Breast Cancer Cell–Neutrophil Interactions Enhance Neutrophil Survival and Pro-Tumorigenic Activities
Simple Summary
Abstract
1. Introduction
2. Results
2.1. A Higher Number of Neutrophils in Tumors from Breast Cancer Patients
2.2. Breast Cancer Patients with Chemotherapy-Resistance Expressed Higher Levels of Neutrophil-Related Genes
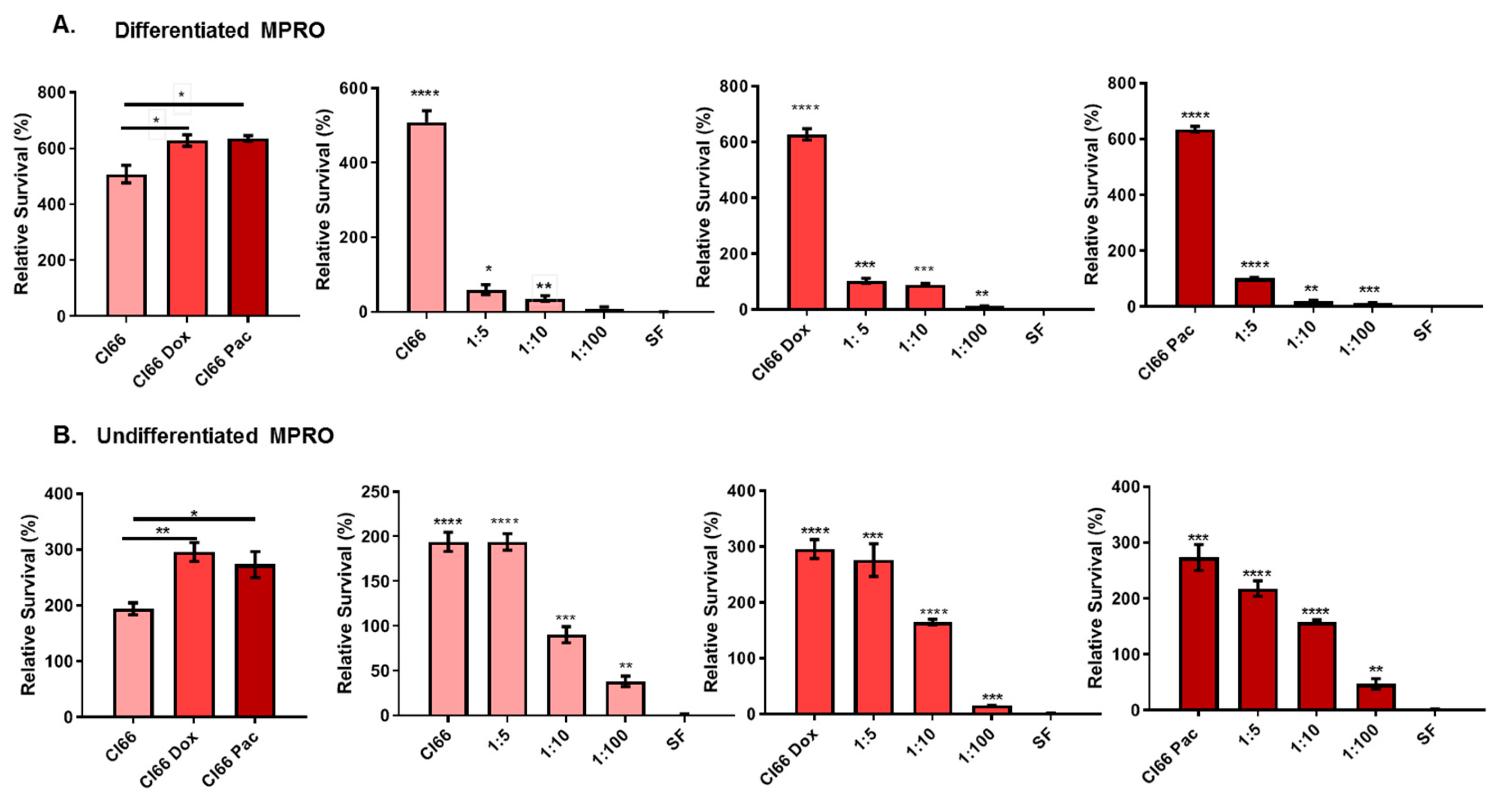
2.3. The Supernatant from Chemotherapy-Resistant Breast Cancer Cell Lines Enhanced Neutrophil Survival
2.4. Breast Cancer Cells Modulated Expression of Neutrophil-Secreted Pro-Tumor Factors
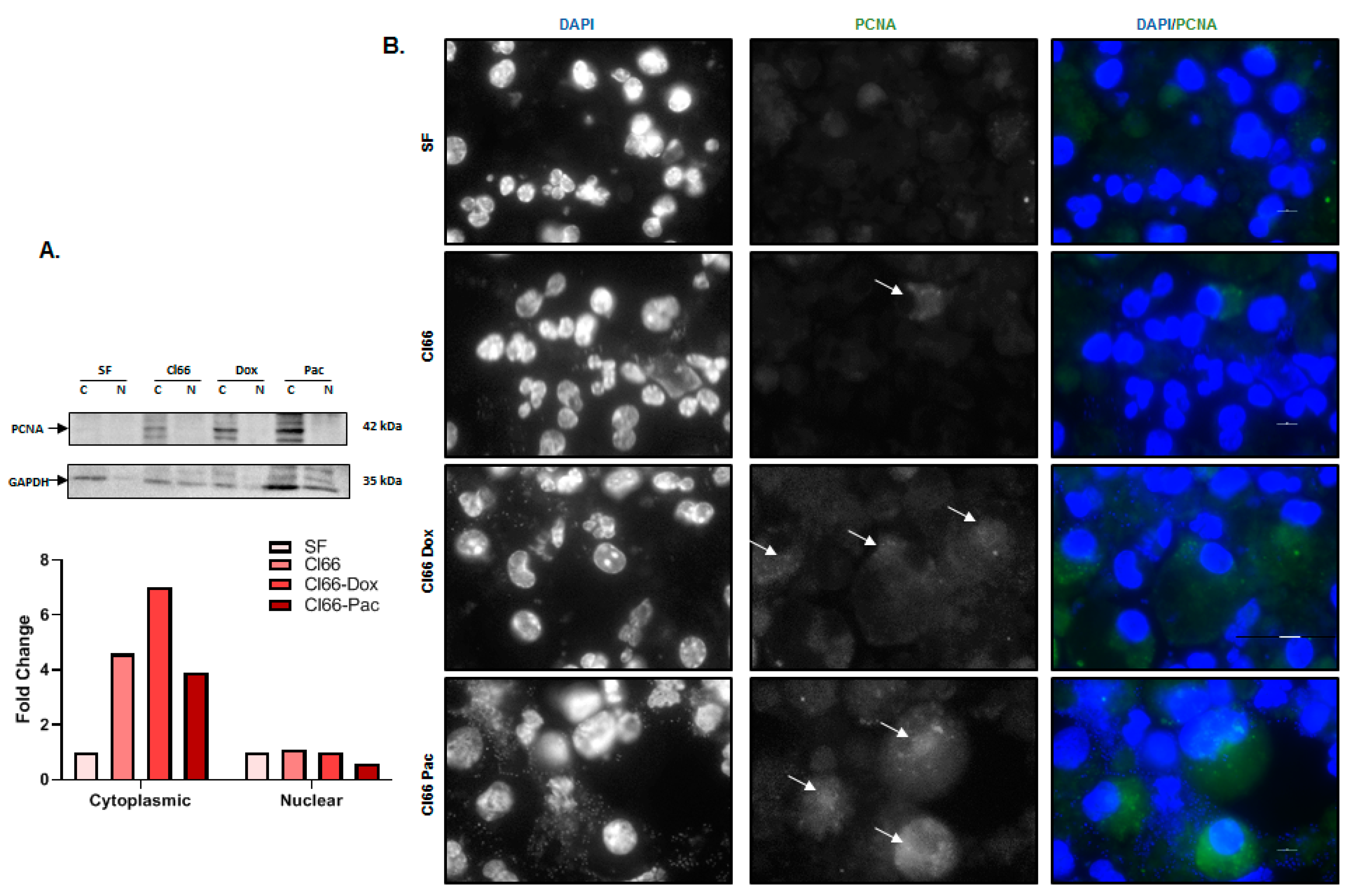
2.5. Neutrophils Treated with the Supernatant of Breast Cancer Cell Lines Formed NETs
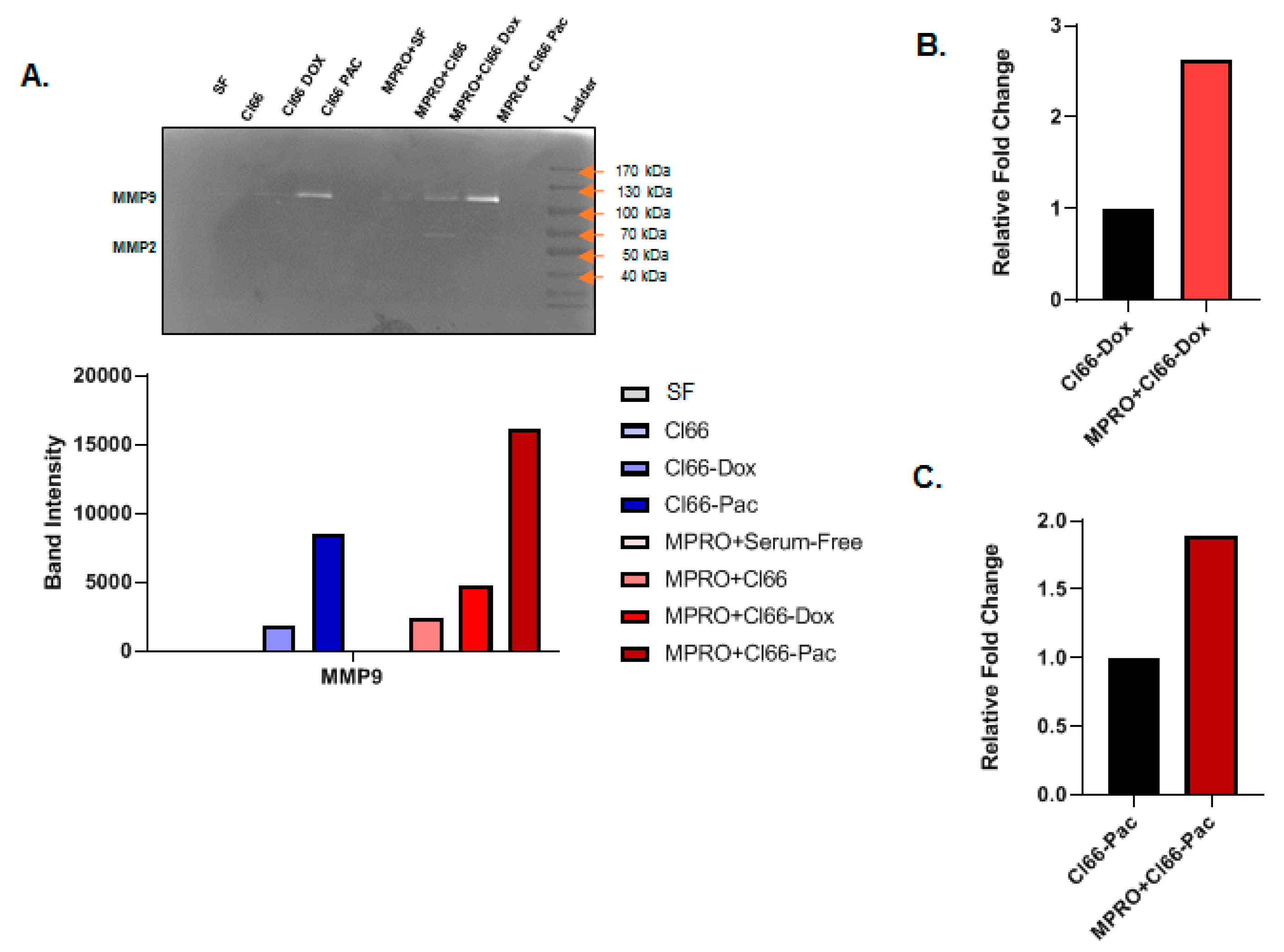
2.6. Neutrophils Treated with the Supernatant of Chemotherapy-Resistant Cells Secreted MMPs
3. Discussion
4. Materials and Methods
4.1. Cell Lines and Reagents
4.2. Human Breast Cancer Specimens
4.3. Immunohistochemistry
4.4. Bioinformatic Analysis
4.5. mRNA Analysis
4.6. Immunofluorescence
4.7. Cell Viability Assay
4.8. Immunoblotting
4.9. Gelatin Zymography
4.10. Statistical analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Siegel, R.L.; Miller, K.D.; Jemal, A. Cancer statistics, 2020. CA Cancer J. Clin. 2020, 70, 7–30. [Google Scholar] [CrossRef]
- Miller, K.D.; Siegel, R.L.; Lin, C.C.; Mariotto, A.B.; Kramer, J.L.; Rowland, J.H.; Stein, K.D.; Alteri, R.; Jemal, A. Cancer treatment and survivorship statistics, 2016. CA Cancer J. Clin. 2016, 66, 271–289. [Google Scholar] [CrossRef] [PubMed]
- Bagnyukova, T.V.; Serebriiskii, I.G.; Zhou, Y.; Hopper-Borge, E.A.; Golemis, E.A.; Astsaturov, I. Chemotherapy and signaling: How can targeted therapies supercharge cytotoxic agents? Cancer Biol. Ther. 2010, 10, 839–853. [Google Scholar] [CrossRef] [PubMed]
- Alfarouk, K.O.; Stock, C.M.; Taylor, S.; Walsh, M.; Muddathir, A.K.; Verduzco, D.; Bashir, A.H.; Mohammed, O.Y.; Elhassan, G.O.; Harguindey, S.; et al. Resistance to cancer chemotherapy: Failure in drug response from ADME to P-gp. Cancer Cell Int. 2015, 15, 71. [Google Scholar] [CrossRef] [PubMed]
- Echeverria, G.V.; Ge, Z.; Seth, S.; Zhang, X.; Jeter-Jones, S.; Zhou, X.; Cai, S.; Tu, Y.; McCoy, A.; Peoples, M.; et al. Resistance to neoadjuvant chemotherapy in triple-negative breast cancer mediated by a reversible drug-tolerant state. Sci. Transl. Med. 2019, 11. [Google Scholar] [CrossRef]
- Chun, K.H.; Park, J.H.; Fan, S. Predicting and overcoming chemotherapeutic resistance in breast cancer. Adv. Exp. Med. Biol. 2017, 1026, 59–104. [Google Scholar] [CrossRef]
- Edwardson, D.W.; Boudreau, J.; Mapletoft, J.; Lanner, C.; Kovala, A.T.; Parissenti, A.M. Inflammatory cytokine production in tumor cells upon chemotherapy drug exposure or upon selection for drug resistance. PLoS ONE 2017, 12, e0183662. [Google Scholar] [CrossRef]
- Sharma, B.; Varney, M.L.; Saxena, S.; Wu, L.; Singh, R.K. Induction of CXCR2 ligands, stem cell-like phenotype, and metastasis in chemotherapy-resistant breast cancer cells. Cancer Lett. 2016, 372, 192–200. [Google Scholar] [CrossRef]
- Wu, S.; Saxena, S.; Varney, M.L.; Singh, R.K. CXCR1/2 chemokine network regulates melanoma resistance to chemotherapies mediated by NF-kappaB. Curr. Mol. Med. 2017, 17, 436–449. [Google Scholar] [CrossRef]
- Wu, L.; Saxena, S.; Awaji, M.; Singh, R.K. Tumor-associated neutrophils in cancer: Going pro. Cancers 2019, 11, 564. [Google Scholar] [CrossRef]
- Ha, H.; Debnath, B.; Neamati, N. Role of the CXCL8-CXCR1/2 axis in cancer and inflammatory diseases. Theranostics 2017, 7, 1543–1588. [Google Scholar] [CrossRef] [PubMed]
- Planaguma, A.; Domenech, T.; Pont, M.; Calama, E.; Garcia-Gonzalez, V.; Lopez, R.; Auli, M.; Lopez, M.; Fonquerna, S.; Ramos, I.; et al. Combined anti CXC receptors 1 and 2 therapy is a promising anti-inflammatory treatment for respiratory diseases by reducing neutrophil migration and activation. Pulm. Pharmacol. Ther. 2015, 34, 37–45. [Google Scholar] [CrossRef] [PubMed]
- Cheng, Y.; Ma, X.L.; Wei, Y.Q.; Wei, X.W. Potential roles and targeted therapy of the CXCLs/CXCR2 axis in cancer and inflammatory diseases. Biochim. Biophys. Acta Rev. Cancer. 2019, 1871, 289–312. [Google Scholar] [CrossRef] [PubMed]
- Sharma, B.; Nawandar, D.M.; Nannuru, K.C.; Varney, M.L.; Singh, R.K. Targeting CXCR2 enhances chemotherapeutic response, inhibits mammary tumor growth, angiogenesis, and lung metastasis. Mol. Cancer Ther. 2013, 12, 799–808. [Google Scholar] [CrossRef]
- Singh, S.; Varney, M.; Singh, R.K. Host CXCR2-dependent regulation of melanoma growth, angiogenesis, and experimental lung metastasis. Cancer Res. 2009, 69, 411–415. [Google Scholar] [CrossRef]
- Sharma, B.; Nannuru, K.C.; Varney, M.L.; Singh, R.K. Host Cxcr2-dependent regulation of mammary tumor growth and metastasis. Clin. Exp. Metastasis 2015, 32, 65–72. [Google Scholar] [CrossRef]
- Sharma, B.; Nannuru, K.C.; Saxena, S.; Varney, M.L.; Singh, R.K. CXCR2: A novel mediator of mammary tumor bone metastasis. Int. J. Mol. Sci. 2019, 20, 1237. [Google Scholar] [CrossRef]
- Guo, F.; Long, L.; Wang, J.; Wang, Y.; Liu, Y.; Wang, L.; Luo, F. Insights on CXC chemokine receptor 2 in breast cancer: An emerging target for oncotherapy. Oncol. Lett. 2019, 18, 5699–5708. [Google Scholar] [CrossRef]
- Nannuru, K.C.; Sharma, B.; Varney, M.L.; Singh, R.K. Role of chemokine receptor CXCR2 expression in mammary tumor growth, angiogenesis and metastasis. J. Carcinog. 2011, 10, 40. [Google Scholar] [CrossRef]
- Sody, S.; Uddin, M.; Gruneboom, A.; Gorgens, A.; Giebel, B.; Gunzer, M.; Brandau, S. Distinct spatio-temporal dynamics of tumor-associated neutrophils in small tumor lesions. Front. Immunol. 2019, 10, 1419. [Google Scholar] [CrossRef]
- Wang, Y.; Wang, K.; Han, G.C.; Wang, R.X.; Xiao, H.; Hou, C.M.; Guo, R.F.; Dou, Y.; Shen, B.F.; Li, Y.; et al. Neutrophil infiltration favors colitis-associated tumorigenesis by activating the interleukin-1 (IL-1)/IL-6 axis. Mucosal. Immunol. 2014, 7, 1106–1115. [Google Scholar] [CrossRef] [PubMed]
- Tsuda, Y.; Fukui, H.; Asai, A.; Fukunishi, S.; Miyaji, K.; Fujiwara, S.; Teramura, K.; Fukuda, A.; Higuchi, K. An immunosuppressive subtype of neutrophils identified in patients with hepatocellular carcinoma. J. Clin. Biochem. Nutr. 2012, 51, 204–212. [Google Scholar] [CrossRef] [PubMed]
- Coffelt, S.B.; Wellenstein, M.D.; de Visser, K.E. Neutrophils in cancer: Neutral no more. Nat. Rev. Cancer 2016, 16, 431–446. [Google Scholar] [CrossRef] [PubMed]
- Felix, K.; Gaida, M.M. Neutrophil-derived proteases in the microenvironment of pancreatic cancer-active players in tumor progression. Int. J. Biol. Sci. 2016, 12, 302–313. [Google Scholar] [CrossRef] [PubMed]
- El Rayes, T.; Catena, R.; Lee, S.; Stawowczyk, M.; Joshi, N.; Fischbach, C.; Powell, C.A.; Dannenberg, A.J.; Altorki, N.K.; Gao, D.; et al. Lung inflammation promotes metastasis through neutrophil protease-mediated degradation of Tsp-1. Proc. Natl. Acad. Sci. USA 2015, 112, 16000–16005. [Google Scholar] [CrossRef] [PubMed]
- Al-Batran, S.; Wirtz, R.M.; Pauligk, C.; Steinmetz, K.; Probst, S.; Hartmann, J.T.; Hofheinz, R.; Altmannsberger, H.M.; Petry, C.; Jäger, E. Association of elevated matrix metalloproteinase-9 (MMP-9) mRNA expression levels with resistance to chemotherapy and survival in patients with metastatic gastric cancer receiving first-line chemotherapy: Results from the FLO versus FLP gastric cancer phase III trial of the AIO. J. Clin. Oncol. 2008, 26, 4544. [Google Scholar] [CrossRef]
- Li, Y.; Cao, X.; Liu, Y.; Zhao, Y.; Herrmann, M. Neutrophil extracellular traps formation and aggregation Orchestrate induction and resolution of sterile crystal-mediated inflammation. Front. Immunol. 2018, 9, 1559. [Google Scholar] [CrossRef]
- Prince, L.R.; Allen, L.; Jones, E.C.; Hellewell, P.G.; Dower, S.K.; Whyte, M.K.; Sabroe, I. The role of interleukin-1beta in direct and toll-like receptor 4-mediated neutrophil activation and survival. Am. J. Pathol. 2004, 165, 1819–1826. [Google Scholar] [CrossRef]
- Papayannopoulos, V. Neutrophil extracellular traps in immunity and disease. Nat. Rev. Immunol. 2018, 18, 134–147. [Google Scholar] [CrossRef]
- Gentles, A.J.; Newman, A.M.; Liu, C.L.; Bratman, S.V.; Feng, W.; Kim, D.; Nair, V.S.; Xu, Y.; Khuong, A.; Hoang, C.D.; et al. The prognostic landscape of genes and infiltrating immune cells across human cancers. Nat. Med. 2015, 21, 938–945. [Google Scholar] [CrossRef]
- Wu, L.; Awaji, M.; Saxena, S.; Varney, M.L.; Sharma, B.; Singh, R.K. IL-17-CXC chemokine receptor 2 axis facilitates breast cancer progression by up-regulating neutrophil recruitment. Am. J. Pathol. 2020, 190, 222–233. [Google Scholar] [CrossRef] [PubMed]
- Witko-Sarsat, V.; Mocek, J.; Bouayad, D.; Tamassia, N.; Ribeil, J.A.; Candalh, C.; Davezac, N.; Reuter, N.; Mouthon, L.; Hermine, O.; et al. Proliferating cell nuclear antigen acts as a cytoplasmic platform controlling human neutrophil survival. J. Exp. Med. 2010, 207, 2631–2645. [Google Scholar] [CrossRef] [PubMed]
- Cools-Lartigue, J.; Spicer, J.; McDonald, B.; Gowing, S.; Chow, S.; Giannias, B.; Bourdeau, F.; Kubes, P.; Ferri, L. Neutrophil extracellular traps sequester circulating tumor cells and promote metastasis. J. Clin. Invest. 2013. [Google Scholar] [CrossRef] [PubMed]
- Bekes, E.M.; Schweighofer, B.; Kupriyanova, T.A.; Zajac, E.; Ardi, V.C.; Quigley, J.P.; Deryugina, E.I. Tumor-recruited neutrophils and neutrophil TIMP-free MMP-9 regulate coordinately the levels of tumor angiogenesis and efficiency of malignant cell intravasation. Am. J. Pathol. 2011, 179, 1455–1470. [Google Scholar] [CrossRef] [PubMed]
- Medeiros, N.I.; Fares, R.C.; Franco, E.P.; Sousa, G.R.; Mattos, R.T.; Chaves, A.T.; Nunes, M.D.; Dutra, W.O.; Correa-Oliveira, R.; Rocha, M.O.; et al. Differential expression of matrix metalloproteinases 2, 9 and cytokines by neutrophils and monocytes in the clinical forms of chagas disease. PLoS Negl. Trop. Dis. 2017, 11, e0005284. [Google Scholar] [CrossRef] [PubMed]
- Mehner, C.; Hockla, A.; Miller, E.; Ran, S.; Radisky, D.C.; Radisky, E.S. Tumor cell-produced matrix metalloproteinase 9 (MMP-9) drives malignant progression and metastasis of basal-like triple negative breast cancer. Oncotarget 2014, 5, 2736–2749. [Google Scholar] [CrossRef] [PubMed]
- Deryugina, E.I.; Quigley, J.P. Tumor angiogenesis: MMP-mediated induction of intravasation- and metastasis-sustaining neovasculature. Matrix Biol. 2015, 44, 94–112. [Google Scholar] [CrossRef] [PubMed]
- Hanahan, D.; Weinberg, R. Hallmarks of cancer: The next generation. Cell 2011, 144, 646–674. [Google Scholar] [CrossRef]
- Wu, L.; Saxena, S.; Singh, R.K. Neutrophils in the Tumor Microenvironment. In Tumor Microenvironment: Hematopoietic Cells—Part A; Birbrair, A., Ed.; Springer International Publishing: Cham, Switzerland, 2020; pp. 1–20. [Google Scholar] [CrossRef]
- Zilio, S.; Serafini, P. Neutrophils and granulocytic MDSC: The janus god of cancer immunotherapy. Vaccines 2016, 4, 31. [Google Scholar] [CrossRef]
- Thorn, C.F.; Oshiro, C.; Marsh, S.; Hernandez-Boussard, T.; McLeod, H.; Klein, T.E.; Altman, R.B. Doxorubicin pathways: Pharmacodynamics and adverse effects. Pharm. Genom. 2011, 21, 440–446. [Google Scholar] [CrossRef]
- Abu Samaan, T.M.; Samec, M.; Liskova, A.; Kubatka, P.; Busselberg, D. Paclitaxel’s mechanistic and clinical effects on breast cancer. Biomolecules 2019, 9, 789. [Google Scholar] [CrossRef] [PubMed]
- Sheng, S.; Zhang, J.; Ai, J.; Hao, X.; Luan, R. Aberrant expression of IL-23/IL-23R in patients with breast cancer and its clinical significance. Mol. Med. Rep. 2018, 17, 4639–4644. [Google Scholar] [CrossRef] [PubMed]
- Jin, Z.; Wang, W.; Jiang, N.; Zhang, L.; Li, Y.; Xu, X.; Cai, S.; Wei, L.; Liu, X.; Chen, G.; et al. Clinical implications of iNOS levels in triple-negative breast cancer responding to neoadjuvant chemotherapy. PLoS ONE 2015, 10, e0130286. [Google Scholar] [CrossRef] [PubMed]
- Takesue, S.; Ohuchida, K.; Shinkawa, T.; Otsubo, Y.; Matsumoto, S.; Sagara, A.; Yonenaga, A.; Ando, Y.; Kibe, S.; Nakayama, H.; et al. Neutrophil extracellular traps promote liver micrometastasis in pancreatic ductal adenocarcinoma via the activation of cancerassociated fibroblasts. Int. J. Oncol. 2020, 56, 596–605. [Google Scholar] [CrossRef] [PubMed]
- Teijeira, A.; Garasa, S.; Gato, M.; Alfaro, C.; Migueliz, I.; Cirella, A.; de Andrea, C.; Ochoa, M.C.; Otano, I.; Etxeberria, I.; et al. CXCR1 and CXCR2 chemokine receptor agonists produced by tumors induce neutrophil extracellular traps that interfere with immune cytotoxicity. Immunity 2020, 52, 856–871, e858. [Google Scholar] [CrossRef]
- Kerros, C.; Tripathi, S.C.; Zha, D.; Mehrens, J.M.; Sergeeva, A.; Philips, A.V.; Qiao, N.; Peters, H.L.; Katayama, H.; Sukhumalchandra, P.; et al. Neuropilin-1 mediates neutrophil elastase uptake and cross-presentation in breast cancer cells. J. Biol. Chem. 2017, 292, 10295–10305. [Google Scholar] [CrossRef]
- Yoon, C.I.; Park, S.; Cha, Y.J.; Lee, H.S.; Bae, S.J.; Cha, C.; Lee, D.Y.; Ahn, S.G.; Jeong, J. Associations between absolute neutrophil count and lymphocyte-predominant breast cancer. Breast 2020, 50, 141–148. [Google Scholar] [CrossRef]
- Wang, Y.; Chen, J.; Yang, L.; Li, J.; Wu, W.; Huang, M.; Lin, L.; Su, S. Tumor-contacted neutrophils promote metastasis by a CD90-TIMP-1 juxtacrine-paracrine loop. Clin. Cancer Res. 2019, 25, 1957–1969. [Google Scholar] [CrossRef]
- Gupta, D.; Shah, H.P.; Malu, K.; Berliner, N.; Gaines, P. Differentiation and characterization of myeloid cells. Curr. Protoc. Immunol. 2014, 104, 29. [Google Scholar] [CrossRef]
- Awaji, M.; Saxena, S.; Wu, L.; Prajapati, D.R.; Purohit, A.; Varney, M.L.; Kumar, S.; Rachagani, S.; Ly, Q.P.; Jain, M.; et al. CXCR2 signaling promotes secretory cancer-associated fibroblasts in pancreatic ductal adenocarcinoma. FASEB J. 2020. [Google Scholar] [CrossRef]
- Chang, J.C.; Wooten, E.C.; Tsimelzon, A.; Hilsenbeck, S.G.; Gutierrez, M.C.; Tham, Y.L.; Kalidas, M.; Elledge, R.; Mohsin, S.; Osborne, C.K.; et al. Patterns of resistance and incomplete response to docetaxel by gene expression profiling in breast cancer patients. J. Clin. Oncol. 2005, 23, 1169–1177. [Google Scholar] [CrossRef] [PubMed]
- Babicki, S.; Arndt, D.; Marcu, A.; Liang, Y.; Grant, J.R.; Maciejewski, A.; Wishart, D.S. Heatmapper: Web-enabled heat mapping for all. Nucleic Acids Res. 2016, 44, 147–153. [Google Scholar] [CrossRef] [PubMed]
- Saxena, S.; Purohit, A.; Varney, M.L.; Hayashi, Y.; Singh, R.K. Semaphorin-5A maintains epithelial phenotype of malignant pancreatic cancer cells. BMC Cancer 2018, 18, 1283. [Google Scholar] [CrossRef] [PubMed]
- Saxena, S.; Hayashi, Y.; Wu, L.; Awaji, M.; Atri, P.; Varney, M.L.; Purohit, A.; Rachagani, S.; Batra, S.K.; Singh, R.K. Pathological and functional significance of Semaphorin-5A in pancreatic cancer progression and metastasis. Oncotarget 2018, 9, 5931–5943. [Google Scholar] [CrossRef]
- Wilson, T.J.; Nannuru, K.C.; Singh, R.K. Cathepsin G-mediated activation of pro-matrix metalloproteinase 9 at the tumor-bone interface promotes transforming growth factor-beta signaling and bone destruction. Mol. Cancer Res. 2009, 7, 1224–1233. [Google Scholar] [CrossRef]




| CXCR2 | CXCR1 | IL17A | NOS2 | MPO | CXCL3 | CXCL5 | CXCL8 | CXCL6 | ELANE | CTSG | CXCL2 | CXCL7 | CXCL1 | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| MEAN(R) | 208.089 | 197.244 | 87.4742 | 98.7889 | 16.2153 | 50.5724 | 19.3574 | 54.4989 | 7.56835 | 38.0585 | 136.787 | 8.87476 | 119.519 | 22.3482 |
| STANDARD DEVIATION | 69.761 | 96.442 | 30.401 | 67.474 | 8.205 | 25.416 | 7.473 | 46.34 | 2.208 | 14.042 | 40.764 | 3.148 | 247.595 | 32.185 |
| MEAN(S) | 136.445 | 115.511 | 85.8337 | 81.5115 | 8.73933 | 36.6042 | 13.4168 | 39.9076 | 9.19468 | 30.0681 | 146.353 | 11.9816 | 54.1024 | 14.949 |
| STANDARD DEVIATION | 19.809 | 41.735 | 18.228 | 49.583 | 3.625 | 14.686 | 3.291 | 51.389 | 3.312 | 16.513 | 40.134 | 7.798 | 98.898 | 17.365 |
| STUDENT T TEST | 0.00625 | 0.02477 | 0.88545 | 0.517 | 0.01692 | 0.14908 | 0.03437 | 0.49398 | 0.18111 | 0.23465 | 0.58968 | 0.21119 | 0.45655 | 0.53308 |
| Gene Name | Forward (5′–3′) | Reverse (3′–5′) |
|---|---|---|
| Il-23 | GCTGTGCCTAGGAGTAGCAG | TGGCTGTTGTCCTTGAGTCC |
| Gapdh | AGCCTCGTCCCGTAGACAAAA | GATGACAAGCTTCCCATTCTCG |
| Il-1β | GCAACTGTTCCTGAACTCAACT | ATCTTTTGGGGTCCGTCAACT |
| Ccl4 | TGACCAAAAGAGGCAGACAG | CTCCCCCAAAAAAACAAAAC |
| Ccl3 | TTCTCTGTACCATGACACTCTGC | CGTGGAATCTTCCGGCTGTAG |
| Ccl2 | TTAAAAACCTGGATCGGAACCAA | GCATTAGCTTCAGATTTACGGGT |
| Inos | GTTCTCAGCCCAACAATACAAGA | GTGGACGGGTCGATGTCAC |
| Protein | Primary Antibody | Source | Application | Dilution |
|---|---|---|---|---|
| PCNA | anti-PCNA | Santa Cruz, TX, USA Cell Signaling, MA, USA | WB IF | 1:1000 1:200 |
| Human neutrophil | Anti-Myeloperoxidase | Abcam, MA, | IHC | 1:100 |
| GAPDH | Anti GAPDH: sc-53;02 | Santa Cruz, TX | WB | 1:2000 |
| Anti-Histone H3 | (citrulline R2 + R8 + R17): ab5103 | Abcam, MA, | IF | 1:200 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wu, L.; Saxena, S.; Goel, P.; Prajapati, D.R.; Wang, C.; Singh, R.K. Breast Cancer Cell–Neutrophil Interactions Enhance Neutrophil Survival and Pro-Tumorigenic Activities. Cancers 2020, 12, 2884. https://doi.org/10.3390/cancers12102884
Wu L, Saxena S, Goel P, Prajapati DR, Wang C, Singh RK. Breast Cancer Cell–Neutrophil Interactions Enhance Neutrophil Survival and Pro-Tumorigenic Activities. Cancers. 2020; 12(10):2884. https://doi.org/10.3390/cancers12102884
Chicago/Turabian StyleWu, Lingyun, Sugandha Saxena, Paran Goel, Dipakkumar R. Prajapati, Cheng Wang, and Rakesh K. Singh. 2020. "Breast Cancer Cell–Neutrophil Interactions Enhance Neutrophil Survival and Pro-Tumorigenic Activities" Cancers 12, no. 10: 2884. https://doi.org/10.3390/cancers12102884
APA StyleWu, L., Saxena, S., Goel, P., Prajapati, D. R., Wang, C., & Singh, R. K. (2020). Breast Cancer Cell–Neutrophil Interactions Enhance Neutrophil Survival and Pro-Tumorigenic Activities. Cancers, 12(10), 2884. https://doi.org/10.3390/cancers12102884






