The Role of TRAIL/DRs in the Modulation of Immune Cells and Responses
Abstract
1. Introduction
2. Expression and Function of TRAIL/DRs in Immune Cells
2.1. Myeloid Cells
2.1.1. Neutrophils
2.1.2. Monocytes and Macrophages
2.1.3. Dendritic Cells
2.1.4. Other Myeloid Cells
Basophils and Mast Cells
Eosinophils
Myeloid Derived Suppressor Cells
2.2. Lymphoid Cells
2.2.1. Conventional Natural Killer Cells and Innate Lymphoid Cells 1
2.2.2. Conventional αβ T cells
2.2.3. Innate-like T cells
Invariant Natural Killer T cells
γδ T cells
2.2.4. B cells
3. Common Themes and Open Questions on the Role of TRAIL/DRs in Immune Cells
3.1. Regulation of TRAIL-Sensitivity
3.1.1. TRAIL Expression
3.1.2. Membrane-Bound vs. Soluble TRAIL
3.1.3. Expression Levels of the Death Receptors
3.1.4. Receptor Interactions
3.1.5. Signaling Pathways
3.2. TRAIL on Exosomes
3.3. TRAIL’s Role in the Resolution of Immune Responses
3.3.1. Removing Effector Cells
3.3.2. Impairing Effector Cells
3.3.3. Limiting Tissue Damage
3.3.4. Avoiding Autoimmunity
4. TRAIL/DRs in the Tumor Microenvironment
4.1. Anti-Tumor Cytotoxicity of TRAIL+ Immune Cells
4.2. TRAIL Susceptibility of Tumors and Immune-Surveillance
4.3. Tumor Mechanisms to Evade TRAIL-Mediated Cytotoxicity
4.4. TRAIL/DR-Related Immune-Tumor Cross-Talk in the Tumor Microenvironment
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| APC | antigen presenting cell |
| AICD | activation-induced cell death |
| AML | acute myeloid leukemia |
| BCR | B cell receptor |
| BM-DCs | bone marrow-derived dendritic cells |
| BMDMs | bone-marrow derived macrophages |
| CAC | colitis-associated colorectal cancer |
| CBMCs | cord blood-derived mast cells |
| CCL | C-C motif chemokine ligand |
| CD | cluster of differentiation |
| CIA | collagen-induced arthritis |
| CML | chronic myeloid leukemia |
| cNK | conventional NK |
| CNS | central nervous system |
| CRP | C-reactive protein |
| CXCR | C-X-C chemokine receptor |
| DC | dendritic cell |
| DcR | decoy receptor |
| DD | death domain |
| DR | death receptor |
| DTx | diphtheria toxin |
| EAE | experimental autoimmune encephalomyelitis |
| EAT | experimental autoimmune thyroiditis |
| EoE | eosinophilic esophagitis |
| Eomes | eomesodermin |
| ER | endoplasmic reticulum |
| fMLP | N-Formylmethionyl-leucyl-phenylalanine |
| GC | germinal center |
| G-CSF | granulocyte-colony stimulating factor |
| GM-CSF | granulocyte-macrophage colony-stimulating factor |
| HBV | hepatitis B virus |
| HCC | hepatocellular carcinoma |
| Hsp | heat shock protein |
| IFN | interferon |
| Ig | immunoglobulin |
| IL | interleukin |
| ILC | innate lymphoid cell |
| LPS | lipopolysaccharide |
| MCA | methylcholanthrene |
| MCMV | murine cytomegalovirus |
| MDSCs | myeloid derived suppressor cells |
| mRNA | messenger RNA |
| MSC | mesenchymal stem cell |
| iNKT | invariant Natural Killer T |
| NK | Natural Killer |
| OPG | osteoprotegerin |
| Pam3C | tri-palmitoyl-S-glyceryl cysteine |
| pDC | plasmacytoid dendritic cell |
| PEDF | pigment epithelium derived factor |
| PHA | phytohemagglutinin |
| Poly I:C | polyinosinic:polycytidylic acid |
| RANKL | Receptor activator of nuclear factor kappa-Β ligand |
| RNA | ribonucleic acid |
| SDF1 | stromal cell-derived factor 1 |
| SLE | systemic lupus erythematosus |
| TAMs | tumor associated macrophages |
| TCR | T cell receptor |
| TGF | transforming growth factor |
| Th | T helper type |
| TLR | toll like receptor |
| TNF | tumor necrosis factor |
| TNFRSF | TNF receptor superfamily |
| TRAIL | tumor necrosis factor–related apoptosis–inducing ligand |
| Treg | regulatory T cell |
| UPR | unfolded protein response. |
References
- von Karstedt, S.; Montinaro, A.; Walczak, H. Exploring the TRAILs less travelled: TRAIL in cancer biology and therapy. Nat. Publ. Group 2017, 17, 352–366. [Google Scholar] [CrossRef]
- O’ Reilly, E.; Tirincsi, A.; Logue, S.E.; Szegezdi, E. The Janus Face of Death Receptor Signaling during Tumor Immunoediting. Front. Immunol. 2016, 7, 273. [Google Scholar] [CrossRef] [PubMed]
- Falschlehner, C.; Schaefer, U.; Walczak, H. Following TRAIL’s path in the immune system. Immunology 2009, 127, 145–154. [Google Scholar] [CrossRef] [PubMed]
- Rossin, A.; Miloro, G.; Hueber, A.O. TRAIL and FasL Functions in Cancer and Autoimmune Diseases: Towards an Increasing Complexity. Cancers 2019, 11, 639. [Google Scholar] [CrossRef] [PubMed]
- Naval, J.; de Miguel, D.; Gallego-Lleyda, A.; Anel, A.; Martinez-Lostao, L. Importance of TRAIL Molecular Anatomy in Receptor Oligomerization and Signaling. Implications for Cancer Therapy. Cancers 2019, 11, 444. [Google Scholar] [CrossRef]
- Vanamee, É.S.; Faustman, D.L. Structural principles of tumor necrosis factor superfamily signaling. Sci. Signal. 2018, 11, eaao4910. [Google Scholar] [CrossRef] [PubMed]
- Lafont, E.; Hartwig, T.; Walczak, H. Paving TRAIL’s Path with Ubiquitin. Trends Biochem. Sci. 2018, 43, 44–60. [Google Scholar] [CrossRef] [PubMed]
- Emery, J.G.; McDonnell, P.; Burke, M.B.; Deen, K.C.; Lyn, S.; Silverman, C.; Dul, E.; Appelbaum, E.R.; Eichman, C.; DiPrinzio, R.; et al. Osteoprotegerin is a receptor for the cytotoxic ligand TRAIL. J. Biol. Chem. 1998, 273, 14363–14367. [Google Scholar] [CrossRef] [PubMed]
- Dubuisson, A.; Micheau, O. Antibodies and Derivatives Targeting DR4 and DR5 for Cancer Therapy. Antibodies 2017, 6, 16. [Google Scholar] [CrossRef]
- Kretz, A.L.; Trauzold, A.; Hillenbrand, A.; Knippschild, U.; Henne-Bruns, D.; von Karstedt, S.; Lemke, J. TRAILblazing Strategies for Cancer Treatment. Cancers 2019, 11, 456. [Google Scholar] [CrossRef] [PubMed]
- Kamohara, H.; Matsuyama, W.; Shimozato, O.; Abe, K.; Galligan, C.; Hashimoto, S.I.; Matsushima, K.; Yoshimura, T. Regulation of tumour necrosis factor-related apoptosis-inducing ligand (TRAIL) and TRAIL receptor expression in human neutrophils. Immunology 2004, 111, 186–194. [Google Scholar] [CrossRef] [PubMed]
- Renshaw, S.A.; Parmar, J.S.; Singleton, V.; Rowe, S.J.; Dockrell, D.H.; Dower, S.K.; Bingle, C.D.; Chilvers, E.R.; Whyte, M.K.B. Acceleration of Human Neutrophil Apoptosis by TRAIL. J. Immunol. 2003, 170, 1027–1033. [Google Scholar] [CrossRef] [PubMed]
- Koga, Y.; Matsuzaki, A.; Suminoe, A.; Hattori, H.; Hara, T. Neutrophil-derived TNF-related apoptosis-inducing ligand (TRAIL): A novel mechanism of antitumor effect by neutrophils. Cancer Res. 2004, 64, 1037–1043. [Google Scholar] [CrossRef] [PubMed]
- Kemp, T.J.; Ludwig, A.T.; Earel, J.K.; Moore, J.M.; Vanoosten, R.L.; Moses, B.; Leidal, K.; Nauseef, W.M.; Griffith, T.S. Neutrophil stimulation with Mycobacterium bovis bacillus Calmette-Guerin (BCG) results in the release of functional soluble TRAIL/Apo-2L. Blood 2005, 106, 3474–3482. [Google Scholar] [CrossRef] [PubMed]
- Cassatella, M.A.; Huber, V.; Calzetti, F.; Margotto, D.; Tamassia, N.; Peri, G.; Mantovani, A.; Rivoltini, L.; Tecchio, C. Interferon-activated neutrophils store a TNF-related apoptosis-inducing ligand (TRAIL/Apo-2 ligand) intracellular pool that is readily mobilizable following exposure to proinflammatory mediators. J. Leukoc. Biol. 2006, 79, 123–132. [Google Scholar] [CrossRef] [PubMed]
- Simons, M.P.; Leidal, K.G.; Nauseef, W.M.; Griffith, T.S. TNF-related apoptosis-inducing ligand (TRAIL) is expressed throughout myeloid development, resulting in a broad distribution among neutrophil granules. J. Leukoc. Biol. 2008, 83, 621–629. [Google Scholar] [CrossRef] [PubMed]
- Tecchio, C.; Huber, V.; Scapini, P.; Calzetti, F.; Margotto, D.; Todeschini, G.; Pilla, L.; Martinelli, G.; Pizzolo, G.; Rivoltini, L.; et al. IFNalpha-stimulated neutrophils and monocytes release a soluble form of TNF-related apoptosis-inducing ligand (TRAIL/Apo-2 ligand) displaying apoptotic activity on leukemic cells. Blood 2004, 103, 3837–3844. [Google Scholar] [CrossRef]
- Tanaka, H.; Ito, T.; Kyo, T.; Kimura, A. Treatment with IFNalpha in vivo up-regulates serum-soluble TNF-related apoptosis inducing ligand (sTRAIL) levels and TRAIL mRNA expressions in neutrophils in chronic myelogenous leukemia patients. Eur. J. Haematol. 2007, 78, 389–398. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.L.; Wang, Y.; Huang, C.Y.; Zhou, Z.Q.; Zhao, J.J.; Zhang, X.F.; Pan, Q.Z.; Wu, J.X.; Weng, D.S.; Tang, Y.; et al. IL-17 induces antitumor immunity by promoting beneficial neutrophil recruitment and activation in esophageal squamous cell carcinoma. Oncoimmunology 2017, 7, e1373234. [Google Scholar] [CrossRef] [PubMed]
- Lum, J.J.; Bren, G.; McClure, R.; Badley, A.D. Elimination of Senescent Neutrophils by TNF-Related Apoptosis-Inducing Ligand. J. Immunol. 2005, 175, 1232–1238. [Google Scholar] [CrossRef] [PubMed]
- McGrath, E.E.; Lawrie, A.; Marriott, H.M.; Mercer, P.; Cross, S.S.; Arnold, N.; Singleton, V.; Thompson, A.A.R.; Walmsley, S.R.; Renshaw, S.A.; et al. Deficiency of tumour necrosis factor-related apoptosis-inducing ligand exacerbates lung injury and fibrosis. Thorax 2012, 67, 796–803. [Google Scholar] [CrossRef] [PubMed]
- Herold, S.; Steinmueller, M.; von Wulffen, W.; Cakarova, L.; Pinto, R.; Pleschka, S.; Mack, M.; Kuziel, W.A.; Corazza, N.; Brunner, T.; et al. Lung epithelial apoptosis in influenza virus pneumonia: The role of macrophage-expressed TNF-related apoptosis-inducing ligand. J. Exp. Med. 2008, 205, 3065–3077. [Google Scholar] [CrossRef] [PubMed]
- Leu, S.W.; Shi, L.; Xu, C.; Zhao, Y.; Liu, B.; Li, Y.; Shiedlin, A.; Xiang, C.; Shen, H.; Quinn, D.A.; et al. TLR4 through IFN-beta Promotes Low Molecular Mass Hyaluronan-Induced Neutrophil Apoptosis. J. Immunol. 2011, 186, 556–562. [Google Scholar] [CrossRef] [PubMed]
- Yan, B.; Wei, J.J.; Yuan, Y.; Sun, R.; Li, D.; Luo, J.; Liao, S.J.; Zhou, Y.H.; Shu, Y.; Wang, Q.; et al. IL-6 Cooperates with G-CSF To Induce Protumor Function of Neutrophils in Bone Marrow by Enhancing STAT3 Activation. J. Immunol. 2013, 190, 5882–5893. [Google Scholar] [CrossRef] [PubMed]
- Zou, J.M.; Qin, J.; Li, Y.C.; Wang, Y.; Li, D.; Shu, Y.; Luo, C.; Wang, S.S.; Chi, G.; Guo, F.; et al. IL-35 induces N2 phenotype of neutrophils to promote tumor growth. Oncotarget 2017, 8, 33501–33514. [Google Scholar] [CrossRef]
- Sun, R.; Luo, J.; Li, D.; Shu, Y.; Luo, C.; Wang, S.S.; Qin, J.; Zhang, G.M.; Feng, Z.H. Neutrophils with protumor potential could efficiently suppress tumor growth after cytokine priming and in presence of normal NK cells. Oncotarget 2014, 5, 12621–12634. [Google Scholar] [CrossRef]
- Hasegawa, H.; Yamada, Y.; Harasawa, H.; Tsuji, T.; Murata, K.; Sugahara, K.; Tsuruda, K.; Masuda, M.; Takasu, N.; Kamihira, S. Restricted expression of tumor necrosis factor-related apoptosis-inducing ligand receptor 4 in human peripheral blood lymphocytes. Cell. Immunol. 2004, 231, 1–7. [Google Scholar] [CrossRef]
- Liguori, M.; Buracchi, C.; Pasqualini, F.; Bergomas, F.; Pesce, S.; Sironi, M.; Grizzi, F.; Mantovani, A.; Belgiovine, C.; Allavena, P. Functional TRAIL receptors in monocytes and tumor-associated macrophages: A possible targeting pathway in the tumor microenvironment. Oncotarget 2016, 7, 41662–41676. [Google Scholar] [CrossRef]
- Daigle, I.; Simon, H.U. Alternative functions for TRAIL receptors in eosinophils and neutrophils. Swiss Med. Wkly 2001, 131, 231–237. [Google Scholar]
- Dominguez, G.A.; Condamine, T.; Mony, S.; Hashimoto, A.; Wang, F.; Liu, Q.; Forero, A.; Bendell, J.; Witt, R.; Hockstein, N.; et al. Selective Targeting of Myeloid-Derived Suppressor Cells in Cancer Patients Using DS-8273a, an Agonistic TRAIL-R2 Antibody. Clin. Cancer Res. 2017, 23, 2942–2950. [Google Scholar] [CrossRef]
- Hoffmann, O.; Priller, J.; Prozorovski, T.; Schulze-Topphoff, U.; Baeva, N.; Lunemann, J.D.; Aktas, O.; Mahrhofer, C.; Stricker, S.; Zipp, F.; et al. TRAIL limits excessive host immune responses in bacterial meningitis. J. Clin. Investig. 2007, 117, 2004–2013. [Google Scholar] [CrossRef] [PubMed]
- Beyer, K.; Poetschke, C.; Partecke, L.I.; von Bernstorff, W.; Maier, S.; Broeker, B.M.; Heidecke, C.D. TRAIL Induces Neutrophil Apoptosis and Dampens Sepsis-Induced Organ Injury in Murine Colon Ascendens Stent Peritonitis. PLoS ONE 2014, 9, e97451. [Google Scholar] [CrossRef] [PubMed]
- Zheng, S.J.; Jiang, J.; Shen, H.; Chen, Y.H. Reduced Apoptosis and Ameliorated Listeriosis in TRAIL-Null Mice. J. Immunol. 2004, 173, 5652–5658. [Google Scholar] [CrossRef] [PubMed]
- Cziupka, K.; Busemann, A.; Partecke, L.I.; Pötschke, C.; Rath, M.; Traeger, T.; Koerner, P.; von Bernstorff, W.; Kessler, W.; Diedrich, S.; et al. Tumor necrosis factor-related apoptosis-inducing ligand (TRAIL) improves the innate immune response and enhances survival in murine polymicrobial sepsis. Crit. Care Med. 2010, 38, 2169–2174. [Google Scholar] [CrossRef] [PubMed]
- Nagase, H.; Miyamasu, M.; Yamaguchi, M.; Imanishi, M.; Tsuno, N.H.; Matsushima, K.; Yamamoto, K.; Morita, Y.; Hirai, K. Cytokine-mediated regulation of CXCR4 expression in human neutrophils. J. Leukoc. Biol. 2002, 71, 711–717. [Google Scholar] [PubMed]
- Liew, P.X.; Kubes, P. The Neutrophil’s Role During Health and Disease. Physiol. Rev. 2019, 99, 1223–1248. [Google Scholar] [CrossRef]
- Vogt, K.L.; Summers, C.; Condliffe, A.M. The clinical consequences of neutrophil priming. Curr. Opin. Hematol. 2019, 26, 22–27. [Google Scholar] [CrossRef]
- Wang, X.; Qiu, L.; Li, Z.; Wang, X.Y.; Yi, H. Understanding the Multifaceted Role of Neutrophils in Cancer and Autoimmune Diseases. Front. Immunol. 2018, 9, 2456. [Google Scholar] [CrossRef]
- McGrath, E.E.; Marriott, H.M.; Lawrie, A.; Francis, S.E.; Sabroe, I.; Renshaw, S.A.; Dockrell, D.H.; Whyte, M.K.B. TNF-related apoptosis-inducing ligand (TRAIL) regulates inflammatory neutrophil apoptosis and enhances resolution of inflammation. J. Leukoc. Biol. 2011, 90, 855–865. [Google Scholar] [CrossRef]
- Koedel, U.; Frankenberg, T.; Kirschnek, S.; Obermaier, B.; Häcker, H.; Paul, R.; Häcker, G. Apoptosis Is Essential for Neutrophil Functional Shutdown and Determines Tissue Damage in Experimental Pneumococcal Meningitis. PLoS Pathog. 2009, 5, e1000461. [Google Scholar] [CrossRef]
- Steinwede, K.; Henken, S.; Bohling, J.; Maus, R.; Ueberberg, B.; Brumshagen, C.; Brincks, E.L.; Griffith, T.S.; Welte, T.; Maus, U.A. TNF-related apoptosis-inducing ligand (TRAIL) exerts therapeutic efficacy for the treatment of pneumococcal pneumonia in mice. J. Exp. Med. 2012, 209, 1937–1952. [Google Scholar] [CrossRef] [PubMed]
- Shinnoh, M.; Horinaka, M.; Yasuda, T.; Yoshikawa, S.; Morita, M.; Yamada, T.; Miki, T.; Sakai, T. Clostridium butyricum MIYAIRI 588 shows antitumor effects by enhancing the release of TRAIL from neutrophils through MMP-8. Int. J. Oncol. 2013, 42, 903–911. [Google Scholar] [CrossRef] [PubMed]
- Ludwig, A.T.; Moore, J.M.; Luo, Y.; Chen, X.; Saltsgaver, N.A.; O’Donnell, M.A.; Griffith, T.S. Tumor necrosis factor-related apoptosis-inducing ligand: A novel mechanism for Bacillus Calmette-Guérin-induced antitumor activity. Cancer Res. 2004, 64, 3386–3390. [Google Scholar] [CrossRef] [PubMed]
- Jablonska, E.; Jablonski, J.; Marcinczyk, M.; Grabowska, Z.; Piotrowski, L. The release of soluble forms of TRAIL and DR5 by neutrophils of oral cavity cancer patients. Folia Histochem. Cytobiol. 2008, 46, 177–183. [Google Scholar] [CrossRef] [PubMed]
- Sawicka-Powierza, J.; Jablonska, E.; Kloczko, J.; Piszcz, J.; Garley, M.; Ratajczk-Wrona, W. Evaluation of TNF superfamily molecules release by neutrophils and B leukemic cells of patients with chronic B-Cell lymphocytic leukemia. Neoplasma 2011, 58, 45–50. [Google Scholar] [CrossRef] [PubMed]
- Lu, G.; Janjic, B.M.; Janjic, J.; Whiteside, T.L.; Storkus, W.J.; Vujanovic, N.L. Innate Direct Anticancer Effector Function of Human Immature Dendritic Cells. II. Role of TNF, Lymphotoxin-α1β2, Fas Ligand, and TNF-Related Apoptosis-Inducing Ligand. J. Immunol. 2002, 168, 1831–1839. [Google Scholar] [CrossRef]
- Griffith, T.S.; Wiley, S.R.; Kubin, M.Z.; Sedger, L.M.; Maliszewski, C.R.; Fanger, N.A. Monocyte-mediated Tumoricidal Activity via the Tumor Necrosis Factor–related Cytokine, TRAIL. J. Exp. Med. 1999, 189, 1343–1354. [Google Scholar] [CrossRef]
- Halaas, O.; Vik, R.; Ashkenazi, A.; Espevik, T. Lipopolysaccharide induces expression of APO2 ligand/TRAIL in human monocytes and macrophages. Scand. J. Immunol. 2000, 51, 244–250. [Google Scholar] [CrossRef]
- Fanger, N.A.; Maliszewski, C.R.; Schooley, K.; Griffith, T.S. Human Dendritic Cells Mediate Cellular Apoptosis via Tumor Necrosis Factor–Related Apoptosis-Inducing Ligand (Trail). J. Exp. Med. 1999, 190, 1155–1164. [Google Scholar] [CrossRef]
- Yamamoto, T.; Nagano, H.; Sakon, M.; Wada, H.; Eguchi, H.; Kondo, M.; Damdinsuren, B.; Ota, H.; Nakamura, M.; Wada, H.; et al. Partial contribution of tumor necrosis factor-related apoptosis-inducing ligand (TRAIL)/TRAIL receptor pathway to antitumor effects of interferon-alpha/5-fluorouracil against Hepatocellular Carcinoma. Clin. Cancer Res. 2004, 10, 7884–7895. [Google Scholar] [CrossRef]
- Kemp, T.J.; Moore, J.M.; Griffith, T.S. Human B Cells Express Functional TRAIL/Apo-2 Ligand after CpG-Containing Oligodeoxynucleotide Stimulation. J. Immunol. 2004, 173, 892–899. [Google Scholar] [CrossRef] [PubMed]
- Washburn, B.; Weigand, M.A.; Grosse-Wilde, A.; Janke, M.; Stahl, H.; Rieser, E.; Sprick, M.R.; Schirrmacher, V.; Walczak, H. TNF-Related Apoptosis-Inducing Ligand Mediates Tumoricidal Activity of Human Monocytes Stimulated by Newcastle Disease Virus. J. Immunol. 2003, 170, 1814–1821. [Google Scholar] [CrossRef]
- Ehrlich, S.; Infante-Duarte, C.; Seeger, B.; Zipp, F. Regulation of soluble and surface-bound TRAIL in human T cells, B cells, and monocytes. Cytokine 2003, 24, 244–253. [Google Scholar] [CrossRef]
- Mehmut, M.; Takeda, K.; Abe, M.; Ogata, H.; Hirose, S.; Okumura, K.; Fujime, M. Fas Ligand and TNF-Related Apoptosis-Inducing Ligand Induction on Infiltrating Lymphocytes in Bladder Carcinoma by Bacillus Calmette-Guérin Treatment. Urol. Int. 2005, 75, 80–87. [Google Scholar] [CrossRef] [PubMed]
- Halin, S.; Rudolfsson, S.H.; Doll, J.A.; Crawford, S.E.; Wikström, P.; Bergh, A. Pigment Epithelium-Derived Factor Stimulates Tumor Macrophage Recruitment and Is Downregulated by the Prostate Tumor Microenvironment. Neoplasia (New York) 2010, 12, 336–345. [Google Scholar] [CrossRef]
- Ho, T.C.; Chen, S.L.; Shih, S.C.; Chang, S.J.; Yang, S.L.; Hsieh, J.W.; Cheng, H.C.; Chen, L.J.; Tsao, Y.P. Pigment Epithelium-derived Factor (PEDF) Promotes Tumor Cell Death by Inducing Macrophage Membrane Tumor Necrosis Factor-related Apoptosis-inducing Ligand (TRAIL). J. Biol. Chem. 2011, 286, 35943–35954. [Google Scholar] [CrossRef]
- Secchiero, P.; Rimondi, E.; di Iasio, M.G.; Agnoletto, C.; Melloni, E.; Volpi, I.; Zauli, G. C-Reactive Protein Downregulates TRAIL Expression in Human Peripheral Monocytes via an Egr-1-Dependent Pathway. Clin. Cancer Res. 2013, 19, 1949–1959. [Google Scholar] [CrossRef]
- Kaplan, M.J.; Ray, D.; Mo, R.R.; Yung, R.L.; Richardson, B.C. TRAIL (Apo2 Ligand) and TWEAK (Apo3 Ligand) Mediate CD4+ T Cell Killing of Antigen-Presenting Macrophages. J. Immunol. 2000, 164, 2897–2904. [Google Scholar] [CrossRef]
- Diehl, G.E.; Yue, H.H.; Hsieh, K.; Kuang, A.A.; Ho, M.; Morici, L.A.; Lenz, L.L.; Cado, D.; Riley, L.W.; Winoto, A. TRAIL-R as a negative regulator of innate immune cell responses. Immunity 2004, 21, 877–889. [Google Scholar] [CrossRef]
- Huang, Y.; Wang, Y.; Li, X.; Chen, Z.; Li, X.; Wang, H.; Ni, M.; Li, J. Molecular mechanism of ER stress-induced gene expression of tumor necrosis factor-related apoptosis-inducing ligand (TRAIL) in macrophages. Febs J. 2015, 282, 2361–2378. [Google Scholar] [CrossRef]
- Martinez, F.O.; Gordon, S. The M1 and M2 paradigm of macrophage activation: Time for reassessment. F1000Prime Rep. 2014, 6, 13. [Google Scholar] [CrossRef] [PubMed]
- Germano, G.; Frapolli, R.; Belgiovine, C.; Anselmo, A.; Pesce, S.; Liguori, M.; Erba, E.; Uboldi, S.; Zucchetti, M.; Pasqualini, F.; et al. Role of Macrophage Targeting in the Antitumor Activity of Trabectedin. Cancer Cell 2013, 23, 249–262. [Google Scholar] [CrossRef]
- Huang, Y.J.; Hsu, S.H. TRAIL-functionalized gold nanoparticles selectively trigger apoptosis in polarized macrophages. Nanotheranostics 2017, 1, 326–337. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Wei, W.; Wang, D.; Shi, J.; Xiang, Y.; Zhang, Y.; Liu, S.; Liu, Y.; Zheng, D. Tumor necrosis factor (TNF)-related apoptosis-inducing ligand (TRAIL) induces chemotactic migration of monocytes via a death receptor 4-mediated RhoGTPase pathway. Mol. Immunol. 2010, 47, 2475–2484. [Google Scholar] [CrossRef]
- Li, J.; Yang, P.; Wu, Q.; Li, H.; Ding, Y.; Hsu, H.C.; Spalding, D.M.; Mountz, J.D. Death receptor 5-targeted depletion of interleukin-23-producing macrophages, Th17, and Th1/17 associated with defective tyrosine phosphatase in mice and patients with rheumatoid arthritis. Arthritis Rheum. 2013, 65, 2594–2605. [Google Scholar] [CrossRef] [PubMed]
- Secchiero, P.; Candido, R.; Corallini, F.; Zacchigna, S.; Toffoli, B.; Rimondi, E.; Fabris, B.; Giacca, M.; Zauli, G. Systemic Tumor Necrosis Factor–Related Apoptosis-Inducing Ligand Delivery Shows Antiatherosclerotic Activity in Apolipoprotein E–Null Diabetic Mice. Circulation 2006, 114, 1522–1530. [Google Scholar] [CrossRef]
- Di Bartolo, B.A.; Chan, J.; Bennett, M.R.; Cartland, S.; Bao, S.; Tuch, B.E.; Kavurma, M.M. TNF-related apoptosis-inducing ligand (TRAIL) protects against diabetes and atherosclerosis in Apoe−/− mice. Diabetologia 2011, 54, 3157–3167. [Google Scholar] [CrossRef]
- Li, J.; Hsu, H.C.; Yang, P.; Wu, Q.; Li, H.; Edgington, L.E.; Bogyo, M.; Kimberly, R.P.; Mountz, J.D. Treatment of arthritis by macrophage depletion and immunomodulation: Testing an apoptosis-mediated therapy in a humanized death receptor mouse model. Arthritis Rheum. 2012, 64, 1098–1109. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.; Li, Z.; Huang, W. Interleukin-4 Enhances the Sensitivity of Human Monocytes to Tumor Necrosis Factor-Related Apoptosis-Inducing Ligand Through Upregulation of Death Receptor 4. J. Interferon Cytokine Res. 2018, 38, 186–194. [Google Scholar] [CrossRef]
- Yao, Z.; Zhang, P.; Guo, H.; Shi, J.; Liu, S.; Liu, Y.; Zheng, D. RIP1 modulates death receptor mediated apoptosis and autophagy in macrophages. Mol. Oncol. 2014, 9, 806–817. [Google Scholar] [CrossRef] [PubMed]
- Strebel, A.; Bachmann, F.; Wernli, M.; Erb, P. Tumor necrosis factor-related, apoptosis-inducing ligand supports growth of mouse mastocytoma tumors by killing tumor-infiltrating macrophages. Int. J. Cancer 2002, 100, 627–634. [Google Scholar] [CrossRef] [PubMed]
- Liu, F.; Cheng, W.; Bi, X.; Zhang, Y.; Zhao, Y.; Jiang, F. Stage-dependent effects of exogenous TRAIL on atherogenesis: Role of ER stress-mediated sensitization of macrophage apoptosis. Clin. Exp. Pharm. Physiol. 2016, 43, 543–551. [Google Scholar] [CrossRef] [PubMed]
- Herbeuval, J.P.; Lambert, C.; Sabido, O.; Cottier, M.; Fournel, P.; Dy, M.; Genin, C. Macrophages from cancer patients: Analysis of TRAIL, TRAIL receptors, and colon tumor cell apoptosis. J. Nat. Cancer Inst. 2003, 95, 611–621. [Google Scholar] [CrossRef] [PubMed]
- Secchiero, P.; Gonelli, A.; Mirandola, P.; Melloni, E.; Zamai, L.; Celeghini, C.; Milani, D.; Zauli, G. Tumor necrosis factor-related apoptosis-inducing ligand induces monocytic maturation of leukemic and normal myeloid precursors through a caspase-dependent pathway. Blood 2002, 100, 2421–2429. [Google Scholar] [CrossRef] [PubMed]
- Gao, J.; Wang, D.; Liu, D.; Liu, M.; Ge, Y.; Jiang, M.; Liu, Y.; Zheng, D. Tumor necrosis factor–related apoptosis-inducing ligand induces the expression of proinflammatory cytokines in macrophages and re-educates tumor-associated macrophages to an antitumor phenotype. Mol. Biol. Cell 2015, 26, 3178–3189. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.Y.; Kim, Y.M.; Park, J.M.; Han, Y.M.; Lee, K.C.; Hahm, K.B.; Hong, S. Cancer preventive effect of recombinant TRAIL by ablation of oncogenic inflammation in colitis-associated cancer rather than anticancer effect. Oncotarget 2018, 9, 1705–1716. [Google Scholar] [CrossRef] [PubMed]
- Vidalain, P.O.; Azocar, O.; Yagita, H.; Rabourdin-Combe, C.; Servet-Delprat, C. Cytotoxic Activity of Human Dendritic Cells Is Differentially Regulated by Double-Stranded RNA and CD40 Ligand. J. Immunol. 2001, 167, 3765–3772. [Google Scholar] [CrossRef]
- Shi, J.; Ikeda, K.; Maeda, Y.; Shinagawa, K.; Ohtsuka, A.; Yamamura, H.; Tanimoto, M. Identification of CD123+ myeloid dendritic cells as an early-stage immature subset with strong tumoristatic potential. Cancer Lett. 2008, 270, 19–29. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Wang, M.; Shi, J.; Wan, Y.; Li, J.; Yuan, Y. A subset of myeloid dendritic cells derived from peripheral blood monocytes represented a predominant subset characterized by their potential tumor-inhibiting activity. Vitr. Cell Dev. Biol. Anim. 2009, 45, 398–404. [Google Scholar] [CrossRef]
- Anguille, S.; Lion, E.; Tel, J.; de Vries, I.J.M.; Couderé, K.; Fromm, P.D.; Van Tendeloo, V.F.; Smits, E.L.; Berneman, Z.N. Interleukin-15-Induced CD56+ Myeloid Dendritic Cells Combine Potent Tumor Antigen Presentation with Direct Tumoricidal Potential. PLoS ONE 2012, 7, e51851. [Google Scholar] [CrossRef]
- Liu, S.; Yu, Y.; Zhang, M.; Wang, W.; Cao, X. The Involvement of TNF- -Related Apoptosis-Inducing Ligand in the Enhanced Cytotoxicity of IFN-β-Stimulated Human Dendritic Cells to Tumor Cells. J. Immunol. 2001, 166, 5407–5415. [Google Scholar] [CrossRef] [PubMed]
- Santini, S.M.; Lapenta, C.; Logozzi, M.; Parlato, S.; Spada, M.; Di Pucchio, T.; Belardelli, F. Type I interferon as a powerful adjuvant for monocyte-derived dendritic cell development and activity in vitro and in Hu-PBL-SCID mice. J. Exp. Med. 2000, 191, 1777–1788. [Google Scholar] [CrossRef] [PubMed]
- Korthals, M.; Safaian, N.; Kronenwett, R.; Maihöfer, D.; Schott, M.; Papewalis, C.; Diaz Blanco, E.; Winter, M.; Czibere, A.; Haas, R.; et al. Monocyte derived dendritic cells generated by IFN-α acquire mature dendritic and natural killer cell properties as shown by gene expression analysis. J. Trans. Med. 2007, 5, 46. [Google Scholar] [CrossRef] [PubMed]
- Kalb, M.L.; Glaser, A.; Stary, G.; Koszik, F.; Stingl, G. TRAIL+ Human Plasmacytoid Dendritic Cells Kill Tumor Cells In Vitro: Mechanisms of Imiquimod- and IFN-α-Mediated Antitumor Reactivity. J. Immunol. 2012, 188, 1583–1591. [Google Scholar] [CrossRef] [PubMed]
- Chaperot, L.; Blum, A.; Manches, O.; Lui, G.; Angel, J.; Molens, J.P.; Plumas, J. Virus or TLR Agonists Induce TRAIL-Mediated Cytotoxic Activity of Plasmacytoid Dendritic Cells. J. Immunol. 2005, 176, 248–255. [Google Scholar] [CrossRef] [PubMed]
- Taieb, J.; Chaput, N.; Ménard, C.; Apetoh, L.; Ullrich, E.; Bonmort, M.; Péquignot, M.; Casares, N.; Terme, M.; Flament, C.; et al. A novel dendritic cell subset involved in tumor immunosurveillance. Nat. Med. 2006, 12, 214–219. [Google Scholar] [CrossRef] [PubMed]
- Papewalis, C.; Jacobs, B.; Wuttke, M.; Ullrich, E.; Baehring, T.; Fenk, R.; Willenberg, H.S.; Schinner, S.; Cohnen, M.; Seissler, J.; et al. IFN-alpha skews monocytes into CD56+-expressing dendritic cells with potent functional activities in vitro and in vivo. J. Immunol. 2008, 180, 1462–1470. [Google Scholar] [CrossRef] [PubMed]
- Hardy, A.W.; Graham, D.R.; Shearer, G.M.; Herbeuval, J.P. HIV turns plasmacytoid dendritic cells (pDC) into TRAIL-expressing killer pDC and down-regulates HIV coreceptors by Toll-like receptor 7-induced IFN-alpha. Proc. Natl. Acad. Sci. USA 2007, 104, 17453–17458. [Google Scholar] [CrossRef] [PubMed]
- Molenkamp, B.G.; van Leeuwen, P.A.M.; Meijer, S.; Sluijter, B.J.R.; Wijnands, P.G.J.T.B.; Baars, A.; van den Eertwegh, A.J.M.; Scheper, R.J.; de Gruijl, T.D. Intradermal CpG-B Activates Both Plasmacytoid and Myeloid Dendritic Cells in the Sentinel Lymph Node of Melanoma Patients. Clin. Cancer Res. 2007, 13, 2961–2969. [Google Scholar] [CrossRef] [PubMed]
- Cho, Y.S.; Challa, S.; Clancy, L.; Chan, F.K.M. Lipopolysaccharide-induced expression of TRAIL promotes dendritic cell differentiation. Immunology 2010, 130, 504–515. [Google Scholar] [CrossRef] [PubMed]
- Achard, C.; Guillerme, J.B.; Bruni, D.; Boisgerault, N.; Combredet, C.; Tangy, F.; Jouvenet, N.; Grégoire, M.; Fonteneau, J.F. Oncolytic measles virus induces tumor necrosis factor-related apoptosis-inducing ligand (TRAIL)-mediated cytotoxicity by human myeloid and plasmacytoid dendritic cells. Oncoimmunology 2017, 6, e1261240. [Google Scholar] [CrossRef] [PubMed]
- Stary, G.; Klein, I.; Kohlhofer, S.; Koszik, F.; Scherzer, T.; Müllauer, L.; Quendler, H.; Kohrgruber, N.; Stingl, G. Plasmacytoid dendritic cells express TRAIL and induce CD4+ T-cell apoptosis in HIV-1 viremic patients. Blood 2009, 114, 3854–3863. [Google Scholar] [CrossRef] [PubMed]
- Thomann, S.; Boscheinen, J.B.; Vogel, K.; Knipe, D.M.; DeLuca, N.; Gross, S.; Schuler-Thurner, B.; Schuster, P.; Schmidt, B. Combined cytotoxic activity of an infectious, but non-replicative herpes simplex virus type 1 and plasmacytoid dendritic cells against tumour cells. Immunology 2015, 146, 327–338. [Google Scholar] [CrossRef] [PubMed]
- Roux, S.; Apetoh, L.; Chalmin, F.; Ladoire, S.; Mignot, G.; Puig, P.E.; Lauvau, G.; Zitvogel, L.; Martin, F.; Chauffert, B.; et al. CD4+CD25+ Tregs control the TRAIL-dependent cytotoxicity of tumor-infiltrating DCs in rodent models of colon cancer. J. Clin. Investig. 2008, 118, 3751–3761. [Google Scholar] [CrossRef] [PubMed]
- Drobits, B.; Holcmann, M.; Amberg, N.; Swiecki, M.; Grundtner, R.; Hammer, M.; Colonna, M.; Sibilia, M. Imiquimod clears tumors in mice independent of adaptive immunity by converting pDCs into tumor-killing effector cells. J. Clin. Investig. 2012, 122, 575–585. [Google Scholar] [CrossRef]
- Wu, J.; Li, S.; Yang, Y.; Zhu, S.; Zhang, M.; Qiao, Y.; Liu, Y.J.; Chen, J. TLR-activated plasmacytoid dendritic cells inhibit breast cancer cell growth in vitro and in vivo. Oncotarget 2017, 8, 11708–11718. [Google Scholar] [CrossRef] [PubMed]
- Arina, A.; Murillo, O.; Dubrot, J.; Azpilikueta, A.; Gabari, I.; Perez-Gracia, J.L.; Alfaro, C.; Berasain, C.; Prieto, J.; Ferrini, S.; et al. Interleukin-15 liver gene transfer increases the number and function of IKDCs and NK cells. Gene Ther. 2008, 15, 473–483. [Google Scholar] [CrossRef]
- Hira, S.K.; Mondal, I.; Bhattacharya, D.; Manna, P.P. Downregulation of endogenous STAT3 augments tumoricidal activity of interleukin 15 activated dendritic cell against lymphoma and leukemia via TRAIL. Exp. Cell Res. 2014, 327, 192–208. [Google Scholar] [CrossRef]
- You, R.I.; Chang, Y.C.; Chen, P.M.; Wang, W.S.; Hsu, T.L.; Yang, C.Y.; Lee, C.T.; Hsieh, S.L. Apoptosis of dendritic cells induced by decoy receptor 3 (DcR3). Blood 2008, 111, 1480–1488. [Google Scholar] [CrossRef]
- Blum, A.; Chaperot, L.; Molens, J.P.; Foissaud, V.; Plantaz, D.; Plumas, J. Mechanisms of TRAIL-induced apoptosis in leukemic plasmacytoid dendritic cells. Exp. Hematol. 2006, 34, 1655–1662. [Google Scholar] [CrossRef]
- Hayakawa, Y.; Screpanti, V.; Yagita, H.; Grandien, A.; Ljunggren, H.G.; Smyth, M.J.; Chambers, B.J. NK cell TRAIL eliminates immature dendritic cells in vivo and limits dendritic cell vaccination efficacy. J. Immunol. 2004, 172, 123–129. [Google Scholar] [CrossRef]
- Ciesek, S.; Liermann, H.; Hadem, J.; Greten, T.; Tillmann, H.L.; Cornberg, M.; Aslan, N.; Manns, M.P.; Wedemeyer, H. Impaired TRAIL-dependent cytotoxicity of CD1c-positive dendritic cells in chronic hepatitis C virus infection. J. Viral Hepat. 2007, 15, 200–211. [Google Scholar] [CrossRef]
- Tyrinova, T.V.; Leplina, O.Y.; Mishinov, S.V.; Tikhonova, M.A.; Shevela, E.Y.; Stupak, V.V.; Pendyurin, I.V.; Shilov, A.G.; Alyamkina, E.A.; Rubtsova, N.V.; et al. Cytotoxic activity of ex-vivo generated IFNα-induced monocyte-derived dendritic cells in brain glioma patients. Cell. Immunol. 2013, 284, 146–153. [Google Scholar] [CrossRef] [PubMed]
- Leplina, O.Y.; Tyrinova, T.V.; Tikhonova, M.A.; Ostanin, A.A.; Chernykh, E.R. Interferon alpha induces generation of semi-mature dendritic cells with high pro-inflammatory and cytotoxic potential. Cytokine 2015, 71, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Tel, J.; Smits, E.L.; Anguille, S.; Joshi, R.N.; Figdor, C.G.; de Vries, I.J.M. Human plasmacytoid dendritic cells are equipped with antigen-presenting and tumoricidal capacities. Blood 2012, 120, 3936–3944. [Google Scholar] [CrossRef] [PubMed]
- Iyori, M.; Zhang, T.; Pantel, H.; Gagne, B.A.; Sentman, C.L. TRAIL/DR5 Plays a Critical Role in NK Cell-Mediated Negative Regulation of Dendritic Cell Cross-Priming of T Cells. J. Immunol. 2011, 187, 3087–3095. [Google Scholar] [CrossRef] [PubMed]
- Förster, A.; Falcone, F.H.; Gibbs, B.F.; Preussner, L.M.; Fiebig, B.S.; Altunok, H.; Seeger, J.M.; Cerny-Reiterer, S.; Rabenhorst, A.; Papenfuss, K.; et al. Anti-Fas/CD95 and tumor necrosis factor-related apoptosis-inducing ligand (TRAIL) differentially regulate apoptosis in normal and neoplastic human basophils. Leuk. Lymphoma 2012, 54, 835–842. [Google Scholar] [CrossRef]
- Florian, S.; Sonneck, K.; Czerny, M.; Hennersdorf, F.; Hauswirth, A.W.; Buhring, H.J.; Valent, P. Detection of novel leukocyte differentiation antigens on basophils and mast cells by HLDA8 antibodies. Allergy 2006, 61, 1054–1062. [Google Scholar] [CrossRef] [PubMed]
- Berent-Maoz, B.; Piliponsky, A.M.; Daigle, I.; Simon, H.U.; Levi-Schaffer, F. Human Mast Cells Undergo TRAIL-Induced Apoptosis. J. Immunol. 2006, 176, 2272–2278. [Google Scholar] [CrossRef]
- Berent-Maoz, B.; Salemi, S.; Mankuta, D.; Simon, H.U.; Levi-Schaffer, F. Original article: TRAIL mediated signaling in human mast cells: The influence of IgE-dependent activation. Allergy 2008, 63, 333–340. [Google Scholar] [CrossRef]
- Vassina, E.; Leverkus, M.; Yousefi, S.; Braathen, L.R.; Simon, H.U.; Simon, D. Increased Expression and a Potential Anti-Inflammatory Role of TRAIL in Atopic Dermatitis. J. Investig. Dermatol. 2005, 125, 746–752. [Google Scholar] [CrossRef] [PubMed]
- Jakiela, B.; Szczeklik, W.; Sokolowska, B.; Mastalerz, L.; Sanak, M.; Plutecka, H.; Szczeklik, A. Intrinsic pathway of apoptosis in peripheral blood eosinophils of Churg–Strauss syndrome. Rheumatology 2009, 48, 1202–1207. [Google Scholar] [CrossRef] [PubMed]
- Robertson, N.M.; Zangrilli, J.G.; Steplewski, A.; Hastie, A.; Lindemeyer, R.G.; Planeta, M.A.; Smith, M.K.; Innocent, N.; Musani, A.; Pascual, R.; et al. Differential Expression of TRAIL and TRAIL Receptors in Allergic Asthmatics Following Segmental Antigen Challenge: Evidence for a Role of TRAIL in Eosinophil Survival. J. Immunol. 2002, 169, 5986–5996. [Google Scholar] [CrossRef] [PubMed]
- Mitsuyama, H.; Matsuyama, W.; Watanabe, M.; Shirahama, Y.; Higashimoto, I.; Wada, T.; Osame, M.; Arimura, K. Increased expression of TRAIL receptor 3 on eosinophils in Churg-Strauss syndrome. Arthritis Rheum. 2007, 56, 662–673. [Google Scholar] [CrossRef][Green Version]
- Noth, I.; Strek, M.E.; Leff, A.R. Churg-Strauss syndrome. Lancet 2003, 361, 587–594. [Google Scholar] [CrossRef]
- Weckmann, M.; Collison, A.; Simpson, J.L.; Kopp, M.V.; Wark, P.A.B.; Smyth, M.J.; Yagita, H.; Matthaei, K.I.; Hansbro, N.; Whitehead, B.; et al. Critical link between TRAIL and CCL20 for the activation of TH2 cells and the expression of allergic airway disease. Nat. Med. 2007, 13, 1308–1315. [Google Scholar] [CrossRef]
- Collison, A.; Hatchwell, L.; Verrills, N.; Wark, P.A.B.; de Siqueira, A.P.; Tooze, M.; Carpenter, H.; Don, A.S.; Morris, J.C.; Zimmermann, N.; et al. The E3 ubiquitin ligase midline 1 promotes allergen and rhinovirus-induced asthma by inhibiting protein phosphatase 2A activity. Nat. Med. 2013, 19, 232–237. [Google Scholar] [CrossRef]
- Mishra, A.; Hogan, S.P.; Brandt, E.B.; Rothenberg, M.E. IL-5 Promotes Eosinophil Trafficking to the Esophagus. J. Immunol. 2002, 168, 2464–2469. [Google Scholar] [CrossRef]
- Flood-Page, P.; Menzies-Gow, A.; Phipps, S.; Ying, S.; Wangoo, A.; Ludwig, M.S.; Barnes, N.; Robinson, D.; Kay, A.B. Anti-IL-5 treatment reduces deposition of ECM proteins in the bronchial subepithelial basement membrane of mild atopic asthmatics. J. Clin. Invest. 2003, 112, 1029–1036. [Google Scholar] [CrossRef]
- Girkin, J.L.; Hatchwell, L.M.; Collison, A.M.; Starkey, M.R.; Hansbro, P.M.; Yagita, H.; Foster, P.S.; Mattes, J. TRAIL signaling is proinflammatory and proviral in a murine model of rhinovirus 1B infection. Am. J. Physiol. Lung Cell. Mol. Physiol. 2017, 312, L89–L99. [Google Scholar] [CrossRef]
- Collison, A.M.; Sokulsky, L.A.; Sherrill, J.D.; Nightingale, S.; Hatchwell, L.; Talley, N.J.; Walker, M.M.; Rothenberg, M.E.; Mattes, J. TNF-related apoptosis-inducing ligand (TRAIL) regulates midline-1, thymic stromal lymphopoietin, inflammation, and remodeling in experimental eosinophilic esophagitis. J. Allergy Clin. Immunol. 2015, 136, 971–982. [Google Scholar] [CrossRef] [PubMed]
- Tisato, V.; Garrovo, C.; Biffi, S.; Petrera, F.; Voltan, R.; Casciano, F.; Meroni, G.; Agnoletto, C.; Zauli, G.; Secchiero, P. Intranasal Administration of Recombinant TRAIL Down-Regulates CXCL-1/KC in an Ovalbumin-Induced Airway Inflammation Murine Model. PLoS ONE 2014, 9, e115387. [Google Scholar] [CrossRef] [PubMed]
- Faustino, L.; Fonseca, D.M.; Florsheim, E.B.; Resende, R.R.; Lepique, A.P.; Faquim-Mauro, E.; Gomes, E.; Silva, J.S.; Yagita, H.; Russo, M. Tumor necrosis factor-related apoptosis-inducing ligand mediates the resolution of allergic airway inflammation induced by chronic allergen inhalation. Mucosal Immunol. 2014, 7, 1199–1208. [Google Scholar] [CrossRef] [PubMed]
- Collison, A.; Li, J.; Pereira de Siqueira, A.; Zhang, J.; Toop, H.D.; Morris, J.C.; Foster, P.S.; Mattes, J. Tumor Necrosis Factor–Related Apoptosis-Inducing Ligand Regulates Hallmark Features of Airways Remodeling in Allergic Airways Disease. Am. J. Respir. Cell Mol. Biol. 2014, 51, 86–93. [Google Scholar] [CrossRef]
- Marvel, D.; Gabrilovich, D.I. Myeloid-derived suppressor cells in the tumor microenvironment: Expect the unexpected. J. Clin. Investig. 2015, 125, 3356–3364. [Google Scholar] [CrossRef] [PubMed]
- Hartwig, T.; Montinaro, A.; von Karstedt, S.; Sevko, A.; Surinova, S.; Chakravarthy, A.; Taraborrelli, L.; Draber, P.; Lafont, E.; Vargas, F.A.; et al. The TRAIL-Induced Cancer Secretome Promotes a Tumor-Supportive Immune Microenvironment via CCR2. Mol. Cell 2017, 65, 730–742. [Google Scholar] [CrossRef]
- Condamine, T.; Kumar, V.; Ramachandran, I.R.; Youn, J.I.; Celis, E.; Finnberg, N.; El-Deiry, W.S.; Winograd, R.; Vonderheide, R.H.; English, N.R.; et al. ER stress regulates myeloid-derived suppressor cell fate through TRAIL-R–mediated apoptosis. J. Clin. Investig. 2014, 124, 2626–2639. [Google Scholar] [CrossRef]
- Kajitani, K.; Tanaka, Y.; Arihiro, K.; Kataoka, T.; Ohdan, H. Mechanistic analysis of the antitumor efficacy of human natural killer cells against breast cancer cells. Breast Cancer Res. Treat. 2012, 134, 139–155. [Google Scholar] [CrossRef]
- Berg, M.; Lundqvist, A.; McCoy, P., Jr.; Samsel, L.; Fan, Y.; Tawab, A.; Childs, R. Clinical-grade ex vivo-expanded human natural killer cells up-regulate activating receptors and death receptor ligands and have enhanced cytolytic activity against tumor cells. Cytotherapy 2009, 11, 341–355. [Google Scholar] [CrossRef]
- Kashii, Y.; Giorda, R.; Herberman, R.B.; Whiteside, T.L.; Vujanovic, N.L. Constitutive expression and role of the TNF family ligands in apoptotic killing of tumor cells by human NK cells. J. Immunol. 1999, 163, 5358–5366. [Google Scholar]
- Stegmann, K.A.; Björkström, N.K.; Veber, H.; Ciesek, S.; Riese, P.; Wiegand, J.; Hadem, J.; Suneetha, P.V.; Jaroszewicz, J.; Wang, C.; et al. Interferon-α–Induced TRAIL on Natural Killer Cells Is Associated With Control of Hepatitis C Virus Infection. Gastroenterology 2010, 138, 1885–1897. [Google Scholar] [CrossRef] [PubMed]
- Allan, D.S.J.; Cerdeira, A.S.; Ranjan, A.; Kirkham, C.L.; Aguilar, O.A.; Tanaka, M.; Childs, R.W.; Dunbar, C.E.; Strominger, J.L.; Kopcow, H.D.; et al. Transcriptome analysis reveals similarities between human blood CD3− CD56bright cells and mouse CD127+ innate lymphoid cells. Sci. Rep. 2017, 7, 4480. [Google Scholar] [CrossRef] [PubMed]
- Stegmann, K.A.; Robertson, F.; Hansi, N.; Gill, U.; Pallant, C.; Christophides, T.; Pallett, L.J.; Peppa, D.; Dunn, C.; Fusai, G.; et al. CXCR6 marks a novel subset of T-bet-lo Eomes-hi natural killer cells residing in human liver. Sci. Rep. 2016, 6, 26157. [Google Scholar] [CrossRef] [PubMed]
- Hwang, S.; Han, J.; Baek, J.S.; Tak, E.; Song, G.W.; Lee, S.G.; Jung, D.H.; Park, G.C.; Ahn, C.S.; Kim, N. Cytotoxicity of Human Hepatic Intrasinusoidal CD56bright Natural Killer Cells against Hepatocellular Carcinoma Cells. Int. J. Mol. Sci. 2019, 20, 1564. [Google Scholar] [CrossRef] [PubMed]
- Ishiyama, K.; Ohdan, H.; Ohira, M.; Mitsuta, H.; Arihiro, K.; Asahara, T. Difference in cytotoxicity against hepatocellular carcinoma between liver and periphery natural killer cells in humans. Hepatology 2006, 43, 362–372. [Google Scholar] [CrossRef] [PubMed]
- Takeda, K.; Hayakawa, Y.; Smyth, M.J.; Kayagaki, N.; Yamaguchi, N.; Kakuta, S.; Iwakura, Y.; Yagita, H.; Okumura, K. Involvement of tumor necrosis factor-related apoptosis-inducing ligand in surveillance of tumor metastasis by liver natural killer cells. Nat. Med. 2001, 7, 94–100. [Google Scholar] [CrossRef]
- Smyth, M.J.; Cretney, E.; Takeda, K.; Wiltrout, R.H.; Sedger, L.M.; Kayagaki, N.; Yagita, H.; Okumura, K. Tumor necrosis factor-related apoptosis-inducing ligand (TRAIL) contributes to interferon gamma-dependent natural killer cell protection from tumor metastasis. J. Exp. Med. 2001, 193, 661–670. [Google Scholar] [CrossRef]
- Ochi, M.; Ohdan, H.; Mitsuta, H.; Onoe, T.; Tokita, D.; Hara, H.; Ishiyama, K.; Zhou, W.; Tanaka, Y.; Asahara, T. Liver NK cells expressing TRAIL are toxic against self hepatocytes in mice. Hepatology 2004, 39, 1321–1331. [Google Scholar] [CrossRef]
- Jiao, Y.; Huntington, N.D.; Belz, G.T.; Seillet, C. Type 1 Innate Lymphoid Cell Biology: Lessons Learnt from Natural Killer Cells. Front. Immunol. 2016, 7, 293. [Google Scholar] [CrossRef]
- Robinette, M.L.; Colonna, M. Immune modules shared by innate lymphoid cells and T cells. J. Allergy Clin. Immunol. 2016, 138, 1243–1251. [Google Scholar] [CrossRef]
- Colonna, M. Innate Lymphoid Cells: Diversity, Plasticity, and Unique Functions in Immunity. Immunity 2018, 48, 1104–1117. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.H.; Shin, J.H.; Haggadone, M.D.; Sunwoo, J.B. The aryl hydrocarbon receptor is required for the maintenance of liver-resident natural killer cells. J. Exp. Med. 2016, 213, 2249–2257. [Google Scholar] [CrossRef] [PubMed]
- Dang, V.T.A.; Tanabe, K.; Tanaka, Y.; Tokumoto, N.; Misumi, T.; Saeki, Y.; Fujikuni, N.; Ohdan, H. Fasting Enhances TRAIL-Mediated Liver Natural Killer Cell Activity via HSP70 Upregulation. PLoS ONE 2014, 9, e110748. [Google Scholar] [CrossRef]
- Peng, H.; Jiang, X.; Chen, Y.; Sojka, D.K.; Wei, H.; Gao, X.; Sun, R.; Yokoyama, W.M.; Tian, Z. Liver-resident NK cells confer adaptive immunity in skin-contact inflammation. J. Clin. Investig. 2013, 123, 1444–1456. [Google Scholar] [CrossRef] [PubMed]
- Robinette, M.L.; Fuchs, A.; Cortez, V.S.; Lee, J.S.; Wang, Y.; Durum, S.K.; Gilfillan, S.; Colonna, M.; Consortium, I.G. Transcriptional programs define molecular characteristics of innate lymphoid cell classes and subsets. Nat. Immunol. 2015, 16, 306–317. [Google Scholar] [CrossRef] [PubMed]
- Cortez, V.S.; Fuchs, A.; Cella, M.; Gilfillan, S.; Colonna, M. Cutting Edge: Salivary Gland NK Cells Develop Independently of Nfil3 in Steady-State. J. Immunol. 2014, 192, 4487–4491. [Google Scholar] [CrossRef] [PubMed]
- Cortez, V.S.; Cervantes-Barragan, L.; Robinette, M.L.; Bando, J.K.; Wang, Y.; Geiger, T.L.; Gilfillan, S.; Fuchs, A.; Vivier, E.; Sun, J.C.; et al. Transforming Growth Factor-b Signaling Guides the Differentiation of Innate Lymphoid Cells in Salivary Glands. Immunity 2016, 44, 1127–1139. [Google Scholar] [CrossRef]
- Picarda, G.; Ghosh, R.; McDonald, B.; Verma, S.; Thiault, N.; El Morabiti, R.; Griffith, T.S.; Benedict, C.A. Cytomegalovirus evades TRAIL-mediated innate lymphoid cell 1 defenses. J. Virol. 2019, 93, e00617-19. [Google Scholar] [CrossRef] [PubMed]
- Daussy, C.; Faure, F.; Mayol, K.; Viel, S.; Gasteiger, G.; Charrier, E.; Bienvenu, J.; Henry, T.; Debien, E.; Hasan, U.A.; et al. T-bet and Eomes instruct the development of two distinct natural killer cell lineages in the liver and in the bone marrow. J. Exp. Med. 2014, 211, 563–577. [Google Scholar] [CrossRef]
- Constantinides, M.G.; Gudjonson, H.; McDonald, B.D.; Ishizuka, I.E.; Verhoef, P.A.; Dinner, A.R.; Bendelac, A. PLZF expression maps the early stages of ILC1 lineage development. Proc. Natl. Acad. Sci. USA 2015, 112, 5123–5128. [Google Scholar] [CrossRef]
- Sheppard, S.; Schuster, I.S.; Andoniou, C.E.; Cocita, C.; Adejumo, T.; Kung, S.K.P.; Sun, J.C.; Degli-Esposti, M.A.; Guerra, N. The Murine Natural Cytotoxic Receptor NKp46/NCR1 Controls TRAIL Protein Expression in NK Cells and ILC1s. CellReports 2018, 22, 3385–3392. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Dong, W.; Zhang, Y.; Caligiuri, M.A.; Yu, J. Dependence of innate lymphoid cell 1 development on NKp46. PLoS Biol 2018, 16, e2004867. [Google Scholar] [CrossRef] [PubMed]
- Turchinovich, G.; Ganter, S.; Bärenwaldt, A.; Finke, D. NKp46 Calibrates Tumoricidal Potential of Type 1 Innate Lymphocytes by Regulating TRAIL Expression. J. Immunol. 2018, 200, 3762–3768. [Google Scholar] [CrossRef] [PubMed]
- Almeida, F.F.; Tognarelli, S.; Marçais, A.; Kueh, A.J.; Friede, M.E.; Liao, Y.; Willis, S.N.; Luong, K.; Faure, F.; Mercier, F.E.; et al. A point mutation in the Ncr1 signal peptide impairs the development of innate lymphoid cell subsets. Oncoimmunology 2018, 7, e1475875. [Google Scholar] [CrossRef] [PubMed]
- Pallmer, K.; Barnstorf, I.; Baumann, N.S.; Borsa, M.; Jonjic, S.; Oxenius, A. NK cells negatively regulate CD8 T cells via natural cytotoxicity receptor (NCR) 1 during LCMV infection. PLoS Pathog. 2019, 15, e1007725. [Google Scholar] [CrossRef] [PubMed]
- Gao, Y.; Souza-Fonseca-Guimaraes, F.; Bald, T.; Ng, S.S.; Young, A.; Ngiow, S.F.; Rautela, J.; Straube, J.; Waddell, N.; Blake, S.J.; et al. Tumor immunoevasion by the conversion of effector NK cells into type 1 innate lymphoid cells. Nat. Immunol. 2017, 146, 1004–1015. [Google Scholar] [CrossRef]
- Cuff, A.O.; Sillito, F.; Dertschnig, S.; Hall, A.; Luong, T.; Chakraverty, R.; Male, V. The obese liver environment mediates conversion of NK cells to a less cytotoxic ILC1-like phenotype. Front. Immunol. 2019, 10, 2180. [Google Scholar] [CrossRef]
- Vivier, E.; Artis, D.; Colonna, M.; Diefenbach, A.; Di Santo, J.P.; Eberl, G.; Koyasu, S.; Locksley, R.M.; McKenzie, A.N.J.; Mebius, R.E.; et al. Innate Lymphoid Cells: 10 Years On. Cell 2018, 174, 1054–1066. [Google Scholar] [CrossRef] [PubMed]
- Zamai, L.; Ahmad, M.; Bennett, I.M.; Azzoni, L.; Alnemri, E.S.; Perussia, B. Natural killer (NK) cell-mediated cytotoxicity: Differential use of TRAIL and Fas ligand by immature and mature primary human NK cells. J. Exp. Med. 1998, 188, 2375–2380. [Google Scholar] [CrossRef] [PubMed]
- Mirandola, P.; Ponti, C.; Gobbi, G.; Sponzilli, I.; Vaccarezza, M.; Cocco, L.; Zauli, G.; Secchiero, P.; Manzoli, F.A.; Vitale, M. Activated human NK and CD8+ T cells express both TNF-related apoptosis-inducing ligand (TRAIL) and TRAIL receptors but are resistant to TRAIL-mediated cytotoxicity. Blood 2004, 104, 2418–2424. [Google Scholar] [CrossRef]
- Kayagaki, N.; Yamaguchi, N.; Nakayama, M.; Takeda, K.; Akiba, H.; Tsutsui, H.; Okamura, H.; Nakanishi, K.; Okumura, K.; Yagita, H. Expression and function of TNF-related apoptosis-inducing ligand on murine activated NK cells. J. Immunol. 1999, 163, 1906–1913. [Google Scholar] [PubMed]
- Zhang, C.; Zhang, J.; Niu, J.; Zhang, J.; Tian, Z. Interleukin-15 improves cytotoxicity of natural killer cells via up-regulating NKG2D and cytotoxic effector molecule expression as well as STAT1 and ERK1/2 phosphorylation. Cytokine 2008, 42, 128–136. [Google Scholar] [CrossRef] [PubMed]
- Lelaidier, M.; Dìaz-Rodriguez, Y.; Cordeau, M.; Cordeiro, P.; Haddad, E.; Herblot, S.; Duval, M. TRAIL-mediated killing of acute lymphoblastic leukemia by plasmacytoid dendritic cell-activated natural killer cells. Oncotarget 2015, 6, 29440–29455. [Google Scholar] [CrossRef] [PubMed]
- Ahlenstiel, G.; Titerence, R.H.; Koh, C.; Edlich, B.; Feld, J.J.; Rotman, Y.; Ghany, M.G.; Hoofnagle, J.H.; Liang, T.J.; Heller, T.; et al. Natural Killer Cells Are Polarized Toward Cytotoxicity in Chronic Hepatitis C in an Interferon-Alfa–Dependent Manner. Gastroenterology 2010, 138, 325–335. [Google Scholar] [CrossRef] [PubMed]
- Glässner, A.; Eisenhardt, M.; Krämer, B.; Körner, C.; Coenen, M.; Sauerbruch, T.; Spengler, U.; Nattermann, J. NK cells from HCV-infected patients effectively induce apoptosis of activated primary human hepatic stellate cells in a TRAIL-, FasL- and NKG2D-dependent manner. Lab. Investig. 2012, 92, 967–977. [Google Scholar] [CrossRef]
- Sato, K.; Hida, S.; Takayanagi, H.; Yokochi, T.; Kayagaki, N.; Takeda, K.; Yagita, H.; Okumura, K.; Tanaka, N.; Taniguchi, T.; et al. Antiviral response by natural killer cells through TRAIL gene induction by IFN-alpha/beta. Eur. J. Immunol. 2001, 31, 3138–3146. [Google Scholar] [CrossRef]
- Tu, Z.; Hamalainen-Laanaya, H.K.; Crispe, I.N.; Orloff, M.S. Synergy between TLR3 and IL-18 promotes IFN-γ dependent TRAIL expression in human liver NK cells. Cell. Immunol. 2011, 271, 286–291. [Google Scholar] [CrossRef]
- Song, D.Z.; Liang, Y.; Xiao, Q.; Yin, J.; Gong, J.L.; Lai, Z.P.; Zhang, Z.F.; Gao, L.X.; Fan, X.H. TRAIL is Involved in the Tumoricidal Activity of Mouse Natural Killer Cells Stimulated by Newcastle Disease Virus in Vitro. Ana. Rec. 2013, 296, 1552–1560. [Google Scholar] [CrossRef]
- Radaeva, S.; Sun, R.; Jaruga, B.; Nguyen, V.T.; Tian, Z.; Gao, B. Natural Killer Cells Ameliorate Liver Fibrosis by Killing Activated Stellate Cells in NKG2D-Dependent and Tumor Necrosis Factor–Related Apoptosis-Inducing Ligand–Dependent Manners. Gastroenterology 2006, 130, 435–452. [Google Scholar] [CrossRef]
- Takeda, K.; Cretney, E.; Hayakawa, Y.; Ota, T.; Akiba, H.; Ogasawara, K.; Yagita, H.; Kinoshita, K.; Okumura, K.; Smyth, M.J. TRAIL identifies immature natural killer cells in newborn mice and adult mouse liver. Blood 2005, 105, 2082–2089. [Google Scholar] [CrossRef]
- Dunn, C.; Brunetto, M.; Reynolds, G.; Christophides, T.; Kennedy, P.T.; Lampertico, P.; Das, A.; Lopes, A.R.; Borrow, P.; Williams, K.; et al. Cytokines induced during chronic hepatitis B virus infection promote a pathway for NK cell–mediated liver damage. J. Exp. Med. 2007, 204, 667–680. [Google Scholar] [CrossRef] [PubMed]
- Micco, L.; Peppa, D.; Loggi, E.; Schurich, A.; Jefferson, L.; Cursaro, C.; Panno, A.M.; Bernardi, M.; Brander, C.; Bihl, F.; et al. Differential boosting of innate and adaptive antiviral responses during pegylated-interferon-alpha therapy of chronic hepatitis B. J. Hepatol. 2013, 58, 225–233. [Google Scholar] [CrossRef] [PubMed]
- Ahlenstiel, G.; Edlich, B.; Hogdal, L.J.; Rotman, Y.; Noureddin, M.; Feld, J.J.; Holz, L.E.; Titerence, R.H.; Liang, T.J.; Rehermann, B. Early changes in natural killer cell function indicate virologic response to interferon therapy for hepatitis C. Gastroenterology 2011, 141, 1231–1239. [Google Scholar] [CrossRef][Green Version]
- Cordeau, M.; Belounis, A.; Lelaidier, M.; Cordeiro, P.; Sartelet, H.; Herblot, S.; Duval, M. Efficient Killing of High Risk Neuroblastoma Using Natural Killer Cells Activated by Plasmacytoid Dendritic Cells. PLoS ONE 2016, 11, e0164401. [Google Scholar] [CrossRef] [PubMed]
- Yoshioka, T.; Tatsumi, T.; Miyagi, T.; Mukai, K.; Nishio, K.; Nishio, A.; Yokoyama, Y.; Suda, T.; Kegasawa, T.; Shigekawa, M.; et al. Frequency and role of NKp46 and NKG2A in hepatitis B virus infection. PLoS ONE 2017, 12, e0174103. [Google Scholar] [CrossRef]
- Peppa, D.; Gill, U.S.; Reynolds, G.; Easom, N.J.W.; Pallett, L.J.; Schurich, A.; Micco, L.; Nebbia, G.; Singh, H.D.; Adams, D.H.; et al. Up-regulation of a death receptor renders antiviral T cells susceptible to NK cell–mediated deletion. J. Exp. Med. 2013, 210, 99–114. [Google Scholar] [CrossRef] [PubMed]
- Schuster, I.S.; Wikström, M.E.; Brizard, G.; Coudert, J.D.; Estcourt, M.J.; Manzur, M.; O’Reilly, L.A.; Smyth, M.J.; Trapani, J.A.; Hill, G.R.; et al. TRAIL+ NK Cells Control CD4+ T Cell Responses during Chronic Viral Infection to Limit Autoimmunity. Immunity 2014, 41, 646–656. [Google Scholar] [CrossRef]
- Paul, S.; Lal, G. The Molecular Mechanism of Natural Killer Cells Function and Its Importance in Cancer Immunotherapy. Front. Immunol. 2017, 8, 347. [Google Scholar] [CrossRef]
- Prager, I.; Watzl, C. Mechanisms of natural killer cell-mediated cellular cytotoxicity. J. Leukoc. Biol. 2019, 105, 1319–1329. [Google Scholar] [CrossRef]
- Martinez-Lostao, L.; Miguel, D.D.; Al-Wasaby, S.; Gallego-Lleyda, A.; Anel, A. Death ligands and granulysin: Mechanisms of tumor cell death induction and therapeutic opportunities. Immunotherapy 2015, 7, 882–883. [Google Scholar] [CrossRef]
- Poli, A.; Michel, T.; Patil, N.; Zimmer, J. Revisiting the Functional Impact of NK Cells. Trends Immunol. 2018, 39, 460–472. [Google Scholar] [CrossRef] [PubMed]
- Schuster, I.S.; Coudert, J.D.; Andoniou, C.E.; Degli-Esposti, M.A. “Natural Regulators”: NK Cells as Modulators of T Cell Immunity. Front. Immunol. 2016, 7, 230. [Google Scholar] [CrossRef] [PubMed]
- Nielsen, N.; Ødum, N.; Ursø, B.; Lanier, L.L.; Spee, P. Cytotoxicity of CD56bright NK Cells towards Autologous Activated CD4+ T Cells Is Mediated through NKG2D, LFA-1 and TRAIL and Dampened via CD94/NKG2A. PLoS ONE 2012, 7, e31959. [Google Scholar] [CrossRef] [PubMed]
- Wendling, U.; Walczak, H.; Dörr, J.; Jaboci, C.; Weller, M.; Krammer, P.H.; Zipp, F. Expression of TRAIL receptors in human autoreactive and foreign antigen-specific T cells. Cell Death Differ. 2000, 7, 637–644. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Rus, V.; Zernetkina, V.; Puliaev, R.; Cudrici, C.; Mathai, S.; Via, C.S. Increased expression and release of functional tumor necrosis factor-related apoptosis-inducing ligand (TRAIL) by T cells from lupus patients with active disease. Clin. Immunol. 2005, 117, 48–56. [Google Scholar] [CrossRef] [PubMed]
- Dorothee, G.; Vergnon, I.; Menez, J.; Echchakir, H.; Grunenwald, D.; Kubin, M.; Chouaib, S.; Mami-Chouaib, F. Tumor-Infiltrating CD4+ T Lymphocytes Express APO2 Ligand (APO2L)/TRAIL upon Specific Stimulation with Autologous Lung Carcinoma Cells: Role of IFN-α on APO2L/TRAIL Expression and -Mediated Cytotoxicity. J. Immunol. 2002, 169, 809–817. [Google Scholar] [CrossRef]
- Kayagaki, N.; Yamaguchi, N.; Nakayama, M.; Eto, H.; Okumura, K.; Yagita, H. Type I Interferons (IFNs) Regulate Tumor Necrosis Factor–related Apoptosis-inducing Ligand (TRAIL) Expression on Human T Cells: A Novel Mechanism for the Antitumor Effects of Type I IFNs. J. Exp. Med. 1999, 189, 1451–1460. [Google Scholar] [CrossRef]
- Dicker, F.; Kater, A.P.; Fukuda, T.; Kipps, T.J. Fas-ligand (CD178) and TRAIL synergistically induce apoptosis of CD40-activated chronic lymphocytic leukemia B cells. Blood 2005, 105, 3193–3198. [Google Scholar] [CrossRef]
- Mariani, S.M.; Krammer, P.H. Surface expression of TRAIL/Apo-2 ligand in activated mouse T and B cells. Eur. J. Immunol. 1998, 28, 1492–1498. [Google Scholar] [CrossRef]
- Ren, X.; Ye, F.; Jiang, Z.; Chu, Y.; Xiong, S.; Wang, Y. Involvement of cellular death in TRAIL/DR5-dependent suppression induced by CD4+CD25+ regulatory T cells. Cell Death Differ. 2007, 14, 2076–2084. [Google Scholar] [CrossRef]
- Zhang, X.R.; Zhang, L.Y.; Devadas, S.; Li, L.; Keegan, A.D.; Shi, Y.F. Reciprocal expression of TRAIL and CD95L in Th1 and Th2 cells: Role of apoptosis in T helper subset differentiation. Cell Death Differ. 2003, 10, 203–210. [Google Scholar] [CrossRef] [PubMed]
- Arbour, N.; Rastikerdar, E.; McCrea, E.; Lapierre, Y.; Dörr, J.; Bar-Or, A.; Antel, J.P. Upregulation of TRAIL expression on human T lymphocytes by interferon beta and glatiramer acetate. Mult. Scler. J. 2005, 11, 652–657. [Google Scholar] [CrossRef] [PubMed]
- Kayagaki, N.; Yamaguchi, N.; Nakayama, M.; Kawasaki, A.; Akiba, H.; Okumura, K.; Yagita, H. Involvement of TNF-related apoptosis-inducing ligand in human CD4+ T cell-mediated cytotoxicity. J. Immunol. 1999, 162, 2639–2647. [Google Scholar] [PubMed]
- Kuwashima, N.; Nishimura, F.; Eguchi, J.; Sato, H.; Hatano, M.; Tsugawa, T.; Sakaida, T.; Dusak, J.E.; Fellows-Mayle, W.K.; Papworth, G.D.; et al. Delivery of dendritic cells engineered to secrete IFN-alpha into central nervous system tumors enhances the efficacy of peripheral tumor cell vaccines: Dependence on apoptotic pathways. J. Immunol. 2005, 175, 2730–2740. [Google Scholar] [CrossRef]
- Ikeda, T.; Hirata, S.; Fukushima, S.; Matsunaga, Y.; Ito, T.; Uchino, M.; Nishimura, Y.; Senju, S. Dual Effects of TRAIL in Suppression of Autoimmunity: The Inhibition of Th1 Cells and the Promotion of Regulatory T Cells. J. Immunol. 2010, 185, 5259–5267. [Google Scholar] [CrossRef] [PubMed]
- Lunemann, J.D.; Waiczies, S.; Ehrlich, S.; Wendling, U.; Seeger, B.; Kamradt, T.; Zipp, F. Death Ligand TRAIL Induces No Apoptosis but Inhibits Activation of Human (Auto)antigen-Specific T Cells. J. Immunol. 2002, 168, 4881–4888. [Google Scholar] [CrossRef] [PubMed]
- Lehnert, C.; Weiswange, M.; Jeremias, I.; Bayer, C.; Grunert, M.; Debatin, K.M.; Strauss, G. TRAIL–Receptor Costimulation Inhibits Proximal TCR Signaling and Suppresses Human T Cell Activation and Proliferation. J. Immunol. 2014, 193, 4021–4031. [Google Scholar] [CrossRef] [PubMed]
- Janssen, E.M.; Droin, N.M.; Lemmens, E.E.; Pinkoski, M.J.; Bensinger, S.J.; Ehst, B.D.; Griffith, T.S.; Green, D.R.; Schoenberger, S.P. CD4+ T-cell help controls CD8+ T-cell memory via TRAIL-mediated activation-induced cell death. Nature 2005, 434, 88–93. [Google Scholar] [CrossRef] [PubMed]
- Janssen, E.M.; Lemmens, E.E.; Wolfe, T.; Christen, U.; von Herrath, M.G.; Schoenberger, S.P. CD4+ T cells are required for secondary expansion and memory in CD8+ T lymphocytes. Nature 2003, 421, 852–856. [Google Scholar] [CrossRef]
- Feau, S.; Garcia, Z.; Arens, R.; Yagita, H.; Borst, J.; Schoenberger, S.P. The CD4+ T-cell help signal is transmitted from APC to CD8+ T-cells via CD27-CD70 interactions. Nat. Commun. 2012, 3, 948. [Google Scholar] [CrossRef] [PubMed]
- Wolkers, M.C.; Bensinger, S.J.; Green, D.R.; Schoenberger, S.P.; Janssen, E.M. Interleukin-2 rescues helpless effector CD8+ T cells by diminishing the susceptibility to TRAIL mediated death. Immunol. Lett. 2011, 139, 25–32. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Oh, S.; Perera, L.P.; Terabe, M.; Ni, L.; Waldmann, T.A.; Berzofsky, J.A. IL-15 as a mediator of CD4+ help for CD8+ T cell longevity and avoidance of TRAIL-mediated apoptosis. Proc. Natl. Acad. Sci. USA 2008, 105, 5201–5206. [Google Scholar] [CrossRef] [PubMed]
- Sun, J.C.; Williams, M.A.; Bevan, M.J. CD4+ T cells are required for the maintenance, not programming, of memory CD8+ T cells after acute infection. Nat. Immunol. 2004, 5, 927–933. [Google Scholar] [CrossRef] [PubMed]
- Wolkers, M.C.; Gerlach, C.; Arens, R.; Janssen, E.M.; Fitzgerald, P.; Schumacher, T.N.; Medema, J.P.; Green, D.R.; Schoenberger, S.P. Nab2 regulates secondary CD8+ T-cell responses through control of TRAIL expression. Blood 2012, 119, 798–804. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Gurung, P.; Kucaba, T.A.; Schoenberger, S.P.; Ferguson, T.A.; Griffith, T.S. TRAIL-expressing CD8 +T cells mediate tolerance following soluble peptide-induced peripheral T cell deletion. J. Leukoc. Biol. 2010, 88, 1217–1225. [Google Scholar] [CrossRef] [PubMed]
- Griffith, T.S.; Kazama, H.; VanOosten, R.L.; Earle, J.K.; Herndon, J.M.; Green, D.R.; Ferguson, T.A. Apoptotic Cells Induce Tolerance by Generating Helpless CD8+ T Cells That Produce TRAIL. J. Immunol. 2007, 178, 2679–2687. [Google Scholar] [CrossRef]
- Kuerten, S.; Asaad, R.J.; Schoenberger, S.P.; Angelov, D.N.; Lehmann, P.V.; Tary-Lehmann, M. The TRAIL of Helpless CD8 +T Cells in HIV Infection. AIDS Res. Hum. Retrovir. 2008, 24, 1175–1183. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.H.; Cao, Z.; Wolf, J.M.; Van Antwerp, M.; Baker, J.; James, R. Death Ligand Tumor Necrosis Factor-Related Apoptosis-Inducing Ligand Inhibits Experimental Autoimmune Thyroiditis. Endocrinology 2005, 146, 4721–4726. [Google Scholar] [CrossRef][Green Version]
- Chou, A.H.; Tsai, H.F.; Lin, L.L.; Hsieh, S.L.; Hsu, P.I.; Hsu, P.N. Enhanced Proliferation and Increased IFN- Production in T Cells by Signal Transduced Through TNF-Related Apoptosis-Inducing Ligand. J. Immunol. 2001, 167, 1347–1352. [Google Scholar] [CrossRef]
- Bosque, A.; Pardo, J.; Martínez-Lorenzo, M.J.; Lasierra, P.; Larrad, L.; Marzo, I.; Naval, J.; Anel, A. Human CD8+ T cell blasts are more sensitive than CD4+ T cell blasts to regulation by APO2L/TRAIL. Eur. J. Immunol. 2005, 35, 1812–1821. [Google Scholar] [CrossRef]
- Song, K.; Chen, Y.; Göke, R.; Wilmen, A.; Seidel, C.; Göke, A.; Hilliard, B.; Chen, Y. Tumor Necrosis Factor–Related Apoptosis-Inducing Ligand (Trail) Is an Inhibitor of Autoimmune Inflammation and Cell Cycle Progression. J. Exp. Med. 2000, 191, 1095–1104. [Google Scholar] [CrossRef] [PubMed]
- Chyuan, I.T.; Tsai, H.F.; Liao, H.J.; Wu, C.S.; Hsu, P.N. An apoptosis-independent role of TRAIL in suppressing joint inflammation and inhibiting T-cell activation in inflammatory arthritis. Cell. Mol. Immunol. 2017, 15, 846. [Google Scholar] [CrossRef] [PubMed]
- Chyuan, I.T.; Tsai, H.F.; Wu, C.S.; Sung, C.C.; Hsu, P.N. TRAIL-Mediated Suppression of T Cell Receptor Signaling Inhibits T Cell Activation and Inflammation in Experimental Autoimmune Encephalomyelitis. Front. Immunol. 2018, 9, 15. [Google Scholar] [CrossRef] [PubMed]
- Chyuan, I.T.; Tsai, H.F.; Wu, C.S.; Hsu, P.N. TRAIL suppresses gut inflammation and inhibits colitogeic T-cell activation in experimental colitis via an apoptosis-independent pathway. Mucosal Immunol. 2019, 12, 980–989. [Google Scholar] [CrossRef] [PubMed]
- Cretney, E.; McQualter, J.L.; Kayagaki, N.; Yagita, H.; Bernard, C.C.; Grewal, I.S.; Ashkenazi, A.; Smyth, M.J. TNF-related apoptosis-inducing ligand (TRAIL)/Apo2L suppresses experimental autoimmune encephalomyelitis in mice. Immunol. Cell Biol. 2005, 83, 511–519. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.H.; Chen, G.H.; Fan, Y.; Van Antwerp, M.; Baker, J.; James, R. Tumor Necrosis Factor-Related Apoptosis-Inducing Ligand Inhibits Experimental Autoimmune Thyroiditis by the Expansion of CD4 +CD25 +Regulatory T Cells. Endocrinology 2009, 150, 2000–2007. [Google Scholar] [CrossRef]
- Hirata, S.; Matsuyoshi, H.; Fukuma, D.; Kurisaki, A.; Uemura, Y.; Nishimura, Y.; Senju, S. Involvement of Regulatory T Cells in the Experimental Autoimmune Encephalomyelitis-Preventive Effect of Dendritic Cells Expressing Myelin Oligodendrocyte Glycoprotein plus TRAIL. J. Immunol. 2007, 178, 918–925. [Google Scholar] [CrossRef] [PubMed]
- Nieda, M.; Nicol, A.; Koezuka, Y.; Kikuchi, A.; Lapteva, N.; Tanaka, Y.; Tokunaga, K.; Suzuki, K.; Kayagaki, N.; Yagita, H.; et al. TRAIL expression by activated human CD4(+)V alpha 24NKT cells induces in vitro and in vivo apoptosis of human acute myeloid leukemia cells. Blood 2001, 97, 2067–2074. [Google Scholar] [CrossRef]
- Nishihori, Y.; Kato, K.; Tanaka, M.; Okamoto, T.; Hagiwara, S.; Araki, N.; Kogawa, K.; Kuribayashi, K.; Nakamura, K.; Niitsu, Y. Interleukin-2 gene transfer potentiates the alpha-galactosylceramide-stimulated antitumor effect by the induction of TRAIL in NKT and NK cells in mouse models of subcutaneous and metastatic carcinoma. Cancer Biol. Ther. 2009, 8, 1763–1770. [Google Scholar] [CrossRef]
- Huang, J.R.; Tsai, Y.C.; Chang, Y.J.; Wu, J.C.; Hung, J.T.; Lin, K.H.; Wong, C.H.; Yu, A.L. α-Galactosylceramide but not phenyl-glycolipids induced NKT cell anergy and IL-33-mediated myeloid-derived suppressor cell accumulation via upregulation of egr2/3. J. Immunol. 2014, 192, 1972–1981. [Google Scholar] [CrossRef]
- Wolf, B.J.; Choi, J.E.; Exley, M.A. Novel Approaches to Exploiting Invariant NKT Cells in Cancer Immunotherapy. Front. Immunol. 2018, 9, 975. [Google Scholar] [CrossRef] [PubMed]
- Kronenberg, M. Toward an understanding of NKT cell biology: Progress and paradoxes. Annu. Rev. Immunol. 2005, 23, 877–900. [Google Scholar] [CrossRef] [PubMed]
- Nicol, A.; Nieda, M.; Koezuka, Y.; Porcelli, S.; Suzuki, K.; Tadokoro, K.; Durrant, S.; Juji, T. Human invariant valpha24+ natural killer T cells activated by alpha-galactosylceramide (KRN7000) have cytotoxic anti-tumour activity through mechanisms distinct from T cells and natural killer cells. Immunology 2000, 99, 229–234. [Google Scholar] [CrossRef]
- Kawano, T.; Cui, J.; Koezuka, Y.; Toura, I.; Kaneko, Y.; Sato, H.; Kondo, E.; Harada, M.; Koseki, H.; Nakayama, T.; et al. Natural killer-like nonspecific tumor cell lysis mediated by specific ligand-activated Valpha14 NKT cells. Proc. Natl. Acad. Sci. USA 1998, 95, 5690–5693. [Google Scholar] [CrossRef] [PubMed]
- Mattarollo, S.R.; Kenna, T.; Nieda, M.; Nicol, A.J. Chemotherapy pretreatment sensitizes solid tumor-derived cell lines to Vα24+ NKT cell-mediated cytotoxicity. Int. J. Cancer 2006, 119, 1630–1637. [Google Scholar] [CrossRef]
- Wingender, G.; Krebs, P.; Beutler, B.; Kronenberg, M. Antigen-Specific Cytotoxicity by Invariant NKT Cells In Vivo Is CD95/CD178-Dependent and Is Correlated with Antigenic Potency. J. Immunol. 2010, 185, 2721–2729. [Google Scholar] [CrossRef] [PubMed]
- Metelitsa, L.S.; Weinberg, K.I.; Emanuel, P.D.; Seeger, R.C. Expression of CD1d by myelomonocytic leukemias provides a target for cytotoxic NKT cells. Leukemia 2003, 17, 1068–1077. [Google Scholar] [CrossRef]
- Cretney, E.; Takeda, K.; Yagita, H.; Glaccum, M.; Peschon, J.J.; Smyth, M.J. Increased Susceptibility to Tumor Initiation and Metastasis in TNF-Related Apoptosis-Inducing Ligand-Deficient Mice. J. Immunol. 2002, 168, 1356–1361. [Google Scholar] [CrossRef]
- Teng, M.W.L.; Westwood, J.A.; Darcy, P.K.; Sharkey, J.; Tsuji, M.; Franck, R.W.; Porcelli, S.A.; Besra, G.S.; Takeda, K.; Yagita, H.; et al. Combined Natural Killer T-Cell Based Immunotherapy Eradicates Established Tumors in Mice. Cancer Res. 2007, 67, 7495–7504. [Google Scholar] [CrossRef]
- Dieli, F.; Vermijlen, D.; Fulfaro, F.; Caccamo, N.; Meraviglia, S.; Cicero, G.; Roberts, A.; Buccheri, S.; D’Asaro, M.; Gebbia, N.; et al. Targeting Human γδ T Cells with Zoledronate and Interleukin-2 for Immunotherapy of Hormone-Refractory Prostate Cancer. Cancer Res. 2007, 67, 7450–7457. [Google Scholar] [CrossRef]
- D’Asaro, M.; La Mendola, C.; Di Liberto, D.; Orlando, V.; Todaro, M.; Spina, M.; Guggino, G.; Meraviglia, S.; Caccamo, N.; Messina, A.; et al. Vgamma9 Vdelta2 T lymphocytes efficiently recognize and kill zoledronate-sensitized, imatinib-sensitive, and imatinib-resistant chronic myelogenous leukemia cells. J. Immunol. 2010, 184, 3260–3268. [Google Scholar] [CrossRef] [PubMed]
- Dokouhaki, P.; Han, M.; Joe, B.; Li, M.; Johnston, M.R.; Tsao, M.S.; Zhang, L. Adoptive immunotherapy of cancer using ex vivo expanded human gammadelta T cells: A new approach. Cancer Lett. 2010, 297, 126–136. [Google Scholar] [CrossRef] [PubMed]
- Todaro, M.; D’Asaro, M.; Caccamo, N.; Iovino, F.; Francipane, M.G.; Meraviglia, S.; Orlando, V.; La Mendola, C.; Gulotta, G.; Salerno, A.; et al. Efficient killing of human colon cancer stem cells by gammadelta T lymphocytes. J. Immunol. 2009, 182, 7287–7296. [Google Scholar] [CrossRef] [PubMed]
- Meraviglia, S.; Eberl, M.; Vermijlen, D.; Todaro, M.; Buccheri, S.; Cicero, G.; La Mendola, C.; Guggino, G.; D’Asaro, M.; Orlando, V.; et al. In vivo manipulation of Vgamma9 Vdelta2 T cells with zoledronate and low-dose interleukin-2 for immunotherapy of advanced breast cancer patients. Clin. Exp. Immunol. 2010, 161, 290–297. [Google Scholar] [CrossRef] [PubMed]
- Dokouhaki, P.; Schuh, N.W.; Joe, B.; Allen, C.A.D.; Der, S.D.; Tsao, M.S.; Zhang, L. NKG2D regulates production of soluble TRAIL by ex vivo expanded human γδ T cells. Eur. J. Immunol. 2013, 43, 3175–3182. [Google Scholar] [CrossRef] [PubMed]
- Todaro, M.; Orlando, V.; Cicero, G.; Caccamo, N.; Meraviglia, S.; Stassi, G.; Dieli, F. Chemotherapy Sensitizes Colon Cancer Initiating Cells to Vγ9Vδ2 T Cell-Mediated Cytotoxicity. PLoS ONE 2013, 8, e65145. [Google Scholar] [CrossRef]
- Caccamo, N.; La Mendola, C.; Orlando, V.; Meraviglia, S.; Todaro, M.; Stassi, G.; Sireci, G.; Fournie, J.J.; Dieli, F. Differentiation, phenotype, and function of interleukin-17-producing human Vγ9Vδ2 T cells. Blood 2011, 118, 129–138. [Google Scholar] [CrossRef] [PubMed]
- Secchiero, P.; Tiribelli, M.; Barbarotto, E.; Celeghini, C.; Michelutti, A.; Masolini, P.; Fanin, R.; Zauli, G. Aberrant expression of TRAIL in B chronic lymphocytic leukemia (B-CLL) cells. J. Cell. Physiol. 2005, 205, 246–252. [Google Scholar] [CrossRef]
- Staniek, J.; Lorenzetti, R.; Heller, B.; Janowska, I.; Schneider, P.; Unger, S.; Warnatz, K.; Seidl, M.; Venhoff, N.; Thiel, J.; et al. TRAIL-R1 and TRAIL-R2 Mediate TRAIL-Dependent Apoptosis in Activated Primary Human B Lymphocytes. Front. Immunol. 2019, 10, 951. [Google Scholar] [CrossRef]
- Guerreiro-Cacais, A.O.; Levitskaya, J.; Levitsky, V. B cell receptor triggering sensitizes human B cells to TRAIL-induced apoptosis. J. Leukoc. Biol. 2010, 88, 937–945. [Google Scholar] [CrossRef]
- Crowder, R.N.; Zhao, H.; Chatham, W.W.; Zhou, T.; Carter, R.H. B lymphocytes are resistant to death receptor 5-induced apoptosis. Clin. Immunol. 2011, 139, 21–31. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Smulski, C.R.; Decossas, M.; Chekkat, N.; Beyrath, J.; Willen, L.; Guichard, G.; Lorenzetti, R.; Rizzi, M.; Eibel, H.; Schneider, P.; et al. Hetero-oligomerization between the TNF receptor superfamily members CD40, Fas and TRAILR2 modulate CD40 signalling. Nat. Publ. Group 2017, 8, e2601. [Google Scholar] [CrossRef] [PubMed]
- Ursini-Siegel, J.; Zhang, W.; Altmeyer, A.; Hatada, E.N.; Do, R.K.G.; Yagita, H.; Chen-Kiang, S. TRAIL/Apo-2 Ligand Induces Primary Plasma Cell Apoptosis. J. Immunol. 2002, 169, 5505–5513. [Google Scholar] [CrossRef] [PubMed]
- Travert, M.; Ame-Thomas, P.; Pangault, C.; Morizot, A.; Micheau, O.; Semana, G.; Lamy, T.; Fest, T.; Tarte, K.; Guillaudeux, T. CD40 Ligand Protects from TRAIL-Induced Apoptosis in Follicular Lymphomas through NF-κB Activation and Up-Regulation of c-FLIP and Bcl-xL. J. Immunol. 2008, 181, 1001–1011. [Google Scholar] [CrossRef] [PubMed]
- Griebel, P.; Beskorwayne, T.; Van den Broeke, A.; Ferrari, G. CD40 signaling induces B cell responsiveness to multiple members of the gamma chain-common cytokine family. Int. Immunol. 1999, 11, 1139–1147. [Google Scholar] [CrossRef]
- van Grevenynghe, J.; Cubas, R.A.; Noto, A.; DaFonseca, S.; He, Z.; Peretz, Y.; Filali-Mouhim, A.; Dupuy, F.P.; Procopio, F.A.; Chomont, N.; et al. Loss of memory B cells during chronic HIV infection is driven by Foxo3a- and TRAIL-mediated apoptosis. J. Clin. Investig. 2011, 121, 3877–3888. [Google Scholar] [CrossRef] [PubMed]
- Kayagaki, N.; Yamaguchi, N.; Abe, M.; Hirose, S.; Shirai, T.; Okumura, K.; Yagita, H. Suppression of antibody production by TNF-related apoptosis-inducing ligand (TRAIL). Cell. Immunol. 2002, 219, 82–91. [Google Scholar] [CrossRef]
- Schneider, P.; Holler, N.; Bodmer, J.L.; Hahne, M.; Frei, K.; Fontana, A.; Tschopp, J. Conversion of Membrane-bound Fas(CD95) Ligand to Its Soluble Form Is Associated with Downregulation of Its Proapoptotic Activity and Loss of Liver Toxicity. J. Exp. Med. 1998, 187, 1205–1213. [Google Scholar] [CrossRef]
- Wajant, H.; Moosmayer, D.; Wüest, T.; Bartke, T.; Gerlach, E.; Schönherr, U.; Peters, N.; Scheurich, P.; Pfizenmaier, K. Differential activation of TRAIL-R1 and -2 by soluble and membrane TRAIL allows selective surface antigen-directed activation of TRAIL-R2 by a soluble TRAIL derivative. Oncogene 2001, 20, 4101–4106. [Google Scholar] [CrossRef]
- Clancy, L.; Mruk, K.; Archer, K.; Woelfel, M.; Mongkolsapaya, J.; Screaton, G.; Lenardo, M.J.; Chan, F.K.M. Preligand assembly domain-mediated ligand-independent association between TRAIL receptor 4 (TR4) and TR2 regulates TRAIL-induced apoptosis. Proc. Natl. Acad. Sci. USA 2005, 102, 18099–18104. [Google Scholar] [CrossRef]
- Truneh, A.; Sharma, S.; Silverman, C.; Khandekar, S.; Reddy, M.P.; Deen, K.C.; Mclaughlin, M.M.; Srinivasula, S.M.; Livi, G.P.; Marshall, L.A.; et al. Temperature-sensitive Differential Affinity of TRAIL for Its Receptors. J. Biol. Chem. 2000, 275, 23319–23325. [Google Scholar] [CrossRef] [PubMed]
- Martinez-Lorenzo, M.J.; Anel, A.; Gamen, S.; Monle n, I.; Lasierra, P.; Larrad, L.; Pineiro, A.; Alava, M.A.; Naval, J. Activated human T cells release bioactive Fas ligand and APO2 ligand in microvesicles. J. Immunol. 1999, 163, 1274–1281. [Google Scholar] [PubMed]
- Monleon, I.; Martinez-Lorenzo, M.J.; Monteagudo, L.; Lasierra, P.; Taules, M.; Iturralde, M.; Pineiro, A.; Larrad, L.; Alava, M.A.; Naval, J.; et al. Differential Secretion of Fas Ligand- or APO2 Ligand/TNF-Related Apoptosis-Inducing Ligand-Carrying Microvesicles During Activation-Induced Death of Human T Cells. J. Immunol. 2001, 167, 6736–6744. [Google Scholar] [CrossRef] [PubMed]
- Bosque, A.; Dietz, L.; Gallego-Lleyda, A.; Sanclemente, M.; Iturralde, M.; Naval, J.; Alava, M.A.; Martinez-Lostao, L.; Thierse, H.J.; Anel, A. Comparative proteomics of exosomes secreted by tumoral Jurkat T cells and normal human T cell blasts unravels a potential tumorigenic role for valosin-containing protein. Oncotarget 2016, 7, 29287–29305. [Google Scholar] [CrossRef] [PubMed]
- Stenqvist, A.C.; Nagaeva, O.; Baranov, V.; Mincheva-Nilsson, L. Exosomes Secreted by Human Placenta Carry Functional Fas Ligand and TRAIL Molecules and Convey Apoptosis in Activated Immune Cells, Suggesting Exosome-Mediated Immune Privilege of the Fetus. J. Immunol. 2013, 191, 5515–5523. [Google Scholar] [CrossRef]
- Munich, S.; Sobo-Vujanovic, A.; Buchser, W.J.; Beer-Stolz, D.; Vujanovic, N.L. Dendritic cell exosomes directly kill tumor cells and activate natural killer cells via TNF superfamily ligands. Oncoimmunology 2014, 1, 1074–1083. [Google Scholar] [CrossRef]
- Huber, V.; Fais, S.; Iero, M.; Lugini, L.; Canese, P.; Squarcina, P.; Zaccheddu, A.; Colone, M.; Arancia, G.; Gentile, M.; et al. Human Colorectal Cancer Cells Induce T-Cell Death Through Release of Proapoptotic Microvesicles: Role in Immune Escape. Gastroenterology 2005, 128, 1796–1804. [Google Scholar] [CrossRef]
- Martínez-Lorenzo, M.J.; Anel, A.; Alava, M.A.; Piñeiro, A.; Naval, J.; Lasierra, P.; Larrad, L. The human melanoma cell line MelJuSo secretes bioactive FasL and APO2L/TRAIL on the surface of microvesicles. Possible contribution to tumor counterattack. Exp. Cell Res. 2004, 295, 315–329. [Google Scholar] [CrossRef]
- Lo Cicero, A.; Schiera, G.; Proia, P.; Saladino, P.; Savettieri, G.; Di Liegro, C.M.; Di Liegro, I. Oligodendroglioma cells shed microvesicles which contain TRAIL as well as molecular chaperones and induce cell death in astrocytes. Int. J. Oncol. 2011, 39, 1353–1357. [Google Scholar] [CrossRef]
- Wan, C.; Fu, J.; Wang, Y.; Miao, S.; Song, W.; Wang, L. Exosome-related multi-pass transmembrane protein TSAP6 is a target of rhomboid protease RHBDD1-induced proteolysis. PLoS ONE 2012, 7, e37452. [Google Scholar] [CrossRef]
- Martínez-Lorenzo, M.J.; Anel, A.; Saez-Gutierrez, B.; Royo-Cañas, M.; Bosque, A.; Alava, M.A.; Piñeiro, A.; Lasierra, P.; Asín-Ungría, J.; Larrad, L. Rheumatoid synovial fluid T cells are sensitive to APO2L/TRAIL. Clin. Immunol. 2007, 122, 28–40. [Google Scholar] [CrossRef] [PubMed]
- Thayanithy, V.; O’Hare, P.; Wong, P.; Zhao, X.; Steer, C.J.; Subramanian, S.; Lou, E. A transwell assay that excludes exosomes for assessment of tunneling nanotube-mediated intercellular communication. Cell Commun. Signal. 2017, 15, 46. [Google Scholar] [CrossRef] [PubMed]
- Munoz, J.L.; Bliss, S.A.; Greco, S.J.; Ramkissoon, S.H.; Ligon, K.L.; Rameshwar, P. Delivery of Functional Anti-miR-9 by Mesenchymal Stem Cell-derived Exosomes to Glioblastoma Multiforme Cells Conferred Chemosensitivity. Mol. Ther. Nucleic Acids 2013, 2, e126. [Google Scholar] [CrossRef] [PubMed]
- Robbins, P.D.; Morelli, A.E. Regulation of immune responses by extracellular vesicles. Nat. Publ. Group 2014, 14, 195–208. [Google Scholar] [CrossRef] [PubMed]
- Anel, A.; Gallego-Lleyda, A.; de Miguel, D.; Naval, J.; Martinez-Lostao, L. Role of Exosomes in the Regulation of T-cell Mediated Immune Responses and in Autoimmune Disease. Cells 2019, 8, 154. [Google Scholar] [CrossRef] [PubMed]
- Bosque, A.; Pardo, J.; Martínez-Lorenzo, M.J.; Iturralde, M.; Marzo, I.; Piñeiro, A.; Alava, M.A.; Naval, J.; Anel, A. Down-regulation of normal human T cell blast activation: Roles of APO2L/TRAIL, FasL, and c- FLIP, Bim, or Bcl-x isoform expression. J. Leukoc. Biol. 2005, 77, 568–578. [Google Scholar] [CrossRef]
- Grootjans, J.; Kaser, A.; Kaufman, R.J.; Blumberg, R.S. The unfolded protein response in immunity and inflammation. Nat. Publ. Group 2016, 16, 469–484. [Google Scholar] [CrossRef]
- Kemp, K.; Poe, C. Stressed: The Unfolded Protein Response in T Cell Development, Activation, and Function. Int. J. Mol. Sci. 2019, 20, 1792. [Google Scholar] [CrossRef]
- Janssens, S.; Pulendran, B.; Lambrecht, B.N. Emerging functions of the unfolded protein response in immunity. Nat. Publ. Group 2014, 15, 910–919. [Google Scholar] [CrossRef]
- Smith, J.A. Regulation of Cytokine Production by the Unfolded Protein Response; Implications for Infection and Autoimmunity. Front. Immunol. 2018, 9, 6. [Google Scholar] [CrossRef]
- Jiang, Y.; Chen, X.; Fan, M.; Li, H.; Zhu, W.; Chen, X.; Cao, C.; Xu, R.; Wang, Y.; Ma, Y. TRAIL facilitates cytokine expression and macrophage migration during hypoxia/reoxygenation via ER stress-dependent NF-κB pathway. Mol. Immunol. 2017, 82, 123–136. [Google Scholar] [CrossRef] [PubMed]
- Chattergoon, M.A.; Muthumani, K.; Tamura, Y.; Ramanathan, M.; Shames, J.P.; Saulino, V.; Robinson, T.M.; Montaner, L.J.; Weiner, D.B. DR5 activation of caspase-8 induces DC maturation and immune enhancement in vivo. Mol. Ther. 2008, 16, 419–426. [Google Scholar] [CrossRef] [PubMed]
- Brincks, E.L.; Katewa, A.; Kucaba, T.A.; Griffith, T.S.; Legge, K.L. CD8 T Cells Utilize TRAIL to Control Influenza Virus Infection. J. Immunol. 2008, 181, 4918–4925. [Google Scholar] [CrossRef] [PubMed]
- Lamhamedi-Cherradi, S.E.; Zheng, S.J.; Maguschak, K.A.; Peschon, J.; Chen, Y.H. Defective thymocyte apoptosis and accelerated autoimmune diseases in TRAIL−/− mice. Nat. Publ. Group 2003, 4, 255–260. [Google Scholar] [CrossRef] [PubMed]
- Lamhamedi-Cherradi, S.E.; Zheng, S.; Tisch, R.M.; Chen, Y.H. Critical roles of tumor necrosis factor-related apoptosis-inducing ligand in type 1 diabetes. Diabetes 2003, 52, 2274–2278. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Liu, Z.; Xu, X.; Hsu, H.C.; Tousson, A.; Yang, P.A.; Wu, Q.; Liu, C.; Yu, S.; Zhang, H.G.; Mountz, J.D. CII-DC-AdTRAIL cell gene therapy inhibits infiltration of CII-reactive T cells and CII-induced arthritis. J. Clin. Investig. 2003, 112, 1332–1341. [Google Scholar] [CrossRef] [PubMed]
- Jin, C.H.; Chae, S.Y.; Kim, T.H.; Yang, H.K.; Lee, E.Y.; Song, Y.W.; Jo, D.G.; Lee, K.C. Effect of Tumor Necrosis Factor-Related Apoptosis-Inducing Ligand on the Reduction of Joint Inflammation in Experimental Rheumatoid Arthritis. J. Pharmacol. Exp. Ther. 2010, 332, 858–865. [Google Scholar] [CrossRef] [PubMed]
- Mi, Q.S.; Ly, D.; Lamhamedi-Cherradi, S.E.; Salojin, K.V.; Zhou, L.; Grattan, M.; Meagher, C.; Zucker, P.; Chen, Y.H.; Nagle, J.; et al. Blockade of tumor necrosis factor-related apoptosis-inducing ligand exacerbates type 1 diabetes in NOD mice. Diabetes 2003, 52, 1967–1975. [Google Scholar] [CrossRef] [PubMed]
- Hirata, S.; Senju, S.; Matsuyoshi, H.; Fukuma, D.; Uemura, Y.; Nishimura, Y. Prevention of experimental autoimmune encephalomyelitis by transfer of embryonic stem cell-derived dendritic cells expressing myelin oligodendrocyte glycoprotein peptide along with TRAIL or programmed death-1 ligand. J. Immunol. 2005, 174, 1888–1897. [Google Scholar] [CrossRef]
- Aktas, O.; Smorodchenko, A.; Brocke, S.; Infante-Duarte, C.; Topphoff, U.S.; Vogt, J.; Prozorovski, T.; Meier, S.; Osmanova, V.; Pohl, E.; et al. Neuronal Damage in Autoimmune Neuroinflammation Mediated by the Death Ligand TRAIL. Neuron 2005, 46, 421–432. [Google Scholar] [CrossRef] [PubMed]
- Hilliard, B.; Wilmen, A.; Seidel, C.; Liu, T.S.T.; Goke, R.; Chen, Y. Roles of TNF-Related Apoptosis-Inducing Ligand in Experimental Autoimmune Encephalomyelitis. J. Immunol. 2001, 166, 1314–1319. [Google Scholar] [CrossRef] [PubMed]
- Takeda, K.; Smyth, M.J.; Cretney, E.; Hayakawa, Y.; Yamaguchi, N.; Yagita, H.; Okumura, K. Involvement of Tumor Necrosis Factor-Related Apoptosis-Inducing Ligand in NK Cell-Mediated and IFN-γ-Dependent Suppression of Subcutaneous Tumor Growth. Cell. Immunol. 2001, 214, 194–200. [Google Scholar] [CrossRef] [PubMed]
- Thomas, W.D.; Hersey, P. TNF-related apoptosis-inducing ligand (TRAIL) induces apoptosis in Fas ligand-resistant melanoma cells and mediates CD4 T cell killing of target cells. J. Immunol. 1998, 161, 2195–2200. [Google Scholar] [PubMed]
- van der Most, R.G.; Currie, A.J.; Cleaver, A.L.; Salmons, J.; Nowak, A.K.; Mahendran, S.; Larma, I.; Prosser, A.; Robinson, B.W.S.; Smyth, M.J.; et al. Cyclophosphamide Chemotherapy Sensitizes Tumor Cells to TRAIL-Dependent CD8 T Cell-Mediated Immune Attack Resulting in Suppression of Tumor Growth. PLoS ONE 2009, 4, e6982. [Google Scholar] [CrossRef] [PubMed]
- Wang, R.; Zhang, L.; Zhang, X.; Moreno, J.; Luo, X.; Tondravi, M.; Shi, Y. Differential Regulation of the Expression of CD95 Ligand, Receptor Activator of Nuclear Factor- B Ligand (RANKL), TNF-Related Apoptosis-Inducing Ligand (TRAIL), and TNF- During T Cell Activation. J. Immunol. 2001, 166, 1983–1990. [Google Scholar] [CrossRef] [PubMed]
- Dörr, J.; Waiczies, S.; Wendling, U.; Seeger, B.; Zipp, F. Induction of TRAIL-mediated glioma cell death by human T cells. J. Neuroimmunol. 2002, 122, 117–124. [Google Scholar] [CrossRef]
- Zerafa, N.; Westwood, J.A.; Cretney, E.; Mitchell, S.; Waring, P.; Iezzi, M.; Smyth, M.J. Cutting Edge: TRAIL Deficiency Accelerates Hematological Malignancies. J. Immunol. 2005, 175, 5586–5590. [Google Scholar] [CrossRef]
- Seki, N.; Hayakawa, Y.; Brooks, A.D.; Wine, J.; Wiltrout, R.H.; Yagita, H.; Tanner, J.E.; Smyth, M.J.; Sayers, T.J. Tumor necrosis factor-related apoptosis-inducing ligand-mediated apoptosis is an important endogenous mechanism for resistance to liver metastases in murine renal cancer. Cancer Res. 2003, 63, 207–213. [Google Scholar]
- Takeda, K.; Smyth, M.J.; Cretney, E.; Hayakawa, Y.; Kayagaki, N.; Yagita, H.; Okumura, K. Critical role for tumor necrosis factor-related apoptosis-inducing ligand in immune surveillance against tumor development. J. Exp. Med. 2002, 195, 161–169. [Google Scholar] [CrossRef]
- Smyth, M.J.; Thia, K.Y.; Street, S.E.; Cretney, E.; Trapani, J.A.; Taniguchi, M.; Kawano, T.; Pelikan, S.B.; Crowe, N.Y.; Godfrey, D.I. Differential tumor surveillance by natural killer (NK) and NKT cells. J. Exp. Med. 2000, 191, 661–668. [Google Scholar] [CrossRef]
- Crowe, N.Y.; Smyth, M.J.; Godfrey, D.I. A Critical Role for Natural Killer T Cells in Immunosurveillance of Methylcholanthrene-induced Sarcomas. J. Exp. Med. 2002, 196, 119–127. [Google Scholar] [CrossRef]
- Smyth, M.J.; Crowe, N.Y.; Godfrey, D.I. NK cells and NKT cells collaborate in host protection from methylcholanthrene-induced fibrosarcoma. Int. Immunol. 2001, 13, 459–463. [Google Scholar] [CrossRef] [PubMed]
- Spencer, S.L.; Gaudet, S.; Albeck, J.G.; Burke, J.M.; Sorger, P.K. Non-genetic origins of cell-to-cell variability in TRAIL-induced apoptosis. Nature 2009, 459, 428–432. [Google Scholar] [CrossRef] [PubMed]
- Tauzin, S.; Debure, L.; Moreau, J.F.; Legembre, P. CD95-mediated cell signaling in cancer: Mutations and post-translational modulations. Cell. Mol. Life Sci. 2011, 69, 1261–1277. [Google Scholar] [CrossRef]
- Rooney, M.S.; Shukla, S.A.; Wu, C.J.; Getz, G.; Hacohen, N. Molecular and Genetic Properties of Tumors Associated with Local Immune Cytolytic Activity. Cell 2015, 160, 48–61. [Google Scholar] [CrossRef]
- Hopkins-Donaldson, S.; Ziegler, A.; Kurtz, S.; Bigosch, C.; Kandioler, D.; Ludwig, C.; Zangemeister-Wittke, U.; Stahel, R. Silencing of death receptor and caspase-8 expression in small cell lung carcinoma cell lines and tumors by DNA methylation. Cell Death Differ. 2003, 10, 356–364. [Google Scholar] [CrossRef] [PubMed]
- Tian, F.; Lu, J.J.; Wang, L.; Li, L.; Yang, J.; Li, Y.; Liu, Y.Q.; Shen, G.X.; Tu, Y.T.; Tao, J. Expression of c-FLIP in malignant melanoma, and its relationship with the clinicopathological features of the disease. Clin. Exp. Dermatol. 2011, 37, 259–265. [Google Scholar] [CrossRef]
- McCourt, C.; Maxwell, P.; Mazzucchelli, R.; Montironi, R.; Scarpelli, M.; Salto-Tellez, M.; O’Sullivan, J.M.; Longley, D.B.; Waugh, D.J.J. Elevation of c-FLIP in Castrate-Resistant Prostate Cancer Antagonizes Therapeutic Response to Androgen Receptor-Targeted Therapy. Clin. Cancer Res. 2012, 18, 3822–3833. [Google Scholar] [CrossRef]
- Moriwaki, K.; Noda, K.; Furukawa, Y.; Ohshima, K.; Uchiyama, A.; Nakagawa, T.; Taniguchi, N.; Daigo, Y.; Nakamura, Y.; Hayashi, N.; et al. Deficiency of GMDS Leads to Escape from NK Cell-Mediated Tumor Surveillance Through Modulation of TRAIL Signaling. Gastroenterology 2009, 137, 188–198. [Google Scholar] [CrossRef] [PubMed]
- Zhang, B.; Roosmalen, I.A.M.; Reis, C.R.; Setroikromo, R.; Quax, W.J. Death receptor 5 is activated by fucosylation in colon cancer cells. FEBS J. 2018, 286, 555–571. [Google Scholar] [CrossRef] [PubMed]
- Micheau, O. Regulation of TNF-Related Apoptosis-Inducing Ligand Signaling by Glycosylation. Inter. J. Mol. Sci. 2018, 19, 715. [Google Scholar] [CrossRef] [PubMed]
- Dufour, F.; Rattier, T.; Shirley, S.; Picarda, G.; Constantinescu, A.A.; Morle, A.; Zakaria, A.B.; Marcion, G.; Causse, S.; Szegezdi, E.; et al. N-glycosylation of mouse TRAIL-R and human TRAIL-R1 enhances TRAIL-induced death. Cell Death Differ 2017, 24, 500–510. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; QI, X.; Wu, M. Effect of DR4 promoter methylation on the TRAIL-induced apoptosis in lung squamous carcinoma cell. Oncol. Rep. 2015, 34, 2115–2125. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Kriegl, L.; Jung, A.; Engel, J.; Jackstadt, R.; Gerbes, A.L.; Gallmeier, E.; Reiche, J.A.; Hermeking, H.; Rizzani, A.; Bruns, C.J.; et al. Expression, Cellular Distribution, and Prognostic Relevance of TRAIL Receptors in Hepatocellular Carcinoma. Clin. Cancer Res. 2010, 16, 5529–5538. [Google Scholar] [CrossRef]
- Twomey, J.; Zhang, B. Circulating Tumor Cells Develop Resistance to TRAIL-Induced Apoptosis Through Autophagic Removal of Death Receptor 5: Evidence from an In Vitro Model. Cancers 2019, 11, 94. [Google Scholar] [CrossRef]
- Haselmann, V.; Kurz, A.; Bertsch, U.; Hübner, S.; Müller, M.O.; Fritsch, J.; Häsler, R.; Pickl, A.; Fritsche, H.; Annewanter, F.; et al. Nuclear Death Receptor TRAIL-R2 Inhibits Maturation of Let-7 and Promotes Proliferation of Pancreatic and Other Tumor Cells. Gastroenterology 2014, 146, 278–290. [Google Scholar] [CrossRef]
- Ganten, T.M.; Sykora, J.; Koschny, R.; Batke, E.; Aulmann, S.; Mansmann, U.; Stremmel, W.; Sinn, H.P.; Walczak, H. Prognostic significance of tumour necrosis factor-related apoptosis-inducing ligand (TRAIL) receptor expression in patients with breast cancer. J. Mol. Med. 2009, 87, 995–1007. [Google Scholar] [CrossRef]
- Koksal, I.T.; Sanlioglu, A.D.; Karacay, B.; Griffith, T.S.; Sanlioglu, S. Tumor necrosis factor-related apoptosis inducing ligand-R4 decoy receptor expression is correlated with high Gleason scores, prostate-specific antigen recurrence, and decreased survival in patients with prostate carcinoma. Urol. Oncol. Semin. Orig. Investig. 2008, 26, 158–165. [Google Scholar] [CrossRef]
- Riccioni, R.; Pasquini, L.; Mariani, G.; Saulle, E.; Rossini, A.; Diverio, D.; Pelosi, E.; Vitale, A.; Chierichini, A.; Cedrone, M.; et al. TRAIL decoy receptors mediate resistance of acute myeloid leukemia cells to TRAIL. Haematologica 2005, 90, 612–624. [Google Scholar]
- Chamuleau, M.E.D.; Ossenkoppele, G.J.; van Rhenen, A.; van Dreunen, L.; Jirka, S.M.G.; Zevenbergen, A.; Schuurhuis, G.J.; van de Loosdrecht, A.A. High TRAIL-R3 expression on leukemic blasts is associated with poor outcome and induces apoptosis-resistance which can be overcome by targeting TRAIL-R2. Leuk. Res. 2011, 35, 741–749. [Google Scholar] [CrossRef]
- Macher-Goeppinger, S.; Aulmann, S.; Tagscherer, K.E.; Wagener, N.; Haferkamp, A.; Penzel, R.; Brauckhoff, A.; Hohenfellner, M.; Sykora, J.; Walczak, H.; et al. Prognostic Value of Tumor Necrosis Factor-Related Apoptosis-Inducing Ligand (TRAIL) and TRAIL Receptors in Renal Cell Cancer. Clin. Cancer Res. 2009, 15, 650–659. [Google Scholar] [CrossRef] [PubMed]
- Imaoka, Y.; Ohira, M.; Yano, T.; Nakano, R.; Tanimine, N.; Shimizu, S.; Kuroda, S.; Tahara, H.; Kobayashi, T.; Ohdan, H. Polymorphisms in TRAIL predict long-term survival and extrahepatic recurrence following initial hepatectomy for hepatocellular carcinoma. J. Hepat. Biliary Pancreat. Sci. 2018, 25, 370–376. [Google Scholar] [CrossRef] [PubMed]
- Ehrhardt, H.; Fulda, S.; Schmid, I.; Hiscott, J.; Debatin, K.M.; Jeremias, I. TRAIL induced survival and proliferation in cancer cells resistant towards TRAIL-induced apoptosis mediated by NF-κB. Oncogene 2003, 22, 3842–3852. [Google Scholar] [CrossRef] [PubMed]
- Ishimura, N.; Isomoto, H.; Bronk, S.F.; Gores, G.J. Trail induces cell migration and invasion in apoptosis-resistant cholangiocarcinoma cells. Am. J. Physiol. Gastroint. Liver Physiol. 2006, 290, G129–G136. [Google Scholar] [CrossRef] [PubMed]
- von Karstedt, S.; Conti, A.; Nobis, M.; Montinaro, A.; Hartwig, T.; Lemke, J.; Legler, K.; Annewanter, F.; Campbell, A.D.; Taraborrelli, L.; et al. Cancer Cell-Autonomous TRAIL-R Signaling Promotes KRAS-Driven Cancer Progression, Invasion, and Metastasis. Cancer Cell 2015, 27, 561–573. [Google Scholar] [CrossRef] [PubMed]
- Trauzold, A.; Siegmund, D.; Schniewind, B.; Sipos, B.; Egberts, J.; Zorenkov, D.; Emme, D.; Röder, C.; Kalthoff, H.; Wajant, H. TRAIL promotes metastasis of human pancreatic ductal adenocarcinoma. Oncogene 2006, 25, 7434–7439. [Google Scholar] [CrossRef] [PubMed]
- Dufour, F.; Rattier, T.; Constantinescu, A.A.; Zischler, L.; Morlé, A.; Ben Mabrouk, H.; Humblin, E.; Jacquemin, G.; Szegezdi, E.; Delacote, F.; et al. TRAIL receptor gene editing unveils TRAIL-R1 as a master player of apoptosis induced by TRAIL and ER stress. Oncotarget 2017, 8, 9974–9985. [Google Scholar] [CrossRef] [PubMed]
- Tang, W.; Wang, W.; Zhang, Y.; Liu, S.; Liu, Y.; Zheng, D. TRAIL receptor mediates inflammatory cytokine release in an NF-κB-dependent manner. Cell Res. 2009, 19, 758–767. [Google Scholar] [CrossRef] [PubMed]
- Varfolomeev, E.; Maecker, H.; Sharp, D.; Lawrence, D.; Renz, M.; Vucic, D.; Ashkenazi, A. Molecular determinants of kinase pathway activation by Apo2 ligand/tumor necrosis factor-related apoptosis-inducing ligand. J. Biol. Chem. 2005, 280, 40599–40608. [Google Scholar] [CrossRef] [PubMed]
- Henry, C.M.; Martin, S.J. Caspase-8 Acts in a Non-enzymatic Role as a Scaffold for Assembly of a Pro-inflammatory “FADDosome“ Complex upon TRAIL Stimulation. Mol. Cell 2017, 65, 715–729. [Google Scholar] [CrossRef]
- Choi, C.; Kutsch, O.; Park, J.; Zhou, T.; Seol, D.W.; Benveniste, E.N. Tumor necrosis factor-related apoptosis-inducing ligand induces caspase-dependent interleukin-8 expression and apoptosis in human astroglioma cells. Mol. Cell. Biol. 2002, 22, 724–736. [Google Scholar] [CrossRef] [PubMed]
- Tuccitto, A.; Shahaj, E.; Vergani, E.; Ferro, S.; Huber, V.; Rodolfo, M.; Castelli, C.; Rivoltini, L.; Vallacchi, V. Immunosuppressive circuits in tumor microenvironment and their influence on cancer treatment efficacy. Virchows Arch. Int. J. Pathol. 2019, 474, 407–420. [Google Scholar] [CrossRef] [PubMed]
- Donnell, J.S.O.x.; Teng, M.W.L.; Smyth, M.J. Cancer immunoediting and resistance to T cell-based immunotherapy. Nat. Rev. Clin. Oncol. 2019, 16, 151–167. [Google Scholar] [CrossRef] [PubMed]
- Domblides, C.; Lartigue, L.; Faustin, B. Control of the Antitumor Immune Response by Cancer Metabolism. Cells 2019, 8, 104. [Google Scholar] [CrossRef] [PubMed]
- Kiaei, M.; Kipiani, K.; Calingasan, N.; Wille, E.; chen, J.; Heissig, B.; Rafii, S.; Lorenzl, S.; Beal, M. Matrix metalloproteinase-9 regulates TNF-α and FasL expression in neuronal, glial cells and its absence extends life in a transgenic mouse model of amyotrophic lateral sclerosis. Exp. Neurol. 2007, 205, 74–81. [Google Scholar] [CrossRef]
- Schulte, M.; Reiss, K.; Lettau, M.; Maretzky, T.; Ludwig, A.; Hartmann, D.; de Strooper, B.; Janssen, O.; Saftig, P. ADAM10 regulates FasL cell surface expression and modulates FasL-induced cytotoxicity and activation-induced cell death. Cell Death Differ. 2007, 14, 1040–1049. [Google Scholar] [CrossRef] [PubMed]
- Guégan, J.P.; Ginestier, C.; Charafe-Jauffret, E.; Ducret, T.; Quignard, J.F.; Vacher, P.; Legembre, P. CD95/Fas and metastatic disease_ What does not kill you makes you stronger. Sem. Cancer Biol. 2019. [Google Scholar] [CrossRef]
- O’Leary, L.; van der Sloot, A.M.; Reis, C.R.; Deegan, S.; Ryan, A.E.; Dhami, S.P.S.; Murillo, L.S.; Cool, R.H.; Correa de Sampaio, P.; Thompson, K.; et al. Decoy receptors block TRAIL sensitivity at a supracellular level: The role of stromal cells in controlling tumour TRAIL sensitivity. Oncogene 2016, 35, 1261–1270. [Google Scholar] [CrossRef]
- Song, M.; Cubillos-Ruiz, J.R. Endoplasmic Reticulum Stress Responses in Intratumoral Immune Cells: Implications for Cancer Immunotherapy. Trends Immunol. 2019, 40, 128–141. [Google Scholar] [CrossRef]
- Wilson, N.S.; Yang, A.; Yang, B.; Couto, S.; Stern, H.; Gogineni, A.; Pitti, R.; Marsters, S.; Weimer, R.M.; Singh, M.; et al. Proapoptotic activation of death receptor 5 on tumor endothelial cells disrupts the vasculature and reduces tumor growth. Cancer Cell 2012, 22, 80–90. [Google Scholar] [CrossRef]
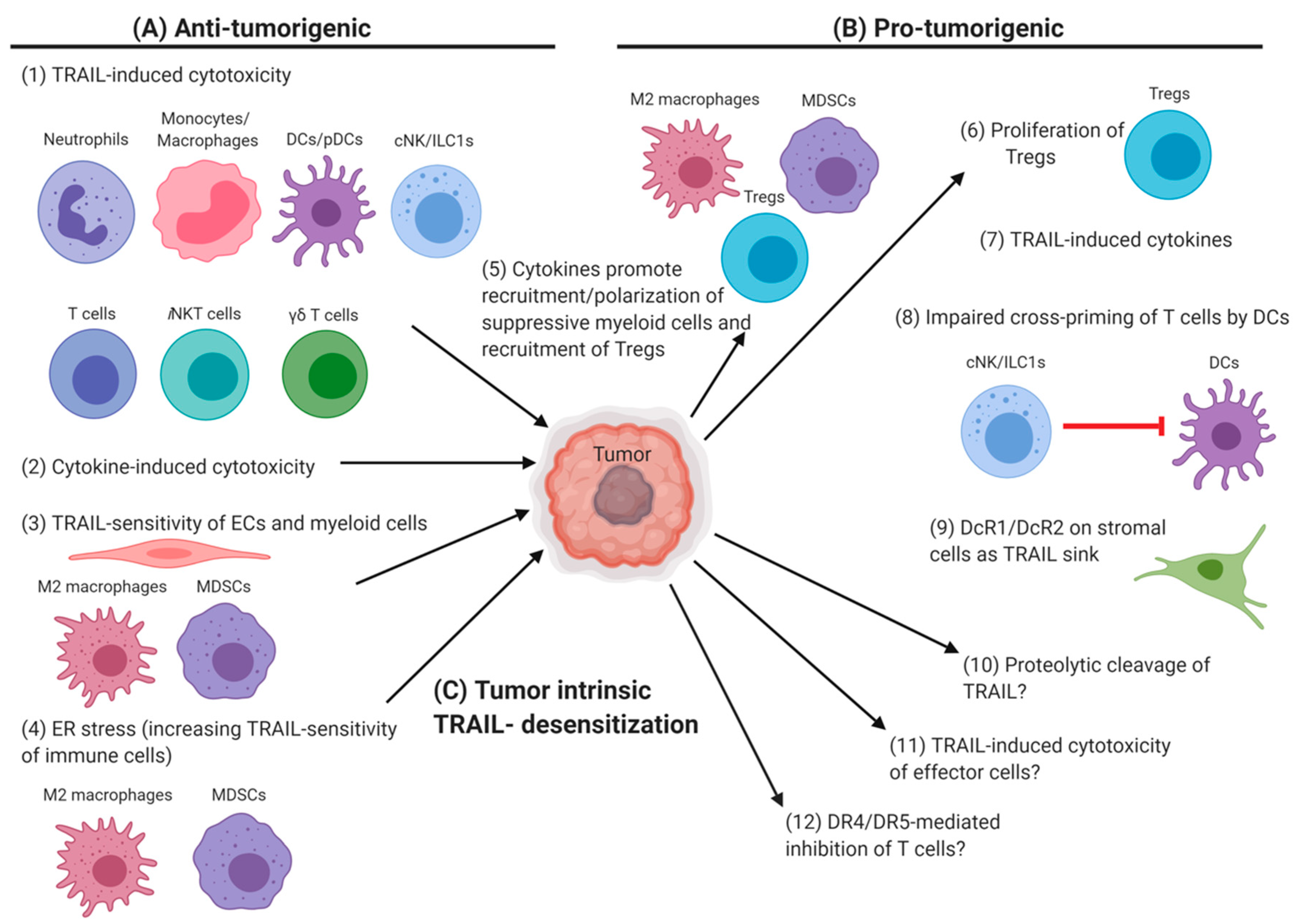
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sag, D.; Ayyildiz, Z.O.; Gunalp, S.; Wingender, G. The Role of TRAIL/DRs in the Modulation of Immune Cells and Responses. Cancers 2019, 11, 1469. https://doi.org/10.3390/cancers11101469
Sag D, Ayyildiz ZO, Gunalp S, Wingender G. The Role of TRAIL/DRs in the Modulation of Immune Cells and Responses. Cancers. 2019; 11(10):1469. https://doi.org/10.3390/cancers11101469
Chicago/Turabian StyleSag, Duygu, Zeynep Ozge Ayyildiz, Sinem Gunalp, and Gerhard Wingender. 2019. "The Role of TRAIL/DRs in the Modulation of Immune Cells and Responses" Cancers 11, no. 10: 1469. https://doi.org/10.3390/cancers11101469
APA StyleSag, D., Ayyildiz, Z. O., Gunalp, S., & Wingender, G. (2019). The Role of TRAIL/DRs in the Modulation of Immune Cells and Responses. Cancers, 11(10), 1469. https://doi.org/10.3390/cancers11101469




