Immunophenotype of T Cells Expressing Programmed Death-1 and Cytotoxic T Cell Antigen-4 in Early Lung Cancer: Local vs. Systemic Immune Response
Abstract
1. Introduction
2. Results
2.1. Patients
2.2. BALF
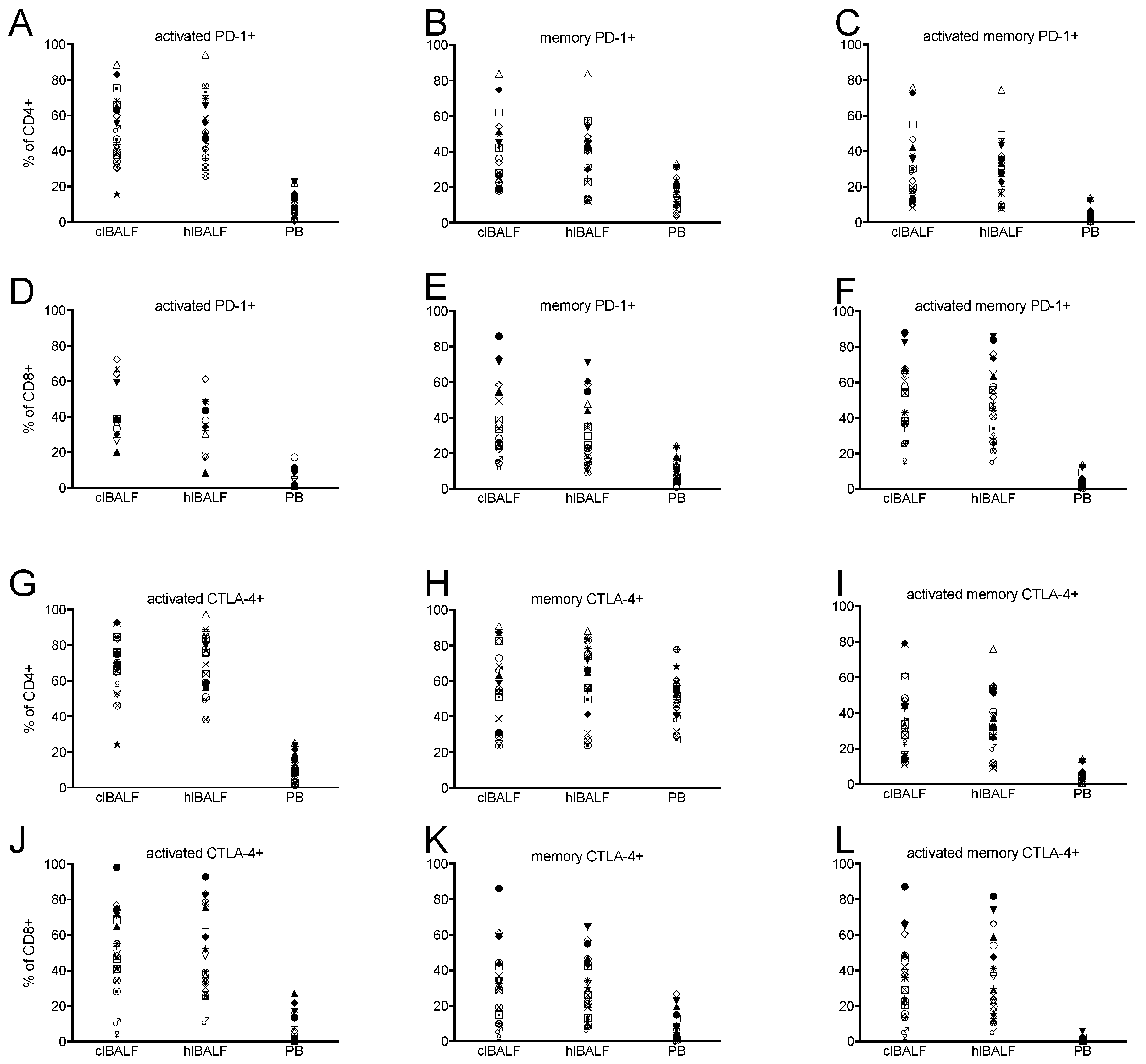
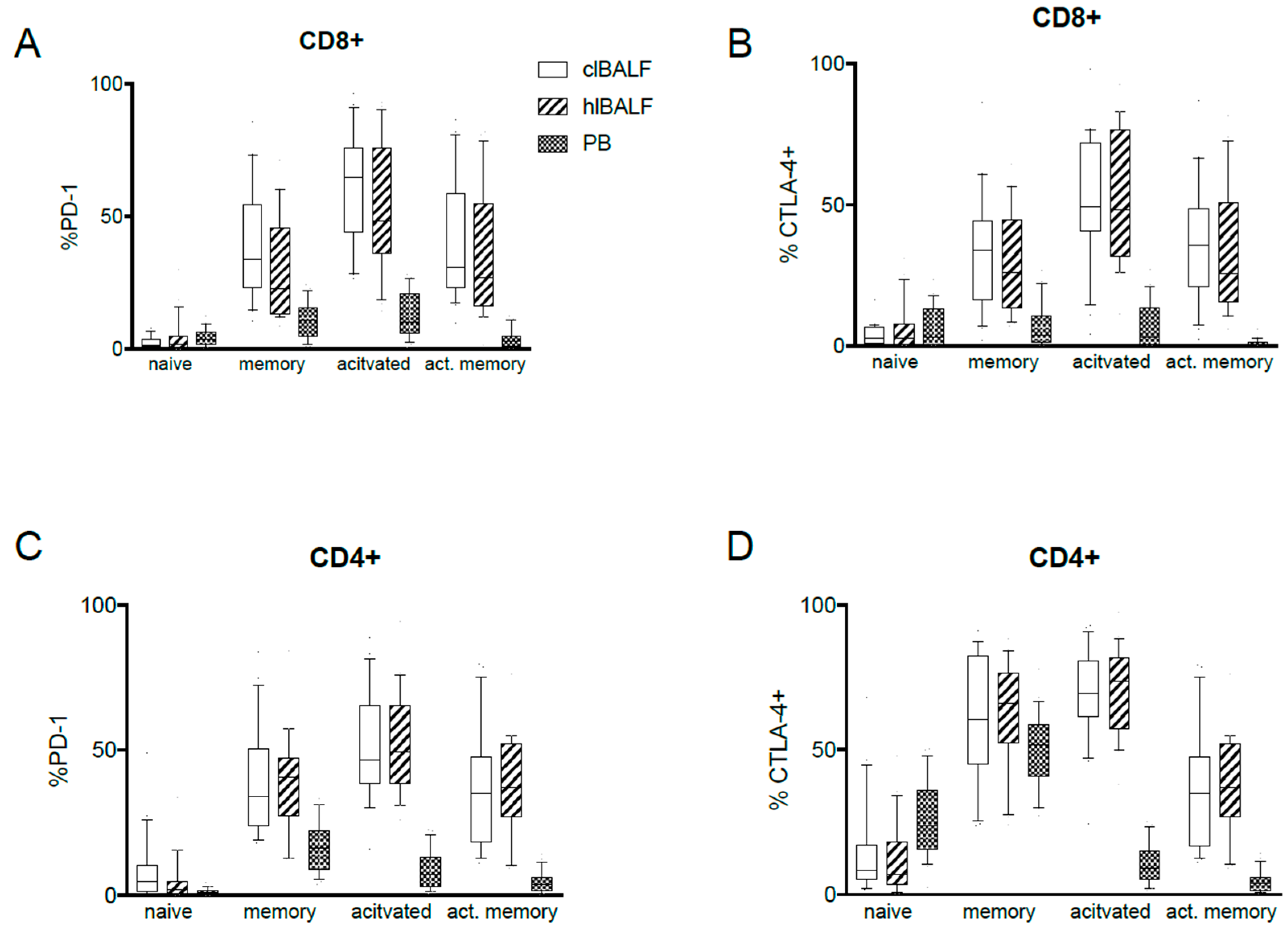
2.3. PD-1 Expression Pattern
2.4. CTLA-4 Expression Pattern
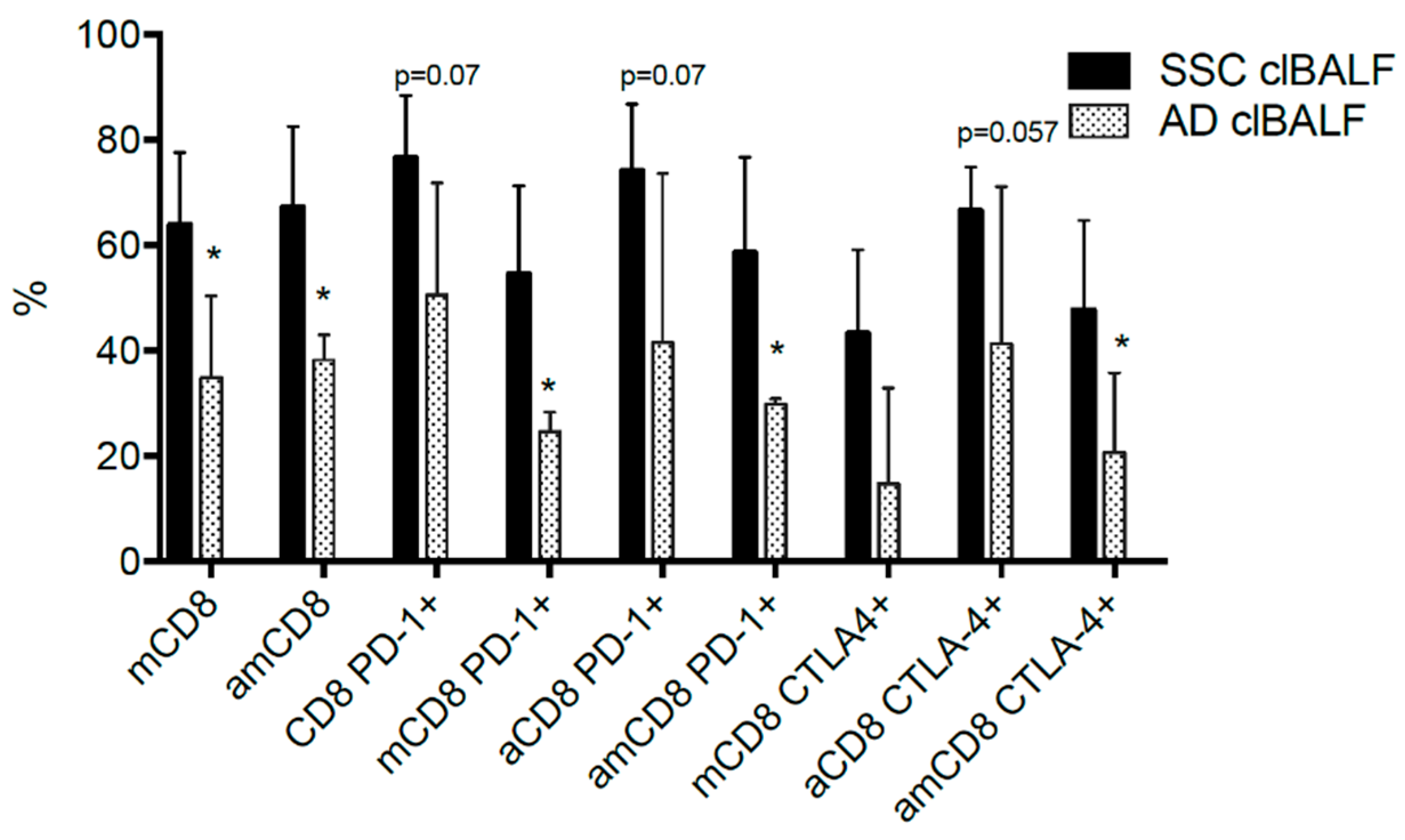
2.5. PD-1 and CTLA-4 Expression in Relation to Clinical Data
3. Discussion
4. Patients and Methods
4.1. Patients
4.2. Bronchoalveolar Lavage
4.3. Flow Cytometry Analysis
4.4. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
Availability of data
Abbreviations
References
- Costantini, A.; Grynovska, M.; Lucibello, F.; Moises, J.; Pages, F.; Tsao, M.S.; Shepherd, F.A.; Bouchaab, H.; Garassino, M.; Aerts, J.G.J.V.; et al. Immunotherapy: A new standard of care in thoracic malignancies? A summary of the European Respiratory Society research seminar of the Thoracic Oncology Assembly. Eur. Respir. J. 2018, 51. [Google Scholar] [CrossRef] [PubMed]
- Dudnik, E.; Moskovitz, M.; Daher, S.; Shamai, S.; Hanovich, E.; Grubstein, A.; Shochat, T.; Wollner, M.; Bar, J.; Merimsky, O.; et al. Effectiveness and safety of nivolumab in advanced non-small cell lung cancer: The real-life data. Lung Cancer 2017. [Google Scholar] [CrossRef]
- Mascaux, C.; Tomasini, P.; Greillier, L.; Barlesi, F. Personalised medicine for nonsmall cell lung cancer. Eur. Respir. Rev. 2017, 26. [Google Scholar] [CrossRef]
- Domagala-Kulawik, J. Immune checkpoint inhibitors in non-small cell lung cancer––Towards daily practice. Adv. Respir. Med. 2018, 86. [Google Scholar] [CrossRef] [PubMed]
- Aerts, J.G.; Hegmans, J.P. Tumor-specific cytotoxic T cells are crucial for efficacy of immunomodulatory antibodies in patients with lung cancer. Cancer Res. 2013, 73, 2381–2388. [Google Scholar] [CrossRef]
- Domagala-Kulawik, J.; Osinska, I.; Hoser, G. Mechanisms of immune response regulation in lung cancer. Transl. Lung Cancer Res. 2014, 3, 15–22. [Google Scholar] [CrossRef]
- Keir, M.E.; Butte, M.J.; Freeman, G.J.; Sharpe, A.H. PD-1 and its ligands in tolerance and immunity. Annu. Rev. Immunol. 2008, 26, 677–704. [Google Scholar] [CrossRef] [PubMed]
- Mahoney, K.M.; Rennert, P.D.; Freeman, G.J. Combination cancer immunotherapy and new immunomodulatory targets. Nat. Rev. Drug Discov. 2015, 14, 561–584. [Google Scholar] [CrossRef]
- Ahmadzadeh, M.; Johnson, L.A.; Heemskerk, B.; Wunderlich, J.R.; Dudley, M.E.; White, D.E.; Rosenberg, S.A. Tumor antigen-specific CD8 T cells infiltrating the tumor express high levels of PD-1 and are functionally impaired. Blood 2009, 114, 1537–1544. [Google Scholar] [CrossRef]
- Pauken, K.E.; Wherry, E.J. SnapShot: T Cell Exhaustion. Cell 2015, 163, 1038. [Google Scholar] [CrossRef] [PubMed]
- Topalian, S.L.; Sznol, M.; McDermott, D.F.; Kluger, H.M.; Carvajal, R.D.; Sharfman, W.H.; Brahmer, J.R.; Lawrence, D.P.; Atkins, M.B.; Powderly, J.D.; et al. Survival, durable tumor remission, and long-term safety in patients with advanced melanoma receiving nivolumab. J. Clin. Oncol. 2014, 32, 1020–1030. [Google Scholar] [CrossRef] [PubMed]
- Wolchok, J.D.; Hoos, A.; O’Day, S.; Weber, J.S.; Hamid, O.; Lebbe, C.; Maio, M.; Binder, M.; Bohnsack, O.; Nichol, G.; et al. Guidelines for the evaluation of immune therapy activity in solid tumors: Immune-related response criteria. Clin. Cancer Res. 2009, 15, 7412–7420. [Google Scholar] [CrossRef] [PubMed]
- Walker, L.S.; Sansom, D.M. Confusing signals: recent progress in CTLA-4 biology. Trends Immunol. 2015, 36, 63–70. [Google Scholar] [CrossRef] [PubMed]
- Sansom, D.M.; Walker, L.S. The role of CD28 and cytotoxic T-lymphocyte antigen-4 (CTLA-4) in regulatory T-cell biology. Immunol. Rev. 2006, 212, 131–148. [Google Scholar] [CrossRef] [PubMed]
- Rudd, C.E. CTLA-4 co-receptor impacts on the function of Treg and CD8+ T-cell subsets. Eur. J. Immunol. 2009, 39, 687–690. [Google Scholar] [CrossRef]
- Krummey, S.M.; Cheeseman, J.A.; Conger, J.A.; Jang, P.S.; Mehta, A.K.; Kirk, A.D.; Larsen, C.P.; Ford, M.L. High CTLA-4 expression on Th17 cells results in increased sensitivity to CTLA-4 coinhibition and resistance to belatacept. Am. J. Transplant. 2014, 14, 607–614. [Google Scholar] [CrossRef]
- Lindsten, T.; Lee, K.P.; Harris, E.S.; Petryniak, B.; Craighead, N.; Reynolds, P.J.; Lombard, D.B.; Freeman, G.J.; Nadler, L.M.; Gray, G.S.; et al. Characterization of CTLA-4 structure and expression on human T cells. J. Immunol. 1993, 151, 3489–3499. [Google Scholar]
- Brunner, M.C.; Chambers, C.A.; Chan, F.K.; Hanke, J.; Winoto, A.; Allison, J.P. CTLA-4-Mediated inhibition of early events of T cell proliferation. J. Immunol. 1999, 162, 5813–5820. [Google Scholar]
- Iida, T.; Ohno, H.; Nakaseko, C.; Sakuma, M.; Takeda-Ezaki, M.; Arase, H.; Kominami, E.; Fujisawa, T.; Saito, T. Regulation of cell surface expression of CTLA-4 by secretion of CTLA-4-containing lysosomes upon activation of CD4+ T cells. J. Immunol. 2000, 165, 5062–5068. [Google Scholar] [CrossRef]
- Wang, X.B.; Zheng, C.Y.; Giscombe, R.; Lefvert, A.K. Regulation of surface and intracellular expression of CTLA-4 on human peripheral T cells. Scand. J. Immunol. 2001, 54, 453–458. [Google Scholar] [CrossRef]
- Kwiecien, I.; Stelmaszczyk-Emmel, A.; Polubiec-Kownacka, M.; Dziedzic, D.; Domagala-Kulawik, J. Elevated regulatory T cells, surface and intracellular CTLA-4 expression and interleukin-17 in the lung cancer microenvironment in humans. Cancer Immunol. Immunother. 2017, 66, 161–170. [Google Scholar] [CrossRef]
- Arrieta, O.; Montes-Servin, E.; Hernandez-Martinez, J.M.; Cardona, A.F.; Casas-Ruiz, E.; Crispin, J.C.; Motola, D.; Flores-Estrada, D.; Barrera, L. Expression of PD-1/PD-L1 and PD-L2 in peripheral T-cells from non-small cell lung cancer patients. Oncotarget 2017, 8, 101994–102005. [Google Scholar] [CrossRef] [PubMed]
- Waki, K.; Yamada, T.; Yoshiyama, K.; Terazaki, Y.; Sakamoto, S.; Matsueda, S.; Komatsu, N.; Sugawara, S.; Takamori, S.; Itoh, K.; et al. PD-1 expression on peripheral blood T-cell subsets correlates with prognosis in non-small cell lung cancer. Cancer Sci. 2014, 105, 1229–1235. [Google Scholar] [CrossRef]
- Rutkowski, J.; Cyman, M.; Slebioda, T.; Bemben, K.; Rutkowska, A.; Gruchala, M.; Kmiec, Z.; Pliszka, A.; Zaucha, R. Evaluation of peripheral blood T lymphocyte surface activation markers and transcription factors in patients with early stage non-small cell lung cancer. Cell Immunol. 2017, 322, 26–33. [Google Scholar] [CrossRef]
- Tassi, E.; Grazia, G.; Vegetti, C.; Bersani, I.; Bertolini, G.; Molla, A.; Baldassari, P.; Andriani, F.; Roz, L.; Sozzi, G.; et al. Early Effector T Lymphocytes Coexpress Multiple Inhibitory Receptors in Primary Non-Small Cell Lung Cancer. Cancer Res. 2017, 77, 851–861. [Google Scholar] [CrossRef] [PubMed]
- Domagala-Kulawik, J.; Hoser, G.; Droszcz, P.; Kawiak, J.; Droszcz, W.; Chazan, R. T-cell subtypes in bronchoalveolar lavage fluid and in peripheral blood from patients with primary lung cancer. Diagn. Cytopathol. 2001, 25, 208–213. [Google Scholar] [CrossRef] [PubMed]
- Domagala-Kulawik, J.; Raniszewska, A. How to evaluate the immune status of lung cancer patients before immunotherapy. Breathe (Sheff.) 2017, 13, 291–296. [Google Scholar] [CrossRef]
- Osinska, I.; Stelmaszczyk-Emmel, A.; Polubiec-Kownacka, M.; Dziedzic, D.; Domagala-Kulawik, J. CD4+/CD25high/FoxP3+/CD127-regulatory T cells in bronchoalveolar lavage fluid of lung cancer patients. Hum. Immunol. 2016. [Google Scholar] [CrossRef]
- Chcialowski, A.; Chorostowska-Wynimko, J.; Fal, A.; Pawlowicz, R.; Domagala-Kulawik, J. Recommendation of the Polish Respiratory Society for bronchoalveolar lavage (BAL) sampling, processing and analysis methods. Pneumonol. Alergol. Pol. 2011, 79, 75–89. [Google Scholar]
- Purwar, R.; Campbell, J.; Murphy, G.; Richards, W.G.; Clark, R.A.; Kupper, T.S. Resident memory T cells (T(RM)) are abundant in human lung: Diversity, function, and antigen specificity. PLoS ONE 2011, 6, e16245. [Google Scholar] [CrossRef]
- Jameson, S.C.; Masopust, D. Understanding Subset Diversity in T Cell Memory. Immunity 2018, 48, 214–226. [Google Scholar] [CrossRef]
- Huang, A.C.; Postow, M.A.; Orlowski, R.J.; Mick, R.; Bengsch, B.; Manne, S.; Xu, W.; Harmon, S.; Giles, J.R.; Wenz, B.; et al. T-cell invigoration to tumour burden ratio associated with anti-PD-1 response. Nature 2017, 545, 60–65. [Google Scholar] [CrossRef]
- Chen, D.S.; Mellman, I. Elements of cancer immunity and the cancer-immune set point. Nature 2017, 541, 321–330. [Google Scholar] [CrossRef]
- Nowicki, T.S.; Hu-Lieskovan, S.; Ribas, A. Mechanisms of Resistance to PD-1 and PD-L1 Blockade. Cancer J. 2018, 24, 47–53. [Google Scholar] [CrossRef] [PubMed]
- Voong, K.R.; Feliciano, J.; Becker, D.; Levy, B. Beyond PD-L1 testing-emerging biomarkers for immunotherapy in non-small cell lung cancer. Ann. Transl. Med. 2017, 5, 376. [Google Scholar] [CrossRef] [PubMed]
- Djenidi, F.; Adam, J.; Goubar, A.; Durgeau, A.; Meurice, G.; de Montpréville, V.; Validire, P.; Besse, B.; Mami-Chouaib, F. CD8+CD103+ tumor-infiltrating lymphocytes are tumor-specific tissue-resident memory T cells and a prognostic factor for survival in lung cancer patients. J. Immunol. 2015, 194, 3475–3486. [Google Scholar] [CrossRef] [PubMed]
- Hoser, G.; Domagala-Kulawik, J.; Droszcz, P.; Droszcz, W.; Kawiak, J. Lymphocyte subsets differences in smokers and nonsmokers with primary lung cancer: A flow cytometry analysis of bronchoalveolar lavage fluid cells. Med. Sci. Monit. 2003, 9, BR310–BR315. [Google Scholar] [PubMed]
- Zikos, T.A.; Donnenberg, A.D.; Landreneau, R.J.; Luketich, J.D.; Donnenberg, V.S. Lung T-cell subset composition at the time of surgical resection is a prognostic indicator in non-small cell lung cancer. Cancer Immunol. Immunother. 2011, 60, 819–827. [Google Scholar] [CrossRef] [PubMed]
- Simoni, Y.; Becht, E.; Fehlings, M.; Loh, C.Y.; Koo, S.L.; Teng, K.W.W.; Yeong, J.P.S.; Nahar, R.; Zhang, T.; Kared, H.; et al. Bystander CD8(+) T cells are abundant and phenotypically distinct in human tumour infiltrates. Nature 2018, 557, 575–579. [Google Scholar] [CrossRef] [PubMed]
- Brahmer, J.R. PD-1-targeted immunotherapy: Recent clinical findings. Clin. Adv. Hematol. Oncol. 2012, 10, 674–675. [Google Scholar]
- Bally, A.P.; Austin, J.W.; Boss, J.M. Genetic and Epigenetic Regulation of PD-1 Expression. J. Immunol. 2016, 196, 2431–2437. [Google Scholar] [CrossRef] [PubMed]
- Buchbinder, E.I.; Desai, A. CTLA-4 and PD-1 Pathways: Similarities, Differences, and Implications of Their Inhibition. Am. J. Clin. Oncol. 2016, 39, 98–106. [Google Scholar] [CrossRef]
- Hellmann, M.D.; Ciuleanu, T.E.; Pluzanski, A.; Lee, J.S.; Otterson, G.A.; Audigier-Valette, C.; Minenza, E.; Linardou, H.; Burgers, S.; Salman, P.; et al. Nivolumab plus Ipilimumab in Lung Cancer with a High Tumor Mutational Burden. N. Engl. J. Med. 2018, 378, 2093–2104. [Google Scholar] [CrossRef] [PubMed]
- Zou, W.; Chen, L. Inhibitory B7-family molecules in the tumour microenvironment. Nat. Rev. Immunol. 2008, 8, 467–477. [Google Scholar] [CrossRef] [PubMed]
- Forde, P.M.; Chaft, J.E.; Smith, K.N.; Anagnostou, V.; Cottrell, T.R.; Hellmann, M.D.; Zahurak, M.; Yang, S.C.; Jones, D.R.; Broderick, S.; et al. Neoadjuvant PD-1 Blockade in Resectable Lung Cancer. N. Engl. J. Med. 2018, 378, 1976–1986. [Google Scholar] [CrossRef] [PubMed]
- Lee, C.K.; Man, J.; Lord, S.; Links, M.; Gebski, V.; Mok, T.; Yang, J.C. Checkpoint Inhibitors in Metastatic EGFR-Mutated Non-Small Cell Lung Cancer-A Meta-Analysis. J. Thorac. Oncol. 2017, 12, 403–407. [Google Scholar] [CrossRef] [PubMed]
| Variable | Patients |
|---|---|
| Sex F/M (n) | 12/9 |
| Age (mean ± SD years) | 66.8 ± 7.6 |
| Women (mean ± SD years) | 66.8 ± 7.6 |
| Men (mean ± SD years) | 66.7 ± 7.5 |
| Smoking history | |
| Smokers/ex-smokers/never-smokers (n, %) | 14 (76.2%)/5 (23.8%)/2 (9.5%) |
| Pack/years (mean ± SD) | 38.9 ± 16.3 |
| Histology (n, %) | |
| Squamous cell carcinoma | 10 (47.6%) |
| Adenocarcinoma | 7 (33.3%) |
| Large cell | 2 (9.5%) |
| NOS | 2 (9.5%) |
| Cancer grade | G2-6 (26%) |
| G3-5 (21%) | |
| NA-53% | |
| EGFR mutation, ALK rearrageemnt | Non confrimed |
| Stage of disease (n,%) | |
| IA | 6 (28.6%) |
| IB | 7 (33.3%) |
| IIB | 4 (19.0%) |
| IIIA | 4 (19%) |
| Metastases | 0 |
| Symptoms of the respiratory system (interview of the patient) (yes/no (n, %)) | 11 (52.4%)/10 (47.6%) |
| Cough | 13 (61.9%)/8 (38.1%) |
| Hemoptysis | 0 (0.0%)/21 (100.0%) |
| Dyspnea | 16 (76.2%)/5 (23.8%) |
| One year follow-up | |
| Surgical resection | 16 (69%) |
| Progression | 1 (5%) |
| Death | 0 |
| No data | 6 (26%) |
| Cell Type | A. clBALF n = 21 | B. hlBALF n = 21 | C. PB n = 21 | p < 0.05 * Group A-B-C ANOVA, Kruskal-Wallis | p < 0.05 * Group in Groups Post-Hoc |
|---|---|---|---|---|---|
| Lymphocytes | 17.4 (10.2–22.1) | 17.1 (12.8–19.7) | 30.7 (25.9–36.3) | 0.0003 | A-C: 0.0009 |
| B-C: 0.0023 | |||||
| T cells CD3+ (% of all cells) | 7.7 (4.5–9.4) | 7.9 (6.4–12.6) | 16.4 (13.5–26.4) | <0.0001 | A-C: <0.0001 |
| B-C: 0.0028 | |||||
| T cells CD3+ (% of all lymphocytes) | 59.0 (52.2–67.8) | 58.4 (36.3–67.3) | 59.0 (52.2–67.8) | 0.1689 | - |
| CD8+ (% of T cells) | 30.2 (22.4–37.0) | 33.7 (23.4–44.1) | 29.8 (25.6–37.7) | 0.7782 | - |
| CD4+ (% of T cells) | 24.2 (15.0–38.9) | 19.7 (14.9–29.1) | 57.8 (48.7–64.1) | <0.0001 | A-C: <0.0001 |
| B-C: <0.0001 | |||||
| Ratio CD4: CD8 | 0.8 (0.5–1.3) | 0.6 (0.4–1.3) | 2.1 (1.3–2.4) | 0.0003 | A-C: 0.0037 |
| B-C: 0.0008 | |||||
| CD8+ subpopulation: (% of CD8+ cells) | |||||
| Naïve CD8+ (CD8+CD45RA+CD127+) | 4.3 (2.5–7.8) | 3.9 (1.4–8.3) | 28.3 (25.3–48.4) | <0.0001 | A-C: <0.0001 |
| B-C: <0.0001 | |||||
| Memory CD8+ (CD8+CD127+CD45RA−) | 53.9 (34.9–62.1) | 50.4 (29.1–57.9) | 29.3 (17.3–42.1) | 0.0120 | A-C: 0.0011 |
| B-C: 0.0347 | |||||
| Activated CD8+ (CD8+CD69+CD127−CD45RA−) | 92.3 (86.3–93.9) | 86.8 (83.4–94.4) | 26.0 (11.7–38.9) | <0.0001 | A-C: <0.0001 |
| B-C: <0.0001 | |||||
| Activated memory CD8+ (CD8+CD69+CD127+CD45RA) | 54.8 (38.0–67.1) | 47.3 (29.7–63.5) | 3.1 (1.4–4.7) | <0.0001 | A-C: <0.0001 |
| B-C: <0.0001 | |||||
| CD4+ subpopulation: (% of CD4+ cells) | |||||
| Naïve CD4+ (CD4+CD45RA+CD127+) | 8.8 (5.7–25.0) | 7.3 (3.7–12.1) | 23.7 (17.0–35.5) | 0.0060 | - |
| B-C: 0.0055 | |||||
| Memory CD4 + (CD4+CD127+CD45RA−) | 33.9 (25.0–49.6) | 40.7 (30.0–46.0) | 16.2 (9.1–21.0) | 0.0341 | - |
| B-C: 0.0505 | |||||
| Activated CD4+ (CD4+CD69+CD127−CD45RA−) | 74.0 (66.7–78.6) | 73.8 (59.0–82.3) | 8.9 (7.2–14.2) | <0.0001 | A-C: <0.0001 |
| B-C: <0.0001 | |||||
| Activated memory CD4+ (CD4+CD69+CD127+CD45RA) | 35.0 (19.7–47.1) | 37.1 (27.3–51.8) | 3.7 (1.8–5.9) | <0.0001 | A-C: <0.0001 |
| B-C: <0.0001 |
| Lymphocyte Subset (%) Median (p25–p75) | A. clBALF n = 21 | B. hlBALF n = 21 | C. PB n = 21 | p < 0.05 * Group A-B-C ANOVA, Kruskal-Wallis | p < 0.05 * Group, in Groups Post-Hoc |
|---|---|---|---|---|---|
| CD8+ subpopulation: (% of CD8+ cells) | |||||
| all CD8+PD1+ (CD8+PD1+) | 68.1 (50.6–79.3) | 51.8 (40.9–78.7) | 25.9 (20.2–33.8) | <0.0001 | A-C: <0.0001 |
| B-C: 0.0001 | |||||
| naïve CD8+ PD-1+ (CD8+CD45RA+CD127+PD-1+) | 1.5 (1.1–3.3) | 1.8 (0.5–4.3) | 3.7 (2.4–5.9) | 0.0608 | - |
| memory CD8+ PD-1+ (CD8+CD127+CD45RA-PD-1+) | 33.8 (24.0–54.2) | 22.9 (13.5–44.0) | 11.0 (5.5–14.5) | <0.0001 | A-C: <0.0001 |
| B-C: 0.0002 | |||||
| activated CD8+ PD-1+ (CD8+CD69+CD127-CD45RA-PD-1+) | 64.7 (47.1–75.1) | 48.4 (37.5–70.2) | 10.2 (6.4–19.4) | <0.0001 | A-C: <0.0001 |
| B-C: <0.0001 | |||||
| activated memory CD8+ PD-1+ (CD8+CD69+CD127+CD45RA-PD1+) | 30.9 (23.7–57.0) | 26.9 (18.1–53.0) | 2.8 (1.1–4.4) | <0.0001 | A-C: <0.0001 |
| B-C: <0.0001 | |||||
| GMF | |||||
| all CD8+PD1+ (CD8+PD1+) | 2202 (1890–2530) | 2007 (1793–2431) | 2158 (1769–2580) | 0.5683 | - |
| naïve CD8+ PD-1+ (CD8+CD45RA+CD127+PD-1+) | 1825.5 (1489.5–2070) | 1886 (1376–2431) | 1500 (1354–1707) | 0.0139 | A-C: 0.0406 |
| B-C: 0.0313 | |||||
| memory CD8+ PD-1+ (CD8+CD127+CD45RA-PD-1+) | 2283 (1933–2685) | 2248 (1746–2556) | 2261 (2003–2646) | 0.7570 | - |
| activated CD8+ PD-1+ (CD8+CD69+CD127-CD45RA-PD-1+) | 2267 (2013–2677) | 2151 (1843–2444) | 3416 (3043–3694) | 0.0001 | A-C: 0.0023 |
| B-C: 0.0001 | |||||
| activated memory CD8+ PD-1+ (CD8+CD69+CD127+CD45RA-PD1+) Geo Mean | 2476 (1974–2771) | 2289 (1918–2572) | 4282 (4013–4472) | <0.0001 | A-C: 0.0001 |
| B-C: <0.0001 | |||||
| CD4+ subpopulation: (% of CD4+ cells) | |||||
| all CD4+PD1+ (CD4+PD1+) | 52.3 (44.3–68.6) | 54.8 (45.4–68.4) | 25.0 (16.3–37.2) | <0.0001 | A-C: <0.0001 |
| B-C: <0.0001 | |||||
| naïve CD4+ PD-1+ (CD4+CD45RA+CD127+PD-1+) | 4.9 (1.5–10.1) | 2.0 (0.5–4.9) | 0.8 (0.3–1.8) | 0.0013 | A-C: 0.0008 |
| A-B:0.07 | |||||
| memory CD4+ PD-1+ (CD4+CD127+CD45RA-PD-1+) | 61.0 (51.0–82.2) | 66.5 (54.9–75.5) | 51.7 (41.2–57.4) | <0.0001 | A-C: 0.0001 |
| B-C: <0.0001 | |||||
| activated CD4+ PD-1+ (CD4+CD69+CD127-CD45RA-PD-1+) | 46.6 (38.9–64.8) | 49.4 (40.5–65.1) | 7.4 (3.3–12.0) | <0.0001 | A-C: <0.0001 |
| B-C: <0.0001 | |||||
| activated memory CD4+ PD-1 (CD4+CD69+CD127+CD45RA-PD1+) | 23.4 (14.4–37.7) | 30.7 (18.2–35.8) | 3.6 (1.8–5.6) | <0.0001 | A-C: <0.0001 |
| B-C: <0.0001 | |||||
| GMF | |||||
| all CD4+PD1+ (CD4+PD1+) | 2471 (2071–3036) | 2658 (2271–3274) | 2511 (2133–2626) | 0.2275 | - |
| naïve CD4+ PD-1+ (CD4+CD45RA+CD127+PD-1+) | 1797 (1665–2034) | 1867 (1693–2134) | 1759 (1575–1957) | 0.5243 | - |
| memory CD4+ PD-1+ (CD4+CD127+CD45RA-PD-1+) | 2775 (2331–3323) | 2737 (2635–3325) | 2310 (2076–2495) | 0.0019 | A-C: 0.0196 |
| B-C: 0.0026 | |||||
| activated CD4+ PD-1+ (CD4+CD69+CD127-CD45RA-PD-1+) | 2699 (2311–3304) | 2839 (2423–3424) | 4933 (4631–5426) | <0.0001 | A-C: <0.0001 |
| B-C: 0.0001 | |||||
| activated memory CD4+ PD-1 (CD4+CD69+CD127+CD45RA-PD1+) | 3271 (2551–3682) | 3117 (2799–3780) | 5112 (4899–5314) | <0.0001 | A-C: <0.0001 |
| B-C: <0.0001 | |||||
| Lymphocyte Subset (%) Median (p25–p75) | A. clBALF n = 21 | B. hlBALF n = 21 | C. PB n = 21 | p < 0.05 * Group A-B-C ANOVA, Kruskal-Wallis | p < 0.05 * Group, in Groups Post-Hoc |
|---|---|---|---|---|---|
| CD8+ subpopulation (% of CD8+ cells) | |||||
| all CD8+CTLA-4+ (CD8+CTLA-4+) | 57.9 (45.6–76.3) | 52.6 (37.9–80.7) | 15.6 (2.8–46.3) | 0.0001 | A-C: 0.0002 |
| B-C: 0.0005 | |||||
| naïve CD8+ CTLA-4+ (CD8+CD45RA+CD127+CTLA-4+) | 2.7 (1.0–6.4) | 2.7 (0.7–6.0) | 3.1 (0.2–12.2) | 0.9720 | - |
| - | |||||
| memory CD8+ CTLA-4+ (CD8+CD127+CD45RA-CTLA-4+) | 33.7 (17.4–44.2) | 26.1 (13.4–43.1) | 3.7 (1.8–8.5) | <0.0001 | A-C: <0.00001 |
| B-C: <0.0001 | |||||
| activated CD8+ CTLA-4+ (CD8+CD69+CD127-CD45RA-CTLA-4+) | 49.4 (40.9–71.0) | 48.4 (33.4–75.5) | 2.9 (0.3–13.1) | <0.0001 | A-C: <0.0001 |
| B-C: <0.0001 | |||||
| activated memory CD8+ CTLA-4+ (CD8+CD69+CD127+CD45RA-CTLA-4+) | 35.7 (21.0–48.3) | 25.7 (15.9–47.5) | 0.2 (0.1–1.3) | <0.0001 | A-C: <0.0001 |
| B-C: <0.0001 | |||||
| GMF | |||||
| all CD8+CTLA-4+ (CD8+CTLA-4) Geo Mean | 859 (832–963) | 839 (794–1001) | 2371 (2279–3311) | <0.0001 | A-C: <0.0001 |
| B-C: <0.0001 | |||||
| naïve CD8+ CTLA-4+ GMF (CD8+CD45RA+CD127+CTLA-4+) Geo Mean | 996.5 (889.5–1225.5) | 926 (869–1251) | 2393 (2236–2692) | <0.0001 | A-C: <0.0001 |
| B-C: <0.0001 | |||||
| memory CD8+ CTLA-4+ GMF (CD8+CD127+CD45RA-CTLA-4+) Geo Mean | 898 (850–934) | 881 (828–1014) | 2482 (2357–3500) | <0.0001 | A-C: <0.0001 |
| B-C: <0.0001 | |||||
| activated CD8+ CTLA-4+ GMF (CD8+CD69+CD127-CD45RA-CTLA-4+) Geo Mean | 873 (848–973) | 860 (791–1022) | 2477 (2318–3231) | <0.0001 | A-C: <0.0001 |
| B-C: <0.0001 | |||||
| activated memory CD8+ CTLA-4+ GMF (CD8+CD69+CD127+CD45RA-CTLA-4+) Geo Mean | 921 (863–983) | 903 (822–1042) | 2506 (2363–3328) | <0.0001 | A-C: <0.0001 |
| B-C: <0.0001 | |||||
| CD4+ subpopulation (% of CD4+ cells) | |||||
| all CD4+CTLA-4+ (CD4+CTLA-4+) | 98.5 (92.1–99.2) | 99.6 (96.8–99.9) | 100.0 (99.9–100.0) | 0.0689 | - |
| naïve CD4+ CTLA-4+ (CD4+CD45RA+CD127+CTLA-4+) | 8.3 (5.4–16.1) | 7.1 (3.6–11.4) | 23.7 (17.0–35.4) | 0.0009 | A-C: 0.0126 |
| B-C: 0.0013 | |||||
| memory CD4+ CTLA-4+ (CD4+CD127+CD45RA-CTLA-4+) | 60.4 (51.0–82.1) | 66.1 (54.8–75.0) | 51.7 (41.2–57.4) | 0.0505 | - |
| activated CD4+ CTLA-4+ (CD4+CD69+CD127-CD45RA-CTLA-4+) | 69.5 (65.0–78.1) | 73.5 (57.8–80.0) | 8.9 (7.2–14.2) | <0.0001 | A-C: <0.0001 |
| B-C: <0.0001 | |||||
| activated memory CD4+ CTLA-4+ (CD4+CD69+CD127+CD45RA-CTLA-4+) | 35.0 (16.7–47.1) | 37.1 (27.3–51.7) | 3.7 (1.8–5.9) | <0.0001 | A-C: <0.0001 |
| B-C: <0.0001 | |||||
| GMF | |||||
| all CD4+CTLA-4+ (CD4+CTLA-4+) Geo Mean | 2555 (2037–3405) | 3134 (2236–3547) | 6451 (4783–7990) | <0.0001 | A-C: <0.0001 |
| B-C: <0.0001 | |||||
| naïve CD4+ CTLA-4+ GMF (CD4+CD45RA+CD127+CTLA-4+) Geo Mean | 1523 (1269–2210) | 1751 (1270–2297) | 5702 (4651–7905) | <0.0001 | A-C: 0.0037 |
| B-C: 0.0008 | |||||
| memory CD4+ CTLA-4+ GMF (CD4+CD127+CD45RA-CTLA-4+) Geo Mean | 3059 (2400–3970) | 2801 (2221–3969) | 6730 (5082–8560) | <0.0001 | A-C: <0.0001 |
| B-C: <0.0001 | |||||
| activated CD4+ CTLA-4+ GMF (CD4+CD69+CD127-CD45RA-CTLA-4+) Geo Mean | 2580 (2208–3591) | 3093 (2230–3604) | 6744 (4848–8264) | <0.0001 | A-C: <0.0001 |
| B-C: <0.0001 | |||||
| activated memory CD4+ CTLA-4+ GMF (CD4+CD69+CD127+CD45RA-CTLA-4+) Geo Mean | 3058 (2529–4358) | 3347 (2569–4187) | 6934 (5145–8633) | <0.0001 | A-C: <0.0001 |
| B-C: <0.0001 | |||||
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kwiecien, I.; Skirecki, T.; Polubiec-Kownacka, M.; Raniszewska, A.; Domagala-Kulawik, J. Immunophenotype of T Cells Expressing Programmed Death-1 and Cytotoxic T Cell Antigen-4 in Early Lung Cancer: Local vs. Systemic Immune Response. Cancers 2019, 11, 567. https://doi.org/10.3390/cancers11040567
Kwiecien I, Skirecki T, Polubiec-Kownacka M, Raniszewska A, Domagala-Kulawik J. Immunophenotype of T Cells Expressing Programmed Death-1 and Cytotoxic T Cell Antigen-4 in Early Lung Cancer: Local vs. Systemic Immune Response. Cancers. 2019; 11(4):567. https://doi.org/10.3390/cancers11040567
Chicago/Turabian StyleKwiecien, Iwona, Tomasz Skirecki, Małgorzata Polubiec-Kownacka, Agata Raniszewska, and Joanna Domagala-Kulawik. 2019. "Immunophenotype of T Cells Expressing Programmed Death-1 and Cytotoxic T Cell Antigen-4 in Early Lung Cancer: Local vs. Systemic Immune Response" Cancers 11, no. 4: 567. https://doi.org/10.3390/cancers11040567
APA StyleKwiecien, I., Skirecki, T., Polubiec-Kownacka, M., Raniszewska, A., & Domagala-Kulawik, J. (2019). Immunophenotype of T Cells Expressing Programmed Death-1 and Cytotoxic T Cell Antigen-4 in Early Lung Cancer: Local vs. Systemic Immune Response. Cancers, 11(4), 567. https://doi.org/10.3390/cancers11040567








