee, J.C.; Wang, S.G.; Son, S.M.; Kim, I.J.; Lee, B.J. Tumor location–dependent skip lateral cervical lymph node metastasis in papillary thyroid cancer. Head Neck 2014, 36, 887–891. [Google Scholar] [CrossRef] [PubMed]
- Park, J.H.; Lee, Y.S.; Kim, B.W.; Chang, H.-S.; Park, C.S. Skip lateral neck node metastases in papillary thyroid carcinoma. World J. Surg. 2012, 36, 743–747. [Google Scholar] [CrossRef] [PubMed]
- Nie, X.; Tan, Z.; Ge, M. Skip metastasis in papillary thyroid carcinoma is difficult to predict in clinical practice. BMC Cancer 2017, 17, 702. [Google Scholar] [CrossRef] [PubMed]
- Lei, J.; Zhong, J.; Jiang, K.; Li, Z.; Gong, R.; Zhu, J. Skip lateral lymph node metastasis leaping over the central neck compartment in papillary thyroid carcinoma. Oncotarget 2017, 8, 27022. [Google Scholar] [CrossRef] [PubMed]
- Lim, Y.C.; Koo, B.S. Predictive factors of skip metastases to lateral neck compartment leaping central neck compartment in papillary thyroid carcinoma. Oral Oncol. 2012, 48, 262–265. [Google Scholar] [CrossRef]
- Sosa, J.A.; Bowman, H.M.; Tielsch, J.M.; Powe, N.R.; Gordon, T.A.; Udelsman, R. The importance of surgeon experience for clinical and economic outcomes from thyroidectomy. Ann. Surg. 1998, 228, 320. [Google Scholar] [CrossRef]
- Loyo, M.; Tufano, R.P.; Gourin, C.G. National trends in thyroid surgery and the effect of volume on short-term outcomes. Laryngoscope 2013, 123, 2056–2063. [Google Scholar] [CrossRef]
- Kandil, E.; Noureldine, S.I.; Abbas, A.; Tufano, R.P. The impact of surgical volume on patient outcomes following thyroid surgery. Surgery 2013, 154, 1346–1353. [Google Scholar] [CrossRef]
- Duclos, A.; Peix, J.-L.; Colin, C.; Kraimps, J.-L.; Menegaux, F.; Pattou, F.; Sebag, F.; Touzet, S.; Bourdy, S.; Voirin, N. Influence of experience on performance of individual surgeons in thyroid surgery: Prospective cross sectional multicentre study. BMJ 2012, 344. [Google Scholar] [CrossRef]
- Hauch, A.; Al-Qurayshi, Z.; Randolph, G.; Kandil, E. Total thyroidectomy is associated with increased risk of complications for low-and high-volume surgeons. Ann. Surg. Oncol. 2014, 21, 3844–3852. [Google Scholar] [CrossRef] [PubMed]
- Feeney, T.; Price, L.L.; Chen, L.; Graham, R.; Chatterjee, A. Resident and fellow participation in thyroid and parathyroid surgery: An acs-nsqip clinical outcomes analysis. J. Surg. Res. 2017, 220, 346–352. [Google Scholar] [CrossRef]
- Ambe, P.C.; Wassenberg, D.R. Is sutureless thyroid surgery safe in the hands of surgical trainees. A single centre retrospective study. BMC Res. Notes 2016, 9, 118. [Google Scholar] [CrossRef]
- Mishra, A.; Agarwal, G.; Agarwal, A.; Mishra, S.K. Safety and efficacy of total thyroidectomy in hands of endocrine surgery trainees. Am. J. Surg. 1999, 178, 377–380. [Google Scholar] [CrossRef]
- Reeve, T.S.; Curtin, A.; Fingleton, L.; Kennedy, P.; Mackie, W.; Porter, T.; Simons, D.; Townend, D.; Delbridge, L. Can total thyroidectomy be performed as safely by general surgeons in provincial centers as by surgeons in specialized endocrine surgical units?: Making the case for surgical training. Arch. Surg. 1994, 129, 834–836. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Qu, S.; Liu, R.; Sheng, C.; Shi, X.; Zhu, G.; Murugan, A.K.; Guan, H.; Yu, H.; Wang, Y.; et al. Tert promoter mutations and their association with braf v600e mutation and aggressive clinicopathological characteristics of thyroid cancer. J. Clin. Endocrinol. Metab. 2014, 99, E1130–E1136. [Google Scholar] [CrossRef] [PubMed]
- Melo, M.; da Rocha, A.G.; Vinagre, J.; Batista, R.; Peixoto, J.; Tavares, C.; Celestino, R.; Almeida, A.; Salgado, C.; Eloy, C.; et al. Tert promoter mutations are a major indicator of poor outcome in differentiated thyroid carcinomas. J. Clin. Endocrinol. Metab. 2014, 99, E754–E765. [Google Scholar] [CrossRef]
- Moon, S.; Song, Y.S.; Kim, Y.A.; Lim, J.A.; Cho, S.W.; Moon, J.H.; Hahn, S.; Park, D.J.; Park, Y.J. Effects of coexistent braf(v600e) and tert promoter mutations on poor clinical outcomes in papillary thyroid cancer: A meta-analysis. Thyroid 2017, 27, 651–660. [Google Scholar] [CrossRef]
| Characteristics | CLN-Positive (n = 165) | CLN-Negative (n = 711) | p-Value |
|---|---|---|---|
| Sex | 0.001 | ||
| Female | 133 (80.6%) | 637 (89.6%) | |
| Male | 32 (19.4%) | 74 (10.4%) | |
| Age (years) | 0.355 | ||
| <55 | 148 (89.7%) | 619 (87.1%) | |
| ≥55 | 17 (10.3%) | 92 (12.9%) | |
| Tumor size (cm) | 0.59 ± 0.23 | 0.52 ± 0.23 | <0.001 |
| Microscopic capsular invasion | 70 (42.4%) | 189 (26.6%) | <0.001 |
| Multifocality | 18 (10.9%) | 77 (10.8%) | 0.976 |
| Recurrence | 13 (7.9%) | 35 (4.9%) | 0.133 |
| Mean follow-up duration (years) | 12.8 ± 4.3 | 13.4 ± 4.4 | 0.166 |
| Variable | N | Recurrence | Univariate Analysis | Multivariate Analysis | ||
|---|---|---|---|---|---|---|
| HR (95% CI) | p-Value | HR (95% CI) | p-Value | |||
| Sex | 0.016 | 0.030 | ||||
| Female | 770 | 43 (5.6%) | 1.000 | 1.000 | ||
| Male | 106 | 5 (4.7%) | 3.307 (1.245–8.787) | 3.043 (1.117–8.288) | ||
| Age (years) | 0.154 | 0.194 | ||||
| <55 | 767 | 42 (5.5%) | 1.000 | 1.000 | ||
| ≥55 | 109 | 6 (5.5%) | 1.887 (0.788–4.523) | 1.793 (0.743–4.326) | ||
| Tumor size | 0.456 | 0.732 | ||||
| ≤5 mm | 505 | 19 (3.8%) | 1.000 | 1.000 | ||
| >5 mm | 371 | 29 (7.8%) | 1.254 (0.691–2.276) | 1.119 (0.587–2.133) | ||
| Multifocality | 0.184 | 0.243 | ||||
| Absent | 781 | 42 (5.4%) | 1.000 | 1.000 | ||
| Present | 95 | 6 (6.3%) | 1.844 (0.773–4.400) | 1.700 (0.698–4.143) | ||
| Microscopic capsular invasion | 0.262 | 0.540 | ||||
| Absent | 617 | 33 (5.3%) | 1.000 | 1.000 | ||
| Present | 259 | 15 (5.8%) | 1.432 (0.765–2.680) | 1.242 (0.621–2.484) | ||
| CLN metastasis | 0.069 | 0.123 | ||||
| Absent | 711 | 35 (4.9%) | 1.000 | 1.000 | ||
| Present | 165 | 13 (7.9%) | 1.815 (0.954–3.452) | 1.687 (0.868–3.275) | ||
| Variable | N | Lateral LN Recurrence | Univariate Analysis | Multivariate Analysis | ||
|---|---|---|---|---|---|---|
| HR (95% CI) | p-Value | HR (95% CI) | p-Value | |||
| Sex | <0.001 | 0.007 | ||||
| Female | 770 | 11 (1.4%) | 1.000 | 1.000 | ||
| Male | 106 | 4 (3.8%) | 10.620 (2.833–39.808) | 7.113 (1.708–29.622) | ||
| Age (years) | 0.087 | 0.090 | ||||
| <55 | 767 | 12 (1.6%) | 1.000 | 1.000 | ||
| ≥55 | 109 | 3 (2.8%) | 3.121 (0.848–11.483) | 3.190 (0.835–12.184) | ||
| Tumor size | 0.055 | 0.048 | ||||
| ≤5 mm | 505 | 3 (0.6%) | 1.000 | 1.000 | ||
| >5 mm | 371 | 12 (3.2%) | 3.515 (0.975–12.675) | 3.790 (1.009–14.236) | ||
| Multifocality | 0.407 | 0.375 | ||||
| Absent | 781 | 13 (1.7%) | 1.000 | 1.000 | ||
| Present | 95 | 2 (2.1%) | 1.895 (0.418–8.595) | 2.023 (0.427–9.591) | ||
| Microscopic capsular invasion | 0.938 | 0.444 | ||||
| Absent | 617 | 11 (1.8%) | 1.000 | 1.000 | ||
| Present | 259 | 4 (1.5%) | 1.047 (0.327–3.348) | 0.616 (0.178–2.134) | ||
| CLN metastasis | 0.007 | 0.023 | ||||
| Absent | 711 | 8 (1.1%) | 1.000 | 1.000 | ||
| Present | 165 | 7 (4.2%) | 4.064 (1.463–11.292) | 3.649 (1.192–11.169) | ||
| Complications | CLN Positive (n = 165) | CLN Negative (n = 711) | p-Value |
|---|---|---|---|
| Complication | 0.519 | ||
| Absent | 158 (95.8%) | 674 (94.8%) | |
| Present | 7 (4.2%) | 37 (5.2%) | |
| Hematoma | 1.000 | ||
| Absent | 165 (100%) | 710 (99.9%) | |
| Present | 0 (0%) | 1 (0.1%) | |
| Seroma | 0.331 | ||
| Absent | 164 (99.4%) | 696 (97.9%) | |
| Present | 1 (0.6%) | 15 (2.1%) | |
| Hoarseness (transient) | 1.000 | ||
| Absent | 163 (98.8%) | 701 (98.6%) | |
| Present | 2 (1.2%) | 10 (1.4%) | |
| Hypocalcemia (transient) | 0.700 | ||
| Absent | 164 (99.4%) | 701 (98.6%) | |
| Present | 1 (0.6%) | 10 (1.4%) | |
| RLN injury | 0.023 | ||
| Absent | 162 (98.2%) | 710 (99.9%) | |
| Present | 3 (1.8%) | 1 (0.1%) |
| Characteristics | First Period 1986–1997 (n = 46) | Second Period 1998–2009 (n = 830) | p-Value |
|---|---|---|---|
| Sex | 0.010 | ||
| Female | 46 (100%) | 724 (87.2%) | |
| Male | 0 (0%) | 106 (12.8%) | |
| Age (years) | 0.030 | ||
| <55 | 45 (97.8%) | 722 (87.0%) | |
| ≥55 | 1 (2.2%) | 108 (13.0%) | |
| Tumor size (cm) | 0.63 ± 0.30 | 0.53 ± 0.23 | 0.020 |
| Microscopic capsular invasion | 10 (21.7%) | 249 (30.0%) | 0.232 |
| Multifocality | 2 (4.3%) | 93 (11.2%) | 0.219 |
| CLN metastasis | 8 (17.4%) | 157 (18.9%) | 0.797 |
| Complication | 1 (2.2%) | 43 (5.2%) | 0.723 |
| Hematoma | 0 (0%) | 1 (0.1%) | 1.000 |
| Seroma | 0 (0%) | 16 (1.9%) | 1.000 |
| Hoarseness (transient) | 0 (0%) | 12 (1.4%) | 1.000 |
| Hypocalcemia (transient) | 1 (2.2%) | 10 (1.2%) | 0.449 |
| RLN injury | 0 (0%) | 4 (0.5%) | 1.000 |
| Mean follow-up duration (years) | 24.2 ± 2.4 | 12.7 ± 3.6 | <0.001 |
| Recurrence | 9 (19.6%) | 39 (4.7%) | <0.001 |
| Recurrence-free survival duration | |||
| ≤5 years | 3 (6.5%) | 16 (1.9%) | 0.073 |
| >5 years | 6 (13.0%) | 23 (2.8%) | 0.003 |
| Mean recurrence-free survival duration (years) | 7.4 ± 3.4 | 5.7 ± 3.6 | 0.227 |
| Characteristics | Total Patients (n = 876) |
|---|---|
| Sex | |
| Female | 770 (87.9%) |
| Male | 106 (12.1%) |
| Age (years) | |
| <55 | 767 (87.6%) |
| ≥55 | 109 (12.4%) |
| Tumor size (cm) | 0.53 ± 0.23 |
| Microscopic capsular invasion | |
| Absent | 617 (70.4%) |
| Present | 259 (29.6%) |
| Multifocality | |
| Absent | 781 (89.2%) |
| Present | 95 (10.8%) |
| CLN metastasis | |
| Absent | 711 (81.2%) |
| Present | 165 (18.8%) |
| Recurrence | |
| Absent | 828 (94.5%) |
| Present | 48 (5.5%) |
| Mean follow-up duration (years) | 13.3 ± 4.4 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Choi, S.M.; Kim, J.K.; Lee, C.R.; Lee, J.; Jeong, J.J.; Nam, K.-H.; Chung, W.Y.; Kang, S.-W. Completion Total Thyroidectomy Is Not Necessary for Papillary Thyroid Microcarcinoma with Occult Central Lymph Node Metastasis: A Long-Term Serial Follow-Up. Cancers 2020, 12, 3032. https://doi.org/10.3390/cancers12103032
Choi SM, Kim JK, Lee CR, Lee J, Jeong JJ, Nam K-H, Chung WY, Kang S-W. Completion Total Thyroidectomy Is Not Necessary for Papillary Thyroid Microcarcinoma with Occult Central Lymph Node Metastasis: A Long-Term Serial Follow-Up. Cancers. 2020; 12(10):3032. https://doi.org/10.3390/cancers12103032
Chicago/Turabian StyleChoi, Soon Min, Jin Kyong Kim, Cho Rok Lee, Jandee Lee, Jong Ju Jeong, Kee-Hyun Nam, Woong Youn Chung, and Sang-Wook Kang. 2020. "Completion Total Thyroidectomy Is Not Necessary for Papillary Thyroid Microcarcinoma with Occult Central Lymph Node Metastasis: A Long-Term Serial Follow-Up" Cancers 12, no. 10: 3032. https://doi.org/10.3390/cancers12103032
APA StyleChoi, S. M., Kim, J. K., Lee, C. R., Lee, J., Jeong, J. J., Nam, K.-H., Chung, W. Y., & Kang, S.-W. (2020). Completion Total Thyroidectomy Is Not Necessary for Papillary Thyroid Microcarcinoma with Occult Central Lymph Node Metastasis: A Long-Term Serial Follow-Up. Cancers, 12(10), 3032. https://doi.org/10.3390/cancers12103032






