Carbon Ion Radiobiology
Simple Summary
Abstract
1. Introduction
2. DNA Damage
2.1. Repair Pathway Choice
2.2. Chromosome Aberrations
2.3. Summary
3. RBE
3.1. RBE Models
3.2. In Vitro Studies
3.3. In Vivo Studies
3.4. Summary
4. Hypoxia
4.1. OER in Radiotherapy
4.2. In Vitro Studies
4.3. In Vivo Studies
4.4. Summary
5. Molecular Radiobiology
5.1. Radiogenomics
5.2. Cancer Stem Cells
5.3. Combined Treatments
5.3.1. Chemotherapy
5.3.2. Targeted Therapy
5.3.3. Nanoparticles
5.3.4. Immunotherapy
5.4. Carcinogenesis
5.5. Summary
6. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Wilson, R.R. Radiological use of fast protons. Radiology 1946, 47, 487–491. [Google Scholar] [CrossRef] [PubMed]
- Tobias, C.A. Failla Memorial lecture. The future of heavy-ion science in biology and medicine. Radiat. Res. 1985, 103, 1–33. [Google Scholar] [CrossRef] [PubMed]
- Durante, M.; Paganetti, H. Nuclear physics in particle therapy: A review. Rep. Prog. Phys. 2016, 79, 096702. [Google Scholar] [CrossRef] [PubMed]
- Schardt, D.; Elsässer, T.; Schulz-Ertner, D. Heavy-ion tumor therapy: Physical and radiobiological benefits. Rev. Mod. Phys. 2010, 82, 383–425. [Google Scholar] [CrossRef]
- Durante, M.; Cucinotta, F.A. Physical basis of radiation protection in space travel. Rev. Mod. Phys. 2011, 83, 1245–1281. [Google Scholar] [CrossRef]
- Sørensen, B.S.; Overgaard, J.; Bassler, N. In vitro RBE-LET dependence for multiple particle types. Acta Oncol. 2011, 50, 757–762. [Google Scholar] [CrossRef]
- Friedrich, T.; Scholz, U.; Elsässer, T.; Durante, M.; Scholz, M. Systematic analysis of RBE and related quantities using a database of cell survival experiments with ion beam irradiation. J. Radiat. Res. 2013, 54, 494–514. [Google Scholar] [CrossRef]
- Blakely, E.A.; Ngo, F.; Curtis, S.; Tobias, C.A. Heavy-ion radiobiology: Cellular studies. Adv. Radiat. Biol. 1984, 11, 295–389. [Google Scholar] [CrossRef]
- Durante, M. New challenges in high-energy particle radiobiology. Br. J. Radiol. 2014, 87, 20130626. [Google Scholar] [CrossRef]
- Skarsgard, L.D. Radiobiology with heavy charged particles: A historical review. Phys. Medica 1998, 14 (Suppl. 1), 1–19. [Google Scholar]
- Durante, M.; Loeffler, J.S. Charged particles in radiation oncology. Nat. Rev. Clin. Oncol. 2010, 7, 37–43. [Google Scholar] [CrossRef]
- Durante, M. Eighth Warren K. Sinclair Keynote Address: Heavy Ions in Therapy and Space: Benefits and Risks. Health Phys. 2012, 103, 532–539. [Google Scholar] [CrossRef]
- Castro, J.R. Results of heavy ion radiotherapy. Radiat. Environ. Biophys. 1995, 34, 45–48. [Google Scholar] [CrossRef]
- Horsman, M.R.; Mortensen, L.S.; Busk, M.; Overgaard, J.; Horsman, M.R.; Mortensen, L.S.; Petersen, J.B.; Busk, M.; Overgaard, J. Imaging hypoxia to improve radiotherapy outcome. Nat. Rev. Clin. Oncol. 2012, 9, 674–687. [Google Scholar] [CrossRef]
- Tommasino, F.; Scifoni, E.; Durante, M. New Ions for Therapy. Int. J. Part. Ther. 2015, 2, 428–438. [Google Scholar] [CrossRef]
- Tsujii, H.; Kamada, T.; Shirai, T.; Al., E. Carbon-Ion Radiotherapy; Tsujii, H., Kamada, T., Shirai, T., Noda, K., Tsuji, H., Karasawa, K., Eds.; Springer: Japan, Tokyo, 2014; ISBN 978-4-431-54456-2. [Google Scholar]
- Kamada, T.; Tsujii, H.; Blakely, E.A.; Debus, J.; De Neve, W.; Durante, M.; Jäkel, O.; Mayer, R.; Orecchia, R.; Pötter, R.; et al. Carbon ion radiotherapy in Japan: An assessment of 20 years of clinical experience. Lancet Oncol. 2015, 16, e93–e100. [Google Scholar] [CrossRef]
- Kraft, G. Tumor therapy with heavy charged particles. Prog. Part. Nucl. Phys. 2000, 45, 473–544. [Google Scholar] [CrossRef]
- Amaldi, U.; Kraft, G. Radiotherapy with beams of carbon ions. Rep. Prog. Phys. 2005, 68, 1861–1882. [Google Scholar] [CrossRef]
- Brower, V. Carbon Ion Therapy To Debut in Europe. JNCI J. Natl. Cancer Inst. 2009, 101, 74–76. [Google Scholar] [CrossRef][Green Version]
- Malouff, T.D.; Mahajan, A.; Krishnan, S.; Beltran, C.; Seneviratne, D.S.; Trifiletti, D.M. Carbon Ion Therapy: A Modern Review of an Emerging Technology. Front. Oncol. 2020, 10. [Google Scholar] [CrossRef]
- Pompos, A.; Durante, M.; Choy, H. Heavy Ions in Cancer Therapy. JAMA Oncol. 2016, 2, 1539. [Google Scholar] [CrossRef]
- Bortfeld, T.R.; Loeffler, J.S. Three ways to make proton therapy affordable. Nature 2017, 549, 451–453. [Google Scholar] [CrossRef] [PubMed]
- Jäkel, O.; Smith, A.R.; Orton, C.G. The more important heavy charged particle radiotherapy of the future is more likely to be with heavy ions rather than protons. Med. Phys. 2013, 40, 090601. [Google Scholar] [CrossRef] [PubMed]
- Uhl, M.; Mattke, M.; Welzel, T.; Roeder, F.; Oelmann, J.; Habl, G.; Jensen, A.; Ellerbrock, M.; Jäkel, O.; Haberer, T.; et al. Highly effective treatment of skull base chordoma with carbon ion irradiation using a raster scan technique in 155 patients: First long-term results. Cancer 2014, 120, 3410–3417. [Google Scholar] [CrossRef]
- Shinoto, M.; Yamada, S.; Terashima, K.; Yasuda, S.; Shioyama, Y.; Honda, H.; Kamada, T.; Tsujii, H.; Saisho, H.; Asano, T.; et al. Carbon Ion Radiation Therapy With Concurrent Gemcitabine for Patients With Locally Advanced Pancreatic Cancer. Int. J. Radiat. Oncol. 2016, 95, 498–504. [Google Scholar] [CrossRef]
- Kawashiro, S.; Yamada, S.; Okamoto, M.; Ohno, T.; Nakano, T.; Shinoto, M.; Shioyama, Y.; Nemoto, K.; Isozaki, Y.; Tsuji, H.; et al. Multi-institutional Study of Carbon-ion Radiotherapy for Locally Advanced Pancreatic Cancer: Japan Carbon-ion Radiation Oncology Study Group (J-CROS) Study 1403 Pancreas. Int. J. Radiat. Oncol. 2018, 101, 1212–1221. [Google Scholar] [CrossRef]
- Shinoto, M.; Yamada, S.; Okamoto, M.; Shioyama, Y.; Ohno, T.; Nakano, T.; Nemoto, K.; Isozaki, Y.; Kawashiro, S.; Tsuji, H.; et al. Carbon-ion radiotherapy for locally recurrent rectal cancer: Japan Carbon-ion Radiation Oncology Study Group (J-CROS) Study 1404 Rectum. Radiother. Oncol. 2019, 132, 236–240. [Google Scholar] [CrossRef] [PubMed]
- Durante, M.; Debus, J. Heavy Charged Particles: Does Improved Precision and Higher Biological Effectiveness Translate to Better Outcome in Patients? Semin. Radiat. Oncol. 2018, 28, 160–167. [Google Scholar] [CrossRef]
- Tommasino, F.; Durante, M. Proton radiobiology. Cancers 2015, 7, 353–381. [Google Scholar] [CrossRef] [PubMed]
- Tracy, B.L.; Stevens, D.L.; Goodhead, D.T.; Hill, M.A. Variation in RBE for Survival of V79-4 Cells as a Function of Alpha-Particle (Helium Ion) Energy. Radiat. Res. 2015, 184, 33. [Google Scholar] [CrossRef]
- Brenner, D.J.; Sachs, R.K. Do low dose-rate bystander effects influence domestic radon risks? Int. J. Radiat. Biol. 2002, 78, 593–604. [Google Scholar] [CrossRef] [PubMed]
- Parker, C.; Lewington, V.; Shore, N.; Kratochwil, C.; Levy, M.; Lindén, O.; Noordzij, W.; Park, J.; Saad, F. Targeted Alpha Therapy, an Emerging Class of Cancer Agents. JAMA Oncol. 2018. [Google Scholar] [CrossRef]
- La Tessa, C.; Sivertz, M.; Chiang, I.H.; Lowenstein, D.; Rusek, A. Overview of the NASA space radiation laboratory. Life Sci. Space Res. 2016, 11, 18–23. [Google Scholar] [CrossRef] [PubMed]
- Walsh, L.; Schneider, U.; Fogtman, A.; Kausch, C.; McKenna-Lawlor, S.; Narici, L.; Ngo-Anh, J.; Reitz, G.; Sabatier, L.; Santin, G.; et al. Research plans in Europe for radiation health hazard assessment in exploratory space missions. Life Sci. Space Res. 2019, 21, 73–82. [Google Scholar] [CrossRef]
- Paganetti, H. Relative biological effectiveness (RBE) values for proton beam therapy. Variations as a function of biological endpoint, dose, and linear energy transfer. Phys. Med. Biol. 2014, 59, R419–R472. [Google Scholar] [CrossRef] [PubMed]
- Underwood, T.S.A.; Grassberger, C.; Bass, R.; MacDonald, S.M.; Meyersohn, N.M.; Yeap, B.Y.; Jimenez, R.B.; Paganetti, H. Asymptomatic Late-phase Radiographic Changes Among Chest-Wall Patients Are Associated with a Proton RBE Exceeding 1.1. Int. J. Radiat. Oncol. 2018, 101, 809–819. [Google Scholar] [CrossRef]
- Haas-Kogan, D.; Indelicato, D.; Paganetti, H.; Esiashvili, N.; Mahajan, A.; Yock, T.; Flampouri, S.; MacDonald, S.; Fouladi, M.; Stephen, K.; et al. National Cancer Institute Workshop on Proton Therapy for Children: Considerations Regarding Brainstem Injury. Int. J. Radiat. Oncol. 2018, 101, 152–168. [Google Scholar] [CrossRef]
- Paganetti, H.; Blakely, E.; Carabe-Fernandez, A.; Carlson, D.J.; Das, I.J.; Dong, L.; Grosshans, D.; Held, K.D.; Mohan, R.; Moiseenko, V.; et al. Report of the AAPM TG-256 on the relative biological effectiveness of proton beams in radiation therapy. Med. Phys. 2019, 46, e53–e78. [Google Scholar] [CrossRef]
- Lea, D.E. Action of Radiations on Living Cells; Cambridge University Press: New York, NY, USA, 1947. [Google Scholar]
- Goodhead, D.T. Initial events in the cellular effects of ionizing radiations: Clustered damage in DNA. Int. J. Radiat. Biol. 1994, 65, 7–17. [Google Scholar] [CrossRef]
- Sokol, O.; Scifoni, E.; Tinganelli, W.; Kraft-Weyrather, W.; Wiedemann, J.; Maier, A.; Boscolo, D.; Friedrich, T.; Brons, S.; Durante, M.; et al. Oxygen beams for therapy: Advanced biological treatment planning and experimental verification. Phys. Med. Biol. 2017, 62, 7798–7813. [Google Scholar] [CrossRef]
- Claesson, K.; Magnander, K.; Kahu, H.; Lindegren, S.; Hultborn, R.; Elmroth, K. RBE of α-particles from 211 At for complex DNA damage and cell survival in relation to cell cycle position. Int. J. Radiat. Biol. 2011, 87, 372–384. [Google Scholar] [CrossRef] [PubMed]
- Pinto, M.; Prise, K.M.; Michael, B.D. Evidence for Complexity at the Nanometer Scale of Radiation-Induced DNA DSBs as a Determinant of Rejoining Kinetics. Radiat. Res. 2005, 164, 73–85. [Google Scholar] [CrossRef] [PubMed]
- Asaithamby, A.; Hu, B.; Chen, D.J. Unrepaired clustered DNA lesions induce chromosome breakage in human cells. Proc. Natl. Acad. Sci. USA 2011, 108, 8293–8298. [Google Scholar] [CrossRef] [PubMed]
- Anderson, J.A.; Harper, J.V.; Cucinotta, F.A.; O’Neill, P. Participation of DNA-PKcs in DSB Repair after Exposure to High- and Low-LET Radiation. Radiat. Res. 2010, 174, 195–205. [Google Scholar] [CrossRef] [PubMed]
- Averbeck, N.B.; Topsch, J.; Scholz, M.; Kraft-Weyrather, W.; Durante, M.; Taucher-Scholz, G. Efficient Rejoining of DNA Double-Strand Breaks despite Increased Cell-Killing Effectiveness following Spread-Out Bragg Peak Carbon-Ion Irradiation. Front. Oncol. 2016, 6, 1–8. [Google Scholar] [CrossRef]
- Durante, M.; Furusawa, Y.; George, K.; Gialanella, G.; Greco, O.; Grossi, G.; Matsufuji, N.; Pugliese, M.; Yang, T.C. Rejoining and misrejoining of radiation-induced chromatin breaks. IV. Charged particles. Radiat. Res. 1998, 149, 446–454. [Google Scholar] [CrossRef]
- Oike, T.; Niimi, A.; Okonogi, N.; Murata, K.; Matsumura, A.; Noda, S.-E.; Kobayashi, D.; Iwanaga, M.; Tsuchida, K.; Kanai, T.; et al. Visualization of complex DNA double-strand breaks in a tumor treated with carbon ion radiotherapy. Sci. Rep. 2016, 6, 22275. [Google Scholar] [CrossRef]
- Depes, D.; Lee, J.-H.; Bobkova, E.; Jezkova, L.; Falkova, I.; Bestvater, F.; Pagacova, E.; Kopecna, O.; Zadneprianetc, M.; Bacikova, A.; et al. Single-molecule localization microscopy as a promising tool for γH2AX/53BP1 foci exploration. Eur. Phys. J. D 2018, 72, 158. [Google Scholar] [CrossRef]
- Bobkova, E.; Depes, D.; Lee, J.-H.; Jezkova, L.; Falkova, I.; Pagacova, E.; Kopecna, O.; Zadneprianetc, M.; Bacikova, A.; Kulikova, E.; et al. Recruitment of 53BP1 Proteins for DNA Repair and Persistence of Repair Clusters Differ for Cell Types as Detected by Single Molecule Localization Microscopy. Int. J. Mol. Sci. 2018, 19, 3713. [Google Scholar] [CrossRef]
- Frese, M.C.; Yu, V.K.; Stewart, R.D.; Carlson, D.J. A Mechanism-Based Approach to Predict the Relative Biological Effectiveness of Protons and Carbon Ions in Radiation Therapy. Int. J. Radiat. Oncol. 2012, 83, 442–450. [Google Scholar] [CrossRef]
- Georgakilas, A.G.; O’Neill, P.; Stewart, R.D. Induction and Repair of Clustered DNA Lesions: What do We Know so far? Radiat. Res. 2013, 180, 100–109. [Google Scholar] [CrossRef]
- Iliakis, G.; Mladenov, E.; Mladenova, V. Necessities in the Processing of DNA Double Strand Breaks and Their Effects on Genomic Instability and Cancer. Cancers 2019, 11, 1671. [Google Scholar] [CrossRef]
- Yajima, H.; Fujisawa, H.; Nakajima, N.I.; Hirakawa, H.; Jeggo, P.A.; Okayasu, R.; Fujimori, A. The complexity of DNA double strand breaks is a critical factor enhancing end-resection. DNA Repair (Amst) 2013, 12, 936–946. [Google Scholar] [CrossRef] [PubMed]
- Iliakis, G.; Murmann, T.; Soni, A. Alternative end-joining repair pathways are the ultimate backup for abrogated classical non-homologous end-joining and homologous recombination repair: Implications for the formation of chromosome translocations. Mutat. Res. Toxicol. Environ. Mutagen. 2015, 793, 166–175. [Google Scholar] [CrossRef] [PubMed]
- Sallmyr, A.; Tomkinson, A.E. Repair of DNA double-strand breaks by mammalian alternative end-joining pathways. J. Biol. Chem. 2018, 293, 10536–10546. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Jasin, M. An essential role for CtIP in chromosomal translocation formation through an alternative end-joining pathway. Nat. Struct. Mol. Biol. 2011, 18, 80–84. [Google Scholar] [CrossRef] [PubMed]
- Lee-Theilen, M.; Matthews, A.J.; Kelly, D.; Zheng, S.; Chaudhuri, J. CtIP promotes microhomology-mediated alternative end joining during class-switch recombination. Nat. Struct. Mol. Biol. 2010, 18, 75–79. [Google Scholar] [CrossRef]
- Averbeck, N.B.; Ringel, O.; Herrlitz, M.; Jakob, B.; Durante, M.; Taucher-Scholz, G. DNA end resection is needed for the repair of complex lesions in G1-phase human cells. Cell Cycle 2014, 13, 2509–2516. [Google Scholar] [CrossRef]
- Jeggo, P.A.; Geuting, V.; Löbrich, M. The role of homologous recombination in radiation-induced double-strand break repair. Radiother. Oncol. 2011, 101, 7–12. [Google Scholar] [CrossRef]
- Gerelchuluun, A.; Manabe, E.; Ishikawa, T.; Sun, L.; Itoh, K.; Sakae, T.; Suzuki, K.; Hirayama, R.; Asaithamby, A.; Chen, D.J.; et al. The Major DNA Repair Pathway after Both Proton and Carbon-Ion Radiation is NHEJ, but the HR Pathway is More Relevant in Carbon Ions. Radiat. Res. 2015, 183, 345–356. [Google Scholar] [CrossRef]
- Takahashi, A.; Kubo, M.; Ma, H.; Nakagawa, A.; Yoshida, Y.; Isono, M.; Kanai, T.; Ohno, T.; Furusawa, Y.; Funayama, T.; et al. Nonhomologous End-Joining Repair Plays a More Important Role than Homologous Recombination Repair in Defining Radiosensitivity after Exposure to High-LET Radiation. Radiat. Res. 2014, 182, 338–344. [Google Scholar] [CrossRef]
- Fontana, A.O.; Augsburger, M.A.; Grosse, N.; Guckenberger, M.; Lomax, A.J.; Sartori, A.A.; Pruschy, M.N. Differential DNA repair pathway choice in cancer cells after proton- and photon-irradiation. Radiother. Oncol. 2015, 116, 374–380. [Google Scholar] [CrossRef]
- Grosse, N.; Fontana, A.O.; Hug, E.B.; Lomax, A.; Coray, A.; Augsburger, M.; Paganetti, H.; Sartori, A.A.; Pruschy, M. Deficiency in homologous recombination renders mammalian cells more sensitive to proton versus photon irradiation. Int. J. Radiat. Oncol. Biol. Phys. 2014, 88, 175–181. [Google Scholar] [CrossRef]
- Soni, A.; Murmann-Konda, T.; Siemann-Loekes, M.; Pantelias, G.E.; Iliakis, G. Chromosome breaks generated by low doses of ionizing radiation in G2-phase are processed exclusively by gene conversion. DNA Repair (Amst.) 2020, 89, 102828. [Google Scholar] [CrossRef] [PubMed]
- Durante, M.; Yamada, S.; Ando, K.; Furusawa, Y.; Kawata, T.; Majima, H.; Nakano, T.; Tsujii, H. Measurements of the equivalent whole-body dose during radiation therapy by cytogenetic methods. Phys. Med. Biol. 1999, 44, 1289–1298. [Google Scholar] [CrossRef]
- George, K.; Durante, M.; Willingham, V.; Wu, H.; Yang, T.C.; Cucinotta, F.A. Biological effectiveness of accelerated particles for the induction of chromosome damage measured in metaphase and interphase human lymphocytes. Radiat. Res. 2003, 160, 425–435. [Google Scholar] [CrossRef] [PubMed]
- Ritter, S.; Durante, M. Heavy-ion induced chromosomal aberrations: A review. Mutat. Res.-Genet. Toxicol. Environ. Mutagen. 2010, 701, 38–46. [Google Scholar] [CrossRef] [PubMed]
- Anderson, R.M.; Stevens, D.L.; Goodhead, D.T. M-FISH analysis shows that complex chromosome aberrations induced by alpha-particle tracks are cumulative products of localized rearrangements. Proc. Natl. Acad. Sci. USA 2002, 99, 12167–12172. [Google Scholar] [CrossRef] [PubMed]
- Durante, M.; George, K.; Wu, H.; Cucinotta, F. a Karyotypes of human lymphocytes exposed to high-energy iron ions. Radiat. Res. 2002, 158, 581–590. [Google Scholar] [CrossRef]
- Hartel, C.; Nikoghosyan, A.; Durante, M.; Sommer, S.; Nasonova, E.; Fournier, C.; Lee, R.; Debus, J.; Schulz-Ertner, D.; Ritter, S. Chromosomal aberrations in peripheral blood lymphocytes of prostate cancer patients treated with IMRT and carbon ions. Radiother. Oncol. 2010, 95, 73–78. [Google Scholar] [CrossRef]
- Pignalosa, D.; Lee, R.; Hartel, C.; Sommer, S.; Nikoghosyan, A.; Debus, J.; Ritter, S.; Durante, M. Chromosome inversions in lymphocytes of prostate cancer patients treated with X-rays and carbon ions. Radiother. Oncol. 2013, 109, 256–261. [Google Scholar] [CrossRef]
- Durante, M.; Yamada, S.; Ando, K.; Furusawa, Y.; Kawata, T.; Majima, H.; Nakano, T.; Tsujii, H. X-rays vs. carbon-ion tumor therapy: Cytogenetic damage in lymphocytes. Int. J. Radiat. Oncol. Biol. Phys. 2000, 47, 793–798. [Google Scholar] [CrossRef]
- Elsässer, T.; Weyrather, W.K.; Friedrich, T.; Durante, M.; Iancu, G.; Krämer, M.; Kragl, G.; Brons, S.; Winter, M.; Weber, K.-J.; et al. Quantification of the relative biological effectiveness for ion beam radiotherapy: Direct experimental comparison of proton and carbon ion beams and a novel approach for treatment planning. Int. J. Radiat. Oncol. Biol. Phys. 2010, 78, 1177–1183. [Google Scholar] [CrossRef]
- IAEA Relative Biological Effectiveness in Ion Beam Therapy. In Techinical Report Sereies No. 461; IAEA: Vienna, Austria, 2008; p. 153.
- ICRU Report 93—Prescribing, Recording and Reporting Light Ion Beam Therapy. J. ICRU 2016, 16. [CrossRef]
- Grün, R.; Friedrich, T.; Krämer, M.; Zink, K.; Durante, M.; Engenhart-Cabillic, R.; Scholz, M. Assessment of potential advantages of relevant ions for particle therapy: A model based study. Med. Phys. 2015, 42, 1037–1047. [Google Scholar] [CrossRef]
- ICRU Report 78-Prescribing, Recording, and Reporting Proton-Beam Therapy: Contents. J. ICRU 2007, 7. [CrossRef]
- Mutter, R.W.; Jethwa, K.R.; Wan Chan Tseung, H.S.; Wick, S.M.; Kahila, M.M.H.; Viehman, J.K.; Shumway, D.A.; Corbin, K.S.; Park, S.S.; Remmes, N.B.; et al. Incorporation of Biologic Response Variance Modeling Into the Clinic: Limiting Risk of Brachial Plexopathy and Other Late Effects of Breast Cancer Proton Beam Therapy. Pract. Radiat. Oncol. 2020, 10, e71–e81. [Google Scholar] [CrossRef]
- Stewart, R.D.; Carlson, D.J.; Butkus, M.P.; Hawkins, R.; Friedrich, T.; Scholz, M. A comparison of mechanism-inspired models for particle relative biological effectiveness (RBE). Med. Phys. 2018, 45, e925–e952. [Google Scholar] [CrossRef]
- Kanai, T.; Matsufuji, N.; Miyamoto, T.; Mizoe, J.; Kamada, T.; Tsuji, H.; Kato, H.; Baba, M.; Tsujii, H. Examination of GyE system for HIMAC carbon therapy. Int. J. Radiat. Oncol. Biol. Phys. 2006, 64, 650–656. [Google Scholar] [CrossRef]
- Hawkins, R.B. A microdosimetric-kinetic model for the effect of non-Poisson distribution of lethal lesions on the variation of RBE with LET. Radiat. Res. 2003, 160, 61–69. [Google Scholar] [CrossRef]
- Scholz, M.; Kellerer, A.M.; Kraft-Weyrather, W.; Kraft, G. Computation of cell survival in heavy ion beams for therapy: The model and its approximation. Radiat. Environ. Biophys. 1997, 36, 59–66. [Google Scholar] [CrossRef]
- Grün, R.; Friedrich, T.; Elsässer, T.; Krämer, M.; Zink, K.; Karger, C.P.; Durante, M.; Engenhart-Cabillic, R.; Scholz, M. Impact of enhancements in the local effect model (LEM) on the predicted RBE-weighted target dose distribution in carbon ion therapy. Phys. Med. Biol. 2012, 57, 7261–7274. [Google Scholar] [CrossRef]
- Gillmann, C.; Jäkel, O.; Karger, C.P. RBE-weighted doses in target volumes of chordoma and chondrosarcoma patients treated with carbon ion radiotherapy: Comparison of local effect models I and IV. Radiother. Oncol. 2019, 141, 234–238. [Google Scholar] [CrossRef]
- Fossati, P.; Molinelli, S.; Matsufuji, N.; Ciocca, M.; Mirandola, A.; Mairani, A.; Mizoe, J.; Hasegawa, A.; Imai, R.; Kamada, T.; et al. Dose prescription in carbon ion radiotherapy: A planning study to compare NIRS and LEM approaches with a clinically-oriented strategy. Phys. Med. Biol. 2012, 57, 7543–7554. [Google Scholar] [CrossRef]
- Steinsträter, O.; Grün, R.; Scholz, U.; Friedrich, T.; Durante, M.; Scholz, M. Mapping of RBE-weighted doses between HIMAC- and LEM-based treatment planning systems for carbon ion therapy. Int. J. Radiat. Oncol. Biol. Phys. 2012, 84, 854–860. [Google Scholar] [CrossRef]
- Mein, S.; Klein, C.; Kopp, B.; Magro, G.; Harrabi, S.; Karger, C.P.; Haberer, T.; Debus, J.; Abdollahi, A.; Dokic, I.; et al. Assessment of RBE-Weighted Dose Models for Carbon Ion Therapy Toward Modernization of Clinical Practice at HIT: In Vitro, in Vivo, and in Patients. Int. J. Radiat. Oncol. 2020. [Google Scholar] [CrossRef]
- Loeffler, J.S.; Durante, M. Charged particle therapy--optimization, challenges and future directions. Nat. Rev. Clin. Oncol. 2013, 10, 411–424. [Google Scholar] [CrossRef]
- Fowler, J.F. 21 Years of biologically effective dose. Br. J. Radiol. 2010, 83, 554–568. [Google Scholar] [CrossRef]
- Salama, J.K.; Kirkpatrick, J.P.; Yin, F.-F. Stereotactic body radiotherapy treatment of extracranial metastases. Nat. Rev. Clin. Oncol. 2012, 9, 654–665. [Google Scholar] [CrossRef]
- Tubin, S.; Yan, W.; Mourad, W.F.; Fossati, P.; Khan, M.K. The future of radiation-induced abscopal response: Beyond conventional radiotherapy approaches. Future Oncol. 2020, 16, 1137–1151. [Google Scholar] [CrossRef]
- Tubin, S.; Popper, H.H.; Brcic, L. Novel stereotactic body radiation therapy (SBRT)-based partial tumor irradiation targeting hypoxic segment of bulky tumors (SBRT-PATHY): Improvement of the radiotherapy outcome by exploiting the bystander and abscopal effects. Radiat. Oncol. 2019, 14, 21. [Google Scholar] [CrossRef]
- Emami, B. Tolerance of Normal Tissue to Therapeutic Radiation. Rep. Radiother. Oncol. 2013, 1, 123–127. [Google Scholar]
- Friedrich, T.; Scholz, U.; Durante, M.; Scholz, M. RBE of ion beams in hypofractionated radiotherapy (SBRT). Phys. Med. 2014, 30, 588–591. [Google Scholar] [CrossRef]
- Chapman, J.D. Can the two mechanisms of tumor cell killing by radiation be exploited for therapeutic gain? J. Radiat. Res. 2014, 55, 2–9. [Google Scholar] [CrossRef]
- Jeong, J.; Taasti, V.T.; Jackson, A.; Deasy, J.O. The Relative Biological Effectiveness of Carbon Ion Radiation Therapy for Early Stage Lung Cancer. Radiother. Oncol. 2020. [Google Scholar] [CrossRef]
- Saager, M.; Glowa, C.; Peschke, P.; Brons, S.; Gr?n, R.; Scholz, M.; Huber, P.E.; Debus, J.; Karger, C.P. The relative biological effectiveness of carbon ion irradiations of the rat spinal cord increases linearly with LET up to 99 keV/micron. Acta Oncol. (Madr.) 2016, 55, 1512–1515. [Google Scholar] [CrossRef]
- Saager, M.; Glowa, C.; Peschke, P.; Brons, S.; Scholz, M.; Huber, P.E.; Debus, J.; Karger, C.P. Carbon Ion Irradiation of the Rat Spinal Cord: Dependence of the Relative Biological Effectiveness on Linear Energy Transfer. Int. J. Radiat. Oncol. Biol. Phys. 2014, 90, 63–70. [Google Scholar] [CrossRef]
- Saager, M.; Glowa, C.; Peschke, P.; Brons, S.; Grün, R.; Scholz, M.; Debus, J.; Karger, C.P. Fractionated carbon ion irradiations of the rat spinal cord: Comparison of the relative biological effectiveness with predictions of the local effect model. Radiat. Oncol. 2020, 15, 6. [Google Scholar] [CrossRef]
- Karger, C.P.; Peschke, P.; Sanchez-Brandelik, R.; Scholz, M.; Debus, J. Radiation tolerance of the rat spinal cord after 6 and 18 fractions of photons and carbon ions: Experimental results and clinical implications. Int. J. Radiat. Oncol. 2006, 66, 1488–1497. [Google Scholar] [CrossRef]
- Saager, M.; Glowa, C.; Peschke, P.; Brons, S.; Grün, R.; Scholz, M.; Huber, P.E.; Debus, J.; Karger, C.P. Split dose carbon ion irradiation of the rat spinal cord: Dependence of the relative biological effectiveness on dose and linear energy transfer. Radiother. Oncol. 2015, 117, 358–363. [Google Scholar] [CrossRef]
- Saager, M.; Peschke, P.; Welzel, T.; Huang, L.; Brons, S.; Grün, R.; Scholz, M.; Debus, J.; Karger, C.P. Late normal tissue response in the rat spinal cord after carbon ion irradiation. Radiat. Oncol. 2018, 13, 5. [Google Scholar] [CrossRef]
- Ando, K.; Koike, S.; Uzawa, A.; Takai, N.; Fukawa, T.; Furusawa, Y.; Aoki, M.; Miyato, Y. Biological Gain of Carbon-ion Radiotherapy for the Early Response of Tumor Growth Delay and against Early Response of Skin Reaction in Mice. J. Radiat. Res. 2005, 46, 51–57. [Google Scholar] [CrossRef]
- Sørensen, B.S.; Horsman, M.R.; Alsner, J.; Overgaard, J.; Durante, M.; Scholz, M.; Friedrich, T.; Bassler, N. Relative biological effectiveness of carbon ions for tumor control, acute skin damage and late radiation-induced fibrosis in a mouse model. Acta Oncol. (Madr.) 2015, 54, 1623–1630. [Google Scholar] [CrossRef]
- Glowa, C.; Karger, C.P.; Brons, S.; Zhao, D.; Mason, R.P.; Huber, P.E.; Debus, J.; Peschke, P. Carbon ion radiotherapy decreases the impact of tumor heterogeneity on radiation response in experimental prostate tumors. Cancer Lett. 2016, 378, 97–103. [Google Scholar] [CrossRef]
- Glowa, C.; Peschke, P.; Brons, S.; Debus, J.; Karger, C.P. Intrinsic and extrinsic tumor characteristics are of minor relevance for the efficacy of split-dose carbon ion irradiation in three experimental prostate tumors. Radiother. Oncol. 2019, 133, 120–124. [Google Scholar] [CrossRef]
- Glowa, C.; Peschke, P.; Brons, S.; Neels, O.C.; Kopka, K.; Debus, J.; Karger, C.P. Carbon ion radiotherapy: Impact of tumor differentiation on local control in experimental prostate carcinomas. Radiat. Oncol. 2017, 12, 174. [Google Scholar] [CrossRef]
- Bert, C.; Engenhart-Cabillic, R.; Durante, M. Particle therapy for noncancer diseases. Med. Phys. 2012, 39, 1716–1727. [Google Scholar] [CrossRef]
- van der Ree, M.H.; Blanck, O.; Limpens, J.; Lee, C.H.; Balgobind, B.V.; Dieleman, E.M.T.; Wilde, A.A.M.; Zei, P.C.; de Groot, J.R.; Slotman, B.J.; et al. Cardiac radioablation—A systematic review. Heart Rhythm 2020, 17, 1381–1392. [Google Scholar] [CrossRef]
- Cuculich, P.S.; Schill, M.R.; Kashani, R.; Mutic, S.; Lang, A.; Cooper, D.; Faddis, M.; Gleva, M.; Noheria, A.; Smith, T.W.; et al. Noninvasive Cardiac Radiation for Ablation of Ventricular Tachycardia. N. Engl. J. Med. 2017, 377, 2325–2336. [Google Scholar] [CrossRef]
- Lehmann, H.I.; Graeff, C.; Simoniello, P.; Constantinescu, A.; Takami, M.; Lugenbiel, P.; Richter, D.; Eichhorn, A.; Prall, M.; Kaderka, R.; et al. Feasibility Study on Cardiac Arrhythmia Ablation Using High-Energy Heavy Ion Beams. Sci. Rep. 2016, 6, 38895. [Google Scholar] [CrossRef]
- Rapp, F.; Simoniello, P.; Wiedemann, J.; Bahrami, K.; Grünebaum, V.; Ktitareva, S.; Durante, M.; Lugenbiel, P.; Thomas, D.; Lehmann, H.I.; et al. Biological Cardiac Tissue Effects of High-Energy Heavy Ions – Investigation for Myocardial Ablation. Sci. Rep. 2019, 9, 5000. [Google Scholar] [CrossRef]
- Uzawa, A.; Ando, K.; Kase, Y.; Hirayama, R.; Matsumoto, Y.; Matsufuji, N.; Koike, S.; Kobashi, G. Designing a ridge filter based on a mouse foot skin reaction to spread out Bragg-peaks for carbon-ion radiotherapy. Radiother. Oncol. 2015, 115, 279–283. [Google Scholar] [CrossRef] [PubMed]
- Ando, S.; Koike, K.; Nojima, Y.-J.; Chen, C.K. Mouse skin reactions following fractionated irradiation with carbon ions. Int. J. Radiat. Biol. 1998, 74, 129–138. [Google Scholar] [CrossRef] [PubMed]
- Kagawa, K.; Murakami, M.; Hishikawa, Y.; Abe, M.; Akagi, T.; Yanou, T.; Kagiya, G.; Furusawa, Y.; Ando, K.; Nojima, K.; et al. Preclinical biological assessment of proton and carbon ion beams at Hyogo Ion Beam Medical Center. Int. J. Radiat. Oncol. 2002, 54, 928–938. [Google Scholar] [CrossRef]
- Raju, M.R.; Carpenter, S.G. A heavy particle comparative study. Part IV: Acute and late reactions. Br. J. Radiol. 1978, 51, 720–727. [Google Scholar] [CrossRef] [PubMed]
- Zhou, C.; Jones, B.; Moustafa, M.; Yang, B.; Brons, S.; Cao, L.; Dai, Y.; Schwager, C.; Chen, M.; Jaekel, O.; et al. Determining RBE for development of lung fibrosis induced by fractionated irradiation with carbon ions utilizing fibrosis index and high-LET BED model. Clin. Transl. Radiat. Oncol. 2019, 14, 25–32. [Google Scholar] [CrossRef]
- Zhou, C.; Jones, B.; Moustafa, M.; Schwager, C.; Bauer, J.; Yang, B.; Cao, L.; Jia, M.; Mairani, A.; Chen, M.; et al. Quantitative assessment of radiation dose and fractionation effects on normal tissue by utilizing a novel lung fibrosis index model. Radiat. Oncol. 2017, 12, 172. [Google Scholar] [CrossRef]
- Brownstein, J.M.; Wisdom, A.J.; Castle, K.D.; Mowery, Y.M.; Guida, P.; Lee, C.; Durante, M.; Kirsch, D.G. Characterizing the Potency and Impact of Carbon Ion Therapy in a Primary Mouse Model of Soft Tissue Sarcoma. Mol. Cancer Ther. 2018, 17, 858–868. [Google Scholar] [CrossRef]
- Koike, S.; Ando, K.; Oohira, C.; Fukawa, T.; Lee, R.; Takai, N.; Monobe, M.; Furusawa, Y.; Aoki, M.; Yamada, S.; et al. Relative Biological Effectiveness of 290 MeV/u Carbon Ions for the Growth Delay of a Radioresistant Murine Fibrosarcoma. J. Radiat. Res. 2002, 43, 247–255. [Google Scholar] [CrossRef] [PubMed]
- Yoshida, Y.; Ando, K.; Ando, K.; Murata, K.; Yoshimoto, Y.; Musha, A.; Kubo, N.; Kawamura, H.; Koike, S.; Uzawa, A.; et al. Evaluation of therapeutic gain for fractionated carbon-ion radiotherapy using the tumor growth delay and crypt survival assays. Radiother. Oncol. 2015, 117, 351–357. [Google Scholar] [CrossRef]
- Takahashi, A.; Yano, T.; Matsumoto, H.; Wang, X.; Ohnishi, K.; Tamamoto, T.; Tsuji, K.; Yukawa, O.; Ohnishi, T. Effects of accelerated carbon-ions on growth inhibition of transplantable human esophageal cancer in nude mice. Cancer Lett. 1998, 122, 181–186. [Google Scholar] [CrossRef]
- Peschke, P.; Karger, C.P.; Scholz, M.; Debus, J.; Huber, P.E. Relative Biological Effectiveness of Carbon Ions for Local Tumor Control of a Radioresistant Prostate Carcinoma in the Rat. Int. J. Radiat. Oncol. 2011, 79, 239–246. [Google Scholar] [CrossRef] [PubMed]
- Schmid, T.E.; Multhoff, G. Preclinical Mouse Models for the Evaluation of Novel Techniques in Particle Therapy. In Advances in Radiation Therapy; Guckenberger, M., Combs, S.E., Zips, D., Eds.; Karger: Basel, Switzerland, 2018; pp. 122–129. ISBN 978-3-318-06361-5. [Google Scholar]
- Hagiwara, Y.; Bhattacharyya, T.; Matsufuji, N.; Isozaki, Y.; Takiyama, H.; Nemoto, K.; Tsuji, H.; Yamada, S. Influence of dose-averaged linear energy transfer on tumour control after carbon-ion radiation therapy for pancreatic cancer. Clin. Transl. Radiat. Oncol. 2020, 21, 19–24. [Google Scholar] [CrossRef] [PubMed]
- Bassler, N.; Toftegaard, J.; Lühr, A.; Sørensen, B.S.; Scifoni, E.; Krämer, M.; Jäkel, O.; Mortensen, L.S.; Overgaard, J.; Petersen, J.B. LET-painting increases tumour control probability in hypoxic tumours. Acta Oncol. 2014, 53, 25–32. [Google Scholar] [CrossRef]
- Bassler, N.; Jäkel, O.; Søndergaard, C.S.; Petersen, J.B. Dose- and LET-painting with particle therapy. Acta Oncol. 2010, 49, 1170–1176. [Google Scholar] [CrossRef] [PubMed]
- Kopp, B.; Mein, S.; Dokic, I.; Harrabi, S.; Böhlen, T.T.; Haberer, T.; Debus, J.; Abdollahi, A.; Mairani, A. Development and Validation of Single Field Multi-Ion Particle Therapy Treatments. Int. J. Radiat. Oncol. Biol. Phys. 2020, 106, 194–205. [Google Scholar] [CrossRef]
- Sokol, O.; Krämer, M.; Hild, S.; Durante, M.; Scifoni, E. Kill painting of hypoxic tumors with multiple ion beams. Phys. Med. Biol. 2019, 64, 045008. [Google Scholar] [CrossRef]
- Pignalosa, D.; Durante, M. Overcoming resistance of cancer stem cells. Lancet Oncol. 2012, 13, e187–e188. [Google Scholar] [CrossRef]
- Bertout, J.A.; Patel, S.A.; Simon, M.C. The impact of O2 availability on human cancer. Nat. Rev. Cancer 2008, 8, 967–975. [Google Scholar] [CrossRef]
- Bristow, R.G.; Hill, R.P. Hypoxia and metabolism. Hypoxia, DNA repair and genetic instability. Nat. Rev. Cancer 2008, 8, 180–192. [Google Scholar] [CrossRef]
- Bayer, C.; Vaupel, P. Acute versus chronic hypoxia in tumors. Strahlenther. und Onkol. 2012, 188, 616–627. [Google Scholar] [CrossRef] [PubMed]
- Saxena; Jolly Acute vs. Chronic vs. Cyclic Hypoxia: Their Differential Dynamics, Molecular Mechanisms, and Effects on Tumor Progression. Biomolecules 2019, 9, 339. [CrossRef] [PubMed]
- Dewhirst, M.W.; Cao, Y.; Moeller, B. Cycling hypoxia and free radicals regulate angiogenesis and radiotherapy response. Nat. Rev. Cancer 2008, 8, 425–437. [Google Scholar] [CrossRef] [PubMed]
- Barker, H.E.; Paget, J.T.E.; Khan, A.A.; Harrington, K.J. The tumour microenvironment after radiotherapy: Mechanisms of resistance and recurrence. Nat. Rev. Cancer 2015, 15, 409–425. [Google Scholar] [CrossRef] [PubMed]
- Brown, J.M.; Wilson, W.R. Exploiting tumour hypoxia in cancer treatment. Nat. Rev. Cancer 2004, 4, 437–447. [Google Scholar] [CrossRef]
- Huang, J.; Li, J.J. Multiple Dynamics in Tumor Microenvironment Under Radiotherapy. Adv. Exp. Med. Biol. 2020, 1263, 175–202. [Google Scholar] [CrossRef] [PubMed]
- Pajonk, F.; Vlashi, E.; McBride, W.H. Radiation resistance of cancer stem cells: The 4 R’s of radiobiology revisited. Stem Cells 2010, 28, 639–648. [Google Scholar] [CrossRef] [PubMed]
- Ashton, T.M.; Fokas, E.; Kunz-Schughart, L.A.; Folkes, L.K.; Anbalagan, S.; Huether, M.; Kelly, C.J.; Pirovano, G.; Buffa, F.M.; Hammond, E.M.; et al. The anti-malarial atovaquone increases radiosensitivity by alleviating tumour hypoxia. Nat. Commun. 2016, 7, 12308. [Google Scholar] [CrossRef] [PubMed]
- McGowan, D.R.; Skwarski, M.; Bradley, K.M.; Campo, L.; Fenwick, J.D.; Gleeson, F.V.; Green, M.; Horne, A.; Maughan, T.S.; McCole, M.G.; et al. Buparlisib with thoracic radiotherapy and its effect on tumour hypoxia: A phase I study in patients with advanced non-small cell lung carcinoma. Eur. J. Cancer 2019, 113, 87–95. [Google Scholar] [CrossRef]
- Ashton, T.M.; McKenna, W.G.; Kunz-Schughart, L.A.; Higgins, G.S. Oxidative Phosphorylation as an Emerging Target in Cancer Therapy. Clin. Cancer Res. 2018, 24, 2482–2490. [Google Scholar] [CrossRef]
- Narayanan, D.; Ma, S.; Özcelik, D. Targeting the Redox Landscape in Cancer Therapy. Cancers 2020, 12, 1706. [Google Scholar] [CrossRef] [PubMed]
- Horsman, M.R.; Overgaard, J. The impact of hypoxia and its modification of the outcome of radiotherapy. J. Radiat. Res. 2016, 57, i90–i98. [Google Scholar] [CrossRef] [PubMed]
- Strigari, L.; Benassi, M.; Sarnelli, A.; Polico, R.; D’Andrea, M. A modified hypoxia-based TCP model to investigate the clinical outcome of stereotactic hypofractionated regimes for early stage non-small-cell lung cancer (NSCLC). Med. Phys. 2012, 39, 4502–4514. [Google Scholar] [CrossRef] [PubMed]
- Carlson, D.J.; Stewart, R.D.; Semenenko, V.A. Effects of oxygen on intrinsic radiation sensitivity: A test of the relationship between aerobic and hypoxic linear-quadratic (LQ) model parameters. Med. Phys. 2006, 33, 3105–3115. [Google Scholar] [CrossRef]
- Toma-Dasu, I.; Sandström, H.; Barsoum, P.; Dasu, A. To fractionate or not to fractionate? That is the question for the radiosurgery of hypoxic tumors. J. Neurosurg. 2014, 121, 110–115. [Google Scholar] [CrossRef]
- Wouters, B.G.; Brown, J.M. Cells at intermediate oxygen levels can be more important than the “hypoxic fraction” in determining tumor response to fractionated radiotherapy. Radiat. Res. 1997, 147, 541–550. [Google Scholar] [CrossRef]
- Furusawa, Y.; Fukutsu, K.; Aoki, M.; Itsukaichi, H.; Eguchi-Kasai, K.; Ohara, H.; Yatagai, F.; Kanai, T.; Ando, K. Inactivation of aerobic and hypoxic cells from three different cell lines by accelerated (3)He-, (12)C- and (20)Ne-ion beams. Radiat. Res. 2000, 154, 485–496. [Google Scholar] [CrossRef]
- Wenzl, T.; Wilkens, J.J. Theoretical analysis of the dose dependence of the oxygen enhancement ratio and its relevance for clinical applications. Radiat. Oncol. 2011, 6, 171. [Google Scholar] [CrossRef]
- McKeown, S.R. Defining normoxia, physoxia and hypoxia in tumours—Implications for treatment response. Br. J. Radiol. 2014, 87, 1–12. [Google Scholar] [CrossRef]
- Castro, J.R.; Saunders, W.M.; Tobias, C.A.; Chen, G.T.Y.; Curtis, S.; Lyman, J.T.; Michael Collier, J.; Pitluck, S.; Woodruff, K.A.; Blakely, E.A.; et al. Treatment of cancer with heavy charged particles. Int. J. Radiat. Oncol. Biol. Phys. 1982, 8, 2191–2198. [Google Scholar] [CrossRef]
- Nakano, T.; Suzuki, Y.; Ohno, T.; Kato, S.; Suzuki, M.; Morita, S.; Sato, S.; Oka, K.; Tsujii, H. Carbon beam therapy overcomes the radiation resistance of uterine cervical cancer originating from hypoxia. Clin. Cancer Res. 2006, 12, 2185–2190. [Google Scholar] [CrossRef]
- Huart, C.; Chen, J.; Le Calvé, B.; Michiels, C.; Wéra, A.-C. Could Protons and Carbon Ions Be the Silver Bullets Against Pancreatic Cancer? Int. J. Mol. Sci. 2020, 21, 4767. [Google Scholar] [CrossRef] [PubMed]
- Koong, A.C.; Mehta, V.K.; Le, Q.T.; Fisher, G.A.; Terris, D.J.; Brown, J.M.; Bastidas, A.J.; Vierra, M. Pancreatic tumors show high levels of hypoxia. Int. J. Radiat. Oncol. Biol. Phys. 2000, 48, 919–922. [Google Scholar] [CrossRef]
- Tinganelli, W.; Durante, M.; Hirayama, R.; Krämer, M.; Maier, A.; Kraft-Weyrather, W.; Furusawa, Y.; Friedrich, T.; Scifoni, E. Kill-painting of hypoxic tumours in charged particle therapy. Sci. Rep. 2015, 5, 17016. [Google Scholar] [CrossRef] [PubMed]
- Scifoni, E.; Tinganelli, W.; Weyrather, W.K.; Durante, M.; Maier, A.; Krämer, M. Including oxygen enhancement ratio in ion beam treatment planning: Model implementation and experimental verification. Phys. Med. Biol. 2013, 58, 3871–3895. [Google Scholar] [CrossRef]
- Dokic, I.; Mairani, A.; Niklas, M.; Zimmermann, F.; Chaudhri, N.; Krunic, D.; Tessonnier, T.; Ferrari, A.; Parodi, K.; Jäkel, O.; et al. Next generation multi-scale biophysical characterization of high precision cancer particle radiotherapy using clinical proton, helium-, carbon- and oxygen ion beams. Oncotarget 2016, 7, 56676–56689. [Google Scholar] [CrossRef] [PubMed]
- Kurz, C.; Mairani, A.; Parodi, K. First experimental-based characterization of oxygen ion beam depth dose distributions at the Heidelberg Ion-Beam Therapy Center. Phys. Med. Biol. 2012, 57, 5017–5034. [Google Scholar] [CrossRef]
- Inaniwa, T.; Kanematsu, N.; Noda, K.; Kamada, T. Treatment planning of intensity modulated composite particle therapy with dose and linear energy transfer optimization. Phys. Med. Biol. 2017, 62, 5180–5197. [Google Scholar] [CrossRef]
- Toma-Dasu, I.; Uhrdin, J.; Antonovic, L.; Dasu, A.; Nuyts, S.; Dirix, P.; Haustermans, K.; Brahme, A. Dose prescription and treatment planning based on FMISO-PET hypoxia. Acta Oncol. (Madr.) 2012, 51, 222–230. [Google Scholar] [CrossRef]
- Mortensen, L.S.; Buus, S.; Nordsmark, M.; Bentzen, L.; Munk, O.L.; Keiding, S.; Overgaard, J. Identifying hypoxia in human tumors: A correlation study between 18F-FMISO PET and the Eppendorf oxygen-sensitive electrode. Acta Oncol. 2010, 49, 934–940. [Google Scholar] [CrossRef]
- Dubois, L.J.; Lieuwes, N.G.; Janssen, M.H.M.; Peeters, W.J.M.; Windhorst, A.D.; Walsh, J.C.; Kolb, H.C.; Ollers, M.C.; Bussink, J.; van Dongen, G.A.M.S.; et al. Preclinical evaluation and validation of [18F]HX4, a promising hypoxia marker for PET imaging. Proc. Natl. Acad. Sci. USA 2011, 108, 14620–14625. [Google Scholar] [CrossRef]
- Tinganelli, W.; Ma, N.-Y.; Von Neubeck, C.; Maier, A.; Schicker, C.; Kraft-Weyrather, W.; Durante, M. Influence of acute hypoxia and radiation quality on cell survival. J. Radiat. Res. 2013, 54, i23–i30. [Google Scholar] [CrossRef]
- Ma, N.-Y.; Tinganelli, W.; Maier, A.; Durante, M.; Kraft-Weyrather, W. Influence of chronic hypoxia and radiation quality on cell survival. J. Radiat. Res. 2013, 54, i13–i22. [Google Scholar] [CrossRef]
- Hirayama, R.; Uzawa, A.; Takase, N.; Matsumoto, Y.; Noguchi, M.; Koda, K.; Ozaki, M.; Yamashita, K.; Li, H.; Kase, Y.; et al. Evaluation of SCCVII tumor cell survival in clamped and non-clamped solid tumors exposed to carbon-ion beams in comparison to X-rays. Mutat. Res. Toxicol. Environ. Mutagen. 2013, 756, 146–151. [Google Scholar] [CrossRef] [PubMed]
- Fukawa, T.; Takematsu, K.; Oka, K.; Koike, S.; Ando, K.; Kobayashi, H.; Tanishita, K. Differences in pO2 Peaks of a Murine Fibrosarcoma between Carbon-ion and X-ray Irradiation. J. Radiat. Res. 2004, 45, 303–308. [Google Scholar] [CrossRef] [PubMed]
- Ando, K.; Koike, S.; Ohira, C.; Chen, Y.J.; Nojima, K.; Ando, S.; Ohbuchi, T.; Kobayashi, N.; Shimizu, W.; Urano, M. Accelerated reoxygenation of a murine fibrosarcoma after carbon-ion radiation. Int. J. Radiat. Biol. 1999, 75, 505–512. [Google Scholar] [CrossRef] [PubMed]
- Bendinger, A.L.; Seyler, L.; Saager, M.; Debus, C.; Peschke, P.; Komljenovic, D.; Debus, J.; Peter, J.; Floca, R.O.; Karger, C.P.; et al. Impact of Single Dose Photons and Carbon Ions on Perfusion and Vascular Permeability: A Dynamic Contrast-Enhanced MRI Pilot Study in the Anaplastic Rat Prostate Tumor R3327-AT1. Radiat. Res. 2019, 193, 34. [Google Scholar] [CrossRef] [PubMed]
- Semenza, G.L. Pharmacologic Targeting of Hypoxia-Inducible Factors. Annu. Rev. Pharmacol. Toxicol. 2019, 59, 379–403. [Google Scholar] [CrossRef] [PubMed]
- Pugh, C.W.; Ratcliffe, P.J. New horizons in hypoxia signaling pathways. Exp. Cell Res. 2017, 356, 116–121. [Google Scholar] [CrossRef] [PubMed]
- Subtil, F.S.B.; Wilhelm, J.; Bill, V.; Westholt, N.; Rudolph, S.; Fischer, J.; Scheel, S.; Seay, U.; Fournier, C.; Taucher-Scholz, G.; et al. Carbon ion radiotherapy of human lung cancer attenuates HIF-1 signaling and acts with considerably enhanced therapeutic efficiency. FASEB J. 2014, 28, 1412–1421. [Google Scholar] [CrossRef]
- Valable, S.; Gérault, A.N.; Lambert, G.; Leblond, M.M.; Anfray, C.; Toutain, J.; Bordji, K.; Petit, E.; Bernaudin, M.; Pérès, E.A. Impact of Hypoxia on Carbon Ion Therapy in Glioblastoma Cells: Modulation by LET and Hypoxia-Dependent Genes. Cancers 2020, 12, 2019. [Google Scholar] [CrossRef]
- Wozny, A.-S.; Lauret, A.; Battiston-Montagne, P.; Guy, J.-B.; Beuve, M.; Cunha, M.; Saintigny, Y.; Blond, E.; Magne, N.; Lalle, P.; et al. Differential pattern of HIF-1α expression in HNSCC cancer stem cells after carbon ion or photon irradiation: One molecular explanation of the oxygen effect. Br. J. Cancer 2017, 116, 1340–1349. [Google Scholar] [CrossRef] [PubMed]
- Wozny, A.-S.; Vares, G.; Alphonse, G.; Lauret, A.; Monini, C.; Magné, N.; Cuerq, C.; Fujimori, A.; Monboisse, J.-C.; Beuve, M.; et al. ROS Production and Distribution: A New Paradigm to Explain the Differential Effects of X-ray and Carbon Ion Irradiation on Cancer Stem Cell Migration and Invasion. Cancers 2019, 11, 468. [Google Scholar] [CrossRef] [PubMed]
- Baumann, M.; Krause, M.; Overgaard, J.; Debus, J.; Bentzen, S.M.; Daartz, J.; Richter, C.; Zips, D.; Bortfeld, T. Radiation oncology in the era of precision medicine. Nat. Rev. Cancer 2016, 16, 234–249. [Google Scholar] [CrossRef] [PubMed]
- Kerns, S.L.; Chuang, K.-H.; Hall, W.; Werner, Z.; Chen, Y.; Ostrer, H.; West, C.; Rosenstein, B. Radiation biology and oncology in the genomic era. Br. J. Radiol. 2018, 91, 20170949. [Google Scholar] [CrossRef]
- Rosenstein, B.S. Radiogenomics: Identification of Genomic Predictors for Radiation Toxicity. Semin. Radiat. Oncol. 2017, 27, 300–309. [Google Scholar] [CrossRef] [PubMed]
- West, C.; Rosenstein, B.S. Establishment of a Radiogenomics Consortium. Int. J. Radiat. Oncol. 2010, 76, 1295–1296. [Google Scholar] [CrossRef]
- Barnett, G.C.; West, C.M.L.; Dunning, A.M.; Elliott, R.M.; Coles, C.E.; Pharoah, P.D.P.; Burnet, N.G. Normal tissue reactions to radiotherapy: Towards tailoring treatment dose by genotype. Nat. Rev. Cancer 2009, 9, 134–142. [Google Scholar] [CrossRef] [PubMed]
- Bergom, C.; West, C.M.; Higginson, D.S.; Abazeed, M.E.; Arun, B.; Bentzen, S.M.; Bernstein, J.L.; Evans, J.D.; Gerber, N.K.; Kerns, S.L.; et al. The Implications of Genetic Testing on Radiation Therapy Decisions: A Guide for Radiation Oncologists. Int. J. Radiat. Oncol. 2019, 105, 698–712. [Google Scholar] [CrossRef]
- Swift, M.; Morrell, D.; Massey, R.B.; Chase, C.L. Incidence of Cancer in 161 Families Affected by Ataxia–Telangiectasia. N. Engl. J. Med. 1991, 325, 1831–1836. [Google Scholar] [CrossRef]
- Barnett, G.C.; Coles, C.E.; Elliott, R.M.; Baynes, C.; Luccarini, C.; Conroy, D.; Wilkinson, J.S.; Tyrer, J.; Misra, V.; Platte, R.; et al. Independent validation of genes and polymorphisms reported to be associated with radiation toxicity: A prospective analysis study. Lancet Oncol. 2012, 13, 65–77. [Google Scholar] [CrossRef]
- Fachal, L.; Gómez-Caamaño, A.; Barnett, G.C.; Peleteiro, P.; Carballo, A.M.; Calvo-Crespo, P.; Kerns, S.L.; Sánchez-García, M.; Lobato-Busto, R.; Dorling, L.; et al. A three-stage genome-wide association study identifies a susceptibility locus for late radiotherapy toxicity at 2q24.1. Nat. Genet. 2014, 46, 891–894. [Google Scholar] [CrossRef] [PubMed]
- Barnett, G.C.; Thompson, D.; Fachal, L.; Kerns, S.; Talbot, C.; Elliott, R.M.; Dorling, L.; Coles, C.E.; Dearnaley, D.P.; Rosenstein, B.S.; et al. A genome wide association study (GWAS) providing evidence of an association between common genetic variants and late radiotherapy toxicity. Radiother. Oncol. 2014, 111, 178–185. [Google Scholar] [CrossRef] [PubMed]
- Andreassen, C.N.; Rosenstein, B.S.; Kerns, S.L.; Ostrer, H.; De Ruysscher, D.; Cesaretti, J.A.; Barnett, G.C.; Dunning, A.M.; Dorling, L.; West, C.M.L.; et al. Individual patient data meta-analysis shows a significant association between the ATM rs1801516 SNP and toxicity after radiotherapy in 5456 breast and prostate cancer patients. Radiother. Oncol. 2016, 121, 431–439. [Google Scholar] [CrossRef]
- Kerns, S.L.; Fachal, L.; Dorling, L.; Barnett, G.C.; Baran, A.; Peterson, D.R.; Hollenberg, M.; Hao, K.; Di Narzo, A.; Ahsen, M.E.; et al. Radiogenomics Consortium Genome-Wide Association Study Meta-Analysis of Late Toxicity After Prostate Cancer Radiotherapy. JNCI J. Natl. Cancer Inst. 2020, 112, 179–190. [Google Scholar] [CrossRef]
- Schumacher, F.R.; Al Olama, A.A.; Berndt, S.I.; Benlloch, S.; Ahmed, M.; Saunders, E.J.; Dadaev, T.; Leongamornlert, D.; Anokian, E.; Cieza-Borrella, C.; et al. Association analyses of more than 140,000 men identify 63 new prostate cancer susceptibility loci. Nat. Genet. 2018, 50, 928–936. [Google Scholar] [CrossRef] [PubMed]
- Morton, L.M.; Ricks-Santi, L.; West, C.M.L.; Rosenstein, B.S. Radiogenomic predictors of adverse effects following charged particle therapy. Int. J. Part. Ther. 2019, 5, 103–113. [Google Scholar] [CrossRef]
- West, C. Translational radiobiology: Radiogenomics. In Proceedings of the PTCOG-58, Manchester, UK, 10–15 June 2019. [Google Scholar]
- Story, M.D.; Durante, M. Radiogenomics. Med. Phys. 2018, 45. [Google Scholar] [CrossRef]
- Akino, Y.; Teshima, T.; Kihara, A.; Kodera-Suzumoto, Y.; Inaoka, M.; Higashiyama, S.; Furusawa, Y.; Matsuura, N. Carbon-Ion Beam Irradiation Effectively Suppresses Migration and Invasion of Human Non?Small-Cell Lung Cancer Cells. Int. J. Radiat. Oncol. 2009, 75, 475–481. [Google Scholar] [CrossRef] [PubMed]
- Kamlah, F.; Hänze, J.; Arenz, A.; Seay, U.; Hasan, D.; Juricko, J.; Bischoff, B.; Gottschald, O.R.; Fournier, C.; Taucher-Scholz, G.; et al. Comparison of the effects of carbon ion and photon irradiation on the angiogenic response in human lung adenocarcinoma cells. Int. J. Radiat. Oncol. Biol. Phys. 2011, 80, 1541–1549. [Google Scholar] [CrossRef]
- Takahashi, Y.; Teshima, T.; Kawaguchi, N.; Hamada, Y.; Mori, S.; Madachi, A.; Ikeda, S.; Mizuno, H.; Ogata, T.; Nojima, K.; et al. Heavy Ion Irradiation Inhibits in Vitro Angiogenesis Even at Sublethal Dose Heavy Ion Irradiation Inhibits in Vitro Angiogenesis Even at Sublethal Dose. Cancer Res. 2003, 63, 4253–4257. [Google Scholar]
- Suetens, A.; Moreels, M.; Quintens, R.; Chiriotti, S.; Tabury, K.; Michaux, A.; Grégoire, V.; Baatout, S. Carbon ion irradiation of the human prostate cancer cell line PC3: A whole genome microarray study. Int. J. Oncol. 2014, 44, 1056–1072. [Google Scholar] [CrossRef] [PubMed]
- Moncharmont, C.; Levy, A.; Guy, J.-B.; Falk, A.T.; Guilbert, M.; Trone, J.-C.; Alphonse, G.; Gilormini, M.; Ardail, D.; Toillon, R.-A.; et al. Radiation-enhanced cell migration/invasion process: A review. Crit. Rev. Oncol. Hematol. 2014, 92, 133–142. [Google Scholar] [CrossRef]
- Widder, J.; Van Der Schaaf, A.; Lambin, P.; Marijnen, C.A.M.; Pignol, J.P.; Rasch, C.R.; Slotman, B.J.; Verheij, M.; Langendijk, J.A. The Quest for Evidence for Proton Therapy: Model-Based Approach and Precision Medicine. Int. J. Radiat. Oncol. Biol. Phys. 2016, 95, 30–36. [Google Scholar] [CrossRef]
- Nowrouzi, A.; Sertorio, M.G.; Akbarpour, M.; Knoll, M.; Krunic, D.; Kuhar, M.; Schwager, C.; Brons, S.; Debus, J.; Wells, S.I.; et al. Personalized assessment of normal tissue radiosensitivity via transcriptome response to photon, proton and carbon irradiation in patient-derived human intestinal organoids. Cancers 2020, 12, 469. [Google Scholar] [CrossRef]
- Luft, S.; Arrizabalaga, O.; Kulish, I.; Nasonova, E.; Durante, M.; Ritter, S.; Schroeder, I.S. Ionizing Radiation Alters Human Embryonic Stem Cell Properties and Differentiation Capacity by Diminishing the Expression of Activin Receptors. Stem Cells Dev. 2017, 26, 341–352. [Google Scholar] [CrossRef] [PubMed]
- Visvader, J.E.; Lindeman, G.J. Cancer stem cells in solid tumours: Accumulating evidence and unresolved questions. Nat. Rev. Cancer 2008, 8, 755–768. [Google Scholar] [CrossRef] [PubMed]
- Clevers, H. The cancer stem cell: Premises, promises and challenges. Nat. Med. 2011, 17, 313–319. [Google Scholar] [CrossRef]
- Clara, J.A.; Monge, C.; Yang, Y.; Takebe, N. Targeting signalling pathways and the immune microenvironment of cancer stem cells—A clinical update. Nat. Rev. Clin. Oncol. 2020, 17, 204–232. [Google Scholar] [CrossRef] [PubMed]
- Baumann, M.; Krause, M.; Hill, R. Exploring the role of cancer stem cells in radioresistance. Nat Rev Cancer 2008, 8, 545–554. [Google Scholar] [CrossRef]
- Rich, J.N. Cancer stem cells in radiation resistance. Cancer Res. 2007, 67, 8980–8984. [Google Scholar] [CrossRef] [PubMed]
- Bao, S.; Wu, Q.; McLendon, R.E.; Hao, Y.; Shi, Q.; Hjelmeland, A.B.; Dewhirst, M.W.; Bigner, D.D.; Rich, J.N. Glioma stem cells promote radioresistance by preferential activation of the DNA damage response. Nature 2006, 444, 756–760. [Google Scholar] [CrossRef] [PubMed]
- Diehn, M.; Cho, R.W.; Lobo, N.A.; Kalisky, T.; Dorie, M.J.; Kulp, A.N.; Qian, D.; Lam, J.S.; Ailles, L.E.; Wong, M.; et al. Association of reactive oxygen species levels and radioresistance in cancer stem cells. Nature 2009, 458, 780–783. [Google Scholar] [CrossRef]
- Najafi, M.; Farhood, B.; Mortezaee, K.; Kharazinejad, E.; Majidpoor, J.; Ahadi, R. Hypoxia in solid tumors: A key promoter of cancer stem cell (CSC) resistance. J. Cancer Res. Clin. Oncol. 2020, 146, 19–31. [Google Scholar] [CrossRef]
- Cui, X.; Oonishi, K.; Tsujii, H.; Yasuda, T.; Matsumoto, Y.; Furusawa, Y.; Akashi, M.; Kamada, T.; Okayasu, R. Effects of carbon ion beam on putative colon cancer stem cells and its comparison with X-rays. Cancer Res. 2011, 71, 3676–3687. [Google Scholar] [CrossRef]
- Oonishi, K.; Cui, X.; Hirakawa, H.; Fujimori, A.; Kamijo, T.; Yamada, S.; Yokosuka, O.; Kamada, T. Different effects of carbon ion beams and X-rays on clonogenic survival and DNA repair in human pancreatic cancer stem-like cells. Radiother. Oncol. 2012, 105, 258–265. [Google Scholar] [CrossRef]
- Sai, S.; Wakai, T.; Vares, G.; Yamada, S.; Kamijo, T.; Kamada, T.; Shirai, T. Combination of carbon ion beam and gemcitabine causes irreparable DNA damage and death of radioresistant pancreatic cancer stem-like cells in vitro and in vivo. Oncotarget 2015, 6, 5517–5535. [Google Scholar] [CrossRef]
- Sai, S.; Vares, G.; Kim, E.H.; Karasawa, K.; Wang, B.; Nenoi, M.; Horimoto, Y.; Hayashi, M. Carbon ion beam combined with cisplatin effectively disrupts triple negative breast cancer stem-like cells in vitro. Mol. Cancer 2015, 14, 166. [Google Scholar] [CrossRef] [PubMed]
- Chiblak, S.; Tang, Z.; Campos, B.; Gal, Z.; Unterberg, A.; Debus, J.; Herold-Mende, C.; Abdollahi, A. Radiosensitivity of Patient-Derived Glioma Stem Cell 3-Dimensional Cultures to Photon, Proton, and Carbon Irradiation. Int. J. Radiat. Oncol. 2016, 95, 112–119. [Google Scholar] [CrossRef]
- Takahashi, M.; Hirakawa, H.; Yajima, H.; Izumi-Nakajima, N.; Okayasu, R.; Fujimori, A. Carbon ion beam is more effective to induce cell death in sphere-type A172 human glioblastoma cells compared with X-rays. Int. J. Radiat. Biol. 2014, 90, 1125–1132. [Google Scholar] [CrossRef]
- Chiblak, S.; Tang, Z.; Lemke, D.; Knoll, M.; Dokic, I.; Warta, R.; Moustafa, M.; Mier, W.; Brons, S.; Rapp, C.; et al. Carbon irradiation overcomes glioma radioresistance by eradicating stem cells and forming an antiangiogenic and immunopermissive niche. JCI Insight 2019, 4. [Google Scholar] [CrossRef]
- Bertrand, G.; Maalouf, M.; Boivin, A.; Battiston-Montagne, P.; Beuve, M.; Levy, A.; Jalade, P.; Fournier, C.; Ardail, D.; Magné, N.; et al. Targeting Head and Neck Cancer Stem Cells to Overcome Resistance to Photon and Carbon Ion Radiation. Stem Cell Rev. Rep. 2014, 10, 114–126. [Google Scholar] [CrossRef]
- Baek, S.-J.; Ishii, H.; Tamari, K.; Hayashi, K.; Nishida, N.; Konno, M.; Kawamoto, K.; Koseki, J.; Fukusumi, T.; Hasegawa, S.; et al. Cancer stem cells: The potential of carbon ion beam radiation and new radiosensitizers (Review). Oncol. Rep. 2015, 34, 2233–2237. [Google Scholar] [CrossRef]
- Meyfour, A.; Pahlavan, S.; Mirzaei, M.; Krijgsveld, J.; Baharvand, H.; Salekdeh, G.H. The quest of cell surface markers for stem cell therapy. Cell. Mol. Life Sci. 2020. [Google Scholar] [CrossRef] [PubMed]
- Gregoire, V.; Baumann, M. Combined Radiotherapy and Chemotherapy. In Basic Clinical Radiobiology, 5th ed.; Joiner, M.C., van der Kogel, A.J., Eds.; CRC Press/Taylor & Francis Group: Boca Raton, FL, USA, 2018; pp. 246–258. ISBN 9780429490606. [Google Scholar]
- Pignon, J.; Bourhis, J.; Domenge, C.; Designé, L. Chemotherapy added to locoregional treatment for head and neck squamous-cell carcinoma: Three meta-analyses of updated individual data. Lancet 2000, 355, 949–955. [Google Scholar] [CrossRef]
- Jones, B.; Dale, R.G. The potential for mathematical modelling in the assessment of the radiation dose equivalent of cytotoxic chemotherapy given concomitantly with radiotherapy. Br. J. Radiol. 2005, 78, 939–943. [Google Scholar] [CrossRef] [PubMed]
- Durante, M.; Tommasino, F.; Yamada, S. Modeling Combined Chemotherapy and Particle Therapy for Locally Advanced Pancreatic Cancer. Front. Oncol. 2015, 5. [Google Scholar] [CrossRef] [PubMed]
- Schlaich, F.; Brons, S.; Haberer, T.; Debus, J.; Combs, S.E.; Weber, K.-J. Comparison of the effects of photon versus carbon ion irradiation when combined with chemotherapy in vitro. Radiat. Oncol. 2013, 8, 260. [Google Scholar] [CrossRef]
- Harrabi, S.B.; Adeberg, S.; Winter, M.; Haberer, T.; Debus, J.; Weber, K.-J. S-phase–specific radiosensitization by gemcitabine for therapeutic carbon ion exposure in vitro. J. Radiat. Res. 2016, 57, 110–114. [Google Scholar] [CrossRef]
- El Shafie, R.A.; Habermehl, D.; Rieken, S.; Mairani, A.; Orschiedt, L.; Brons, S.; Haberer, T.; Weber, K.-J.; Debus, J.; Combs, S.E. In vitro evaluation of photon and raster-scanned carbon ion radiotherapy in combination with gemcitabine in pancreatic cancer cell lines. J. Radiat. Res. 2013, 54, i113–i119. [Google Scholar] [CrossRef]
- Harrabi, S.; Combs, S.E.; Brons, S.; Haberer, T.; Debus, J.; Weber, K.-J. Temozolomide in combination with carbon ion or photon irradiation in glioblastoma multiforme cell lines – does scheduling matter? Int. J. Radiat. Biol. 2013, 89, 692–697. [Google Scholar] [CrossRef]
- Combs, S.E.; Zipp, L.; Rieken, S.; Habermehl, D.; Brons, S.; Winter, M.; Haberer, T.; Debus, J.; Weber, K.-J. In vitro evaluation of photon and carbon ion radiotherapy in combination with chemotherapy in glioblastoma cells. Radiat. Oncol. 2012, 7, 9. [Google Scholar] [CrossRef]
- Dehne, S.; Fritz, C.; Rieken, S.; Baris, D.; Brons, S.; Haberer, T.; Debus, J.; Weber, K.-J.; Schmid, T.E.; Combs, S.E.; et al. Combination of Photon and Carbon Ion Irradiation with Targeted Therapy Substances Temsirolimus and Gemcitabine in Hepatocellular Carcinoma Cell Lines. Front. Oncol. 2017, 7. [Google Scholar] [CrossRef] [PubMed]
- Falzone, L.; Salomone, S.; Libra, M. Evolution of Cancer Pharmacological Treatments at the Turn of the Third Millennium. Front. Pharmacol. 2018, 9. [Google Scholar] [CrossRef] [PubMed]
- Chae, Y.K.; Pan, A.P.; Davis, A.A.; Patel, S.P.; Carneiro, B.A.; Kurzrock, R.; Giles, F.J. Path toward Precision Oncology: Review of Targeted Therapy Studies and Tools to Aid in Defining “Actionability” of a Molecular Lesion and Patient Management Support. Mol. Cancer Ther. 2017, 16, 2645–2655. [Google Scholar] [CrossRef] [PubMed]
- Gatzka, M. Targeted Tumor Therapy Remixed—An Update on the Use of Small-Molecule Drugs in Combination Therapies. Cancers 2018, 10, 155. [Google Scholar] [CrossRef]
- Konings, K.; Vandevoorde, C.; Baselet, B.; Baatout, S.; Moreels, M. Combination Therapy With Charged Particles and Molecular Targeting: A Promising Avenue to Overcome Radioresistance. Front. Oncol. 2020, 10. [Google Scholar] [CrossRef]
- Moncharmont, C.; Guy, J.-B.; Wozny, A.-S.; Gilormini, M.; Battiston-Montagne, P.; Ardail, D.; Beuve, M.; Alphonse, G.; Simoëns, X.; Rancoule, C.; et al. Carbon ion irradiation withstands cancer stem cells’ migration/invasion process in Head and Neck Squamous Cell Carcinoma (HNSCC). Oncotarget 2016, 7, 47738–47749. [Google Scholar] [CrossRef]
- Vares, G.; Ahire, V.; Sunada, S.; Ho Kim, E.; Sai, S.; Chevalier, F.; Romeo, P.-H.; Yamamoto, T.; Nakajima, T.; Saintigny, Y. A multimodal treatment of carbon ions irradiation, miRNA-34 and mTOR inhibitor specifically control high-grade chondrosarcoma cancer stem cells. Radiother. Oncol. 2020, 150, 253–261. [Google Scholar] [CrossRef]
- Hirai, T.; Shirai, H.; Fujimori, H.; Okayasu, R.; Sasai, K.; Masutani, M. Radiosensitization effect of poly(ADP-ribose) polymerase inhibition in cells exposed to low and high liner energy transfer radiation. Cancer Sci. 2012, 103, 1045–1050. [Google Scholar] [CrossRef]
- Ghorai, A.; Bhattacharyya, N.P.; Sarma, A.; Ghosh, U. Radiosensitivity and Induction of Apoptosis by High LET Carbon Ion Beam and Low LET Gamma Radiation: A Comparative Study. Scientifica (Cairo) 2014, 2014, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Lesueur, P.; Chevalier, F.; El-Habr, E.A.; Junier, M.-P.; Chneiweiss, H.; Castera, L.; Müller, E.; Stefan, D.; Saintigny, Y. Radiosensitization Effect of Talazoparib, a Parp Inhibitor, on Glioblastoma Stem Cells Exposed to Low and High Linear Energy Transfer Radiation. Sci. Rep. 2018, 8, 3664. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Li, P.; Hirayama, R.; Niu, Y.; Liu, X.; Chen, W.; Jin, X.; Zhang, P.; Ye, F.; Zhao, T.; et al. Genistein sensitizes glioblastoma cells to carbon ions via inhibiting DNA-PKcs phosphorylation and subsequently repressing NHEJ and delaying HR repair pathways. Radiother. Oncol. 2018, 129, 84–94. [Google Scholar] [CrossRef] [PubMed]
- Yang, L.; Liu, Y.; Sun, C.; Yang, X.; Yang, Z.; Ran, J.; Zhang, Q.; Zhang, H.; Wang, X.; Wang, X. Inhibition of DNA-PKcs enhances radiosensitivity and increases the levels of ATM and ATR in NSCLC cells exposed to carbon ion irradiation. Oncol. Lett. 2015, 10, 2856–2864. [Google Scholar] [CrossRef]
- Zhou, X.; Zhang, X.; Xie, Y.; Tanaka, K.; Wang, B.; Zhang, H. DNA-PKcs Inhibition Sensitizes Cancer Cells to Carbon-Ion Irradiation via Telomere Capping Disruption. PLoS ONE 2013, 8, e72641. [Google Scholar] [CrossRef]
- Lee, Y.; Sunada, S.; Hirakawa, H.; Fujimori, A.; Nickoloff, J.A.; Okayasu, R. TAS-116, a Novel Hsp90 Inhibitor, Selectively Enhances Radiosensitivity of Human Cancer Cells to X-rays and Carbon Ion Radiation. Mol. Cancer Ther. 2017, 16, 16–24. [Google Scholar] [CrossRef]
- Lee, Y.; Li, H.K.; Masaoka, A.; Sunada, S.; Hirakawa, H.; Fujimori, A.; Nickoloff, J.A.; Okayasu, R. The purine scaffold Hsp90 inhibitor PU-H71 sensitizes cancer cells to heavy ion radiation by inhibiting DNA repair by homologous recombination and non-homologous end joining. Radiother. Oncol. 2016, 121, 162–168. [Google Scholar] [CrossRef]
- Konings, K.; Vandevoorde, C.; Belmans, N.; Vermeesen, R.; Baselet, B.; Van Walleghem, M.; Janssen, A.; Isebaert, S.; Baatout, S.; Haustermans, K.; et al. The Combination of Particle Irradiation With the Hedgehog Inhibitor GANT61 Differently Modulates the Radiosensitivity and Migration of Cancer Cells Compared to X-Ray Irradiation. Front. Oncol. 2019, 9. [Google Scholar] [CrossRef]
- Konings, K.; Belmans, N.; Vermeesen, R.; Baselet, B.; Lamers, G.; Janssen, A.; Isebaert, S.; Baatout, S.; Haustermans, K.; Moreels, M. Targeting the Hedgehog pathway in combination with X-ray or carbon ion radiation decreases migration of MCF-7 breast cancer cells. Int. J. Oncol. 2019. [Google Scholar] [CrossRef]
- Jain, S.; Hirst, D.G.; O’Sullivan, J.M. Gold nanoparticles as novel agents for cancer therapy. Br. J. Radiol. 2012, 85, 101–113. [Google Scholar] [CrossRef]
- Norouzi, M. Gold Nanoparticles in Glioma Theranostics. Pharmacol. Res. 2020, 156, 104753. [Google Scholar] [CrossRef]
- Penninckx, S.; Heuskin, A.-C.; Michiels, C.; Lucas, S. Gold Nanoparticles as a Potent Radiosensitizer: A Transdisciplinary Approach from Physics to Patient. Cancers 2020, 12, 2021. [Google Scholar] [CrossRef] [PubMed]
- Howard, D.; Sebastian, S.; Le, Q.V.-C.; Thierry, B.; Kempson, I. Chemical Mechanisms of Nanoparticle Radiosensitization and Radioprotection: A Review of Structure-Function Relationships Influencing Reactive Oxygen Species. Int. J. Mol. Sci. 2020, 21, 579. [Google Scholar] [CrossRef]
- Jeremic, B.; Aguerri, A.R.; Filipovic, N. Radiosensitization by gold nanoparticles. Clin. Transl. Oncol. 2013, 15, 593–601. [Google Scholar] [CrossRef] [PubMed]
- Schuemann, J.; Berbeco, R.; Chithrani, D.B.; Cho, S.H.; Kumar, R.; McMahon, S.J.; Sridhar, S.; Krishnan, S. Roadmap to Clinical Use of Gold Nanoparticles for Radiation Sensitization. Int. J. Radiat. Oncol. 2016, 94, 189–205. [Google Scholar] [CrossRef]
- Schuemann, J.; Bagley, A.; Berbeco, R.; Bromma, K.; Butterworth, K.T.; Byrne, H.; Chithrani, D.B.; Cho, S.H.; Cook, J.R.; Favaudon, V.; et al. Roadmap for metal nanoparticles in radiation therapy: Current status, translational challenges, and future directions. Phys. Med. Biol. 2020. [Google Scholar] [CrossRef]
- Peukert, D.; Kempson, I.; Douglass, M.; Bezak, E. Metallic nanoparticle radiosensitisation of ion radiotherapy: A review. Phys. Med. 2018, 47, 121–128. [Google Scholar] [CrossRef] [PubMed]
- Lacombe, S.; Porcel, E.; Scifoni, E. Particle therapy and nanomedicine: State of art and research perspectives. Cancer Nanotechnol. 2017, 8, 9. [Google Scholar] [CrossRef]
- Fuss, M.C.; Boscolo, D.; Durante, M.; Scifoni, E.; Krämer, M. Systematic quantification of nanoscopic dose enhancement of gold nanoparticles in ion beams. Phys. Med. Biol. 2020, 65, 075008. [Google Scholar] [CrossRef] [PubMed]
- Rudek, B.; McNamara, A.; Ramos-Méndez, J.; Byrne, H.; Kuncic, Z.; Schuemann, J. Radio-enhancement by gold nanoparticles and their impact on water radiolysis for x-ray, proton and carbon-ion beams. Phys. Med. Biol. 2019, 64, 175005. [Google Scholar] [CrossRef]
- Kirkwood, J.M.; Butterfield, L.H.; Tarhini, A.A.; Zarour, H.; Kalinski, P.; Ferrone, S. Immunotherapy of cancer in 2012. CA Cancer J Clin 2012, 62, 309–335. [Google Scholar] [CrossRef]
- Robert, C.; Schachter, J.; Long, G.V.; Arance, A.; Grob, J.J.; Mortier, L.; Daud, A.; Carlino, M.S.; McNeil, C.; Lotem, M.; et al. Pembrolizumab versus Ipilimumab in Advanced Melanoma. N. Engl. J. Med. 2015, 372, 2521–2532. [Google Scholar] [CrossRef]
- Postow, M.A.; Sidlow, R.; Hellmann, M.D. Immune-Related Adverse Events Associated with Immune Checkpoint Blockade. N. Engl. J. Med. 2018, 378, 158–168. [Google Scholar] [CrossRef]
- Whiteside, T.L.; Demaria, S.; Rodriguez-Ruiz, M.E.; Zarour, H.M.; Melero, I. Emerging Opportunities and Challenges in Cancer Immunotherapy. Clin. Cancer Res. 2016, 22, 1845–1855. [Google Scholar] [CrossRef]
- Formenti, S.C.; Demaria, S. Systemic effects of local radiotherapy. Lancet Oncol. 2009, 10, 718–726. [Google Scholar] [CrossRef]
- Ngwa, W.; Irabor, O.C.; Schoenfeld, J.D.; Hesser, J.; Demaria, S.; Formenti, S.C. Using immunotherapy to boost the abscopal effect. Nat. Rev. Cancer 2018, 18, 313–322. [Google Scholar] [CrossRef] [PubMed]
- Demaria, S.; Golden, E.B.; Formenti, S.C. Role of Local Radiation Therapy in Cancer Immunotherapy. JAMA Oncol. 2015, 1, 1325–1332. [Google Scholar] [CrossRef] [PubMed]
- Formenti, S.C.; Demaria, S. Combining radiotherapy and cancer immunotherapy: A paradigm shift. J. Natl. Cancer Inst. 2013, 105, 256–265. [Google Scholar] [CrossRef]
- Weichselbaum, R.R.; Liang, H.; Deng, L.; Fu, Y.-X. Radiotherapy and immunotherapy: A beneficial liaison? Nat. Rev. Clin. Oncol. 2017, 14, 365–379. [Google Scholar] [CrossRef] [PubMed]
- Durante, M.; Reppingen, N.; Held, K.D. Immunologically augmented cancer treatment using modern radiotherapy. Trends Mol. Med. 2013, 19, 565–582. [Google Scholar] [CrossRef]
- Van Limbergen, E.J.; De Ruysscher, D.K.; Olivo Pimentel, V.; Marcus, D.; Berbee, M.; Hoeben, A.; Rekers, N.; Theys, J.; Yaromina, A.; Dubois, L.J.; et al. Combining radiotherapy with immunotherapy: The past, the present and the future. Br. J. Radiol. 2017, 90, 20170157. [Google Scholar] [CrossRef]
- Herrera, F.G.; Bourhis, J.; Coukos, G. Radiotherapy combination opportunities leveraging immunity for the next oncology practice. CA Cancer J. Clin. 2017, 67, 65–85. [Google Scholar] [CrossRef] [PubMed]
- Keam, S.; Gill, S.; Ebert, M.A.; Nowak, A.K.; Cook, A.M. Enhancing the efficacy of immunotherapy using radiotherapy. Clin. Transl. Immunol. 2020, 9. [Google Scholar] [CrossRef]
- Zhao, X.; Shao, C. Radiotherapy-Mediated Immunomodulation and Anti-Tumor Abscopal Effect Combining Immune Checkpoint Blockade. Cancers 2020, 12, 2762. [Google Scholar] [CrossRef] [PubMed]
- Lubas, M.J.; Kumar, S.S. The Combined Use of SBRT and Immunotherapy—a Literature Review. Curr. Oncol. Rep. 2020, 22, 117. [Google Scholar] [CrossRef] [PubMed]
- Victor, C.T.-S.; Rech, A.J.; Maity, A.; Rengan, R.; Pauken, K.E.; Stelekati, E.; Benci, J.L.; Xu, B.; Dada, H.; Odorizzi, P.M.; et al. Radiation and dual checkpoint blockade activate non-redundant immune mechanisms in cancer. Nature 2015, 520, 373–377. [Google Scholar] [CrossRef] [PubMed]
- Postow, M.A.; Callahan, M.K.; Barker, C.A.; Yamada, Y.; Yuan, J.; Kitano, S.; Mu, Z.; Rasalan, T.; Adamow, M.; Ritter, E.; et al. Immunologic correlates of the abscopal effect in a patient with melanoma. N. Engl. J. Med. 2012, 366, 925–931. [Google Scholar] [CrossRef] [PubMed]
- Hiniker, S.M.; Chen, D.S.; Knox, S.J. Abscopal Effect in a Patient with Melanoma. N. Engl. J. Med. 2012, 366, 2035–2036. [Google Scholar] [CrossRef] [PubMed]
- Golden, E.B.; Chhabra, A.; Chachoua, A.; Adams, S.; Donach, M.; Fenton-Kerimian, M.; Friedman, K.; Ponzo, F.; Babb, J.S.; Goldberg, J.; et al. Local radiotherapy and granulocyte-macrophage colony-stimulating factor to generate abscopal responses in patients with metastatic solid tumours: A proof-of-principle trial. Lancet Oncol. 2015, 16, 795–803. [Google Scholar] [CrossRef]
- Ko, E.C.; Raben, D.; Formenti, S.C. The Integration of Radiotherapy with Immunotherapy for the Treatment of Non–Small Cell Lung Cancer. Clin. Cancer Res. 2018, 24, 5792–5806. [Google Scholar] [CrossRef]
- Antonia, S.J.; Villegas, A.; Daniel, D.; Vicente, D.; Murakami, S.; Hui, R.; Yokoi, T.; Chiappori, A.; Lee, K.H.; de Wit, M.; et al. Durvalumab after Chemoradiotherapy in Stage III Non–Small-Cell Lung Cancer. N. Engl. J. Med. 2017, 377, 1919–1929. [Google Scholar] [CrossRef]
- Antonia, S.J.; Villegas, A.; Daniel, D.; Vicente, D.; Murakami, S.; Hui, R.; Kurata, T.; Chiappori, A.; Lee, K.H.; de Wit, M.; et al. Overall Survival with Durvalumab after Chemoradiotherapy in Stage III NSCLC. N. Engl. J. Med. 2018, 379, 2342–2350. [Google Scholar] [CrossRef]
- Formenti, S.C.; Rudqvist, N.-P.; Golden, E.; Cooper, B.; Wennerberg, E.; Lhuillier, C.; Vanpouille-Box, C.; Friedman, K.; Ferrari de Andrade, L.; Wucherpfennig, K.W.; et al. Radiotherapy induces responses of lung cancer to CTLA-4 blockade. Nat. Med. 2018, 24, 1845–1851. [Google Scholar] [CrossRef] [PubMed]
- Formenti, S.C. Optimizing Dose Per Fraction: A New Chapter in the Story of the Abscopal Effect? Int. J. Radiat. Oncol. Biol. Phys. 2017, 99, 677–679. [Google Scholar] [CrossRef]
- Brooks, E.D.; Chang, J.Y. Time to abandon single-site irradiation for inducing abscopal effects. Nat. Rev. Clin. Oncol. 2019, 16, 123–135. [Google Scholar] [CrossRef] [PubMed]
- Durante, M.; Brenner, D.J.; Formenti, S.C. Does heavy ion therapy work through the immune system? Int. J. Radiat. Oncol. Biol. Phys. 2016, 96, 934–936. [Google Scholar] [CrossRef] [PubMed]
- Durante, M.; Formenti, S. Harnessing radiation to improve immunotherapy: Better with particles? Br. J. Radiol. 2020, 93, 20190224. [Google Scholar] [CrossRef]
- Davuluri, R.; Jiang, W.; Fang, P.; Xu, C.; Komaki, R.; Gomez, D.R.; Welsh, J.; Cox, J.D.; Crane, C.H.; Hsu, C.C.; et al. Lymphocyte Nadir and Esophageal Cancer Survival Outcomes After Chemoradiation Therapy. Int. J. Radiat. Oncol. 2017, 99, 128–135. [Google Scholar] [CrossRef]
- Venkatesulu, B.P.; Mallick, S.; Lin, S.H.; Krishnan, S. A systematic review of the influence of radiation-induced lymphopenia on survival outcomes in solid tumors. Crit. Rev. Oncol. Hematol. 2018, 123, 42–51. [Google Scholar] [CrossRef] [PubMed]
- Chen, D.; Patel, R.R.; Verma, V.; Ramapriyan, R.; Barsoumian, H.B.; Cortez, M.A.; Welsh, J.W. Interaction between lymphopenia, radiotherapy technique, dosimetry, and survival outcomes in lung cancer patients receiving combined immunotherapy and radiotherapy. Radiother. Oncol. 2020, 150, 114–120. [Google Scholar] [CrossRef]
- Bakhoum, S.F.; Ngo, B.; Laughney, A.M.; Cavallo, J.-A.; Murphy, C.J.; Ly, P.; Shah, P.; Sriram, R.K.; Watkins, T.B.K.; Taunk, N.K.; et al. Chromosomal instability drives metastasis through a cytosolic DNA response. Nature 2018, 553, 467–472. [Google Scholar] [CrossRef]
- Harding, S.M.; Benci, J.L.; Irianto, J.; Discher, D.E.; Minn, A.J.; Greenberg, R.A. Mitotic progression following DNA damage enables pattern recognition within micronuclei. Nature 2017, 548, 466–470. [Google Scholar] [CrossRef] [PubMed]
- Galluzzi, L.; Vanpouille-Box, C.; Bakhoum, S.F.; Demaria, S. SnapShot: CGAS-STING Signaling. Cell 2018, 173, 276–276.e1. [Google Scholar] [CrossRef]
- Durante, M.; Formenti, S.C. Radiation-Induced Chromosomal Aberrations and Immunotherapy: Micronuclei, Cytosolic DNA, and Interferon-Production Pathway. Front. Oncol. 2018, 8. [Google Scholar] [CrossRef] [PubMed]
- Sato, H.; Niimi, A.; Yasuhara, T.; Permata, T.B.M.; Hagiwara, Y.; Isono, M.; Nuryadi, E.; Sekine, R.; Oike, T.; Kakoti, S.; et al. DNA double-strand break repair pathway regulates PD-L1 expression in cancer cells. Nat. Commun. 2017, 8. [Google Scholar] [CrossRef]
- Wang, W.; Chapman, N.M.; Zhang, B.; Li, M.; Fan, M.; Laribee, R.N.; Zaidi, M.R.; Pfeffer, L.M.; Chi, H.; Wu, Z.-H. Upregulation of PD-L1 via HMGB1-Activated IRF3 and NF-κB Contributes to UV Radiation-Induced Immune Suppression. Cancer Res. 2019. [Google Scholar] [CrossRef]
- Iijima, M.; Okonogi, N.; Nakajima, N.I.; Morokoshi, Y.; Kanda, H.; Yamada, T.; Kobayashi, Y.; Banno, K.; Wakatsuki, M.; Yamada, S.; et al. Significance of PD-L1 expression in carbon-ion radiotherapy for uterine cervical adeno/adenosquamous carcinoma. J. Gynecol. Oncol. 2020, 31. [Google Scholar] [CrossRef] [PubMed]
- Shevtsov, M.; Sato, H.; Multhoff, G.; Shibata, A. Novel Approaches to Improve the Efficacy of Immuno-Radiotherapy. Front. Oncol. 2019, 9. [Google Scholar] [CrossRef]
- Ebner, D.K.; Tinganelli, W.; Helm, A.; Bisio, A.; Simoniello, P.; Natale, F.; Yamada, S.; Kamada, T.; Shimokawa, T.; Durante, M. Generating and grading the abscopal effect: Proposal for comprehensive evaluation of combination immunoradiotherapy in mouse models. Transl. Cancer Res. 2017, 6, S892–S899. [Google Scholar] [CrossRef]
- Ando, K.; Fujita, H.; Hosoi, A.; Ma, L.; Wakatsuki, M.; Seino, K.; Kakimi, K.; Imai, T.; Shimokawa, T.; Nakano, T. Intravenous dendritic cell administration enhances suppression of lung metastasis induced by carbon-ion irradiation. J. Radiat. Res. 2017, 58, 446–455. [Google Scholar] [CrossRef]
- Ohkubo, Y.; Iwakawa, M.; Seino, K.-I.; Nakawatari, M.; Wada, H.; Kamijuku, H.; Nakamura, E.; Nakano, T.; Imai, T. Combining Carbon Ion Radiotherapy and Local Injection of α-Galactosylceramide–Pulsed Dendritic Cells Inhibits Lung Metastases in an In Vivo Murine Model. Int. J. Radiat. Oncol. 2010, 78, 1524–1531. [Google Scholar] [CrossRef]
- Shimokawa, T.; Ma, L.; Ando, K.; Sato, K.; Imai, T. The Future of Combining Carbon-Ion Radiotherapy with Immunotherapy: Evidence and Progress in Mouse Models. Int. J. Part. Ther. 2016, 3, 61–70. [Google Scholar] [CrossRef]
- Takahashi, Y.; Yasui, T.; Minami, K.; Tamari, K.; Hayashi, K.; Otani, K.; Seo, Y.; Isohashi, F.; Koizumi, M.; Ogawa, K. Carbon ion irradiation enhances the antitumor efficacy of dual immune checkpoint blockade therapy both for local and distant sites in murine osteosarcoma. Oncotarget 2019, 10, 633–646. [Google Scholar] [CrossRef] [PubMed]
- Helm, A.; Tinganelli, W.; Simoniello, P.; Kurosawa, F.; Fournier, C.; Shimokawa, T.; Durante, M. Reduction of lung metastases in a mouse osteosarcoma model treated with carbon ions and immune checkpoint inhibitors. Int. J. Radiat. Oncol. 2020. [Google Scholar] [CrossRef] [PubMed]
- Durante, M.; Cucinotta, F.A. Heavy ion carcinogenesis and human space exploration. Nat. Rev. Cancer 2008, 8, 465–472. [Google Scholar] [CrossRef]
- Chancellor, J.; Scott, G.; Sutton, J. Space Radiation: The Number One Risk to Astronaut Health beyond Low Earth Orbit. Life 2014, 4, 491–510. [Google Scholar] [CrossRef] [PubMed]
- Newhauser, W.D.; Durante, M. Assessing the risk of second malignancies after modern radiotherapy. Nat. Rev. Cancer 2011, 11, 438–448. [Google Scholar] [CrossRef]
- Gondi, V.; Yock, T.I.; Mehta, M.P. Proton therapy for paediatric CNS tumours—Improving treatment-related outcomes. Nat. Rev. Neurol. 2016, 12, 334–345. [Google Scholar] [CrossRef]
- Rieber, J.G.; Kessel, K.A.; Witt, O.; Behnisch, W.; Kulozik, A.E.; Debus, J.; Combs, S.E. Treatment tolerance of particle therapy in pediatric patients. Acta Oncol. 2015, 54, 1049–1055. [Google Scholar] [CrossRef]
- Mohamad, O.; Tabuchi, T.; Nitta, Y.; Nomoto, A.; Sato, A.; Kasuya, G.; Makishima, H.; Choy, H.; Yamada, S.; Morishima, T.; et al. Risk of subsequent primary cancers after carbon ion radiotherapy, photon radiotherapy, or surgery for localised prostate cancer: A propensity score-weighted, retrospective, cohort study. Lancet Oncol. 2019, 20, 674–685. [Google Scholar] [CrossRef]
- maoka, T.; Nishimura, M.; Daino, K.; Takabatake, M.; Moriyama, H.; Nishimura, Y.; Morioka, T.; Shimada, Y.; Kakinuma, S. Risk of second cancer after ion beam radiotherapy: Insights from animal carcinogenesis studies. Int. J. Radiat. Biol. 2019, 95, 1431–1440. [Google Scholar] [CrossRef]
- Goodhead, D.T. Neutrons are forever! Historical perspectives. Int. J. Radiat. Biol. 2019, 95, 957–984. [Google Scholar] [CrossRef]
- Suman, S.; Kumar, S.; Moon, B.-H.; Strawn, S.J.; Thakor, H.; Fan, Z.; Shay, J.W.; Fornace, A.J.; Datta, K. Relative Biological Effectiveness of Energetic Heavy Ions for Intestinal Tumorigenesis Shows Male Preponderance and Radiation Type and Energy Dependence in APC1638N/+ Mice. Int. J. Radiat. Oncol. 2016, 95, 131–138. [Google Scholar] [CrossRef] [PubMed]
- Brenner, D.J.; Hall, E.J. Secondary neutrons in clinical proton radiotherapy: A charged issue. Radiother. Oncol. 2008, 86, 165–170. [Google Scholar] [CrossRef] [PubMed]
- Münter, M.W.; Wengenroth, M.; Fehrenbacher, G.; Schardt, D.; Nikoghosyan, A.; Durante, M.; Debus, J. Heavy ion radiotherapy during pregnancy. Fertil. Steril. 2010, 94, 2329.e5–2329.e7. [Google Scholar] [CrossRef]
- Stokkevåg, C.H.; Fukahori, M.; Nomiya, T.; Matsufuji, N.; Engeseth, G.M.; Hysing, L.B.; Ytre-Hauge, K.S.; Rørvik, E.; Szostak, A.; Muren, L.P. Modelling of organ-specific radiation-induced secondary cancer risks following particle therapy. Radiother. Oncol. 2016, 120, 300–306. [Google Scholar] [CrossRef]
- Eley, J.G.; Friedrich, T.; Homann, K.L.; Howell, R.M.; Scholz, M.; Durante, M.; Newhauser, W.D. Comparative Risk Predictions of Second Cancers after Carbon-Ion Therapy Versus Proton Therapy. Int. J. Radiat. Oncol. Biol. Phys. 2016, 95, 279–286. [Google Scholar] [CrossRef] [PubMed]
- Favaudon, V.; Caplier, L.; Monceau, V.; Pouzoulet, F.; Sayarath, M.; Fouillade, C.; Poupon, M.-F.; Brito, I.; Hupe, P.; Bourhis, J.; et al. Ultrahigh dose-rate FLASH irradiation increases the differential response between normal and tumor tissue in mice. Sci. Transl. Med. 2014, 6, 245ra93. [Google Scholar] [CrossRef] [PubMed]
- Bourhis, J.; Sozzi, W.J.; Jorge, P.G.; Gaide, O.; Bailat, C.; Duclos, F.; Patin, D.; Ozsahin, M.; Bochud, F.; Germond, J.-F.; et al. Treatment of a first patient with FLASH-radiotherapy. Radiother. Oncol. 2019, 139, 18–22. [Google Scholar] [CrossRef]
- Harrington, K.J. Ultrahigh Dose-rate Radiotherapy: Next Steps for FLASH-RT. Clin. Cancer Res. 2019, 25, 3–5. [Google Scholar] [CrossRef]
- Schültke, E.; Balosso, J.; Breslin, T.; Cavaletti, G.; Djonov, V.; Esteve, F.; Grotzer, M.; Hildebrandt, G.; Valdman, A.; Laissue, J. Microbeam radiation therapy—Grid therapy and beyond: A clinical perspective. Br. J. Radiol. 2017, 90, 20170073. [Google Scholar] [CrossRef] [PubMed]
- Dilmanian, F.A.; Zhong, Z.; Bacarian, T.; Benveniste, H.; Romanelli, P.; Wang, R.; Welwart, J.; Yuasa, T.; Rosen, E.M.; Anschel, D.J. Interlaced x-ray microplanar beams: A radiosurgery approach with clinical potential. Proc. Natl. Acad. Sci. USA 2006, 103, 9709–9714. [Google Scholar] [CrossRef] [PubMed]
- Prezado, Y.; Jouvion, G.; Patriarca, A.; Nauraye, C.; Guardiola, C.; Juchaux, M.; Lamirault, C.; Labiod, D.; Jourdain, L.; Sebrie, C.; et al. Proton minibeam radiation therapy widens the therapeutic index for high-grade gliomas. Sci. Rep. 2018, 8, 16479. [Google Scholar] [CrossRef] [PubMed]
- Durante, M.; Golubev, A.; Park, W.-Y.; Trautmann, C. Applied nuclear physics at the new high-energy particle accelerator facilities. Phys. Rep. 2019, 800, 1–37. [Google Scholar] [CrossRef]
- Dilmanian, F.A.; Rusek, A.; Fois, G.R.; Olschowka, J.; Desnoyers, N.R.; Park, J.Y.; Dioszegi, I.; Dane, B.; Wang, R.; Tomasi, D.; et al. Interleaved carbon minibeams: An experimental radiosurgery method with clinical potential. Int. J. Radiat. Oncol. Biol. Phys. 2012, 84, 514–519. [Google Scholar] [CrossRef]
- Zakaria, A.M.; Colangelo, N.W.; Meesungnoen, J.; Azzam, E.I.; Plourde, M.-É.; Jay-Gerin, J.-P. Ultra-High Dose-Rate, Pulsed (FLASH) Radiotherapy with Carbon Ions: Generation of Early, Transient, Highly Oxygenated Conditions in the Tumor Environment. Radiat. Res. 2020. [Google Scholar] [CrossRef]
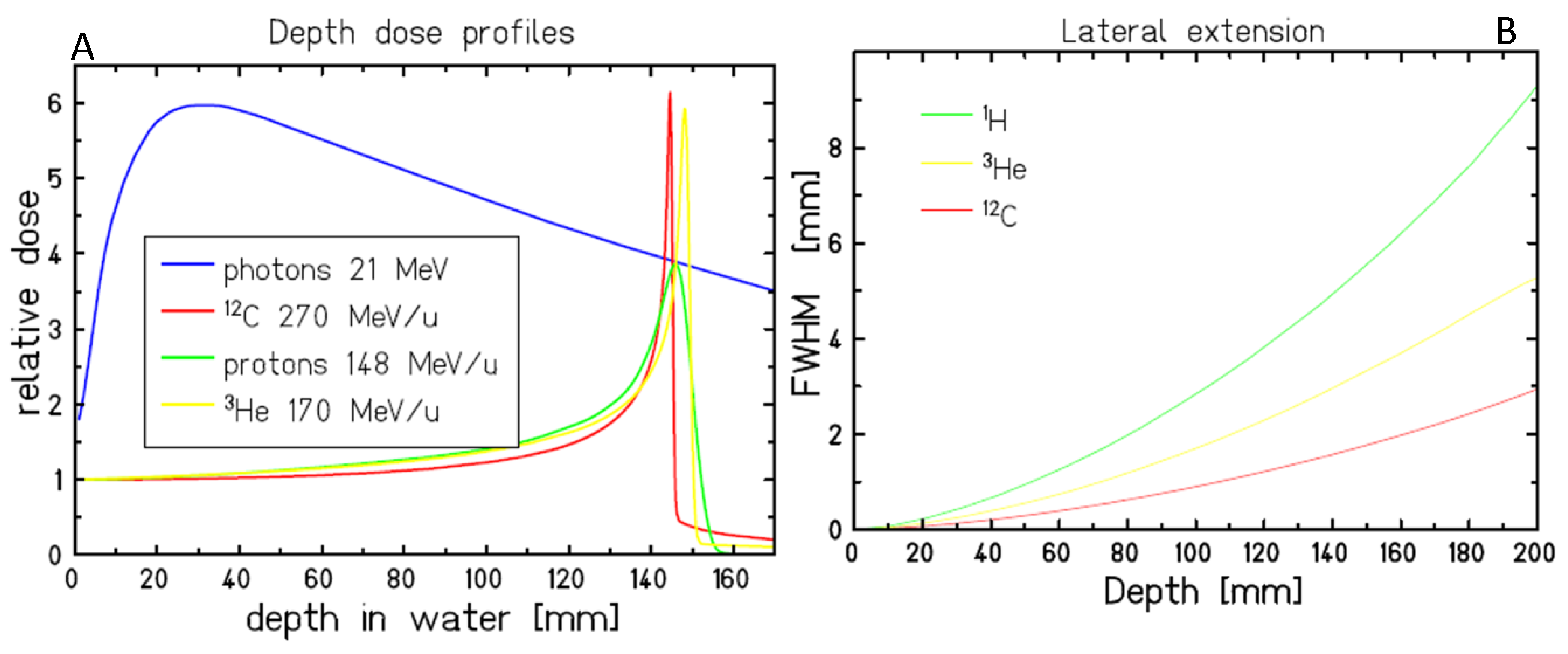
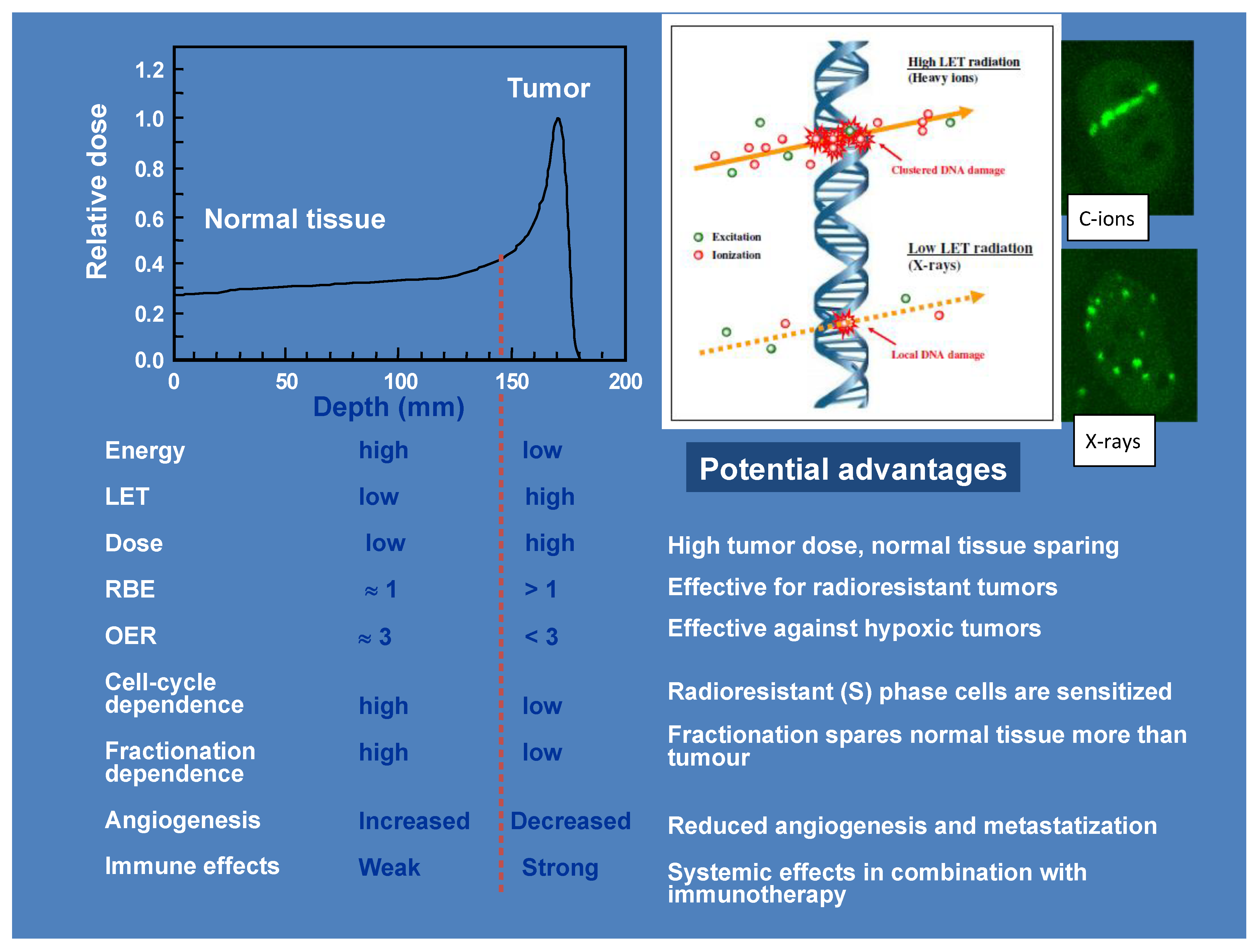
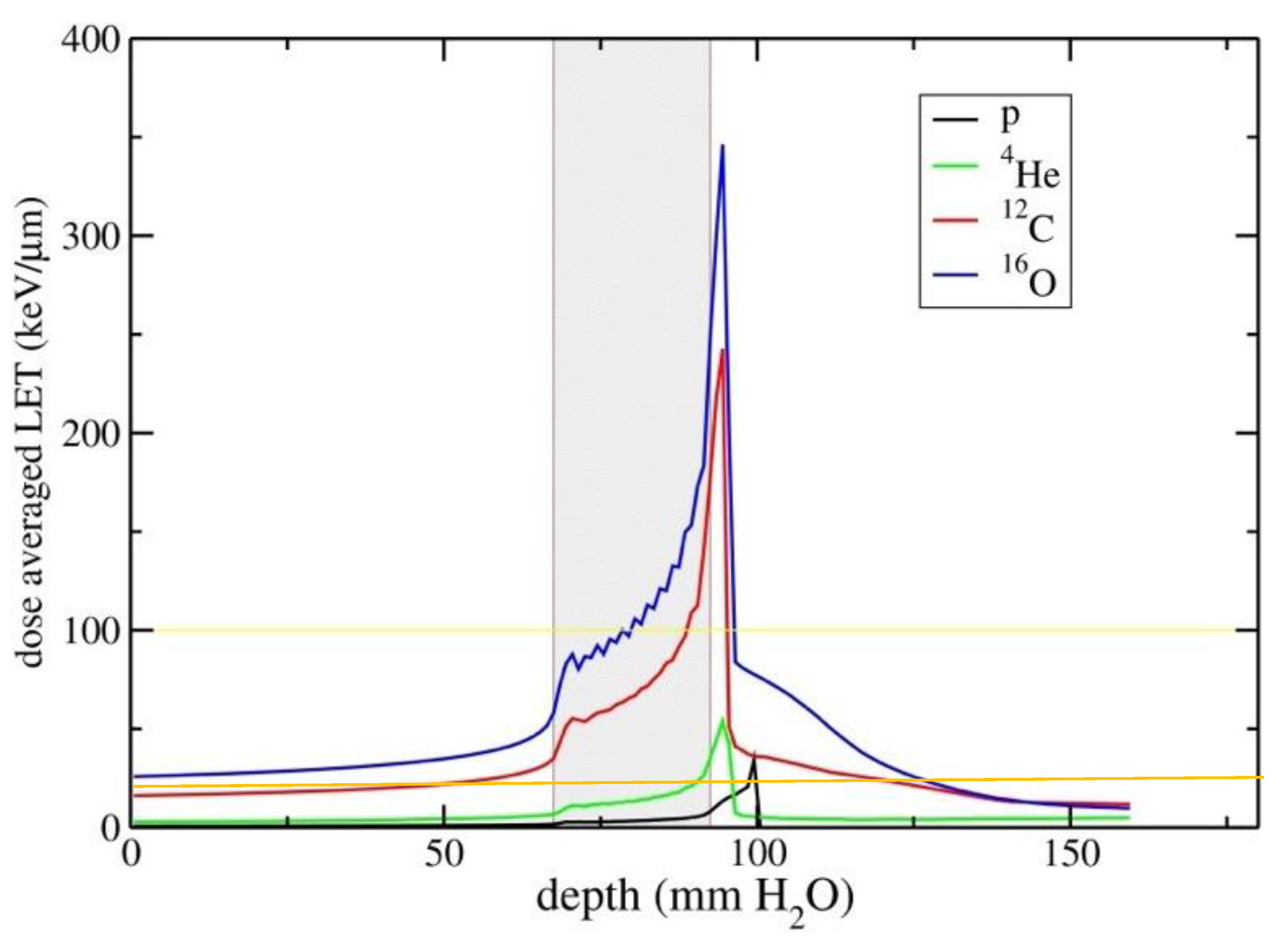
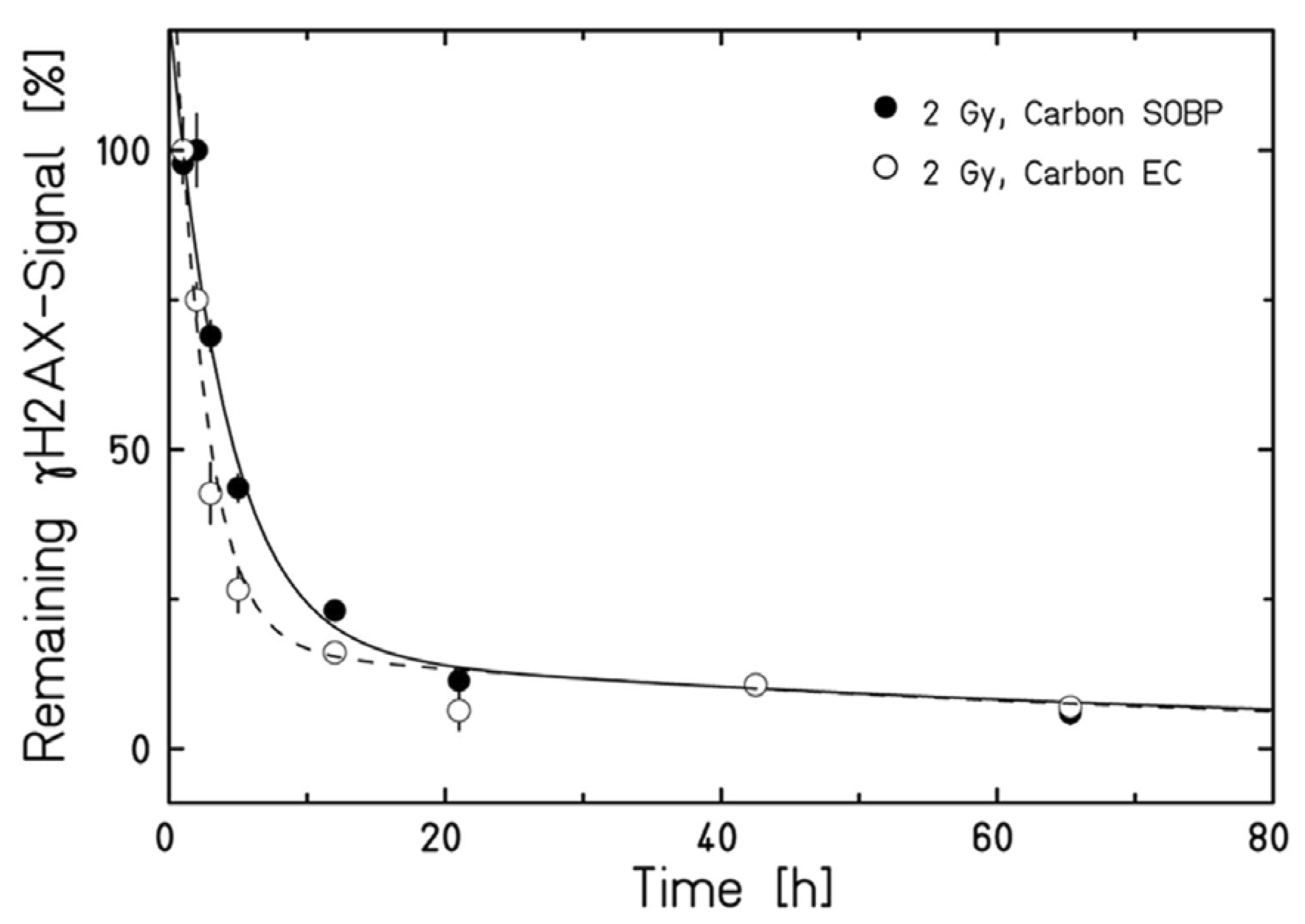
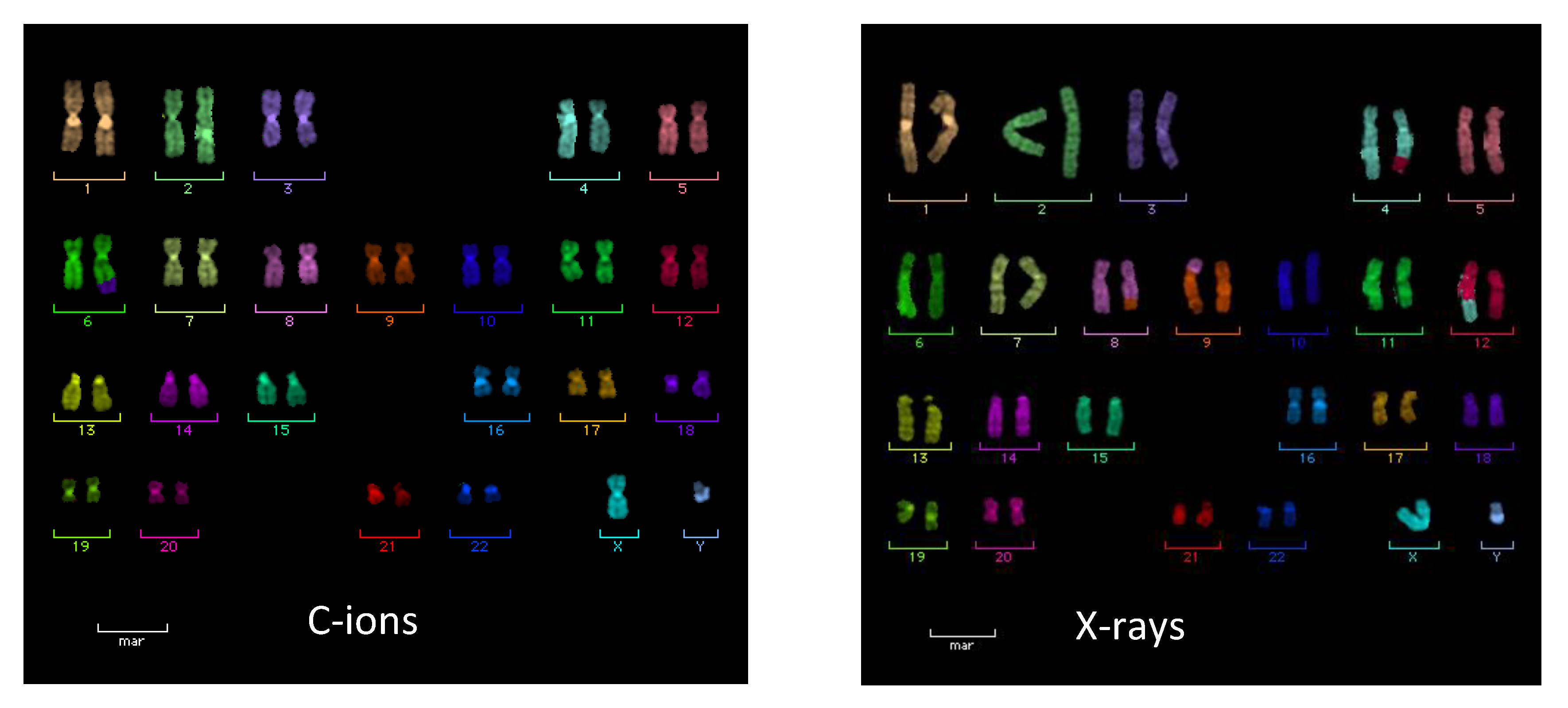
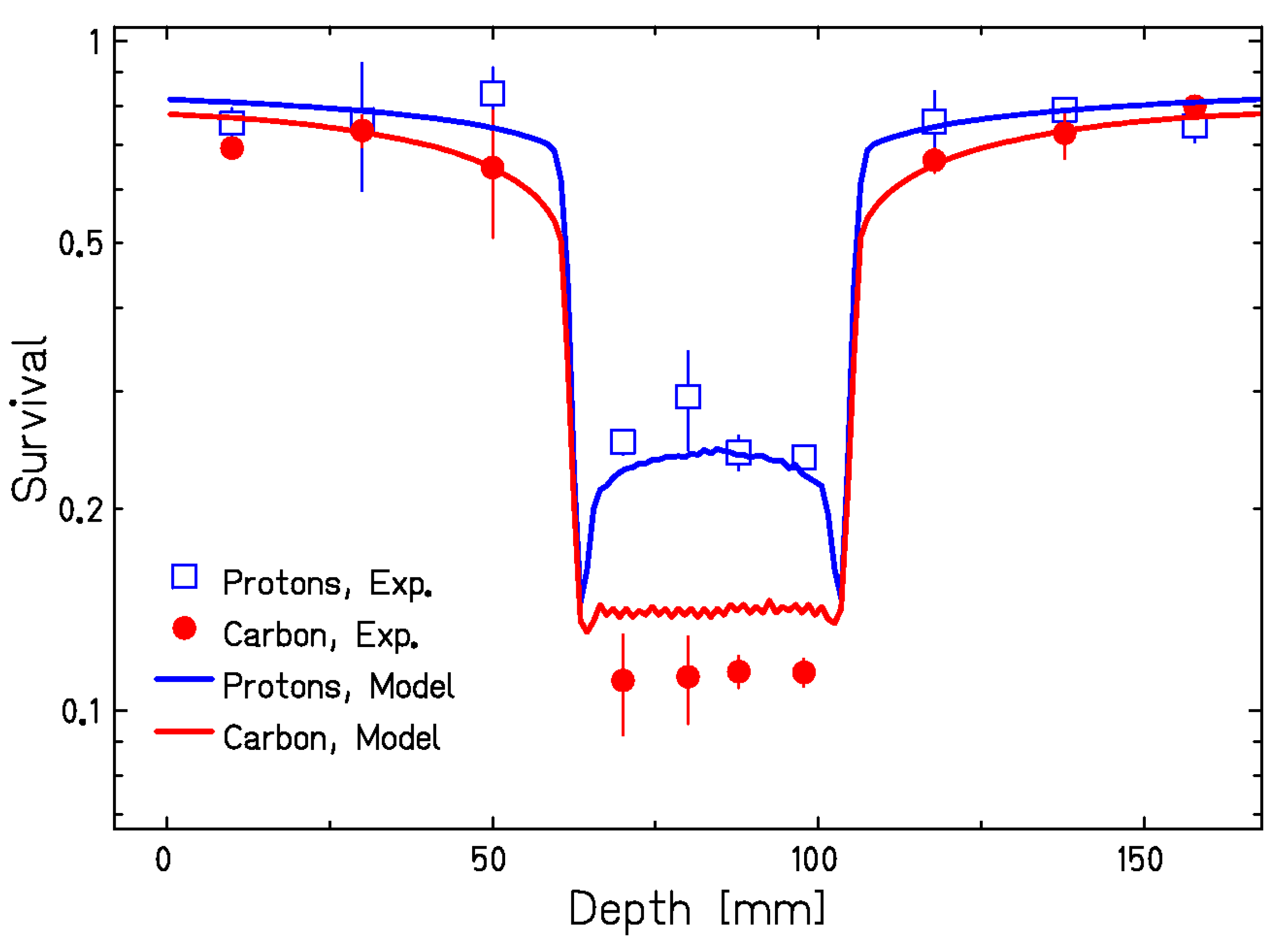
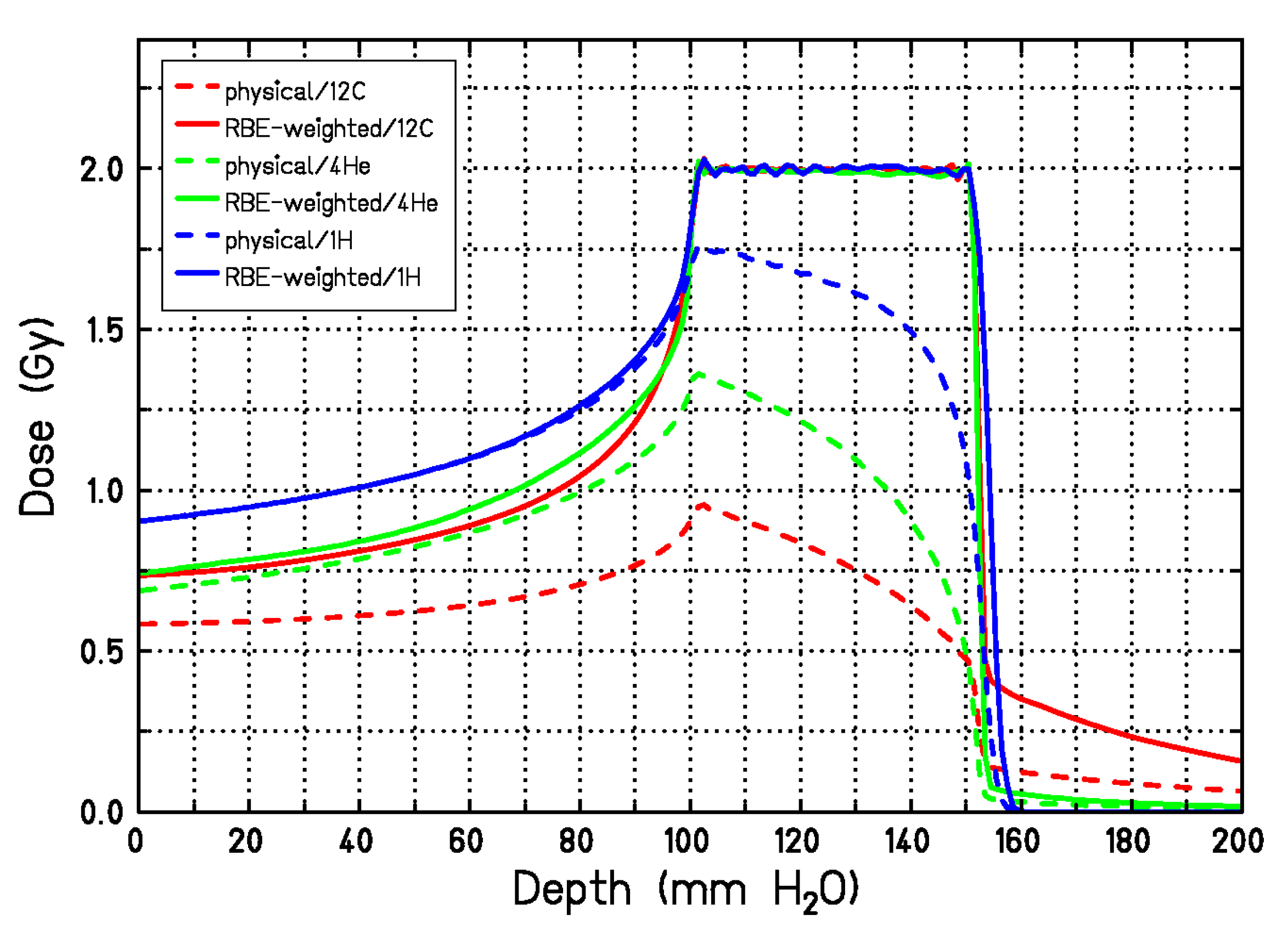
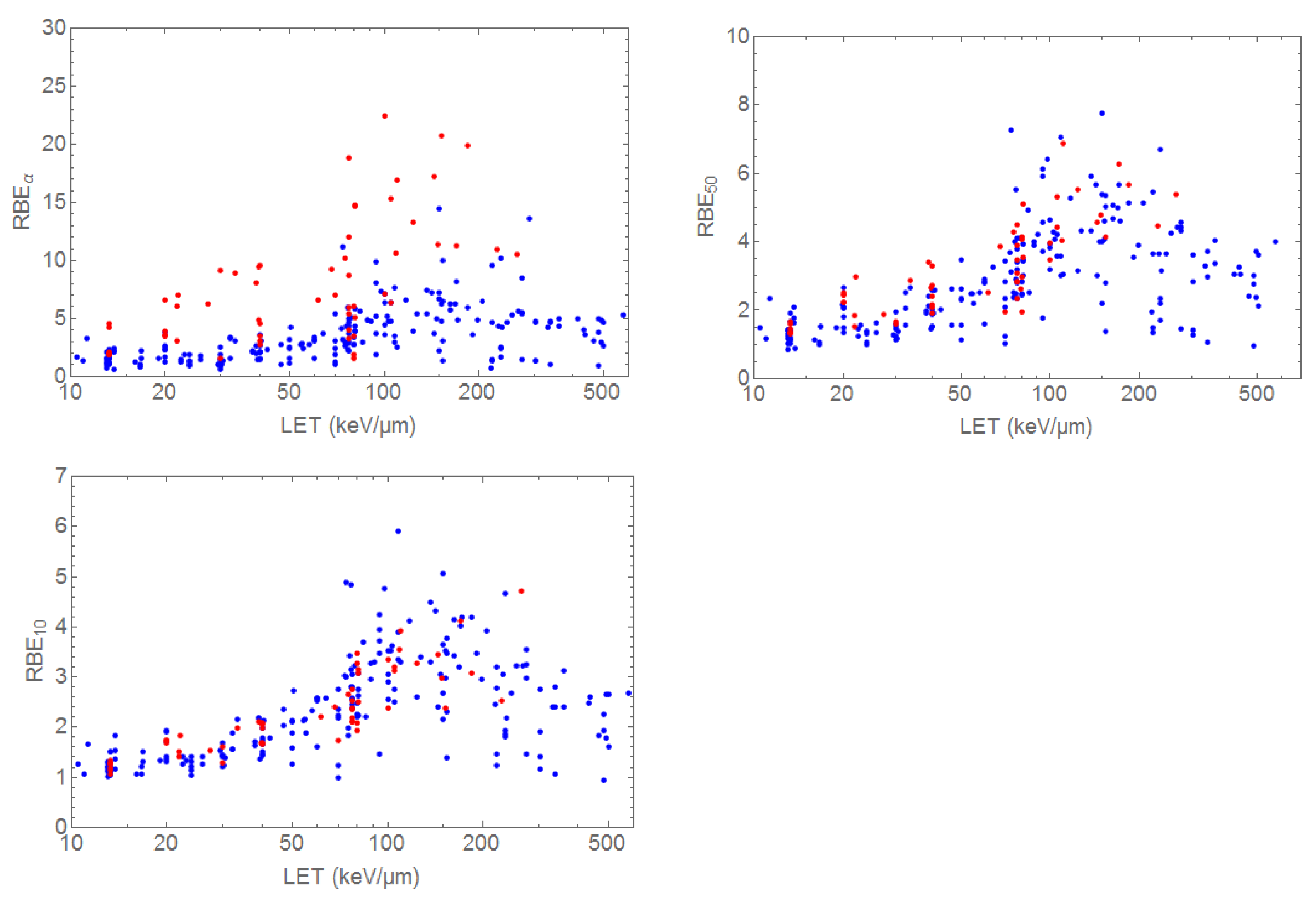
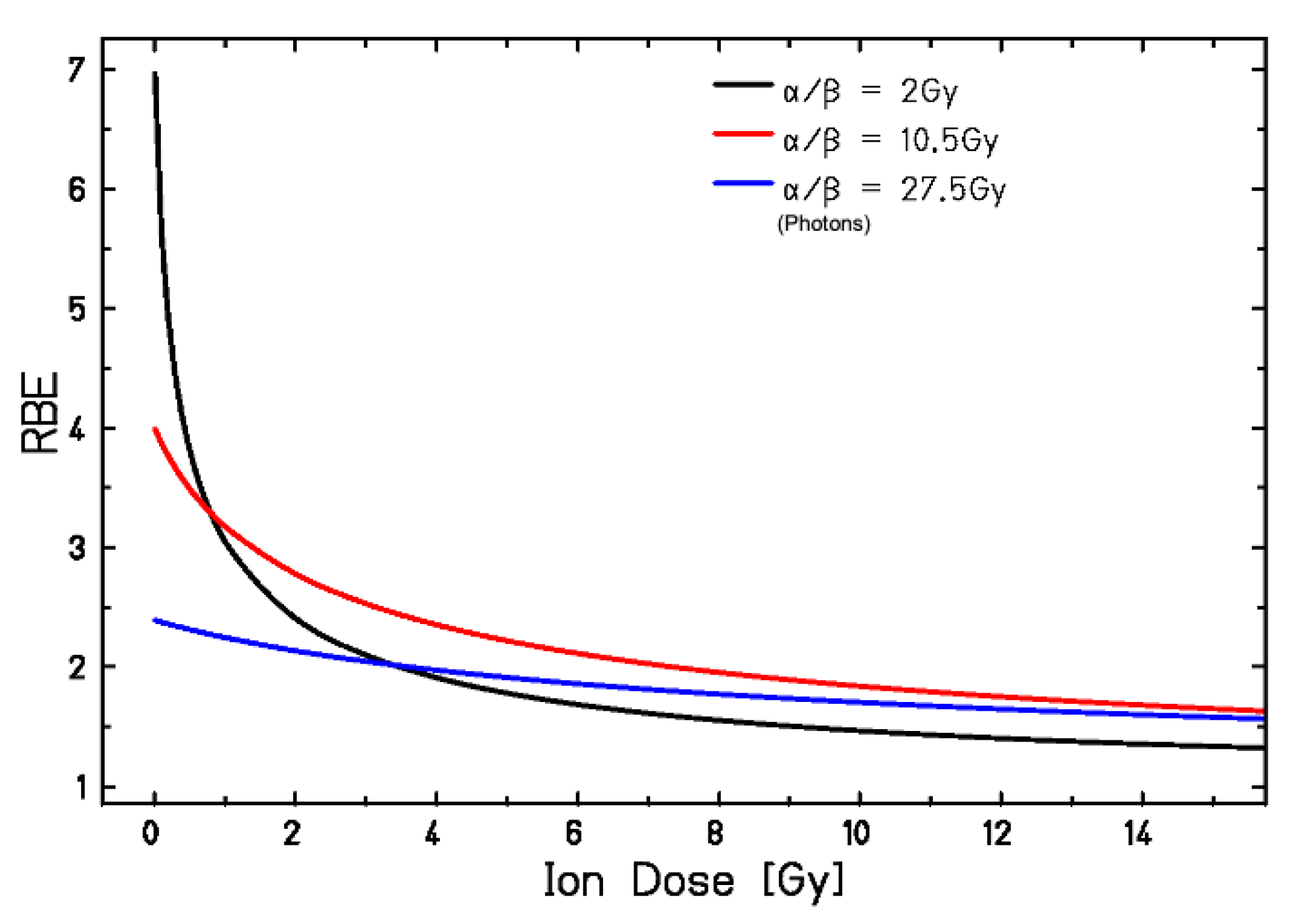
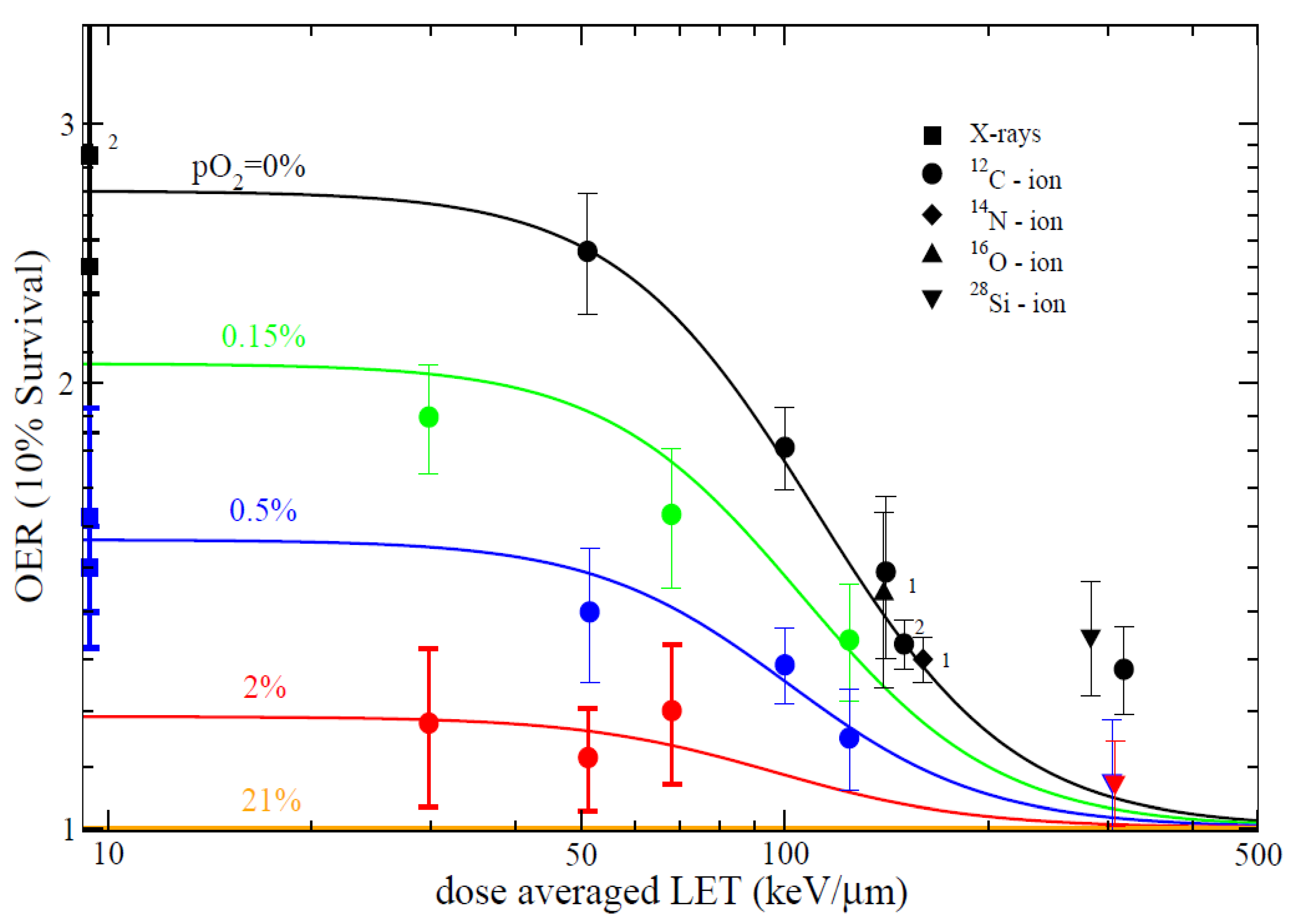
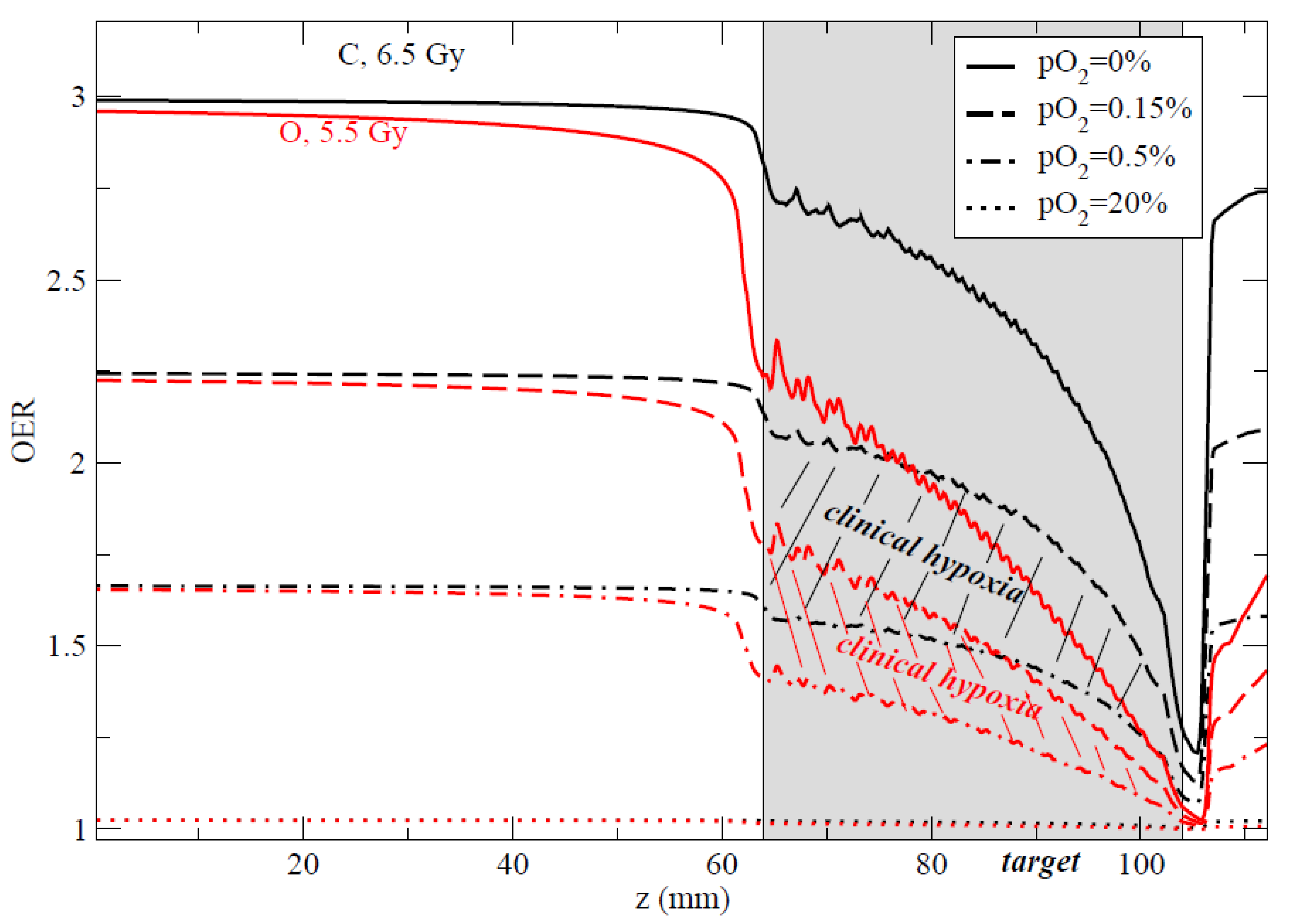
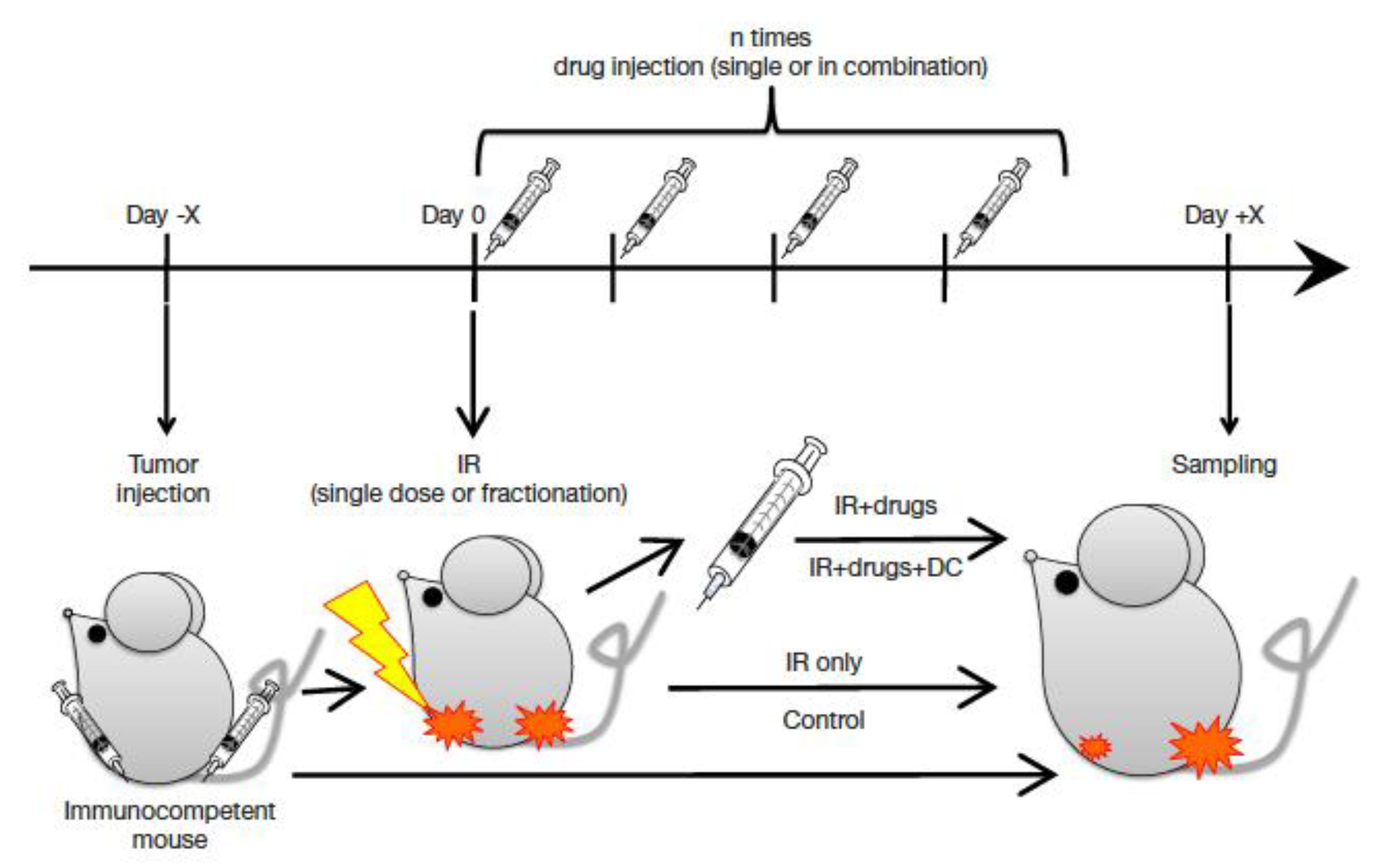
| Brief Title | ID | Sponsors | Phase | Condition | Arm 2 |
|---|---|---|---|---|---|
| Trial of proton versus carbon ion in patients with chondrosarcoma | NCT01182753 | Heidelberg University, Germany | III | Chondro-sarcoma of the skull base | Protons |
| Randomised trial of proton vs. carbon ion in patients with chordoma | NCT01182779 | Heidelberg University, Germany | III | Chordoma of the skull base | Protons |
| C-ion radiotherapy for glioblastoma | NCT01165671 CLEOPATRA | Heidelberg University, Germany | II | Primary glio-blastoma | Protons *,$ |
| Ion prostate irradiation | NCT01641185 IPI | Heidelberg University, Germany | II | Prostate cancer | Protons |
| Ion irradiation of sacrococcygeal chordoma | NCT01811394 ISAC | Heidelberg University, Germany | II | Sacrococcygeal chordoma | Protons |
| Neoadjuvant Irradiation of Retroperitoneal Soft Tissue Sarcoma | NCT04219202 Retro-Ion | Heidelberg University, Germany | II | Soft tissue sarcoma | Protons |
| Randomized C-ions vs. IMRT for radioresistant tumors | NCT02838602 ETOILE | Lyon University Hospitals, France | II | Adenoid cystic carcinoma and sarcomas | IMRT |
| Prospective trial comparing carbon ions to IMRT in pancreatic cancer | BAA-N01CM51007-51 | NCI, USA | I/III | Locally advanced pancreatic cancer | X-rays * |
| Prospective multicenter randomized trial for pancreas cancer | NCT03536182 CIPHER | Toshiba and UT Southwestern, Dallas, TX | III | Locally advanced pancreatic cancer | X-rays * |
| Newly Diagnosed Glioblastoma | NCT04536649 | Shanghai Proton and Heavy Ion Center | III | Primary glio-blastoma | X-rays * |
| Proton Versus Photon Radiotherapy for Nasopharyngeal Carcinoma | NCT04528394 | Shanghai Proton and Heavy Ion Center | II | Naso-pharyngeal Carcinoma | Protons § |
| Animal | Tissue | Doses (Gy) | Fractions | RBE | References |
|---|---|---|---|---|---|
| Rat | Spinal cord | 15–50 | 1–18 | 1.5–3.5 | [99,100,101,102,103,104] |
| Mouse | Skin reaction | 10–80 | 1–6 | 1.2–3.0 | [105,115,116] |
| 10–42 | 1 | 1.36 | [106] | ||
| 10–60 | 1 | 1.3–2.16 | [117] | ||
| 10–35 | 1 | 1.2–1.5 | [118] | ||
| Mouse | Lung fibrosis | 2–20 | 1–5 | 2.7–2.8 | [119,120] |
| 10–42 | 1 | 1.51 | [106] | ||
| Mouse | Mammary carcinoma | 10–42 | 1 | 1.48 | [106] |
| Mouse | Soft tissue sarcoma | 10 | 1 | 3 | [121] |
| Mouse | Fibrosarcoma | 10–80 | 1–12 | 1.4–3.0 | [105,122,123] |
| Nude mouse | Human esophageal cancer | 2–20 | 1 | 2.02 | [124] |
| Rat | Prostate carcinoma | 10–60 | 1–2 | 1.6–2.3 | [107,108,109,125] |
| Target | Small Molecule Inhibitor | Doses (Gy) | Cells | Outcome | Ref. |
|---|---|---|---|---|---|
| EGFR | Cetuximab | 1–4 | Human laryngeal squamous cell carcinoma | Inhibition of invasion | [234] |
| mTOR | Temsirolimus | 0.1–3 | Hepatocellular carcinoma | Additive effects in cell killing | [229] |
| Rapamycin | 1–5 | Chondrosarcoma cells | Sensitization of the C-ion effects | [235] | |
| PARP1/2 | Olaparib | 1–5 | Human pancreatic cancer cells | Sensitization of the C-ion effects | [236] |
| PARP-1 knockdown | 1–4 | HeLa cells | Sensitization of the C-ion effects | [237] | |
| Talazoparib | 2 | Human glioblastoma stem-like cells | Sensitization of the C-ion effects | [238] | |
| DNA-PKcs | Genistein | 2–6 | Human glioblastoma cell lines | Sensitization by inhibition of NHEJ | [239] |
| NU7026 | 2 | Human lung normal and cancer cells | Sensitization by inhibition of NHEJ | [240] | |
| NU7026 | 1–4 | Hela cells, human breast cancer cells | Sensitization mediated by telomere-end capping | [241] | |
| Hsp90 | TAS-116 | 1–5 | Hela, lung cancer and normal human fibroblasts, tumor xenografts | Radio-sensitization of both X-rays and C-ions | [242] |
| PU-H71 | 1–7 | HeLa derivative, human lung normal and cancer cell lines | Sensitization of cancer cell but not of normal cells | [243] | |
| Hedgehog | GANT61 | 0.2–4 | Prostate cancer cells Pediatric medulloblastoma | Sensitization and reduced migration | [244] |
| GANT61 | 0.2–4 | Human breast cancer cells | Reduced cell migration | [245] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tinganelli, W.; Durante, M. Carbon Ion Radiobiology. Cancers 2020, 12, 3022. https://doi.org/10.3390/cancers12103022
Tinganelli W, Durante M. Carbon Ion Radiobiology. Cancers. 2020; 12(10):3022. https://doi.org/10.3390/cancers12103022
Chicago/Turabian StyleTinganelli, Walter, and Marco Durante. 2020. "Carbon Ion Radiobiology" Cancers 12, no. 10: 3022. https://doi.org/10.3390/cancers12103022
APA StyleTinganelli, W., & Durante, M. (2020). Carbon Ion Radiobiology. Cancers, 12(10), 3022. https://doi.org/10.3390/cancers12103022







