Cortactin: A Major Cellular Target of the Gastric Carcinogen Helicobacter pylori
Abstract
1. Introduction
2. Cortactin Activity Depends on Its Phosphorylation States and Is Involved in Tumor Development
3. H. pylori Virulence Factors
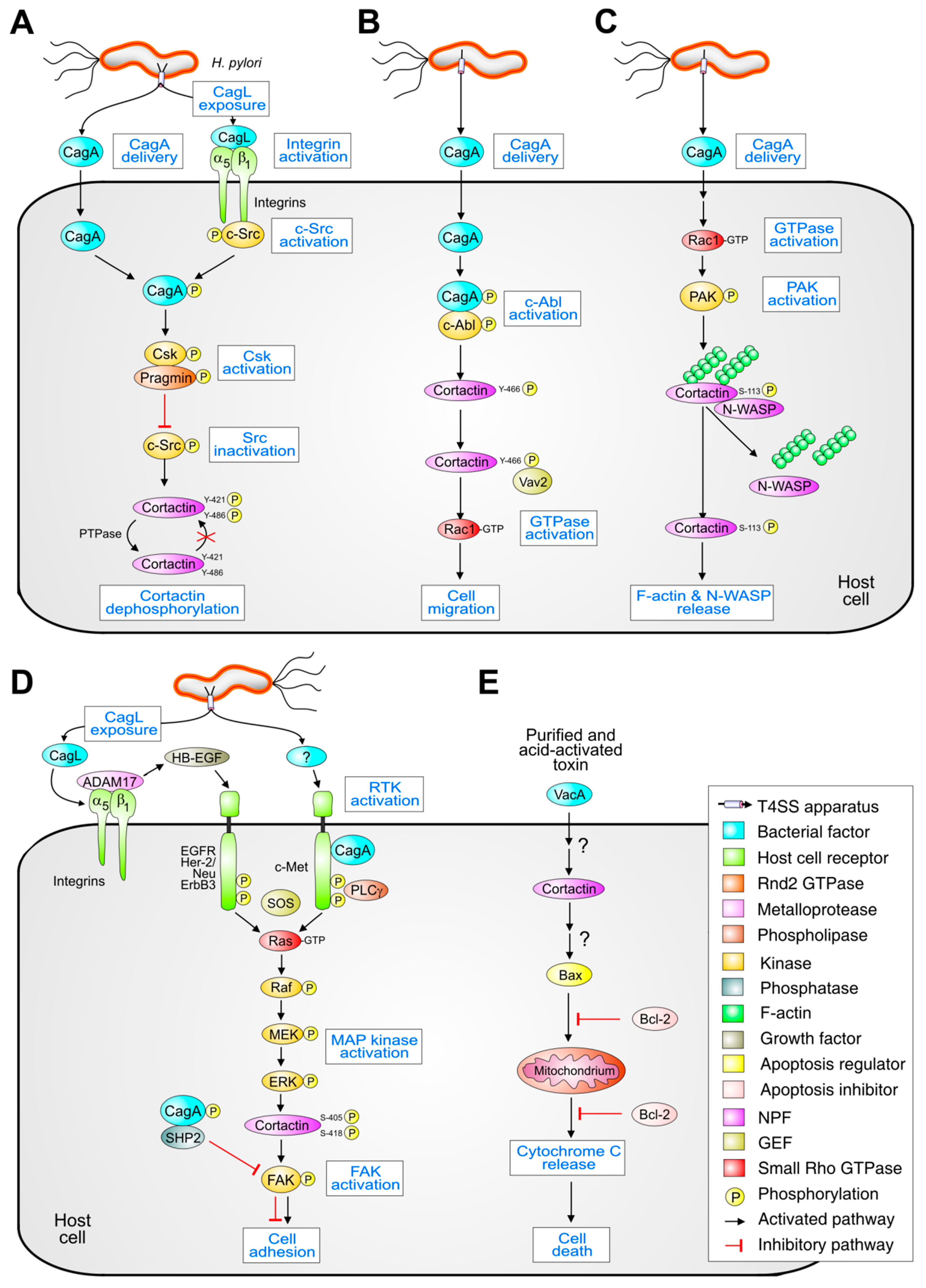
4. H. pylori Affects Host Kinases that Control Cortactin
5. Role of Tyrosine-Phosphorylated Cortactin Generated by H. pylori
6. Function of Serine-Phosphorylated Cortactin by H. pylori
7. Role of Cortactin in VacA-Induced Apoptosis
8. Potential Role of Cortactin in Podosome and Invadopodia Formation
9. Conclusions and Perspectives
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| ABA | F-actin binding assay |
| APA | Actin polymerization assay |
| AR | Autoradiography |
| CCAA | Cell–Cell Adhesion Assays |
| CIA | Collagen invasion assay |
| CLSM | Confocal Laser Scanning Microscope |
| CMA | Cell motility assay |
| EM | Electron microscopy |
| FRET | Fluorescence Resonance Energy Transfer assay |
| GEF AA | GEF activity assay |
| GST-BA | GST-binding assay |
| ICC | Immunocytochemistry |
| IFM | Immunofluorescence Microscopy |
| IHC | Immunohistochemistry |
| In vitro | In vitro actin branching assay |
| In vitro KA | In vitro Kinase Assay |
| IP | Immunoprecipitation |
| IPA | Intestinal permeability assay |
| KA | Kymograph Analysis |
| LSM | Laser Scanning Microscope |
| MDA | Matrix degradation assay |
| MS | Mass Spectroscopy |
| NB | Northern Blot |
| PA | Proliferation assay |
| PALM | Single-Molecule Photoactivated Localization Microscopy |
| STORM | Stochastic Optical Reconstruction Microscopy |
| SMA | Synapse Morphology Analysis |
| WB | Western Blotting |
| YTHA | Yeast Two-Hybrid Analyses |
References
- Salama, N.R.; Hartung, M.L.; Muller, A. Life in the human stomach: Persistence strategies of the bacterial pathogen Helicobacter pylori. Nat. Rev. Microbiol. 2013, 11, 385–399. [Google Scholar] [CrossRef]
- Yamaoka, Y.; Graham, D.Y. Helicobacter pylori virulence and cancer pathogenesis. Future Oncol. 2014, 10, 1487–1500. [Google Scholar] [CrossRef] [PubMed]
- Amieva, M.; Peek, R.M. Pathobiology of Helicobacter pylori-induced gastric cancer. Gastroenterology 2016, 150, 64–78. [Google Scholar] [CrossRef] [PubMed]
- Hatakeyama, M. Malignant Helicobacter pylori-associated diseases: Gastric cancer and MALT lymphoma. Adv. Exp. Med. Biol. 2019, 1149, 135–149. [Google Scholar] [CrossRef] [PubMed]
- Tegtmeyer, N.; Wessler, S.; Necchi, V.; Rohde, M.; Harrer, A.; Rau, T.T.; Asche, C.I.; Boehm, M.; Loessner, H.; Figueiredo, C.; et al. Helicobacter pylori employs a unique basolateral type IV secretion mechanism for CagA delivery. Cell Host Microbe 2017, 22, 552–560.e5. [Google Scholar] [CrossRef]
- Kusters, J.G.; van Vliet, A.H.M.; Kuipers, E.J. Pathogenesis of Helicobacter pylori infection. Clin. Microbiol. Rev. 2006, 19, 449–490. [Google Scholar] [CrossRef]
- Wallasch, C.; Crabtree, J.E.; Bevec, D.; Robinson, P.A.; Wagner, H.; Ullrich, A. Helicobacter pylori-stimulated EGF receptor transactivation requires metalloprotease cleavage of HB-EGF. Biochem. Biophys. Res. Commun. 2002, 295, 695–701. [Google Scholar] [CrossRef]
- Keates, S.; Sougioultzis, S.; Keates, A.C.; Zhao, D.Z.; Peek, R.M.; Shaw, L.M.; Kelly, C.P. cag+ Helicobacter pylori induce transactivation of the epidermal growth factor receptor in AGS gastric epithelial cells. J. Biol. Chem. 2001, 276, 48127–48134. [Google Scholar] [CrossRef]
- Ashktorab, H.; Daremipouran, M.; Wilson, M.; Siddiqi, S.; Lee, E.L.; Rakhshani, N.; Malekzadeh, R.; Johnson, A.C.; Hewitt, S.M.; Smoot, D.T. Transactivation of the EGFR by AP-1 is induced by Helicobacter pylori in gastric cancer. Am. J. Gastroenterol. 2007, 102, 2135–2146. [Google Scholar] [CrossRef]
- Hartung, M.L.; Gruber, D.C.; Koch, K.N.; Gruter, L.; Rehrauer, H.; Tegtmeyer, N.; Backert, S.; Muller, A.H. H. pylori-induced DNA strand breaks are introduced by nucleotide excision repair endonucleases and promote NF-κB target gene expression. Cell Rep. 2015, 13, 70–79. [Google Scholar] [CrossRef]
- Naumann, M.; Sokolova, O.; Tegtmeyer, N.; Backert, S. Helicobacter pylori: A paradigm pathogen for subverting host cell signal transmission. Trends Microbiol. 2017, 25, 316–328. [Google Scholar] [CrossRef] [PubMed]
- Costa, L.; Corre, S.; Michel, V.; Le Luel, K.; Fernandes, J.; Ziveri, J.; Jouvion, G.; Danckaert, A.; Mouchet, N.; Da Silva Barreira, D.; et al. USF1 defect drives p53 degradation during Helicobacter pylori infection and accelerates gastric carcinogenesis. Gut 2019. [Google Scholar] [CrossRef] [PubMed]
- Zucca, E.; Copie-Bergman, C.; Ricardi, U.; Thieblemont, C.; Raderer, M.; Ladetto, M.; Grp, E.G.W. Gastric marginal zone lymphoma of MALT type: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann. Oncol. 2013, 24, 144–148. [Google Scholar] [CrossRef] [PubMed]
- Floch, P.; Megraud, F.; Lehours, P. Helicobacter pylori strains and gastric MALT lymphoma. Toxins 2017, 9, 132. [Google Scholar] [CrossRef] [PubMed]
- Treiber, G.; Lambert, J.R. The impact of Helicobacter pylori eradication on peptic ulcer healing. Am. J. Gastroenterol. 1998, 93, 1080–1084. [Google Scholar] [CrossRef] [PubMed]
- Kuo, S.H.; Wu, M.S.; Yeh, K.H.; Lin, C.W.; Hsu, P.N.; Chen, L.T.; Cheng, A.L. Novel insights of lymphomagenesis of Helicobacter pylori-dependent gastric mucosa-associated lymphoid tissue lymphoma. Cancers 2019, 11, 547. [Google Scholar] [CrossRef]
- Ferlay, J.; Colombet, M.; Soerjomataram, I.; Mathers, C.; Parkin, D.M.; Pineros, M.; Znaor, A.; Bray, F. Estimating the global cancer incidence and mortality in 2018: GLOBOCAN sources and methods. Int. J. Cancer 2019, 144, 1941–1953. [Google Scholar] [CrossRef]
- Kim, M.A.; Lee, H.S.; Yang, H.K.; Kim, W.H. Clinicopathologic and protein expression differences between cardia carcinoma and noncardia carcinoma of the stomach. Cancer 2005, 103, 1439–1446. [Google Scholar] [CrossRef]
- Riihimaki, M.; Hemminki, A.; Sundquist, K.; Sundquist, J.; Hemminki, K. Metastatic spread in patients with gastric cancer. Oncotarget 2016, 7, 52307–52316. [Google Scholar] [CrossRef]
- Schnoor, M.; Stradal, T.E.; Rottner, K. Cortactin: Cell functions of a multifaceted actin-binding protein. Trends Cell Biol. 2018, 28, 79–98. [Google Scholar] [CrossRef]
- Weaver, A.M. Cortactin in tumor invasiveness. Cancer Lett. 2008, 265, 157–166. [Google Scholar] [CrossRef] [PubMed]
- Uruno, T.; Liu, J.L.; Zhang, P.J.; Fan, Y.X.; Egile, C.; Li, P.; Mueller, S.C.; Zhan, X. Activation of Arp2/3 complex-mediated actin polymerization by cortactin. Nat. Cell Biol. 2001, 3, 259–266. [Google Scholar] [CrossRef] [PubMed]
- Kelley, L.C.; Hayes, K.E.; Ammer, A.G.; Martin, K.H.; Weed, S.A. Cortactin phosphorylated by ERK1/2 localizes to sites of dynamic actin regulation and is required for carcinoma lamellipodia persistence. PLoS ONE 2010, 5, 13. [Google Scholar] [CrossRef]
- Tegtmeyer, N.; Wittelsberger, R.; Hartig, R.; Wessler, S.; Martinez-Quiles, N.; Backert, S. Serine phosphorylation of cortactin controls focal adhesion kinase activity and cell scattering induced by Helicobacter pylori. Cell Host Microbe 2011, 9, 520–531. [Google Scholar] [CrossRef]
- Selbach, M.; Backert, S. Cortactin: An Achilles’ heel of the actin cytoskeleton targeted by pathogens. Trends Microbiol. 2005, 13, 181–189. [Google Scholar] [CrossRef]
- Mizutani, K.; Miki, H.; He, H.; Maruta, H.; Takenawa, T. Essential role of neural Wiskott-Aldrich syndrome protein in podosome formation and degradation of extracellular matrix in src-transformed fibroblasts. Cancer Res. 2002, 62, 669–674. [Google Scholar]
- Kinley, A.W.; Weed, S.A.; Weaver, A.M.; Karginov, A.V.; Bissonette, E.; Cooper, J.A.; Parsons, J.T. Cortactin interacts with WIP in regulating Arp2/3 activation and membrane protrusion. Curr. Biol. 2003, 13, 384–393. [Google Scholar] [CrossRef]
- Dudek, S.M.; Birukov, K.G.; Zhan, X.; Garcia, J.G.N. Novel interaction of cortactin with endothelial cell myosin light chain kinase. Biochem. Biophys. Res. Commun. 2002, 298, 511–519. [Google Scholar] [CrossRef]
- Yamada, H.; Takeda, T.; Michiue, H.; Abe, T.; Takei, K. Actin bundling by dynamin 2 and cortactin is implicated in cell migration by stabilizing filopodia in human non-small cell lung carcinoma cells. Int. J. Oncol. 2016, 49, 877–886. [Google Scholar] [CrossRef]
- Abe, T.; La, T.M.; Miyagaki, Y.; Oya, E.; Wei, F.Y.; Sumida, K.; Fujise, K.; Takeda, T.; Tomizawa, K.; Takei, K.; et al. Phosphorylation of cortactin by cyclin-dependent kinase 5 modulates actin bundling by the dynamin 1-cortactin ring-like complex and formation of filopodia and lamellipodia in NG108-15 glioma-derived cells. Int. J. Oncol. 2019, 54, 550–558. [Google Scholar] [CrossRef]
- Kim, K.; Hou, P.; Gorski, J.L.; Cooper, J.A. Effect of Fgd1 on cortactin in Arp2/3 complex-mediated actin assembly. Biochemistry 2004, 43, 2422–2427. [Google Scholar] [CrossRef] [PubMed]
- Hou, P.; Estrada, L.; Kinley, A.W.; Parsons, J.T.; Vojtek, A.B.; Gorski, J.L. Fgd1, the Cdc42 GEF responsible for Faciogenital Dysplasia, directly interacts with cortactin and mAbp1 to modulate cell shape. Hum. Mol. Genet. 2003, 12, 1981–1993. [Google Scholar] [CrossRef] [PubMed]
- Katsube, T.; Takahisa, M.; Ueda, R.; Hashimoto, N.; Kobayashi, M.; Togashi, S. Cortactin associates with the cell-cell junction protein ZO-1 in both Drosophila and mouse. J. Biol. Chem. 1998, 273, 29672–29677. [Google Scholar] [CrossRef] [PubMed]
- MacGillavry, H.D.; Kerr, J.M.; Kassner, J.; Frost, N.A.; Blanpied, T.A. Shank-cortactin interactions control actin dynamics to maintain flexibility of neuronal spines and synapses. Eur. J. Neurosci. 2016, 43, 179–193. [Google Scholar] [CrossRef]
- Webb, B.A.; Zhou, S.T.; Eves, R.; Shen, L.; Jia, L.; Mak, A.S. Phosphorylation of cortactin by p21-activated kinase. Arch. Biochem. Biophys. 2006, 456, 183–193. [Google Scholar] [CrossRef]
- Eiseler, T.; Hausser, A.; De Kimpe, L.; Van Lint, J.; Pfizenmaier, K. Protein kinase D controls actin polymerization and cell motility through phosphorylation of cortactin. J. Biol. Chem. 2010, 285, 18672–18683. [Google Scholar] [CrossRef]
- Sroka, R.; Van Lint, J.; Katz, S.F.; Schneider, M.R.; Kleger, A.; Paschke, S.; Seufferlein, T.; Eiseler, T. Cortactin is a scaffolding platform for the E-cadherin adhesion complex and is regulated by protein kinase D1 phosphorylation. J. Cell Sci. 2016, 129, 2416–2429. [Google Scholar] [CrossRef]
- Janjanam, J.; Chandaka, G.K.; Kotla, S.; Rao, G.N. PLC beta 3 mediates cortactin interaction with WAVE2 in MCP1-induced actin polymerization and cell migration. Mol. Biol. Cell 2015, 26, 4589–4606. [Google Scholar] [CrossRef]
- Stock, K.; Borrink, R.; Mikesch, J.H.; Hansmeier, A.; Rehkamper, J.; Trautmann, M.; Wardelmann, E.; Hartmann, W.; Sperveslage, J.; Steinestel, K. Overexpression and Tyr421-phosphorylation of cortactin is induced by three-dimensional spheroid culturing and contributes to migration and invasion of pancreatic ductal adenocarcinoma (PDAC) cells. Cancer Cell Int. 2019, 19, 77. [Google Scholar] [CrossRef]
- Oser, M.; Mader, C.C.; Gil-Henn, H.; Magalhaes, M.; Bravo-Cordero, J.J.; Koleske, A.J.; Condeelis, J. Specific tyrosine phosphorylation sites on cortactin regulate Nck1-dependent actin polymerization in invadopodia. J. Cell Sci. 2010, 123, 3662–3673. [Google Scholar] [CrossRef]
- Rosenberg, B.J.; Gil-Henn, H.; Mader, C.C.; Halo, T.; Yin, T.F.; Condeelis, J.; Machida, K.; Wu, Y.I.; Koleske, A.J. Phosphorylated cortactin recruits Vav2 guanine nucleotide exchange factor to activate Rac3 and promote invadopodial function in invasive breast cancer cells. Mol. Biol. Cell 2017, 28, 1347–1360. [Google Scholar] [CrossRef] [PubMed]
- Huang, C.; Liu, J.L.; Haudenschild, C.C.; Zhan, X. The role of tyrosine phosphorylation of cortactin in the locomotion of endothelial cells. J. Biol. Chem. 1998, 273, 25770–25776. [Google Scholar] [CrossRef] [PubMed]
- Boyle, S.N.; Michaud, G.A.; Schweitzer, B.; Predki, P.F.; Koleske, A.J. A critical role for cortactin phosphorylation by Abl-family kinases in PDGF-induced dorsal-wave formation. Curr. Biol. 2007, 17, 445–451. [Google Scholar] [CrossRef] [PubMed]
- Ren, Y.; He, Y.; Brown, S.; Zbornik, E.; Mlodzianoski, M.J.; Ma, D.; Huang, F.; Mattoo, S.; Suter, D.M. A single tyrosine phosphorylation site in cortactin is important for filopodia formation in neuronal growth cones. Mol. Biol. Cell 2019, 30, 1817–1833. [Google Scholar] [CrossRef] [PubMed]
- Yamada, H.; Abe, T.; Satoh, A.; Okazaki, N.; Tago, S.; Kobayashi, K.; Yoshida, Y.; Oda, Y.; Watanabe, M.; Tomizawa, K.; et al. Stabilization of actin bundles by a dynamin 1/cortactin ring complex is necessary for growth cone filopodia. J. Neurosci. 2013, 33, 4514–4526. [Google Scholar] [CrossRef] [PubMed]
- McNiven, M.A.; Kim, L.; Krueger, E.W.; Orth, J.D.; Cao, H.; Wong, T.W. Regulated interactions between dynamin and the actin-binding protein cortactin modulate cell shape. J. Cell Biol. 2000, 151, 187–198. [Google Scholar] [CrossRef]
- Gray, N.W.; Kruchten, A.E.; Chen, J.; McNiven, M.A. A dynamin-3 spliced variant modulates the actin/cortactin-dependent morphogenesis of dendritic spines. J. Cell Sci. 2005, 118, 1279–1290. [Google Scholar] [CrossRef]
- Ohoka, Y.; Takai, Y. Isolation and characterization of cortactin isoforms and a novel cortactin-binding protein, CBP90. Genes Cells 1998, 3, 603–612. [Google Scholar] [CrossRef]
- Chen, Y.K.; Hsueh, Y.P. Cortactin-binding protein 2 modulates the mobility of cortactin and regulates dendritic spine formation and maintenance. J. Neurosci. 2012, 32, 1043–1055. [Google Scholar] [CrossRef]
- Du, Y.R.; Weed, S.A.; Xiong, W.C.; Marshall, T.D.; Parsons, J.T. Identification of a novel cortactin SH3 domain-binding protein and its localization to growth cones of cultured neurons. Mol. Cell. Biol. 1998, 18, 5838–5851. [Google Scholar] [CrossRef]
- Olofsson, A.; Skalman, L.N.; Obi, I.; Lundmark, R.; Arnqvist, A. Uptake of Helicobacter pylori vesicles is facilitated by clathrin-dependent and clathrin-independent endocytic pathways. mBio 2014, 5. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, J.; Ohnsihi, H.; Shibata, H.; Wada, A.; Hirayama, T.; Iiri, T.; Ueda, N.; Kanamaru, C.; Tsuchida, T.; Mashima, H.; et al. Dynamin is involved in human epithelial cell vacuolation caused by the Helicobacter pylori-produced cytotoxin VacA. J. Clin. Investig. 2001, 107, 363–370. [Google Scholar] [CrossRef] [PubMed]
- Krueger, S.; Hundertmark, T.; Kuester, D.; Kalinski, T.; Peitz, U.; Roessner, A. Helicobacter pylori alters the distribution of ZO-1 and p120ctn in primary human gastric epithelial cells. Pathol. Res. Pract. 2007, 203, 433–444. [Google Scholar] [CrossRef] [PubMed]
- Martinez-Quiles, N.; Ho, H.Y.H.; Kirschner, M.W.; Ramesh, N.; Geha, R.S. Erk/Src phosphorylation of cortactin acts as a switch on-switch off mechanism that controls its ability to activate N-WASP. Mol. Cell. Biol. 2004, 24, 5269–5280. [Google Scholar] [CrossRef]
- Cowieson, N.P.; King, G.; Cookson, D.; Ross, I.; Huber, T.; Hume, D.A.; Kobe, B.; Martin, J.L. Cortactin adopts a globular conformation and bundles actin into sheets. J. Biol. Chem. 2008, 283, 16187–16193. [Google Scholar] [CrossRef]
- Lua, B.L.; Low, B.C. Cortactin phosphorylation as a switch for actin cytoskeletal network and cell dynamics control. FEBS Lett. 2005, 579, 577–585. [Google Scholar] [CrossRef]
- Kelley, L.; Hayes, K.; Ammer, A.; Martin, K.; Weed, S. Revisiting the ERK/Src cortactin switch. Commun. Integr. Biol. 2011, 4, 205–207. [Google Scholar] [CrossRef]
- Tegtmeyer, N.; Backert, S. Role of Abl and Src family kinases in actin-cytoskeletal rearrangements induced by the Helicobacter pylori CagA protein. Eur. J. Cell Biol. 2011, 90, 880–890. [Google Scholar] [CrossRef]
- Drazic, A.; Myklebust, L.M.; Ree, R.; Arnesen, T. The world of protein acetylation. Biochim. Biophys. Acta Proteins Proteom. 2016, 1864, 1372–1401. [Google Scholar] [CrossRef]
- Zhang, X.H.; Yuan, Z.G.; Zhang, Y.T.; Yong, S.; Salas-Burgos, A.; Koomen, J.; Olashaw, N.; Parsons, J.T.; Yang, X.J.; Dent, S.R.; et al. HDAC6 modulates cell motility by altering the acetylation level of cortactin. Mol. Cell 2007, 27, 197–213. [Google Scholar] [CrossRef]
- Kozyreva, V.K.; McLaughlin, S.L.; Livengood, R.H.; Calkins, R.A.; Kelley, L.C.; Rajulapati, A.; Ice, R.J.; Smolkin, M.B.; Weed, S.A.; Pugacheva, E.N. NEDD9 regulates actin dynamics through cortactin deacetylation in an AURKA/HDAC6-dependent manner. Mol. Cancer Res. 2014, 12, 681–693. [Google Scholar] [CrossRef] [PubMed]
- Meiler, E.; Nieto-Pelegrin, E.; Martinez-Quiles, N. Cortactin tyrosine phosphorylation promotes its deacetylation and inhibits cell spreading. PLoS ONE 2012, 7, e33662. [Google Scholar] [CrossRef] [PubMed]
- Ramos-Garcia, P.; Gonzalez-Moles, M.A.; Ayen, A.; Gonzalez-Ruiz, L.; Ruiz-Avila, I.; Gil-Montoya, J.A. Prognostic and clinicopathological significance of CTTN/cortactin alterations in head and neck squamous cell carcinoma: Systematic review and meta-analysis. Head Neck J. Sci. Spec. Head Neck 2019, 41, 1963–1978. [Google Scholar] [CrossRef] [PubMed]
- Jing, X.Q.; Wu, H.; Ji, X.P.; Wu, H.X.; Shi, M.M.; Zhao, R. Cortactin promotes cell migration and invasion through upregulation of the dedicator of cytokinesis 1 expression in human colorectal cancer. Oncol. Rep. 2016, 36, 1946–1952. [Google Scholar] [CrossRef]
- Wei, J.; Zhao, Z.X.; Li, Y.; Zhou, Z.Q.; You, T.G. Cortactin expression confers a more malignant phenotype to gastric cancer SGC-7901 cells. World J. Gastroenterol. 2014, 20, 3287–3300. [Google Scholar] [CrossRef]
- Chuma, M.; Sakamoto, M.; Yasuda, J.; Fujii, G.; Nakanishi, K.; Tsuchiya, A.; Ohta, T.; Asaka, M.; Hirohashi, S. Overexpression of cortactin is involved in motility and metastasis of hepatocellular carcinoma. J. Hepatol. 2004, 41, 629–636. [Google Scholar] [CrossRef]
- Dedes, K.J.; Lopez-Garcia, M.A.; Geyer, F.C.; Lambros, M.B.K.; Savage, K.; Vatcheva, R.; Wilkerson, P.; Wetterskog, D.; Lacroix-Triki, M.; Natrajan, R.; et al. Cortactin gene amplification and expression in breast cancer: A chromogenic in situ hybridisation and immunohistochemical study. Breast Cancer Res. Treat. 2010, 124, 653–666. [Google Scholar] [CrossRef]
- Li, A.; Zhang, L.; Zhang, X.; Jin, W.; Ren, Y. Expression and clinical significance of cortactin protein in ovarian neoplasms. Clin. Transl. Oncol. 2016, 18, 220–227. [Google Scholar] [CrossRef]
- Yin, M.; Ma, W.Q.; An, L.G. Cortactin in cancer cell migration and invasion. Oncotarget 2017, 8, 88232–88243. [Google Scholar] [CrossRef]
- Rodrigo, J.P.; Garcia-Carracedo, D.; Garcia, L.A.; Menendez, S.T.; Allonca, E.; Gonzalez, M.V.; Fresno, M.F.; Suarez, C.; Garcia-Pedrero, J.M. Distinctive clinicopathological associations of amplification of the cortactin gene at 11q13 in head and neck squamous cell carcinomas. J. Pathol. 2009, 217, 516–523. [Google Scholar] [CrossRef]
- Backert, S.; Haas, R.; Gerhard, M.; Naumann, M. The Helicobacter pylori type IV secretion system encoded by the cag pathogenicity island: Architecture, function, and signaling. In Type IV Secretion in Gram-Negative and Gram-Positive Bacteria; Current Topics in Microbiology and Immunology Book Series Volume 413; Backert, S., Grohmann, E., Eds.; Springer: Cham, Switzerland, 2018; pp. 187–220. [Google Scholar]
- Bugaytsova, J.A.; Bjornham, O.; Chernov, Y.A.; Gideonsson, P.; Henriksson, S.; Mendez, M.; Sjostrom, R.; Mahdavi, J.; Shevtsova, A.; Ilver, D.; et al. Helicobacter pylori adapts to chronic infection and gastric disease via pH-responsive BabA-mediated adherence. Cell Host Microbe 2017, 21, 376–389. [Google Scholar] [CrossRef] [PubMed]
- McClain, M.S.; Beckett, A.C.; Cover, T.L. Helicobacter pylori vacuolating toxin and gastric cancer. Toxins 2017, 9, 316. [Google Scholar] [CrossRef] [PubMed]
- Loh, J.T.; Torres, V.J.; Algood, H.M.S.; McClain, M.S.; Cover, T.L. Helicobacter pylori HopQ outer membrane protein attenuates bacterial adherence to gastric epithelial cells. FEMS Microbiol. Lett. 2008, 289, 53–58. [Google Scholar] [CrossRef] [PubMed]
- Hatakeyama, M. Structure and function of Helicobacter pylori CagA, the first-identified bacterial protein involved in human cancer. Proc. Jpn. Acad. Ser. B Phys. Biol. Sci. 2017, 93, 196–219. [Google Scholar] [CrossRef]
- Su, Y.L.; Huang, H.L.; Huang, B.S.; Chen, P.C.; Chen, C.S.; Wang, H.L.; Lin, P.H.; Chieh, M.S.; Wu, J.J.; Yang, J.C.; et al. Combination of OipA, BabA, and SabA as candidate biomarkers for predicting Helicobacter pylori-related gastric cancer. Sci. Rep. 2016, 6, 12. [Google Scholar] [CrossRef]
- Grohmann, E.; Christie, P.J.; Waksman, G.; Backert, S. Type IV secretion in Gram-negative and Gram-positive bacteria. Mol. Microbiol. 2018, 107, 455–471. [Google Scholar] [CrossRef]
- Knorr, J.; Ricci, V.; Hatakeyama, M.; Backert, S. Classification of Helicobacter pylori virulence factors: Is CagA a toxin or not? Trends Microbiol. 2019, 27, 731–738. [Google Scholar] [CrossRef]
- Chauhan, N.; Tay, A.C.Y.; Marshall, B.J.; Jain, U. Helicobacter pylori VacA, a distinct toxin exerts diverse functionalities in numerous cells: An overview. Helicobacter 2019, 24, e12544. [Google Scholar] [CrossRef]
- Harrer, A.; Boehm, M.; Backert, S.; Tegtmeyer, N. Overexpression of serine protease HtrA enhances disruption of adherens junctions, paracellular transmigration and type IV secretion of CagA by Helicobacter pylori. Gut Pathog. 2017, 9, 40. [Google Scholar] [CrossRef]
- Backert, S.; Boehm, M.; Harrer, A.; Tegtmeyer, N. Helicobacter pylori serine protease HtrA exhibits a crucial role in oxygen stress resistance, heat tolerance and epithelial barrier disruption. Helicobacter 2017, 22. [Google Scholar]
- Hoy, B.; Lower, M.; Weydig, C.; Carra, G.; Tegtmeyer, N.; Geppert, T.; Schroder, P.; Sewald, N.; Backert, S.; Schneider, G.; et al. Helicobacter pylori HtrA is a new secreted virulence factor that cleaves E-cadherin to disrupt intercellular adhesion. EMBO Rep. 2010, 11, 798–804. [Google Scholar] [CrossRef] [PubMed]
- Backert, S.; Bernegger, S.; Skorko-Glonek, J.; Wessler, S. Extracellular HtrA serine proteases: An emerging new strategy in bacterial pathogenesis. Cell. Microbiol. 2018, 20. [Google Scholar] [CrossRef] [PubMed]
- Head, J.A.; Jiang, D.Y.; Li, M.; Zorn, L.J.; Schaefer, E.M.; Parsons, J.T.; Weed, S.A. Cortactin tyrosine phosphorylation requires Rac1 activity and association with the cortical actin cytoskeleton. Mol. Biol. Cell 2003, 14, 3216–3229. [Google Scholar] [CrossRef] [PubMed]
- Selbach, M.; Moese, S.; Hurwitz, R.; Hauck, C.R.; Meyer, T.F.; Backert, S. The Helicobacter pylori CagA protein induces cortactin dephosphorylation and actin rearrangement by c-Src inactivation. EMBO J. 2003, 22, 515–528. [Google Scholar] [CrossRef]
- Knorr, J.; Backert, S.; Tegtmeyer, N. SHP2-independent dephosphorylation of cortactin and vinculin during infection with Helicobacter pylori. Cancers 2020. submitted. [Google Scholar]
- Keates, S.; Keates, A.C.; Warny, M.; Peek, R.M.; Murray, P.G.; Kelly, C.P. Differential activation of mitogen-activated protein kinases in AGS gastric epithelial cells by cag+ and cag− Helicobacter pylori. J. Immunol. 1999, 163, 5552–5559. [Google Scholar]
- Meyer-ter-Vehn, T.; Covacci, A.; Kist, M.; Pahl, H.L. Helicobacter pylori activates mitogen-activated protein kinase cascades and induces expression of the proto-oncogenes c-fos and c-jun. J. Biol. Chem. 2000, 275, 16064–16072. [Google Scholar] [CrossRef]
- Brandt, S.; Kwok, T.; Hartig, R.; Konig, W.; Backert, S. NF-κB activation and potentiation of proinflammatory responses by the Helicobacter pylori CagA protein. Proc. Natl. Acad. Sci. USA 2005, 102, 9300–9305. [Google Scholar] [CrossRef]
- Tegtmeyer, N.; Hartig, R.; Delahay, R.M.; Rohde, M.; Brandt, S.; Conradi, J.; Takahashi, S.; Smolka, A.J.; Sewald, N.; Backert, S. A small fibronectin-mimicking protein from bacteria induces cell spreading and focal adhesion formation. J. Biol. Chem. 2010, 285, 23513–23524. [Google Scholar] [CrossRef]
- Senda, Y.; Murata-Kamiya, N.; Hatakeyama, M. C-terminal Src kinase-mediated EPIYA phosphorylation of Pragmin creates a feed-forward C-terminal Src kinase activation loop that promotes cell motility. Cancer Sci. 2016, 107, 972–980. [Google Scholar] [CrossRef]
- Tammer, I.; Brandt, S.; Hartig, R.; Konig, W.; Backert, S. Activation of Abl by Helicobacter pylori: A novel kinase for CagA and crucial mediator of host cell scattering. Gastroenterology 2007, 132, 1309–1319. [Google Scholar] [CrossRef] [PubMed]
- Cottet, S.; Corthesy-Theulaz, I.; Spertini, F.; Corthesy, B. Microaerophilic conditions permit to mimic in vitro events occurring during in vivo Helicobacter pylori infection and to identify Rho/Ras-associated proteins in cellular signaling. J. Biol. Chem. 2002, 277, 33978–33986. [Google Scholar] [CrossRef] [PubMed]
- Tegtmeyer, N.; Harrer, A.; Backert, S. Helicobacter pylori CagA targets cortactin and guanine nucleotide exchange factor Vav2 and Rac1 to control cell scattering. Manuscript in preparation.
- Saha, A.; Backert, S.; Hammond, C.E.; Gooz, M.; Smolka, A.J. Helicobacter pylori CagL activates ADAM17 to induce repression of the gastric H, K-ATPase α subunit. Gastroenterology 2010, 139, 239–248. [Google Scholar] [CrossRef] [PubMed]
- Churin, Y.; Al-Ghoul, L.; Kepp, O.; Meyer, T.E.; Birchmeier, W.; Naumann, M. Helicobacter pylori CagA protein targets the c-Met receptor and enhances the motogenic response. J. Cell Biol. 2003, 161, 249–255. [Google Scholar] [CrossRef]
- Tsutsumi, R.; Takahashi, A.; Azuma, T.; Higashi, H.; Hatakeyama, M. Focal adhesion kinase is a substrate and downstream effector of SHP-2 complexed with Helicobacter pylori CagA. Mol. Cell. Biol. 2006, 26, 261–276. [Google Scholar] [CrossRef]
- Chang, H.; Chen, D.F.; Ni, B.S.; Zuo, Q.F.; Wang, C.H.; Han, R.; Lan, C.H. Cortactin mediates apoptosis of gastric epithelial cells induced by VacA protein of Helicobacter pylori. Dig. Dis. Sci. 2016, 61, 80–90. [Google Scholar] [CrossRef]
- Weidmann, M.D.; Surve, C.R.; Eddy, R.J.; Chen, X.M.; Gertler, F.B.; Sharma, V.P.; Condeelis, J.S. MenaINV dysregulates cortactin phosphorylation to promote invadopodium maturation. Sci. Rep. 2016, 6, 36142. [Google Scholar] [CrossRef]
- Radhakrishnan, V.M.; Kojs, P.; Young, G.; Ramalingam, R.; Jagadish, B.; Mash, E.A.; Martinez, J.D.; Ghishan, F.K.; Kiela, P.R. pTyr421 cortactin is overexpressed in colon cancer and is dephosphorylated by curcumin: Involvement of non-receptor type 1 protein tyrosine phosphatase (PTPN1). PLoS ONE 2014, 9, e85796. [Google Scholar] [CrossRef]
- Zhang, L.H.; Tian, B.; Diao, L.R.; Xiong, Y.Y.; Tian, S.F.; Zhang, B.H.; Li, W.M.; Ren, H.; Li, Y.; Ji, J.F. Dominant expression of 85-kDa form of cortactin in colorectal cancer. J. Cancer Res. Clin. Oncol. 2006, 132, 113–120. [Google Scholar] [CrossRef]
- Yamasaki, E.; Wada, A.; Kumatori, A.; Nakagawa, I.; Funao, J.; Nakayama, M.; Hisatsune, J.; Kimura, M.; Moss, J.; Hirayama, T. Helicobacter pylori vacuolating cytotoxin induces activation of the proapoptotic proteins Bax and Bak, leading to cytochrome C release and cell death, independent of vacuolation. J. Biol. Chem. 2006, 281, 11250–11259. [Google Scholar] [CrossRef]
- Jain, P.; Luo, Z.Q.; Blanke, S.R. Helicobacter pylori vacuolating cytotoxin A (VacA) engages the mitochondrial fission machinery to induce host cell death. Proc. Natl. Acad. Sci. USA 2011, 108, 16032–16037. [Google Scholar] [CrossRef] [PubMed]
- Gauthier, N.C.; Monzo, P.; Gonzalez, T.; Doye, A.; Oldani, A.; Gounon, P.; Ricci, V.; Cormont, M.; Boquet, P. Early endosomes associated with dynamic F-actin structures are required for late trafficking of H. pylori VacA toxin. J. Cell Biol. 2007, 177, 343–354. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.; Huang, J.; Xie, X.Q.; He, Y.; Mo, F.; Luo, Z.X. Quercetin from Polygonum capitatum protects against gastric inflammation and apoptosis associated with Helicobacter pylori infection by affecting the levels of p38MAPK, BCL-2 and BAX. Molecules 2017, 22, 744. [Google Scholar] [CrossRef] [PubMed]
- Le Roux-Goglin, E.; Varon, C.; Spuul, P.; Asencio, C.; Megraud, F.; Genot, E. Helicobacter infection induces podosome assembly in primary hepatocytes in vitro. Eur. J. Cell Biol. 2012, 91, 161–170. [Google Scholar] [CrossRef]
- Flynn, D.C.; Cho, Y.; Vincent, D.; Cunnick, J.M. Podosomes and invadopodia: Related structures with common protein components that may promote breast cancer cellular invasion. Breast Cancer Basic Clin. Res. 2008, 2, 17–29. [Google Scholar] [CrossRef]
- Wang, H.; Tao, L.; Jin, F.; Gu, H.; Dai, X.; Ni, T.; Feng, J.; Ding, Y.; Xiao, W.; Qian, Y.; et al. Cofilin 1 induces the epithelial-mesenchymal transition of gastric cancer cells by promoting cytoskeletal rearrangement. Oncotarget 2017, 8, 39131–39142. [Google Scholar] [CrossRef]
- Xie, R.; Wang, H.; Jin, H.; Wen, G.; Tuo, B.; Xu, J. NHE1 is upregulated in gastric cancer and regulates gastric cancer cell proliferation, migration and invasion. Oncol. Rep. 2017, 37, 1451–1460. [Google Scholar] [CrossRef][Green Version]
- Magalhaes, M.A.O.; Larson, D.R.; Mader, C.C.; Bravo-Cordero, J.J.; Gil-Henn, H.; Oser, M.; Chen, X.; Koleske, A.J.; Condeelis, J. Cortactin phosphorylation regulates cell invasion through a pH-dependent pathway. J. Cell Biol. 2011, 195, 903–920. [Google Scholar] [CrossRef]
- Beaty, B.T.; Wang, Y.; Bravo-Cordero, J.J.; Sharma, V.P.; Miskolci, V.; Hodgson, L.; Condeelis, J. Talin regulates moesin-NHE-1 recruitment to invadopodia and promotes mammary tumor metastasis. J. Cell Biol. 2014, 205, 737–751. [Google Scholar] [CrossRef]
- Markwell, S.M.; Ammer, A.G.; Interval, E.T.; Allen, J.L.; Papenberg, B.W.; Hames, R.A.; Castano, J.E.; Schafer, D.A.; Weed, S.A. Cortactin phosphorylation by casein kinase 2 regulates actin-related protein 2/3 complex activity, invadopodia function, and tumor cell invasion. Mol. Cancer Res. 2019, 17, 987–1001. [Google Scholar] [CrossRef]
- Lee, Y.S.; Lee, D.Y.; Yu, D.Y.; Kim, S.; Lee, Y.C. Helicobacter pylori induces cell migration and invasion through casein kinase 2 in gastric epithelial cells. Helicobacter 2014, 19, 465–475. [Google Scholar] [CrossRef]
- Yamada, H.; Kikuchi, T.; Masumoto, T.; Wei, F.Y.; Abe, T.; Takeda, T.; Nishiki, T.; Tomizawa, K.; Watanabe, M.; Matsui, H.; et al. Possible role of cortactin phosphorylation by protein kinase C in actin-bundle formation at growth cone. Biol. Cell 2015, 107, 319–330. [Google Scholar] [CrossRef]
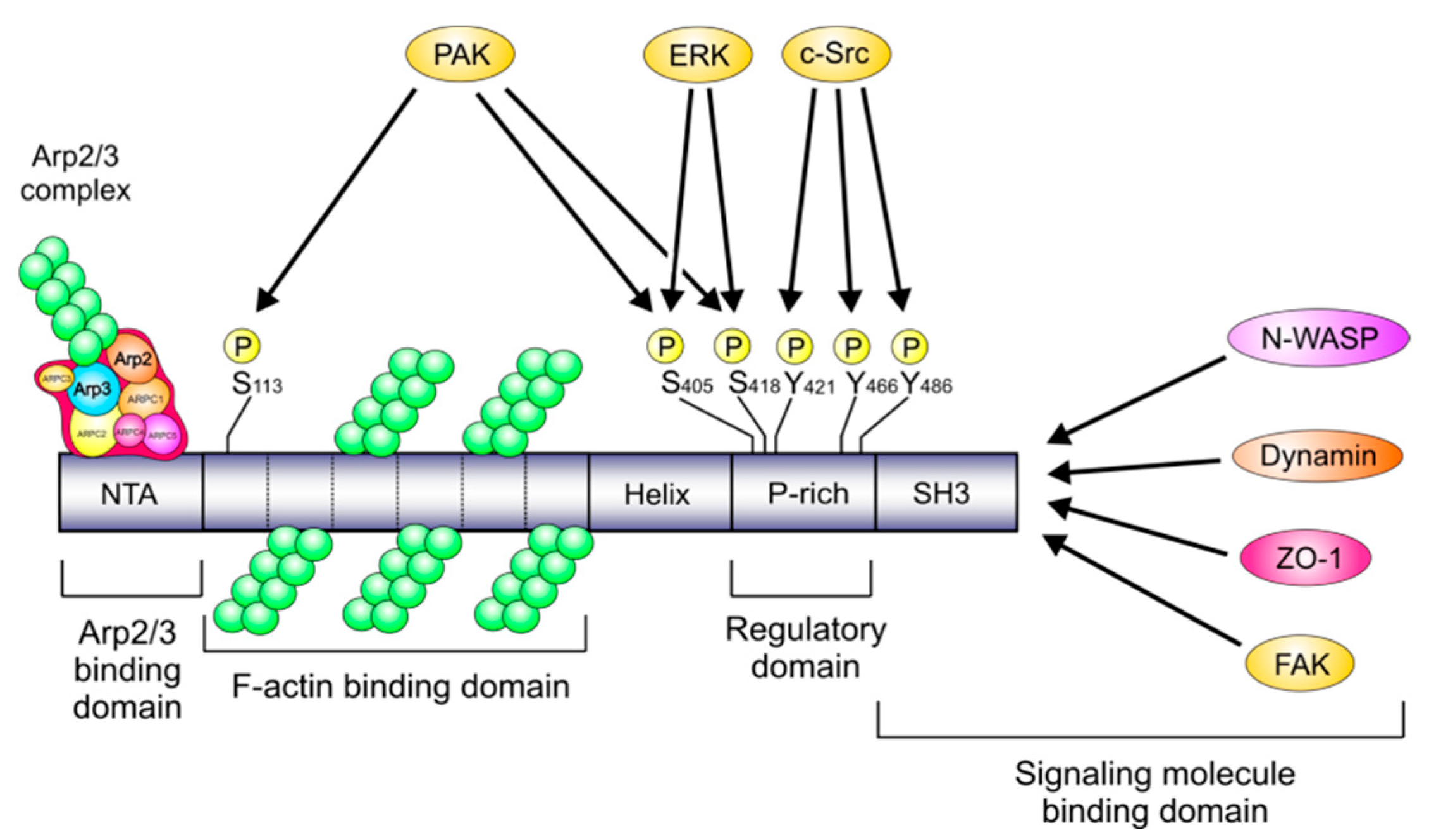
| Phospho-Sites in Cortactin | Interaction Partners | Cortactin Kinase | Proposed Function | Host Cell | Applied Methods | References |
|---|---|---|---|---|---|---|
| unknown | WIP | unknown | Arp2/3 complex activation | HEK293 | IP, WB, IFM, APA, GST-BA, YTHA | [27] |
| Fgd1 | In vitro | APA, GST-BA | [31] | |||
| MC3T3-E1 (Mouse osteoblast cells) and COS-7 (monkey kidney fibroblast-like cells) | YTHA, GST-BA, IP, ICC, WB, IFM | [32] | ||||
| ZO-1 | Cell-cell junction formation | Drosophila melanogaster (Canton-S wild type) embryo | YTHA, NB, GST-BA, WB, IFM, IP | [33] | ||
| Shank | Synapse morphology and function | Dissociated hippocampal cultures | WB, ICC, CLSM, PALM, SMA | [34] | ||
| S-113 | unknown | PAK1 | Reduced binding of cortactin to F-actin | A7r5 (Pancreatic ductal adenocarcinoma cells) | In vitro KA, MS, GST-BA, IFM | [35] |
| S-298 | WAVE2 | PKD1 | Generation of a 14-3-3 binding motif; binding to F-actin; Arp2/3 complex activation | Panc89 (PDAC), MCF-7 and HEK293T cells | IP, IHC, ABA, APA, CMA, GST-BA, In vitro ABA, CLSM, FRET, KA | [36] |
| β-catenin and vinculin | Destabilization of adherence junctions | HEK293T and Caco-2 cells | IP, IHC, ABA, CLSM, IPA, FRET, CCAA, | [37] | ||
| S-405 | FAK | ERK1/2 | FAK activation; cell motility and elongation; p85 phenotype | AGS (human gastric adenocarcinoma cells) | IP, WB, In vitro KA, IFM | [24] |
| S-418 | unknown | p80 phenotype | ||||
| unknown | Co-localization with F-actin in invadopodia | UMSCC1 (Head and Neck Squamous Cell Carcinomas) | WB, IP, IHC, IFM, CMA | [23] | ||
| unknown | Localization in lamellipodia | UMSCC2 (Head and Neck Squamous Cell Carcinomas) | ||||
| S-405, S-418 | N-WASP | Lamellipodia dynamics and motility | HNSCC (Head and Neck Squamous Cell Carcinomas) | |||
| WAVE2 | PKCδ | G-actin polymerization, F-actin stress fiber formation; cell migration | HASMC (Human Aortic Smooth Muscle Cells) | WB, IP, IFM, APA | [38] | |
| Y-421 | Gelsolin | Src | Increase of migratory capacity (migration and invasion; prometastatic, migratory phenotype | PDAC (Pancreatic ductal adenocarcinoma cells) | IHC, WB, PA, CMA, Scratch assay, CIA, MDA, IFM, IP | [39] |
| Y-421, Y-466 | Adaptor protein NCK1 | Nck1-dependent Arp2/3 activation | MDA-MB-231 (epithelial cell line from human breast cancer) | WB, IP, APA, FRET, LSM | [40] | |
| Vav2 | Invadopodium maturation; actin polymerization, matrix degradation, and invasive migratory behavior | MDA-MB-231 | WB, IP, GST-BA, IFM, APA, GST-BA, GEF AA, FRET | [41] | ||
| Y-421, Y-466, Y-482 | unknown | Inhibition of cortactin’s F-actin cross-linking activity; Enhanced cell migration | ECV304 (Human endothelial cells) | CMA, AR, IP, CLSM | [42] | |
| Endothelial myosin light chain kinase (MLCK) | Inhibition of MLCK binding to F-actin; abolished cortactin-mediated augmentation of Arp2/3-stimulated actin polymerization | In vitro | GST-BA, WB, In vitro KA | [28] | ||
| unknown | Abl | Dorsal-wave formation leading to lamellipodial protrusion | Mouse embryonic fibroblast | WB, IP, In vitro KA | [43] | |
| Y-499 | Arp2/3 | Srk | Filopodia formation | Bag cell neurons from Aplysia californica | ICC, IFM, STORM, WB, In vitro KA | [44] |
| T-145, T-219 | Dynamin | CDK5 | Inhibition of actin-bundling activity; reduction of pseudopodal formation | NG108-15 (glioma-derived cells) | CMA, In vitro KA, ABA, GST-BA, WB, EM, MS | [30] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sharafutdinov, I.; Backert, S.; Tegtmeyer, N. Cortactin: A Major Cellular Target of the Gastric Carcinogen Helicobacter pylori. Cancers 2020, 12, 159. https://doi.org/10.3390/cancers12010159
Sharafutdinov I, Backert S, Tegtmeyer N. Cortactin: A Major Cellular Target of the Gastric Carcinogen Helicobacter pylori. Cancers. 2020; 12(1):159. https://doi.org/10.3390/cancers12010159
Chicago/Turabian StyleSharafutdinov, Irshad, Steffen Backert, and Nicole Tegtmeyer. 2020. "Cortactin: A Major Cellular Target of the Gastric Carcinogen Helicobacter pylori" Cancers 12, no. 1: 159. https://doi.org/10.3390/cancers12010159
APA StyleSharafutdinov, I., Backert, S., & Tegtmeyer, N. (2020). Cortactin: A Major Cellular Target of the Gastric Carcinogen Helicobacter pylori. Cancers, 12(1), 159. https://doi.org/10.3390/cancers12010159







