A Transgenic Mouse Model of Pacak–Zhuang Syndrome with An Epas1 Gain-of-Function Mutation
Abstract
1. Introduction
2. Results
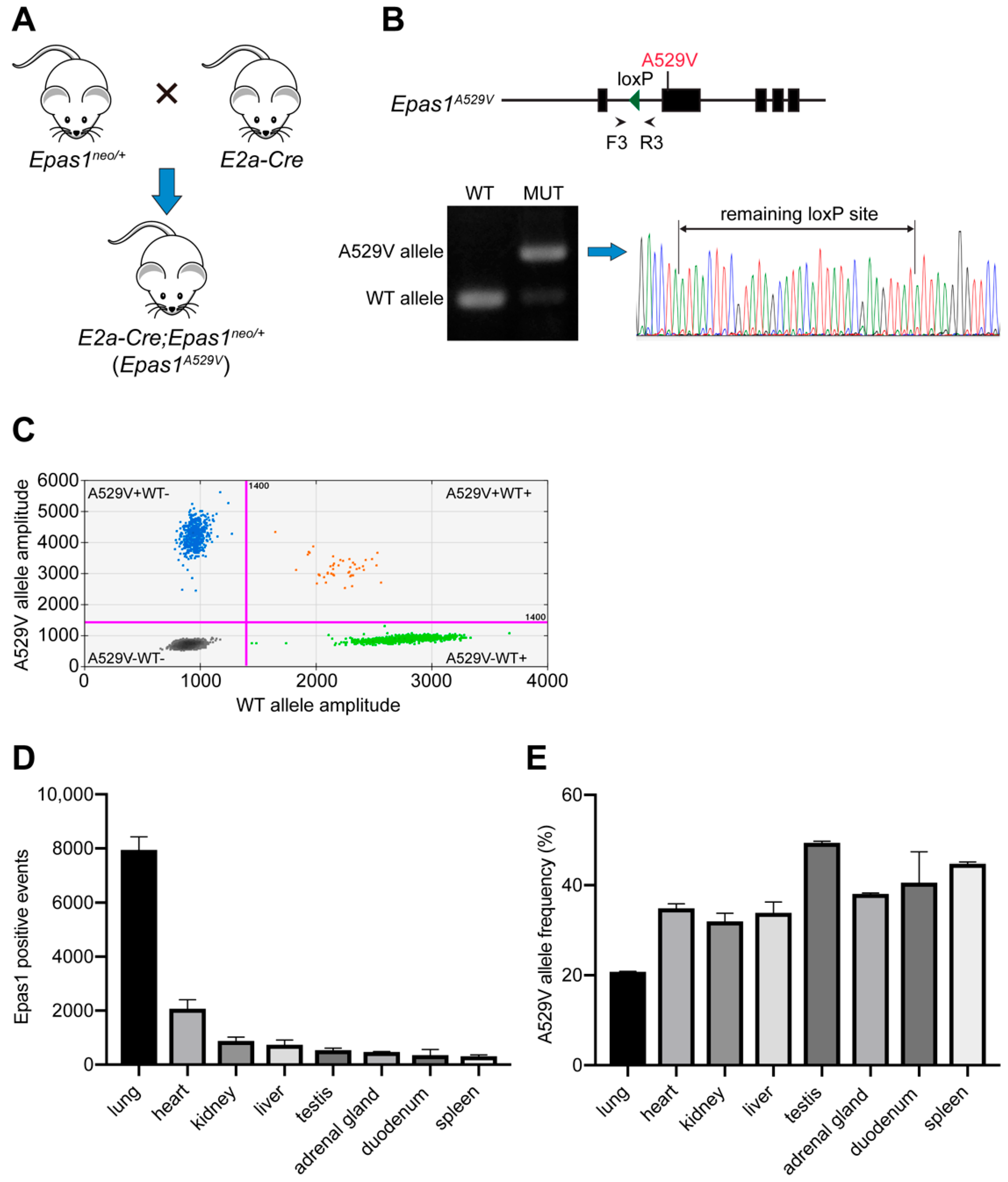
2.1. Establishment of A Somatic Epas1A529V Animal Model
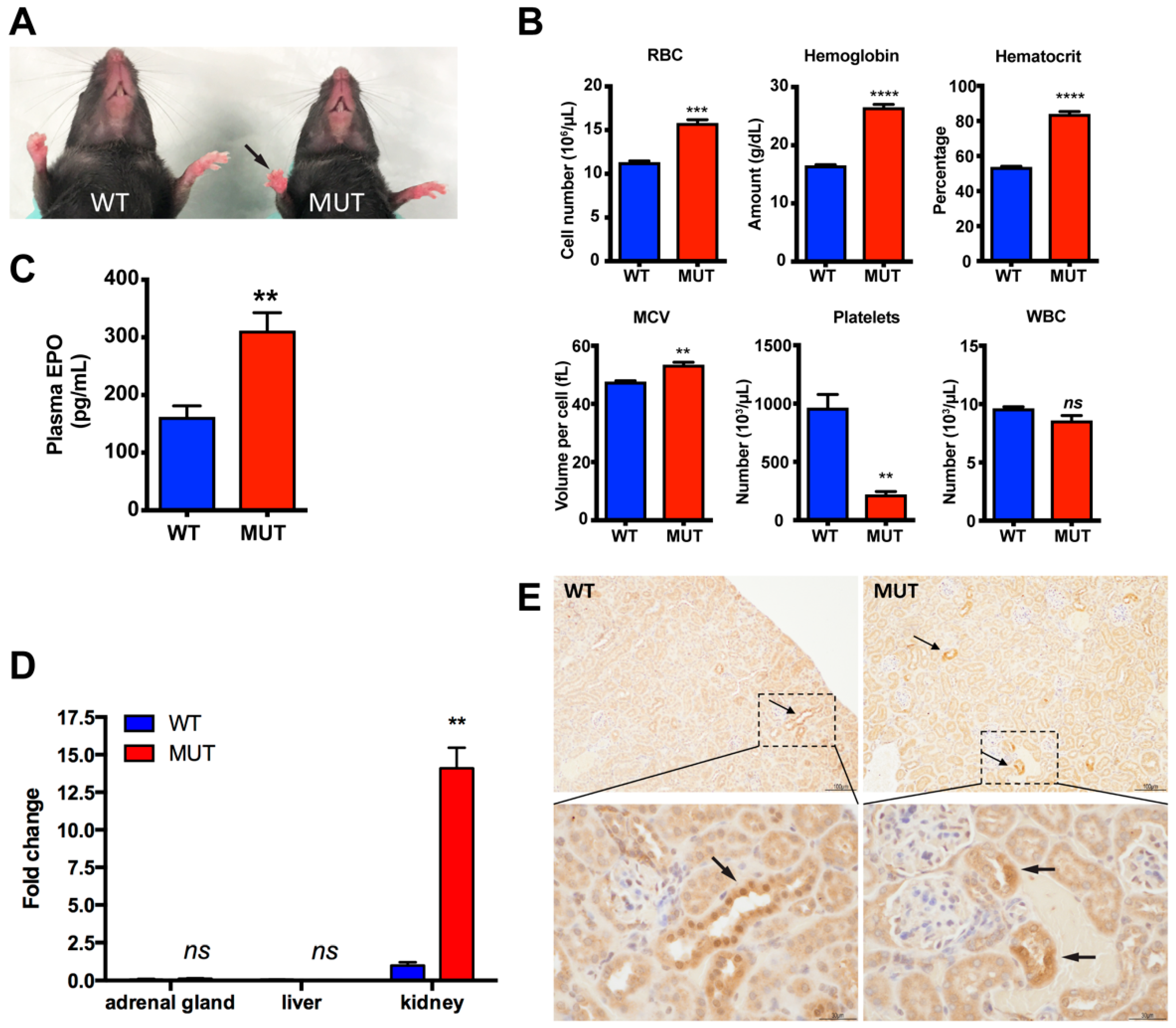
2.2. Polycythemia and Elevated EPO in Epas1A529V Mutant Mice
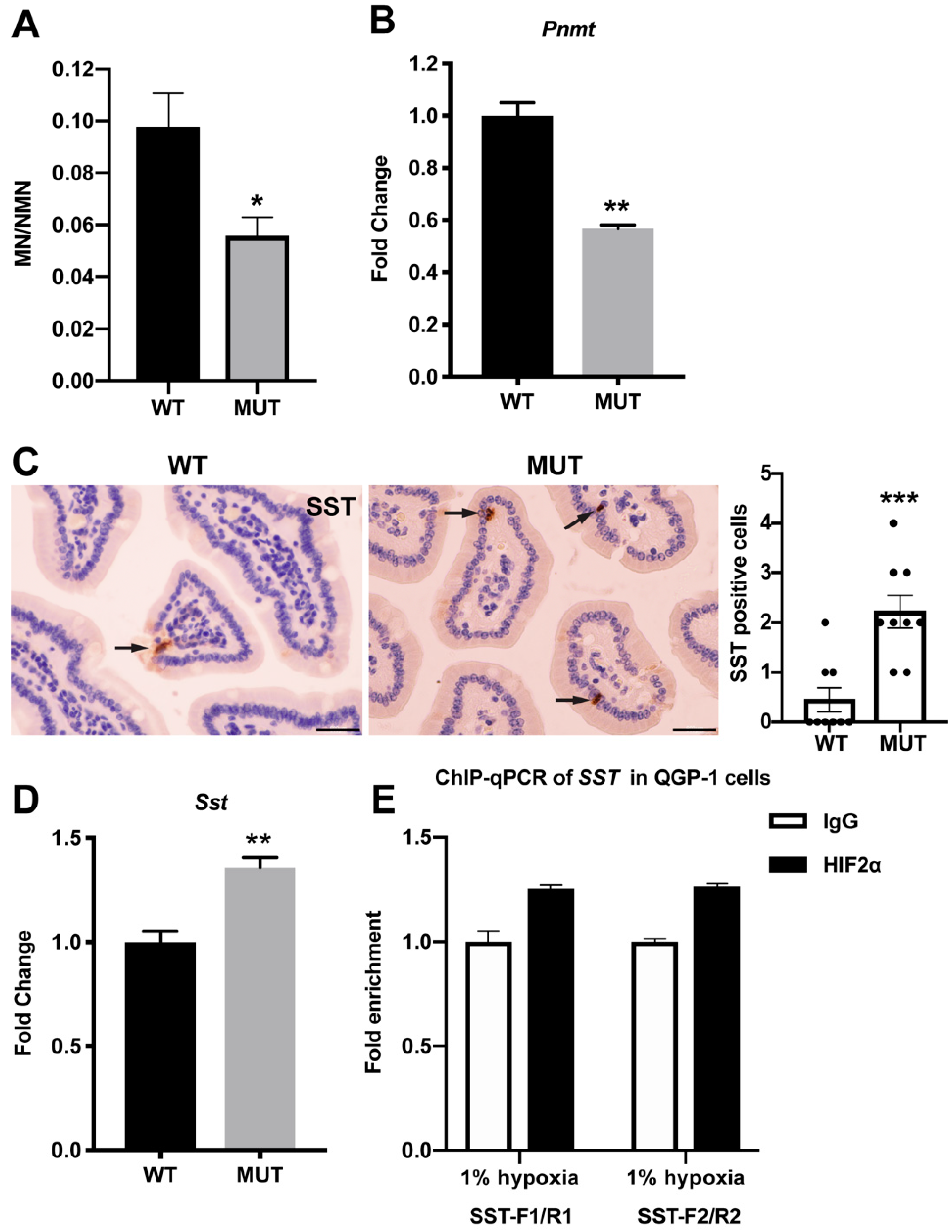
2.3. Biochemistry Characteristics of Epas1A529V Mutant Mice
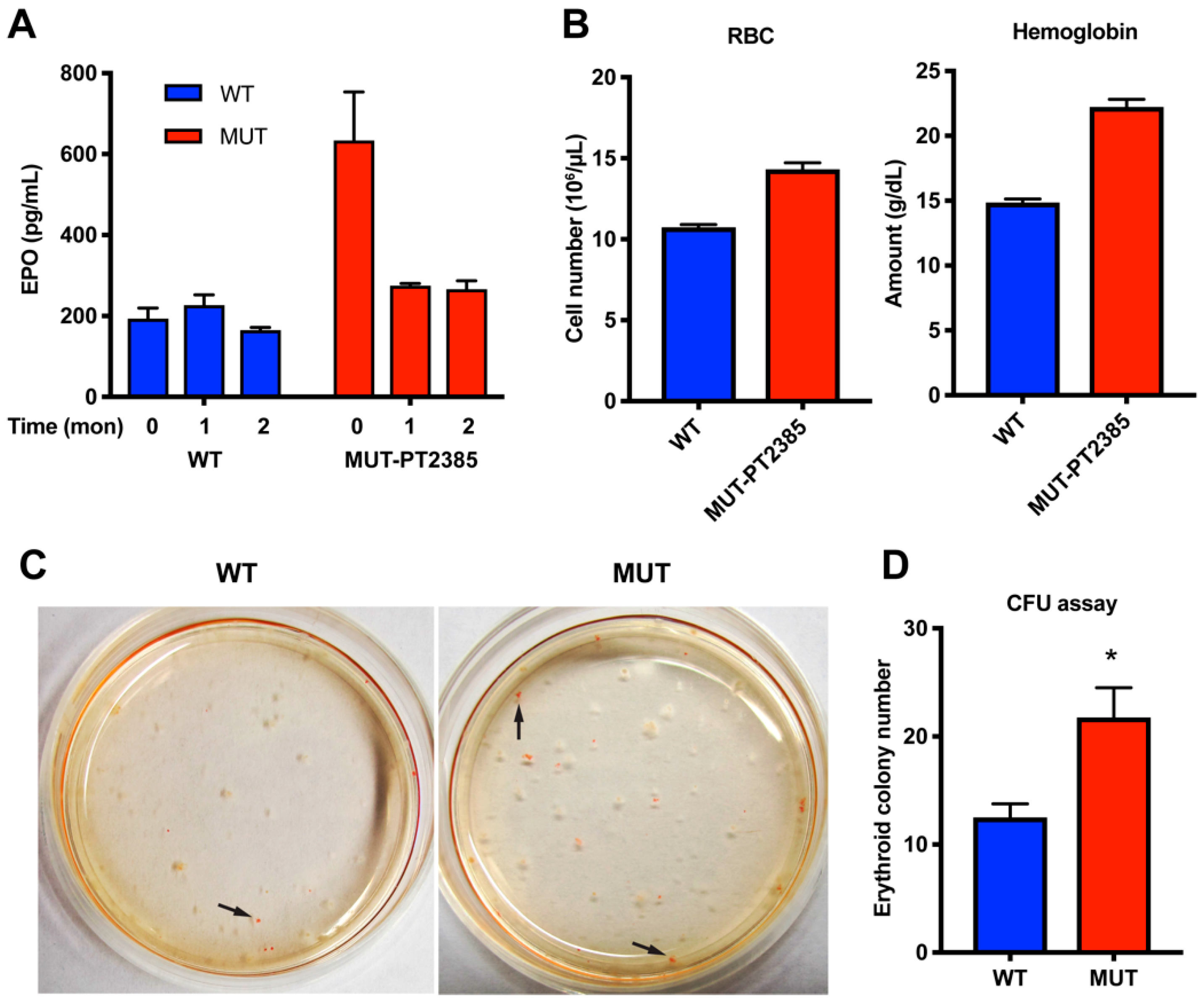
2.4. Inhibition of HIF2α Reduced EPO but Not Polycythemia in Epas1A529V Mutant Mice
3. Discussion
4. Materials and Methods
4.1. Mouse Model and Genotyping
4.2. Complete Blood Count (CBC)
4.3. Enzyme-Linked Immunosorbent Assay (ELISA)
4.4. Quantitative Real-Time Polymerase Chain Reaction (qRT-PCR)
4.5. Immunohistochemistry (IHC) Staining
4.6. Droplet Digital PCR (ddPCR)
4.7. PT2385 Treatment
4.8. Determinations of Urinary Catecholamines and Metanephrines
4.9. ChIP-qPCR
4.10. Colony-Forming Unit (CFU) Assay
4.11. Statistics
4.12. Study Approval
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Zhuang, Z.; Yang, C.; Lorenzo, F.; Merino, M.; Fojo, T.; Kebebew, E.; Popovic, V.; Stratakis, C.A.; Prchal, J.T.; Pacak, K. Somatic HIF2A gain-of-function mutations in paraganglioma with polycythemia. N. Engl. J. Med. 2012, 367, 922–930. [Google Scholar] [CrossRef] [PubMed]
- Pacak, K.; Jochmanova, I.; Prodanov, T.; Yang, C.; Merino, M.J.; Fojo, T.; Prchal, J.T.; Tischler, A.S.; Lechan, R.M.; Zhuang, Z. New syndrome of paraganglioma and somatostatinoma associated with polycythemia. J. Clin. Oncol. 2013, 31, 1690–1698. [Google Scholar] [CrossRef]
- Qin, N.; de Cubas, A.A.; Garcia-Martin, R.; Richter, S.; Peitzsch, M.; Menschikowski, M.; Lenders, J.W.; Timmers, H.J.; Mannelli, M.; Opocher, G.; et al. Opposing effects of HIF1alpha and HIF2alpha on chromaffin cell phenotypic features and tumor cell proliferation: Insights from MYC-associated factor X. Int. J. Cancer 2014, 135, 2054–2064. [Google Scholar] [CrossRef] [PubMed]
- Barba, T.; Boileau, J.C.; Pasquet, F.; Hot, A.; Pavic, M. [Inherited primitive and secondary polycythemia]. Rev. Med. Interne. 2016, 37, 460–465. [Google Scholar] [CrossRef] [PubMed]
- Prchal, J.T. Secondary polycythemia (Erythrocytosis). In Williams Hematology, 9th ed.; Kaushansky, K., Lichtman, M.A., Prchal, J.T., Levi, M., Press, O.W., Burns, L.J., Caligiuri, M.A., Eds.; McGraw-Hill: New York, NY, USA, 2015; pp. 871–888. [Google Scholar]
- Li, Z.; Bao, S.; Wu, Q.; Wang, H.; Eyler, C.; Sathornsumetee, S.; Shi, Q.; Cao, Y.; Lathia, J.; McLendon, R.E.; et al. Hypoxia-inducible factors regulate tumorigenic capacity of glioma stem cells. Cancer Cell 2009, 15, 501–513. [Google Scholar] [CrossRef]
- Schonenberger, D.; Harlander, S.; Rajski, M.; Jacobs, R.A.; Lundby, A.K.; Adlesic, M.; Hejhal, T.; Wild, P.J.; Lundby, C.; Frew, I.J. Formation of Renal Cysts and Tumors in Vhl/Trp53-Deficient Mice Requires HIF1alpha and HIF2alpha. Cancer Res. 2016, 76, 2025–2036. [Google Scholar] [CrossRef] [PubMed]
- Ryan, H.E.; Lo, J.; Johnson, R.S. HIF-1 alpha is required for solid tumor formation and embryonic vascularization. EMBO J. 1998, 17, 3005–3015. [Google Scholar] [CrossRef] [PubMed]
- Tian, H.; Hammer, R.E.; Matsumoto, A.M.; Russell, D.W.; McKnight, S.L. The hypoxia-responsive transcription factor EPAS1 is essential for catecholamine homeostasis and protection against heart failure during embryonic development. Genes Dev. 1998, 12, 3320–3324. [Google Scholar]
- Percy, M.J.; Furlow, P.W.; Lucas, G.S.; Li, X.; Lappin, T.R.; McMullin, M.F.; Lee, F.S. A gain-of-function mutation in the HIF2A gene in familial erythrocytosis. N. Engl. J. Med. 2008, 358, 162–168. [Google Scholar] [CrossRef]
- Gale, D.P.; Harten, S.K.; Reid, C.D.; Tuddenham, E.G.; Maxwell, P.H. Autosomal dominant erythrocytosis and pulmonary arterial hypertension associated with an activating HIF2 alpha mutation. Blood 2008, 112, 919–921. [Google Scholar] [CrossRef]
- Tan, Q.; Kerestes, H.; Percy, M.J.; Pietrofesa, R.; Chen, L.; Khurana, T.S.; Christofidou-Solomidou, M.; Lappin, T.R.; Lee, F.S. Erythrocytosis and pulmonary hypertension in a mouse model of human HIF2A gain of function mutation. J. Biol. Chem. 2013, 288, 17134–17144. [Google Scholar] [CrossRef]
- Richter, S.; Qin, N.; Pacak, K.; Eisenhofer, G. Role of hypoxia and HIF2alpha in development of the sympathoadrenal cell lineage and chromaffin cell tumors with distinct catecholamine phenotypic features. Adv. Pharmacol. 2013, 68, 285–317. [Google Scholar] [CrossRef] [PubMed]
- Jelkmann, W. Regulation of erythropoietin production. J. Physiol. 2011, 589, 1251–1258. [Google Scholar] [CrossRef] [PubMed]
- Franke, K.; Gassmann, M.; Wielockx, B. Erythrocytosis: The HIF pathway in control. Blood 2013, 122, 1122–1128. [Google Scholar] [CrossRef]
- Powers, J.F.; Pacak, K.; Tischler, A.S. Pathology of Human Pheochromocytoma and Paraganglioma Xenografts in NSG Mice. Endocr. Pathol. 2017, 28, 2–6. [Google Scholar] [CrossRef]
- Lepoutre-Lussey, C.; Thibault, C.; Buffet, A.; Morin, A.; Badoual, C.; Benit, P.; Rustin, P.; Ottolenghi, C.; Janin, M.; Castro-Vega, L.J.; et al. From Nf1 to Sdhb knockout: Successes and failures in the quest for animal models of pheochromocytoma. Mol. Cell. Endocrinol. 2016, 421, 40–48. [Google Scholar] [CrossRef]
- Eisenhofer, G.; Huynh, T.T.; Pacak, K.; Brouwers, F.M.; Walther, M.M.; Linehan, W.M.; Munson, P.J.; Mannelli, M.; Goldstein, D.S.; Elkahloun, A.G. Distinct gene expression profiles in norepinephrine- and epinephrine-producing hereditary and sporadic pheochromocytomas: Activation of hypoxia-driven angiogenic pathways in von Hippel-Lindau syndrome. Endocr. Relat. Cancer 2004, 11, 897–911. [Google Scholar] [CrossRef]
- Jimenez, K.; Khare, V.; Evstatiev, R.; Kulnigg-Dabsch, S.; Jambrich, M.; Strobl, H.; Gasche, C. Increased expression of HIF2alpha during iron deficiency-associated megakaryocytic differentiation. J. Thromb. Haemost. 2015, 13, 1113–1127. [Google Scholar] [CrossRef] [PubMed]
- Patel, S.A.; Simon, M.C. Biology of hypoxia-inducible factor-2alpha in development and disease. Cell Death Differ. 2008, 15, 628–634. [Google Scholar] [CrossRef]
- Fishbein, L.; Leshchiner, I.; Walter, V.; Danilova, L.; Robertson, A.G.; Johnson, A.R.; Lichtenberg, T.M.; Murray, B.A.; Ghayee, H.K.; Else, T.; et al. Comprehensive Molecular Characterization of Pheochromocytoma and Paraganglioma. Cancer Cell 2017, 31, 181–193. [Google Scholar] [CrossRef]
- Peitzsch, M.; Pelzel, D.; Glockner, S.; Prejbisz, A.; Fassnacht, M.; Beuschlein, F.; Januszewicz, A.; Siegert, G.; Eisenhofer, G. Simultaneous liquid chromatography tandem mass spectrometric determination of urinary free metanephrines and catecholamines, with comparisons of free and deconjugated metabolites. Clin. Chim. Acta 2013, 418, 50–58. [Google Scholar] [CrossRef] [PubMed]

© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, H.; Cui, J.; Yang, C.; Rosenblum, J.S.; Zhang, Q.; Song, Q.; Pang, Y.; Fang, F.; Sun, M.; Dmitriev, P.; et al. A Transgenic Mouse Model of Pacak–Zhuang Syndrome with An Epas1 Gain-of-Function Mutation. Cancers 2019, 11, 667. https://doi.org/10.3390/cancers11050667
Wang H, Cui J, Yang C, Rosenblum JS, Zhang Q, Song Q, Pang Y, Fang F, Sun M, Dmitriev P, et al. A Transgenic Mouse Model of Pacak–Zhuang Syndrome with An Epas1 Gain-of-Function Mutation. Cancers. 2019; 11(5):667. https://doi.org/10.3390/cancers11050667
Chicago/Turabian StyleWang, Herui, Jing Cui, Chunzhang Yang, Jared S. Rosenblum, Qi Zhang, Qi Song, Ying Pang, Francia Fang, Mitchell Sun, Pauline Dmitriev, and et al. 2019. "A Transgenic Mouse Model of Pacak–Zhuang Syndrome with An Epas1 Gain-of-Function Mutation" Cancers 11, no. 5: 667. https://doi.org/10.3390/cancers11050667
APA StyleWang, H., Cui, J., Yang, C., Rosenblum, J. S., Zhang, Q., Song, Q., Pang, Y., Fang, F., Sun, M., Dmitriev, P., Gilbert, M. R., Eisenhofer, G., Pacak, K., & Zhuang, Z. (2019). A Transgenic Mouse Model of Pacak–Zhuang Syndrome with An Epas1 Gain-of-Function Mutation. Cancers, 11(5), 667. https://doi.org/10.3390/cancers11050667





