Targeting the Tetraspanins with Monoclonal Antibodies in Oncology: Focus on Tspan8/Co-029
Abstract
:1. Introduction
2. Mechanisms and Requirements for the Use of Therapeutic Anti-Tetraspanin Antibodies
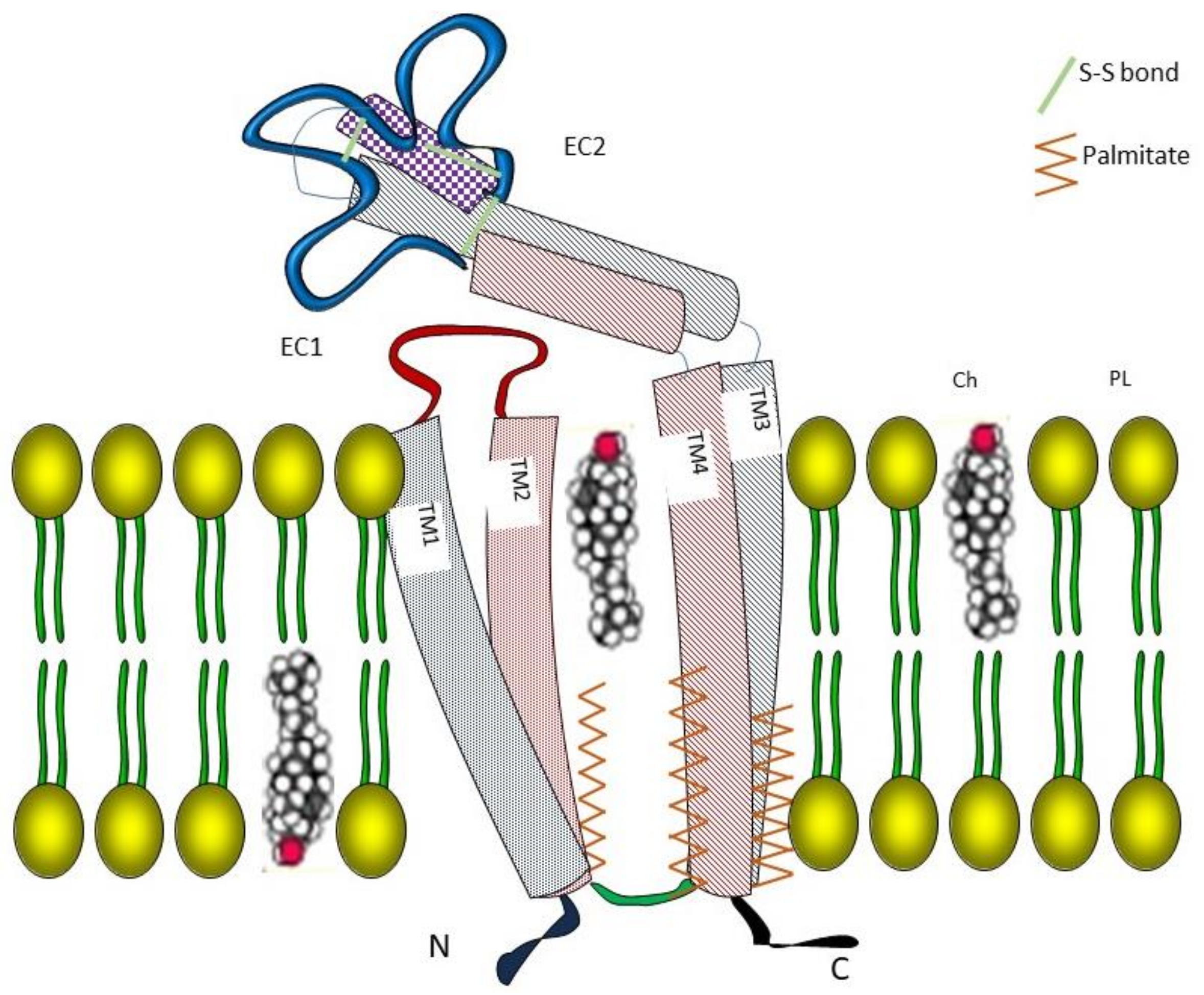
2.1. Molecular Mechanisms
2.1.1. Blocking Antibodies
2.1.2. Cytotoxic Antibodies
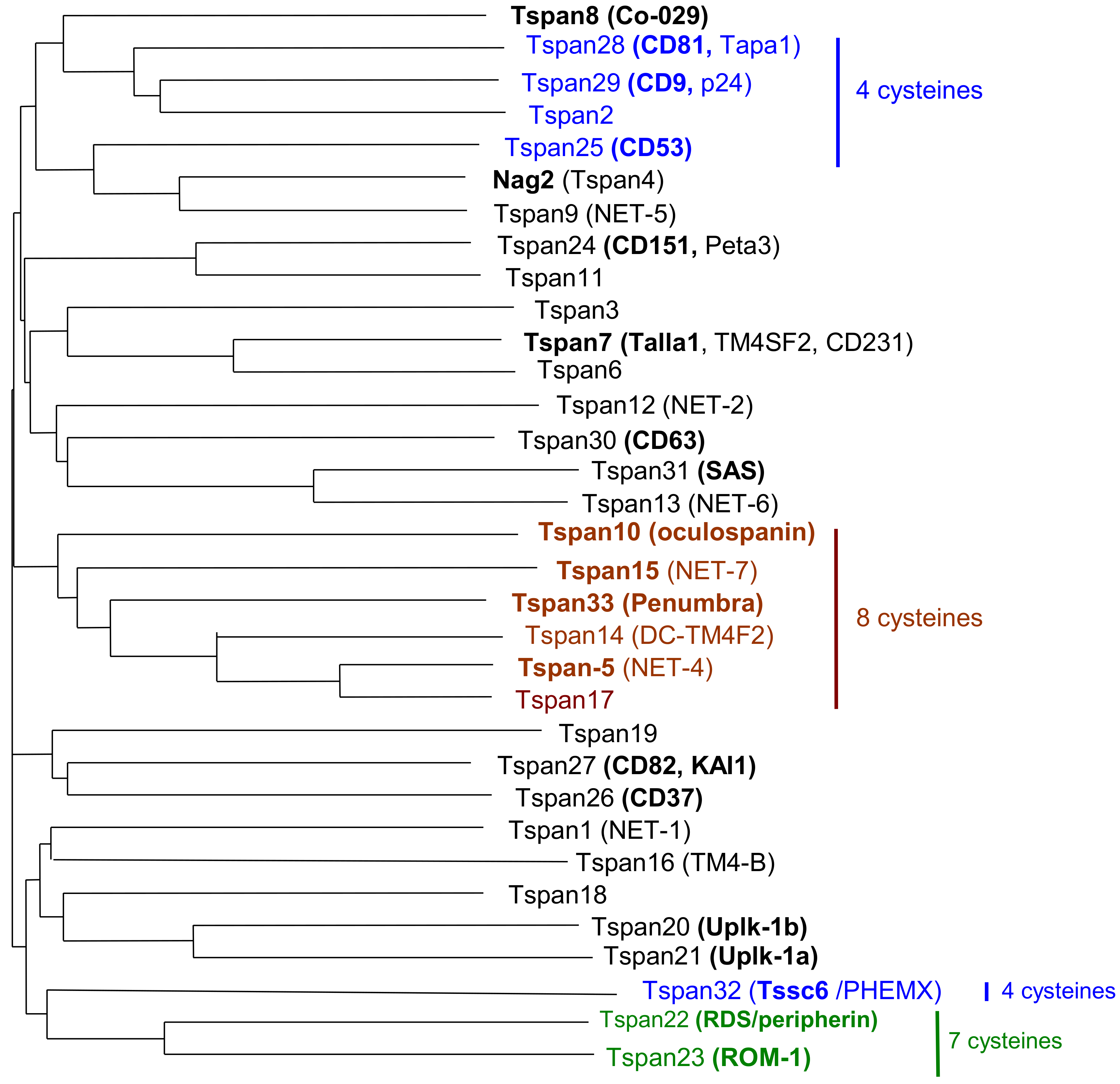
2.2. Pattern of Expression
2.3. Dodging Immune Neutralization
3. Therapeutic Antibodies Directed toward the Tetraspanins CD9, CD151 and CD37 in Cancer
4. Treatment with Unconjugated Anti-Tspan8 Antibodies
5. Treatment with Radionuclide-Conjugated Anti-Tspan8 Antibodies
5.1. Radiolabeling of Antibodies
5.2. Targeting Tspan8 with Radiolabeled Antibodies
5.3. Pros and Cons of RIT for Human Cancers: Focus on Targeting Tspan8
6. Conclusions
Funding
Conflicts of Interest
References
- Boucheix, C.; Rubinstein, E. Tetraspanins. Cell. Mol. Life Sci. 2001, 58, 1189–1205. [Google Scholar] [CrossRef] [PubMed]
- Levy, S.; Shoham, T. The tetraspanin web modulates immune-signalling complexes. Nat. Rev. Immunol. 2005, 5, 136–148. [Google Scholar] [CrossRef] [PubMed]
- Hemler, M.E. Tetraspanin proteins promote multiple cancer stages. Nat. Rev. Cancer 2014, 14, 49–60. [Google Scholar] [CrossRef] [PubMed]
- Charrin, S.; Jouannet, S.; Boucheix, C.; Rubinstein, E. Tetraspanins at a glance. J. Cell Sci. 2014, 127, 3641–3648. [Google Scholar] [CrossRef]
- Masse, I.; Agaësse, G.; Berthier-Vergnes, O. Tetraspanins in cutaneous physiopathology. Med. Sci. (Paris) 2016, 32, 267–273. (In French) [Google Scholar] [CrossRef] [PubMed]
- Van Deventer, S.J.; Dunlock, V.-M.E.; van Spriel, A.B. Molecular interactions shaping the tetraspanin web. Biochem. Soc. Trans. 2017, 45, 741–750. [Google Scholar] [CrossRef] [PubMed]
- Abache, T.; Le Naour, F.; Planchon, S.; Harper, F.; Boucheix, C.; Rubinstein, E. The transferrin receptor and the tetraspanin web molecules CD9, CD81 and CD9P-1 are differentially sorted into exosomes after TPA treatment of K562 cells. J. Cell. Biochem. 2007, 102, 650–664. [Google Scholar] [CrossRef]
- Wang, H.-X.; Sharma, C.; Knoblich, K.; Granter, S.R.; Hemler, M.E. EWI-2 negatively regulates TGF-β signaling leading to altered melanoma growth and metastasis. Cell Res. 2015, 25, 370–385. [Google Scholar] [CrossRef]
- Lammerding, J.; Kazarov, A.R.; Huang, H.; Lee, R.T.; Hemler, M.E. Tetraspanin CD151 regulates alpha6beta1 integrin adhesion strengthening. Proc. Natl. Acad. Sci. USA 2003, 100, 7616–7621. [Google Scholar] [CrossRef]
- Shoham, T.; Rajapaksa, R.; Boucheix, C.; Rubinstein, E.; Poe, J.C.; Tedder, T.F.; Levy, S. The tetraspanin CD81 regulates the expression of CD19 during B cell development in a postendoplasmic reticulum compartment. J. Immunol. 2003, 171, 4062–4072. [Google Scholar] [CrossRef]
- Saint-Pol, J.; Eschenbrenner, E.; Dornier, E.; Boucheix, C.; Charrin, S.; Rubinstein, E. Regulation of the trafficking and the function of the metalloprotease ADAM10 by tetraspanins. Biochem. Soc. Trans. 2017, 45, 937–944. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Y.; Ailane, N.; Sala-Valdés, M.; Haghighi-Rad, F.; Billard, M.; Nguyen, V.; Saffroy, R.; Lemoine, A.; Rubinstein, E.; Boucheix, C.; et al. Multi-factorial modulation of colorectal carcinoma cells motility—Partial coordination by the tetraspanin Co-029/Tspan8. Oncotarget 2017, 8, 27454–27470. [Google Scholar] [CrossRef] [PubMed]
- Zimmerman, B.; Kelly, B.; McMillan, B.J.; Seegar, T.C.M.; Dror, R.O.; Kruse, A.C.; Blacklow, S.C. Crystal structure of a full-length human Tetraspanin reveals a cholesterol-binding pocket. Cell 2016, 167, 1041–1051. [Google Scholar] [CrossRef] [PubMed]
- Zöller, M. Tetraspanins: Push and pull in suppressing and promoting metastasis. Nat. Rev. Cancer 2009, 9, 40–55. [Google Scholar] [CrossRef] [PubMed]
- Charrin, S.; le Naour, F.; Silvie, O.; Milhiet, P.-E.; Boucheix, C.; Rubinstein, E. Lateral organization of membrane proteins: Tetraspanins spin their web. Biochem. J. 2009, 420, 133–154. [Google Scholar] [CrossRef] [PubMed]
- Ashman, L.K.; Zoller, M. Tetraspanins in Cancer. In Tetraspanins; Berditchevski, F., Rubinstein, E., Eds.; Springer: Heidelberg, Germany, 2013; pp. 257–298. [Google Scholar]
- De Winde, C.M.; Veenbergen, S.; Young, K.H.; Xu-Monette, Z.Y.; Wang, X.-X.; Xia, Y.; Jabbar, K.J.; van den Brand, M.; van der Schaaf, A.; Elfrink, S.; et al. Tetraspanin CD37 protects against the development of B cell lymphoma. J. Clin. Investig. 2016, 126, 653–666. [Google Scholar] [CrossRef] [PubMed]
- Andreu, Z.; Yáñez-Mó, M. Tetraspanins in extracellular vesicle formation and function. Front. Immunol. 2014, 5, 442. [Google Scholar] [CrossRef] [PubMed]
- Greco, C.; Bralet, M.-P.; Ailane, N.; Dubart-Kupperschmitt, A.; Rubinstein, E.; Le Naour, F.; Boucheix, C. E-cadherin/p120-catenin and tetraspanin Co-029 cooperate for cell motility control in human colon carcinoma. Cancer Res. 2010, 70, 7674–7683. [Google Scholar] [CrossRef]
- Claas, C.; Seiter, S.; Claas, A.; Savelyeva, L.; Schwab, M.; Zöller, M. Association between the rat homologue of CO-029, a metastasis-associated tetraspanin molecule and consumption coagulopathy. J. Cell Biol. 1998, 141, 267–280. [Google Scholar] [CrossRef]
- Gesierich, S.; Berezovskiy, I.; Ryschich, E.; Zöller, M. Systemic induction of the angiogenesis switch by the tetraspanin D6.1A/CO-029. Cancer Res. 2006, 66, 7083–7094. [Google Scholar] [CrossRef]
- Yue, S.; Mu, W.; Erb, U.; Zöller, M. The tetraspanins CD151 and Tspan8 are essential exosome components for the crosstalk between cancer initiating cells and their surrounding. Oncotarget 2015, 6, 2366–2384. [Google Scholar] [CrossRef] [PubMed]
- Maisonial-Besset, A.; Witkowski, T.; Navarro-Teulon, I.; Berthier-Vergnes, O.; Fois, G.; Zhu, Y.; Besse, S.; Bawa, O.; Briat, A.; Quintana, M.; et al. Tetraspanin 8 (TSPAN 8) as a potential target for radio-immunotherapy of colorectal cancer. Oncotarget 2017, 8, 22034–22047. [Google Scholar] [CrossRef] [PubMed]
- Ailane, N.; Greco, C.; Zhu, Y.; Sala-Valdés, M.; Billard, M.; Casal, I.; Bawa, O.; Opolon, P.; Rubinstein, E.; Boucheix, C. Effect of an anti-human Co-029/Tspan8 mouse monoclonal antibody on tumor growth in a nude mouse model. Front. Physiol. 2014, 5. [Google Scholar] [CrossRef] [PubMed]
- Park, C.S.; Kim, T.-K.; Kim, H.G.; Kim, Y.-J.; Jeoung, M.H.; Lee, W.R.; Go, N.K.; Heo, K.; Lee, S. Therapeutic targeting of tetraspanin8 in epithelial ovarian cancer invasion and metastasis. Oncogene 2016, 35, 4540–4548. [Google Scholar] [CrossRef] [PubMed]
- Hemler, M.E. Targeting of tetraspanin proteins—Potential benefits and strategies. Nat. Rev. Drug Discov. 2008, 7, 747–758. [Google Scholar] [CrossRef] [PubMed]
- Maeda, H.; Nakamura, H.; Fang, J. The EPR effect for macromolecular drug delivery to solid tumors: Improvement of tumor uptake, lowering of systemic toxicity and distinct tumor imaging in vivo. Adv. Drug Deliv. Rev. 2013, 65, 71–79. [Google Scholar] [CrossRef] [PubMed]
- Matsumura, Y.; Maeda, H. A new concept for macromolecular therapeutics in cancer chemotherapy: Mechanism of tumoritropic accumulation of proteins and the antitumor agent smancs. Cancer Res. 1986, 46, 6387–6392. [Google Scholar]
- Martin, C.; Kizlik-Masson, C.; Pèlegrin, A.; Watier, H.; Viaud-Massuard, M.-C.; Joubert, N. Antibody-drug conjugates: Design and development for therapy and imaging in and beyond cancer, LabEx MAbImprove industrial workshop, July 27–28, 2017, Tours, France. MAbs 2018, 10, 210–221. [Google Scholar] [CrossRef]
- Lin, J.; Sagert, J. Targeting Drug Conjugates. In Innovations for Next-Generation Antibody-Drug Conjugates; Cancer Drug Discovery and Development; Humana Press: New York, NY, USA, 2018; pp. 281–298. [Google Scholar]
- Berthier-Vergnes, O.; Kharbili, M.E.; de la Fouchardière, A.; Pointecouteau, T.; Verrando, P.; Wierinckx, A.; Lachuer, J.; Le Naour, F.; Lamartine, J. Gene expression profiles of human melanoma cells with different invasive potential reveal TSPAN8 as a novel mediator of invasion. Br. J. Cancer 2011, 104, 155–165. [Google Scholar] [CrossRef]
- Kanetaka, K.; Sakamoto, M.; Yamamoto, Y.; Yamasaki, S.; Lanza, F.; Kanematsu, T.; Hirohashi, S. Overexpression of tetraspanin CO-029 in hepatocellular carcinoma. J. Hepatol. 2001, 35, 637–642. [Google Scholar] [CrossRef]
- Gesierich, S.; Paret, C.; Hildebrand, D.; Weitz, J.; Zgraggen, K.; Schmitz-Winnenthal, F.H.; Horejsi, V.; Yoshie, O.; Herlyn, D.; Ashman, L.K.; et al. Colocalization of the tetraspanins, CO-029 and CD151, with integrins in human pancreatic adenocarcinoma: Impact on cell motility. Clin. Cancer Res. 2005, 11, 2840–2852. [Google Scholar] [CrossRef] [PubMed]
- Ahmadzadeh, V.; Farajnia, S.; Feizi, M.A.H.; Nejad, R.A.K. Antibody humanization methods for development of therapeutic applications. Monoclon. Antib. Immunodiagn. Immunother. 2014, 33, 67–73. [Google Scholar] [CrossRef] [PubMed]
- Vences-Catalán, F.; Levy, S. Immune Targeting of Tetraspanins Involved in Cell Invasion and Metastasis. Front. Immunol. 2018, 9, 1277. [Google Scholar] [CrossRef] [PubMed]
- Nakamoto, T.; Murayama, Y.; Oritani, K.; Boucheix, C.; Rubinstein, E.; Nishida, M.; Katsube, F.; Watabe, K.; Kiso, S.; Tsutsui, S.; et al. A novel therapeutic strategy with anti-CD9 antibody in gastric cancers. J. Gastroenterol. 2009, 44, 889–896. [Google Scholar] [CrossRef] [PubMed]
- Worthington, R.E.; Carroll, R.C.; Boucheix, C. Platelet activation by CD9 monoclonal antibodies is mediated by the Fc gamma II receptor. Br. J. Haematol. 1990, 74, 216–222. [Google Scholar] [CrossRef] [PubMed]
- Uckun, F.M.; Kersey, J.H.; Haake, R.; Weisdorf, D.; Ramsay, N.K. Autologous bone marrow transplantation in high-risk remission B-lineage acute lymphoblastic leukemia using a cocktail of three monoclonal antibodies (BA-1/CD24, BA-2/CD9 and BA-3/CD10) plus complement and 4-hydroperoxycyclophosphamide for ex vivo bone marrow purging. Blood 1992, 79, 1094–1104. [Google Scholar] [PubMed]
- Press, O.W.; Eary, J.F.; Appelbaum, F.R.; Martin, P.J.; Badger, C.C.; Nelp, W.B.; Glenn, S.; Butchko, G.; Fisher, D.; Porter, B. Radiolabeled-antibody therapy of B-cell lymphoma with autologous bone marrow support. N. Engl. J. Med. 1993, 329, 1219–1224. [Google Scholar] [CrossRef]
- Witkowska, M.; Smolewski, P.; Robak, T. Investigational therapies targeting CD37 for the treatment of B-cell lymphoid malignancies. Expert Opin. Investig. Drugs 2018, 27, 171–177. [Google Scholar] [CrossRef]
- Palmer, T.D.; Martínez, C.H.; Vasquez, C.; Hebron, K.E.; Jones-Paris, C.; Arnold, S.A.; Chan, S.M.; Chalasani, V.; Gomez-Lemus, J.A.; Williams, A.K.; et al. Integrin-free tetraspanin CD151 can inhibit tumor cell motility upon clustering and is a clinical indicator of prostate cancer progression. Cancer Res. 2014, 74, 173–187. [Google Scholar] [CrossRef]
- Haeuw, J.-F.; Goetsch, L.; Bailly, C.; Corvaia, N. Tetraspanin CD151 as a target for antibody-based cancer immunotherapy. Biochem. Soc. Trans. 2011, 39, 553–558. [Google Scholar] [CrossRef]
- Larson, S.M.; Carrasquillo, J.A.; Cheung, N.-K.V.; Press, O.W. Radioimmunotherapy of human tumours. Nat. Rev. Cancer 2015, 15, 347–360. [Google Scholar] [CrossRef] [PubMed]
- Ferrero, S.; Pastore, A.; Scholz, C.W.; Forstpointner, R.; Pezzutto, A.; Bergmann, L.; Trümper, L.; Finke, J.; Keller, U.; Ghione, P.; et al. Radioimmunotherapy in relapsed/refractory mantle cell lymphoma patients: Final results of a European MCL Network Phase II Trial. Leukemia 2016, 30, 984–987. [Google Scholar] [CrossRef] [PubMed]
- Navarro-Teulon, I.; Lozza, C.; Pèlegrin, A.; Vivès, E.; Pouget, J.-P. General overview of radioimmunotherapy of solid tumors. Immunotherapy 2013, 5, 467–487. [Google Scholar] [CrossRef] [PubMed]
- Makvandi, M.; Dupis, E.; Engle, J.W.; Nortier, F.M.; Fassbender, M.E.; Simon, S.; Birnbaum, E.R.; Atcher, R.W.; John, K.D.; Rixe, O.; et al. Alpha-Emitters and Targeted Alpha Therapy in Oncology: From Basic Science to Clinical Investigations. Target Oncol. 2018, 13, 189–203. [Google Scholar] [CrossRef] [PubMed]
- Allen, B.J.; Singla, A.A.; Rizvi, S.M.A.; Graham, P.; Bruchertseifer, F.; Apostolidis, C.; Morgenstern, A. Analysis of patient survival in a Phase I trial of systemic targeted α-therapy for metastatic melanoma. Immunotherapy 2011, 3, 1041–1050. [Google Scholar] [CrossRef] [PubMed]
- Rosenblat, T.L.; McDevitt, M.R.; Mulford, D.A.; Pandit-Taskar, N.; Divgi, C.R.; Panageas, K.S.; Heaney, M.L.; Chanel, S.; Morgenstern, A.; Sgouros, G.; et al. Sequential cytarabine and alpha-particle immunotherapy with bismuth-213-lintuzumab (HuM195) for acute myeloid leukemia. Clin. Cancer Res. 2010, 16, 5303–5311. [Google Scholar] [CrossRef] [PubMed]
- Kraeber-Bodéré, F.; Rousseau, C.; Bodet-Milin, C.; Frampas, E.; Faivre-Chauvet, A.; Rauscher, A.; Sharkey, R.M.; Goldenberg, D.M.; Chatal, J.-F.; Barbet, J. A pretargeting system for tumor PET imaging and radioimmunotherapy. Front. Pharmacol. 2015, 6, 54. [Google Scholar]
- Van de Watering, F.C.J.; Rijpkema, M.; Robillard, M.; Oyen, W.J.G.; Boerman, O.C. Pretargeted imaging and radioimmunotherapy of cancer using antibodies and bioorthogonal chemistry. Front. Med. (Lausanne) 2014, 1, 44. [Google Scholar] [CrossRef]
- Venkatanarasimha, N.; Gogna, A.; Tong, K.T.A.; Damodharan, K.; Chow, P.K.H.; Lo, R.H.G.; Chandramohan, S. Radioembolisation of hepatocellular carcinoma: A primer. Clin. Radiol. 2017, 72, 1002–1013. [Google Scholar] [CrossRef]
- Pruszynski, M.; D’Huyvetter, M.; Bruchertseifer, F.; Morgenstern, A.; Lahoutte, T. Evaluation of an Anti-HER2 Nanobody Labeled with 225Ac for Targeted α-Particle Therapy of Cancer. Mol. Pharm. 2018, 15, 1457–1466. [Google Scholar] [CrossRef]
- Sörensen, J.; Sandberg, D.; Sandström, M.; Wennborg, A.; Feldwisch, J.; Tolmachev, V.; Åström, G.; Lubberink, M.; Garske-Román, U.; Carlsson, J.; et al. First-in-human molecular imaging of HER2 expression in breast cancer metastases using the 111In-ABY-025 affibody molecule. J. Nucl. Med. 2014, 55, 730–735. [Google Scholar] [CrossRef] [PubMed]
- Rondon, A.; Ty, N.; Bequignat, J.-B.; Quintana, M.; Briat, A.; Witkowski, T.; Bouchon, B.; Boucheix, C.; Miot-Noirault, E.; Pouget, J.-P.; et al. Antibody PEGylation in bioorthogonal pretargeting with trans-cyclooctene/tetrazine cycloaddition: In vitro and in vivo evaluation in colorectal cancer models. Sci. Rep. 2017, 7, 14918. [Google Scholar] [CrossRef] [PubMed]
- Houghton, J.L.; Membreno, R.; Abdel-Atti, D.; Cunanan, K.M.; Carlin, S.; Scholz, W.W.; Zanzonico, P.B.; Lewis, J.S.; Zeglis, B.M. Establishment of the In Vivo Efficacy of Pretargeted Radioimmunotherapy Utilizing Inverse Electron Demand Diels-Alder Click Chemistry. Mol. Cancer Ther. 2017, 16, 124–133. [Google Scholar] [CrossRef] [PubMed]
- Heiler, S.; Wang, Z.; Zöller, M. Pancreatic cancer stem cell markers and exosomes—The incentive push. World J. Gastroenterol. 2016, 22, 5971–6007. [Google Scholar] [CrossRef] [PubMed]
- Norain, A.; Dadachova, E. Targeted Radionuclide Therapy of Melanoma. Semin. Nucl. Med. 2016, 46, 250–259. [Google Scholar] [CrossRef]
- Raposo, G.; Stoorvogel, W. Extracellular vesicles: Exosomes, microvesicles and friends. J. Cell Biol. 2013, 200, 373–383. [Google Scholar] [CrossRef] [PubMed]
- Rossin, R.; Robillard, M.S. Pretargeted imaging using bioorthogonal chemistry in mice. Curr. Opin. Chem. Biol. 2014, 21, 161–169. [Google Scholar] [CrossRef]
| Preclinical and clinical models | Effect of Tspan8 Expression and Modulation by Antibody or Tspan8-LEL Targeting on Migration/Invasion/Metastasis, Proliferation/Tumor Growth, Angiogenesis | References |
|---|---|---|
| Rat pancreatic adenocarcinoma cells (AS-Tspan8 vs. AS) | In vitro: similar proliferation of two AS cell lines Inhibition by anti-rat Tspan8 mAb D6.1 of AS-Tspan8 In vivo: increased metastasis formation of AS-Tspan8 (i.v., s.c. or i.f.p. injection) | [20] |
| Rat pancreatic adenocarcinoma cells (AS-Tspan8 vs. AS) | In vitro: increased endothelial cell branching blocked by mAb D6.1 In vivo: peritoneal carcinosis—increased vessel density (intravital microscopy) abolished by mAb D6 | [21] |
| Highly metastatic rat pancreatic adenocarcinoma BSp73ASML (+/−Tspan8 knockdown) | In vitro: transwell migration and wound healing: reduced in BSp73ASML-Tspan8kd and by mAb D6.1 in BSp73AMSL No effect of D6.1 on BSp73ASML single-cell motility In vivo: delayed metastasis and prolonged survival in BSp73ASML-Tspan8kd | [22] |
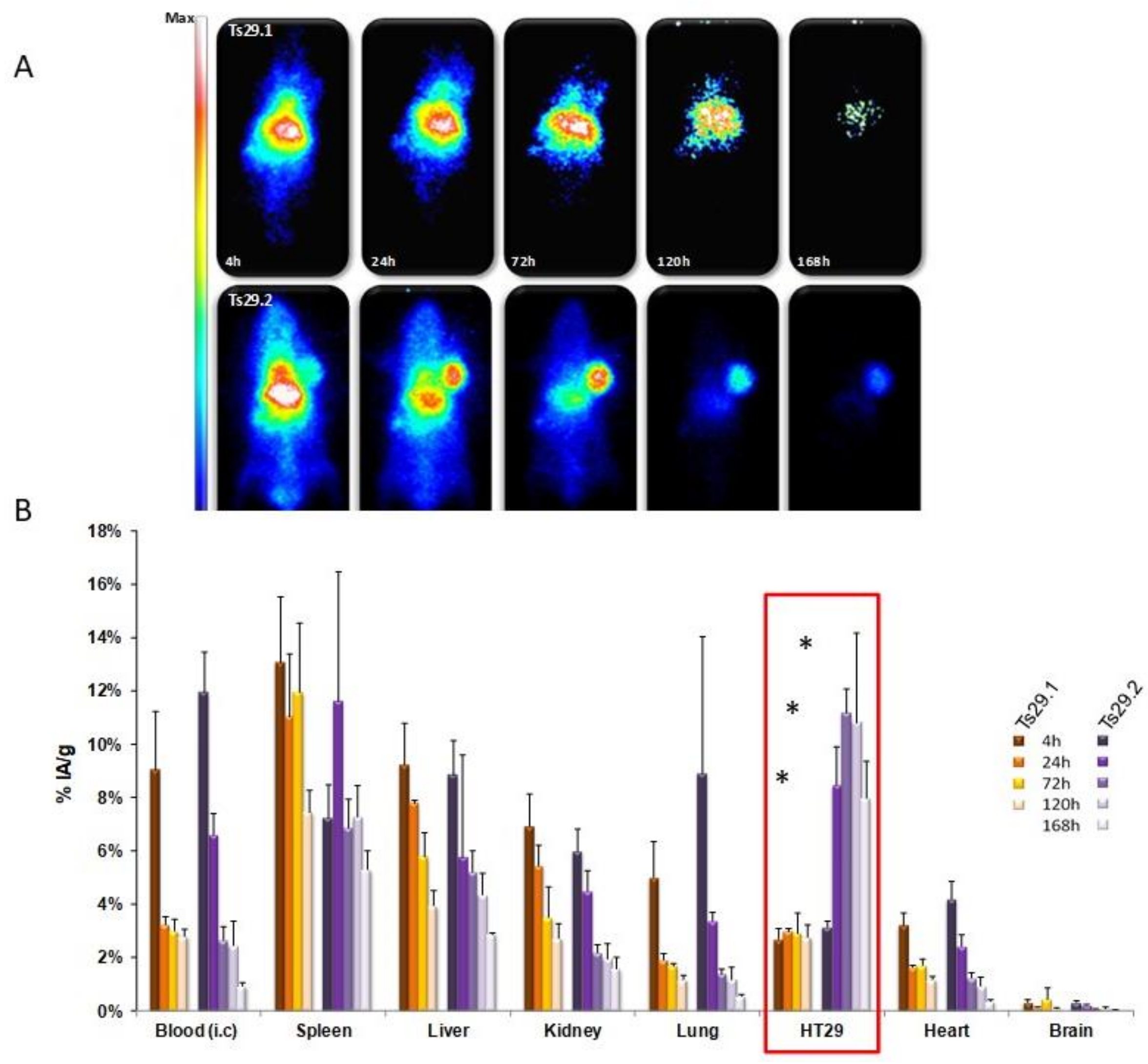
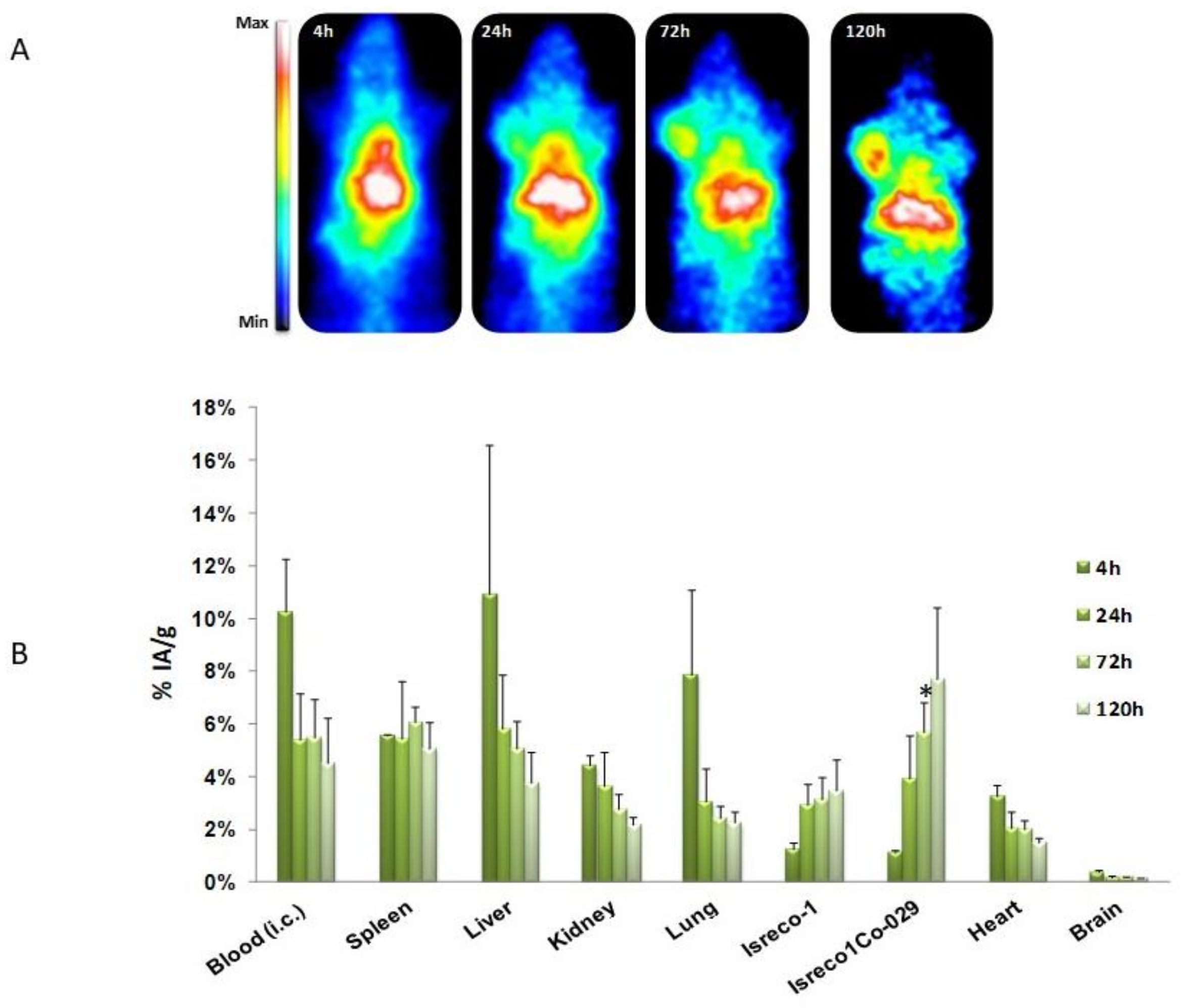
| Human colorectal cancer cell lines: Isreco1 and Is-Co029(Tspan8) HT-29, SW480 and SW480-Tspan8 | Patients: IHC: Tspan8 high expression correlated with worse prognosis In vitro: single cell-motility on collagen I increased by Ecad, p120ctn and EGFR RNAi when Tspan8 is expressed. This effect is reversed by anti-mouse Tspan8 mAb Ts29.1. No effect of mAb Ts29.1 or Ts29.2 on proliferation In vivo (nude mice): tumor growth reduced by i.p. injection of mAb Ts29.2 No effect on angiogenesis (IHC–CD34 labeling) Tumor growth inhibition by i.v. injection of [177Lu]DOTA-Ts29.2 | [12,19,23,24] |
| Human ovarian cell line—effect of Tspan8 RNAi, Tspan8-LEL-Fc, Tspan8-LEL IgG (human Ab selected by phage display) | In vitro: invasion in Matrigel-coated Transwell is inhibited by the 3 reagents In vivo: partial metastasis inhibition (SK-OV3-Luc) by i.v. injection of Tspan8-LEL IgG | [25] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bonnet, M.; Maisonial-Besset, A.; Zhu, Y.; Witkowski, T.; Roche, G.; Boucheix, C.; Greco, C.; Degoul, F. Targeting the Tetraspanins with Monoclonal Antibodies in Oncology: Focus on Tspan8/Co-029. Cancers 2019, 11, 179. https://doi.org/10.3390/cancers11020179
Bonnet M, Maisonial-Besset A, Zhu Y, Witkowski T, Roche G, Boucheix C, Greco C, Degoul F. Targeting the Tetraspanins with Monoclonal Antibodies in Oncology: Focus on Tspan8/Co-029. Cancers. 2019; 11(2):179. https://doi.org/10.3390/cancers11020179
Chicago/Turabian StyleBonnet, Mathilde, Aurélie Maisonial-Besset, Yingying Zhu, Tiffany Witkowski, Gwenaëlle Roche, Claude Boucheix, Céline Greco, and Françoise Degoul. 2019. "Targeting the Tetraspanins with Monoclonal Antibodies in Oncology: Focus on Tspan8/Co-029" Cancers 11, no. 2: 179. https://doi.org/10.3390/cancers11020179
APA StyleBonnet, M., Maisonial-Besset, A., Zhu, Y., Witkowski, T., Roche, G., Boucheix, C., Greco, C., & Degoul, F. (2019). Targeting the Tetraspanins with Monoclonal Antibodies in Oncology: Focus on Tspan8/Co-029. Cancers, 11(2), 179. https://doi.org/10.3390/cancers11020179






