Cancer Vaccines Co-Targeting HER2/Neu and IGF1R
Abstract
1. Introduction
2. Results
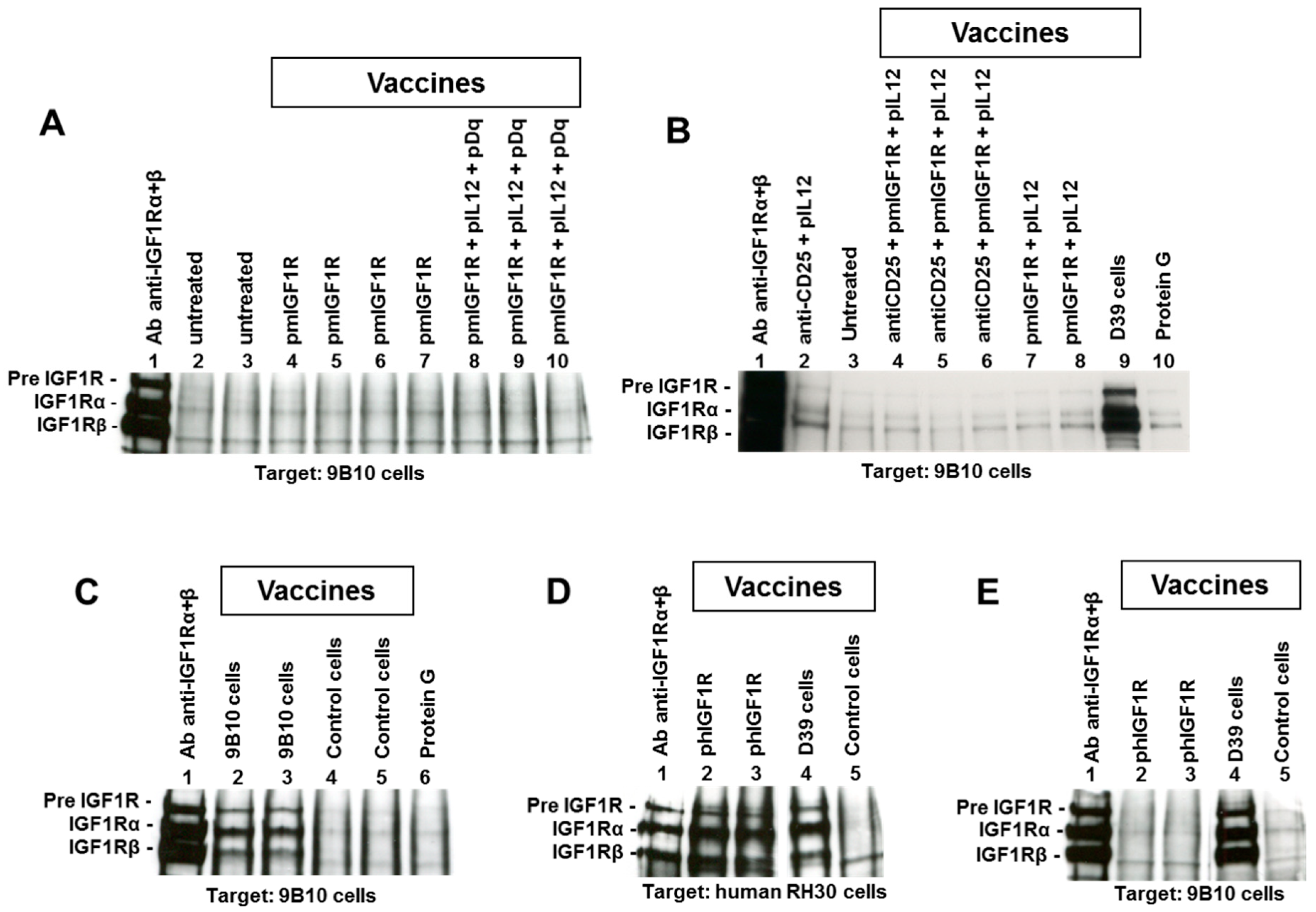
2.1. DNA Vaccines Against IGF1R
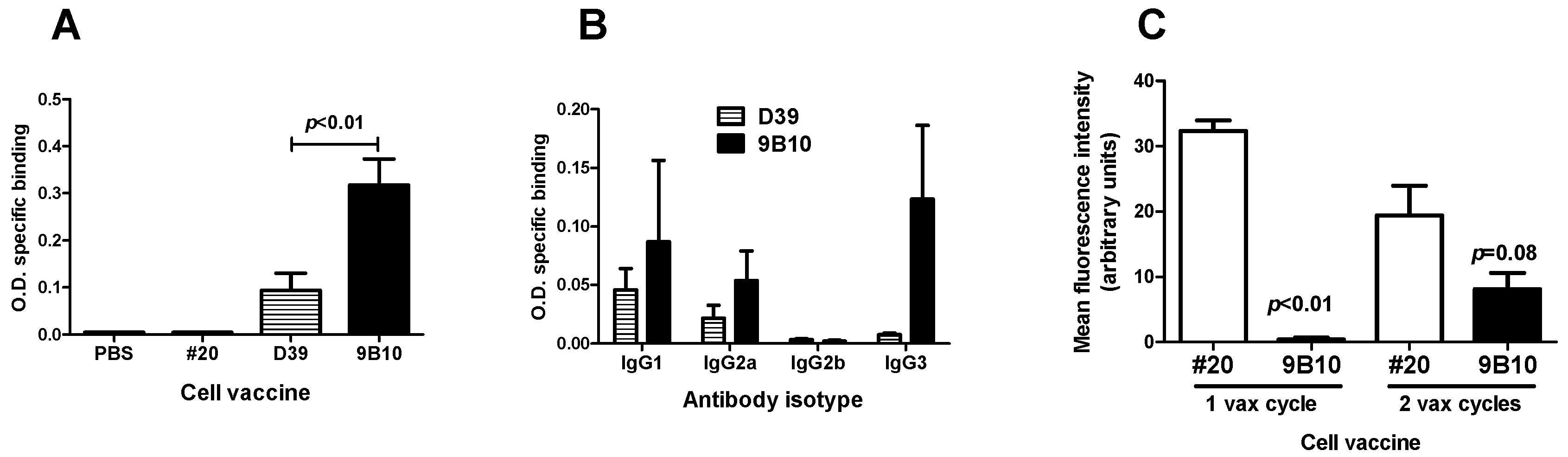
2.2. Cell Vaccines Co-Targeting HER2/neu and IGF1R
3. Discussion
4. Materials and Methods
4.1. Cells, Culture Conditions, and Reagents
4.2. Plasmids and Transfections
4.3. Ethics Statement
4.4. Mice
4.5. DNA and Cell Vaccinations
4.6. Immunoprecipitation/Western Blot and Elisa Assays
4.7. Indirect Immunofluorescence
4.8. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Weiner, L.M. Building better magic bullets-improving unconjugated monoclonal antibody therapy for cancer. Nat. Rev. Cancer 2007, 7, 701–706. [Google Scholar] [CrossRef]
- Lollini, P.L.; Cavallo, F.; Nanni, P.; Forni, G. Vaccines for tumour prevention. Nat. Rev. Cancer 2006, 6, 204–216. [Google Scholar] [CrossRef]
- Smit, M.A.; Jaffee, E.M.; Lutz, E.R. Cancer immunoprevention—The next frontier. Cancer Prev. Res. 2014, 7, 1072–1080. [Google Scholar] [CrossRef]
- Finn, O.J.; Beatty, P.L. Cancer immunoprevention. Curr. Opin. Immunol. 2016, 39, 52–58. [Google Scholar] [CrossRef]
- Lollini, P.L.; De Giovanni, C.; Nanni, P. Preclinical HER-2 Vaccines: From Rodent to Human HER-2. Front Oncol. 2013, 3, 151. [Google Scholar] [CrossRef]
- Nanni, P.; Nicoletti, G.; De Giovanni, C.; Landuzzi, L.; Di Carlo, E.; Cavallo, F.; Pupa, S.M.; Rossi, I.; Colombo, M.P.; Ricci, C.; et al. Combined allogeneic tumor cell vaccination and systemic interleukin 12 prevents mammary carcinogenesis in HER-2/neu transgenic mice. J. Exp. Med. 2001, 194, 1195–1205. [Google Scholar] [CrossRef] [PubMed]
- Nanni, P.; Landuzzi, L.; Nicoletti, G.; De Giovanni, C.; Rossi, I.; Croci, S.; Astolfi, A.; Iezzi, M.; Di Carlo, E.; Musiani, P.; et al. Immunoprevention of mammary carcinoma in HER-2/neu transgenic mice is IFN-gamma and B cell dependent. J. Immunol. 2004, 173, 2288–2296. [Google Scholar] [CrossRef] [PubMed]
- Benavides, L.C.; Gates, J.D.; Carmichael, M.G.; Patil, R.; Holmes, J.P.; Hueman, M.T.; Mittendorf, E.A.; Craig, D.; Stojadinovic, A.; Ponniah, S.; et al. The impact of HER2/neu expression level on response to the E75 vaccine: From U.S. Military Cancer Institute Clinical Trials Group Study I-01 and I-02. Clin. Cancer Res. 2009, 15, 2895–2904. [Google Scholar] [CrossRef] [PubMed]
- Mittendorf, E.A.; Clifton, G.T.; Holmes, J.P.; Schneble, E.; van Echo, D.; Ponniah, S.; Peoples, G.E. Final report of the phase I/II clinical trial of the E75 (nelipepimut-S) vaccine with booster inoculations to prevent disease recurrence in high-risk breast cancer patients. Ann. Oncol. 2014, 25, 1735–1742. [Google Scholar] [CrossRef]
- Diaz, C.M.; Chiappori, A.; Aurisicchio, L.; Bagchi, A.; Clark, J.; Dubey, S.; Fridman, A.; Fabregas, J.C.; Marshall, J.; Scarselli, E.; et al. Phase 1 studies of the safety and immunogenicity of electroporated HER2/CEA DNA vaccine followed by adenoviral boost immunization in patients with solid tumors. J. Transl. Med. 2013, 11, 62. [Google Scholar] [CrossRef] [PubMed]
- Sanabria-Figueroa, E.; Donnelly, S.M.; Foy, K.C.; Buss, M.C.; Castellino, R.C.; Paplomata, E.; Taliaferro-Smith, L.; Kaumaya, P.T.; Nahta, R. Insulin-like growth factor-1 receptor signaling increases the invasive potential of human epidermal growth factor receptor 2-overexpressing breast cancer cells via Src-focal adhesion kinase and forkhead box protein M1. Mol. Pharmacol. 2015, 87, 150–161. [Google Scholar] [CrossRef] [PubMed]
- Huang, F.; Greer, A.; Hurlburt, W.; Han, X.; Hafezi, R.; Wittenberg, G.M.; Reeves, K.; Chen, J.; Robinson, D.; Li, A.; et al. The mechanisms of differential sensitivity to an insulin-like growth factor-1 receptor inhibitor (BMS-536924) and rationale for combining with EGFR/HER2 inhibitors. Cancer Res. 2009, 69, 161–170. [Google Scholar] [CrossRef] [PubMed]
- Simpson, A.; Petnga, W.; Macaulay, V.M.; Weyer-Czernilofsky, U.; Bogenrieder, T. Insulin-Like Growth Factor (IGF) Pathway Targeting in Cancer: Role of the IGF Axis and Opportunities for Future Combination Studies. Target Oncol. 2017, 12, 571–597. [Google Scholar] [CrossRef] [PubMed]
- Kalebic, T.; Tsokos, M.; Helman, L.J. In vivo treatment with antibody against IGF-1 receptor suppresses growth of human rhabdomyosarcoma and down-regulates p34cdc2. Cancer Res. 1994, 54, 5531–5534. [Google Scholar]
- De Giovanni, C.; Melani, C.; Nanni, P.; Landuzzi, L.; Nicoletti, G.; Frabetti, F.; Griffoni, C.; Colombo, M.P.; Lollini, P.L. Redundancy of autocrine loops in human rhabdomyosarcoma cells: Induction of differentiation by suramin. Br. J. Cancer 1995, 72, 1224–1229. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Martins, A.S.; Olmos, D.; Missiaglia, E.; Shipley, J. Targeting the insulin-like growth factor pathway in rhabdomyosarcomas: Rationale and future perspectives. Sarcoma 2011, 2011, 209736. [Google Scholar] [CrossRef]
- Cecil, D.L.; Park, K.H.; Gad, E.; Childs, J.S.; Higgins, D.M.; Plymate, S.R.; Disis, M.L. T-helper I immunity, specific for the breast cancer antigen insulin-like growth factor-I receptor (IGF-IR), is associated with increased adiposity. Breast Cancer Res. Treat. 2013, 139, 657–665. [Google Scholar] [CrossRef] [PubMed]
- Pappo, A.S.; Vassal, G.; Crowley, J.J.; Bolejack, V.; Hogendoorn, P.C.; Chugh, R.; Ladanyi, M.; Grippo, J.F.; Dall, G.; Staddon, A.P.; et al. A phase 2 trial of R1507, a monoclonal antibody to the insulin-like growth factor-1 receptor (IGF-1R), in patients with recurrent or refractory rhabdomyosarcoma, osteosarcoma, synovial sarcoma, and other soft tissue sarcomas: Results of a Sarcoma Alliance for Research Through Collaboration study. Cancer 2014, 120, 2448–2456. [Google Scholar]
- Wan, X.; Yeung, C.; Heske, C.; Mendoza, A.; Helman, L.J. IGF-1R Inhibition Activates a YES/SFK Bypass Resistance Pathway: Rational Basis for Co-Targeting IGF-1R and Yes/SFK Kinase in Rhabdomyosarcoma. Neoplasia 2015, 17, 358–366. [Google Scholar] [CrossRef]
- Chakraborty, A.; Hatzis, C.; DiGiovanna, M.P. Co-targeting the HER and IGF/insulin receptor axis in breast cancer, with triple targeting with endocrine therapy for hormone-sensitive disease. Breast Cancer Res. Treat. 2017, 163, 37–50. [Google Scholar] [CrossRef]
- Disis, M.L.; Gad, E.; Herendeen, D.R.; Lai, V.P.; Park, K.H.; Cecil, D.L.; O’Meara, M.M.; Treuting, P.M.; Lubet, R.A. A multiantigen vaccine targeting neu, IGFBP-2, and IGF-IR prevents tumor progression in mice with preinvasive breast disease. Cancer Prev. Res. 2013, 6, 1273–1282. [Google Scholar] [CrossRef] [PubMed]
- Nanni, P.; Nicoletti, G.; De Giovanni, C.; Croci, S.; Astolfi, A.; Landuzzi, L.; Di Carlo, E.; Iezzi, M.; Musiani, P.; Lollini, P.L. Development of rhabdomyosarcoma in HER-2/neu transgenic p53 mutant mice. Cancer Res. 2003, 63, 2728–2732. [Google Scholar]
- De Giovanni, C.; Landuzzi, L.; Nicoletti, G.; Lollini, P.L.; Nanni, P. Molecular and cellular biology of rhabdomyosarcoma. Future Oncol. 2009, 5, 1449–1475. [Google Scholar] [CrossRef] [PubMed]
- De Giovanni, C.; Nanni, P.; Landuzzi, L.; Ianzano, M.L.; Nicoletti, G.; Croci, S.; Palladini, A.; Lollini, P.L. Immune targeting of autocrine IGF2 hampers rhabdomyosarcoma growth and metastasis. BMC Cancer 2019, 19, 126. [Google Scholar] [CrossRef] [PubMed]
- De Giovanni, C.; Nicoletti, G.; Palladini, A.; Croci, S.; Landuzzi, L.; Antognoli, A.; Murgo, A.; Astolfi, A.; Ferrini, S.; Fabbi, M.; et al. A multi-DNA preventive vaccine for p53/Neu-driven cancer syndrome. Hum. Gene Ther. 2009, 20, 453–464. [Google Scholar] [CrossRef] [PubMed]
- Stevenson, F.K.; Ottensmeier, C.H.; Rice, J. DNA vaccines against cancer come of age. Curr. Opin. Immunol. 2010, 22, 264–270. [Google Scholar] [CrossRef] [PubMed]
- Yang, B.; Jeang, J.; Yang, A.; Wu, T.C.; Hung, C.F. DNA vaccine for cancer immunotherapy. Hum. Vaccines Immunother. 2014, 10, 3153–3164. [Google Scholar] [CrossRef]
- Croci, S.; Nanni, P.; Palladini, A.; Nicoletti, G.; Grosso, V.; Benegiamo, G.; Landuzzi, L.; Lamolinara, A.; Ianzano, M.L.; Ranieri, D.; et al. Interleukin-15 is required for immunosurveillance and immunoprevention of HER2/neu-driven mammary carcinogenesis. Breast Cancer Res. 2015, 17, 70. [Google Scholar] [CrossRef] [PubMed]
- Ianzano, M.L.; Croci, S.; Nicoletti, G.; Palladini, A.; Landuzzi, L.; Grosso, V.; Ranieri, D.; Dall’Ora, M.; Santeramo, I.; Urbini, M.; et al. Tumor suppressor genes promote rhabdomyosarcoma progression in p53 heterozygous, HER-2/neu transgenic mice. Oncotarget 2014, 5, 108–119. [Google Scholar] [CrossRef] [PubMed]
- Croci, S.; Nicoletti, G.; Landuzzi, L.; De Giovanni, C.; Astolfi, A.; Marini, C.; Di Carlo, E.; Musiani, P.; Forni, G.; Nanni, P.; et al. Immunological prevention of a multigene cancer syndrome. Cancer Res. 2004, 64, 8428–8434. [Google Scholar] [CrossRef] [PubMed]
- Palladini, A.; Nicoletti, G.; Pappalardo, F.; Murgo, A.; Grosso, V.; Stivani, V.; Ianzano, M.L.; Antognoli, A.; Croci, S.; Landuzzi, L.; et al. In silico modeling and in vivo efficacy of cancer-preventive vaccinations. Cancer Res. 2010, 70, 7755–7763. [Google Scholar] [CrossRef] [PubMed]
- Nanni, P.; Nicoletti, G.; Palladini, A.; Croci, S.; Murgo, A.; Antognoli, A.; Landuzzi, L.; Fabbi, M.; Ferrini, S.; Musiani, P.; et al. Antimetastatic activity of a preventive cancer vaccine. Cancer Res. 2007, 67, 11037–11044. [Google Scholar] [CrossRef] [PubMed]
- Quaglino, E.; Mastini, C.; Amici, A.; Marchini, C.; Iezzi, M.; Lanzardo, S.; De Giovanni, C.; Montani, M.; Lollini, P.L.; Masucci, G.; et al. A better immune reaction to Erbb-2 tumors is elicited in mice by DNA vaccines encoding rat/human chimeric proteins. Cancer Res. 2010, 70, 2604–2612. [Google Scholar] [CrossRef] [PubMed]
- Denies, S.; Cicchelero, L.; Polis, I.; Sanders, N.N. Immunogenicity and safety of xenogeneic vascular endothelial growth factor receptor-2 DNA vaccination in mice and dogs. Oncotarget 2016, 7, 10905–10916. [Google Scholar] [CrossRef] [PubMed]
- Geenen, V. The appearance of the thymus and the integrated evolution of adaptive immune and neuroendocrine systems. Acta Clin. Belg. 2012, 67, 209–213. [Google Scholar]
- Niethammer, A.G.; Xiang, R.; Becker, J.C.; Wodrich, H.; Pertl, U.; Karsten, G.; Eliceiri, B.P.; Reisfeld, R.A. A DNA vaccine against VEGF receptor 2 prevents effective angiogenesis and inhibits tumor growth. Nat. Med. 2002, 8, 1369–1375. [Google Scholar] [CrossRef] [PubMed]
- Balañá, M.E.; Labriola, L.; Salatino, M.; Movsichoff, F.; Peters, G.; Charreau, E.H.; Elizalde, P.V. Activation of ErbB-2 via a hierarchical interaction between ErbB-2 and type I insulin-like growth factor receptor in mammary tumor cells. Oncogene 2001, 20, 34–47. [Google Scholar]
- De Giovanni, C.; Nicoletti, G.; Landuzzi, L.; Romani, F.; Croci, S.; Palladini, A.; Murgo, A.; Antognoli, A.; Ianzano, M.L.; Stivani, V.; et al. Human responses against HER-2-positive cancer cells in human immune system-engrafted mice. Br. J. Cancer 2012, 107, 1302–1309. [Google Scholar] [CrossRef] [PubMed][Green Version]
- De Giovanni, C.; Nicoletti, G.; Landuzzi, L.; Astolfi, A.; Croci, S.; Comes, A.; Ferrini, S.; Meazza, R.; Iezzi, M.; Di Carlo, E.; et al. Immunoprevention of HER-2/neu transgenic mammary carcinoma through an interleukin 12-engineered allogeneic cell vaccine. Cancer Res. 2004, 64, 4001–4009. [Google Scholar] [CrossRef] [PubMed]
- Croci, S.; Landuzzi, L.; Astolfi, A.; Nicoletti, G.; Rosolen, A.; Sartori, F.; Follo, M.Y.; Oliver, N.; De Giovanni, C.; Nanni, P.; et al. Inhibition of connective tissue growth factor (CTGF/CCN2) expression decreases the survival and myogenic differentiation of human rhabdomyosarcoma cells. Cancer Res. 2004, 64, 1730–1736. [Google Scholar] [CrossRef]
- Sell, C.; Dumenil, G.; Deveaud, C.; Miura, M.; Coppola, D.; De Angelis, T.; Rubin, R.; Efstratiadis, A.; Baserga, R. Effect of a null mutation of the insulin-like growth factor I receptor gene on growth and transformation of mouse embryo fibroblasts. Mol. Cell Biol. 1994, 14, 3604–3612. [Google Scholar] [CrossRef] [PubMed]
- Peretz, S.; Jensen, R.; Baserga, R.; Glazer, P.M. ATM-dependent expression of the insulin-like growth factor-I receptor in a pathway regulating radiation response. Proc. Natl. Acad. Sci. USA 2001, 98, 1676–1681. [Google Scholar] [CrossRef] [PubMed]
- De Giovanni, C.; Nicoletti, G.; Quaglino, E.; Landuzzi, L.; Palladini, A.; Ianzano, M.L.; Dall’Ora, M.; Grosso, V.; Ranieri, D.; Laranga, R.; et al. Vaccines against human HER2 prevent mammary carcinoma in mice transgenic for human HER2. Breast Cancer Res. 2014, 16, R10. [Google Scholar] [CrossRef] [PubMed]
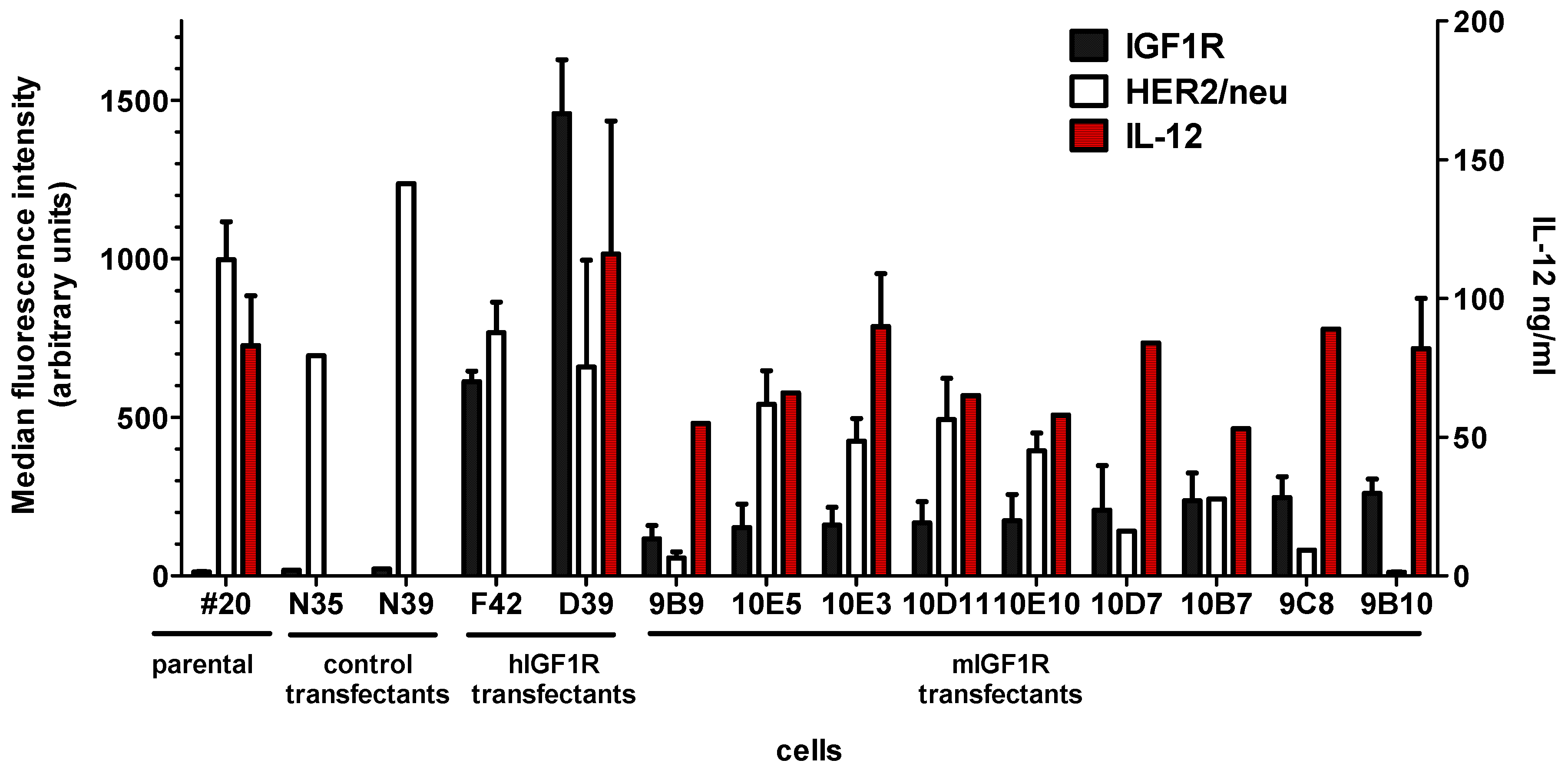

| Cells | Vector | Resistance Gene | IGF1R Gene | IGF1R Membrane Expression (mfi) | HER2/neu Membrane Expression (mfi) | IL12 in 72 h Culture (106 Cells Seeded) ng/mL |
|---|---|---|---|---|---|---|
| #20 | - | - | - | 12 ± 2 | 997 ± 120 | 88 ± 29 |
| #20/N | pcDNA3.1 | neo | - | 10 ± 4 | 966 ± 271 | nd |
| D39 | pCVNIGF1R | neo | Human | 1458 ± 170 | 659 ± 337 | 116 ± 48 |
| 9B10 | pmIGF1Ropt | neo | Murine (optimized) | 260 ± 45 | 11 ± 2 | 83 ± 18 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
De Giovanni, C.; Landuzzi, L.; Palladini, A.; Ianzano, M.L.; Nicoletti, G.; Ruzzi, F.; Amici, A.; Croci, S.; Nanni, P.; Lollini, P.-L. Cancer Vaccines Co-Targeting HER2/Neu and IGF1R. Cancers 2019, 11, 517. https://doi.org/10.3390/cancers11040517
De Giovanni C, Landuzzi L, Palladini A, Ianzano ML, Nicoletti G, Ruzzi F, Amici A, Croci S, Nanni P, Lollini P-L. Cancer Vaccines Co-Targeting HER2/Neu and IGF1R. Cancers. 2019; 11(4):517. https://doi.org/10.3390/cancers11040517
Chicago/Turabian StyleDe Giovanni, Carla, Lorena Landuzzi, Arianna Palladini, Marianna Lucia Ianzano, Giordano Nicoletti, Francesca Ruzzi, Augusto Amici, Stefania Croci, Patrizia Nanni, and Pier-Luigi Lollini. 2019. "Cancer Vaccines Co-Targeting HER2/Neu and IGF1R" Cancers 11, no. 4: 517. https://doi.org/10.3390/cancers11040517
APA StyleDe Giovanni, C., Landuzzi, L., Palladini, A., Ianzano, M. L., Nicoletti, G., Ruzzi, F., Amici, A., Croci, S., Nanni, P., & Lollini, P.-L. (2019). Cancer Vaccines Co-Targeting HER2/Neu and IGF1R. Cancers, 11(4), 517. https://doi.org/10.3390/cancers11040517







