Wnt-11 as a Potential Prognostic Biomarker and Therapeutic Target in Colorectal Cancer
Abstract
1. Introduction
2. Results
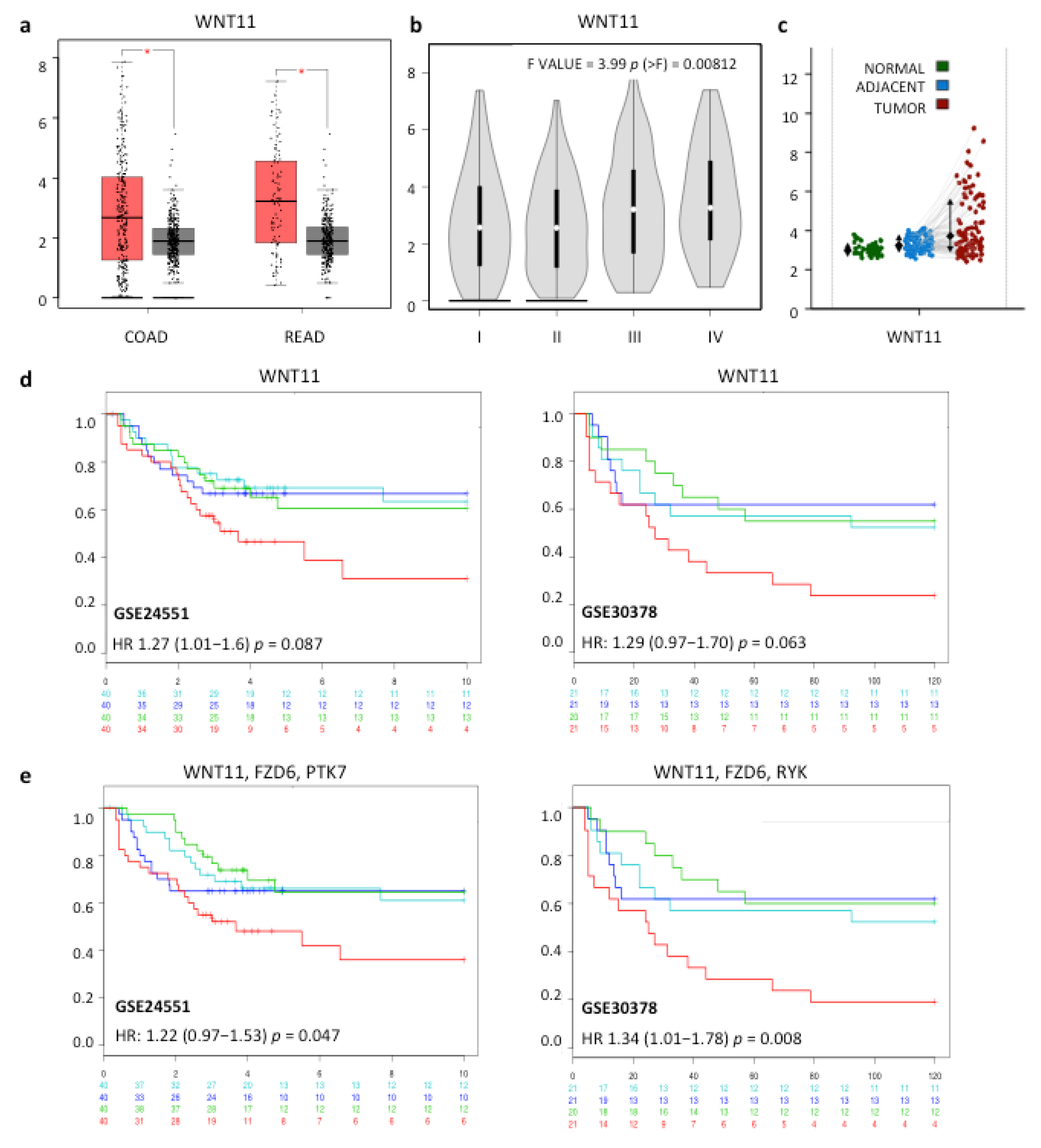
2.1. Increased Expression of WNT11 and Potential Wnt-11 Receptors is Associated with Poor Prognosis in CRC
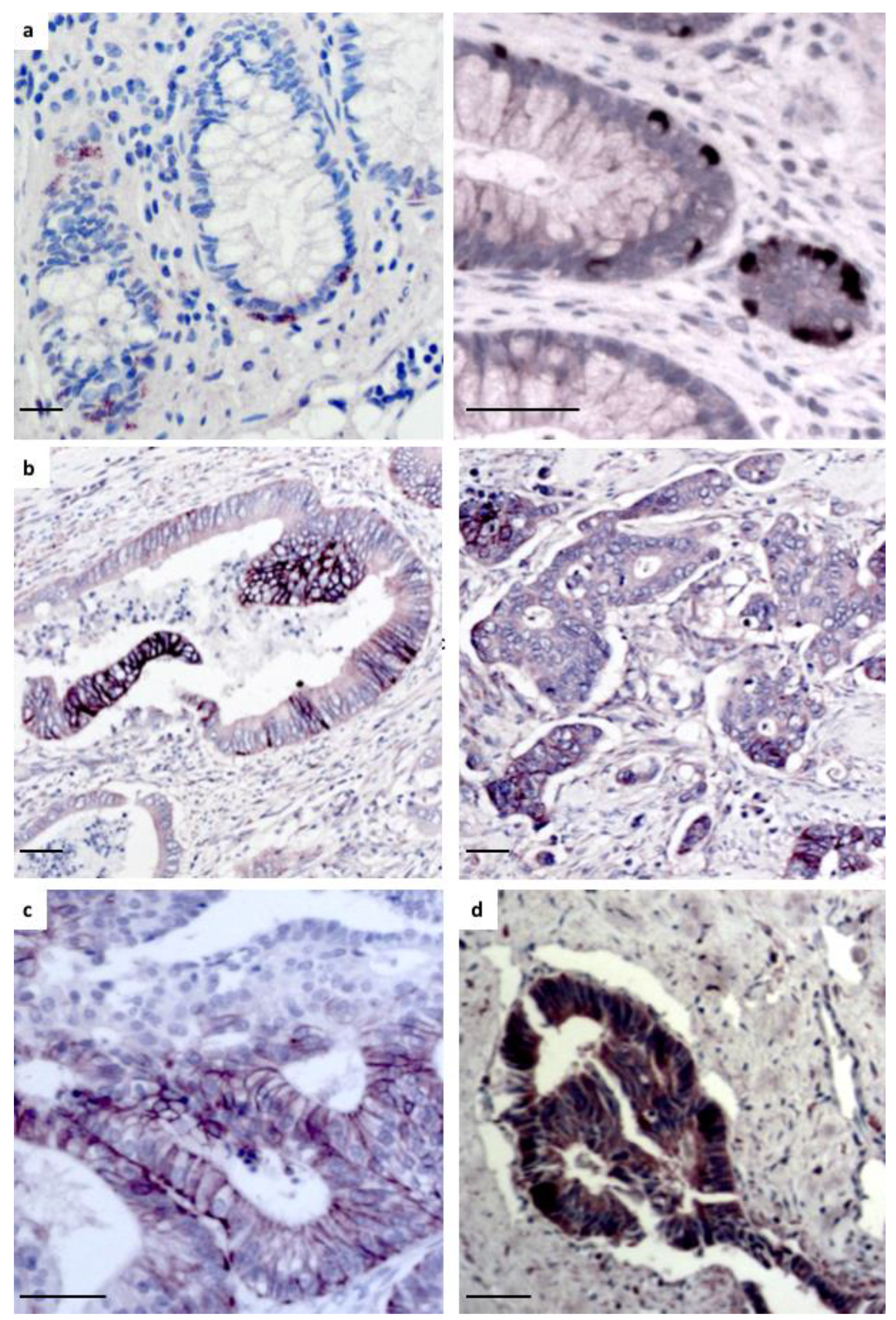
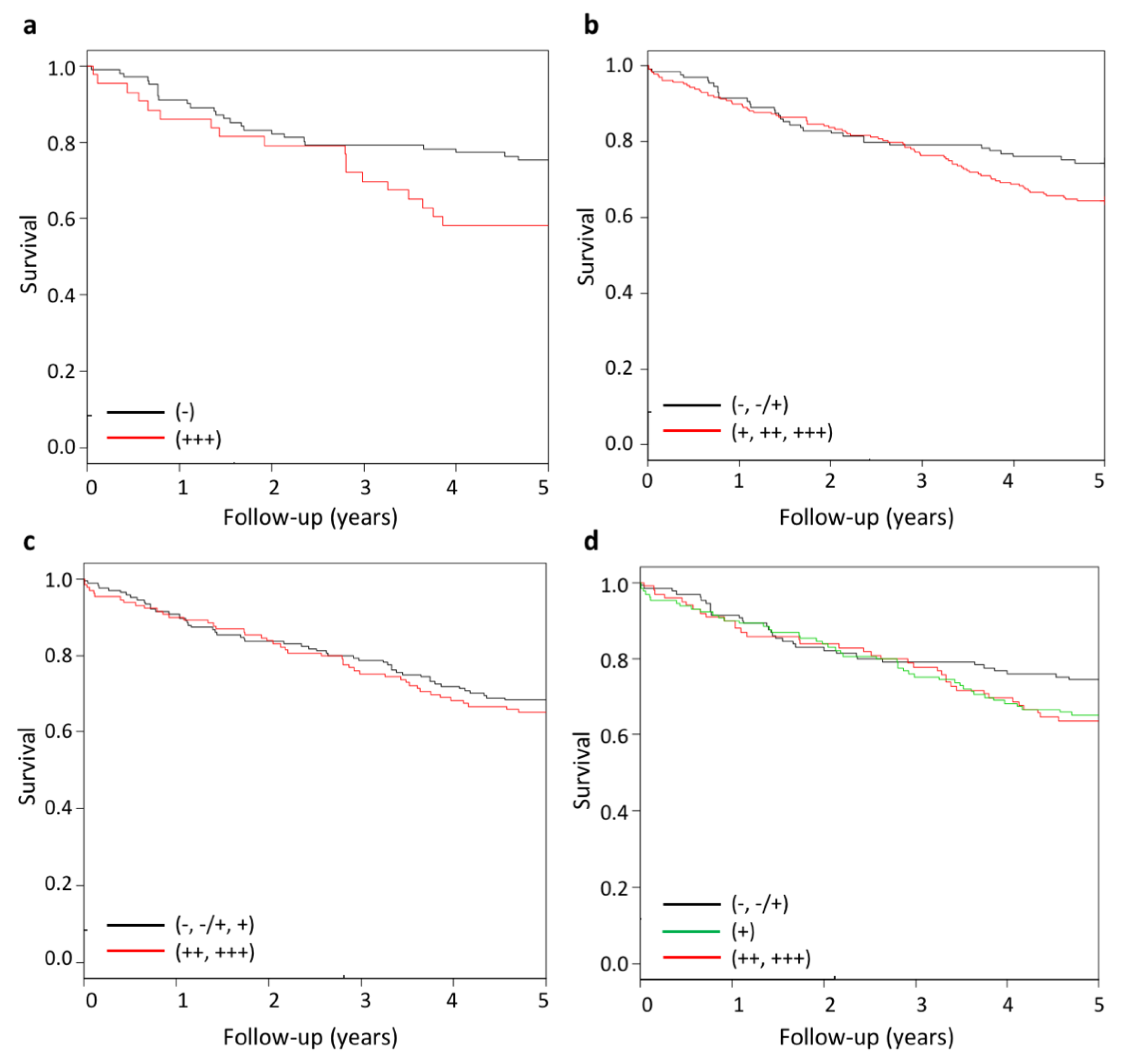
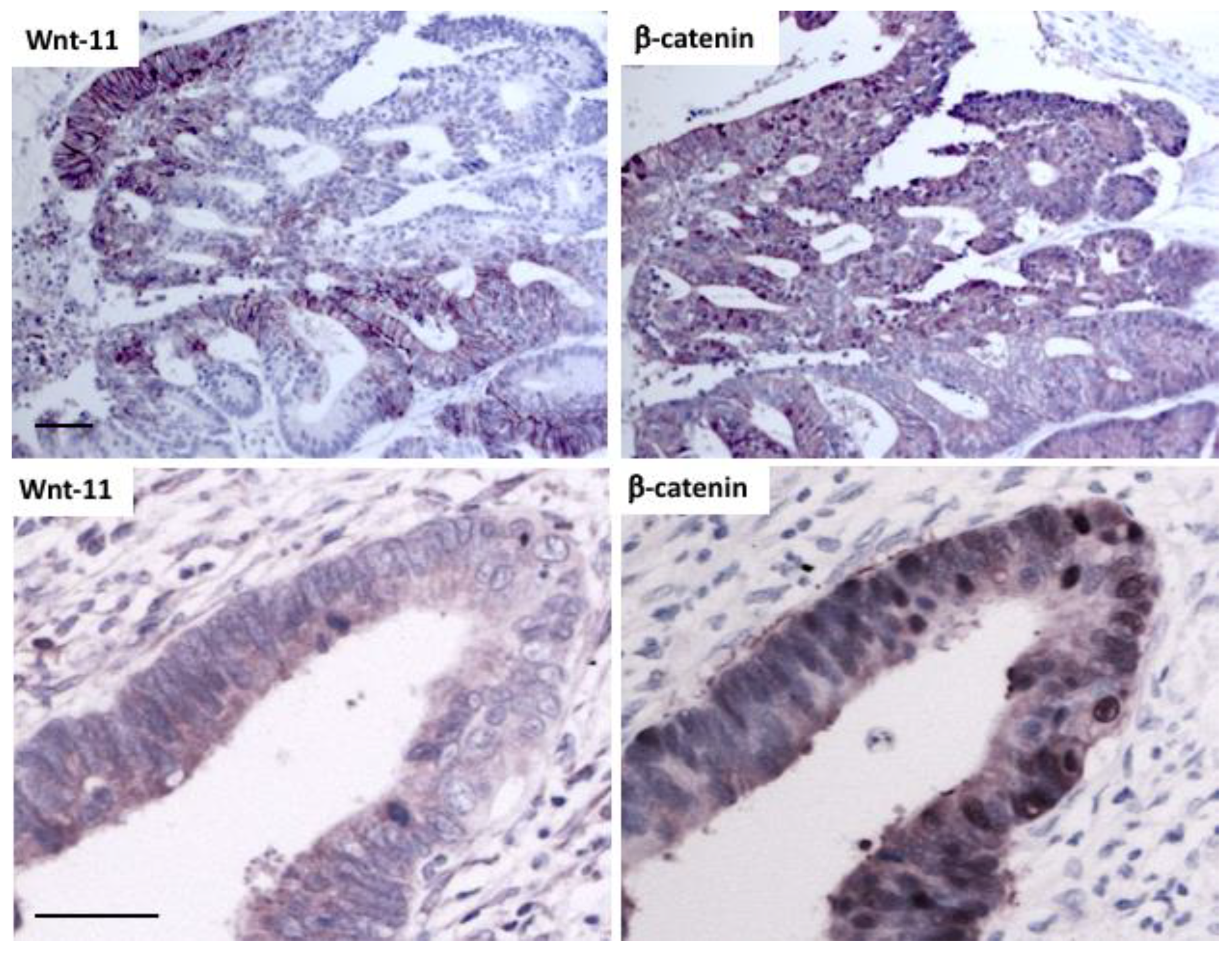
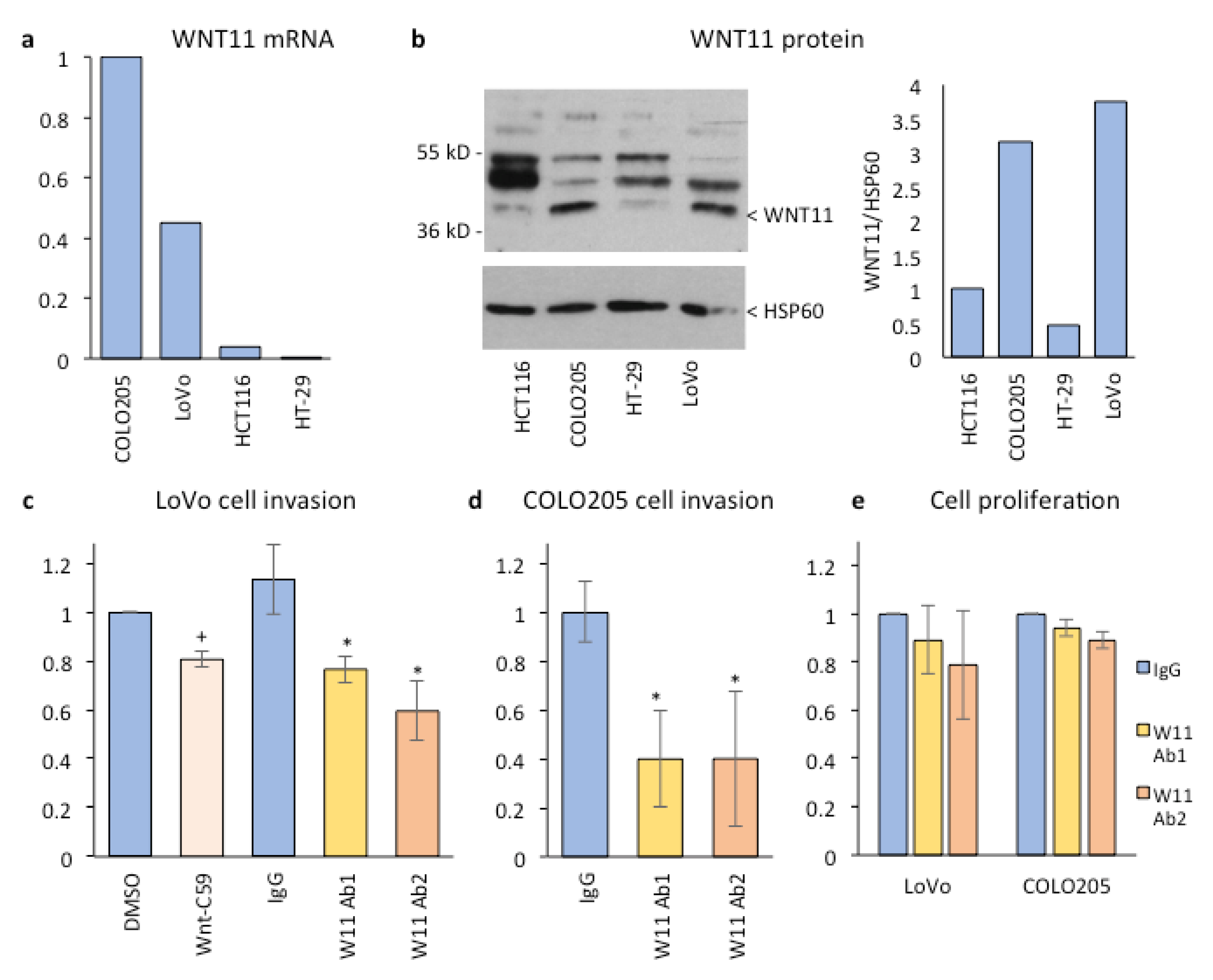
2.2. Increased Wnt-11 Protein Expression Associates with Poor Prognosis in Colorectal Cancer.
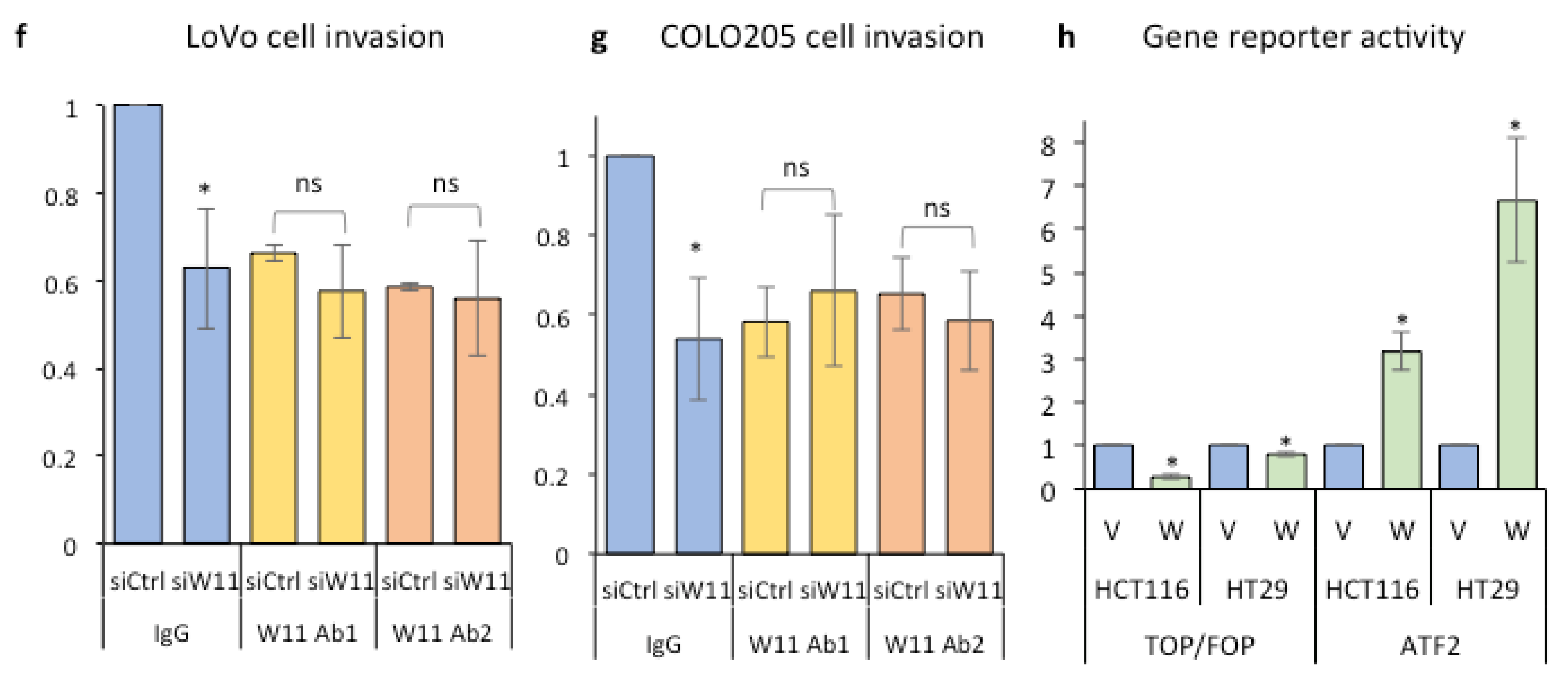
2.3. Inhibition of Wnt-11 Reduces Colorectal Cancer Cell Invasion
3. Discussion
4. Materials and Methods
4.1. Cell Culture and Reagents
4.2. RNA Extraction, cDNA Synthesis, and Quantitative Real Time PCR
4.3. Protein Extraction and Western Blotting
4.4. Clinical Samples
4.5. Immunohistochemistry
4.6. Invasion and Proliferation Assays
4.7. Transient Transfections and Gene Reporter Assays
4.8. Bioinformatics Analysis of Gene Expression Data
4.9. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Uysal-Onganer, P.; Kypta, R.M. Wnt11 in 2011—The regulation and function of a non-canonical Wnt. Acta Physiol. (Oxf.) 2012, 204, 52–64. [Google Scholar] [CrossRef] [PubMed]
- Mori, H.; Yao, Y.; Learman, B.S.; Kurozumi, K.; Ishida, J.; Ramakrishnan, S.K.; Overmyer, K.A.; Xue, X.; Cawthorn, W.P.; Reid, M.A.; et al. Induction of WNT11 by hypoxia and hypoxia-inducible factor-1alpha regulates cell proliferation, migration and invasion. Sci. Rep. 2016, 6, 21520. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Wu, S.; Xia, Y.; Li, X.E.; Xia, Y.; Zhou, Z.D.; Sun, J. Wingless homolog Wnt11 suppresses bacterial invasion and inflammation in intestinal epithelial cells. Am. J. Physiol. Gastrointest. Liver Physiol. 2011, 301, G992–G1003. [Google Scholar] [CrossRef] [PubMed]
- Neumann, P.A.; Koch, S.; Hilgarth, R.S.; Perez-Chanona, E.; Denning, P.; Jobin, C.; Nusrat, A. Gut commensal bacteria and regional Wnt gene expression in the proximal versus distal colon. Am. J. Pathol. 2014, 184, 592–599. [Google Scholar] [CrossRef] [PubMed]
- Malcomson, F.C.; Willis, N.D.; McCallum, I.; Xie, L.; Kelly, S.; Bradburn, D.M.; Belshaw, N.J.; Johnson, I.T.; Mathers, J.C. Adherence to the World Cancer Research Fund/American Institute for Cancer Research cancer prevention recommendations and WNT-pathway-related markers of bowel cancer risk. Br. J. Nutr. 2018, 10, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Nishioka, M.; Ueno, K.; Hazama, S.; Okada, T.; Sakai, K.; Suehiro, Y.; Okayama, N.; Hirata, H.; Oka, M.; Imai, K.; et al. Possible involvement of Wnt11 in colorectal cancer progression. Mol. Carcinog. 2013, 52, 207–217. [Google Scholar] [CrossRef] [PubMed]
- Ouko, L.; Ziegler, T.R.; Gu, L.H.; Eisenberg, L.M.; Yang, V.W. Wnt11 signaling promotes proliferation, transformation, and migration of IEC6 intestinal epithelial cells. J. Biol. Chem. 2004, 279, 26707–26715. [Google Scholar] [CrossRef]
- Dwyer, M.A.; Joseph, J.D.; Wade, H.E.; Eaton, M.L.; Kunder, R.S.; Kazmin, D.; Chang, C.Y.; McDonnell, D.P. WNT11 expression is induced by estrogen-related receptor alpha and beta-catenin and acts in an autocrine manner to increase cancer cell migration. Cancer Res. 2010, 70, 9298–9308. [Google Scholar] [CrossRef]
- Tian, S.; Hu, J.; Tao, K.; Wang, J.; Chu, Y.; Li, J.; Liu, Z.; Ding, X.; Xu, L.; Li, Q.; et al. Secreted AGR2 promotes invasion of colorectal cancer cells via Wnt11-mediated non-canonical Wnt signaling. Exp. Cell. Res. 2018, 364, 198–207. [Google Scholar] [CrossRef]
- Ueno, K.; Hiura, M.; Suehiro, Y.; Hazama, S.; Hirata, H.; Oka, M.; Imai, K.; Dahiya, R.; Hinoda, Y. Frizzled-7 as a potential therapeutic target in colorectal cancer. Neoplasia 2008, 10, 697–705. [Google Scholar] [CrossRef]
- Voloshanenko, O.; Gmach, P.; Winter, J.; Kranz, D.; Boutros, M. Mapping of Wnt-Frizzled interactions by multiplex CRISPR targeting of receptor gene families. FASEB J. 2017, 31, 4832–4844. [Google Scholar] [CrossRef] [PubMed]
- Dijksterhuis, J.P.; Baljinnyam, B.; Stanger, K.; Sercan, H.O.; Ji, Y.; Andres, O.; Rubin, J.S.; Hannoush, R.N.; Schulte, G. Systematic mapping of WNT-FZD protein interactions reveals functional selectivity by distinct WNT-FZD pairs. J. Biol. Chem. 2015, 290, 6789–6798. [Google Scholar] [CrossRef] [PubMed]
- Voloshanenko, O.; Schwartz, U.; Kranz, D.; Rauscher, B.; Linnebacher, M.; Augustin, I.; Boutros, M. beta-catenin-independent regulation of Wnt target genes by RoR2 and ATF2/ATF4 in colon cancer cells. Sci. Rep. 2018, 8, 3178. [Google Scholar] [CrossRef] [PubMed]
- Schwab, R.H.M.; Amin, N.; Flanagan, D.J.; Johanson, T.M.; Phesse, T.J.; Vincan, E. Wnt is necessary for mesenchymal to epithelial transition in colorectal cancer cells. Dev. Dyn. 2018, 247, 521–530. [Google Scholar] [CrossRef] [PubMed]
- Murillo-Garzon, V.; Gorrono-Etxebarria, I.; Akerfelt, M.; Puustinen, M.C.; Sistonen, L.; Nees, M.; Carton, J.; Waxman, J.; Kypta, R.M. Frizzled-8 integrates Wnt-11 and transforming growth factor-beta signaling in prostate cancer. Nat. Commun. 2018, 9, 1747. [Google Scholar] [CrossRef] [PubMed]
- Lhoumeau, A.C.; Martinez, S.; Boher, J.M.; Monges, G.; Castellano, R.; Goubard, A.; Doremus, M.; Poizat, F.; Lelong, B.; de Chaisemartin, C.; et al. Overexpression of the Promigratory and Prometastatic PTK7 Receptor Is Associated with an Adverse Clinical Outcome in Colorectal Cancer. PLoS ONE 2015, 10, e0123768. [Google Scholar] [CrossRef] [PubMed]
- Tian, X.; Yan, L.; Zhang, D.; Guan, X.; Dong, B.; Zhao, M.; Hao, C. PTK7 overexpression in colorectal tumors: Clinicopathological correlation and prognosis relevance. Oncol. Rep. 2016, 36, 1829–1836. [Google Scholar] [CrossRef][Green Version]
- Zhou, J.K.; Zheng, Y.Z.; Liu, X.S.; Gou, Q.; Ma, R.; Guo, C.L.; Croce, C.M.; Liu, L.; Peng, Y. ROR1 expression as a biomarker for predicting prognosis in patients with colorectal cancer. Oncotarget 2017, 8, 32864–32872. [Google Scholar] [CrossRef]
- Tang, Z.; Li, C.; Kang, B.; Gao, G.; Li, C.; Zhang, Z. GEPIA: A web server for cancer and normal gene expression profiling and interactive analyses. Nucleic Acids Res. 2017, 45, W98–W102. [Google Scholar] [CrossRef]
- Cordero, D.; Sole, X.; Crous-Bou, M.; Sanz-Pamplona, R.; Pare-Brunet, L.; Guino, E.; Olivares, D.; Berenguer, A.; Santos, C.; Salazar, R.; et al. Large differences in global transcriptional regulatory programs of normal and tumor colon cells. BMC Cancer 2014, 14, 708. [Google Scholar] [CrossRef]
- Sveen, A.; Agesen, T.H.; Nesbakken, A.; Rognum, T.O.; Lothe, R.A.; Skotheim, R.I. Transcriptome instability in colorectal cancer identified by exon microarray analyses: Associations with splicing factor expression levels and patient survival. Genome Med. 2011, 3, 32. [Google Scholar] [CrossRef]
- Agesen, T.H.; Sveen, A.; Merok, M.A.; Lind, G.E.; Nesbakken, A.; Skotheim, R.I.; Lothe, R.A. ColoGuideEx: A robust gene classifier specific for stage II colorectal cancer prognosis. Gut 2012, 61, 1560–1567. [Google Scholar] [CrossRef]
- Aguirre-Gamboa, R.; Gomez-Rueda, H.; Martinez-Ledesma, E.; Martinez-Torteya, A.; Chacolla-Huaringa, R.; Rodriguez-Barrientos, A.; Tamez-Pena, J.G.; Trevino, V. SurvExpress: An online biomarker validation tool and database for cancer gene expression data using survival analysis. PLoS ONE 2013, 8, e74250. [Google Scholar] [CrossRef]
- Uysal-Onganer, P.; Kawano, Y.; Caro, M.; Walker, M.M.; Diez, S.; Darrington, R.S.; Waxman, J.; Kypta, R.M. Wnt-11 promotes neuroendocrine-like differentiation, survival and migration of prostate cancer cells. Mol. Cancer 2010, 9, 55. [Google Scholar] [CrossRef]
- Ying, J.; Li, H.; Yu, J.; Ng, K.M.; Poon, F.F.; Wong, S.C.; Chan, A.T.; Sung, J.J.; Tao, Q. WNT5A exhibits tumor-suppressive activity through antagonizing the Wnt/beta-catenin signaling, and is frequently methylated in colorectal cancer. Clin. Cancer Res. 2008, 14, 55–61. [Google Scholar] [CrossRef]
- Zhang, Y.; Lin, L.; Jin, Y.; Lin, Y.; Cao, Y.; Zheng, C. Overexpression of WNT5B promotes COLO 205 cell migration and invasion through the JNK signaling pathway. Oncol. Rep. 2016, 36, 23–30. [Google Scholar] [CrossRef]
- Gorroño-Etxebarria, I.; Sanchez, S.; Kypta, R.M. Personal observation. 2019. [Google Scholar]
- Richards, M.H.; Seaton, M.S.; Wallace, J.; Al-Harthi, L. Porcupine is not required for the production of the majority of Wnts from primary human astrocytes and CD8+ T cells. PLoS ONE 2014, 9, e92159. [Google Scholar] [CrossRef]
- Rao, D.M.; Bordeaux, E.K.; Yamamoto, T.M.; Bitler, B.G.; Sikora, M.J. WNT4 and WNT3A activate cell autonomous Wnt signaling independent of PORCN or secretion. Biorxiv 2019, 333906. [Google Scholar] [CrossRef]
- Ohkawara, B.; Niehrs, C. An ATF2-based luciferase reporter to monitor non-canonical Wnt signaling in Xenopus embryos. Dev. Dyn. 2011, 240, 188–194. [Google Scholar] [CrossRef]
- Zhu, H.; Mazor, M.; Kawano, Y.; Walker, M.M.; Leung, H.Y.; Armstrong, K.; Waxman, J.; Kypta, R.M. Analysis of Wnt gene expression in prostate cancer: Mutual inhibition by WNT11 and the androgen receptor. Cancer Res. 2004, 64, 7918–7926. [Google Scholar] [CrossRef]
- Lin, Z.; Reierstad, S.; Huang, C.C.; Bulun, S.E. Novel estrogen receptor-alpha binding sites and estradiol target genes identified by chromatin immunoprecipitation cloning in breast cancer. Cancer Res. 2007, 67, 5017–5024. [Google Scholar] [CrossRef]
- Slattery, M.L.; Pellatt, D.F.; Mullany, L.E.; Wolff, R.K.; Herrick, J.S. Gene expression in colon cancer: A focus on tumor site and molecular phenotype. Genes Chromosomes Cancer 2015, 54, 527–541. [Google Scholar] [CrossRef]
- Quintana, J.M.; Gonzalez, N.; Lazaro, S.; Bare, M.; Fernandez-de-Larrea, N.; Redondo, M.; Briones, E.; Escobar, A.; Sarasqueta, C.; Garcia-Gutierrez, S.; et al. Predictors of 1- and 2-year mortality in patients with rectal cancer. Colorectal Dis. 2018, 20, 676–687. [Google Scholar] [CrossRef]
- Schuijers, J.; Mokry, M.; Hatzis, P.; Cuppen, E.; Clevers, H. Wnt-induced transcriptional activation is exclusively mediated by TCF/LEF. EMBO J. 2014, 33, 146–156. [Google Scholar] [CrossRef]
- Zhang, P.; Cai, Y.; Soofi, A.; Dressler, G.R. Activation of Wnt11 by transforming growth factor-beta drives mesenchymal gene expression through non-canonical Wnt protein signaling in renal epithelial cells. J. Biol. Chem. 2012, 287, 21290–21302. [Google Scholar] [CrossRef]
- Zhou, L.; Wang, L.; Lu, L.; Jiang, P.; Sun, H.; Wang, H. Inhibition of miR-29 by TGF-beta-Smad3 signaling through dual mechanisms promotes transdifferentiation of mouse myoblasts into myofibroblasts. PLoS ONE 2012, 7, e33766. [Google Scholar] [CrossRef]
- Zhang, Q.; Bai, X.; Chen, W.; Ma, T.; Hu, Q.; Liang, C.; Xie, S.; Chen, C.; Hu, L.; Xu, S.; et al. Wnt/beta-catenin signaling enhances hypoxia-induced epithelial-mesenchymal transition in hepatocellular carcinoma via crosstalk with hif-1alpha signaling. Carcinogenesis 2013, 34, 962–973. [Google Scholar] [CrossRef]
- Ulrich, F.; Krieg, M.; Schotz, E.M.; Link, V.; Castanon, I.; Schnabel, V.; Taubenberger, A.; Mueller, D.; Puech, P.H.; Heisenberg, C.P. Wnt11 functions in gastrulation by controlling cell cohesion through Rab5c and E-cadherin. Dev. Cell. 2005, 9, 555–564. [Google Scholar] [CrossRef]
- Witzel, S.; Zimyanin, V.; Carreira-Barbosa, F.; Tada, M.; Heisenberg, C.P. Wnt11 controls cell contact persistence by local accumulation of Frizzled 7 at the plasma membrane. J. Cell. Biol. 2006, 175, 791–802. [Google Scholar] [CrossRef]
- Caldwell, G.M.; Jones, C.E.; Soon, Y.; Warrack, R.; Morton, D.G.; Matthews, G.M. Reorganisation of Wnt-response pathways in colorectal tumorigenesis. Br. J. Cancer 2008, 98, 1437–1442. [Google Scholar] [CrossRef]
- Kim, B.K.; Yoo, H.I.; Kim, I.; Park, J.; Kim Yoon, S. FZD6 expression is negatively regulated by miR-199a-5p in human colorectal cancer. BMB Rep. 2015, 48, 360–366. [Google Scholar] [CrossRef]
- Jung, Y.S.; Jun, S.; Lee, S.H.; Sharma, A.; Park, J.I. Wnt2 complements Wnt/beta-catenin signaling in colorectal cancer. Oncotarget 2015, 6, 37257–37268. [Google Scholar] [CrossRef]
- Bakker, E.R.; Das, A.M.; Helvensteijn, W.; Franken, P.F.; Swagemakers, S.; van der Valk, M.A.; ten Hagen, T.L.; Kuipers, E.J.; van Veelen, W.; Smits, R. Wnt5a promotes human colon cancer cell migration and invasion but does not augment intestinal tumorigenesis in Apc1638N mice. Carcinogenesis 2013, 34, 2629–2638. [Google Scholar] [CrossRef]
- Smith, K.; Bui, T.D.; Poulsom, R.; Kaklamanis, L.; Williams, G.; Harris, A.L. Up-regulation of macrophage wnt gene expression in adenoma-carcinoma progression of human colorectal cancer. Br. J. Cancer 1999, 81, 496–502. [Google Scholar] [CrossRef]
- Cheng, R.; Sun, B.; Liu, Z.; Zhao, X.; Qi, L.; Li, Y.; Gu, Q. Wnt5a suppresses colon cancer by inhibiting cell proliferation and epithelial-mesenchymal transition. J. Cell. Physiol. 2014, 229, 1908–1917. [Google Scholar] [CrossRef]
- Shojima, K.; Sato, A.; Hanaki, H.; Tsujimoto, I.; Nakamura, M.; Hattori, K.; Sato, Y.; Dohi, K.; Hirata, M.; Yamamoto, H.; et al. Wnt5a promotes cancer cell invasion and proliferation by receptor-mediated endocytosis-dependent and -independent mechanisms, respectively. Sci. Rep. 2015, 5, 8042. [Google Scholar] [CrossRef]
- Bengoa-Vergniory, N.; Kypta, R.M. Canonical and noncanonical Wnt signaling in neural stem/progenitor cells. Cell. Mol. Life Sci. 2015, 72, 4157–4172. [Google Scholar] [CrossRef]
- Bengoa-Vergniory, N.; Gorrono-Etxebarria, I.; Lopez-Sanchez, I.; Marra, M.; Di Chiaro, P.; Kypta, R. Identification of Noncanonical Wnt Receptors Required for Wnt-3a-Induced Early Differentiation of Human Neural Stem Cells. Mol. Neurobiol. 2017, 54, 6213–6224. [Google Scholar] [CrossRef]
- Quintana, J.M.; Gonzalez, N.; Anton-Ladislao, A.; Redondo, M.; Bare, M.; Fernandez de Larrea, N.; Briones, E.; Escobar, A.; Sarasqueta, C.; Garcia-Gutierrez, S.; et al. Colorectal cancer health services research study protocol: The CCR-CARESS observational prospective cohort project. BMC Cancer 2016, 16, 435. [Google Scholar] [CrossRef]
- Bengoa-Vergniory, N.; Gorrono-Etxebarria, I.; Gonzalez-Salazar, I.; Kypta, R.M. A switch from canonical to noncanonical Wnt signaling mediates early differentiation of human neural stem cells. Stem Cells 2014, 32, 3196–3208. [Google Scholar] [CrossRef]
- Goswami, C.P.; Nakshatri, H. PROGgeneV2: Enhancements on the existing database. BMC Cancer 2014, 14, 970. [Google Scholar] [CrossRef]
| Dataset | Patients | Fold Change | p-Value |
|---|---|---|---|
| Bittner Colon | 373 | 1.513 | 0.058 |
| Gaedke Colorectal | 130 | 1.75 | 8.8 × 10−9 |
| Gaspar Colon | 78 | 1.187 | 0.021 |
| Hong Colorectal | 82 | 4.914 | 5.18 × 10−11 |
| Jorissen Colorectal 3 | 154 | 1.468 | 0.19 |
| Kaiser Colon | 105 | 1.301 | 0.002 |
| Ki Colon | 123 | −1.251 | 0.997 |
| Sabates-Beliver Colon | 64 | 1.3 | 0.168 |
| Skrzypczak Colorectal | 105 | 1.268 | 0.003 |
| Skrzypczak Colorectal 2 | 40 | 1.404 | 0.000165 |
| TGCA | 237 | 1.713 | 1.2 × 10−14 |
| All | 1491 | 1.46 | 8.88 × 10−98.9 |
| Gene | p (Normal vs. Adjacent) 1 | p (Normal vs. Tumor)2 |
| WNT11 | 0.12 | 8.9 × 10−11, 1.21 × 10−14 |
| FZD6 | 0.034 | 1 × 10−12, 2.32 × 10−18 |
| FZD7 | 5.3 × 10−12 | 2.7 × 10−9, 2.24 × 10−8 |
| RYK PTK7 | 0.0015 0.81 | <2 × 10−16, 4.24 × 10−10 <2 × 10−16, 6.3 × 10−18 |
| Gene Pair | Correlation (Pearson) | p-Value |
| WNT11/FZD6 | 0.311, 0.17 | 0.00181, 0.00093 |
| WNT11/FZD7 | 0.242, 0.11 | 0.0161, 0.028 |
| WNT11/RYK WNT11/PTK7 | 0.239, 0.29 0.283, 0.27 | 0.0176, 0.000000024 0.00475, 0.00000017 |
| WNT11 | Comparison | p-Value |
| LEFT vs. RIGHT | Tumor 4.51, 3.87 | 0.047 |
| LEFT vs. RIGHT | Adjacent 3.42, 3.19 | 0.002 |
| LEFT vs. RIGHT | Normal 3.17, 2.95 | 0.007 |
| Gender | Males 4.28, Females 4.2 | NS |
| k-RAS | Yes 4.3, No 4.15 | NS |
| Age | Lower in older patients | 0.0073 |
| Wnt-11 N (%) | (−) 101 (28) | (±) 28 (8) | (+) 99 (28) | (++) 86 (24) | (+++) 43 (12) | Total 357 | p |
|---|---|---|---|---|---|---|---|
| Gender | 0.07 | ||||||
| Male | 65 (64.4) | 19 (67.9) | 76 (76.8) | 49 (57) | 27 (62.8) | 236 (66.1) | |
| Female | 36 (35.6) | 9 (32.1) | 23 (23.2) | 37 (43) | 16 (37.2) | 121 (33.9) | |
| Age, Mean (s.d.) | 69.9 (11.5) | 69.4 (12.6) | 70.5 (10.0) | 69.1 (11.1) | 71.7 (10.6) | 70.0 (11) | 0.79 |
| Age | 0.72 | ||||||
| <49 | 5 (4.95) | 2 (7.1) | 3 (3) | 6 (7) | 0 (0) | 16 (4.4) | |
| 50–59 | 16 (15.8) | 3 (10.7) | 10 (10.1) | 11 (12.8) | 8 (18.6) | 48 (13.4) | |
| 60–69 | 19 (18.8) | 6 (21.4) | 21 (21.2) | 22 (25.6) | 8 (18.6) | 76 (21.3) | |
| 70–79 | 42 (41.6) | 11 (39.3) | 49 (49.5) | 33 (38.4) | 15 (34.9) | 150 (42) | |
| ≥80 | 19 (18.8) | 6 (21.4) | 16 (16.2) | 14 (16.3) | 12 (27.9) | 67 (18.8) | |
| CCI | 0.04 | ||||||
| ≤2 | 50 (49.5) | 14 (50.0) | 46 (46.5) | 44 (51.2) | 25 (58.1) | 179 (50.1) | |
| 3 | 23 (22.8) | 3 (10.7) | 36 (36.4) | 17 (19.8) | 11 (25.6) | 90 (25.2) | |
| ≥4 | 28 (27.7) | 11 (39.3) | 17 (17.2) | 25 (29.1) | 7 (16.3) | 88 (24.7) | |
| Adenocarcinoma | 88 (87.1) | 22 (78.6) | 95 (96) | 83 (96.5) | 40 (93.0) | 328 (91.9) | 0.006 |
| Mucinous Ad. | 9 (8.9) | 6 (21.4) | 4 (4) | 4 (4.7) | 3 (7) | 26 (7.3) | 0.025 |
| Tumor Location | 0.04 | ||||||
| Rectal | 36 (35.6) | 7 (25) | 16 (16.2) | 21 (24.4) | 10 (23.3) | 90 (25.2) | |
| Colon | 65 (64.4) | 21 (75) | 83 (83.8) | 65 (75.6) | 33 (76.7) | 267 (74.8) | |
| Tumor Invasion | 0.61 | ||||||
| No invasion | 90 (89.1) | 25 (89.3) | 83 (83.8) | 75 (87.2) | 39 (90.7) | 312 (87.4) | |
| 1 organ | 10 (9.9) | 3 (10.7) | 16 (16.2) | 9 (10.5) | 3 (7) | 41 (11.5) | |
| >1 organ | 1 (1) | 0 (0) | 0 (0) | 2 (2.3) | 1 (2.3) | 4 (1.1) | |
| K-ras Mutation | 0.72 | ||||||
| Not determined | 95 (94) | 27 (96) | 92 (93) | 79 (92) | 41 (95) | 334 (94) | |
| negative | 2 (2) | 0 (0) | 5 (5) | 3 (3.5) | 0 (0) | 10 (2.8) | |
| positive | 4 (4) | 1 (4) | 2 (2) | 4 (4.6) | 2 (4.7) | 13 (3.6) | |
| Mortality | |||||||
| 3 years | 21 (20.8) | 6 (21.4) | 22 (22.2) | 19 (22.1) | 13 (30.2) | 81 (22.7) | 0.80 |
| 5 years | 25 (24.8) | 8 (28.6) | 36 (36.4) | 28 (32.6) | 18 (41.9) | 115 (32.2) | 0.25 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gorroño-Etxebarria, I.; Aguirre, U.; Sanchez, S.; González, N.; Escobar, A.; Zabalza, I.; Quintana, J.M.; Vivanco, M.d.; Waxman, J.; Kypta, R.M. Wnt-11 as a Potential Prognostic Biomarker and Therapeutic Target in Colorectal Cancer. Cancers 2019, 11, 908. https://doi.org/10.3390/cancers11070908
Gorroño-Etxebarria I, Aguirre U, Sanchez S, González N, Escobar A, Zabalza I, Quintana JM, Vivanco Md, Waxman J, Kypta RM. Wnt-11 as a Potential Prognostic Biomarker and Therapeutic Target in Colorectal Cancer. Cancers. 2019; 11(7):908. https://doi.org/10.3390/cancers11070908
Chicago/Turabian StyleGorroño-Etxebarria, Irantzu, Urko Aguirre, Saray Sanchez, Nerea González, Antonio Escobar, Ignacio Zabalza, José Maria Quintana, Maria dM Vivanco, Jonathan Waxman, and Robert M. Kypta. 2019. "Wnt-11 as a Potential Prognostic Biomarker and Therapeutic Target in Colorectal Cancer" Cancers 11, no. 7: 908. https://doi.org/10.3390/cancers11070908
APA StyleGorroño-Etxebarria, I., Aguirre, U., Sanchez, S., González, N., Escobar, A., Zabalza, I., Quintana, J. M., Vivanco, M. d., Waxman, J., & Kypta, R. M. (2019). Wnt-11 as a Potential Prognostic Biomarker and Therapeutic Target in Colorectal Cancer. Cancers, 11(7), 908. https://doi.org/10.3390/cancers11070908






