Metabolic Syndrome and the Risk of Breast Cancer and Subtypes by Race, Menopause and BMI
Abstract
1. Introduction
2. Methods
2.1. Study Participants
2.2. Main Exposure
2.3. Main Outcome
2.4. Study Covariates
2.5. Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Author contributions
Funding
Conflicts of Interest
References
- Becker, S. A historic and scientific review of breast cancer: The next global healthcare challenge. Int. J. Gynecol. Obstet. 2015, 131, S36–S39. [Google Scholar] [CrossRef] [PubMed]
- Siegel, R.L.; Miller, K.D.; Jemal, A. Cancer statistics, 2018. CA A Cancer J. Clin. 2018, 68, 7–30. [Google Scholar] [CrossRef] [PubMed]
- Ferlay, J.; Soerjomataram, I.; Dikshit, R.; Eser, S.; Mathers, C.; Rebelo, M.; Parkin, D.M.; Forman, D.; Bray, F. Cancer incidence and mortality worldwide: Sources, methods and major patterns in globocan 2012. Int. J. Cancer 2015, 136, E359–E386. [Google Scholar] [CrossRef] [PubMed]
- Bowen, R.L.; Stebbing, J.; Jones, L.J. A review of the ethnic differences in breast cancer. Pharmacogenomics 2006, 7, 935–942. [Google Scholar] [CrossRef] [PubMed]
- Chlebowski, R.T.; Chen, Z.; Anderson, G.L.; Rohan, T.; Aragaki, A.; Lane, D.; Dolan, N.C.; Paskett, E.D.; McTiernan, A.; Hubbell, F.A.; et al. Ethnicity and breast cancer: Factors influencing differences in incidence and outcome. JNCI 2005, 97, 439–448. [Google Scholar] [CrossRef] [PubMed]
- Renehan, A.G.; Tyson, M.; Egger, M.; Heller, R.F.; Zwahlen, M. Body-mass index and incidence of cancer: A systematic review and meta-analysis of prospective observational studies. Lancet 2008, 371, 569–578. [Google Scholar] [CrossRef]
- Ogundiran, T.O.; Huo, D.; Adenipekun, A.; Campbell, O.; Oyesegun, R.; Akang, E.; Adebamowo, C.; Olopade, O.I. Body fat distribution and breast cancer risk: Findings from the nigerian breast cancer study. Cancer Causes Control 2012, 23, 565–574. [Google Scholar] [CrossRef] [PubMed]
- Nicolucci, A. Epidemiological aspects of neoplasms in diabetes. Acta Diabetol. 2010, 47, 87–95. [Google Scholar] [CrossRef] [PubMed]
- Pereira, A.; Garmendia, M.L.; Alvarado, M.E.; Albala, C. Hypertension and the risk of breast cancer in chilean women: A case-control study. APJCP 2012, 13, 5829–5834. [Google Scholar] [CrossRef] [PubMed]
- Soler, M.; Chatenoud, L.; Negri, E.; Parazzini, F.; Franceschi, S.; la Vecchia, C. Hypertension and hormone-related neoplasms in women. Hypertension 1999, 34, 320–325. [Google Scholar] [CrossRef] [PubMed]
- Porto, L.A.; Lora, K.J.; Soares, J.C.; Costa, L.O. Metabolic syndrome is an independent risk factor for breast cancer. Arch. Gynecol Obstet. 2011, 284, 1271–1276. [Google Scholar] [CrossRef] [PubMed]
- Rosato, V.; Bosetti, C.; Talamini, R.; Levi, F.; Montella, M.; Giacosa, A.; Negri, E.; La Vecchia, C. Metabolic syndrome and the risk of breast cancer in postmenopausal women. Ann. Oncol. 2011, 22, 2687–2692. [Google Scholar] [CrossRef] [PubMed]
- Capasso, I.; Esposito, E.; de Laurentiis, M.; Maurea, N.; Cavalcanti, E.; Botti, G.; Petrillo, A.; Montella, M.; D’Aiuto, M.; Coppola, C.; et al. Metabolic syndrome-breast cancer link varies by intrinsic molecular subtype. Diabetol. Metab. Syndr. 2014, 6, 105. [Google Scholar] [CrossRef] [PubMed]
- Maiti, B.; Kundranda, M.N.; Spiro, T.P.; Daw, H.A. The association of metabolic syndrome with triple-negative breast cancer. Breast Cancer Res. Tr. 2010, 121, 479–483. [Google Scholar] [CrossRef] [PubMed]
- Calip, G.S.; Malone, K.E.; Gralow, J.R.; Stergachis, A.; Hubbard, R.A.; Boudreau, D.M. Metabolic syndrome and outcomes following early-stage breast cancer. Breast Cancer Res. Treat. 2014, 148, 363–377. [Google Scholar] [CrossRef] [PubMed]
- Healy, L.A.; Ryan, A.M.; Carroll, P.; Ennis, D.; Crowley, V.; Boyle, T.; Kennedy, M.J.; Connolly, E.; Reynolds, J.V. Metabolic syndrome, central obesity and insulin resistance are associated with adverse pathological features in postmenopausal breast cancer. Clin. Oncol. (R. Coll. Radiol.) 2010, 22, 281–288. [Google Scholar] [CrossRef] [PubMed]
- Berrino, F.; Villarini, A.; Traina, A.; Bonanni, B.; Panico, S.; Mano, M.P.; Mercandino, A.; Galasso, R.; Barbero, M.; Simeoni, M.; et al. Metabolic syndrome and breast cancer prognosis. Breast Cancer Res. Treat. 2014, 147, 159–165. [Google Scholar] [CrossRef] [PubMed]
- Yang, X.R.; Chang-Claude, J.; Goode, E.L.; Couch, F.J.; Nevanlinna, H.; Milne, R.L.; Gaudet, M.; Schmidt, M.K.; Broeks, A.; Cox, A.L.; et al. Associations of breast cancer risk factors with tumor subtypes: A pooled analysis from the breast cancer association consortium studies. J. Natl. Cancer I 2011, 103, 250–263. [Google Scholar] [CrossRef] [PubMed]
- Pierobon, M.; Frankenfeld, C.L. Obesity as a risk factor for triple-negative breast cancers: A systematic review and meta-analysis. Breast Cancer Res. Treat. 2013, 137, 307–314. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, R.; Iwasaki, M.; Inoue, M.; Sasazuki, S.; Sawada, N.; Yamaji, T.; Shimazu, T.; Tsugane, S. Body weight at age 20 years, subsequent weight change and breast cancer risk defined by estrogen and progesterone receptor status--the japan public health center-based prospective study. Int. J. Cancer 2011, 129, 1214–1224. [Google Scholar] [CrossRef] [PubMed]
- Mitrou, P.N.; Kipnis, V.; Thiébaut, A.M.; Reedy, J.; Subar, A.F.; Wirfält, E.; Flood, A.; Mouw, T.; Hollenbeck, A.R.; Leitzmann, M.F.; et al. Mediterranean dietary pattern and prediction of all-cause mortality in a us population: Results from the nih-aarp diet and health study. Arch. Intern. Med. 2007, 167, 2461–2468. [Google Scholar] [CrossRef] [PubMed]
- Schatzkin, A.; Subar, A.F.; Thompson, F.E.; Harlan, L.C.; Tangrea, J.; Hollenbeck, A.R.; Hurwitz, P.E.; Coyle, L.; Schussler, N.; Michaud, D.S.; et al. Design and serendipity in establishing a large cohort with wide dietary intake distributions the national institutes of health–american association of retired persons diet and health study. Am. J. Epidemiol. 2001, 154, 1119–1125. [Google Scholar] [CrossRef] [PubMed]
- Brinton, L.A.; Smith, L.; Gierach, G.L.; Pfeiffer, R.M.; Nyante, S.J.; Sherman, M.E.; Park, Y.; Hollenbeck, A.R.; Dallal, C.M. Breast cancer risk in older women: Results from the nih-aarp diet and health study. CCC 2014, 25, 843–857. [Google Scholar] [CrossRef] [PubMed]
- Alberti, K.G.; Eckel, R.H.; Grundy, S.M.; Zimmet, P.Z.; Cleeman, J.I.; Donato, K.A.; Fruchart, J.C.; James, W.P.; Loria, C.M.; Smith, S.C., Jr.; et al. Harmonizing the metabolic syndrome: A joint interim statement of the international diabetes federation task force on epidemiology and prevention; national heart, lung, and blood institute; american heart association; world heart federation; international atherosclerosis society; and international association for the study of obesity. Circulation 2009, 120, 1640–1645. [Google Scholar] [PubMed]
- Park, Y.; Leitzmann, M.F.; Subar, A.F.; Hollenbeck, A.; Schatzkin, A. Dairy food, calcium, and risk of cancer in the nih-aarp diet and health study. Arch. Intern. Med. 2009, 169, 391–401. [Google Scholar] [CrossRef] [PubMed]
- Fine, J.P.; Gray, R.J. A proportional hazards model for the subdistribution of a competing risk. J. Am. Stat. Assoc. 1999, 94, 496–509. [Google Scholar] [CrossRef]
- Hiller, L.; Marshall, A.; Dunn, J. Assessing violations of the proportional hazards assumption in cox regression: Does the chosen method matter? Trials 2015, 16, P134. [Google Scholar] [CrossRef]
- Rong, S.S.; Chen, L.J.; Leung, C.K.S.; Matsushita, K.; Jia, L.; Miki, A.; Chiang, S.W.Y.; Tam, P.O.S.; Hashida, N.; Young, A.L.; et al. Ethnic specific association of the cav1/cav2 locus with primary open-angle glaucoma. Sci. Rep.-UK 2016, 6, 27837. [Google Scholar] [CrossRef] [PubMed]
- Park, Y.M.; White, A.J.; Nichols, H.B.; O’Brien, K.M.; Weinberg, C.R.; Sandler, D.P. The association between metabolic health, obesity phenotype and the risk of breast cancer. Int. J. Cancer 2017, 140, 2657–2666. [Google Scholar] [CrossRef] [PubMed]
- Esposito, K.; Chiodini, P.; Colao, A.; Lenzi, A.; Giugliano, D. Metabolic syndrome and risk of cancer. A systematic review and meta-analysis. Diabetes Care 2012, 35, 2402–2411. [Google Scholar] [CrossRef] [PubMed]
- Capasso, I.; Esposito, E.; Pentimalli, F.; Crispo, A.; Montella, M.; Grimaldi, M.; De Marco, M.; Cavalcanti, E.; D’Aiuto, M.; Fucito, A.; et al. Metabolic syndrome affects breast cancer risk in postmenopausal women: National cancer institute of naples experience. Cancer Biol. Ther. 2010, 10, 1240–1243. [Google Scholar] [CrossRef] [PubMed]
- Agnoli, C.; Berrino, F.; Abagnato, C.A.; Muti, P.; Panico, S.; Crosignani, P.; Krogh, V. Metabolic syndrome and postmenopausal breast cancer in the ordet cohort: A nested case-control study. NMCD 2010, 20, 41–48. [Google Scholar] [CrossRef] [PubMed]
- Kabat, G.C.; Kim, M.; Chlebowski, R.T.; Khandekar, J.; Ko, M.G.; McTiernan, A.; Neuhouser, M.L.; Parker, D.R.; Shikany, J.M.; Stefanick, M.L.; et al. A longitudinal study of the metabolic syndrome and risk of postmenopausal breast cancer. Cancer Epidemiol. Biomark. Prev. 2009, 18, 2046–2053. [Google Scholar] [CrossRef] [PubMed]
- Bjorge, T.; Lukanova, A.; Jonsson, H.; Tretli, S.; Ulmer, H.; Manjer, J.; Stocks, T.; Selmer, R.; Nagel, G.; Almquist, M.; et al. Metabolic syndrome and breast cancer in the me-can (metabolic syndrome and cancer) project. Cancer Epidemiol. Biomark. Prev. 2010, 19, 1737–1745. [Google Scholar] [CrossRef] [PubMed]
- Osaki, Y.; Taniguchi, S.; Tahara, A.; Okamoto, M.; Kishimoto, T. Metabolic syndrome and incidence of liver and breast cancers in japan. Cancer Epidemiol. 2012, 36, 141–147. [Google Scholar] [CrossRef] [PubMed]
- Esposito, K.; Chiodini, P.; Capuano, A.; Bellastella, G.; Maiorino, M.I.; Rafaniello, C.; Giugliano, D. Metabolic syndrome and postmenopausal breast cancer: Systematic review and meta-analysis. Menopause 2013, 20, 1301–1309. [Google Scholar] [CrossRef] [PubMed]
- Wani, B.; Aziz, S.A.; Ganaie, M.A.; Mir, M.H. Metabolic syndrome and breast cancer risk. Indian J. Med. Paediatr. Oncol. 2017, 38, 434–439. [Google Scholar] [CrossRef] [PubMed]
- Agnoli, C.; Grioni, S.; Sieri, S.; Sacerdote, C.; Ricceri, F.; Tumino, R.; Frasca, G.; Pala, V.; Mattiello, A.; Chiodini, P.; et al. Metabolic syndrome and breast cancer risk: A case-cohort study nested in a multicentre italian cohort. PLoS ONE 2015, 10, e0128891. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.; Lu, F.; Liu, S.J.; Du, J.B.; Wang, J.M.; Qian, Y.; Shen, C.; Jin, G.F.; Hu, Z.B.; Shen, H.B. Cancer risk and key components of metabolic syndrome: A population-based prospective cohort study in Chinese. Chin. Med. J.-Peking 2012, 125, 481–485. [Google Scholar]
- Lamar, M.; Rubin, L.H.; Ajilore, O.; Charlton, R.; Zhang, A.; Yang, S.; Cohen, J.; Kumar, A. What metabolic syndrome contributes to brain outcomes in african american & caucasian cohorts. Curr. Alzheimer Res. 2015, 12, 640–647. [Google Scholar] [PubMed]
- Zeller, M.; Steg, P.; Ravisy, J.; Laurent, Y.; Janin-Manificat, L.; L’Huillier, I.; Beer, J.C.; Oudot, A.; Rioufol, G.; Makki, H.; et al. Prevalence and impact of metabolic syndrome on hospital outcomes in acute myocardial infarction. Arch. Intern. Med. 2005, 165, 1192–1198. [Google Scholar] [CrossRef] [PubMed]
- Djiogue, S.; Nwabo Kamdje, A.H.; Vecchio, L.; Kipanyula, M.J.; Farahna, M.; Aldebasi, Y.; Seke Etet, P.F. Insulin resistance and cancer: The role of insulin and igfs. Endocr.-Relat. Cancer 2013, 20, R1–R17. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Wen, Y.-Y.; Li, Z.-R.; Luo, D.-L.; Zhang, X.-H. The molecular mechanisms between metabolic syndrome and breast cancer. Biochem. Bioph. Res. Commun. 2016, 471, 391–395. [Google Scholar] [CrossRef] [PubMed]
- Yun, J.E.; Kimm, H.; Jo, J.; Jee, S.H. Serum leptin is associated with metabolic syndrome in obese and nonobese korean populations. Metabolism 2010, 59, 424–429. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.W.; Jo, H.H.; Kim, M.R.; You, Y.O.; Kim, J.H. Association between metabolic syndrome and serum leptin levels in postmenopausal women. J. Obstet. Gynaecol. 2012, 32, 73–77. [Google Scholar] [CrossRef] [PubMed]
- Niu, J.; Jiang, L.; Guo, W.; Shao, L.; Liu, Y.; Wang, L. The association between leptin level and breast cancer: A meta-analysis. PLoS ONE 2013, 8, e67349. [Google Scholar] [CrossRef] [PubMed]
- Assiri, A.M.A.; Kamel, H.F.M.; Hassanien, M.F.R. Resistin, visfatin, adiponectin, and leptin: Risk of breast cancer in pre- and postmenopausal saudi females and their possible diagnostic and predictive implications as novel biomarkers. Dis. Mark. 2015, 2015, 9. [Google Scholar] [CrossRef] [PubMed]
- Hauner, D.; Hauner, H. Metabolic syndrome and breast cancer: Is there a link? Breast Care 2014, 9, 277–281. [Google Scholar] [CrossRef] [PubMed]
- Nelson, E.R.; Chang, C.-Y.; McDonnell, D.P. Cholesterol and breast cancer pathophysiology. TEM 2014, 25, 649–655. [Google Scholar] [CrossRef] [PubMed]
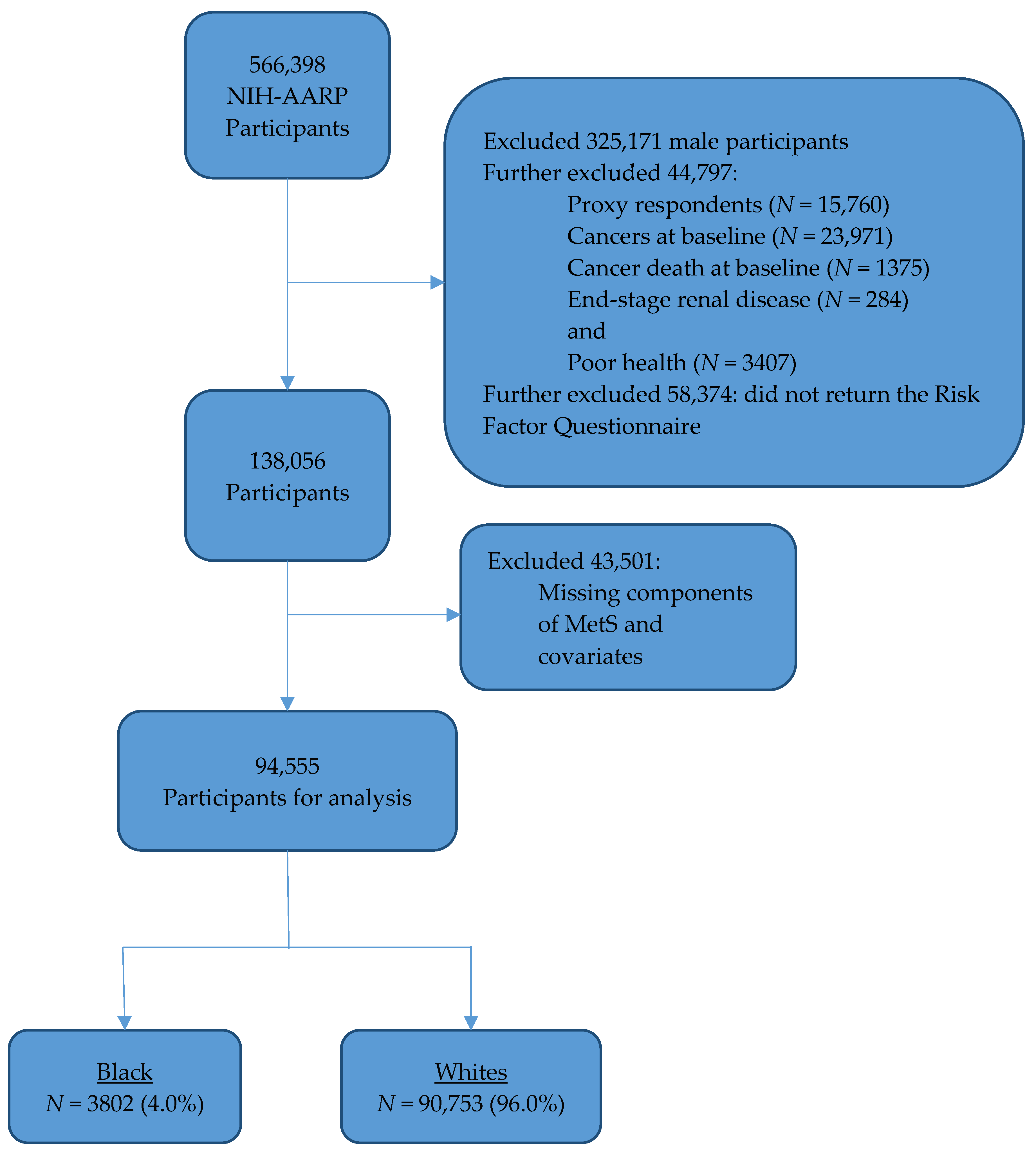
| Study Characteristics | Total | Breast Cancer | No Breast Cancer | p-Value |
|---|---|---|---|---|
| N (%) | N (col%) | N (col%) | ||
| Ethnicity | ||||
| White | 90,753 (96.0) | 5214 (96.9) | 85,539 (95.9) | 0.0003 |
| Black | 3802 (4.0) | 166 (3.1) | 3636 (4.1) | |
| Age at entry | ||||
| 50–59 | 35,805 (37.9) | 1899 (35.3) | 33,906 (38) | <0.0001 |
| 60–69 | 55,638 (58.8) | 3317 (61.7) | 52,321 (58.7) | |
| 70–79 | 3112 (3.3) | 164 (3.0) | 2948 (3.3) | |
| Education | ||||
| <High school | 4058 (4.3) | 189 (3.5) | 3869 (4.3) | <0.0001 |
| High school or GED | 33,226 (35.1) | 1762 (32.8) | 31,464 (35.3) | |
| Some college | 24,563 (26.0) | 1401 (26) | 23,162 (26) | |
| ≥College | 32,708 (34.6) | 2028 (37.7) | 30,680 (34.4) | |
| Menopausal status | ||||
| Premenopausal | 3693 (3.9) | 196 (3.6) | 3497 (3.9) | 0.304 |
| Postmenopausal | 90,662 (96.1) | 5174 (96.4) | 85,488 (96.1) | |
| BMI | ||||
| <18.5 | 1450 (1.5) | 2344 (43.7) | 39,752 (44.7) | 0.047 |
| 18.5–24.9 | 42,096 (44.6) | 1719 (32) | 28,617 (32.2) | |
| 25.0–29.9 | 30,336 (32.2) | 776 (14.5) | 12,174 (13.7) | |
| 30.0–34.9 | 12,950 (13.7) | 460 (8.6) | 7039 (7.9) | |
| ≥35 | 7499 (8.0) | 556 (8.4) | 6943 (7.9) | |
| Metabolic syndrome | ||||
| Yes (%) | 4956 (5.2) | 293 (5.4) | 4663 (5.2) | 0.091 |
| Current hormone therapy | ||||
| Yes (%) | 44,387 (46.9) | 2863 (53.2) | 41,524 (46.6) | <0.0001 |
| Family history of breast cancer | ||||
| Yes (%) | 11,949 (12.6) | 951 (17.7) | 10,998 (12.3) | <0.0001 |
| N (Events) | All 94,555 (5380) b | Black 3802 (166) b | White 90,753 (5214) b |
|---|---|---|---|
| Overall c | |||
| MetS | 1.13 (1.00, 1.27) | 0.79(0.43, 1.43) | 1.10 (0.97, 1.25) |
| Components | |||
| High WC | 1.13 (1.05, 1.21) | 0.65 (0.42, 1.02) | 1.11 (1.02, 1.21) |
| Elevated Cholesterol | 1.06 (1.01, 1.12) | 1.03 (0.76, 1.39) | 1.08 (1.02, 1.14) |
| High blood pressure | 1.11 (1.04, 1.17) | 1.28 (0.92, 1.78) | 1.08 (1.02, 1.15) |
| Diabetes | 1.03 (0.92, 1.16) | 0.83 (0.52, 1.34) | 1.02 (0.90, 1.15) |
| Number of MetS Components | |||
| 0 (Ref) | 1.00 | 1.00 | 1.00 |
| 1 | 1.14 (1.03, 1.25) | 1.08 (0.56, 2.11) | 1.13 (1.03, 1.25) |
| 2 | 1.26 (1.13, 1.40) | 0.78 (0.38, 1.62) | 1.25 (1.12, 1.39) |
| 3 | 1.30 (1.12, 1.51) | 0.84 (0.36, 1.99) | 1.29 (1.10, 1.51) |
| 4 | 1.45 (0.99, 2.13) | 0.51 (0.06, 4.11) | 1.48 (1.00, 2.19) |
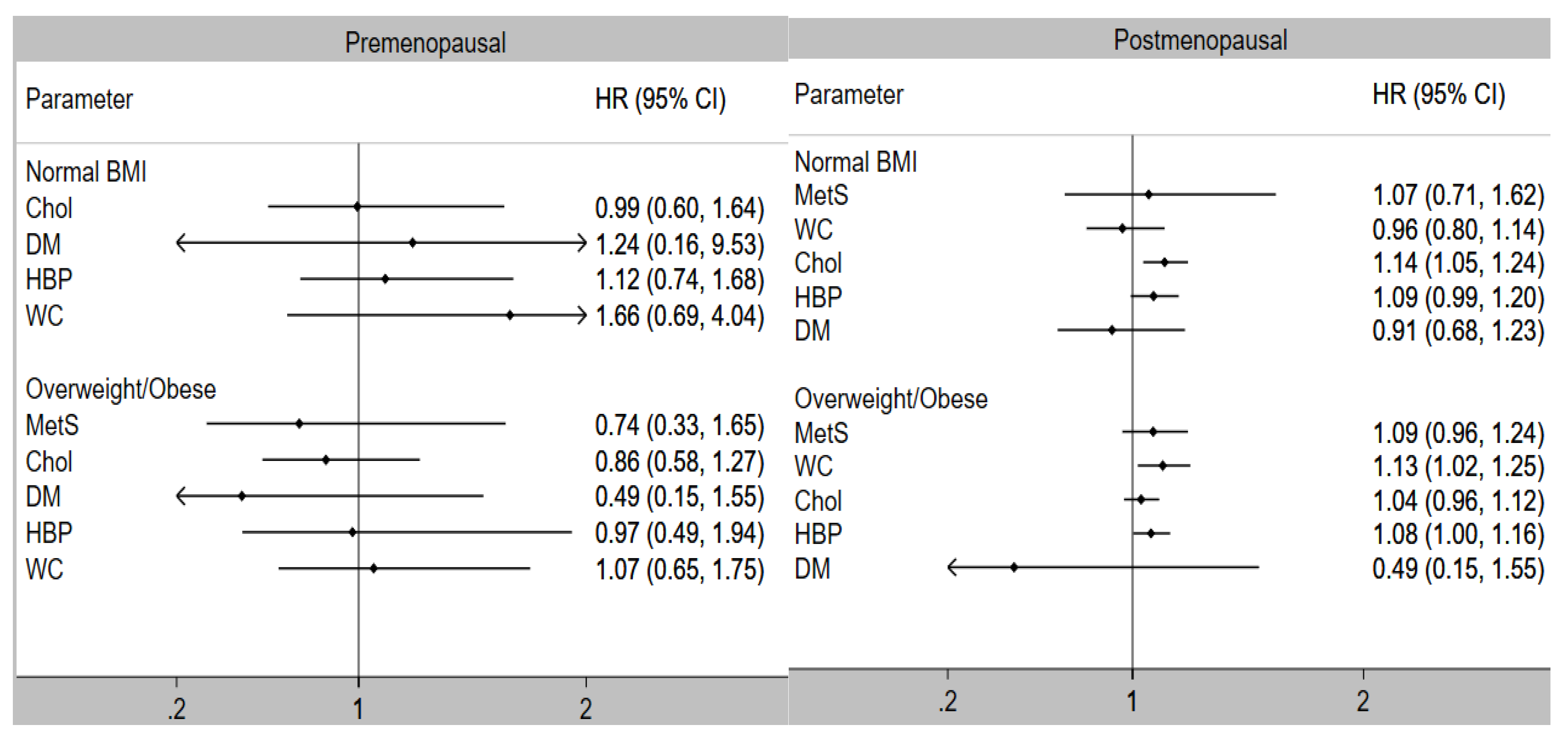
| Normal BMI | |||
| MetS | 1.04 (0.69, 1.58) | NE | 1.10 (0.73, 1.67) |
| Components | |||
| High WC | 0.97 (0.82, 1.16) | NE | 1.00 (0.84, 1.19) |
| Elevated Cholesterol | 1.13 (1.04, 1.23) | 1.12 (0.59, 2.15) | 1.13(1.04, 1.23) |
| High blood pressure | 1.09 (0.99, 1.20) | 1.22 (0.64, 2.31) | 1.09 (0.99, 1.20) |
| Diabetes | 0.91 (0.68, 1.22) | 0.28 (0.04, 1.99) | 0.95 (0.71, 1.28) |
| Number of MetS Components | |||
| 0 (Ref) | |||
| 1 | 1.15 (1.02, 1.29) | 0.87 (0.32, 2.33) | 1.15 (1.02, 1.29) |
| 2 | 1.27 (1.09, 1.48) | 0.88 (0.27, 2.86) | 1.27 (1.09, 1.49) |
| 3 | 1.15 (0.75, 1.78) | NE | 1.21 (0.78, 1.87) |
| 4 | 1.16 (0.16, 8.40) | NE | 1.47 (0.20, 10.62) |
| Overweight/Obese | |||
| MetS | 1.08 (0.95, 1.23) | 0.83 (0.45, 1.52) | 1.09 (0.96, 1.24) |
| Components | |||
| High WC | 1.13 (1.03, 1.25) | 0.73 (0.44, 1.20) | 1.15 (1.04, 1.27) |
| Elevated Cholesterol | 1.03 (0.96, 1.11) | 0.96 (0.67, 1.37) | 1.04 (0.96, 1.12) |
| High blood pressure | 1.08 (1.01, 1.16) | 1.31 (0.88, 1.95) | 1.07 (1.00, 1.16) |
| Diabetes | 1.01 (0.89, 1.16) | 0.93 (0.56, 1.55) | 1.02 (0.89, 1.17) |
| Number of MetS Components | |||
| 0 (Ref) | |||
| 1 | 1.10 (0.92, 1.32) | 1.24 (0.48, 3.24) | 1.10 (0.92, 1.32) |
| 2 | 1.18 (0.99, 1.41) | 0.74 (0.28, 1.95) | 1.20 (1.00, 1.44) |
| 3 | 1.24 (1.01, 1.53) | 0.95 (0.33, 2.76) | 1.25 (1.01, 1.55) |
| 4 | 1.35 (0.89, 2.05) | 0.57 (0.06, 5.06) | 1.40 (0.92, 2.15) |
| Overall | Postmenopausal | Premenopausal | |
|---|---|---|---|
| N (Events) | ER− vs. ER+ 4392 (685) § | ER− vs. ER+ 4212 (664) | ER− vs. ER+ 169 (21) |
| MetS | 0.88 (0.60, 1.28) | 0.97 (0.66, 1.43) | NE |
| Components | |||
| High WC | 0.71 (0. 57, 0.88) | 0.82 (0.62, 1.08) | 0.15 (0.02, 1.52) |
| Elevated Cholesterol | 1.12 (0.95, 1.33) | 1.09 (0.92, 1.29) | 2.04 (0.53, 7.88) |
| High blood pressure | 0.93 (0.78, 1.11) | 0.98 (0.82, 1.17) | 0.44 (0.09, 2.22) |
| Diabetes | 0.98 (0.68, 1.41) | 1.02 (0.71, 1.48) | 2.92 (0.11, 78.03) |
| Number of MetS Components | |||
| 0 | Ref | Ref | Ref |
| 1 | 1.05 (0.78, 1.41) | 1.00 (0.74, 1.34) | NE |
| 2 | 0.85 (0.61, 1.18) | 0.94 (0.67, 1.33) | NE |
| 3 | 0.85 (0.53, 1.36) | 0.99 (0.61, 1.63) | NE |
| 4 | 1.18 (0.38, 3.63) | 1.50 (0.48, 4.71) | NE |
| N (Events) | Postmenopausal 90,662 (5174) b | Premenopausal 3693 (196) b |
|---|---|---|
| MetS | 1.14 (1.01, 1.29) | 0.83 (0.38, 1.78) |
| Components | ||
| High WC | 1.12 (1.04, 1.20) | 1.31 (0.90, 1.90) |
| Elevated Cholesterol | 1.07 (1.01, 1.13) | 0.88 (0.65, 1.19) |
| High blood pressure | 1.11 (1.05, 1.17) | 1.14 (0.82, 1.59) |
| Diabetes | 1.05 (0.93, 1.18) | 0.60 (0.22, 1.63) |
| Number of MetS Components | ||
| 0 (Ref) | 1.00 | 1.00 |
| 1 | 1.15 (1.0, 1.27) | 0.79 (0.48, 1.30) |
| 2 | 1.26 (1.14, 1.41) | 1.01 (0.59, 1.78) |
| 3 | 1.32 (1.13, 1.53) | 0.70 (0.28, 1.75) |
| 4 | 1.44 (0.97, 2.12) | 2.00 (0.23, 17.74) |
| MetS Component Combinations | Overall (N = 94,555, Cases = 5380) b | Postmenopausal (N = 90,662, Cases = 5174) | Premenopausal (N = 3693, Cases = 196) |
|---|---|---|---|
| High blood pressure, high WC and diabetes | 1.15 (0.93, 1.43) | 1.11 (0.90, 1.38) | 1.18 (0.28, 4.92) |
| Elevated cholesterol, diabetes, and high WC | 1.15 (0.86, 1.53) | 1.10 (0.82, 1.48) | 1.14 (0.15, 8.87) |
| High blood pressure, diabetes and elevated cholesterol | 1.04 (0.80, 1.34) | 1.02 (0.79, 1.32) | 0.60 (0.08, 4.42) |
| High blood pressure, elevated cholesterol and high WC | 1.12 (0.97, 1.29) | 1.09(0.94, 1.26) | 0.79 (0.34, 1.79) |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dibaba, D.T.; Braithwaite, D.; Akinyemiju, T. Metabolic Syndrome and the Risk of Breast Cancer and Subtypes by Race, Menopause and BMI. Cancers 2018, 10, 299. https://doi.org/10.3390/cancers10090299
Dibaba DT, Braithwaite D, Akinyemiju T. Metabolic Syndrome and the Risk of Breast Cancer and Subtypes by Race, Menopause and BMI. Cancers. 2018; 10(9):299. https://doi.org/10.3390/cancers10090299
Chicago/Turabian StyleDibaba, Daniel T., Dejana Braithwaite, and Tomi Akinyemiju. 2018. "Metabolic Syndrome and the Risk of Breast Cancer and Subtypes by Race, Menopause and BMI" Cancers 10, no. 9: 299. https://doi.org/10.3390/cancers10090299
APA StyleDibaba, D. T., Braithwaite, D., & Akinyemiju, T. (2018). Metabolic Syndrome and the Risk of Breast Cancer and Subtypes by Race, Menopause and BMI. Cancers, 10(9), 299. https://doi.org/10.3390/cancers10090299






