Breath Analysis: A Systematic Review of Volatile Organic Compounds (VOCs) in Diagnostic and Therapeutic Management of Pleural Mesothelioma
Abstract
1. Introduction
1.1. Mesothelioma and Asbestos-Related Diseases
1.2. Breath Analysis As Clinical Diagnostic and Disease Monitoring Tools
1.2.1. Breath Composition and Characteristics
1.2.2. Breath Analysis: Sampling and Analytical Methods
1.3. Goals of Systematic Review
1.4. Methodological Approach for Review Writing
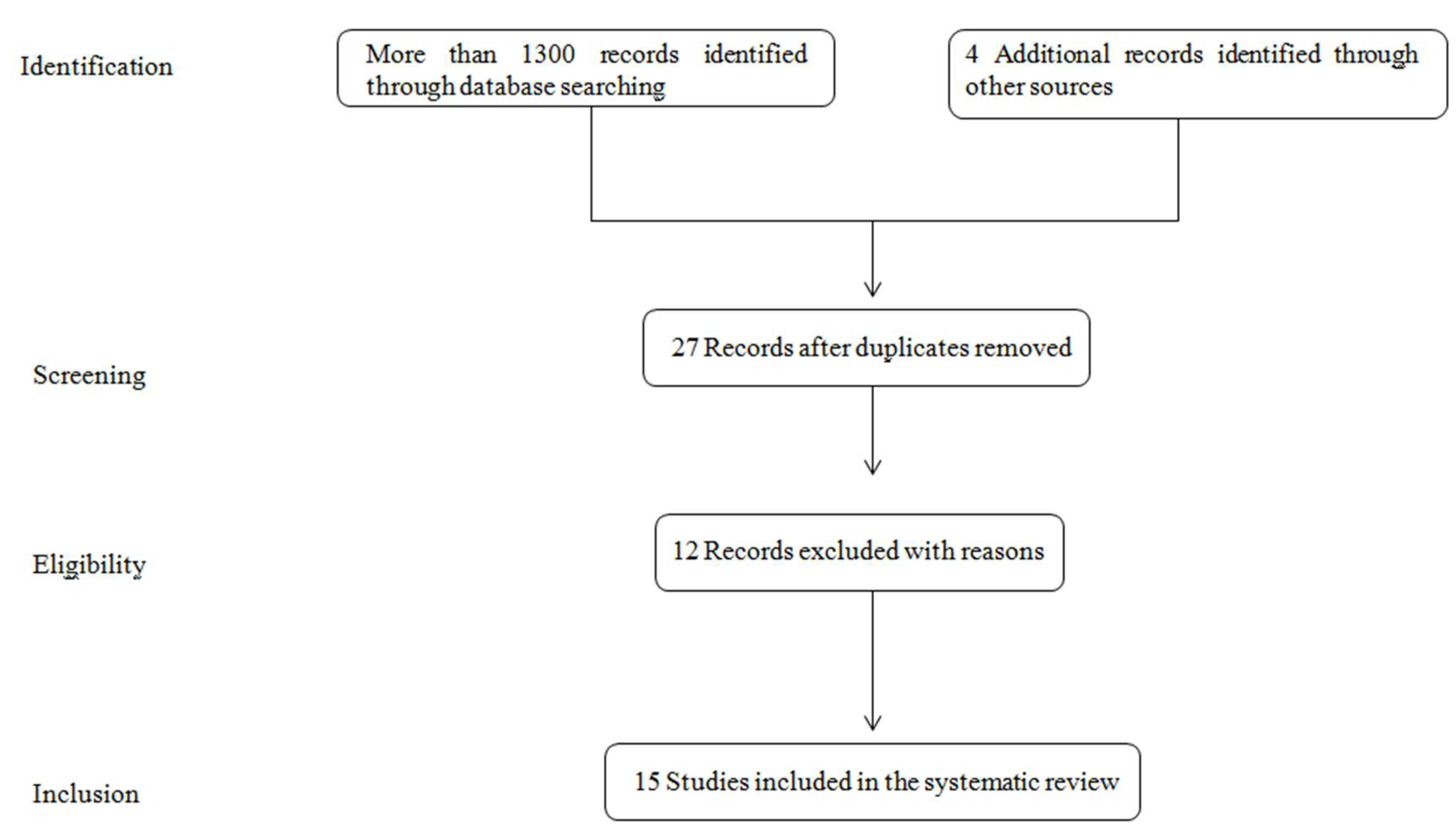
1.4.1. Literature Search
1.4.2. Search Strategy and Literature Selection
1.4.3. Data Collection and Analysis
2. Results and Discussion
2.1. Gas Chromatography and Liquid Chromatography Coupled to Mass Spectrometry (GC/MS, LC/MS)
2.2. Ion Mobility Spectrometry
2.3. Sensor Technology
2.4. Canine Scent
3. Conclusions and Future Perspectives
Author Contributions
Funding
Conflicts of Interest
References
- Liu, B.; van Gerwen, M.; Bonassi, S.; Taioli, E. On behalf of the International Association for the study of Lung Cancer Mesothelioma Task Force. Epidemiology of environmental exposure and malignant mesothelioma. J. Thorac. Oncol. 2017, 12, 1031–1045. [Google Scholar] [CrossRef] [PubMed]
- Roe, O.D.; Stella, G.M. Malignant pleural mesothelioma: History, controversy and future of a manmade epidemic. Eur. Respir. Rev. 2015, 24, 115–131. [Google Scholar] [CrossRef] [PubMed]
- Forde, P.M.; Scherpeerel, A.; Tsao, A. Use of Immune Checkpoint Inhibitors in Mesothelioma. Curr. Treat. Options Oncol. 2019, 20, 18. [Google Scholar] [CrossRef] [PubMed]
- Baas, P.; Fennell, D.; Kerr, K.M.; Felip, E.; Van Schil, P.E.; Hass, R.L.; Peters, S. Malignant pleural mesothelioma: ESMO clinical practice guidelines for diagnosis, treatment and follow up. Ann. Oncol. 2015, 26, v31–v39. [Google Scholar] [CrossRef] [PubMed]
- Travis, W.D.; Brambilla, E.; Burke, A.P.; Marx, A.; Nicholson, A.G. Tumours of the Lung, Pleura, Thymus and Hearth; Iarc Press: Lyon, France, 2015. [Google Scholar]
- Baumann, F.; Ambrosi, J.P.; Carbone, M. Asbestos is not just asbestos: An unrecognized health hazard. Lancet Oncol. 2013, 14, 576–578. [Google Scholar] [CrossRef]
- Jarvholm, B.; Burdorf, A. Emerging evidence that the ban on asbestos use is reducing the occurrence of pleural mesothelioma in Sweden. Scand. J. Public Health 2015, 43, 875–881. [Google Scholar] [CrossRef] [PubMed]
- Krupoves, A.; Camus, M.; De Guire, L. Incidence of malignant mesothelioma of the pleura in Quebec and Canada from 1984 to 2007, and projections from 2008 to 2032. Am. J. Ind. Med. 2015, 58, 473–482. [Google Scholar] [CrossRef]
- Soeberg, M.J.; Leigh, J.; Driscoli, T.; Armstrong, B.; Young, J.M.; Van Zandwijck, N. Incidence and survival trends for malignant pleural and peritoneal mesothelioma, Australia, 1982–2009. Occup. Environ. Med. 2016, 73, 187–194. [Google Scholar] [CrossRef]
- Langniau, S.; Lamote, K.; van Meerbeeck, J.P.; Vermaelen, K. Biomarkers for early diagnosis of malignant mesothelioma: Do we need another moonshot? Oncotarget 2017, 8, 1–11. [Google Scholar]
- Cowan, D.M.; Cheng, T.J.; Ground, M.; Shamel, J.; Varghese, A.; Madl, A.K. Analysis of work place compliance measurement of asbestos by the U.S. Occupational Safety and Health Administration (1984-2011). Regul. Toxicol. Pharm. 2015, 72, 615–629. [Google Scholar] [CrossRef]
- Carbone, M.; Ly, B.H.; Dodson, R.F.; Pagano, I.; Morris, P.T.; Dogan, UA.; Gazdar, A.F.; Pass, H.I.; Yang, H. Malignant mesothelioma: Facts, myths, and hypotheses. J. Cell. Physiol. 2012, 227, 44–58. [Google Scholar] [CrossRef] [PubMed]
- Linton, A.; Vardy, J.; Clarke, S.; van Zandwijk, N. The ticking time-bomb of asbestos: Its insidious role in the development of malignant mesothelioma. Crit. Rev. Oncol. Hematol. 2012, 84, 200–212. [Google Scholar] [CrossRef] [PubMed]
- Takahashi, K.; Landrigan, P.J. The global health dimensions of asbestos and asbestos-related diseases. Ann. Glob. Health 2016, 82, 209–213. [Google Scholar] [CrossRef] [PubMed]
- Mirabelli, D.; Cavone, D.; Merler, E.; Gennaro, V.; Romanelli, A.; Mensi, C.; Chellini, E.; Nicita, C.; Marinaccio, A.; Magnani, C.; et al. Non-occupational exposure to asbestos and malignant mesothelioma in the Italian National Registry of Mesothelioma. Occup. Environ. Med. 2010, 67, 792–794. [Google Scholar] [CrossRef] [PubMed]
- Carbone, M.; Kanodia, S.; Chao, A.; Miller, A.; Wali, A.; Weissman, D.; Adjei, A.; Baumann, F.; Boffetta, P.; Buck, B.; et al. Consensus report of the 2015 Weinman International Conference on Mesothelioma. J. Thorac. Oncol. 2016, 11, 1246–1262. [Google Scholar] [CrossRef] [PubMed]
- De Rienzo, A.; Archer, M.A.; Yeap, B.Y.; Dao, N.; Sciaranghella, D.; Sideris, A.C.; Zheng, Y.; Holman, A.G.; Wang, Y.E.; Dal Cin, P.S.; et al. Gender-specific molecular and clinical features underlie malignant pleural mesothelioma. Cancer Res. 2016, 76, 319–328. [Google Scholar] [CrossRef] [PubMed]
- Van Gerwen, M.; Alpert, N.; Wolf, A.; Ohri, N.; Lewis, E.; Rosenzweig, K.E.; Flores, R.; Taioli, E. Prognostic factors of survival in patients with malignant pleural mesothelioma: An analysis of the National Cancer Database. Carcinogenesis 2019, 40, 529–536. [Google Scholar] [CrossRef] [PubMed]
- Matullo, G.; Guarrera, S.; Betti, M.; Fiorito, G.; Ferrante, D.; Voglino, F.; Cadby, G.; Di Gaetano, C.; Rosa, F.; Russo, A.; et al. Genetic variants associated with increased risk of malignant pleural mesothelioma: A genome-wide association study. PLoS ONE 2013, 8, e61253. [Google Scholar] [CrossRef]
- Betti, M.; Aspesi, A.; Ferrante, D.; Sculco, M.; Righi, L.; Mirabelli, D.; Napoli, F.; Rondón-Lagos, M.; Casalone, E.; Vignolo Lutati, F.; et al. Sensitivity to asbestos is increased in patients with mesothelioma and pathogenic germline variants in BAP1 or other DNA repair genes. Genes Chromosomes Cancer 2018, 57, 573–583. [Google Scholar] [CrossRef]
- Cadby, G.; Mukherjee, S.; Musk, A.W.B.; Reid, A.; Garlepp, M.; Dic, I.; Robinson, C.; Hui, J.; Fiorito, G.; Guarrera, S.; et al. A genome-wide association study for malignant mesothelioma risk. Lung Cancer 2013, 82, 1–8. [Google Scholar] [CrossRef]
- Sezgi, C.; Taylan, M.; Sen, H.S.; Evliyaoğlu, O.; Kaya, H.; Abakay, O.; Abakay, A.; Tanrıkulu, A.C.; Senyiğit, A. Oxidative Status and Acute Phase Reactants in Patients with Environmental Asbestos Exposure and Mesothelioma. Sci. World J. 2014, 2014, 902748. [Google Scholar] [CrossRef] [PubMed]
- Demir, M.; Kaya, H.; Taylan, M.; Ekinci, A.; Yılmaz, S.; Teke, F.; Sezgi, C.; Tanrikulu, A.C.; Meteroglu, F.; Senyigit, A. Evaluation of new biomarkers in the prediction of malignant mesothelioma in subjects with environmental asbestos exposure. Lung 2016, 194, 409–417. [Google Scholar] [CrossRef]
- Creaney, J.; Olsen, N.J.; Brims, F.; Dick, I.M.; Musk, A.W.; De Clerck, N.H.; Skates, S.J.; Robinson, B.W.S. Serum mesothelin for early detection of asbestos-induced cancer malignant mesothelioma. Cancer Epidemiol. Biomark. Prev. 2010, 19, 2238–2246. [Google Scholar] [CrossRef] [PubMed]
- Napolitano, A.; Antoine, D.J.; Pellegrini, L.; Baumann, F.; Pagano, I.; Pastorino, S.; Goparaju, C.M.; Prokrym, K.; Canino, C.; Pass, H.I.; et al. HMGB1 and its hyperacetylated isoform are sensitive and specific biomarkers to detect asbestos exposure and to identify mesothelioma patients. Clin. Cancer Res. 2016, 22, 3087–3096. [Google Scholar] [CrossRef] [PubMed]
- Chen, Z.; Gaudino, G.; Pass, H.I.; Carbone, M.; Yang, H. Diagnostic and prognostic biomarkers for malignant mesothelioma: An update. Transl. Lung Cancer Res. 2017, 6, 259–269. [Google Scholar] [CrossRef] [PubMed]
- Ostroff, R.M.; Mehan, M.R.; Stewart, A.; Ayers, D.; Brody, E.N.; Williams, S.A.; Levin, S.; Black, B.; Harbut, M.; Carbone, M.; et al. Early detection of malignant pleural mesothelioma in asbestos-exposed individuals with a noninvasive proteomics-based surveillance tool. PLoS ONE 2012, 7, e46091. [Google Scholar] [CrossRef] [PubMed]
- Arnold, D.T.; De Fonseka, D.; Hamilton, F.W.; Rahman, N.M.; Maskell, N.A. Prognostication and monitoring of mesothelioma using biomarkers: A systematic review. Br. J. Cancer 2017, 116, 731–741. [Google Scholar] [CrossRef] [PubMed]
- Guarrera, S.; Viberti, C.; Cugliari, G.; Allione, A.; Casalone, E.; Betti, M.; Ferrante, D.; Aspesi, A.; Casadio, C.; Grosso, F.; et al. Peripheral Blood DNA Methylation as Potential Biomarker of Malignant Pleural Mesothelioma in Asbestos-Exposed Subjects. J. Thorac. Oncol. 2019, 14, 527–539. [Google Scholar] [CrossRef] [PubMed]
- Betta, P.G.; Magnani, C.; Bensi, T.; Trincheri, N.F.; Orecchia, S. Immunohistochemistry and molecular diagnostics of pleural malignant mesothelioma. Arch. Pathol. Lab. Med. 2012, 136, 253–261. [Google Scholar] [CrossRef]
- Churg, A.; Sheffield, B.S.; Galateau-Salle, F. New Markers for Separating Benign From Malignant Mesothelial Proliferations: Are We There Yet? Arch. Pathol. Lab. Med. 2016, 140, 318–321. [Google Scholar] [CrossRef] [PubMed]
- Novello, S.; Pinto, C.; Torri, V.; Porcu, L.; Di Maio, M.; Toseo, M.; Ceresoli, G.; Magnani, C.; Silvestri, S.; Veltri, A.; et al. The third Italian consensus conference for malignant pleural mesothelioma: State of the art and recommendations. Crit. Rev. Oncol. Hematol. 2016, 104, 9–20. [Google Scholar] [CrossRef] [PubMed]
- Cinausero, M.; Rihawi, K.; Sperandi, F.; Melotti, B.; Ardizzoni, A. Chemotherapy treatment in malignant pleural mesothelioma: A difficult history. J. Thorac. Dis. 2018, 10, S304–S310. [Google Scholar] [CrossRef] [PubMed]
- Scherpereel, A.; Mazieres, J.; Greillier, L.; Lantuejoul, S.; Dô, P.; Bylicki, O.; Monnet, I.; Corre, R.; Audigier-Valette, C.; Locatelli-Sanchez, M. French Cooperative Thoracic Intergroup. Nivolumab or nivolumab plus ipilimumab in patients with relapsed malignant pleural mesothelioma (IFCT-1501 MAPS2): A multicentre, open-label, randomised, non-comparative, phase 2 trial. Lancet Oncol. 2019, 20, 239–253. [Google Scholar] [CrossRef]
- Disselhorst, M.J.; Quispel-Janssen, J.; Lalezari, F.; Monkhorst, K.; de Vries, J.F.; van der Noort, V.; Harms, E.; Burgers, S.; Baas, P. Ipilimumab and nivolumab in the treatment of recurrent malignant pleural mesothelioma(INITIATE): Results of a prospective, single-arm, phase 2 trial. Lancet Respir. Med. 2019, 7, 260–270. [Google Scholar] [CrossRef]
- Ye, L.; Ma, S.; Robinson, B.W.; Creaney, J. Immunotherapy strategies for mesothelioma—The role of tumor specific neoantigens in a new era of precision medicine. Expert Rev. Respir. Med. 2019, 13, 181–192. [Google Scholar] [CrossRef] [PubMed]
- Gisbert, J.P.; Pajares, J.M. Review article: 13C-urea breath test in the diagnosis of Helicobacter pylori infection—A critical. Aliment. Pharm. 2004, 20, 1001–1017. [Google Scholar] [CrossRef]
- Miekisch, W.; Schubert, J.K.; Noeldge-Schomburg, G.F.E. Diagnostic potential of breath analysis—Focus on volatile organic compounds. Clin. Chim. Acta 2004, 347, 25–39. [Google Scholar] [CrossRef]
- Rosias, P.R.; Dompeling, E.; Dentener, M.A.; Pennings, H.J.; Hendriks, H.J.E.; Van Iersel, M.P.A.; Jöbsis, Q. Childhood asthma: Exhaled markers of airway inflammation, asthma control score, and lung function tests. Pediatr. Pulmonol. 2004, 38, 107–114. [Google Scholar] [CrossRef]
- Buszewski, B.; Kęsy, M.; Ligor, T.; Amann, A. Human exhaled air analytics: Biomarkers of diseases. Biomed. Chromatogr. 2007, 21, 553–566. [Google Scholar] [CrossRef]
- Solga, S.F.; Risby, T. What is Normal Breath? Challenge and Opportunity. IEEE Sens. J. 2010, 10, 7–9. [Google Scholar] [CrossRef]
- Španěl, P.; Smith, D. Progress in SIFT-MS: Breath analysis and other applications. Mass Spec. Rev. 2011, 30, 236–267. [Google Scholar] [CrossRef] [PubMed]
- Philips, M.; Herrera, J.; Krishnan, S.; Zain, M.; Greenberg, J.; Cataneo, R.N. Variation in volatile organic compounds in the breath of normal humans. J. Chromatogr. B 1999, 729, 75–78. [Google Scholar] [CrossRef]
- Horvath, I.; Lazar, Z.; Gyulai, N.; Kollai, M.; Losonczy, G. Exhaled biomarkers in lung cancer. Eur. Respir. J. 2009, 34, 261–275. [Google Scholar] [CrossRef]
- De Lacy, C.B.; Amann, A.; Al-Kateb, H.; Flynn, C.; Filipiak, W.; Khalid, T.; Osborne, D.; Ratcliffe, N.M. A review of the volatiles from the healthy human body. J. Breath Res. 2014, 8, 014001. [Google Scholar] [CrossRef] [PubMed]
- Wallace, M.A.G.; Pleil, J.D. Evolution of clinical and environmental health applications of exhaled breath research: Review of methods and instrumentation for gas-phase, condensate, and aerosols. Anal. Chim. Acta 2018, 1024, 18–38. [Google Scholar] [CrossRef] [PubMed]
- Amann, A.; de Lacy, C.B.; Miekisch, W.; Schubert, J.; Buszewski, B.; Pleil, J.; Ratcliffe, N.; Risby, T. The human volatilome: Volatile organic compounds (VOCs) in exhaled breath, skin emanations, urine, feces and saliva. J. Breath Res. 2014, 8, 1–17. [Google Scholar] [CrossRef]
- Pleil, J.D.; Fisher, J.W.; Lindstrom, A.B. Trichloroethene levels in human blood and exhaled breath from controlled inhalation exposure. Environ. Health Perspect. 1998, 106, 573–580. [Google Scholar] [CrossRef] [PubMed]
- Bajtarevic, A.; Ager, C.; Pienz, M.; Klieber, M.; Schwarz, K.; Ligor, M.; Ligor, T.; Filipiak, W.; Denz, H.; Fiegl, M.; Hilbe, W.; et al. Noninvasive detection of lung cancer by analysis of exhaled breath. BMC Cancer 2009, 9, 348. [Google Scholar] [CrossRef]
- Ligor, M.; Ligor, T.; Bajtarevic, A.; Ager, C.; Pienz, M.; Klieber, M.; Denz, H.; Fiegl, M.; Hilbe, W.; Weiss, W.; et al. Determination of volatile organic compounds in exhaled breath of patients with lung cancer using solid phase microextraction and gas chromatography mass spectrometry. Clin. Chem. Lab. Med. 2009, 47, 550–560. [Google Scholar] [CrossRef]
- Lamote, K.; Nackaerts, K.; van Meerbeeck, J.P. Strengths, weaknesses, and opportunities of diagnostic breathomics in pleural mesothelioma-a hypothesis. Cancer Epidemiol. Biomark. Prev. 2014, 23, 898–908. [Google Scholar] [CrossRef]
- Miekisch, W.; Schubert, J. From highly sophisticated analytical techniques to life-saving diagnostics: Technical developments in breath analysis. Trends Anal. Chem. 2006, 25, 665–673. [Google Scholar]
- Cao, W.Q.; Duan, Y.X. Current status of methods and techniques for breath analysis. Crit. Rev. Anal. Chem. 2007, 37, 3–13. [Google Scholar] [CrossRef]
- Alonso, M.; Sanchez, J.M. Analytical challenges in breath analysis and its application to exposure monitoring. Trac. Trends Anal. Chem. 2013, 44, 78–89. [Google Scholar] [CrossRef]
- Steeghs, M.M.; Cristescu, S.M.; Harren, F.J. The suitability of Tedlar bags for breath sampling in medical diagnostic research. Physiol. Meas. 2006, 28, 73–84. [Google Scholar] [PubMed]
- Pleil, J.D.; Lindstrom, A.B. Measurement of volatile organic compounds in exhaled breath as collected in evacuated electropolished canister. J. Chromatogr. B Biomed. Sci. Appl. 1995, 665, 271–279. [Google Scholar] [CrossRef]
- Giungato, P.; Di Gilio, A.; Palmisani, J.; Marzocca, A.; Mazzone, A.; Brattoli, M.; Giua, R.; de Gennaro, G. Synergistic approaches for odor active compounds monitoring and identification: State of the art, integration, limits and potentialities of analytical and sensorial techniques (Review). Trends Anal. Chem. 2018, 107, 116–129. [Google Scholar] [CrossRef]
- Smith, D.; Spanel, P. Selected ion flow tube mass spectrometry (SIFT-MS) for on-line trace gas analysis. Mass Spectrom. Rev. 2005, 24, 661–700. [Google Scholar] [CrossRef]
- Risby, T.H.; Amann, A.; Smith, D. Breath analysis for clinical diagnostics and therapeutic monitoring. World Sci. Lond. 2005, 251–267. [Google Scholar]
- Awano, S.; Koshimune, S.; Kurihara, E.; Gohara, K.; Sakai, A.; Soh, I.; Hamasaki, T.; Takehara, T. The assessment of methyl mercaptan, an important clinical marker for the diagnosis of oral malodor. J. Dent. 2004, 32, 555–559. [Google Scholar] [CrossRef]
- Chouinard, C.D.; Wei, M.S.; Beekman, C.R.; Kemperman, R.H.; Yost, R.A. Ion Mobility in Clinical Analysis: Current Progress and Future Perspectives. Clin. Chem. 2016, 62, 124–133. [Google Scholar] [CrossRef]
- Moser, B.; Bodrogi, F.; Eibl, G.; Lechner, M.; Rieder, J.; Lirk, P. Mass spectrometric profile of exhaled breath—Field study by PTR-MS. Respir. Physiol. Neurobiol. 2005, 145, 295–300. [Google Scholar] [CrossRef] [PubMed]
- Lirk, P.; Bodrogi, F.; Raifer, H.; Greiner, K.; Ulmer, H.; Rieder, J. Elective haemodialysis increases exhaled isoprene. Nephrol. Dial. Transpl. 2003, 18, 937–941. [Google Scholar] [CrossRef] [PubMed]
- Lindinger, C.; Pollien, P.; Ali, S.; Yeretzian, C.; Blank, I.; Mark, T. Unambiguous identification of volatile organic compounds by proton-transfer reaction mass spectrometry coupled with GC/MS. Anal. Chem. 2005, 77, 4117–4124. [Google Scholar] [CrossRef] [PubMed]
- Blake, R.S.; Whyte, C.; Hughes, C.O.; Ellis, A.M.; Monks, P.S. Demonstration of proton-transfer reaction time-of-flight mass spectrometry for real-time analysis of trace volatile organic compounds. Anal. Chem. 2004, 76, 3841–3845. [Google Scholar] [CrossRef] [PubMed]
- Baumbach, J.I. Ion mobility spectrometry coupled with multi-capillary columns for metabolic profiling of human breath. J. Breath Res. 2009, 3, 034001. [Google Scholar] [CrossRef]
- Westhoff, M.; Litterst, P.; Freitag, L.; Urfer, W.; Bader, S.; Baumbach, J.-I. Ion mobility spectrometry for the detection of volatile organic compounds in exhaled breath of patients with lung cancer: Results of a pilot study. Thorax 2009, 64, 744–748. [Google Scholar] [CrossRef]
- Fitzgerald, J.E.; Bui, E.T.; Simon, N.M.; Fenniri, H. Artificial Nose Technology: Status and Prospects in Diagnostics. Trends Biotechnol. 2017, 35, 33–42. [Google Scholar] [CrossRef]
- Li, F.; Xie, Z.; Schmidt, H.; Sielemann, S.; Baumbach, J. Ion mobility spectrometerfor online monitoring of trace compounds. Spectrochim. Acta 2002, 57, 1563–1574. [Google Scholar] [CrossRef]
- Xie, Z.; Ruzsanyi, V.; Sielemann, S.; Schmidt, H.; Baumbach, J. Determination of pentane, isoprene and acetone Using HSCC-UV-IMS. Int. J. Ion. Mobil. Spectrom. 2001, 4, 88–91. [Google Scholar]
- Steiner, W.E.; Clowers, B.H.; English, W.A.; Hill Jr, H.H. Atmospheric pressure matrix-assisted laser desorption/ionization with analysis by ion mobility time-of-flight mass spectrometry. Rapid Commun. Mass Spectrom. 2004, 18, 882–888. [Google Scholar] [CrossRef]
- Ruzsanyi, V.; Baumbach, J.; Sielemann, S.; Litterst, P.; Westhoff, M.; Freitag, L. Detection of human metabolites using multi-capillary columns coupled to ion mobility spectrometers. J. Chromatogr. A 2005, 1084, 145–151. [Google Scholar] [CrossRef]
- Machado, R.F.; Laskowski, D.; Deffenderfer, O.; Burch, T.; Zheng, S.; Mazzone, P.J.; Mekhail, T.; Jennings, C.; Stoller, J.K.; Pyle, J.; et al. Detection of lung cancer by sensor array analyses of exhaled breath. Am. J. Respir. Crit. Care Med. 2005, 171, 1286–1291. [Google Scholar] [CrossRef] [PubMed]
- Princivalle, A.; Monasta, L.; Butturini, G.; Bassi, C.; Perbellini, L. Pancreatic ductal adenocarcinoma can be detected by analysis of volatile organic compounds (VOCs) in alveolar air. BMC Cancer 2018, 18, 529. [Google Scholar] [CrossRef] [PubMed]
- Netzer, M.; Millonig, G.; Osl, M.; Pfeifer, B.; Praun, S.; Villinger, J.; Vogel, W.; Baumgartner, C. A new ensemble-based algorithm for identifying breath gas marker candidates in liver disease using ion molecule reaction mass spectrometry. Bioinformatics 2009, 25, 941–947. [Google Scholar] [CrossRef] [PubMed]
- Millonig, G.; Praun, S.; Netzer, M.; Baumgartner, C.; Dornauer, A.; Mueller, S.; Villinger, J.; Vogel, W. Non-invasive diagnosis of liver diseases by breath analysis using an optimized ion–molecule reaction mass spectrometry approach: A pilot study. Biomarkers 2010, 15, 297–306. [Google Scholar] [CrossRef] [PubMed]
- Mohamed, E.I.; Bruno, E.; Linder, R.; Alessandrini, M.; Di Girolamo, A.; Poppl, S.J.; Puija, A.; De Lorenzo, A. A novel method for diagnosing chronic rhinosinusitis based on an electronic nose. Otorrinolaringol. Ibero Am. 2003, 30, 447–457. [Google Scholar]
- Groves, W.A.; Zellers, E.T. Analysis of solvent vapors in breath and ambient air with a surface acoustic wave sensor array. Ann. Occup. Hyg. 2001, 45, 609–623. [Google Scholar] [CrossRef]
- Di Natale, C.; Macagnano, A.; Martinelli, E.; Paolesse, R.; DArcangelo, G.; Roscioni, C.; Finazzi-Agro, A.; D’Amico, A. Lung cancer identification by the analysis of breath by means of an array of non-selective gas sensors. Biosens. Bioelectron. 2003, 18, 1209–1218. [Google Scholar] [CrossRef]
- Kamp, D.W.; Graceffa, P.; Pryor, W.A.; Weitzman, S.A. The role of free radicals in asbestos-induced diseases. Free Radic. Biol. Med. 1992, 12, 293–315. [Google Scholar] [CrossRef]
- Kamp, D.W.; Weitzman, S.A. The molecular basis of asbestos induced lung injury. Thorax 1999, 54, 638–652. [Google Scholar] [CrossRef]
- Cugell, D.W.; Kamp, D.W. Asbestos and the pleura—A review. Chest 2004, 125, 1103–1117. [Google Scholar] [CrossRef]
- Murata, M.; Thanan, R.; Ma, N.; Kawanishi, S. Role of nitrative and oxidative DNA damage in inflammation-related carcinogenesis. J. Biomed. Biotechnol. 2012, 2012, 623019. [Google Scholar] [CrossRef] [PubMed]
- Yang, H.; Rivera, Z.; Jube, S.; Nasu, M.; Bertino, P.; Goparaju, C.; Franzoso, G.; Lotze, M.T.; Pass, H.I.; Bianchi, M.E.; et al. Programmed necrosis induced by asbestos in human mesothelial cells causes high-mobility group box 1 protein release and resultant inflammation. Proc. Natl. Acad. Sci. USA 2010, 107, 12611–12616. [Google Scholar] [CrossRef] [PubMed]
- Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) standards & guidelines. Available online: http://www.prisma-statement.org/ (accessed on 1 April 2019).
- Green, S.; Higgins, J.P.T.; Alderson, P.; Clarke, M.; Mulrow, C.D.; Oxman, A.D. Chapter 1: What is a systematic review. In Cochrane Handbook of Systematic Reviews of Interventions; Cochrane Book Series; John Wiley & Sons: Chichester, UK, 2008; pp. 1–9. [Google Scholar]
- Lamote, K.; Brinkman, P.; Vandermeersch, L.; Vynck, M.; Sterk, P.J.; Van Langenhove, H.; Thas, O.; Van Cleemput, J.; Nackaerts, K.; Van Meerbeeck, J.P. Breath analysis by gas chromatography-mass spectrometry and electronic nose to screen for pleural mesothelioma: A cross-sectional case control study. Oncotarget 2017, 8, 91593–91602. [Google Scholar] [CrossRef] [PubMed]
- Lamote, K.; Vynck, M.; Thas, O.; Van Cleemput, J.; Nackaerts, K.; van Meerbeeck, J.P. Exhaled breath to screen for malignant pleural mesothelioma: A validation study. Eur. Respir. J. 2017, 50, 1700919. [Google Scholar] [CrossRef] [PubMed]
- Karvonen, T.; Kankaanranta, H.; Saarelainen, S.; Moilanen, E.; Lehtimäki, L. Comparison of feasibility and estimates of central and peripheral nitric oxide parameters by different mathematical models. J. Breath Res. 2017, 11, 047102. [Google Scholar] [CrossRef] [PubMed]
- Lamote, K.; Vynck, M.; Van Cleemput, J.; Thas, O.; Nackaerts, K.; Van Meerbeeck, J.P. Detection of malignant pleural mesothelioma in exhaled breath by multicapillary column/ion mobility spectrometry (MCC/IMS). J. Breath Res. 2016, 10, 046001. [Google Scholar] [CrossRef]
- Lamote, K.; Hiddinga, B.; Van Cleemput, J.; Nackaerts, K.; Thas, O.; Van Meerbeeck, J.P. A breath test for diagnosing malignant pleural mesothelioma. Ann. Oncol. 2014, 25, 542–545. [Google Scholar] [CrossRef][Green Version]
- Cakir, Y.; Métrailler, L.; Baumbach, J.I.; Ktaus, T. Signals in asbestos related diseases in human breath—Preliminary results. Int. J. Ion Mobil. Spec. 2014, 17, 87–94. [Google Scholar] [CrossRef]
- Chapman, E.A.; Thomas, P.S.; Stone, E.; Lewis, C.; Yates, D.H. A breath test for malignant mesothelioma using an electronic nose. Eur. Respir. J. 2012, 40, 448–454. [Google Scholar] [CrossRef]
- Dragonieri, S.; Van der Schee, M.P.; Massaro, T.; Schiavulli, N.; Brinkman, P.; Pinca, A.; Carratú, P.; Spanevello, A.; Resta, O.; Musti, M.; et al. An electronic nose distinguishes exhaled breath of patients with Malignant Pleural Mesothelioma from controls. Lung Cancer 2012, 75, 326–331. [Google Scholar] [CrossRef] [PubMed]
- De Gennaro, G.; Dragonieri, S.; Longobardi, F.; Musti, M.; Stallone, G.; Trizio, L.; Tutino, M. Chemical characterization of exhaled breath to differentiate between patients with malignant plueral mesothelioma from subjects with similar professional asbestos exposure. Anal. Bioanal. Chem. 2010, 398, 3043–3050. [Google Scholar] [CrossRef] [PubMed]
- Syslová, K.; Kačer, P.; Kuzma, M.; Pankracová, A.; Fenclová, Z.; Vlčková, Š.; Lebedová, J.; Pelclová, D. LC-ESI-MS/MS method for oxidative stress multimarker screening in the exhaled breath condensate of asbestosis/silicosis patients. J. Breath Res. 2010, 4, 017104. [Google Scholar] [CrossRef] [PubMed]
- Chow, S.; Campbell, C.; Sandrini, A.; Thomas, P.S.; Johnson, A.R.; Yates, D.H. Exhaled breath condensate biomarkers in asbestos-related lung disorders. Respir. Med. 2009, 103, 1091–1097. [Google Scholar] [CrossRef] [PubMed]
- Syslová, K.; Kačer, P.; Kuzma, M.; Najmanová, V.; Fenclová, Z.; Vlčková, Š.; Lebedová, J.; Pelclová, D. Rapid and easy method for monitoring oxidative stress markers in body fluids of patients with asbestos or silica-induced lung diseases. J. Chromatogr. B 2009, 877, 2477–2486. [Google Scholar] [CrossRef] [PubMed]
- Pelclova, D.; Fenclova, Z.; Kacer, P.; Kuzma, M.; Navratil, T.; Lebedova, J. Increased 8-isoprostane, a Marker of Oxidative Stress in Exhaled Breath Condensate in Subjects with Asbestos Exposure. Ind. Health 2008, 46, 484–489. [Google Scholar] [CrossRef] [PubMed]
- Lehtonen, H.; Oksa, P.; Lehtimäki, L.; Sepponen, A.; Nieminen, R.; Kankaanranta, H.; Saarelainen, S.; Järvenpaä, R.; Uitti, J.; Moilanen, E. Increased alveolar nitric oxide concentration and high levels of leukotriene B4 and 8-isoprostane in exhaled breath condensate in patients with asbestosis. Thorax 2007, 62, 602–607. [Google Scholar] [CrossRef] [PubMed]
- Sandrini, A.; Johnson, A.R.; Thomas, P.S.; Yates, D.H. Fractional exhaled nitric oxide concentration is increased in asbestosis and pleural plaques. Respirology 2006, 11, 325–329. [Google Scholar] [CrossRef]
- Amundsen, T.; Stein Sundstr, Ø.M.; Buvik, T.; Arna Gederaas, O.; Haaverstad, R. Can dogs smell lung cancer? First study using exhaled breath and urine screening in unselected patients with suspected lung cancer. Acta Oncol. 2014, 53, 307–315. [Google Scholar] [CrossRef]
- Buszewski, B.; Ligor, T.; Jezierski, T.; Wenda-Piesik, A.; Walczak, M.; Rudnicka, J. Identification of volatile lung cancer markers by gas chromatography–mass spectrometry: Comparison with discrimination by canines. Anal. Bioanal. Chem. 2012, 404, 141–146. [Google Scholar] [CrossRef]
- Ehmann, R.; Boedeker, E.; Friedrich, U.; Sagert, J.; Dippon, J.; Friedel, G.; Walles, T. Canine scent detection in the diagnosis of lung cancer: Revisiting a puzzling phenomenon. Eur. Respir. J. 2012, 39, 669–676. [Google Scholar] [CrossRef] [PubMed]
| First Author, Year [Ref] | Sample Tipology | Analytical Technique | Storage Container | Subjects Involved (Cancer Stage) | Classifier | Results (Discriminating VOCs) | Concentration Range |
|---|---|---|---|---|---|---|---|
| Lamote K., 2017 [87] | exhaled breath samples | GC/MS e-nose | 10 L Tedlar bags | 14 MPM, 19 asymptomatic former EXP, 15 ARD, 14 HC | Pearson Chi2-test, Shapiro-Wilk test, logistic regression (lasso) | diethyl ether, limonene, nonanal, methylcyclopentane and cyclohexane | not reported |
| Lamote K., 2017 [88] | exhaled breath samples | MCC-IMS | 52 MPM, 56 LC, 70 BLD, 41 ARD, 59 EXP and 52 HC | Logistic regression (lasso), Fisher’s exact test, Kolmogorov-Smirnov test, ANOVA or Kruskal-Wallis test | P1, P3, P7, P9, P21, and P26 | not reported | |
| Karvonen T., 2017 [89] | exhaled breath samples | Sievers NOA 280 chemiluminescence analyser | 69 HC adults, 66 HC children, 73 asbestos-exposed and 72 COPD | 5 mathematical methods (Tsoukias and George, Pietropaoli, Condorelli, Högman and Meriläinen, and Silkoff) to estimate alveolar and bronchial NO parameters | NO | not reported | |
| Lamote K., 2016 [90] | exhaled breath samples | MCC-IMS | 23 MPM, 22 asymptomatic former asbestos workers and 21 HC | Logistic least absolute shrinkage and selection operator (lasso) regression, chi-squared test or Fisher’s exact, Kolmogorov–Smirnov test to assess normality, t-test or analysis of variance (ANOVA), Wilcoxon–Mann–Whitney test or Kruskal–Wallis test | P3, P5, P50 and P71 | not reported | |
| Lamote K., 2014b [91] | exhaled breath samples | MCC-IMS | 20 MPM patients, 10 asbestos-exposed and 10 HC | Logistic LASSO regression | P5, P3, P83, P1 and P67 | not reported | |
| Cakir Y., 2014 [92] | exhaled breath samples | MCC-IMS | 25 MPM and 12 HC | Box and Whisker plot and decision tree | 4-ethytoluol and alpha pinene | not reported | |
| Chapman E.A., 2012 [93] | exhaled breath samples | e-nose (Cyranose320) | 2-L gas impermeable bag | 20 MPM (19:stage 2, 1:stage 1b), 13 Pleural disease and 42 HC | PCA, linear canonical discriminant analysis and Mahalanobis distance | not reported | |
| Dragonieri S., 2012 [94] | exhaled breath samples | e-nose (Cyranose 320) | 5-L Tedlar bag | 13 MPM (7:stage 1b; 1:stage 1a; 3: stage 2; 2: stage 3), 13 asbestos-exposed and 13 HC | PCA and CDA | not reported | |
| de Gennaro G., 2010 [95] | exhaled breath samples | TD-GC-MS | 5-L Tedlar bag | 13 MPM, 13 EXP and 13 HC | Anova, PCA, DFA, CP-ANN | cyclopentane, cyclohexane | cyclopentane median value: MPM patients 120.42 ng/L vs. asbestosis 605.49 ng/L vs. HC 34.83 ng/L.; cyclohexane median value: MPM patients 251.79 ng/L vs. asbestosis 69.31 ng/L vs. HC 33.08 ng/L. |
| Syslová K., 2010* [96] | EBC | LC-ESI-MS/MS | 10 patients occupational lung diseases (either silica or asbestos exposure) and 10 HC | Student’s t-test | 8-iso-PGF2α, o-Tyr and 8-OHdG | Patients vs. HC (median pg ml−1): 8-iso-PGF2α 106.1 vs. 86.7; o-Tyr 61.8 vs. 47.5; 8-OHdG 46.5 vs. 14.8. | |
| Chow S., 2009 * [97] | EBC | fluorimetric modification of the Greiss reaction (NOx), Quantipro BCA assay kit (total protein), enzyme-immunoassay (EIA) kit (8-Isoprostane), enzyme immunoassay (EIA) (3-nitrotyrosine) and H2O2 measured spectrophotometrically | 18 Asbestosis, 26 Pleural plaques, 16 diffuse pleural thickening (DPT) and 26 HC | Anova and Pearson’s correlation coefficient | 8-isoprostane, leukotrienes B4, C4, D4, and E4, hydrogen peroxide, EBC total protein, fractional FeNO | asbestosis vs. HC: 8-isoprostane (geometric mean (95% CI) 0.51 (0.17-1.51) vs. 0.07 (0.04–0.13) ng/mL); hydrogen peroxide (13.68 (8.63–21.68) vs. 5.89 (3.99–8.69); EBC total protein (17.27 (10.57–28.23) vs. 7.62(5.13–11.34) mg/mL; FeNO (mean +/− SD) (9.67 +/− 3.26 vs. 7.57 +/− 1.89 ppb). | |
| Syslová K., 2009 * [98] | EBC | LC-ESI_MS/MS | 20 patients occupational exposure to asbestos/silica dust (for 24 years in average) and 10 HC | Student’s t-test | 8-isoprostaglandin F, malondialdehyde(MDA) and 4-hydroxynonenal (HNE) | asbestosis vs HC: 8-iso (mean 71(66–77) vs. 52 (45–61) pg/mL); MDA (mean 72 (65–86) vs. 45 (36-55) ng/mL); HNE (mean 233 (188–267) vs. 165 (140–186) ng/mL). | |
| Pelclova D., 2008 * [99] | EBC | LC-ESI-MS | 92 asbestos-exposed and 46 HC | Student’s t-test, F-test, ANOVA and linear regression | 8-isoprostane | 8-isoprostane asbestos exposed 69.5 ± 6.6 pg/mL vs. HC 47.0 ± 7.8 pg/mL | |
| Lehtonen H., 2007* [100] | EBC | Sievers NOA 280 analyser and immunoassay kit | 15 Asbestosis and 15 HC | Mean values | NO, LTB4 and 8-isoprostane | asbestosis vs HC: NO (3.2 (0.4) vs. 2.0 (0.2) ppb); LTB4 (39.5 (6.0) vs 15.4 (2.9) pg/mL);8-isoprostane (33.5 (9.6) vs. 11.9 (2.8) pg/mL). | |
| Sandrini A., 2006 [101] | exhaled breath samples | chemiluminescence NO and CO analyser LR 2500 (I) | 56 subjects with asbestos-related disorders and 35 HC | Anova and multiple comparison post hoc test (Scheffe) | NO, CO | FENO: asbestosis (7.9 (6.6–15.7) ppb), pleural plaques (6.3 (5.3–-9) ppb), HC (4.6 (3.5–6) ppb). |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Catino, A.; de Gennaro, G.; Di Gilio, A.; Facchini, L.; Galetta, D.; Palmisani, J.; Porcelli, F.; Varesano, N. Breath Analysis: A Systematic Review of Volatile Organic Compounds (VOCs) in Diagnostic and Therapeutic Management of Pleural Mesothelioma. Cancers 2019, 11, 831. https://doi.org/10.3390/cancers11060831
Catino A, de Gennaro G, Di Gilio A, Facchini L, Galetta D, Palmisani J, Porcelli F, Varesano N. Breath Analysis: A Systematic Review of Volatile Organic Compounds (VOCs) in Diagnostic and Therapeutic Management of Pleural Mesothelioma. Cancers. 2019; 11(6):831. https://doi.org/10.3390/cancers11060831
Chicago/Turabian StyleCatino, Annamaria, Gianluigi de Gennaro, Alessia Di Gilio, Laura Facchini, Domenico Galetta, Jolanda Palmisani, Francesca Porcelli, and Niccolò Varesano. 2019. "Breath Analysis: A Systematic Review of Volatile Organic Compounds (VOCs) in Diagnostic and Therapeutic Management of Pleural Mesothelioma" Cancers 11, no. 6: 831. https://doi.org/10.3390/cancers11060831
APA StyleCatino, A., de Gennaro, G., Di Gilio, A., Facchini, L., Galetta, D., Palmisani, J., Porcelli, F., & Varesano, N. (2019). Breath Analysis: A Systematic Review of Volatile Organic Compounds (VOCs) in Diagnostic and Therapeutic Management of Pleural Mesothelioma. Cancers, 11(6), 831. https://doi.org/10.3390/cancers11060831








