Outcomes of Capecitabine and Temozolomide (CAPTEM) in Advanced Neuroendocrine Neoplasms (NENs)
Abstract
1. Introduction
2. Methods
3. Results
4. Discussion
5. Limitations
6. Conclusions
Author Contributions
Funding
Conflicts of Interest
Disclosures
References
- Kruljac, I.; Pape, U. The classification of neuroendocrine neoplasms: “Neuroendocrine carcinomas” revisited—A 2017 update and future perspectives. Endocr. Oncol. Metab. 2017, 3, 37–42. [Google Scholar]
- Mahjoub, A.R.; O’Reilly, E.M. Emerging therapies for pancreas neuroendocrine cancers. Chin. Clin. Oncol. 2013, 2, e23. [Google Scholar]
- Vélayoudom-Céphise, F.; Duvillard, P.; Foucan, L.; Hadoux, J.; Chougnet, C.N.; Leboulleux, S.; Malka, D.; Guigay, J.; Goere, D.; Debaere, T. Are G3 ENETS neuroendocrine neoplasms heterogeneous? Endocr. Relat. Cancer 2013, 20, 649–657. [Google Scholar] [CrossRef]
- Bosman, F.T.; Carneiro, F.; Hruban, R.H.; Theise, N.D. WHO Classification of Tumours of the Digestive System., 4th ed.; World Health Organization: Geneva, Switzerland, 2010; p. 417. [Google Scholar]
- National Comprehensive Cancer Network. NCCN Clinical Practice Guidelines in Oncology Neuroendocrine Tumors. Available online: https://www.nccn.org/professionals/physician_gls/pdf/neuroendocrine_blocks.pdf (accessed on 4 December 2019).
- Kaltsas, G.A.; Besser, G.M.; Grossman, A.B. The diagnosis and medical management of advanced neuroendocrine tumors. Endocr. Rev. 2004, 25, 458–511. [Google Scholar] [CrossRef]
- Modlin, I.M.; Öberg, K.; Chung, D.C.; Jensen, R.T.; de Herder, W.W.; Thakker, R.V.; Caplin, M.; Delle Fave, G.; Kaltsas, G.A.; Krenning, E.P. Gastroenteropancreatic neuroendocrine tumours. Lancet Oncol. 2008, 9, 61–72. [Google Scholar] [CrossRef]
- Sorbye, H.; Welin, S.; Langer, S.W.; Vestermark, L.W.; Holt, N.; Osterlund, P.; Dueland, S.; Hofsli, E.; Guren, M.G.; Ohrling, K. Predictive and prognostic factors for treatment and survival in 305 patients with advanced gastrointestinal neuroendocrine carcinoma (WHO G3): The NORDIC NEC study. Ann. Oncol. 2012, 24, 152–160. [Google Scholar] [CrossRef] [PubMed]
- Ahlman, H.; Nilsson, O.; McNicol, A.M.; Ruszniewski, P.; Niederle, B.; Ricke, J.; Jensen, R.; Kos-Kudła, B.; Öberg, K.; O’Connor, J.M. Poorly-differentiated endocrine carcinomas of midgut and hindgut origin. Neuroendocrinology 2008, 87, 40–46. [Google Scholar] [CrossRef] [PubMed]
- Nilsson, O.; Van Cutsem, E.; Delle Fave, G.; Yao, J.C.; Pavel, M.E.; McNicol, A.M.; Garcia, M.S.; Knapp, W.H.; Keleştimur, F.; Sauvanet, A. Poorly differentiated carcinomas of the foregut (gastric, duodenal and pancreatic). Neuroendocrinology 2006, 84, 212–215. [Google Scholar] [CrossRef]
- Mitry, E.; Baudin, E.; Ducreux, M.; Sabourin, J.C.; Rufie, P.; Aparicio, T.; Aparicio, T.; Lasser, P.; Elias, D.; Duvillard, P.; et al. Treatment of poorly differentiated neuroendocrine tumours with etoposide and cisplatin. Br. J. Cancer 1999, 81, 1351–1355. [Google Scholar] [CrossRef]
- Spada, F.; Antonuzzo, L.; Marconcini, R.; Gelsomino, F.; Fumagalli, C.; Messerini, L.; Di Costanzo, F.; Ricci, S.; Luppi, G.; Frezza, A.M.; et al. Chemotherapy with capecitabine plus temozolomide (CAP-TEM) in patients with advanced neuroendocrine neoplasms (NENs): An italian multicenter retrospective analysis. J. Clin. Oncol. 2015, 33, e15174. [Google Scholar] [CrossRef]
- Fazio, N.; Spada, F.; Giovannini, M. Chemotherapy in gastroenteropancreatic (GEP) neuroendocrine carcinomas (NEC): A critical view. Cancer Treat. Rev. 2013, 39, 270–274. [Google Scholar] [CrossRef] [PubMed]
- Johnson, L.A.; Lavin, P.; Moertel, C.G.; Weiland, L.; Dayal, Y.; Doos, W.G.; Geller, S.A.; Cooper, H.S.; Nime, F.; Masse, S. Carcinoids: The association of histologic growth pattern and survival. Cancer 1983, 51, 882–889. [Google Scholar] [CrossRef]
- Auernhammer, C.J.; Göke, B. Therapeutic strategies for advanced neuroendocrine carcinomas of jejunum/ileum and pancreatic origin. Gut 2011, 60, 1009–1021. [Google Scholar] [CrossRef] [PubMed]
- Chan, J.A.; Kulke, M.H. New treatment options for patients with advanced neuroendocrine tumors. Curr. Treat Options Oncol. 2011, 12, e136. [Google Scholar] [CrossRef]
- Fine, R.L.; Gulati, A.P.; Tsushima, D.; Mowatt, K.B.; Oprescu, A.; Bruce, J.N.; Chabot, J.A. Prospective phase II study of capecitabine and temozolomide (CAPTEM) for progressive, moderately, and well-differentiated metastatic neuroendocrine tumors. J. Clin. Oncol. 2014, 32, e179. [Google Scholar] [CrossRef]
- Cros, J.; Hentic, O.; Rebours, V.; Zappa, M.; Gille, N.; Theou-Anton, N.; Vernerey, D.; Maire, F.; Levy, P.; Bedossa, P. MGMT expression predicts response to temozolomide in pancreatic neuroendocrine tumors. Endocr. Relat. Cancer 2016, 23, 625–633. [Google Scholar] [CrossRef]
- Kotteas, E.A.; Syrigos, K.N.; Saif, M.W. Profile of capecitabine/temozolomide combination in the treatment of well-differentiated neuroendocrine tumors. Onco. Targets Ther. 2016, 9, e699. [Google Scholar]
- Strosberg, J.R.; Fine, R.L.; Choi, J.; Nasir, A.; Coppola, D.; Chen, D.; Helm, J.; Kvols, L. First-line chemotherapy with capecitabine and temozolomide in patients with metastatic pancreatic endocrine carcinomas. Cancer 2011, 117, 268–275. [Google Scholar] [CrossRef]
- Kunz, P.L.; Catalano, P.J.; Nimeiri, H.; Fisher, G.A.; Longacre, T.A.; Suarez, C.J.; Yao, J.C.; Kulke, M.H.; Hendifar, A.E.; Shanks, J.C.; et al. A randomized study of temozolomide or temozolomide and capecitabine in patients with advanced pancreatic neuroendocrine tumors: A trial of the ECOG-ACRIN cancer research group (E2211). J. Clin. Oncol. 2018, 36, e4004. [Google Scholar] [CrossRef]
- Ramirez, R.A.; Beyer, D.T.; Chauhan, A.; Boudreaux, J.P.; Wang, Y.; Woltering, E.A. The role of capecitabine/temozolomide in metastatic neuroendocrine tumors. Oncologist 2016, 21, 671–675. [Google Scholar] [CrossRef]
- Dasari, A.; Shen, C.; Halperin, D.; Zhao, B.; Zhou, S.; Xu, Y.; Shih, T.; Yao, J.C. Trends in the incidence, prevalence, and survival outcomes in patients with neuroendocrine tumors in the United States. JAMA Oncol. 2017, 3, 1335–1342. [Google Scholar] [CrossRef] [PubMed]
- Owen, D.H.; Alexander, A.J.; Konda, B.; Wei, L.; Hemminger, J.A.; Schmidt, C.R.; Abdel-Misih, S.R.; Dillhoff, M.E.; Sipos, J.A.; Kirschner, L.S. Combination therapy with capecitabine and temozolomide in patients with low and high grade neuroendocrine tumors, with an exploratory analysis of O6-methylguanine DNA methyltransferase as a biomarker for response. Oncotarget 2017, 8, e104046. [Google Scholar] [CrossRef] [PubMed]
- Peixoto, R.D.; Noonan, K.L.; Pavlovich, P.; Kennecke, H.F.; Lim, H.J. Outcomes of patients treated with capecitabine and temozolamide for advanced pancreatic neuroendocrine tumors (PNETs) and non-PNETs. J. Gastrointest. Oncol. 2014, 5, e247. [Google Scholar] [CrossRef]
- Murakami, J.; Lee, Y.; Kokeguchi, S.; Tsujigiwa, H.; Asaumi, J.; Nagatsuka, H.; Fukui, K.; Kuroda, M.; Tanaka, N.; Matsubara, N. Depletion of O6-methylguanine-DNA methyltransferase by O6-benzylguanine enhances 5-FU cytotoxicity in colon and oral cancer cell lines. Oncol. Rep. 2007, 17, 1461–1467. [Google Scholar] [CrossRef]
- Kulke, M.H.; Hornick, J.L.; Frauenhoffer, C.; Hooshmand, S.; Ryan, D.P.; Enzinger, P.C.; Meyerhardt, J.A.; Clark, J.W.; Stuart, K.; Fuchs, C.S. O6-methylguanine DNA methyltransferase deficiency and response to temozolomide-based therapy in patients with neuroendocrine tumors. Clin. Cancer Res. 2009, 15, 338–345. [Google Scholar] [CrossRef]
- Moertel, C.G.; Kvols, L.K.; O’Connell, M.J.; Rubin, J. Treatment of neuroendocrine carcinomas with combined etoposide and cisplatin. evidence of major therapeutic activity in the anaplastic variants of these neoplasms. Cancer 1991, 68, 227–232. [Google Scholar] [CrossRef]
- Sahu, A.; Jefford, M.; Lai-Kwon, J.; Thai, A.; Hicks, R.J.; Michael, M. CAPTEM in metastatic well-differentiated intermediate to high grade neuroendocrine tumors: A single centre experience. J. Oncol. 2019, 2019, e9032753. [Google Scholar] [CrossRef]
- Chatzellis, E.; Angelousi, A.; Daskalakis, K.; Tsoli, M.; Alexandraki, K.I.; Wachuła, E.; Meirovitz, A.; Maimon, O.; Grozinsky-Glasberg, S.; Gross, D. Activity and safety of standard and prolonged capecitabine/temozolomide administration in patients with advanced neuroendocrine neoplasms. Neuroendocrinology 2019, 109, 333–345. [Google Scholar] [CrossRef]
- De Mestier, L.; Walter, T.; Brixi, H.; Evrard, C.; Legoux, J.; de Boissieu, P.; Hentic, O.; Cros, J.; Hammel, P.; Tougeron, D. Comparison of temozolomide-capecitabine to 5-fluorouracile-dacarbazine in 247 patients with advanced digestive neuroendocrine tumors using propensity score analyses. Neuroendocrinology 2019, 108, 343–353. [Google Scholar] [CrossRef]
- Al-Toubah, T.; Morse, B.; Strosberg, J. Capecitabine and temozolomide in advanced lung neuroendocrine neoplasms. Oncologist 2019. [Google Scholar] [CrossRef]
- Wang, X.; Li, Y.; Duan, J.; Chen, Y.; Yuan, B.; Qi, Z.; Tan, H. Capecitabine and temozolomide as a promising therapy for advanced thymic atypical carcinoid. Oncologist 2019, 24, 798–802. [Google Scholar] [CrossRef] [PubMed]
- Isacoff, W.H.; Moss, R.A.; Pecora, A.L.; Fine, R.L. Temozolomide/capecitabine therapy for metastatic neuroendocrine tumors of the pancreas. a retrospective review. J. Clin. Oncol. 2006, 24, e14023. [Google Scholar]
- Saif, M.W.; Kaley, K.; Brennan, M.; Garcon, M.C.; Rodriguez, G.; Rodriguez, T. A retrospective study of capecitabine/temozolomide (CAPTEM) regimen in the treatment of metastatic pancreatic neuroendocrine tumors (pNETs) after failing previous therapy. J. Pancreas 2013, 14, 498–501. [Google Scholar]
- La Salvia, A.; Brizzi, M.P.; Muratori, L.; Trevisi, E.; Di Maio, M.; Scagliotti, G.V. Capecitabine plus temozolomide in well-or moderately-differentiated primary atypical neuroendocrine tumours—Single-centre experience of two cases. Endokrynol. Pol. 2019, 70, 380–383. [Google Scholar] [CrossRef] [PubMed]
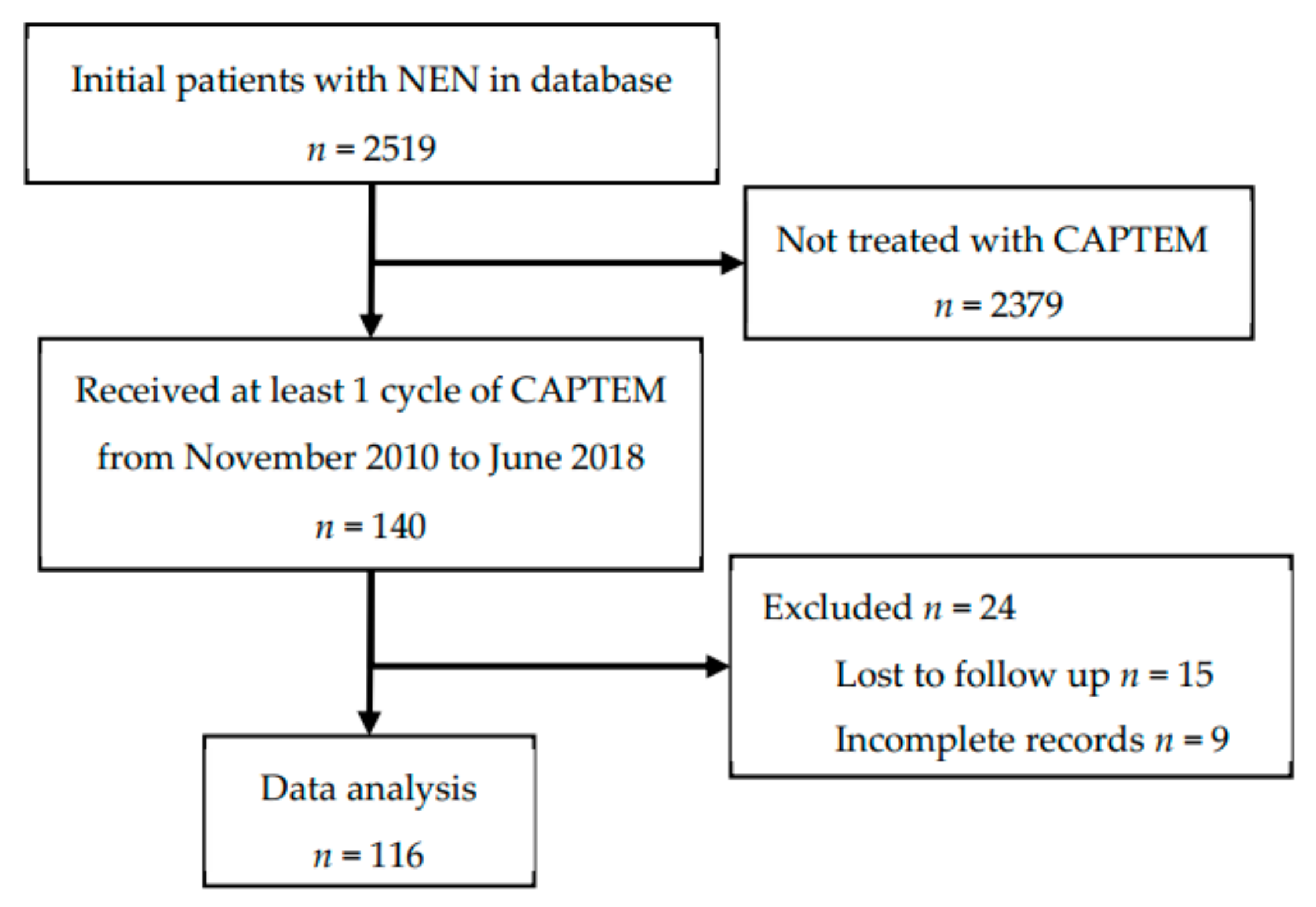
| Characteristic | n (%) |
|---|---|
| Sex | |
| Female | 48 (41) |
| Male | 68 (59) |
| Tumor Differentiation | |
| Poor | 18 (16) |
| Well | 94 (81) |
| Unknown | 4 (3) |
| Primary Tumor Site | |
| Pancreas | 47 (41) |
| Small Intestines | 37 (32) |
| Unknown Primary Site | 12 (10) |
| Lung | 12 (10) |
| Colon/Rectum | 7 (6) |
| Kidney | 1 (1) |
| Metastatic Disease | |
| None | 3 (3) |
| Regional Lymph Node | 4 (3) |
| Hepatic only | 59 (51) |
| Extrahepatic only | 6 (5) |
| Hepatic & Extrahepatic | 44 (38) |
| Prior Treatment | |
| None | 11 (9) |
| Somatostatin Analogs | 54 (47) |
| Surgery | 67 (58) |
| Chemotherapy | 36 (31) |
| Liver Directed Therapy | 35 (30) |
| Targeted Therapy | 19 (16) |
| Radionuclide therapy | 4 (4) |
| Radiation Therapy | 4 (4) |
| Treatment Lines Prior to CAPTEM | |
| 1 | 34 (30) |
| 2 | 43 (37) |
| 3 | 22 (19) |
| 4 | 12 (10) |
| 5 | 5 (4) |
| Tumor Characteristics | Radiographic Response, n (%) | ORR | DCR | ||||||
|---|---|---|---|---|---|---|---|---|---|
| n | CR | PR | SD | PD | n (%) | p Value | n (%) | p Value | |
| All patients | 116 | 1 (1) | 23 (20) | 61 (53) | 31 (27) | 24 (21) | 85 (73) | ||
| Primary Site (n = 116) | |||||||||
| pNEN | 47 | 1 (2) | 17 (36) | 18 (38) | 11 (23) | 18 (38) | 0.0001 | 36 (77) | 0.5049 |
| Non-pNEN | 69 | - | 6 (9) | 43 (62) | 20 (29) | 6 (9) | 49 (71) | ||
| Ki-67 (n = 106) * | |||||||||
| Ki-67 < 3% | 24 | - | 5 (21) | 17 (71) | 2 (8) | 5 (21) | 0.5241 | 22 (92) | 0.0084 |
| Ki-67 3–20% | 50 | 1 (2) | 12 (24) | 24 (48) | 13 (26) | 13 (26) | 37 (74) | ||
| Ki-67 20–55% | 26 | - | 3 (12) | 12 (46) | 11 (42) | 3 (12) | 15 (58) | ||
| Ki-67 > 55% | 6 | - | 1 (17) | 1 (17) | 3 (50) | 1 (17) | 2 (33) | ||
| Prior Treatments (n = 109) | |||||||||
| No SSA | 58 | 0 (0) | 12 (21) | 32 (55) | 14 (24) | 12 (21) | 0.6877 | 44 (76) | 0.6928 |
| SSA | 51 | 1 (2) | 8 (16) | 28 (55) | 14 (27) | 9 (18) | 37 (73) | ||
| No Surgery | 47 | 0 (0) | 11 (23) | 22 (47) | 15 (32) | 11 (23) | 0.6487 | 33 (70) | 0.2195 |
| Surgery | 62 | 1 (2) | 11 (18) | 37 (60) | 13 (21) | 12 (19) | 49 (79) | ||
| No Chemotherapy | 75 | 1 (1) | 14 (19) | 45 (60) | 15 (20) | 15 (20) | 0.6020 | 60 (80) | 0.0666 |
| Chemotherapy | 34 | 0 (0) | 6 (18) | 17 (50) | 1 (3) | 6 (18) | 23 (68) | ||
| No Targeted Therapy | 91 | 1 (1) | 16 (18) | 52 (57) | 22 (24) | 17 (19) | 0.8400 | 69 (76) | 0.7463 |
| Targeted Therapy | 18 | 0 (0) | 3 (17) | 10 (56) | 5 (28) | 3 (17) | 13 (72) | ||
| No PRRT | 106 | 1 (1) | 20 (19) | 58 (55) | 27 (25) | 21 (19) | 0.3909 | 79 (75) | 0.3135 |
| PRRT | 3 | 0 (0) | 0 (0) | 3 (100) | 0 (0) | 0 (0) | 3 (100) | ||
| Toxicity | All Grades, n (%) | Grade 1 | Grade 2 | Grade 3 | Grade 4 |
|---|---|---|---|---|---|
| Anemia | 8 (7) | - | 3 | 5 | - |
| Thrombocytopenia | 11 (9) | - | 2 | 6 | 3 |
| Lymphopenia | 8 (7) | - | 3 | 5 | - |
| Neutropenia | 6 (5) | - | 1 | 4 | 1 |
| Fatigue | 26 (22) | 7 | 12 | 6 | 1 |
| Nausea/Vomiting | 30 (26) | 10 | 14 | 5 | 1 |
| Diarrhea | 10 (9) | 3 | 3 | 2 | 2 |
| PPE | 13 (11) | 1 | 4 | 7 | 1 |
| Weight loss | 3 (3) | 3 | - | - | - |
| Other | 14 (12) | 7 | 2 | 3 | 2 |
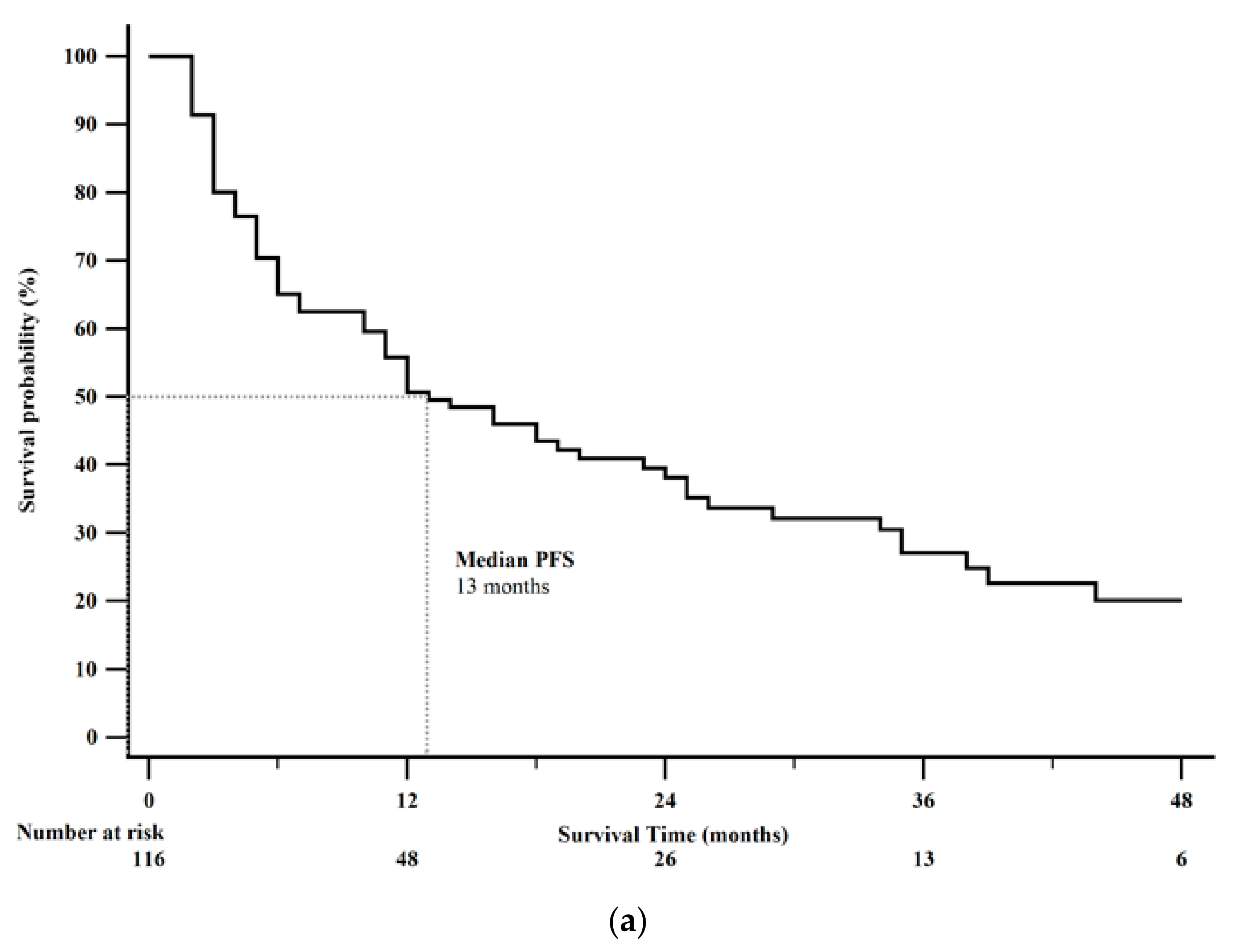
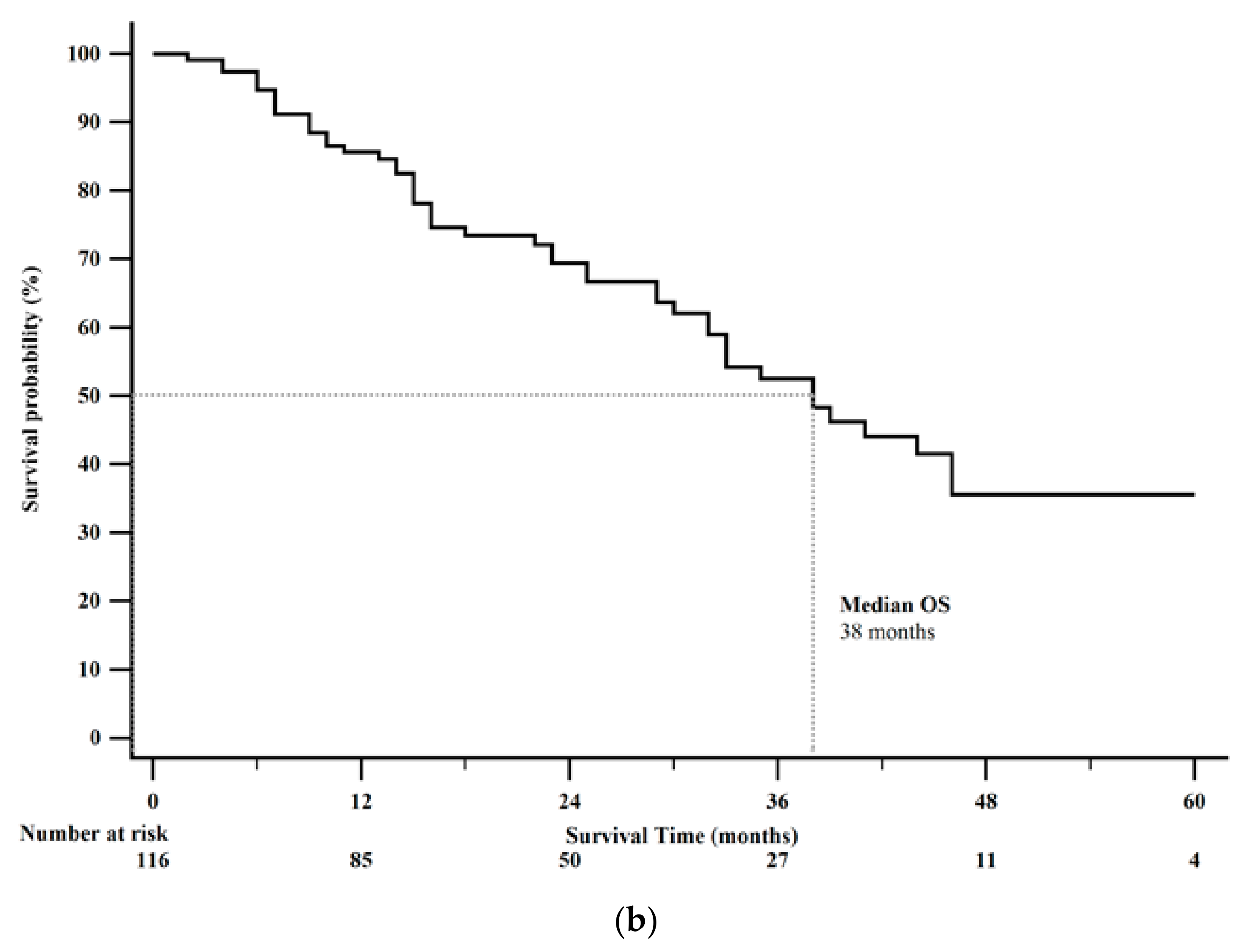
| Factors | n | Median PFS (95% CI) | p Value (Log-Rank) | Median OS (95% CI) | p Value (Log-Rank) |
|---|---|---|---|---|---|
| All patients | 116 | 13 (11–23) | 38 (32–46) | ||
| Sex | 0.1713 | 0.1166 | |||
| Female | 48 | 25 (10–35) | NR | ||
| Male | 68 | 12 (7–18) | 33 (29–41) | ||
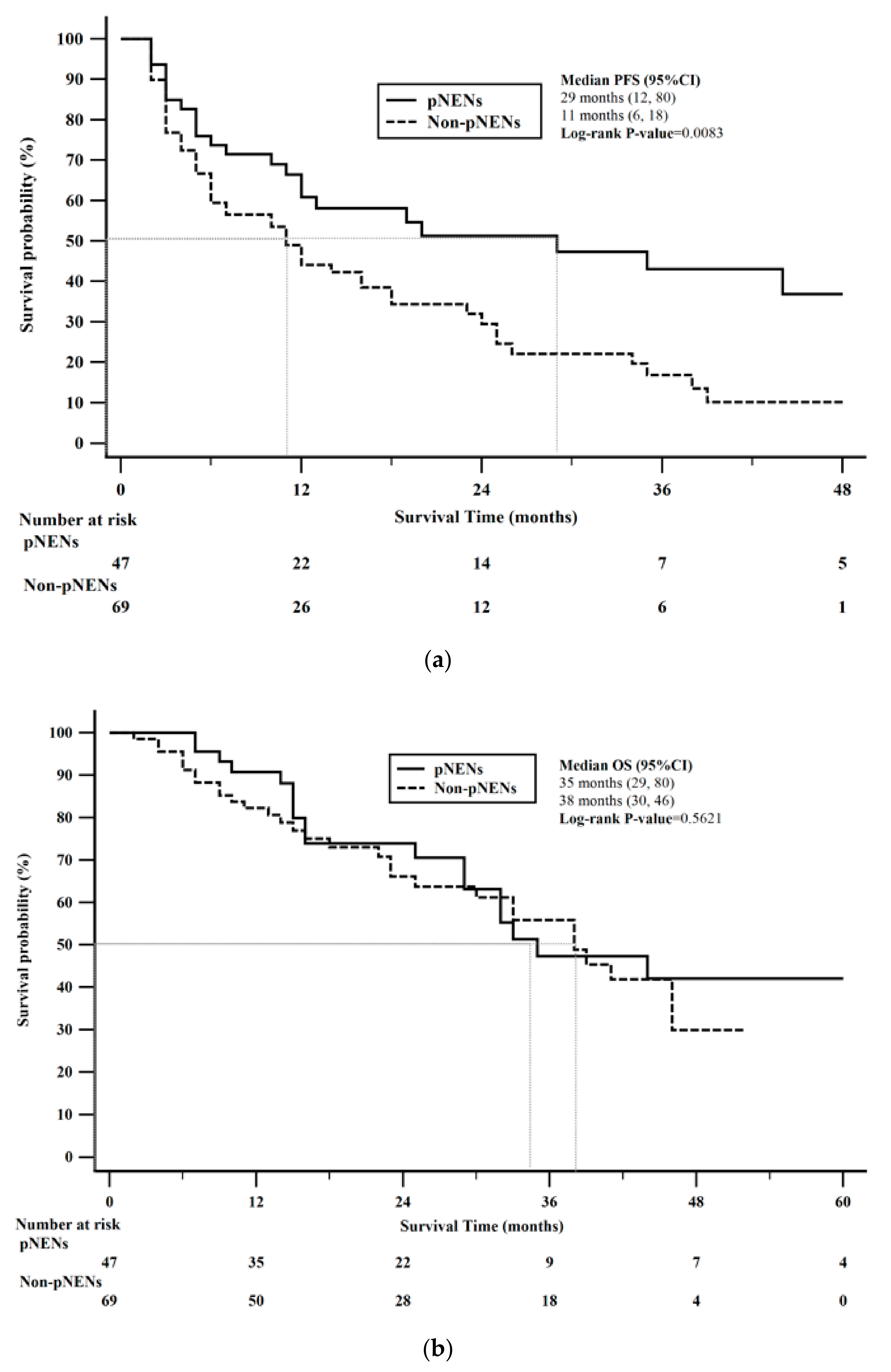
| Primary Site (n = 116) | 0.0083 | 0.5621 | |||
| pNEN | 47 | 29 (12–80) | 35 (29–80) | ||
| Non-pNEN | 69 | 11 (6–18) | 38 (30–46) | ||
| Tumor Differentiation (n = 112) | 0.0192 | 0.0192 | |||
| Well | 94 | 16 (11–26) | 41 (33–46) | ||
| Poor | 18 | 5 (5–12) | 23 (13–25) | ||
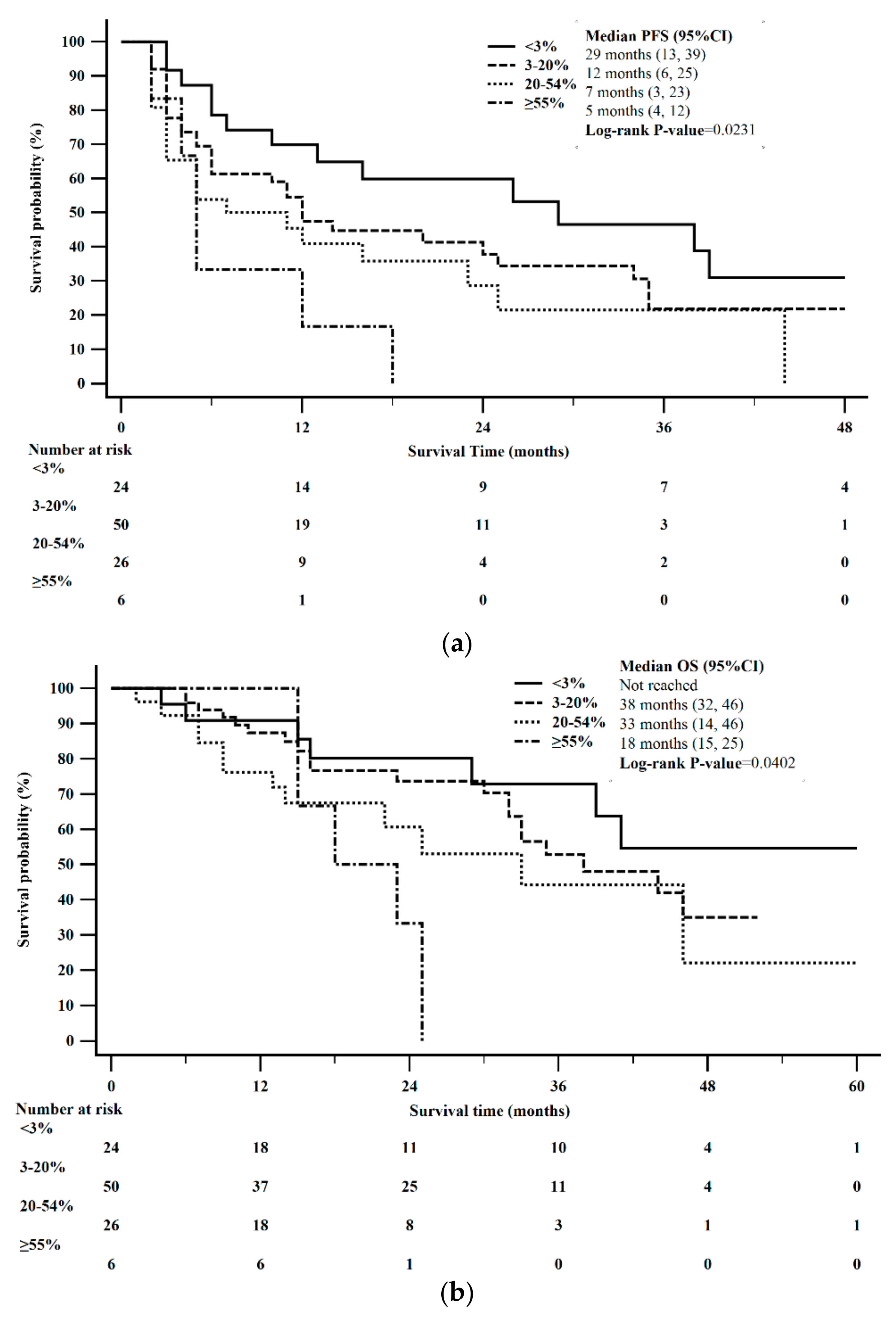
| Ki-67 (n = 106) * | 0.0231 | 0.0402 | |||
| Ki-67 < 3% | 24 | 29 (13–39) | NR | ||
| Ki-67 3–20% | 50 | 12 (6–25) | 33 (30–46) | ||
| Ki-67 20–55% | 26 | 7 (3–25) | 33 (14–46) | ||
| Ki-67 > 55% | 6 | 5 (4–12) | 18 (15–25) | ||
| Tumor Characteristics | Univariate Analysis | Multivariate Analysis † | ||
|---|---|---|---|---|
| HR (95% CI) | p Value | HR (95% CI) | p Value | |
| Primary Site | ||||
| PNEN | 1 | 1 | ||
| Non-PNEN | 1.89 (1.15–3.11) | 0.0116 | 2.04 (1.20–3.47) | 0.0088 |
| Tumor Differentiation | ||||
| Well | 1 | 1 | ||
| Poor | 1.92 (1.08–3.41) | 0.0263 | 1.07 (0.48–2.37) | 0.8682 |
| Ki-67 * | ||||
| Ki-67 < 3% | 1 | 1 | ||
| Ki-67 3–20% | 1.62 (0.84–3.09) | 0.1477 | 11.35 (2.42–53.29) | 0.0021 |
| Ki-67 > 20% | 2.36 (1.20–4.65) | 0.0129 | 7.76 (0.60–100.50) | 0.1167 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Thomas, K.; Voros, B.A.; Meadows-Taylor, M.; Smeltzer, M.P.; Griffin, R.; Boudreaux, J.P.; Thiagarajan, R.; Woltering, E.A.; Ramirez, R.A. Outcomes of Capecitabine and Temozolomide (CAPTEM) in Advanced Neuroendocrine Neoplasms (NENs). Cancers 2020, 12, 206. https://doi.org/10.3390/cancers12010206
Thomas K, Voros BA, Meadows-Taylor M, Smeltzer MP, Griffin R, Boudreaux JP, Thiagarajan R, Woltering EA, Ramirez RA. Outcomes of Capecitabine and Temozolomide (CAPTEM) in Advanced Neuroendocrine Neoplasms (NENs). Cancers. 2020; 12(1):206. https://doi.org/10.3390/cancers12010206
Chicago/Turabian StyleThomas, Katharine, Brianne A. Voros, Meghan Meadows-Taylor, Matthew P. Smeltzer, Ryan Griffin, J. Philip Boudreaux, Ramcharan Thiagarajan, Eugene A. Woltering, and Robert A. Ramirez. 2020. "Outcomes of Capecitabine and Temozolomide (CAPTEM) in Advanced Neuroendocrine Neoplasms (NENs)" Cancers 12, no. 1: 206. https://doi.org/10.3390/cancers12010206
APA StyleThomas, K., Voros, B. A., Meadows-Taylor, M., Smeltzer, M. P., Griffin, R., Boudreaux, J. P., Thiagarajan, R., Woltering, E. A., & Ramirez, R. A. (2020). Outcomes of Capecitabine and Temozolomide (CAPTEM) in Advanced Neuroendocrine Neoplasms (NENs). Cancers, 12(1), 206. https://doi.org/10.3390/cancers12010206




