Development of A New Mouse Model for Intrahepatic Cholangiocellular Carcinoma: Accelerating Functions of Pecam-1
Abstract
1. Introduction
2. Results
2.1. Gross Anatomy and Microscopy of Liver in WT- and Pecam-1-KO-Mice after TAA-Administration
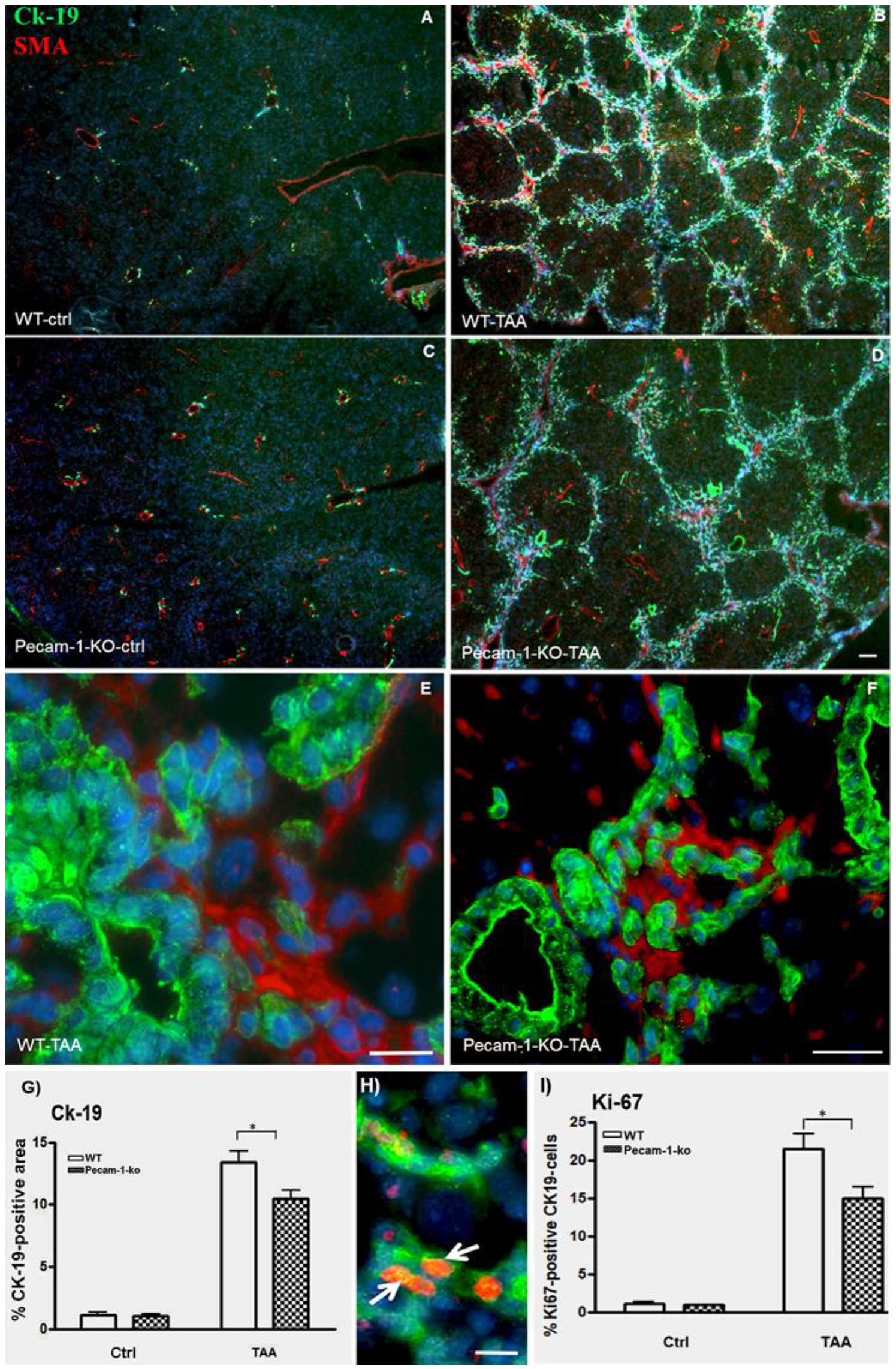
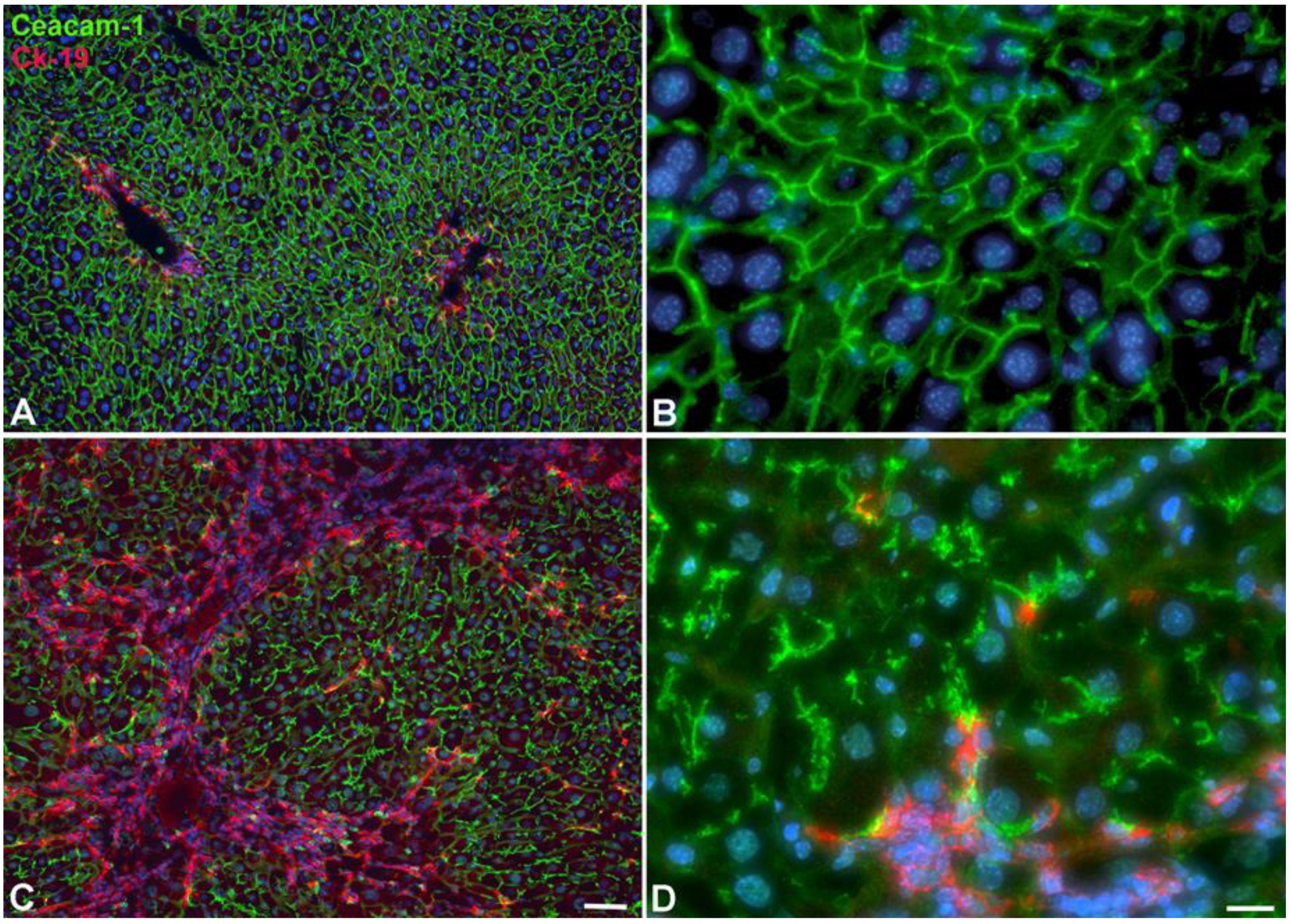
2.2. Immunohistological Identification of ICC in Murine Liver after TAA-Administration
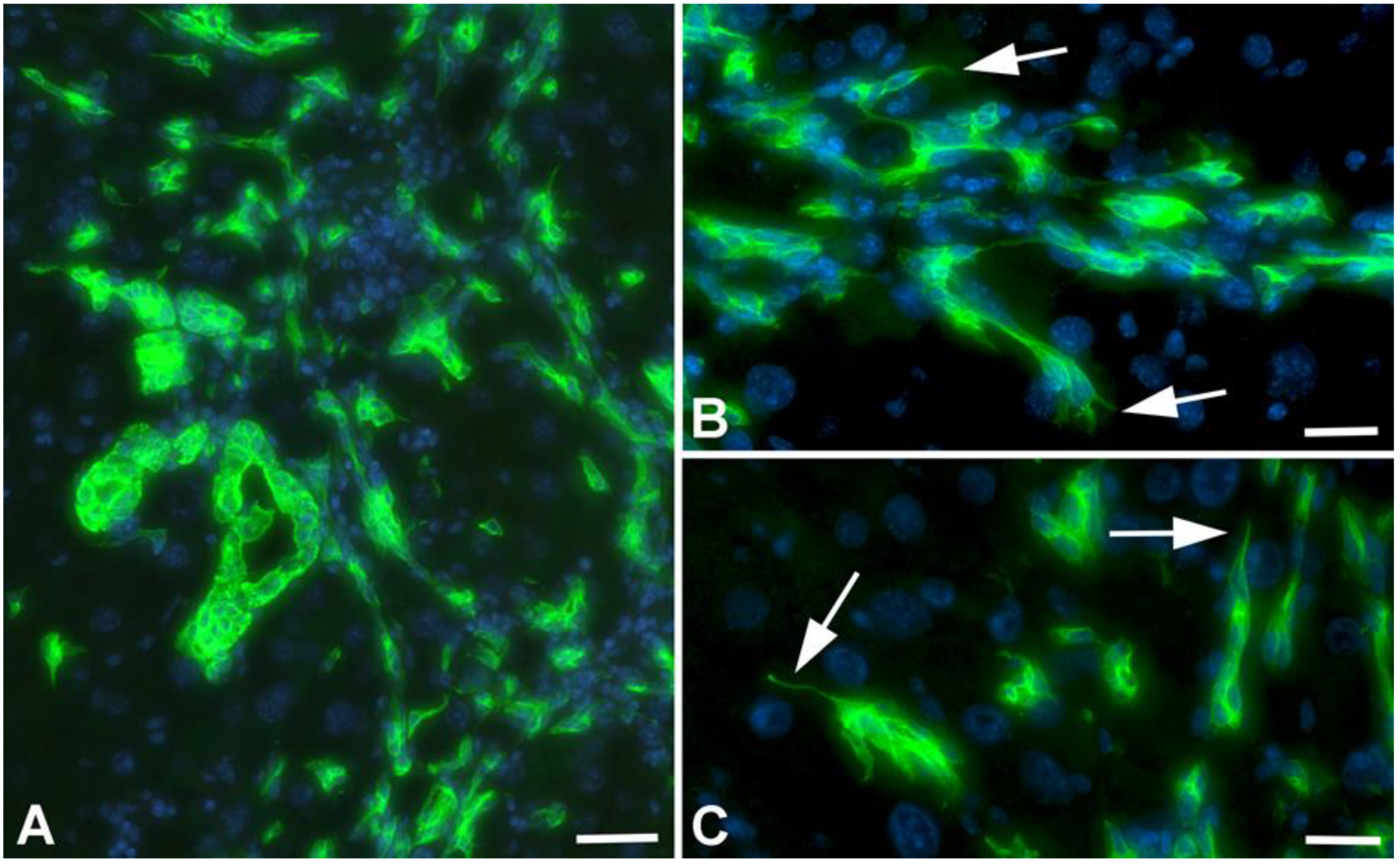
2.3. Infiltration of Neutrophilic Granulocytes in Liver after TAA-Administration
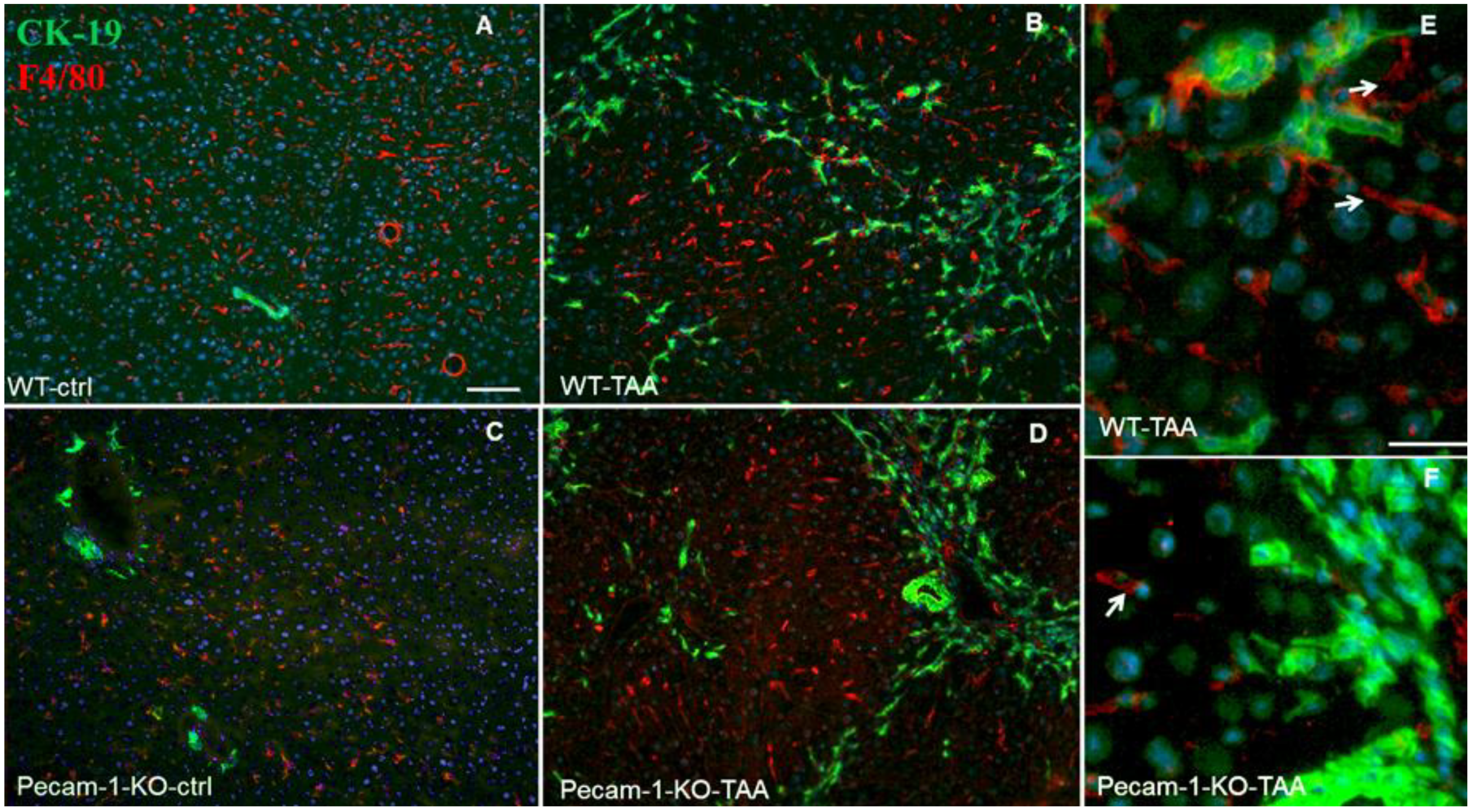
2.4. Localization of Macrophages in Liver after TAA-Administration
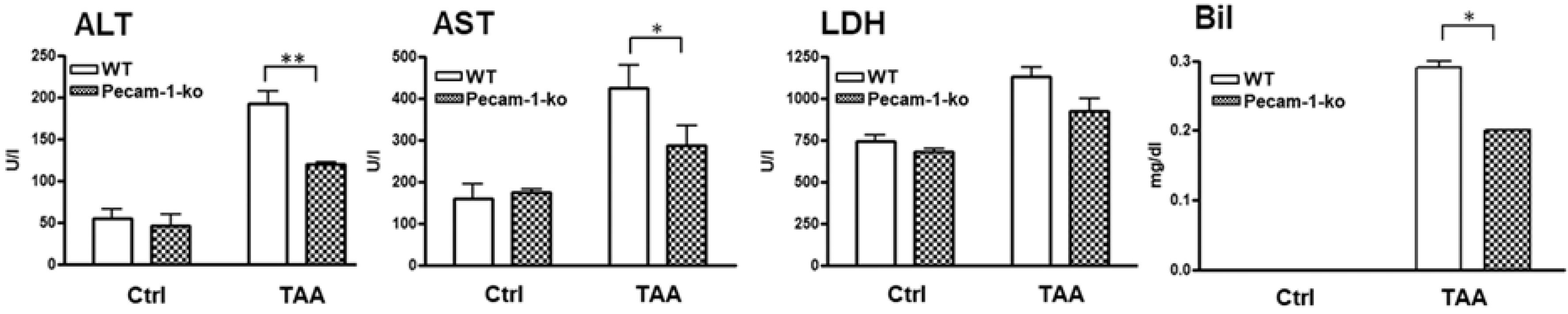
2.5. Elevated Liver Markers in Serum of WT and Pecam-1 KO Mice after TAA Administration
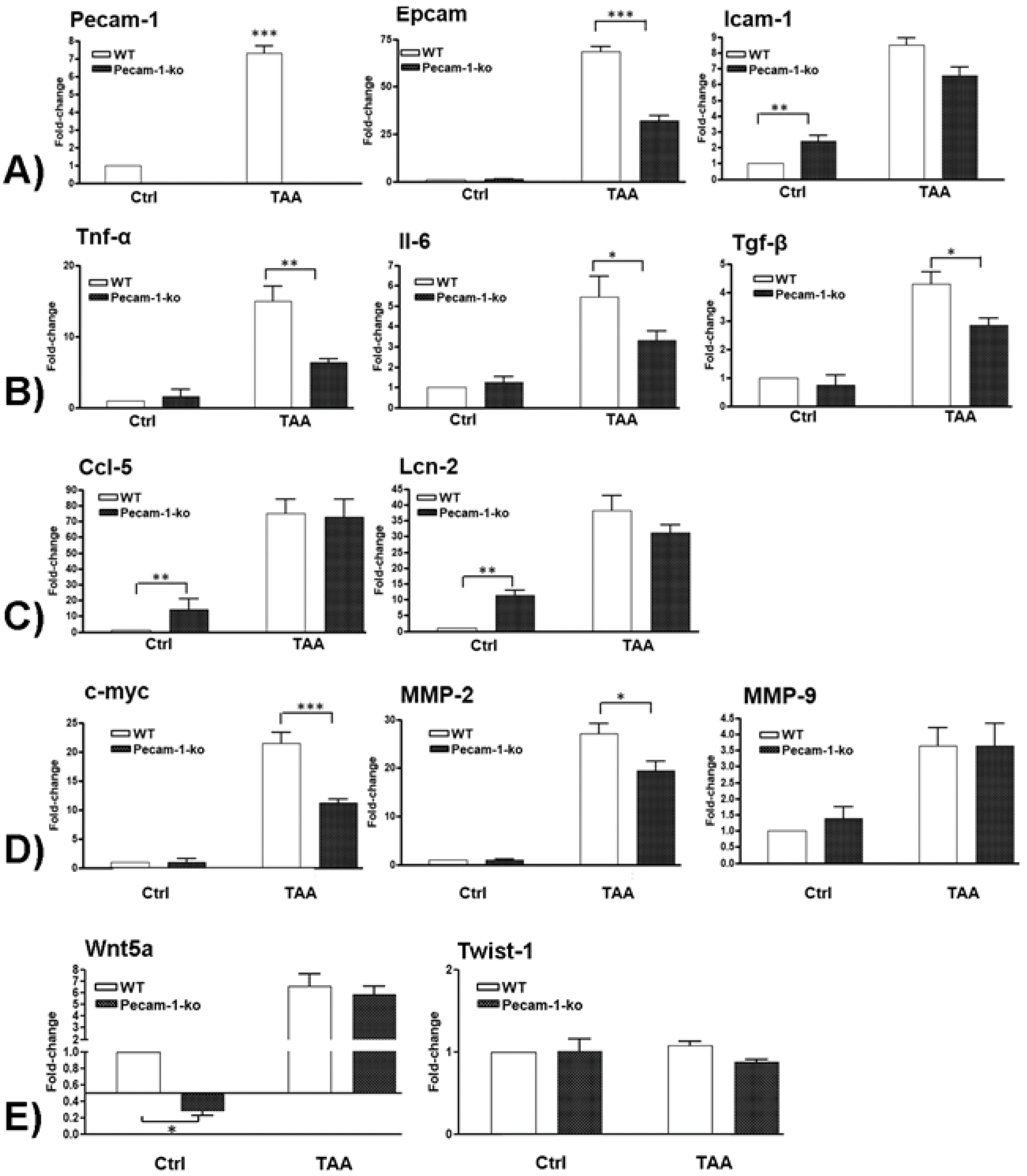
2.6. Changes in Hepatic Gene Expression of Adhesion Molecules
2.7. Changes in Hepatic Gene Expression of Pro-Inflammatory Cytokines
2.8. Changes in Hepatic Gene Expression of Ccl5 and Lcn-2
2.9. Changes in Hepatic Gene Expression of c-Myc, Mmp-2, and Mmp9
2.10. Changes in Hepatic Gene Expression of EMT Markers
3. Discussion
3.1. Animal Models of Intrahepatic Cholangiocellular Carcinoma
3.2. Role of PECAM-1 in Inflammation-Induced Cancer
4. Material and Methods
4.1. Materials
4.2. Animals
4.3. Quantification of Hepatic Markers in Murine Serum
4.4. Immunofluorescence of Murine Liver Sections
4.5. RNA Isolation and Real-Time PCR Analysis
- Pecam-1: forward, AACAGAAACCCGTGGAGATG, reverse, GTCTCTGTGGCTCTCGTTCC;
- Icam-1: forward, ATTCGTTTCCGGAGAGTGTG, reverse, CAGCACCGTGAATGTGATCT;
- EpCAM: forward, GCTGTCATTGTGGTGGTGTC, reverse, TGGATCTCACCCATCTCCTT;
- Tgf-β: forward, TTGCTTCAAGCTCCACAGAGA, reverse, CAGAAGTTGGCATGGTAGCC;
- Tnf-α: forward, CAAACCACCAAGTGGAGGAG, reverse, GTGGGTGAGGAGCACGTAGT;
- Il-6: forward, CAAAGCCAGAGTCCTTCAGAG, reverse, GAGCATTGGAAATTGGGGTA;
- Ccl5/Lix: forward, GGTCCACAGTGCCCTACG, reverse, GCGAGTGCATTCCGCTTA;
- Lcn-2: forward, AAATTGCACAGGTATCCTCAG, reverse, CAGAGAAGATGATGTTGTCGT;
- C-Myc: forward, CGGACACACAACGTCTTGGAA, reverse, AGGATGTAGGCGGTGGCTTTT;
- Mmp-2: forward, GCTGTATTCCCGACCGTTG, reverse, TGGTCCGCGTAAAGTATGGG;
- Mmp-9: forward, ACGACATAGACGGCATCCAGTATC, reverse, AGGTATAGTGGCACACATAGTGGG;
- Wnt-5a: forward, AAGCAGGCCGTAGGACAGTA, reverse, CAGCACGTCTTGAGGCTACA;
- Twist-1: forward, GAGCAAGATTCAGACCCTCAA, reverse, CATCTTGGAGTCCAGCTCGT;
- B2m: forward, ATTCACCCCCACTGAGACTG, reverse, ATCCCAGTAGACGGTCTTGG;
- GAPDH: forward, AGAACATCATCCCTGCATCC, reverse, CACATTGGGGGTAGGAACAC.
4.6. Statistical Analysis
4.7. Data Availability
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Massarweh, N.N.; El-Serag, H.B. Epidemiology of Hepatocellular Carcinoma and Intrahepatic Cholangiocarcinoma. Cancer Control 2017, 24, 1–11. [Google Scholar]
- Kulik, L.; El-Serag, H.B. Epidemiology and Management of Hepatocellular Carcinoma. Gastroenterology 2019, 156, 477–491.e1. [Google Scholar] [PubMed]
- American Cancer Society. 2018. Available online: https://www.cancer.org/cancer/bile-duct-cancer/detection-diagnosis-staging/survival-by-stage.html (accessed on 22 July 2019).
- Kobayashi, S.; Werneburg, N.W.; Bronk, S.F.; Kaufmann, S.H.; Gores, G.J. Interleukin-6 Contributes to Mcl-1 Up-regulation and TRAIL Resistance via an Akt-Signaling Pathway in Cholangiocarcinoma Cells. Gastroenterology 2005, 128, 2054–2065. [Google Scholar] [PubMed]
- Ehling, J.; Tacke, F. Role of chemokine pathways in hepatobiliary cancer. Cancer Lett. 2016, 379, 173–183. [Google Scholar] [PubMed]
- Nguyen, T.; Bui, K.; Scholta, T.; Riebold, M.; Xing, J.; Bhuria, V.; Nguyen, L.; Le, H.; Velavan, T.; Beckert, S.; et al. The role of IL-6 signaling pathway in cholangiocarcinoma. J. Hepatol. 2018, 68, S684–S685. [Google Scholar]
- Yang, J.W.; Murray, B.; Barbier-Torres, L.; Liu, T.; Liu, Z.; Yang, H.; Fan, W.; Wang, J.; Li, Y.; Seki, E.; et al. The mitochondrial chaperone Prohibitin 1 negatively regulates interleukin-8 in human liver cancers. J. Biol. Chem. 2019, 294, 1984–1996. [Google Scholar]
- Heymann, F.; Tacke, F. Immunology in the liver—From homeostasis to disease. Nat. Rev. Gastroenterol. Hepatol. 2016, 13, 88–110. [Google Scholar] [PubMed]
- Vestweber, D. How leukocytes cross the vascular endothelium. Nat. Rev. Immunol. 2015, 15, 692–704. [Google Scholar] [PubMed]
- Privratsky, J.R.; Newman, D.K.; Newman, P.J. PECAM-1: Conflicts of Interest in Inflammation. Life Sci. 2010, 87, 69–82. [Google Scholar]
- Privratsky, J.R.; Tilkens, S.B.; Newman, D.K.; Newman, P.J. PECAM-1 dampens cytokine levels during LPS-induced endotoxemia by regulating leukocyte trafficking. Life Sci. 2012, 90, 177–184. [Google Scholar]
- Paddock, C.; Zhou, D.; Lertkiatmongkol, P.; Newman, P.J.; Zhu, J. Structural basis for PECAM-1 homophilic binding. Blood 2016, 127, 1052–1061. [Google Scholar] [PubMed]
- Thompson, R.D.; Noble, K.E.; Larbi, K.Y.; Dewar, A.; Duncan, G.S.; Mak, T.W.; Nourshargh, S. Platelet-endothelial cell adhesion molecule-1 (PECAM-1)-deficient mice demonstrate a transient and cytokine-specific role for PECAM-1 in leukocyte migration through the perivascular basement membrane. Blood 2001, 97, 1854–1860. [Google Scholar] [PubMed]
- Delisser, H.; Liu, Y.; Desprez, P.-Y.; Thor, A.; Briasouli, P.; Handumrongkul, C.; Wilfong, J.; Yount, G.; Nosrati, M.; Fong, S.; et al. Vascular endothelial platelet endothelial cell adhesion molecule 1 (PECAM-1) regulates advanced metastatic progression. Proc. Natl. Acad. Sci. USA 2010, 107, 18616–18621. [Google Scholar] [PubMed]
- Aroca, F.; Renaud, W.; Bartoli, C.; Bouvier-Labit, C.; Figarella-Branger, D. Expression of PECAM-1/CD31 Isoforms in Human Brain Gliomas. J. Neuro Oncol. 1999, 43, 19–25. [Google Scholar]
- Ledingham, M.A.; Thomson, A.J.; Jordan, F.; Young, A.; Crawford, M.; Norman, J.E. Cell Adhesion Molecule Expression in the Cervix and Myometrium during Pregnancy and Parturition. Obstet. Gynecol. 2001, 97, 235–242. [Google Scholar]
- Anastassiou, G.; Duensing, S.; Steinhoff, G.; Zorn, U.; Grosse, J.; Dallmann, I.; Kirchner, H.; Ganser, A.; Atzpodien, J. Platelet Endothelial Cell Adhesion Molecule-1 (PECAM-1): A Potential Prognostic Marker Involved in Leukocyte Infiltration of Renal Cell Carcinoma. Oncology 1996, 53, 127–132. [Google Scholar]
- Zhou, Z.; Christofidou-Solomidou, M.; Garlanda, C.; Delisser, H.M. Antibody against murine PECAM-1 inhibits tumor angiogenesis in mice. Angiogenesis 1999, 3, 181–188. [Google Scholar]
- Righi, L.; Deaglio, S.; Pecchioni, C.; Gregorini, A.; Horenstein, A.L.; Bussolati, G.; Sapino, A.; Malavasi, F. Role of CD31/Platelet Endothelial Cell Adhesion Molecule-1 Expression in in Vitro and in vivo Growth and Differentiation of Human Breast Cancer Cells. Am. J. Pathol. 2003, 162, 1163–1174. [Google Scholar]
- Abraham, V.; Cao, G.; Parambath, A.; Lawal, F.; Handumrongkul, C.; Debs, R.; Delisser, H.M. Involvement of TIMP-1 in PECAM-1-mediated tumor dissemination. Int. J. Oncol. 2018, 53, 488–502. [Google Scholar]
- Ko, K.S.; Peng, J.; Yang, H. Animal models of cholangiocarcinoma. Curr. Opin. Gastroenterol. 2013, 29, 312–318. [Google Scholar]
- Hajovsky, H.; Hu, G.; Koen, Y.; Sarma, D.; Cui, W.; Moore, D.S.; Staudinger, J.L.; Hanzlik, R.P.; Hajovsky, L. Metabolism and Toxicity of Thioacetamide and Thioacetamide S-Oxide in Rat Hepatocytes. Chem. Res. Toxicol. 2012, 25, 1955–1963. [Google Scholar]
- Amanzada, A.; Moriconi, F.; Mansuroglu, T.; Cameron, S.; Ramadori, G.; Malik, I.A. Induction of chemokines and cytokines before neutrophils and macrophage recruitment in different regions of rat liver after TAA administration. Lab. Investig. 2014, 94, 235–247. [Google Scholar] [PubMed]
- Malik, G.; Wilting, J.; Hess, C.F.; Ramadori, G.; Malik, I.A. PECAM-1 modulates liver damage induced by synergistic effects of TNF-α and irradiation. J. Cell. Mol. Med. 2019, 23, 3336–3344. [Google Scholar] [PubMed]
- Malik, I.A.; Stange, I.; Martius, G.; Cameron, S.; Rave-Fränk, M.; Hess, C.F.; Ellenrieder, V.; Wolff, H.A. Role of PECAM-1 in radiation-induced liver inflammation. J. Cell. Mol. Med. 2015, 19, 2441–2452. [Google Scholar] [PubMed]
- Moriconi, F.; Malik, I.A.; Amanzada, A.; Blaschke, M.; Raddatz, D.; Khan, S.; Ramadori, G. The anti-TNF-α antibody infliximab indirectly regulates PECAM-1 gene expression in two models of in vitro blood cell activation. Lab. Investig. 2012, 92, 166–177. [Google Scholar] [PubMed]
- Yang, H.; Li, T.W.; Peng, J.; Tang, X.; Ko, K.S.; Xia, M.; Aller, M. A Mouse Model of Cholestasis-Associated Cholangiocarcinoma and Transcription Factors Involved in Progression. Gastroenterology 2011, 141, 378–388.e4. [Google Scholar] [PubMed]
- Ikenoue, T.; Terakado, Y.; Nakagawa, H.; Hikiba, Y.; Fujii, T.; Matsubara, D.; Noguchi, R.; Zhu, C.; Yamamoto, K.; Kudo, Y.; et al. A novel mouse model of intrahepatic cholangiocarcinoma induced by liver-specific Kras activation and Pten deletion. Sci. Rep. 2016, 6, 23899. [Google Scholar]
- Mansuroglu, T.; Ramadori, P.; Dudás, J.; Malik, I.; Hammerich, K.; Füzesi, L.; Ramadori, G. Expression of stem cell factor and its receptor c-Kit during the development of intrahepatic cholangiocarcinoma. Lab. Investig. 2009, 89, 562–574. [Google Scholar]
- Dejana, E. The Role of Wnt Signaling in Physiological and Pathological Angiogenesis. Circ. Res. 2010, 107, 943–952. [Google Scholar]
- Lutze, G.; Haarmann, A.; Toukam, J.A.D.; Buttler, K.; Wilting, J.; Becker, J. Non-canonical WNT-signaling controls differentiation of lymphatics and extension lymphangiogenesis via RAC and JNK signaling. Sci. Rep. 2019, 9, 4739. [Google Scholar]
- Linke, F.; Zaunig, S.; Nietert, M.M.; von Bonin, F.; Lutz, S.; Dullin, C.; Janovská, P.; Beissbarth, T.; Alves, F.; Klapper, W.; et al. WNT5A: A motility-promoting factor in Hodgkin lymphoma. Oncogene 2017, 36, 13–23. [Google Scholar] [PubMed]
- Vijgen, S.; Terris, B.; Rubbia-Brandt, L. Pathology of intrahepatic cholangiocarcinoma. Hepatobiliary Surg. Nutr. 2017, 6, 22–34. [Google Scholar] [PubMed]
- Janan, M.; Proungvitaya, S.; Limpaiboon, T.; Proungvitaya, T.; Roytrakul, S.; Wongkham, C.; Jearanaikoon, P.; Chur-In, S.; Wongkham, S. Serum adhesion molecule-1 (ICAM-1) as a potential prognostic marker for cholangiocarcinoma patients. Asian Pac. J. Cancer Prev. 2012, 13, 107–114. [Google Scholar] [PubMed]
- Sulpice, L.; Rayar, M.; Turlin, B.; Boucher, E.; Bellaud, P.; Desille, M.; Meunier, B.; Clément, B.; Boudjema, K.; Coulouarn, C. Epithelial cell adhesion molecule is a prognosis marker for intrahepatic cholangiocarcinoma. J. Surg. Res. 2014, 192, 117–123. [Google Scholar] [PubMed]
- Wang, J.; Xie, C.; Pan, S.; Liang, Y.; Han, J.; Lan, Y.; Sun, J.; Li, K.; Yang, G.; Shi, H.; et al. N-myc downstream-regulated gene 2 inhibits human cholangiocarcinoma progression and is regulated by leukemia inhibitory factor/MicroRNA-181c negative feedback pathway. Hepatology 2016, 64, 1606–1622. [Google Scholar] [PubMed]
- Liu, T.; Yang, H.; Fan, W.; Tu, J.; Li, T.W.; Wang, J.; Shen, H.; Yang, J.; Xiong, T.; Steggerda, J.; et al. Mechanisms of MAFG Dysregulation in Cholestatic Liver Injury and Development of Liver Cancer. Gastroenterology 2018, 155, 557–571.e14. [Google Scholar] [PubMed]
- Page-McCaw, A.; Ewald, A.J.; Werb, Z. Matrix metalloproteinases and the regulation of tissue remodelling. Nat. Rev. Mol. Cell Boil. 2007, 8, 221–233. [Google Scholar]
- Itatsu, K.; Sasaki, M.; Yamaguchi, J.; Ohira, S.; Ishikawa, A.; Ikeda, H.; Sato, Y.; Harada, K.; Zen, Y.; Sato, H.; et al. Cyclooxygenase-2 Is Involved in the Up-Regulation of Matrix Metalloproteinase-9 in Cholangiocarcinoma Induced by Tumor Necrosis Factor-α. Am. J. Pathol. 2009, 174, 829–841. [Google Scholar]
- Sachs, U.J.H.; Andrei-Selmer, C.L.; Maniar, A.; Weiss, T.; Paddock, C.; Orlova, V.V.; Choi, E.Y.; Newman, P.J.; Preissner, K.T.; Chavakis, T.; et al. The Neutrophil-specific Antigen CD177 Is a Counter-receptor for Platelet Endothelial Cell Adhesion Molecule-1 (CD31). J. Boil. Chem. 2007, 282, 23603–23612. [Google Scholar]
- Malik, I.A.; Moriconi, F.; Sheikh, N.; Naz, N.; Khan, S.; Dudas, J.; Mansuroglu, T.; Hess, C.F.; Rave-Fränk, M.; Christiansen, H.; et al. Single-dose gamma-irradiation induces up-regulation of chemokine gene expression and recruitment of granulocytes into the portal area but not into other regions of rat hepatic tissue. Am. J. Pathol. 2010, 176, 1801–1815. [Google Scholar]
- Aldinucci, D.; Casagrande, N. Inhibition of the CCL5/CCR5 Axis against the Progression of Gastric Cancer. Int. J. Mol. Sci. 2018, 19, 1477. [Google Scholar]
- Borkham-Kamphorst, E.; Van De Leur, E.; Haas, U.; Weiskirchen, R. Liver parenchymal cells lacking Lipocalin 2 (LCN2) are prone to endoplasmic reticulum stress and unfolded protein response. Cell. Signal. 2019, 55, 90–99. [Google Scholar] [PubMed]
- Giese, M.A.; Hind, L.E.; Huttenlocher, A. Neutrophil plasticity in the tumor microenvironment. Blood 2019, 133, 2159–2167. [Google Scholar] [PubMed]
- Venkataramani, V.; Küffer, S.; Cheung, K.C.P.; Jiang, X.; Trümper, L.; Wulf, G.G.; Ströbel, P. CD31 Expression Determines Redox Status and Chemoresistance in Human Angiosarcomas. Clin. Cancer Res. 2018, 24, 460–473. [Google Scholar] [PubMed]
- Raskopf, E.; Carmona, M.A.G.; Van Cayzeele, C.J.; Strassburg, C.; Sauerbruch, T.; Schmitz, V. Toxic Damage Increases Angiogenesis and Metastasis in Fibrotic Livers via PECAM-1. BioMed Res. Int. 2014, 2014. [Google Scholar] [CrossRef]
- Ouyang, J.-S.; Li, Y.-P.; Chen, C.-S.; Chen, J.-J.; Chen, T.-K.; Cai, C.; Yang, L. Inhibition of lung tumor growth in nude mice by siRNACD31 targeting PECAM-1. Oncol. Lett. 2014, 8, 33–40. [Google Scholar] [PubMed]
- Schneider, C.A.; Rasband, W.S.; Eliceiri, K.W. NIH Image to ImageJ: 25 years of image analysis. Nat. Methods 2012, 9, 671–675. [Google Scholar] [PubMed]



| Mouse | Liver Disease | Stage | Inflammation Grade |
|---|---|---|---|
| WT | Cirrhosis | 4 | 1–2 |
| WT | Cirrhosis | 4 | 1–2 |
| WT | Cirrhosis | 4 | 1–2 |
| WT | Cirrhosis | 4 | 1–2 |
| WT | Cirrhosis | 4 | 1–2 |
| WT | Cirrhosis | 4 | 1–2 |
| Pecam-1-KO | Cirrhosis | 3 | 1 |
| Pecam-1-KO | Cirrhosis | 3 | 1 |
| Pecam-1-KO | Cirrhosis | 3 | 1 |
| Pecam-1-KO | Cirrhosis | 3 | 1 |
| Pecam-1-KO | Cirrhosis | 3 | 1 |
| Pecam-1-KO | Cirrhosis | 3 | 2 |
| Antibody | Company | Cat. Number | IHC |
|---|---|---|---|
| rb-anti-ms-CK-19 | Abcam | ab 76539 | 0.180556 |
| rt-anti-F4/80 (BM8) | Abcam | ab 16911 | 1:10 |
| rt-anti-ms Ly-6G/ly-6C (Gr-1) | Invitrogen | 14-5931-81 | 0.180556 |
| sh-anti-ms-CEACAM-1 | R &D systems | AF-6480 | 0.111111 |
| ms-anti-SMA | Sigma | A-2547 | 0.180556 |
| Secondary Antibodies: | |||
| gt-anti-rt | |||
| gt-anti-rb | Molecular Probes | --- | 0.180556 |
| dk-anti-rb | |||
| dk-anti-sh | Dianova | 713-225-147 | 0.180556 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Malik, I.A.; Malik, G.; Ströbel, P.; Wilting, J. Development of A New Mouse Model for Intrahepatic Cholangiocellular Carcinoma: Accelerating Functions of Pecam-1. Cancers 2019, 11, 1045. https://doi.org/10.3390/cancers11081045
Malik IA, Malik G, Ströbel P, Wilting J. Development of A New Mouse Model for Intrahepatic Cholangiocellular Carcinoma: Accelerating Functions of Pecam-1. Cancers. 2019; 11(8):1045. https://doi.org/10.3390/cancers11081045
Chicago/Turabian StyleMalik, Ihtzaz Ahmed, Gesa Malik, Philipp Ströbel, and Jörg Wilting. 2019. "Development of A New Mouse Model for Intrahepatic Cholangiocellular Carcinoma: Accelerating Functions of Pecam-1" Cancers 11, no. 8: 1045. https://doi.org/10.3390/cancers11081045
APA StyleMalik, I. A., Malik, G., Ströbel, P., & Wilting, J. (2019). Development of A New Mouse Model for Intrahepatic Cholangiocellular Carcinoma: Accelerating Functions of Pecam-1. Cancers, 11(8), 1045. https://doi.org/10.3390/cancers11081045





