Astragalus polysaccharide (PG2) Ameliorates Cancer Symptom Clusters, as well as Improves Quality of Life in Patients with Metastatic Disease, through Modulation of the Inflammatory Cascade
Abstract
1. Introduction
2. Materials and Methods
2.1. Drugs
2.2. Patient Selection and Study Design
2.3. Randomization and Blinding
2.4. Drug Safety Evaluation
2.5. Study Endpoints
2.6. Determination of Serum Inflammatory Cytokine Levels
2.7. Statistical Analysis
3. Results
3.1. Patient Characteristics
3.2. PG2 Elicits Significant Improvement in Patients’ QoL
3.3. PG2 Infusion Down-Modulate Patients’ Pro-Inflammatory Cytokine Profiles
3.4. PG2-Induced Improvement in QoL Is Associated with Changes in Patients’ Inflammatory Cytokine Profiles
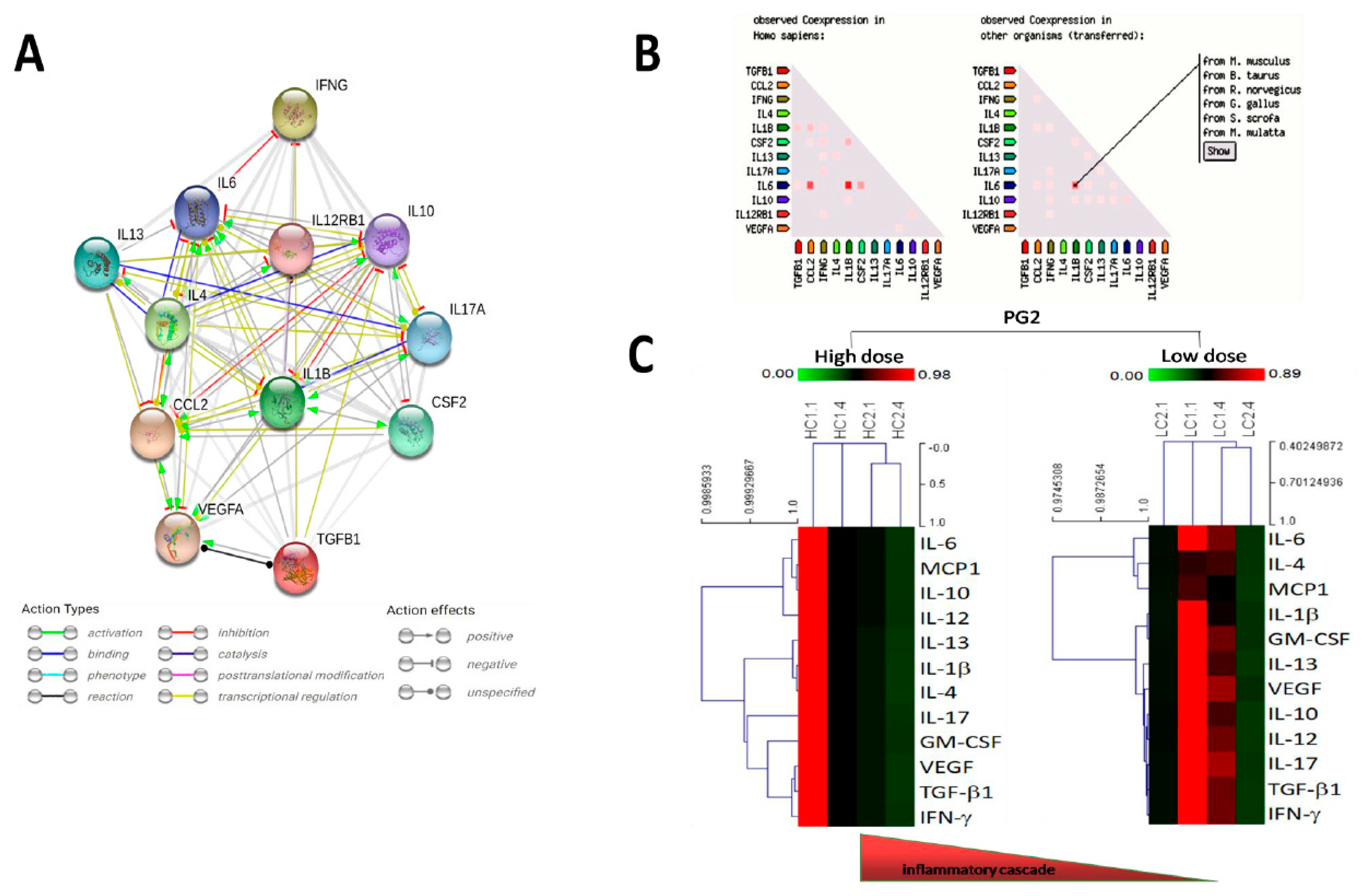
3.5. PG2, the Inflammatory Cascade, and the QoL of Patients with Metastatic Cancer
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Kirkova, J.; Aktas, A.; Walsh, D.; Rybicki, L.; Davis, M.P. Consistency of symptom clusters in advanced cancer. Am. J. Hosp. Palliat. Care 2010, 27, 342–346. [Google Scholar] [CrossRef] [PubMed]
- Yennurajalingam, S.; Kwon, J.H.; Urbauer, D.L.; Hui, D.; Reyes-Gibby, C.C.; Bruera, E. Consistency of symptom clusters among advanced cancerpatients seen at an outpatient supportive care clinic in a tertiary cancer center. Palliat. Support Care 2013, 11, 473–480. [Google Scholar] [CrossRef]
- Chen, E.; Nguyen, J.; Khan, L.; Zhang, L.; Cramarossa, G.; Tsao, M.; Danjoux, C.; Barnes, E.; Sahgal, A.; Holden, L.; et al. Symptom clusters in patients with advanced cancer: A reanalysis comparing different statistical methods. J. Pain Symptom Manag. 2012, 44, 23–32. [Google Scholar] [CrossRef]
- Dong, S.T.; Costa, D.S.; Butow, P.N.; Lovell, M.R.; Agar, M.; Velikova, G.; Teckle, P.; Tong, A.; Tebbutt, N.C.; Clarke, S.J.; et al. Symptom Clusters in Advanced Cancer Patients: An Empirical Comparison of Statistical Methods and the Impact on Quality of Life. J. Pain Symptom Manag. 2016, 51, 88–98. [Google Scholar] [CrossRef] [PubMed]
- Fallowfield, L. Quality of life: A new perspective for cancer patients. Nat. Rev. Cancer 2002, 2, 873–879. [Google Scholar] [CrossRef]
- Jiménez, A.; Madero, R.; Alonso, A.; Martínez-Marín, V.; Vilches, Y.; Martínez, B.; Feliu, M.; Díaz, L.; Espinosa, E.; Feliu, J. Symptom clusters in advanced cancer. J. Pain Symptom Manag. 2011, 42, 24–31. [Google Scholar] [CrossRef] [PubMed]
- Walsh, D.; Rybicki, L. Symptom clustering in advanced cancer. Support Care Cancer 2006, 14, 831–836. [Google Scholar] [CrossRef]
- Baldo, P.; Fornasier, G.; Ciolfi, L.; Sartor, I.; Francescon, S. Pharmacovigilance in oncology. Int. J. Clin. Pharm. 2018, 40, 832–841. [Google Scholar] [CrossRef] [PubMed]
- Rolfes, L.; van Hunsel, F.; Taxis, K.; van Puijenbroek, E. The Impact of Experiencing Adverse Drug Reactions on the Patient’s Quality of Life: A Retrospective Cross-Sectional Study in the Netherlands. Drug Saf. 2016, 39, 769–776. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.S.; Shi, Q.; Williams, L.A.; Mao, L.; Cleeland, C.S.; Komaki, R.R.; Mobley, G.M.; Liao, Z. Inflammatory cytokines are associated with the development of symptom burden in patients with NSCLC undergoing concurrent chemoradiation therapy. Brain Behav. Immun. 2010, 24, 968–974. [Google Scholar] [CrossRef]
- Seruga, B.; Zhang, H.; Bernstein, L.J.; Tannock, I.F. Cytokines and their relationship to the symptoms and outcome of cancer. Nat. Rev. Cancer 2008, 8, 887–899. [Google Scholar] [CrossRef] [PubMed]
- Paulsen, Ø.; Laird, B.; Aass, N.; Lea, T.; Fayers, P.; Kaasa, S.; Klepstad, P. The relationship between pro-inflammatory cytokines and pain, appetite and fatigue in patients with advanced cancer. PLoS ONE 2017, 12, 0177620. [Google Scholar] [CrossRef] [PubMed]
- Liu, Q.Y.; Yao, Y.M.; Zhang, S.W.; Sheng, Z.Y. Astragalus polysaccharides regulate T cell-mediated immunity via CD11c(high)CD45RB (low) DCs in vitro. J. Ethnopharmacol. 2011, 136, 457–464. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Dong, B.; Tan, Y.; Yu, S.; Bao, Y.-X. A study on the immunomodulation of polysaccharopeptide through the TLR4-TIRAP/MAL-MyD88 signaling pathway in PBMCs from breast cancer patients. Immunopharmacol. Immunotoxicol. 2013, 35, 497–504. [Google Scholar] [CrossRef] [PubMed]
- Feng, Z.; Wang, Z.; Yang, M.; Zhou, L.; Bao, Y. Polysaccharopeptide exerts immunoregulatory effects via MyD88-dependent signaling pathway. Int. J. Boil. Macromol. 2016, 82, 201–207. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.H.; Lin, C.Y.; Chen, J.S.; Ho, C.L.; Rau, K.M.; Tsai, J.T.; Chang, C.S.; Yeh, S.P.; Cheng, C.F.; Lai, Y.L. Karnofsky Performance Status as A Predictive Factor for Cancer-Related Fatigue Treatment with Astragalus Polysaccharides (PG2) Injection-A Double Blind, Multi-Center, Randomized Phase IV Study. Cancers 2019, 11, 128. [Google Scholar] [CrossRef]
- Prigerson, H.G.; Bao, Y.; Shah, M.A.; Paulk, M.E.; Leblanc, T.W.; Schneider, B.J.; Garrido, M.M.; Reid, M.C.; Berlin, D.A.; Adelson, K.B.; et al. Chemotherapy Use, Performance Status, and Quality of Life at the End of Life. JAMA Oncol. 2015, 1, 778–784. [Google Scholar] [CrossRef]
- Ribeiro, M.R.; Motta, A.A.; Marcondes-Fonseca, L.A.; Kalil-Filho, J.; Giavina-Bianchi, P. Increase of 10% in the Rate of Adverse Drug Reactions for Each Drug Administered in Hospitalized Patients. Clin. Sao Paulo 2018, 73, e185. [Google Scholar] [CrossRef]
- Chopra, D.; Rehan, H.S.; Sharma, V.; Mishra, R. Chemotherapy-induced adverse drug reactions in oncology patients: A prospective observational survey. Indian J. Med Paediatr. Oncol. 2016, 37, 42–46. [Google Scholar] [CrossRef]
- Laird, B.J.; Kaasa, S.; McMillan, D.C.; Fallon, M.T.; Hjermstad, M.J.; Fayers, P.; Klepstad, P. Prognostic Factors in Patients with Advanced Cancer: A Comparison of Clinicopathological Factors and the Development of an Inflammation-Based Prognostic System. Clin. Cancer Res. 2013, 19, 5456–5464. [Google Scholar] [CrossRef]
- Laird, B.J.; Scott, A.C.; Colvin, L.A.; McKeon, A.-L.; Murray, G.D.; Fearon, K.C.; Fallon, M.T. Pain, Depression, and Fatigue as a Symptom Cluster in Advanced Cancer. J. Pain Symptom Manag. 2011, 42, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Trajkovic-Vidakovic, M.; De Graeff, A.; Voest, E.E.; Teunissen, S.C. Symptoms tell it all: A systematic review of the value of symptom assessment to predict survival in advanced cancer patients. Crit. Rev. Oncol. 2012, 84, 130–148. [Google Scholar] [CrossRef] [PubMed]
- Laird, B.J.; McMillan, D.C.; Fayers, P.; Fearon, K.; Kaasa, S.; Fallon, M.T.; Klepstad, P. The Systemic Inflammatory Response and Its Relationship to Pain and Other Symptoms in Advanced Cancer. Oncologist 2013, 18, 1050–1055. [Google Scholar] [CrossRef] [PubMed]
- Giesinger, J.M.; Wintner, L.M.; Zabernigg, A.; Gamper, E.-M.; Oberguggenberger, A.S.; Sztankay, M.J.; Kemmler, G.; Holzner, B. Assessing quality of life on the day of chemotherapy administration underestimates patients’ true symptom burden. BMC Cancer 2014, 14, 758. [Google Scholar] [CrossRef] [PubMed]
- Laird, B.J.; Fallon, M.; Hjermstad, M.J.; Tuck, S.; Kaasa, S.; Klepstad, P.; McMillan, D.C. Quality of Life in Patients with Advanced Cancer: Differential Association With Performance Status and Systemic Inflammatory Response. J. Clin. Oncol. 2016, 34, 2769–2775. [Google Scholar] [CrossRef] [PubMed]
| Variable | Astragalus Polysaccharides (PG2) Dosage | |
|---|---|---|
| High (500 mg) | Low (250 mg) | |
| Gender (n,%) | ||
| Male | 3 (27.27%) | 7 (58.33%) |
| Female | 8 (72.73%) | 5 (41.67%) |
| Age (years) | ||
| Mean (SD) | 67.27 (12.31) | 63.67 (14.65) |
| Medial (IQR) | 64 (20) | 62 (20.5) |
| 95% CI | (59.00–75.54) | (54.36–72.97) |
| Body mass index (BMI)(kg/m2) | ||
| Mean (SD) | 21.22 (3.38) | 21.08 (4.13) |
| Medial (IQR) | 21.17 (5) | 20.67 (5.81) |
| 95% CI | (18.95–23.50) | (18.45–23.70) |
| Baseline BFI score (Cycle 1 visit 1) | ||
| Mean (SD) | 8.37 (1.18) | 7.79 (1.67) |
| Medial (IQR) | 8.30 (2.20) | 8.25 (2.70) |
| 95% CI | (7.58–9.16) | (6.72–8.87) |
| Baseline Karnofsky Performance status score (Cycle 1 visit 1) | ||
| Mean (SD) | 70.71 (10.44) | 65.00 (15.08) |
| Medial (IQR) | 70 (20) | 70 (25) |
| 95% CI | (63.89–77.93) | (55.42–74.58) |
| Domains | PG2 | Baseline | Visit 1 | Visit 2 | Visit 3 | p-value for change |
|---|---|---|---|---|---|---|
| Pain | High | 48.48 ± 26.30 | 26.67 ± 27.89 | 21.78 ± 22.77 | 15.00 ± 16.67 | 0.038 |
| Low | 50.00 ± 36.24 | 34.44 ± 17.21 | 23.33 ± 17.89 | 13.33 ± 33.33 | 0.014 | |
| Sleep | High | 78.79 ± 22.47 | 40.00 ± 43.46 | 33.33 ± 31.62 | 8.33 ± 16.67 | 0.003 |
| Low | 56.94 ± 41.72 | 44.44 ± 17.21 | 35.33 ± 34.56 | 22.22 ± 19.25 | 0.057 | |
| Appetite | High | 66.67 ± 29.81 | 33.33 ± 23.57 | 30.56 ± 19.48 | 16.67 ± 19.25 | 0.013 |
| Low | 68.06 ± 34.42 | 38.89 ± 25.09 | 30.00 ± 34.56 | 22.22 ± 38.49 | 0.095 | |
| Nausea | High | 27.27 ± 25.03 | 13.33 ± 18.26 | 10.89 ± 19.48 | 6.89 ± 19.48 | 0.046 |
| Low | 30.56 ± 28.28 | 22.22 ± 27.22 | 16.67 ± 43.46 | 16.67 ± 43.46 | 0.093 | |
| Vomiting | High | 12.12 ± 23.68 | 7.33 ± 18.26 | 5.56 ± 13.61 | 0.00 ± 0 | 0.363 |
| Low | 11.11 ± 16.41 | 11.11 ± 17.21 | 20.00 ± 44.72 | 0.00 ± 0 | 0.841 | |
| Fatigue | High | 86.36 ± 17.98 | 40.00 ± 14.91 | 33.33 ± 14.91 | 8.33 ± 16.67 | 0.003 |
| Low | 86.11 ± 21.12 | 33.33 ± 21.08 | 43.33 ± 32.49 | 22.22 ± 19.25 | 0.08 | |
| Global QoL | High | 30.30 ± 15.49 | 56.67 ± 25.28 | 70.83 ± 13.69 | 83.33 ± 13.69 | 0.012 |
| Low | 24.31 ± 18.28 | 61.11 ± 17.21 | 60.00 ± 25.95 | 77.78 ± 9.62 | 0.02 | |
| Total score | High | 54.82 ± 8.06 | 39.09 ± 10.62 | 38.63 ± 8.07 | 31.06 ± 6.72 | 0.006 |
| Low | 50.56 ± 9.11 | 43.68 ± 5.28 | 44.54 ± 17.78 | 34.34 ± 6.99 | 0.004 |
| Cytokines | Baseline | Visit 1 | Visit 2 | Visit 3 | p-value for change |
|---|---|---|---|---|---|
| IL-1β | 155.5 ± 44.3 | 131.8 ± 40.4 | 119.02 ± 2.15 | 104.4 ± 3.8 | 0.014 |
| IL-4 | 143.1 ± 28.6 | 132.4 ± 39.9 | 120.54 ± 4.3 | 102.8 ± 2.7 | 0.04 |
| IL-6 | 147.4 ± 28 | 133.1 ± 39.4 | 122.15 ± 1.16 | 103.6 ± 3.2 | 0.012 |
| MCP-1 | 143.5 ± 28.3 | 131.2 ± 40.9 | 121.63 ± 0.42 | 103.6 ± 3.3 | 0.017 |
| IL-10 | 152.6 ± 45.3 | 132.4 ± 39.9 | 122.78 ± 2.06 | 103.6 ± 3.3 | 0.019 |
| IL-12 | 151.1 ± 46.4 | 133 ± 39.5 | 122.62 ± 1.82 | 104.1 ± 2.8 | 0.03 |
| IL-13 | 154.3 ± 46.1 | 132.4 ± 39.9 | 120.00 ± 3.54 | 102.8 ± 2.8 | 0.025 |
| IL-17 | 153.4 ± 44.9 | 133.7 ± 38.9 | 120.23 ± 3.87 | 102.9 ± 2.7 | 0.018 |
| GM-CSF | 160.3 ± 45.8 | 133 ± 39.5 | 119.54 ± 2.88 | 105.2 ± 3 | 0.014 |
| VEGF | 153.7 ± 44.8 | 133.6 ± 39 | 120.23 ± 3.87 | 105.6 ± 3.6 | 0.023 |
| TGF-β1 | 153.3 ± 44.9 | 132.9 ± 39.5 | 120.17 ± 3.78 | 105.1 ± 3.2 | 0.024 |
| IFN-γ | 151.3 ± 46 | 133 ± 39.5 | 120.17 ± 3.77 | 104.7 ± 2.7 | 0.031 |
| Cytokines | Univariate | Multivariate | ||
|---|---|---|---|---|
| β | Pr > |t| | β | Pr > |t| | |
| IL-1β | 0.043 | 0.0004 | 0.2531 | 0.0013 |
| IL-4 | 0.051 | 0.0006 | ||
| IL-6 | 0.051 | 0.0002 | ||
| MCP-1 | 0.046 | 0.0007 | ||
| IL-10 | 0.037 | 0.0017 | ||
| IL-12 | 0.034 | 0.0041 | ||
| IL-13 | 0.04 | 0.0013 | −0.085 | 0.0442 |
| IL-17 | 0.041 | 0.001 | ||
| GM-CSF | 0.047 | <0.001 | 0.1266 | 0.002 |
| VEGF | 0.041 | 0.0009 | ||
| TGF-β1 | 0.041 | 0.0011 | ||
| IFN-γ | 0.038 | 0.002 | ||
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Huang, W.-C.; Kuo, K.-T.; Bamodu, O.A.; Lin, Y.-K.; Wang, C.-H.; Lee, K.-Y.; Wang, L.-S.; Yeh, C.-T.; Tsai, J.-T. Astragalus polysaccharide (PG2) Ameliorates Cancer Symptom Clusters, as well as Improves Quality of Life in Patients with Metastatic Disease, through Modulation of the Inflammatory Cascade. Cancers 2019, 11, 1054. https://doi.org/10.3390/cancers11081054
Huang W-C, Kuo K-T, Bamodu OA, Lin Y-K, Wang C-H, Lee K-Y, Wang L-S, Yeh C-T, Tsai J-T. Astragalus polysaccharide (PG2) Ameliorates Cancer Symptom Clusters, as well as Improves Quality of Life in Patients with Metastatic Disease, through Modulation of the Inflammatory Cascade. Cancers. 2019; 11(8):1054. https://doi.org/10.3390/cancers11081054
Chicago/Turabian StyleHuang, Wen-Chien, Kuang-Tai Kuo, Oluwaseun Adebayo Bamodu, Yen-Kuang Lin, Chun-Hua Wang, Kang-Yun Lee, Liang-Shun Wang, Chi-Tai Yeh, and Jo-Ting Tsai. 2019. "Astragalus polysaccharide (PG2) Ameliorates Cancer Symptom Clusters, as well as Improves Quality of Life in Patients with Metastatic Disease, through Modulation of the Inflammatory Cascade" Cancers 11, no. 8: 1054. https://doi.org/10.3390/cancers11081054
APA StyleHuang, W.-C., Kuo, K.-T., Bamodu, O. A., Lin, Y.-K., Wang, C.-H., Lee, K.-Y., Wang, L.-S., Yeh, C.-T., & Tsai, J.-T. (2019). Astragalus polysaccharide (PG2) Ameliorates Cancer Symptom Clusters, as well as Improves Quality of Life in Patients with Metastatic Disease, through Modulation of the Inflammatory Cascade. Cancers, 11(8), 1054. https://doi.org/10.3390/cancers11081054






