Effect of Perioperative Lidocaine, Propofol and Steroids on Pulmonary Metastasis in a Murine Model of Breast Cancer Surgery
Abstract
1. Introduction
2. Results
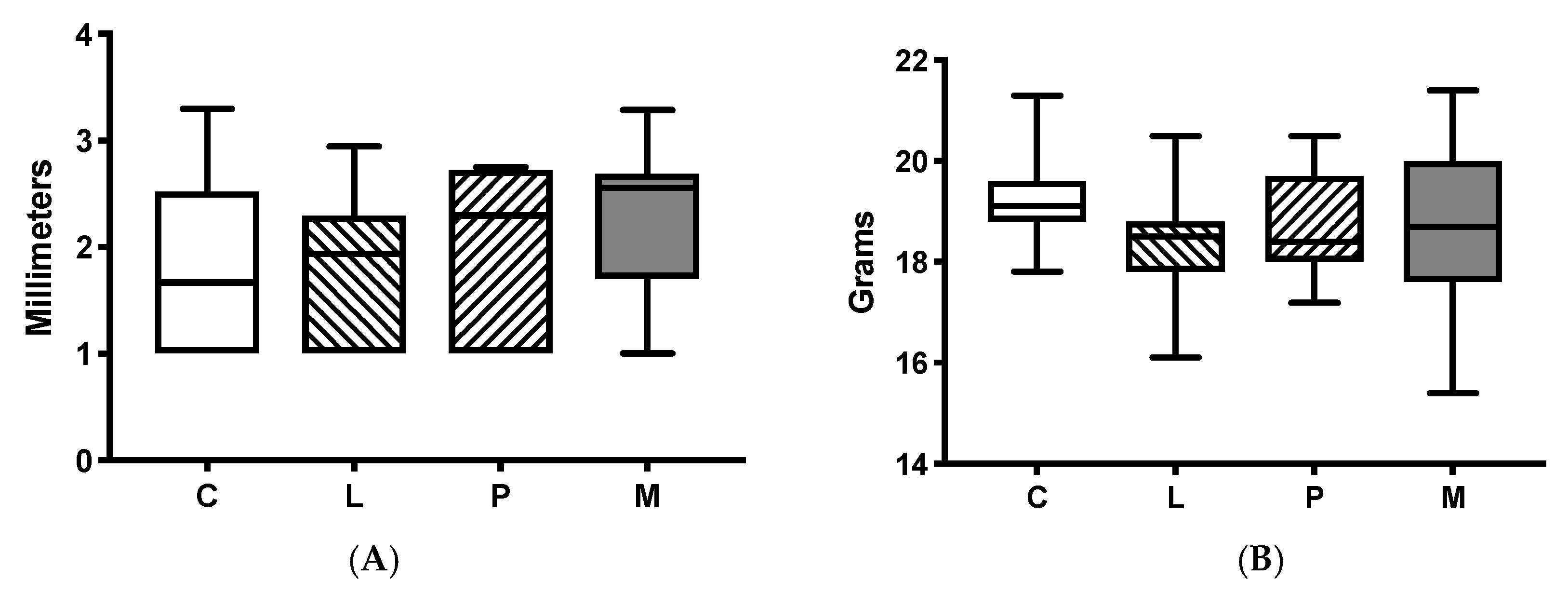
2.1. Baseline Characteristics
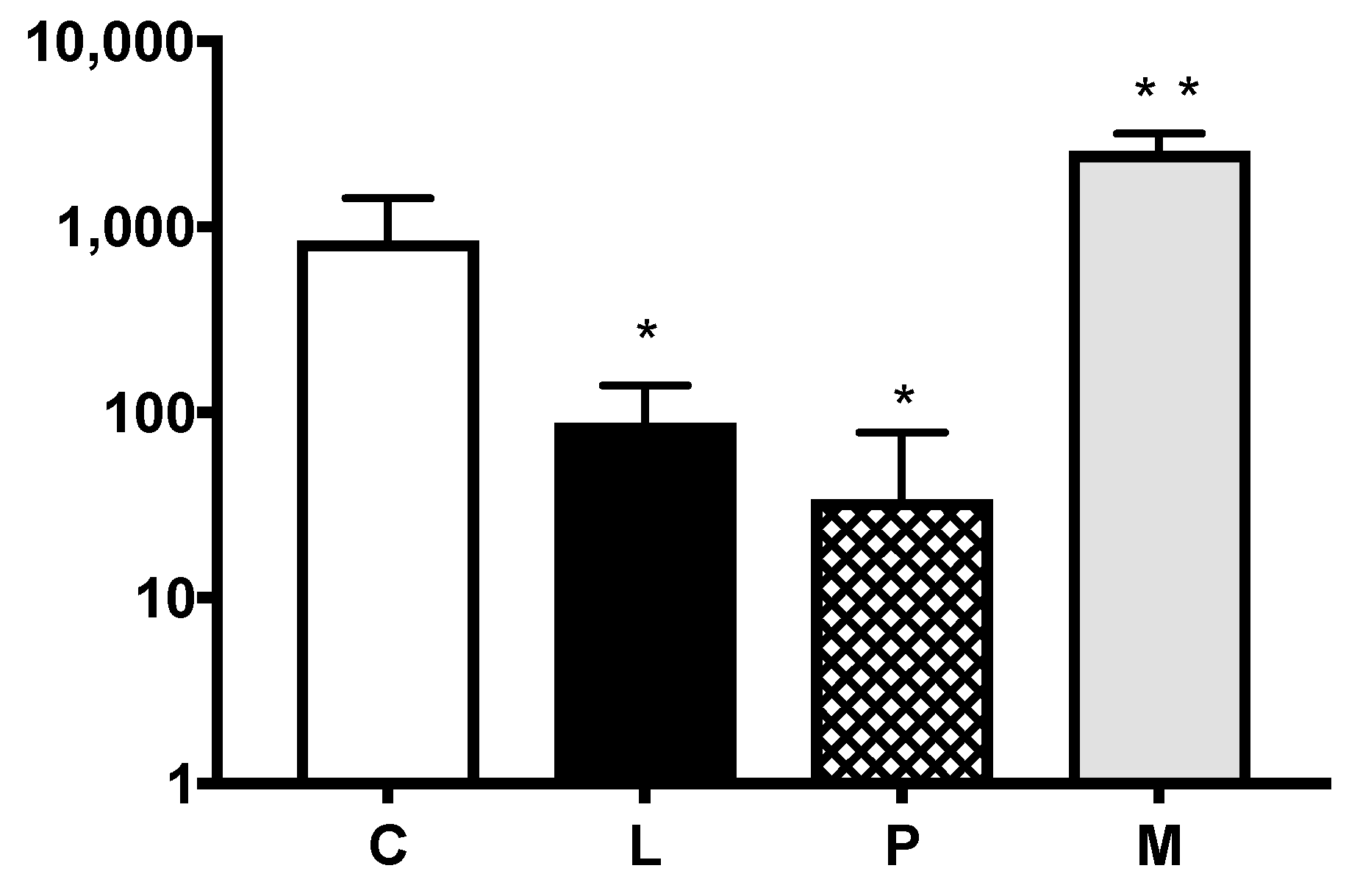
2.2. Pulmonary Colony Counts
2.3. Hepatic Colony Counts
2.4. Cytokine Analysis
3. Discussion
4. Methodology
4.1. Group Enrolment
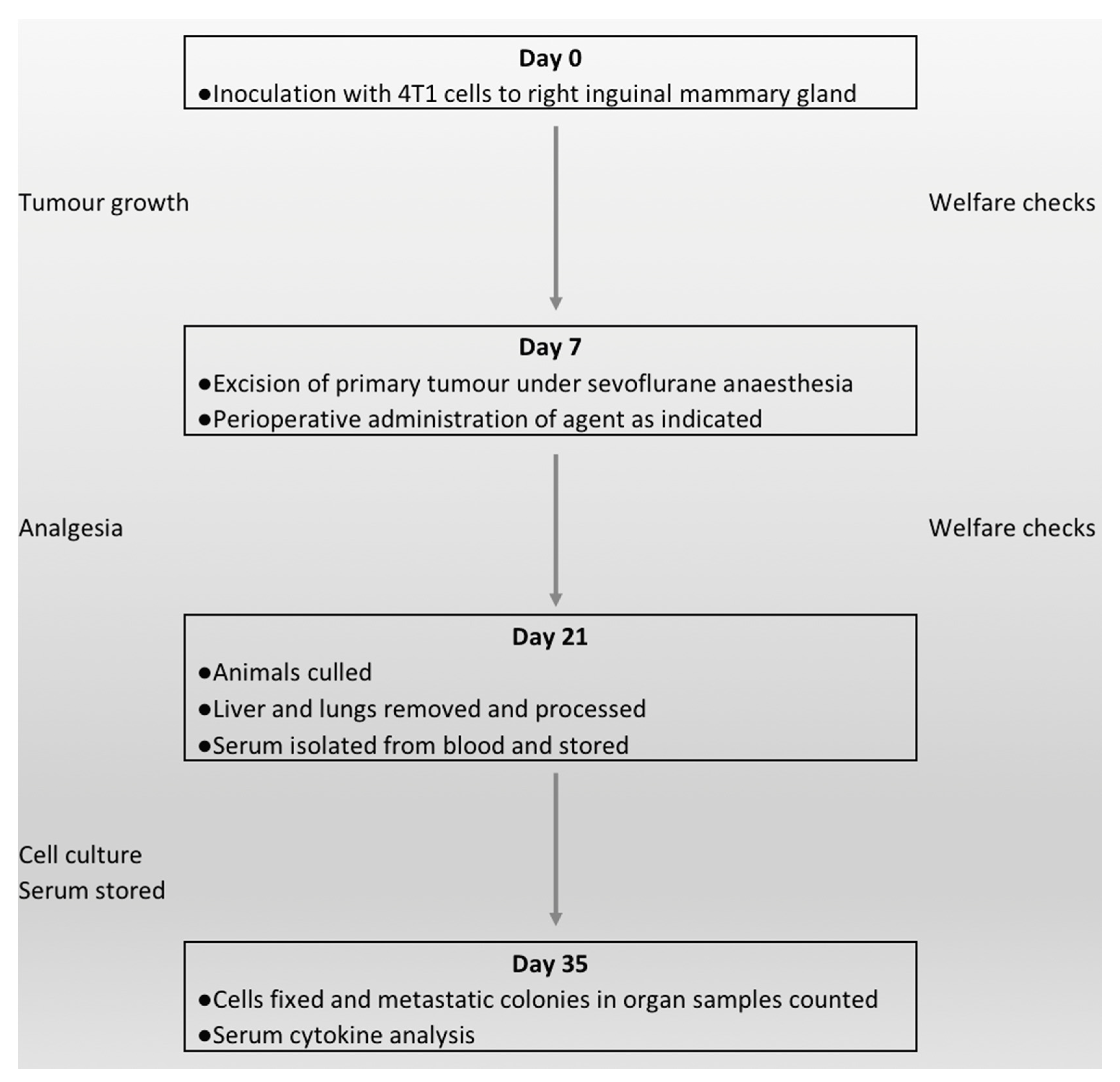
4.2. Conduct of Study
4.3. Study Day 0
4.4. Study Day 7
4.5. Study Day 7 to Study Day 21
4.6. Study Day 21 to Study Day 35
4.7. Cytokine Analysis
4.8. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- American Cancer Society. How Common is Breast Cancer? American Cancer Society (US). 2018. Available online: https://www.cancer.org/cancer/breast-cancer/about/how-common-is-breast-cancer.html (accessed on 18 July 2018).
- Gupta, G.P.; Massagué, J. Cancer metastasis: Building a framework. Cell 2006, 127, 679–695. [Google Scholar] [CrossRef] [PubMed]
- Narod, S.A.; Iqbal, J.; Miller, A.B. Why have breast cancer mortality rates declined? J. Cancer Policy 2015, 5, 8–17. [Google Scholar] [CrossRef]
- NCRI. Female Breast Cancer—Cancer Trends. National Cancer Registry Ireland. 2016. Available online: https://www.ncri.ie/publications/cancer-trends-and-projections/cancer-trends-29-breast-cancer (accessed on 2 June 2018).
- Exadaktylos, A.K.; Buggy, D.J.; Moriarty, D.C.; Mascha, E.; Sessler, D.I. Can anesthetic technique for primary breast cancer surgery affect recurrence or metastasis? Anesthesiology 2006, 105, 660–664. [Google Scholar] [CrossRef]
- Biki, B.; Mascha, E.; Moriarty, D.C.; Fitzpatrick, J.M.; Sessler, D.I.; Buggy, D.J. Anesthetic Technique for Radical Prostatectomy Surgery Affects Cancer Recurrence. A Retrospective Analysis. Anesthesiology 2008, 109, 180–187. [Google Scholar] [CrossRef]
- Heaney, A.; Buggy, D.J. Can anaesthetic and analgesic techniques affect cancer recurrence or metastasis? Br. J. Anaesth. 2012, 109, i17–i28. [Google Scholar] [CrossRef]
- Melamed, R.; Bar-Yosef, S.; Shakhar, G.; Shakhar, K.; Ben-Eliyahu, S. Suppression of Natural Killer Cell Activity and Promotion of Tumor Metastasis by Ketamine, Thiopental, and Halothane, but Not by Propofol: Mediating Mechanisms and Prophylactic Measures. Anesth. Analg. 2003, 97, 1331–1339. [Google Scholar] [CrossRef] [PubMed]
- Byrne, K.; Levins, K.J.; Buggy, D.J. Can anesthetic-analgesic technique during primary cancer surgery affect recurrence or metastasis? Can. J. Anesth. 2016, 63, 184–192. [Google Scholar] [CrossRef]
- Buggy, D.J.; Borgeat, A.; Cata, J.; Doherty, D.G.; Doornebal, C.W.; Forget, P.; Gottumukkala, V.; Gottschalk, A.; Gupta, A.; Gupta, K.; et al. Consensus statement from the BJA Workshop on Cancer and Anaesthesia. Br. J. Anaesth. 2015, 114, 2–3. [Google Scholar] [CrossRef]
- Johnson, M.; Crowley, P.; Foley, A.; Xue, C.; Gallagher, H.; Buggy, D. Does perioperative IV lidocaine infusion during tumour resection surgery reduce metastatic disease in the 4T1 moss model of breast cancer? Br. J. Anaesth. 2018, 120, e1–e2. [Google Scholar] [CrossRef]
- Song, J.; Shen, Y.; Zhang, J.; Lian, Q. Mini Profile of Potential Anticancer Properties of Propofol. PloS ONE 2014, 9, e114440. [Google Scholar] [CrossRef]
- Wigmore, T.J.; Mohammed, K.; Jhanji, S. Long-term survival for patients undergoing volatile versus IV anesthesia for cancer surgery: A retrospective analysis. Anesthesiology 2016, 124, 69–79. [Google Scholar] [CrossRef] [PubMed]
- Li, R.; Liu, H.; Dilger, J.P.; Lin, J. Effect of Propofol on breast Cancer cell, the immune system, and patient outcome. BMC Anesthesiol. 2018, 18, 77. [Google Scholar] [CrossRef] [PubMed]
- Inada, T.; Yamanouchi, Y.; Jomura, S.; Sakamoto, S.; Takahashi, M.; Kambara, T.; Shingu, K. Effect of propofol and isoflurane anaesthesia on the immune response to surgery. Anaesthesia 2004, 59, 954–959. [Google Scholar] [CrossRef]
- Lim, J.-A.; Oh, C.-S.; Yoon, T.-G.; Lee, J.-Y.; Lee, S.-H.; Yoo, Y.-B.; Yang, J.-H.; Kim, S.-H. The effect of propofol and sevoflurane on cancer cell, natural killer cell, and cytotoxic T lymphocyte function in patients undergoing breast cancer surgery: An in vitro analysis. BMC Cancer 2018, 18, 159. [Google Scholar] [CrossRef]
- Inada, T.; Kubo, K.; Shingu, K. Possible link between cyclooxygenase-inhibiting and antitumor properties of propofol. J. Anesth. 2011, 25, 569–575. [Google Scholar] [CrossRef]
- Ke, J.J.; Zhan, J.; Feng, X.B.; Wu, Y.; Rao, Y.; Wang, Y.L. A Comparison of the Effect of total Intravenous Anaesthesia with Propofol and Remifentanil and Inhalational Anaesthesia with Isoflurane on the Release of Pro-and Anti-Inflammatory Cytokines in Patients Undergoing Open Cholecystectomy. Anaesth. Intensiv. Care 2008, 36, 74–78. [Google Scholar] [CrossRef] [PubMed]
- Huang, H.; Benzonana, L.L.; Zhao, H.; Watts, H.R.; Perry, N.J.S.; Bevan, C.; Brown, R.; Ma, D. Prostate cancer cell malignancy via modulation of HIF-1α pathway with isoflurane and propofol alone and in combination. Br. J. Cancer 2014, 111, 1338–1349. [Google Scholar] [CrossRef]
- Coutinho, A.E.; Chapman, K.E. The anti-inflammatory and immunosuppressive effects of glucocorticoids, recent developments and mechanistic insights. Mol. Cell. Endocrinol. 2011, 335, 2–13. [Google Scholar] [CrossRef]
- De la Motte, L.; Kehlet, H.; Vogt, K.; Nielsen, C.H.; Groenvall, J.B.; Nielsen, H.B.; Andersen, A.; Schroeder, T.V.; Lönn, L. Preoperative methylprednisolone enhances recovery after endovascular aortic repair: A randomized, double-blind, placebo-controlled clinical trial. Ann. Surg. 2014, 26, 540–549. [Google Scholar] [CrossRef] [PubMed]
- Lunn, T.H.; Kehlet, H. Perioperative glucocorticoids in hip and knee surgery–benefit vs. harm? A review of randomized clinical trials. Acta Anaesthesiol. Scand. 2013, 57, 823–834. [Google Scholar] [CrossRef] [PubMed]
- Gao, Q.; Hsiao-Pei, M.; Wang, W.P.; Zuo, X.F.; Chen, L.Q. Effect of perioperative glucocorticoid administration on postoperative complications following esophagectomy: A meta-analysis. Oncol. Lett. 2014, 7, 349–356. [Google Scholar] [CrossRef]
- Freeman, J.; Buggy, D.J. Modelling the effects of perioperative interventions on cancer outcome: Lessons from dexmedetomidine. Br. J. Anaesth. 2018, 120, 15–17. [Google Scholar] [CrossRef] [PubMed]
- Pulaski, B.A.; Ostrand-Rosenberg, S. Reduction of established spontaneous mammary carcinoma metastases following immunotherapy with major histocompatibility complex class II and B7. 1 cell-based tumor vaccines. Cancer Res. 1998, 58, 1486–1493. [Google Scholar] [PubMed]
- Pulaski, B.A.; Ostrand-Rosenberg, S. Mouse 4T1 breast tumor model. Curr. Protoco. Immunol. 2000, 39, 20–22. [Google Scholar]
- Jaura, A.I.; Flood, G.; Gallagher, H.C.; Buggy, D.J. Differential effects of serum from patients administered distinct anaesthetic techniques on apoptosis in breast cancer cells in vitro: A pilot study. Br. J. Anaesth. 2014, 113, i63–i67. [Google Scholar] [CrossRef] [PubMed]
- Fan, W.; Zhu, X.; Wu, L.; Wu, Z.; Li, D.; Huang, F.; He, H. Propofol: An anesthetic possessing neuroprotective effects. Eur. Rev. Med. Pharmacol. Sci. 2015, 19, 1520–1529. [Google Scholar] [PubMed]
- Liu, S.; Gu, X.; Zhu, L.; Wu, G.; Zhou, H.; Song, Y.; Wu, C. Effects of propofol and sevoflurane on perioperative immune response in patients undergoing laparoscopic radical hysterectomy for cervical cancer. Medicine 2016, 95, e5479. [Google Scholar] [CrossRef]
- Meng, C.; Song, L.; Wang, J.; Li, D.; Liu, Y.; Cui, X. Propofol induces proliferation partially via downregulation of p53 protein and promotes migration via activation of the Nrf2 pathway in human breast cancer cell line MDA-MB-231. Oncol. Rep. 2017, 37, 841–848. [Google Scholar] [CrossRef]
- Kanto, J.; Gepts, E. Pharmacokinetic Implications for the Clinical Use of Propofol. Clin. Pharmacokinet. 1989, 17, 308–326. [Google Scholar] [CrossRef]
- Eipe, N.; Gupta, S.; Penning, J. Intravenous lidocaine for acute pain: An evidence-based clinical update. BJA Educ. 2016, 16, 292–298. [Google Scholar] [CrossRef]
- Xing, W.; Chen, D.-T.; Pan, J.-H.; Chen, Y.-H.; Yan, Y.; Li, Q.; Xue, R.-F.; Yuan, Y.-F.; Zeng, W.-A. Lidocaine Induces Apoptosis and Suppresses Tumor Growth in Human Hepatocellular Carcinoma Cells In Vitro and in a Xenograft Model In Vivo. Anesthesiology 2017, 126, 868–881. [Google Scholar] [CrossRef] [PubMed]
- Martin, O.A.; Anderson, R.L.; Narayan, K.; MacManus, M.P. Does the mobilization of circulating tumour cells during cancer therapy cause metastasis? Nat. Rev. Clin. Oncol. 2017, 14, 32–44. [Google Scholar] [CrossRef]
- Ames, E.; Murphy, W.J. Advantages and clinical applications of natural killer cells in cancer immunotherapy. Cancer Immunol. Immunother. 2014, 63, 21–28. [Google Scholar] [CrossRef][Green Version]
- Zargar-Shoshtari, K.; Sammour, T.; Kahokehr, A.; Connolly, A.B.; Hill, A.G. Randomized clinical trial of the effect of glucocorticoids on peritoneal inflammation and postoperative recovery after colectomy. Br. J. Surg. 2009, 96, 1253–1261. [Google Scholar] [CrossRef] [PubMed]
- Singh, P.P.; Lemanu, D.P.; Taylor, M.H.; Hill, A.G. Association between preoperative glucocorticoids and long-term survival and cancer recurrence after colectomy: Follow-up analysis of a previous randomized controlled trial. Br. J. Anaesth. 2014, 113, i68–i73. [Google Scholar] [CrossRef] [PubMed]
- Kilkenny, C.; Browne, W.J.; Cuthill, I.C.; Emerson, M.; Altman, D.G. Improving bioscience research reporting: The ARRIVE guidelines for reporting animal research. PLoS Biol. 2010, 8, e1000412. [Google Scholar] [CrossRef] [PubMed]
- Langford, D.J.; Bailey, A.L.; Chanda, M.L.; Clarke, S.E.; Drummond, T.E.; Echols, S.; Glick, S.; Ingrao, J.; Klassen-Ross, T.; Lacroix-Fralish, M.L.; et al. Coding of facial expressions of pain in the laboratory mouse. Nat. Methods 2010, 7, 447–449. [Google Scholar] [CrossRef] [PubMed]
| C | L | P | M | Total | |
|---|---|---|---|---|---|
| Mice Inoculated | 17 | 18 | 20 | 17 | 72 |
| −No tumor | 1 | 2 | 2 | 1 | 6 |
| −I.P tumor | 1 | 1 | 1 | 3 | |
| −Excessive Score | 1 | 1 | |||
| −Damaged plates | 2 | 2 | |||
| Animals Available | 15 | 15 | 15 | 15 | 60 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Freeman, J.; Crowley, P.D.; Foley, A.G.; Gallagher, H.C.; Iwasaki, M.; Ma, D.; Buggy, D.J. Effect of Perioperative Lidocaine, Propofol and Steroids on Pulmonary Metastasis in a Murine Model of Breast Cancer Surgery. Cancers 2019, 11, 613. https://doi.org/10.3390/cancers11050613
Freeman J, Crowley PD, Foley AG, Gallagher HC, Iwasaki M, Ma D, Buggy DJ. Effect of Perioperative Lidocaine, Propofol and Steroids on Pulmonary Metastasis in a Murine Model of Breast Cancer Surgery. Cancers. 2019; 11(5):613. https://doi.org/10.3390/cancers11050613
Chicago/Turabian StyleFreeman, James, Peter D. Crowley, Andrew G. Foley, Helen C. Gallagher, Masae Iwasaki, Daqing Ma, and Donal J. Buggy. 2019. "Effect of Perioperative Lidocaine, Propofol and Steroids on Pulmonary Metastasis in a Murine Model of Breast Cancer Surgery" Cancers 11, no. 5: 613. https://doi.org/10.3390/cancers11050613
APA StyleFreeman, J., Crowley, P. D., Foley, A. G., Gallagher, H. C., Iwasaki, M., Ma, D., & Buggy, D. J. (2019). Effect of Perioperative Lidocaine, Propofol and Steroids on Pulmonary Metastasis in a Murine Model of Breast Cancer Surgery. Cancers, 11(5), 613. https://doi.org/10.3390/cancers11050613






