Use of Antibiotics and Risk of Cancer: A Systematic Review and Meta-Analysis of Observational Studies
Abstract
1. Introduction
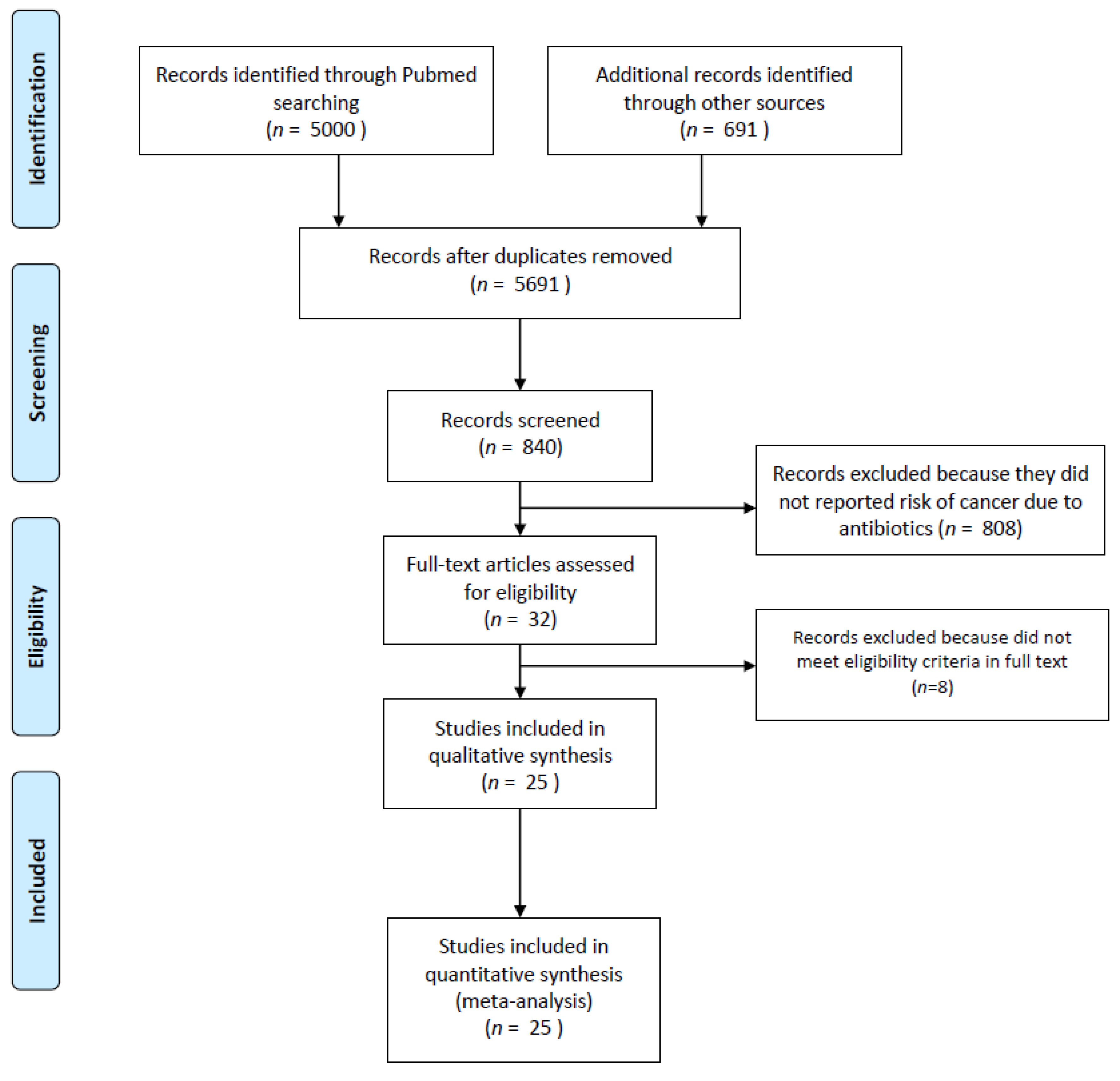
2. Materials and Methods
2.1. Study Selection and Inclusion Criteria
2.2. Data Extraction
2.3. Statistical Analysis
3. Results
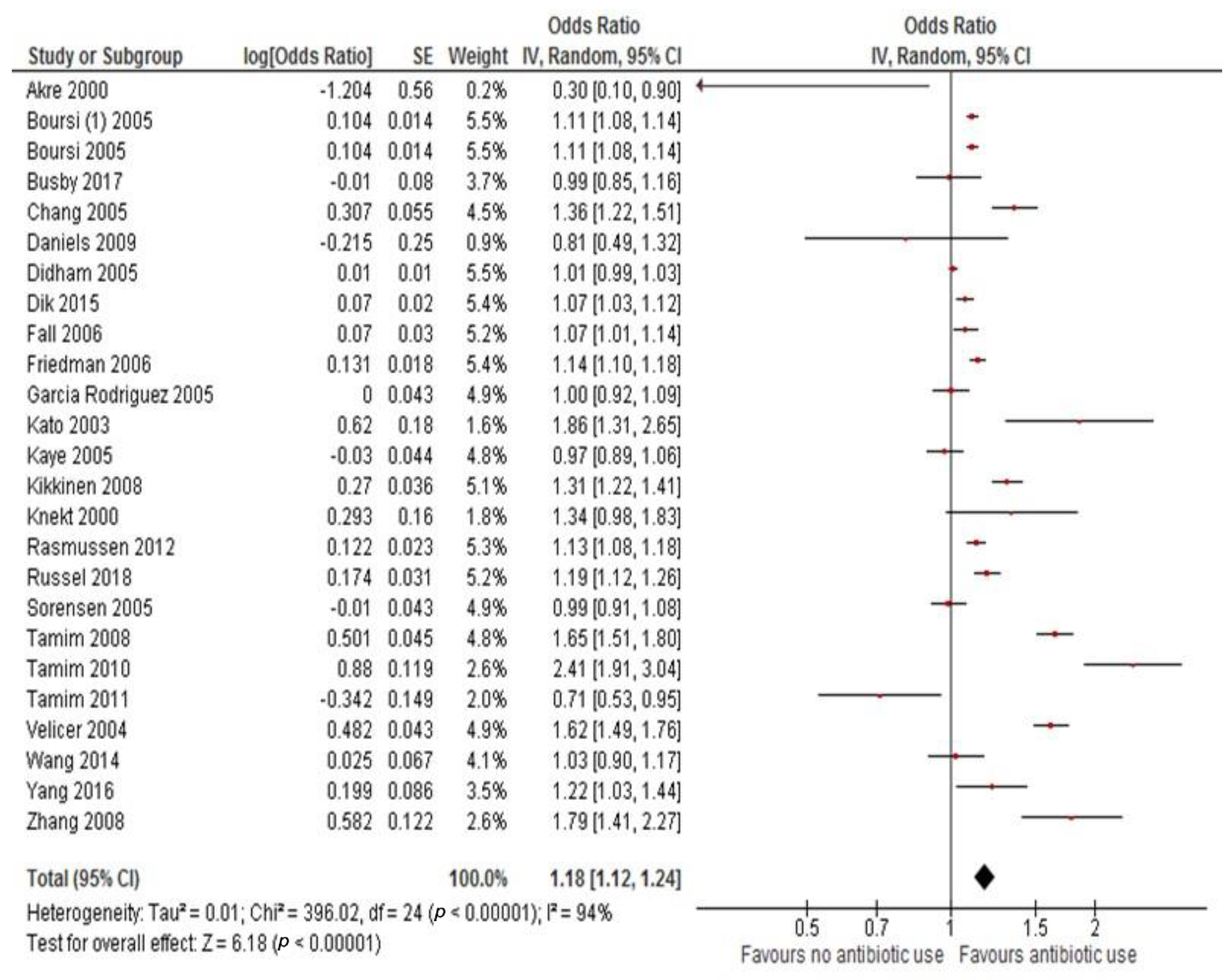
3.1. Primary Analysis: Overall Cancer Incidence
3.2. Secondary Analysis: Latency Period and Risk of Cancer
3.3. Tertiary Analysis: Correlation with Prescriptions and Risk of Exposure
3.4. Subgroup Analysis
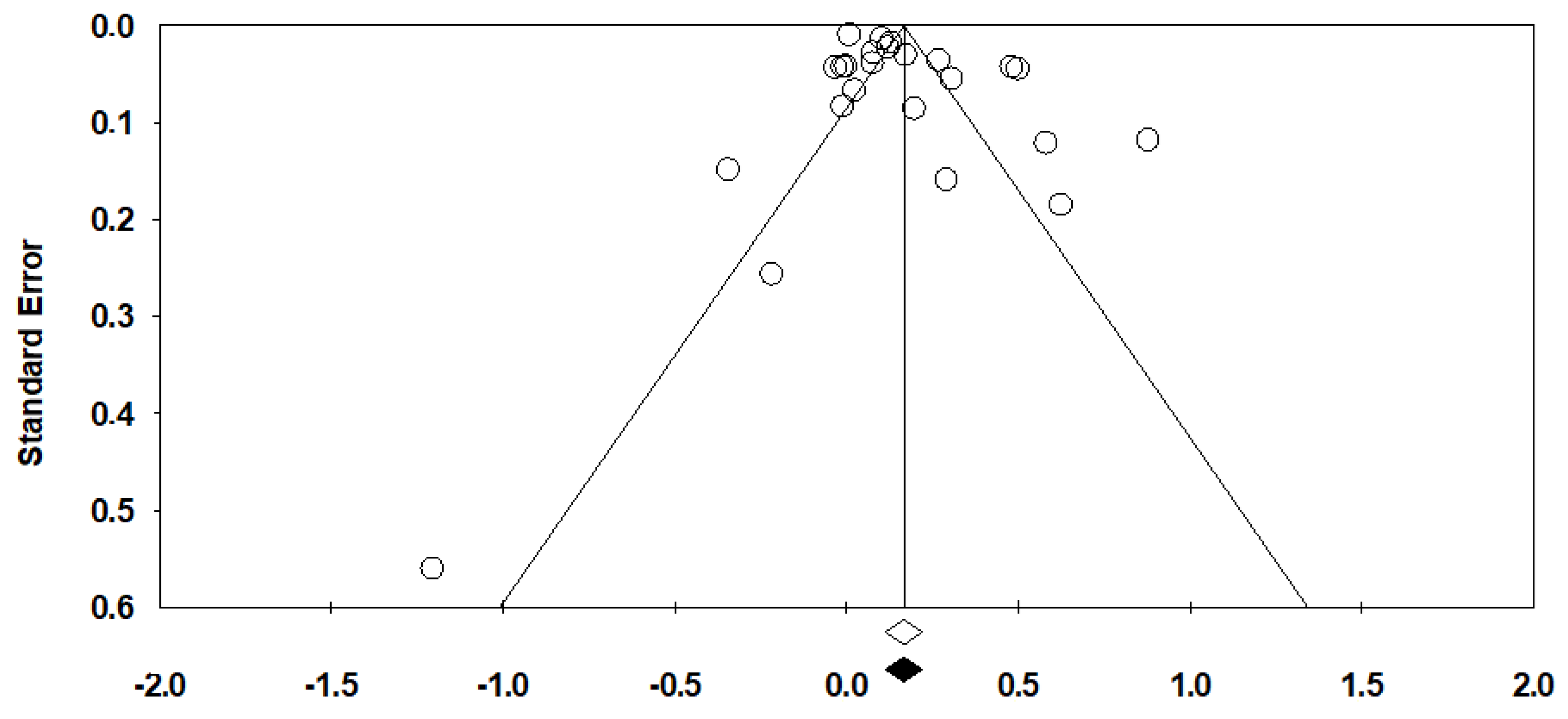
3.5. Publication Bias
3.6. Strength of Evidence
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Blaser, M.J. Antibiotic use and its consequences for the normal microbiome. Science 2016, 352, 544–545. [Google Scholar] [CrossRef] [PubMed]
- Rinninella, E.; Raoul, P.; Cintoni, M.; Franceschi, F.; Miggiano, G.A.D.; Gasbarrini, A.; Mele, M.C. What is the Healthy Gut Microbiota Composition? A Changing Ecosystem across Age, Environment, Diet, and Diseases. Microorganisms 2019, 7, 14. [Google Scholar] [CrossRef] [PubMed]
- Wang, T.; Cai, G.; Qiu, Y.; Fei, N.; Zhang, M.; Pang, X.; Jia, W.; Cai, S.; Zhao, L. Structural segregation of gut microbiota between colorectal cancer patients and healthy volunteers. ISME J. 2012, 6, 320–329. [Google Scholar] [CrossRef] [PubMed]
- Kostic, A.D.; Gevers, D.; Pedamallu, C.S.; Michaud, M.; Duke, F.; Earl, A.M.; Ojesina, A.I.; Jung, J.; Bass, A.J.; Tabernero, J.; et al. Genomic analysis identifies association of Fusobacterium with colorectal carcinoma. Genome Res. 2012, 22, 292–298. [Google Scholar] [CrossRef] [PubMed]
- Becattini, S.; Taur, Y.; Pamer, E.G. Antibiotic-Induced Changes in the Intestinal Microbiota and Disease. Trends Mol. Med. 2016, 22, 458–478. [Google Scholar] [CrossRef]
- Ubeda, C.; Pamer, E.G. Antibiotics, microbiota, and immune defense. Trends Immunol. 2012, 33, 459–466. [Google Scholar] [CrossRef] [PubMed]
- Stroup, D.F.; Berlin, J.A.; Morton, S.C.; Olkin, I.; Williamson, G.D.; Rennie, D.; Moher, D.; Becker, B.J.; Sipe, T.A.; Thacker, S.B. Meta-analysis of observational studies in epidemiology: A proposal for reporting. Meta-analysis of Observational Studies in Epidemiology (MOOSE) group. JAMA 2000, 283, 2008–2012. [Google Scholar] [CrossRef]
- Wells, G.; Shea, B.; O’Connell, D. The Newcastle-Ottawa Scale (NOS) for Assessing the Quality of Nonrandomized Studies in Meta-Analyses. 2009. Available online: http://www. ohri. ca/programs/clinical_epidemiology/oxford. htm (accessed on 19 October 2009).
- Egger, M.; Davey Smith, G.; Schneider, M.; Minder, C. Bias in meta-analysis detected by a simple, graphical test. BMJ 1997, 315, 629–634. [Google Scholar] [CrossRef]
- Akre, K.; Signorello, L.B.; Engstrand, L.; Bergstrom, R.; Larsson, S.; Eriksson, B.I.; Nyren, O. Risk for gastric cancer after antibiotic prophylaxis in patients undergoing hip replacement. Cancer Res. 2000, 60, 6376–6380. [Google Scholar]
- Boursi, B.; Mamtani, R.; Haynes, K.; Yang, Y.X. Recurrent antibiotic exposure may promote cancer formation--Another step in understanding the role of the human microbiota? Eur. J. Cancer 2015, 51, 2655–2664. [Google Scholar] [CrossRef]
- Tamim, H.M.; Hanley, J.A.; Hajeer, A.H.; Boivin, J.F.; Collet, J.P. Risk of breast cancer in relation to antibiotic use. Pharmacoepidemiol. Drug Saf. 2008, 17, 144–150. [Google Scholar] [CrossRef] [PubMed]
- Boursi, B.; Haynes, K.; Mamtani, R.; Yang, Y.X. Impact of antibiotic exposure on the risk of colorectal cancer. Pharmacoepidemiol. Drug Saf. 2015, 24, 534–542. [Google Scholar] [CrossRef] [PubMed]
- Busby, J.; Murchie, P.; Murray, L.; Iversen, L.; Lee, A.J.; Spence, A.; Watson, M.C.; Cardwell, C.R. The effect of medications which cause inflammation of the gastro-oesophageal tract on cancer risk: A nested case-control study of routine Scottish data. Int. J. Cancer 2017, 140, 1828–1835. [Google Scholar] [CrossRef] [PubMed]
- Chang, E.T.; Smedby, K.E.; Hjalgrim, H.; Schollkopf, C.; Porwit-MacDonald, A.; Sundstrom, C.; Tani, E.; d’Amore, F.; Melbye, M.; Adami, H.O.; et al. Medication use and risk of non-Hodgkin’s lymphoma. Am. J. Epidemiol. 2005, 162, 965–974. [Google Scholar] [CrossRef] [PubMed]
- Daniels, N.A.; Chen, Y.H.; Bent, S. Antibiotic and anti-inflammatory use and the risk of prostate cancer. BMC Res. Notes 2009, 2, 57. [Google Scholar] [CrossRef] [PubMed]
- Dik, V.K.; van Oijen, M.G.; Smeets, H.M.; Siersema, P.D. Frequent Use of Antibiotics Is Associated with Colorectal Cancer Risk: Results of a Nested Case-Control Study. Dig. Dis. Sci. 2016, 61, 255–264. [Google Scholar] [CrossRef] [PubMed]
- Fall, K.; Ye, W.; Nyren, O. Antibiotic treatment and risk of gastric cancer. Gut 2006, 55, 793–796. [Google Scholar] [CrossRef]
- Friedman, G.D.; Oestreicher, N.; Chan, J.; Quesenberry, C.P., Jr.; Udaltsova, N.; Habel, L.A. Antibiotics and risk of breast cancer: Up to 9 years of follow-up of 2.1 million women. Cancer Epidemiol. Biomark. Prev. 2006, 15, 2102–2106. [Google Scholar] [CrossRef]
- Garcia Rodriguez, L.A.; Gonzalez-Perez, A. Use of antibiotics and risk of breast cancer. Am. J. Epidemiol. 2005, 161, 616–619. [Google Scholar] [CrossRef]
- Kato, I.; Koenig, K.L.; Baptiste, M.S.; Lillquist, P.P.; Frizzera, G.; Burke, J.S.; Watanabe, H.; Shore, R.E. History of antibiotic use and risk of non-Hodgkin’s lymphoma (NHL). Int. J. Cancer 2003, 107, 99–105. [Google Scholar] [CrossRef]
- Kaye, J.A.; Jick, H. Antibiotics and the risk of breast cancer. Epidemiology 2005, 16, 688–690. [Google Scholar] [CrossRef]
- Kilkkinen, A.; Rissanen, H.; Klaukka, T.; Pukkala, E.; Heliovaara, M.; Huovinen, P.; Mannisto, S.; Aromaa, A.; Knekt, P. Antibiotic use predicts an increased risk of cancer. Int. J. Cancer 2008, 123, 2152–2155. [Google Scholar] [CrossRef]
- Rasmussen, M.L.; Hjalgrim, H.; Molgaard-Nielsen, D.; Wohlfahrt, J.; Melbye, M. Antibiotic use and risk of non-Hodgkin lymphomas. Int. J. Cancer 2012, 131, E1158–E1165. [Google Scholar] [CrossRef]
- Russell, B.; Garmo, H.; Beckmann, K.; Stattin, P.; Adolfsson, J.; Van Hemelrijck, M. A case-control study of lower urinary-tract infections, associated antibiotics and the risk of developing prostate cancer using PCBaSe 3.0. PLoS ONE 2018, 13, e0195690. [Google Scholar] [CrossRef]
- Tamim, H.M.; Musallam, K.M.; Al Kadri, H.M.; Boivin, J.F.; Collet, J.P. Antibiotic use and risk of gynecological cancer. Eur. J. Obstet. Gynecol. Reprod. Biol. 2011, 159, 388–393. [Google Scholar] [CrossRef]
- Velicer, C.M.; Heckbert, S.R.; Lampe, J.W.; Potter, J.D.; Robertson, C.A.; Taplin, S.H. Antibiotic use in relation to the risk of breast cancer. JAMA 2004, 291, 827–835. [Google Scholar] [CrossRef]
- Sorensen, H.T.; Skriver, M.V.; Friis, S.; McLaughlin, J.K.; Blot, W.J.; Baron, J.A. Use of antibiotics and risk of breast cancer: A population-based case-control study. Br. J. Cancer 2005, 92, 594–596. [Google Scholar] [CrossRef]
- Yang, B.; Hagberg, K.W.; Chen, J.; Sahasrabuddhe, V.V.; Graubard, B.I.; Jick, S.; McGlynn, K.A. Associations of antibiotic use with risk of primary liver cancer in the Clinical Practice Research Datalink. Br. J. Cancer 2016, 115, 85–89. [Google Scholar] [CrossRef]
- Zhang, H.; Garcia Rodriguez, L.A.; Hernandez-Diaz, S. Antibiotic use and the risk of lung cancer. Cancer Epidemiol. Biomark. Prev. 2008, 17, 1308–1315. [Google Scholar] [CrossRef]
- Tamim, H.M.; Hajeer, A.H.; Boivin, J.F.; Collet, J.P. Association between antibiotic use and risk of prostate cancer. Int. J. Cancer 2010, 127, 952–960. [Google Scholar] [CrossRef]
- Didham, R.C.; Reith, D.M.; McConnell, D.W.; Harrison, K.S. Antibiotic exposure and breast cancer in New Zealand. Breast Cancer Res. Treat. 2005, 92, 163–167. [Google Scholar] [CrossRef]
- Wang, J.L.; Chang, C.H.; Lin, J.W.; Wu, L.C.; Chuang, L.M.; Lai, M.S. Infection, antibiotic therapy and risk of colorectal cancer: A nationwide nested case-control study in patients with Type 2 diabetes mellitus. Int. J. Cancer 2014, 135, 956–967. [Google Scholar] [CrossRef]
- Knekt, P.; Adlercreutz, H.; Rissanen, H.; Aromaa, A.; Teppo, L.; Heliovaara, M. Does antibacterial treatment for urinary tract infection contribute to the risk of breast cancer? Br. J. Cancer 2000, 82, 1107–1110. [Google Scholar] [CrossRef][Green Version]
- Yang, J.H.; Bhargava, P.; McCloskey, D.; Mao, N.; Palsson, B.O.; Collins, J.J. Antibiotic-Induced Changes to the Host Metabolic Environment Inhibit Drug Efficacy and Alter Immune Function. Cell Host Microbe 2017, 22, 757–765. [Google Scholar] [CrossRef]
- Sergentanis, T.N.; Zagouri, F.; Zografos, G.C. Is antibiotic use a risk factor for breast cancer? A meta-analysis. Pharmacoepidemiol. Drug Saf. 2010, 19, 1101–1107. [Google Scholar] [CrossRef]
- Khosravi, A.; Mazmanian, S.K. Disruption of the gut microbiome as a risk factor for microbial infections. Curr. Opin. Microbiol. 2013, 16, 221–227. [Google Scholar] [CrossRef]
- Routy, B.; Le Chatelier, E.; Derosa, L.; Duong, C.P.M.; Alou, M.T.; Daillere, R.; Fluckiger, A.; Messaoudene, M.; Rauber, C.; Roberti, M.P.; et al. Gut microbiome influences efficacy of PD-1-based immunotherapy against epithelial tumors. Science 2018, 359, 91–97. [Google Scholar] [CrossRef]
- Vetizou, M.; Pitt, J.M.; Daillere, R.; Lepage, P.; Waldschmitt, N.; Flament, C.; Rusakiewicz, S.; Routy, B.; Roberti, M.P.; Duong, C.P.; et al. Anticancer immunotherapy by CTLA-4 blockade relies on the gut microbiota. Science 2015, 350, 1079–1084. [Google Scholar] [CrossRef]
- Aragon, I.M.; Herrera-Imbroda, B.; Queipo-Ortuno, M.I.; Castillo, E.; Del Moral, J.S.; Gomez-Millan, J.; Yucel, G.; Lara, M.F. The Urinary Tract Microbiome in Health and Disease. Eur. Urol. Focus 2018, 4, 128–138. [Google Scholar] [CrossRef]
- Mao, Q.; Jiang, F.; Yin, R.; Wang, J.; Xia, W.; Dong, G.; Ma, W.; Yang, Y.; Xu, L.; Hu, J. Interplay between the lung microbiome and lung cancer. Cancer Lett. 2018, 415, 40–48. [Google Scholar] [CrossRef]
- Whiteside, S.A.; Razvi, H.; Dave, S.; Reid, G.; Burton, J.P. The microbiome of the urinary tract—A role beyond infection. Nat. Rev. Urol. 2015, 12, 81–90. [Google Scholar] [CrossRef]
- Yamamoto, M.L.; Schiestl, R.H. Intestinal microbiome and lymphoma development. Cancer J. 2014, 20, 190–194. [Google Scholar] [CrossRef]
- Mima, K.; Nakagawa, S.; Sawayama, H.; Ishimoto, T.; Imai, K.; Iwatsuki, M.; Hashimoto, D.; Baba, Y.; Yamashita, Y.I.; Yoshida, N.; et al. The microbiome and hepatobiliary-pancreatic cancers. Cancer Lett. 2017, 402, 9–15. [Google Scholar] [CrossRef]
- Bullman, S.; Pedamallu, C.S.; Sicinska, E.; Clancy, T.E.; Zhang, X.; Cai, D.; Neuberg, D.; Huang, K.; Guevara, F.; Nelson, T.; et al. Analysis of Fusobacterium persistence and antibiotic response in colorectal cancer. Science 2017, 358, 1443–1448. [Google Scholar] [CrossRef]
- Zhu, H.; Shen, Z.; Luo, H.; Zhang, W.; Zhu, X. Chlamydia Trachomatis Infection-Associated Risk of Cervical Cancer: A Meta-Analysis. Medicine 2016, 95, e3077. [Google Scholar] [CrossRef]
- Langdon, A.; Crook, N.; Dantas, G. The effects of antibiotics on the microbiome throughout development and alternative approaches for therapeutic modulation. Genome Med. 2016, 8, 39. [Google Scholar] [CrossRef]
- De La Cochetiere, M.F.; Durand, T.; Lepage, P.; Bourreille, A.; Galmiche, J.P.; Dore, J. Resilience of the dominant human fecal microbiota upon short-course antibiotic challenge. J. Clin. Microbiol. 2005, 43, 5588–5592. [Google Scholar] [CrossRef]
- Dethlefsen, L.; Relman, D.A. Incomplete recovery and individualized responses of the human distal gut microbiota to repeated antibiotic perturbation. Proc. Natl. Acad. Sci. USA 2011, 108 (Suppl. 1), 4554–4561. [Google Scholar] [CrossRef]
- Fasugba, O.; Gardner, A.; Mitchell, B.G.; Mnatzaganian, G. Ciprofloxacin resistance in community- and hospital-acquired Escherichia coli urinary tract infections: A systematic review and meta-analysis of observational studies. BMC Infect. Dis. 2015, 15, 545. [Google Scholar] [CrossRef]
- Park, C.H.; Eun, C.S.; Han, D.S. Intestinal microbiota, chronic inflammation, and colorectal cancer. Intest. Res. 2018, 16, 338–345. [Google Scholar] [CrossRef]
- Meeker, D.; Linder, J.A.; Fox, C.R.; Friedberg, M.W.; Persell, S.D.; Goldstein, N.J.; Knight, T.K.; Hay, J.W.; Doctor, J.N. Effect of Behavioral Interventions on Inappropriate Antibiotic Prescribing Among Primary Care Practices: A Randomized Clinical Trial. JAMA 2016, 315, 562–570. [Google Scholar] [CrossRef]
- Sikkens, J.J.; van Agtmael, M.A.; Peters, E.J.G.; Lettinga, K.D.; van der Kuip, M.; Vandenbroucke-Grauls, C.; Wagner, C.; Kramer, M.H.H. Behavioral Approach to Appropriate Antimicrobial Prescribing in Hospitals: The Dutch Unique Method for Antimicrobial Stewardship (DUMAS) Participatory Intervention Study. JAMA Intern. Med. 2017, 177, 1130–1138. [Google Scholar] [CrossRef]
- Zitvogel, L.; Daillere, R.; Roberti, M.P.; Routy, B.; Kroemer, G. Anticancer effects of the microbiome and its products. Nat. Rev. Microbiol. 2017, 15, 465–478. [Google Scholar] [CrossRef]
| Author/Year | Type of Study | Country | N° pts | Cases | Controls | OR/RR for Risk | Type of Analysis | Adjustment Covariates | NOS | RoB |
|---|---|---|---|---|---|---|---|---|---|---|
| Akre/2000 [10] | Case-control | Sweden | 636 | 174 | 462 | 0.3 (0.1–0.7) | - | Gender, age, history of gastric resection, and regular use of aspirin | 7 | Mod |
| Boursi/2015 [11] | Case-control | UK | 103,044 | 20,990 | 82,054 | 1.11 (1.08–1.14) | Days of use, type of antibiotics, n° prescriptions | Diabetes mellitus, BMI, smoking history, alcohol consumption, chronic use of Aspirin/NSAIDs, and performance of screening colonoscopy. | 6 | Mod |
| Boursi/2015 [13] | Case-control | UK | 615,951 | 125,441 | 490,510 | 1.11 (1.08–1.14) | Time from 1st antibiotic use, type of antibiotics, n° prescriptions | Different according to cancer type (see full text) | 6 | Low |
| Busby/2017 [14] | Case-control | Scotland | 18,035 | 3098 | 14,937 | 0.99 (0.84–1.17) | N° prescriptions | Statin and aspirin use, and the presence of myocardial infarction, heart failure, peripheral vascular disease, cerebrovascular disease, connective tissue disease, dementia, chronic obstructive pulmonary disease, rheumatoid arthritis, diabetes, renal disease and liver disease, age, general practice and year of diagnosis | 6 | Mod |
| Chang/2005 [15] | Case-control | Denmark and Sweden | 6242 | 3055 | 3187 | 1.36 (1.22–1.53) | N° prescriptions | Age, sex, country | 5 | Mod |
| Daniels/2009 [16] | Case-control | New Zealand | 260 | 65 | 195 | 0.806 (0.487–1.33) | N° prescriptions | Age group, race, years of enrollment, and number of visits | 5 | Mod |
| Didham/2005 [32] | Case-control | USA | 12,00,000 | 6500 | 1.193.500 | 1.01 (0.99–1.02) | Years of use, type of antibiotics | Age | 5 | Mod |
| Dik/2016 [17] | Case-control | Netherland | 20,017 | 4029 | 15,988 | 1.08 (1.023–1.14) | Days of use, n° prescriptions | Age, sex, insulin-independent diabetes, insulin-dependent diabetes, and the use of proton pump inhibitors, acetylsalicylic acid, nonsteroidal anti-inflammatory drugs, blood lipid-lowering agents, estrogens, and immunosuppressive drugs | 6 | Low |
| Fall/2006 [18] | Retrospective cohort | Sweden | 501,757 | 645 | - | 1.08 (1–1.17) | Sex, age, follow up, type of infection, type of bacteria | Comorbidities | 8 | High |
| Friedman/2006 [19] | Retrospective cohort | US | 2,130,829 | 18521 | - | 1.14 (1.1–1.18) | Days of use, type of antibiotics, hormone use | Days of use, hormone use | 8 | High |
| Garcia Rodriguez/2005 [20] | Case control | Spain | 23,708 | 3708 | 20,000 | 1 (0.92–1.09) | Days of use, n° prescriptions, type of infection | Age, calendar year, body mass index, alcohol intake, hormone replacement therapy, use of NSAIDs, prior benign breast disease, time under observation, and utilization of healthcare services. | 7 | Mod |
| Kato/2003 [21] | Case control | US | 839 | 376 | 463 | 1.87 (1.3–2.7) | N° prescriptions, type of infection | Age, family history of hematologic cancer, college education, smoking status, average frequency of use of pain-relieving drugs, surrogate status and year of interview. | 8 | Low |
| Kaye/2005 [22] | Case control | US | 7559 | 1268 | 7291 | 0.97 (0.89–1.06) | N° prescriptions, type of antibiotics | BMI, use of hormone replacement therapy, history of benign proliferative breast disease, frequency of mammograms, and frequency of visits to the general practice | 7 | Mod |
| Kikkinen/2008 [23] | Retrospective cohort | Finland | 3,112,624 | 134,070 | - | 1.31 (1.22–1.42) | Type of cancer, n° prescriptions, years of duration, time from 1st antibiotic use | Age, sex | 7 | Low |
| Knekt/2000 [34] | Retrospective cohort | Finland | 9461 | 157 | - | 1.34 (0.98–1.83) | Age, bacteriuria, follow up | Age, region type, education, marital status, body mass index, parity, smoking, height, alcohol use and screening positive for bacteriuria. | 10 | Mod |
| Rasmussen/2012 [24] | Retrospective cohort | Denmark | 13,602 | 13,602 | - | 1.13 (1.08–1.19) | Type of antibiotics, n° prescriptions, time from 1st antibiotic | Age, sex, calendar period | 9 | Low |
| Russel/2018 [25] | Case-control | Sweden | 52,568 | 8762 | 43,806 | 1.19 (1.12–1.27) | Type of antibiotics, n° prescriptions, time from 1st antibiotic | Civil status, education, CCI and time between 1st antibiotic and event | 6 | High |
| Sorensen/2005 [28] | Case-control | Denmark | 30,008 | 2728 | 27,280 | 0.99 (0.91–1.06) | Type of antibiotics, n° prescriptions | Age at first birth, parity, and use of postmenopausal hormone replacement therapy | 5 | High |
| Tamim/2008 [12] | Case-control | Canada | 15,495 | 3099 | 12,396 | 1.65 (1.51–1.80) | N° prescriptions, type of antibiotic | Age, time of diagnosis and exposure to antibiotics during the other time periods | 5 | High |
| Tamim/2010 [31] | Case-control | Canada | 20,260 | 4052 | 16,208 | 2.41 (1.91–3.04) | N° prescriptions, type of antibiotic | Age and time of diagnosis | 5 | High |
| Tamim/2011 [26] | Case-control | Canada | 6125 | 1225 | 4900 | 0.71 (0.53–0.95) | N° prescriptions, type of antibiotic | Age, time of diagnosis, and antibiotic exposure in other periods | 5 | High |
| Velicer/2004 [27] | Case-control | US | 10,219 | 2266 | 7953 | 1.62 (1.48–1.76) | N° prescriptions, days of used, type of antibiotic | Age, level of education, race, length of enrollment, number of primary and specialty health care visits, pharmacy co-payment status, age at menarche, parity, age at first birth, body mass index, first-degree family history of breast cancer, mammographic breast density, prior hysterectomy, menopausal status, age at menopause, and use of oral contraceptives and postmenopausal hormones | 5 | High |
| Wang/2014 [33] | Case-control | Taiwan | 27,860 | 5572 | 22,288 | 1.02 (0.89–1.17) | N° prescriptions, type of antibiotic | Age, gender, socioeconomic status and numbers of stool occult blood tested | 5 | High |
| Yang/2016 [35] | Case-control | UK | 5835 | 1195 | 4640 | 1.22 (1.03–1.44) | N° prescriptions, type of antibiotic | BMI, smoking status, alcohol-related disorders, hepatitis B or C virus infection, diabetes, rare metabolic disorders, and use of anti-diabetic medications, paracetamol, and statins | 5 | High |
| Zhang/2008 [30] | Case-control | UK | 14,336 | 4336 | 10,000 | 1.79 (1.41–2.26) | N° prescriptions, type of antibiotic | Smoking status, smoking cessation interventions, episodes of different types of infection, history of COPD, asthma, body mass index, alcohol intake, and indicators of health care utilization | 5 | High |
| Author/Year | Median Follow Up | N° of Prescriptions (Duration of Treatment) | Antibiotics Considered | Cancers Analyzed | Different Time Intervals from Last Antibiotic Use and Cancer Events (Years) |
|---|---|---|---|---|---|
| Akre/2000 [10] | 8 years | NR | NR | Gastric | NR |
| Boursi/2015 [11] | 6.5 years | 1–5, 5–10, >10 course (1–14, 14-56, 56+ day duration) | Nitroimidazoles, penicillins, tetracyclines, macrolides, quinolones, cephalosporins, sulfonamides | Colorectal | 0–1; >1 |
| Boursi/2015 [13] | 4.7–7 years | 1, 2–5, >5 courses | Penicillins, cephalosporins, macrolides, tetracyclines, sulfonamides, quinolones and nitroimidazole | Breast, Oesophagus, Gastric, HCC, Biliary, Gallbladder, Pancreas, Prostate, Renal, Bladder, Melanoma, Cervix, Osteosarcoma, MM | 1–5, 5–10, >10 |
| Busby/2017 [14] | 5.5 years | 1, 2+ | Tetracyclines | Gastroesophageal | NR |
| Chang/2005 [15] | NR | 1–2, 3–5, 6–10, 11+ | NR | NHL | >2 |
| Daniels/2009 [16] | NR | 1–25, 26–50, 51–100, 100+ | Macrolides, tetracyclines, penicillins, sulfonamides, ciprofloxacin, levofloxacin (data not reported separately) | Prostate | NR |
| Didham/2005 [32] | NR | NR (≥2 years) | Macrolides, tetracyclines, penicillins, cephalosporins, sulfonamides, nitrofurantoin, others | Bladder and renal, brain and central nervous system, breast, colorectal, female reproductive system, leukemia, liver, pancreas and other digestive, lung and respiratory, lymphoma (non hodgkin’s), oral cavity, pharynx, oesophagus, other, prostate, skin (melanoma), skin (neoplasms), stomach and small intestine | NR |
| Dik/2016 [17] | 5 years | 1.2, 3–4, 5–7, ≥8 | Tetracyclines, penicillins, sulfonamides, macrolides, quinolones, nitrofurantoin | Colorectal | |
| Fall/2006 [18] | 11.8 years | < vs. ≥3/times year | NR | Non-cardia gastric cancer | 1–4, 5–9, 10–14, 15–19, 20+ |
| Friedman/2006 [19] | 9.4 years | NR (<50, 51–100, 101–500, 501–1000, >1000 days duration) | Penicillins, Tetracyclines, Macrolides, Quinolones, Cephalosporins, Lincosamides, Aminoglycosides, Sulfonamides, Metronidazole, Isoniazid, Rifampin, Nitrofurantoin | Breast | |
| Garcia Rodriguez/2005 [20] | At least 1 year | 1–10, 11–25, 26+ | NR | Breast | NR |
| Kato/2003 [21] | 2–20 years | 1, 2–4, 5–8, 9–17, 18–35, 36+ | NR | NHL | >2 |
| Kaye/2005 [22] | 94 months | NR (1–50, 51–100, 101–500, 500+ days duration) | Penicillins, Tetracyclines, Macrolides, Cephalosporins | Breast | NR |
| Kikkinen/2008 [23] | 7 years | 0–1, 1–5, ≥6 (1–3 years duration) | NR | Hematological, head & neck, gastrointestinal, thoracic, genitourinary, SNC, skin, bone, endocrine, breast, gynecological | NR |
| Knekt/2000 [34] | 18 years | NR | NR | Breast | NR |
| Rasmussen/2012 [24] | 13 years | 1, 2, 3, 4, 5+ | Tetracyclines, sulfonamides, penicillins, macrolides, quinolones | NHL, MM | |
| Russel/2018 [25] | NR | 1–3, 4–6, 7–9, 10+ | Sulfonamides, cephalexin, doxycycline, nitrofurantoin, quinolones, amoxicilline/clavulanate. | Prostate | 6–12 months, 1–2, 3–4, 5+ |
| Sorensen/2005 [28] | NR | 1–5, 6–10, >10 | Penicillins, tetracyclines, macrolides, quinolones, cephalosporins, sulfonamides | Breast | NR |
| Tamim/2008 [12] | NR | 1–3, 4–7, 8–13, 14+ | Penicillins, tetracyclines, macrolides, cephalosporins, sulfonamides, others | Breast | 1–5, 6–10, 11–15 |
| Tamim/2010 [31] | NR | 1–2, 3–5, 6–11, 12+ | Penicillins, tetracyclines, macrolides, cephalosporins, sulfonamides, others | Prostate | 1–5, 6–10, 11–15 |
| Tamim/2011 [26] | NR | Q1, Q2, Q3, Q4 | Penicillins, tetracyclines, macrolides, cephalosporins, sulfonamides, others | Cervical, ovarian, uterine | 1–5, 6–10, 11–15 |
| Velicer/2004 [27] | NR | 1–10, 11–25, 26–50, ≥51 (1–50, 51–100, 101–500, 501–1000, 1000+ days) | Penicillins, tetracyclines, macrolides, cephalosporins, sulfonamides, nitrofurantoins | Breast | NR |
| Wang/2014 [33] | NR | Highest vs. second vs. lowest tertile (<7, 7–14, 14+ days) | Beta-lactam, cephalosporins, carbapenems, lincosamides, imidazoles, moxifloxacin | Colorectal | |
| Yang/2016 [35] | NR | 0–1, 2–4, 5–9, 10–19, 20+ | Penicillins, Cephalosporins, Monobactams, Carbapenems, Glycopeptides, Fosfomycin trometamol, Inhibitors of mycobacterial cell wall, Pyrazinamide Combo, Lipopeptide, Aminoglycosides, Tetracyclines, Macrolides, Chloramphenicol. Oxazolidonones, Sulfonamides, Dapsone, Quinolones, Metronidazole, Nitrofurantoins, Ansamycins, Rifabutin, Clofazimine | Liver | <2, 2–5, >5 |
| Zhang/2008 [30] | NR | 1–4, 5–9, ≥10 | Penicillins, tetracyclines, macrolides, quinolones, cephalosporins, sulfonamides | Lung | NR |
| Subgroup Analysis | N° | Adjusted OR (95% CI) | p | I2 | p for Hetereogeneity | Analysis |
|---|---|---|---|---|---|---|
| All antibiotic use vs. none | 25 | 1.18 (1.12–1.24) | <0.001 | 94% | <0.001 | Random |
| N° prescriptions: higher vs. none/lower | 21 | 1.28 (1.14–1.44) | <0.001 | 96% | <0.001 | Random |
| Duration of use: higher vs. lower | 6 | 1.31 (1.11–1.54) | <0.001 | 95% | <0.001 | Random |
| Diseases: | ||||||
| ➢ Breast | 10 | 1.15 (1.06–1.24) | <0.001 | 96% | <0.001 | Random |
| ➢ Colorectal | 5 | 1.08 (1.007–1.17) | 0.03 | 92% | <0.001 | Random |
| ➢ Gastric | 6 | 1.06 (1.02–1.1) | 0.001 | 51% | 0.06 | Fixed |
| ➢ Esophagus | 4 | 0.98 (0.93–1.04) | 0.6 | 0% | 0.7 | Fixed |
| ➢ Lung | 4 | 1.29 (1.03–1.61) | 0.02 | 89% | <0.001 | Random |
| ➢ Lymphoma | 4 | 1.31 (1.13–1.51) | <0.001 | 90% | <0.001 | Random |
| ➢ Central Nervous System | 2 | Not analyzed | ||||
| ➢ Pancreatic | 4 | 1.28 (1.04–1.57) | 0.019 | 89% | <0.001 | Random |
| ➢ Bladder | 3 | 1.22 (1.08–1.37) | 0.001 | 91% | <0.001 | Random |
| ➢ Renal | 3 | 1.28 (1.1–1.5) | 0.001 | 89% | <0.001 | Random |
| ➢ Prostate | 6 | 1.25 (1.1–1.41) | <0.001 | 97% | <0.001 | Random |
| ➢ Melanoma | 3 | 1.08 (1–1.17) | 0.045 | 83% | <0.001 | Random |
| ➢ Skin non melanoma | 2 | Not analyzed | ||||
| ➢ Uterine | 3 | 0.97 (0.94–1.01) | 0.3 | 4% | 0.39 | Fixed |
| ➢ Ovarian | 3 | 0.95 (0.92–0.99) | 0.027 | 0% | 0.86 | Fixed |
| ➢ Cervix | 4 | 0.75 (0.58–0.96) | 0.025 | 85% | <0.001 | Random |
| ➢ Head and neck | 2 | Not analyzed | ||||
| ➢ Liver | 4 | 1.22 (1.05.1.41) | 0.008 | 85% | <0.001 | Random |
| ➢ Biliary tract | 4 | 1.05 (1.01–1.1) | 0.009 | 20% | 0.25 | Fixed |
| ➢ Myeloma | 3 | 1.36 (1.18–1.56) | <0.001 | 76% | 0.001 | Random |
| ➢ Sarcoma | 1 | Not analyzed | ||||
| Type of antibiotics: | ||||||
| ➢ Beta-lactams | 16 | 1.15 (1.12–1.19) | <0.001 | 89% | <0.001 | Random |
| ➢ Cephalosporins | 14 | 1.19 (1.13–1.25) | <0.001 | 81% | <0.001 | Random |
| ➢ Carbapenems | 2 | Not analyzed | ||||
| ➢ Macrolides | 14 | 1.11 (1.06–1.16) | <0.001 | 69% | <0.001 | Random |
| ➢ Tetracyclines | 15 | 1.06 (1.04–1.09) | <0.001 | 66% | <0.001 | Random |
| ➢ Quinolones | 10 | 1.15 (1.09–1.21) | <0.001 | 80% | <0.001 | Random |
| ➢ Nitrofurantoins | 6 | 1.05 (0.990–1.1) | 0.01 | 24% | 0.28 | Random |
| ➢ Sulfonamides | 14 | 1.07 (1.03–1.11) | <0.001 | 74% | <0.001 | Fixed |
| ➢ Aminoglicosydes | 2 | Not analyzed | Random | |||
| ➢ Nitroimidazoles | 4 | 1.09 (1.01–1.17) | 0.015 | 54% | <0.001 | |
| ➢ Lincosamides | 2 | Not analyzed | Random | |||
| Time elapsed from antibiotic use and incident cancer | 8 | 1.14 (1.05–1.24) | 0.001 | 89 | <0.001 | Random |
| Type of study: | ||||||
| ➢ retrospective cohort | 5 | 1.16 (1.09–1.23) | <0.001 | 95% | <0.001 | Random |
| ➢ case-control | 20 | 1.18 (1.1–1.26) | <0.001 | 94% | <0.001 | Random |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Petrelli, F.; Ghidini, M.; Ghidini, A.; Perego, G.; Cabiddu, M.; Khakoo, S.; Oggionni, E.; Abeni, C.; Hahne, J.C.; Tomasello, G.; et al. Use of Antibiotics and Risk of Cancer: A Systematic Review and Meta-Analysis of Observational Studies. Cancers 2019, 11, 1174. https://doi.org/10.3390/cancers11081174
Petrelli F, Ghidini M, Ghidini A, Perego G, Cabiddu M, Khakoo S, Oggionni E, Abeni C, Hahne JC, Tomasello G, et al. Use of Antibiotics and Risk of Cancer: A Systematic Review and Meta-Analysis of Observational Studies. Cancers. 2019; 11(8):1174. https://doi.org/10.3390/cancers11081174
Chicago/Turabian StylePetrelli, Fausto, Michele Ghidini, Antonio Ghidini, Gianluca Perego, Mary Cabiddu, Shelize Khakoo, Emanuela Oggionni, Chiara Abeni, Jens Claus Hahne, Gianluca Tomasello, and et al. 2019. "Use of Antibiotics and Risk of Cancer: A Systematic Review and Meta-Analysis of Observational Studies" Cancers 11, no. 8: 1174. https://doi.org/10.3390/cancers11081174
APA StylePetrelli, F., Ghidini, M., Ghidini, A., Perego, G., Cabiddu, M., Khakoo, S., Oggionni, E., Abeni, C., Hahne, J. C., Tomasello, G., & Zaniboni, A. (2019). Use of Antibiotics and Risk of Cancer: A Systematic Review and Meta-Analysis of Observational Studies. Cancers, 11(8), 1174. https://doi.org/10.3390/cancers11081174







