Allogeneic Hematopoietic Stem Cell Transplantation in Cutaneous T-Cell Lymphomas
Simple Summary
Abstract
1. Introduction
2. Selection of Patients for Allo-HSCT
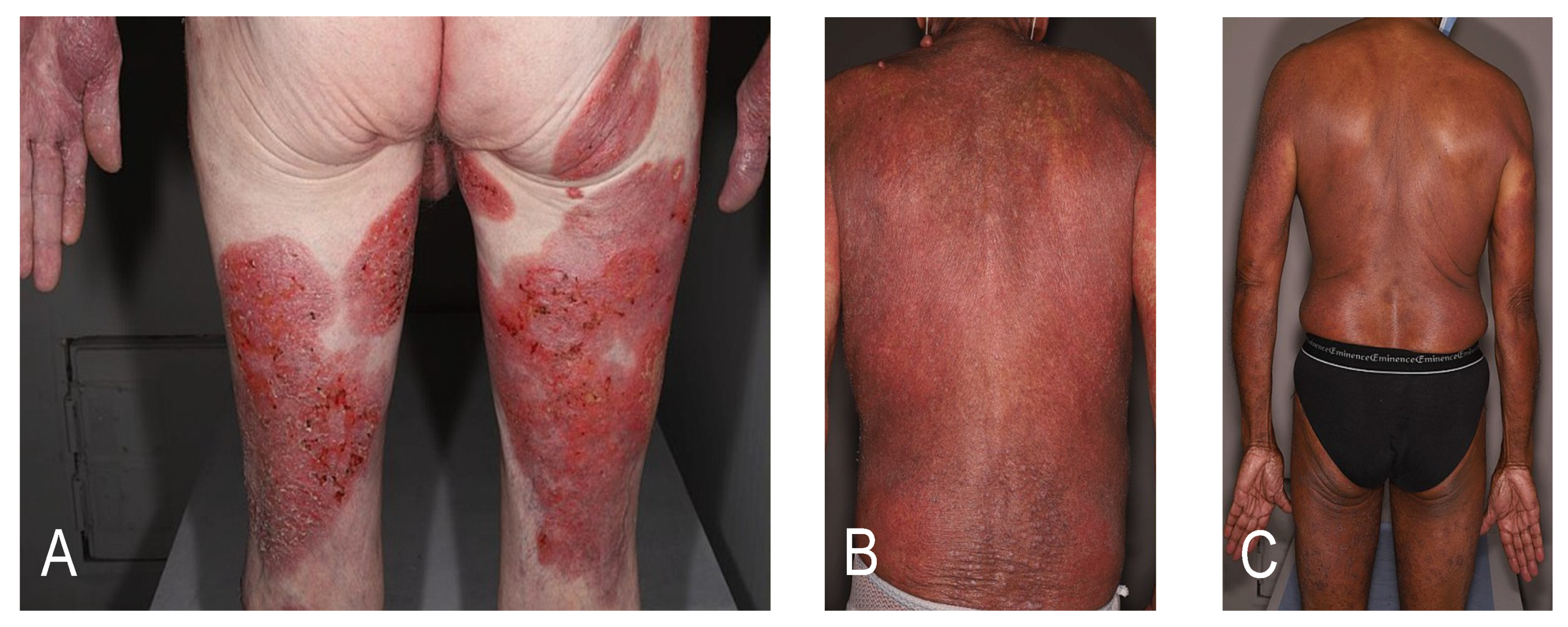
3. Bridging Therapies to Allo-HSCT
4. Different Conditioning Approaches and Types of Donors
5. Evolution Post Allo-HSCT
6. Best Practice Set of Recommendations
7. Conclusions and Perspectives
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Willemze, R.; Cerroni, L.; Kempf, W.; Berti, E.; Facchetti, F.; Swerdlow, S.H.; Jaffe, E.S. The 2018 update of the WHO-EORTC classification for primary cutaneous lymphomas. Blood 2019, 133, 1703–1714. [Google Scholar] [CrossRef]
- Gilson, D.; Whittaker, S.J.; Child, F.J.; Scarisbrick, J.J.; Illidge, T.M.; Parry, E.J.; Mohd Mustapa, M.F.; Exton, L.S.; Kanfer, E.; Rezvani, K.; et al. British Association of Dermatologists and U.K. Cutaneous Lymphoma Group guidelines for the management of primary cutaneous lymphomas 2018. Br. J. Dermatol. 2019, 180, 496–526. [Google Scholar] [CrossRef] [PubMed]
- Olsen, E.A.; Vonderheid, E.; Pimpinelli, N.; Willemze, R.; Kim, Y.; Knobler, R.; Zackheim, H.; Duvic, M.; Estrach, T.; Lamberg, S.; et al. Revisions to the staging and classification of mycosis fungoides and Sézary syndrome: A proposal of the international society for cutaneous lymphomas (ISCL) and the cutaneous lymphoma task force of the european organization of research and treatment of cancer (EORTC). Blood 2007, 110, 1713–1722. [Google Scholar] [CrossRef] [PubMed]
- Agar, N.S.; Wedgeworth, E.; Crichton, S.; Mitchell, T.J.; Cox, M.; Ferreira, S.; Robson, A.; Calonje, E.; Stefanato, C.M.; Wain, E.M.; et al. Survival outcomes and prognostic factors in mycosis fungoides/sézary syndrome: Validation of the revised international society for cutaneous lymphomas/european organisation for research and treatment of cancer staging proposal. J. Clin. Oncol. 2010, 28, 4730–4739. [Google Scholar] [CrossRef] [PubMed]
- Scarisbrick, J.; Prince, H.M.; Vermeer, M.H.; Quaglino, P.; Horwitz, S.; Porcu, P.; Stadler, R.; Wood, G.S.; Beylot-Barry, M.; Pham-Ledard, A.; et al. Cutaneous lymphoma international consortium study of outcome in advanced stages of mycosis fungoides and sézary syndrome: Effect of specific prognostic markers on survival and development of a prognostic model. J. Clin. Oncol. 2015, 33, 3766–3773. [Google Scholar] [CrossRef] [PubMed]
- Prince, H.M.; Querfeld, C. Integrating novel systemic therapies for the treatment of mycosis fungoides and Sézary syndrome. Best Pract. Res. Clin. Haematol. 2018, 31, 322–335. [Google Scholar] [CrossRef] [PubMed]
- Trautinger, F.; Eder, J.; Assaf, C.; Bagot, M.; Cozzio, A.; Dummer, R.; Gniadecki, R.; Klemke, C.-D.; Ortiz-Romero, P.L.; Papadavid, E.; et al. European Organisation for Research and Treatment of Cancer consensus recommendations for the treatment of mycosis fungoides/Sézary syndrome—Update 2017. Eur. J. Cancer 2017, 77, 57–74. [Google Scholar] [CrossRef] [PubMed]
- Iqbal, M.; Reljic, T.; Ayala, E.; Sher, T.; Murthy, H.S.; Roy, V.; Foran, J.; Tun, H.; Kumar, A.; Kharfan-Dabaja, M.A. Efficacy of allogeneic hematopoietic cell transplantation in cutaneous T cell lymphoma: Results of a systematic review and meta-analysis. Biol. Blood Marrow Transpl. 2020, 26, 76–82. [Google Scholar] [CrossRef]
- Burt, R.; Guitart, J.; Traynor, A.; Link, C.; Rosen, S.; Pandolfino, T.; Kuzel, T. Allogeneic hematopoietic stem cell transplantation for advanced mycosis fungoides: Evidence of a graft-versus-tumor effect. Bone Marrow Transplant 2000, 25, 111–113. [Google Scholar] [CrossRef]
- Herbert, K.E.; Spencer, A.; Grigg, A.; Ryan, G.; McCormack, C.; Prince, H.M. Graft-versus-lymphoma effect in refractory cutaneous T-cell lymphoma after reduced-intensity HLA-matched sibling allogeneic stem cell transplantation. Bone Marrow Transpl. 2004, 34, 521–525. [Google Scholar] [CrossRef]
- Duarte, R.F.; Canals, C.; Onida, F.; Gabriel, I.H.; Arranz, R.; Arcese, W.; Ferrant, A.; Kobbe, G.; Narni, F.; Deliliers, G.L.; et al. Allogeneic hematopoietic cell transplantation for patients with mycosis fungoides and sézary syndrome: A retrospective analysis of the lymphoma working party of the European group for blood and marrow transplantation. J. Clin. Oncol. 2010, 28, 4492–4499. [Google Scholar] [CrossRef] [PubMed]
- Cudillo, L.; Cerretti, R.; Picardi, A.; Mariotti, B.; De Angelis, G.; Cantonetti, M.; Postorino, M.; Ceresoli, E.; De Santis, G.; Nasso, D.; et al. Allogeneic hematopoietic stem cell transplantation in primary cutaneous T cell lymphoma. Ann. Hematol. 2018, 97, 1041–1048. [Google Scholar] [CrossRef] [PubMed]
- Quaglino, P.; Maule, M.; Prince, H.M.; Porcu, P.; Horwitz, S.; Duvic, M.; Talpur, R.; Vermeer, M.; Bagot, M.; Guitart, J.; et al. Global patterns of care in advanced stage mycosis fungoides/Sezary syndrome: A multicenter retrospective follow-up study from the Cutaneous Lymphoma International Consortium. Ann. Oncol. 2017, 28, 2517–2525. [Google Scholar] [CrossRef] [PubMed]
- NCCN Guidelines on Mycosis Fungoides/Sézary Syndrome [Internet]. Available online: https://www.nccn.org/professionals/physician_gls/default_nojava.aspx#site (accessed on 6 January 2020).
- De Masson, A.; Beylot-Barry, M.; Bouaziz, J.D.; De Latour, R.P.; Aubin, F.; Garciaz, S.; D’Incan, M.; Dereure, O.; Dalle, S.; Dompmartin, A.; et al. Allogeneic stem cell transplantation for advanced cutaneous T-cell lymphomas: A study from the french society of bone marrow transplantation and french study group on cutaneous lymphomas. Haematologica 2014, 99, 527–534. [Google Scholar] [CrossRef] [PubMed]
- Weng, W.-K.; Arai, S.; Rezvani, A.; Johnston, L.; Lowsky, R.; Miklos, D.; Shizuru, J.; Muffly, L.; Meyer, E.; Negrin, R.S.; et al. Nonmyeloablative allogeneic transplantation achieves clinical and molecular remission in cutaneous T-cell lymphoma. Blood Adv. 2020, 4, 4474–4482. [Google Scholar] [CrossRef] [PubMed]
- Duarte, R.F.; Boumendil, A.; Onida, F.; Gabriel, I.; Arranz, R.; Arcese, W.; Poiré, X.; Kobbe, G.; Narni, F.; Cortelezzi, A.; et al. Long-term outcome of allogeneic hematopoietic cell transplantation for patients with mycosis fungoides and sézary syndrome: A European society for blood and marrow transplantation lymphoma working party extended analysis. J. Clin. Oncol. 2014, 32, 3347–3348. [Google Scholar] [CrossRef]
- ElSawy, M.; Sorror, M.L. Up-to-date tools for risk assessment before allogeneic hematopoietic cell transplantation. Bone Marrow Transpl. 2016, 51, 1283–1300. [Google Scholar] [CrossRef] [PubMed]
- Lansigan, F.; Horwitz, S.M.; Pinter-Brown, L.C.; Carson, K.R.; Shustov, A.R.; Rosen, S.T.; Pro, B.; Hsi, E.D.; Federico, M.; Gisselbrecht, C.; et al. Outcomes of patients with transformed mycosis fungoides: Analysis from a prospective multicenter US cohort study. Clin. Lymphoma Myeloma Leuk. 2020. [Google Scholar] [CrossRef]
- Weng, W.K.; Armstrong, R.; Arai, S.; Desmarais, C.; Hoppe, R.; Kim, Y.H. Minimal residual disease monitoring with high-throughput sequencing of T cell receptors in cutaneous T cell lymphoma. Sci. Transl. Med. 2013, 5, 214ra171. [Google Scholar] [CrossRef]
- De Masson, A.; O’Malley, J.T.; Elco, C.P.; Garcia, S.S.; DiVito, S.J.; Lowry, E.L.; Tawa, M.; Fisher, D.C.; Devlin, P.M.; Teague, J.E.; et al. High-throughput sequencing of the T cell receptor β gene identifies aggressive early-stage mycosis fungoides. Sci. Transl. Med. 2018, 10, eaar5894. [Google Scholar] [CrossRef]
- Duarte, R.F.; Ebmt, O.B.O.T.L.W.P.O.T.; Schmitz, N.; Servitje, O.; Sureda, A. Haematopoietic stem cell transplantation for patients with primary cutaneous T-cell lymphoma. Bone Marrow Transpl. 2008, 41, 597–604. [Google Scholar] [CrossRef] [PubMed]
- Duvic, M.; Donato, M.; Dabaja, B.; Richmond, H.; Singh, L.; Wei, W.; Acholonu, S.; Khouri, I.; Champlin, R.; Hosing, C. Total skin electron beam and non-myeloablative allogeneic hematopoietic stem-cell transplantation in advanced mycosis fungoides and sézary syndrome. J. Clin. Oncol. 2010, 28, 2365–2372. [Google Scholar] [CrossRef] [PubMed]
- Hosing, C.; Bassett, R.; Dabaja, B.; Talpur, R.; Alousi, A.; Ciurea, S.; Popat, U.; Qazilbash, M.; Shpall, E.J.; Oki, Y.; et al. Allogeneic stem-cell transplantation in patients with cutaneous lymphoma: Updated results from a single institution. Ann. Oncol. 2015, 26, 2490–2495. [Google Scholar] [CrossRef] [PubMed]
- O’Malley, J.T.; De Masson, A.; Lowry, E.L.; Giobbie-Hurder, A.; Leboeuf, N.R.; LaRocca, C.; Gehad, A.; Seger, E.; Teague, J.E.; Fisher, D.C.; et al. Radiotherapy eradicates malignant T cells and is associated with improved survival in early-stage mycosis fungoides. Clin. Cancer Res. 2020, 26, 408–418. [Google Scholar] [CrossRef] [PubMed]
- Prince, H.M.; Kim, Y.H.; Horwitz, S.M.; Dummer, R.; Scarisbrick, J.; Quaglino, P.; Zinzani, P.L.; Wolter, P.; Sanches, J.A.; Ortiz-Romero, P.L.; et al. Brentuximab vedotin or physician’s choice in CD30-positive cutaneous T-cell lymphoma (ALCANZA): An international, open-label, randomised, phase 3, multicentre trial. Lancet 2017, 390, 555–566. [Google Scholar] [CrossRef]
- Schneeweiss, M.; Porpaczy, E.; Koch, M.; Jonak, C.; Schiefer, A.-I.; Simonitsch-Klupp, I.; Sillaber, C.; Mayerhöfer, M.; Jäger, U. Transformed mycosis fungoides: Bridging to allogeneic stem cell transplantation with brentuximab vedotin. Leuk. Lymphoma 2016, 57, 206–208. [Google Scholar] [CrossRef] [PubMed]
- Mariotto, S.; Tecchio, C.; Sorio, M.; Bertolasi, L.; Turatti, M.; Tozzi, M.C.; Benedetti, F.; Cavaletti, G.; Monaco, S.; Ferrari, S. Clinical and neurophysiological serial assessments of brentuximab vedotin-associated peripheral neuropathy. Leuk. Lymphoma 2019, 60, 2806–2809. [Google Scholar] [CrossRef]
- Garciaz, S.; Loschi, M.; De Masson, A.; Biard, L.; Mercier, M.; Tomowiak, C.; Delage, J.; Labussiere-Wallet, H.; Sibon, D.; Cassuto, O.; et al. Brentuximab vedotin as a bridge to allogeneic stem-cell transplantation for refractory or relapsing patients with CD30 positive anaplastic or T-cell non-Hodgkin lymphomas: A study on behalf of the SFGM-TC. Leuk. Lymphoma 2019, 60, 2802–2805. [Google Scholar] [CrossRef]
- André, R.; Ram-Wolff, C.; Battistella, M.; De Latour, R.P.; Petit, A.; Bouaziz, J.-D.; Brice, P.; Bagot, M.; De Masson, A. Effectiveness of brentuximab vedotin before and after allogeneic stem-cell transplantation in the management of transformed mycosis fungoides. Br. J. Dermatol. 2020, 182, 1503–1504. [Google Scholar] [CrossRef]
- Dumont, M.; Ram-Wolff, C.; Roelens, M.; Brice, P.; De Latour, R.P.; Battistella, M.; Madelaine, I.; Delyon, J.; Herms, F.; Bouaziz, J.-D.; et al. Efficacy and safety of brentuximab vedotin plus bendamustine in advanced-stage primary cutaneous T-cell lymphomas. Br. J. Dermatol. 2019, 181, 1315–1317. [Google Scholar] [CrossRef]
- Mahevas, T.; Ram-Wolff, C.; Battistella, M.; Pennamen, M.; Rivet, J.; Brice, P.; Bagot, M. Dramatic response to brentuximab vedotin in refractory nontransformed CD 30—Mycosis fungoides allowing allogeneic stem cell transplant and long-term complete remission. Br. J. Dermatol. 2019, 180, 1517–1520. [Google Scholar] [CrossRef] [PubMed]
- Dobos, G.; Battistella, M.; Jouenne, F.; Mourah, S.; Vignon-Pennamen, M.; Ram-Wolff, C.; Herms, F.; Dauendorffer, J.; Rivet, J.; Cayuela, J.; et al. Challenges in the diagnosis of primary cutaneous CD 30 + anaplastic large-cell lymphoma. Br. J. Dermatol. 2020, 182, 233–234. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.H.; Bagot, M.; Pinter-Brown, L.; Rook, A.H.; Porcu, P.; Horwitz, S.M.; Whittaker, S.; Tokura, Y.; Vermeer, M.; Zinzani, P.L.; et al. Mogamulizumab versus vorinostat in previously treated cutaneous T-cell lymphoma (MAVORIC): An international, open-label, randomised, controlled phase 3 trial. Lancet Oncol. 2018, 19, 1192–1204. [Google Scholar] [CrossRef]
- Bonnet, P.; Battistella, M.; Roelens, M.; Ram-Wolff, C.; Herms, F.; Frumholtz, L.; Bouaziz, J.D.; Brice, P.; Moins-Teisserenc, H.; Bagot, M.; et al. Association of autoimmunity and long-term complete remission in patients with Sézary syndrome treated with mogamulizumab. Br. J. Dermatol. 2019, 180, 419–420. [Google Scholar] [CrossRef]
- Alho, A.C.; Kim, H.T.; Chammas, M.J.; Reynolds, C.G.; Matos, T.R.; Forcade, E.; Whangbo, J.; Nikiforow, S.; Cutler, C.S.; Koreth, J.; et al. Unbalanced recovery of regulatory and effector T cells after allogeneic stem cell transplantation contributes to chronic GVHD. Blood 2016, 127, 646–657. [Google Scholar] [CrossRef] [PubMed]
- Inoue, Y.; Fuji, S.; Tanosaki, R.; Fukuda, T. Pretransplant mogamulizumab against ATLL might increase the risk of acute GVHD and non-relapse mortality. Bone Marrow Transpl. 2015, 51, 725–727. [Google Scholar] [CrossRef] [PubMed]
- Dai, J.; Almazan, T.H.; Hong, E.K.; Khodadoust, M.S.; Arai, S.; Weng, W.K.; Kim, Y.H. Potential association of anti-CCR4 antibody mogamulizumab and graft-vs-host disease in patients with mycosis fungoides and sézary syndrome. JAMA Dermatol. 2018, 154, 728–730. [Google Scholar] [CrossRef] [PubMed]
- Wu, P.A.; Kim, Y.H.; Lavori, P.W.; Hoppe, R.T.; Stockerl-Goldstein, K.E. A Meta-analysis of patients receiving allogeneic or autologous hematopoietic stem cell transplant in mycosis fungoides and sézary syndrome. Biol. Blood Marrow Transpl. 2009, 15, 982–990. [Google Scholar] [CrossRef] [PubMed]
- Gabriel, I.H.; Olavarria, E.; Jones, R.R.; Whittaker, S.; Chaidos, A.; Apperley, J.F. Graft versus lymphoma effect after early relapse following reduced-intensity sibling allogeneic stem cell transplantation for relapsed cytotoxic variant of mycosis fungoides. Bone Marrow Transplant 2007, 40, 401–403. [Google Scholar] [CrossRef]
- Plachouri, K.-M.; Weishaupt, C.; Metze, D.; Evers, G.; Berdel, W.E.; Kempf, W.; Sunderkötter, C.; Stelljes, M. Complete durable remission of a fulminant primary cutaneous aggressive epidermotropic CD8+ cytotoxic T-cell lymphoma after autologous and allogeneic hematopoietic stem cell transplantation. JAAD Case Reports 2017, 3, 196–199. [Google Scholar] [CrossRef]
- Lechowicz, M.J.; Lazarus, H.M.; Carreras, J.; Laport, G.G.; Cutler, C.S.; Wiernik, P.H.; Hale, G.A.; Maharaj, D.; Gale, R.P.; Rowlings, P.A.; et al. Allogeneic hematopoietic cell transplantation for mycosis fungoides and Sezary syndrome. Bone Marrow Transpl. 2014, 49, 1360–1365. [Google Scholar] [CrossRef] [PubMed]
- Gagelmann, N.; Bacigalupo, A.; Rambaldi, A.; Hoelzer, D.; Halter, J.; Sanz, J.; Bonifazi, F.; Meijer, E.; Itälä-Remes, M.; Marková, M.; et al. Haploidentical stem cell transplantation with posttransplant cyclophosphamide therapy vs other donor transplantations in adults with hematologic cancers: A systematic review and meta-analysis. JAMA Oncol. 2019. [Google Scholar] [CrossRef] [PubMed]
- Nakaike, T.; Kato, K.; Oku, S.; Hayashi, M.; Kikushige, Y.; Kuroiwa, M.; Takenaka, K.; Iwasaki, H.; Miyamoto, T.; Teshima, T.; et al. Reduced-intensity conditioning followed by cord blood transplantation in a patient with refractory folliculotropic mycosis fungoides. Int. J. Hematol. 2013, 98, 491–495. [Google Scholar] [CrossRef] [PubMed]
- Tsuji, H.; Wada, T.; Murakami, M.; Kashiwagi, T.; Ito, Y.; Ishida-Yamamoto, A.; Jimbo, J.; Shindo, M.; Sato, K.; Kohgo, Y.; et al. Two cases of mycosis fungoides treated by reduced-intensity cord blood transplantation: Two cases of mycosis fungoides. J. Dermatol. 2010, 37, 1040–1045. [Google Scholar] [CrossRef] [PubMed]
- Lundin, J.; Hagberg, H.; Repp, R.; Cavallin-Ståhl, E.; Fredén, S.; Juliusson, G.; Rosenblad, E.; Tjønnfjord, G.; Wiklund, T.; Österborg, A. Phase 2 study of alemtuzumab (anti-CD52 monoclonal antibody) in patients with advanced mycosis fungoides/Sézary syndrome. Blood 2003, 101, 4267–4272. [Google Scholar] [CrossRef] [PubMed]
- Bernengo, M.G.; Quaglino, P.; Comessatti, A.; Ortoncelli, M.; Novelli, M.; Lisa, F.; Fierro, M.T. Low-dose intermittent alemtuzumab in the treatment of Sézary syndrome: Clinical and immunologic findings in 14 patients. Haematologica 2007, 92, 784–794. [Google Scholar] [CrossRef] [PubMed]
- De Masson, A.; Guitera, P.; Brice, P.; Moulonguet, I.; Mouly, F.; Bouaziz, J.-D.; Battistella, M.; Madelaine, I.; Roux, J.; Ram-Wolff, C.; et al. Long-term efficacy and safety of alemtuzumab in advanced primary cutaneous T-cell lymphomas. Br. J. Dermatol. 2014, 170, 720–724. [Google Scholar] [CrossRef]
- Clark, R.A.; Watanabe, R.; Teague, J.E.; Schlapbach, C.; Tawa, M.C.; Adams, N.; Dorosario, A.A.; Chaney, K.S.; Cutler, C.S.; Leboeuf, N.R.; et al. Skin effector memory T cells do not recirculate and provide immune protection in alemtuzumab-treated CTCL patients. Sci. Transl. Med. 2012, 4, 117ra7. [Google Scholar] [CrossRef]
- Hosoi, H.; Hatanaka, K.; Murata, S.; Mushino, T.; Kuriyama, K.; Nishikawa, A.; Hanaoka, N.; Tamura, S.; Nakakuma, H.; Sonoki, T. Long-term complete remission of early hematological relapse after discontinuation of immunosuppressants following allogeneic transplantation for Sezary syndrome. Hematol. Rep. 2018, 10, 7497. [Google Scholar] [CrossRef]
- Scarisbrick, J.J.; Dignan, F.L.; Tulpule, S.; Gupta, E.D.; Kolade, S.; Shaw, B.; Evison, F.; Shah, G.; Tholouli, E.; Mufti, G.; et al. A multicentre UK study of GVHD following DLI: Rates of GVHD are high but mortality from GVHD is infrequent. Bone Marrow Transpl. 2014, 50, 62–67. [Google Scholar] [CrossRef]
- Moritz, R.K.C.; Ditschkowski, M.; Klemke, C.D.; Terras, S.; Schlaak, M.; Knorr, M.; Theurich, S.; Hegenbart, U.; Kremens, B.; Beelen, D.W.; et al. Allogeneic stem cell transplantation in patients with aggressive primary cutaneous T-cell lymphoma—A case series of the ADF working group “cutaneous lymphomas”. J. Dtsch. Dermatol. Ges. 2014, 12, 39–46. [Google Scholar] [CrossRef] [PubMed]
- Faiz, S.; Rached, H.A.; Forcade, E.; Milpied, N.; Beylot-Barry, M.; Yakoub-Agha, I.; Fegueux, N.; Dereure, O.; Chevallier, P.; Huynh, A.; et al. Donor lymphocyte infusion for primary cutaneous T cell lymphomas: A study from the francophone society of bone marrow transplantation and cellular therapy (SFGM-TC) and the french study group on cutaneous lymphomas (GFLEC). Blood 2018, 132, 3341. [Google Scholar] [CrossRef]
- Schlaak, M.; Kurschat, P.; Shimabukuro-Vornhagen, A.; Scheid, C.; Chemnitz, J.; Stadler, R.; Von Bergwelt-Baildon, M.; Theurich, S. Donor lymphocyte infusions combined with systemic PUVA/bexarotene as an effective bimodal immunologic approach in a patient with relapsed cutaneous T cell lymphoma after allogeneic stem cell transplantation. Transpl. Immunol. 2011, 25, 163–166. [Google Scholar] [CrossRef] [PubMed]
- De Masson, A.; Bouaziz, J.D.; Socié, G. Who needs a skin exam? Skin cancers in allogeneic hematopoietic stem cell transplant recipients in the contemporary era. J. Investig. Dermatol. 2019, 139, 512–514. [Google Scholar] [CrossRef]
- Choi, S.W.; Braun, T.; Chang, L.; Ferrara, J.L.; Pawarode, A.; Magenau, J.M.; Hou, G.; Beumer, J.H.; Levine, J.E.; Goldstein, S.; et al. Vorinostat plus tacrolimus and mycophenolate to prevent graft-versus-host disease after related-donor reduced-intensity conditioning allogeneic haemopoietic stem-cell transplantation: A phase 1/2 trial. Lancet Oncol. 2014, 15, 87–95. [Google Scholar] [CrossRef]
- Shiratori, S.; Fujimoto, K.; Nishimura, M.; Hatanaka, K.C.; Kosugi-Kanaya, M.; Okada, K.; Sugita, J.; Shigematsu, A.; Hashimoto, D.; Endo, T.; et al. Allogeneic hematopoietic stem cell transplantation following reduced-intensity conditioning for mycosis fungoides and Sezary syndrome. Hematol. Oncol. 2016, 34, 9–16. [Google Scholar] [CrossRef]
- Väkevä, L.; Niittyvuopio, R.; Leppä, S.; Heiskanen, J.; Lindström, V.; Ranki, A.; Volin, L. Allogeneic haematopoietic stem cell transplantation for patients with cutaneous T-cell lymphoma. Acta Derm. Venereol. 2014, 96, 816–817. [Google Scholar] [CrossRef]
- Dijk, M.R.C.V.; Sanders, C.J.; Verdonck, L.F.; Fijnheer, R.; Tweel, J.G.V.D. Differential diagnosis of skin lesions after allogeneic haematopoietic stem cell transplantation. Histopathology 2003, 42, 313–330. [Google Scholar] [CrossRef]
- Menter, T.; Medani, H.; Olavarria, E.; Kanfer, E.; Naresh, K.N. Pathology findings in patients with cutaneous T-cell lymphomas treated with allogeneic haematopoietic stem cell transplantation. Br. J. Haematol. 2016, 180, 904–908. [Google Scholar] [CrossRef]
- Herbaux, C.; Merryman, R.; Devine, S.; Armand, P.; Houot, R.; Morschhauser, F.; Haverkos, B. Recommendations for managing PD-1 blockade in the context of allogeneic HCT in Hodgkin lymphoma: Taming a necessary evil. Blood 2018, 132, 9–16. [Google Scholar] [CrossRef]
- Khodadoust, M.S.; Rook, A.H.; Porcu, P.; Foss, F.; Moskowitz, A.J.; Shustov, A.; Shanbhag, S.; Sokol, L.; Fling, S.P.; Ramchurren, N.; et al. Pembrolizumab in relapsed and refractory mycosis fungoides and sézary syndrome: A multicenter phase II study. J. Clin. Oncol. 2020, 38, 20–28. [Google Scholar] [CrossRef] [PubMed]
- El Cheikh, J.; Massoud, R.; Abudalle, I.; Haffar, B.; Mahfouz, R.; Kharfan-Dabaja, M.A.; Jisr, T.; Mougharbel, A.; Ibrahim, A.; Bazarbachi, A. Nivolumab salvage therapy before or after allogeneic stem cell transplantation in Hodgkin lymphoma. Bone Marrow Transpl. 2017, 52, 1074–1077. [Google Scholar] [CrossRef] [PubMed]
- Scarfò, I.; Frigault, M.J.; Maus, M.V. CAR-based approaches to cutaneous T-cell lymphoma. Front. Oncol. 2019, 9, 259. [Google Scholar] [CrossRef] [PubMed]
| Ref. | Number of Patients | Median Age (Range) | Pathology | Source of Stem Cells | Conditioning Regimen | Outcomes | Complications | Median Follow-Up |
|---|---|---|---|---|---|---|---|---|
| Duarte et al., J Clin Oncol, 2010 and 2014 [11,17] | 60 | 44 (13–63) | MF = 36 SS = 24 | PB = 50 BM = 10 | MAC = 16 RIC = 44 | 7 year, OS 44% PFS 30% | 7 year, Relapse 45% NRM 22% | 7 years |
| Duvic et al., J Clin Oncol, 2010 [23] | 19 | 50 (21–63) | MF = 5 SS = 14 | PB = 14 BM = 5 | NMA = 19 | 2 year, OS 79% PFS 53% | 1.7 year, Relapse 37% NRM 21% | 1.7 years |
| De Masson et al., Haematologica, 2014 [15] | 37 | 44 (9–63) | MF = 26 SS = 55 Others = 6 | PB = 32 BM = 5 CB = 2 | MAC = 12 RIIC = 25 | 2 year, OS 57% PFS 31% | 2 year, Relapse 56% NRM 18% | 2.4 years |
| Lechowicz et al., Bone Marrow Transplant, 2014 [42] | 129 | 48 (22–72) | Not available | PB = 107 BM = 18 CB = 4 | MAC = 46 RIC/NMA = 83 | 5 year, OS 32% PFS 17% | 5 year, Relapse 61% 2 year, NRM 22% | 3.3 years |
| Hosing et al., Ann Oncol, 2015 [24] | 47 | 51.5 (19–72) | MF = 34 SS = 13 | PB = 35 BM = 12 | MAC = 3 NMA = 2 RIC = 42 | 4 year, OS 51% PFS 26% | 2 year, Relapse 50% NRM 17% | 2 years |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dumont, M.; Peffault de Latour, R.; Ram-Wolff, C.; Bagot, M.; de Masson, A. Allogeneic Hematopoietic Stem Cell Transplantation in Cutaneous T-Cell Lymphomas. Cancers 2020, 12, 2856. https://doi.org/10.3390/cancers12102856
Dumont M, Peffault de Latour R, Ram-Wolff C, Bagot M, de Masson A. Allogeneic Hematopoietic Stem Cell Transplantation in Cutaneous T-Cell Lymphomas. Cancers. 2020; 12(10):2856. https://doi.org/10.3390/cancers12102856
Chicago/Turabian StyleDumont, Maëlle, Régis Peffault de Latour, Caroline Ram-Wolff, Martine Bagot, and Adèle de Masson. 2020. "Allogeneic Hematopoietic Stem Cell Transplantation in Cutaneous T-Cell Lymphomas" Cancers 12, no. 10: 2856. https://doi.org/10.3390/cancers12102856
APA StyleDumont, M., Peffault de Latour, R., Ram-Wolff, C., Bagot, M., & de Masson, A. (2020). Allogeneic Hematopoietic Stem Cell Transplantation in Cutaneous T-Cell Lymphomas. Cancers, 12(10), 2856. https://doi.org/10.3390/cancers12102856





