Sperm Protein 17 Expression by Murine Epithelial Ovarian Cancer Cells and Its Impact on Tumor Progression
Abstract
1. Introduction
2. Results
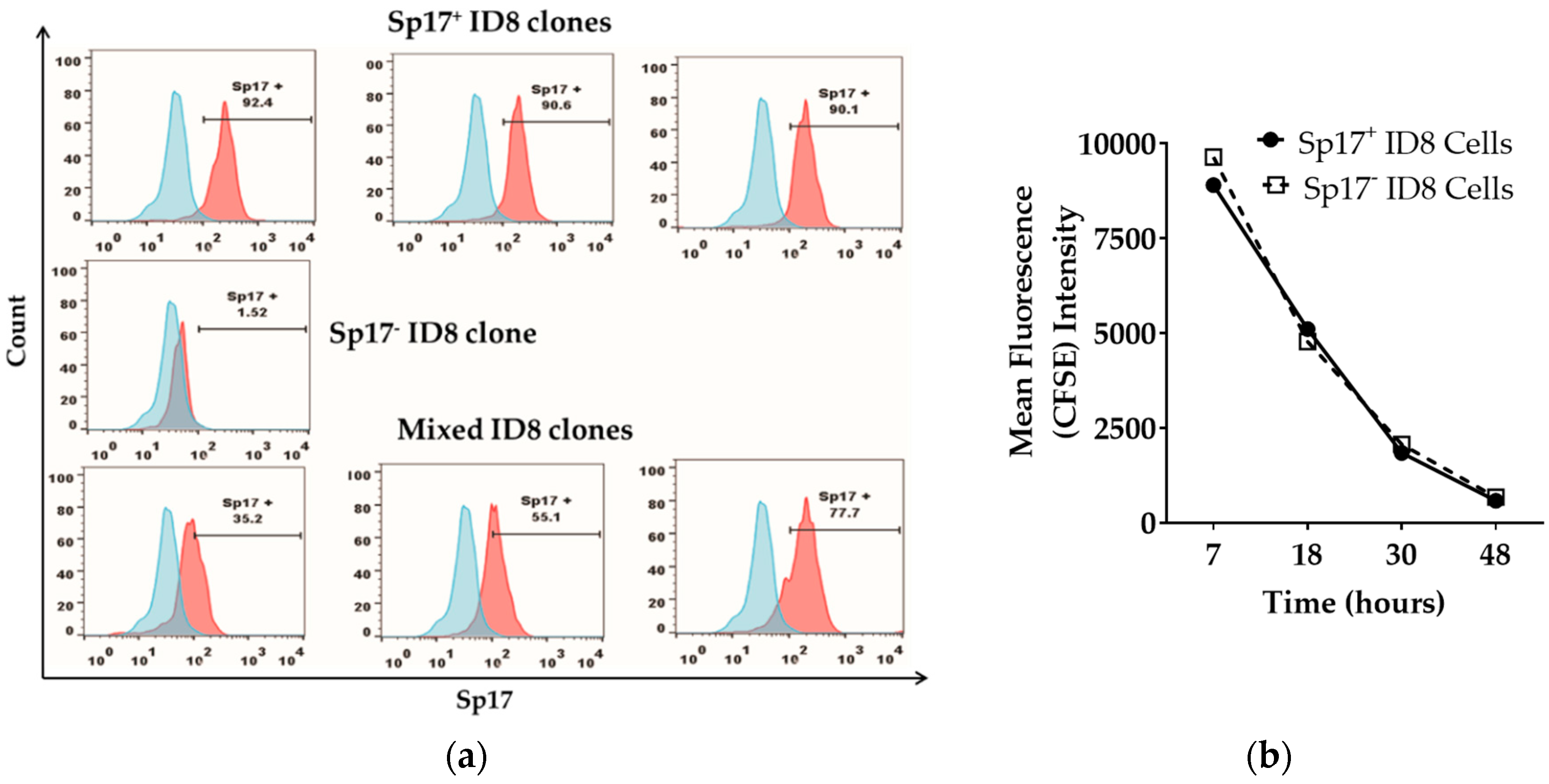
2.1. Sperm Protein 17 Is Expressed in a Subset of ID8 Cells In Vitro
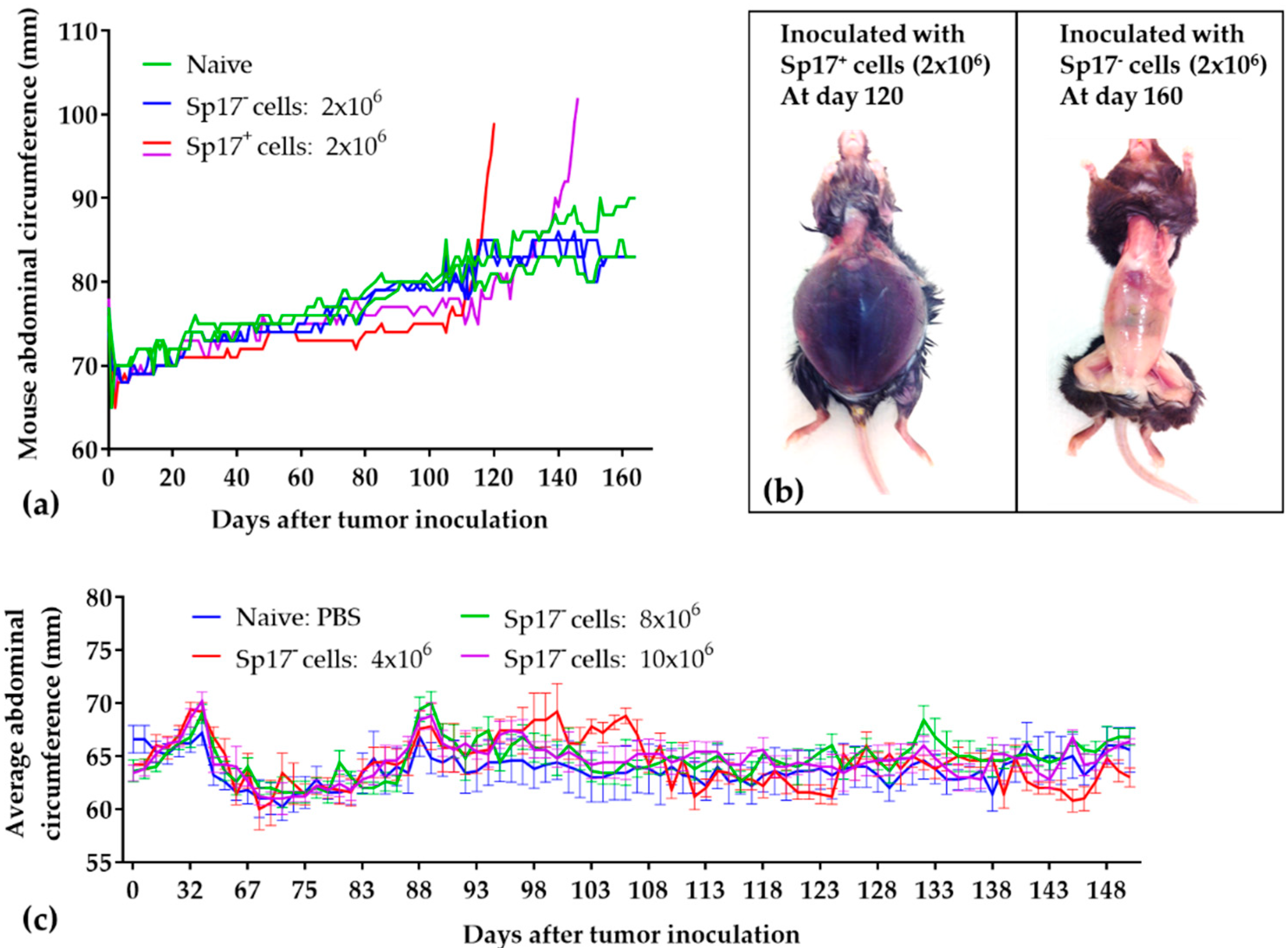
2.2. Sperm Protein 17 Expression Is Required for Tumor Formation In Vivo
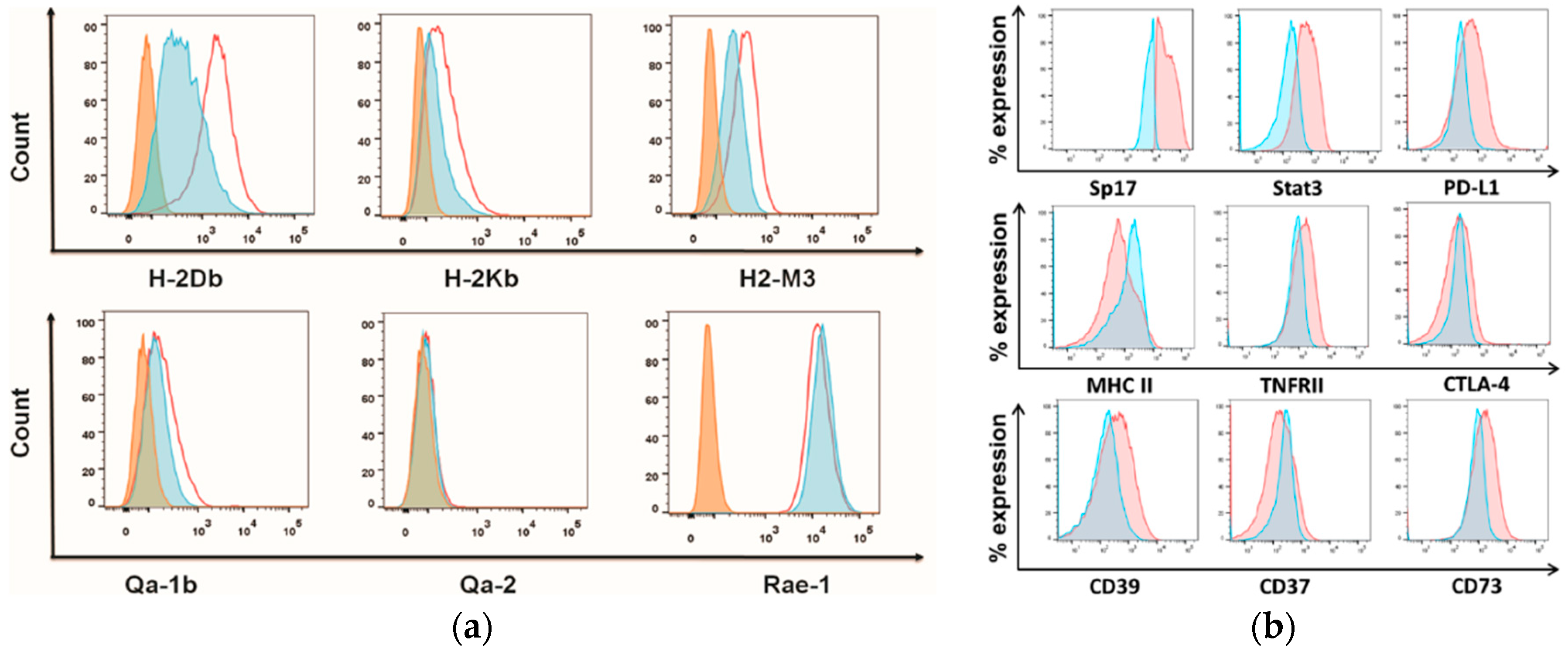
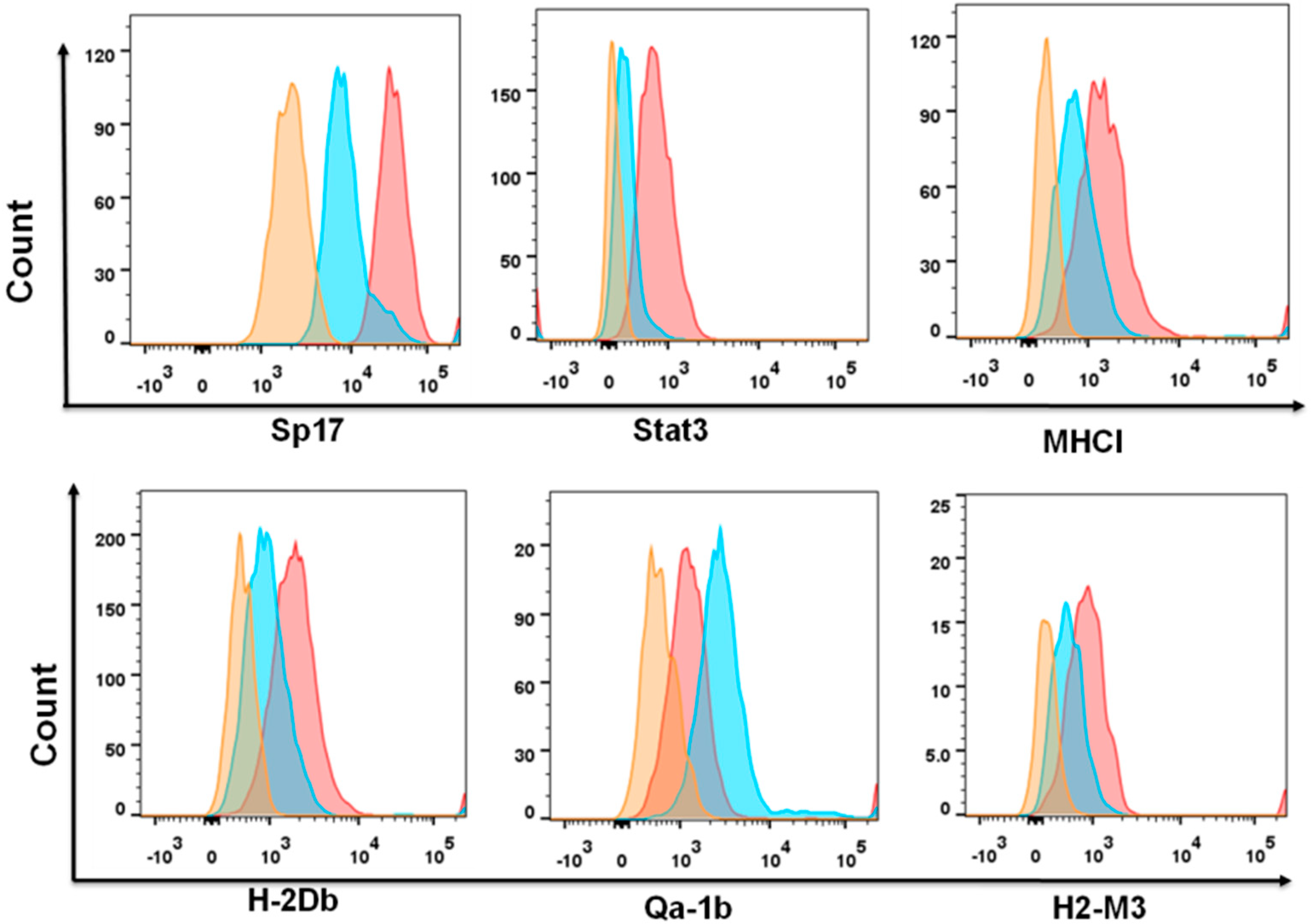
2.3. Sperm Protein 17 Expression Correlates with Altered Immunosuppressive Potential in ID8 Cells
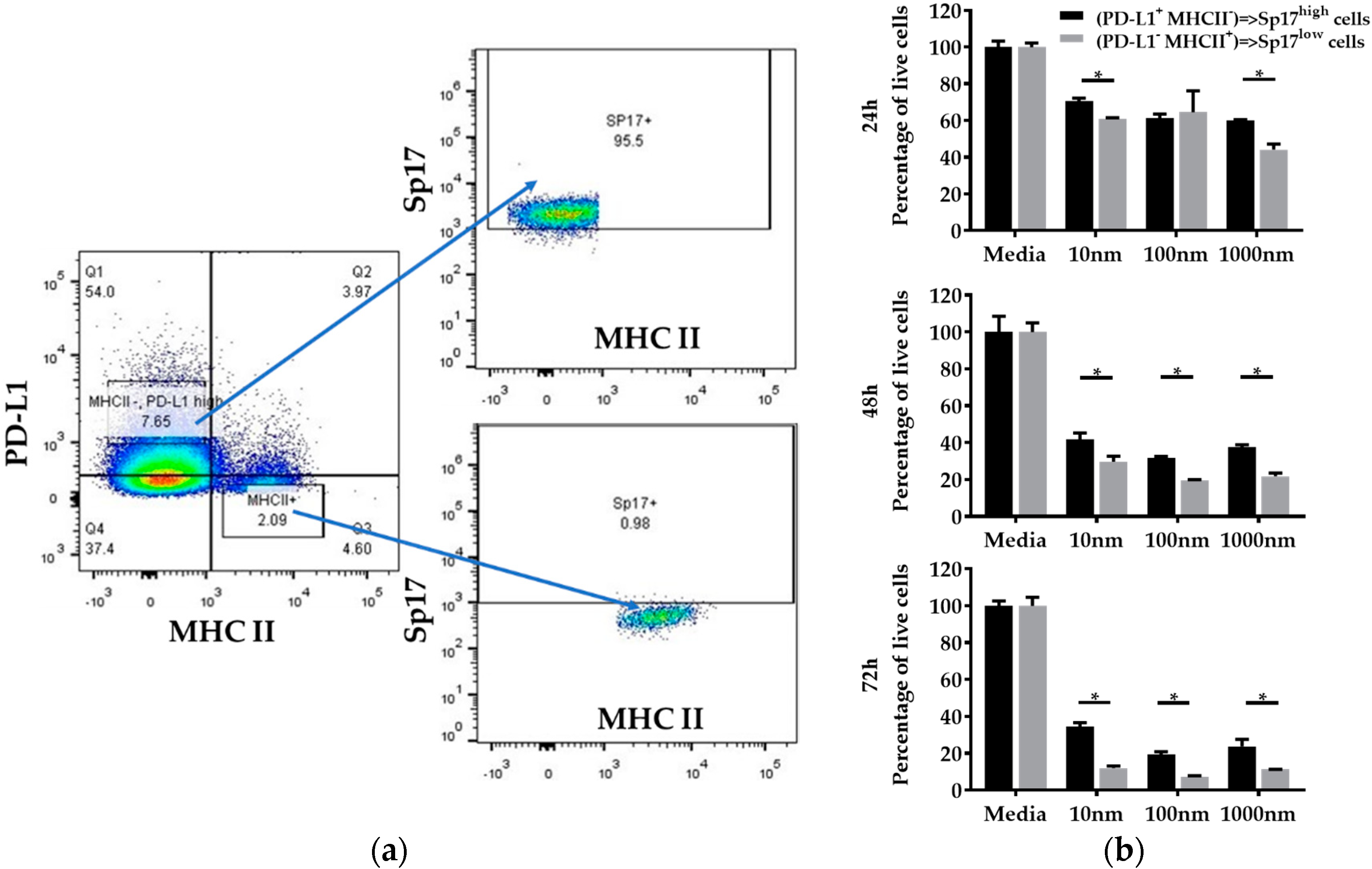
2.4. PD-L1highMHCII− (Sp17high) Cells Display Increased Resistance to Paclitaxel In Vitro
3. Discussion
4. Materials and Methods
4.1. Isolation of Sp17+ ID8 Clones
4.2. ID8 Murine Ovarian Cancer Model
4.3. Flow Cytometry
4.4. Cytotoxicity Assay
4.5. Carboxyfluorescein Succinimidyl Ester Proliferation Assay
4.6. Statistical Analyses
5. Conclusions
6. Patents
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| HEPES | 2-[4-(2-Hydroxyethyl)-1-piperazinyl] ethanesulfonic acid |
| MTT | 3-(4,5-Dimethylthiazol-2-yl)-2,5-Diphenyltetrazolium Bromide |
| CTAs | Cancer testes antigens |
| CD | Clusters of Differentiation |
| CTLA-4 | Cytotoxic T lymphocyte associated antigen-4 |
| EOC | Epithelial ovarian cancer |
| FACS | Fluorescent-activated cell sorting |
| Her2 | Human epidermal growth factor receptor 2 |
| MHC I | Major histocompatibility class I molecules |
| MUC16 | Mucin 16 |
| NHMRC | National health and medical research council |
| NK | Natural killer |
| NY-ESO-1 | New York esophageal squamous cell carcinoma 1 |
| OC | Ovarian cancer |
| PBS | Phosphate buffered saline |
| PD-L1 | Programed cell death ligand-1 |
| PD-1 | Programed cell death protein-1 |
| RPMI | Roswell Park Memorial Institute |
| STAT3 | Signal transducer and activator of transcription 3 |
| Sp17 | Sperm protein 17 |
| TNFRII | Tumor necrosis factor receptor II |
| TP53 | Tumor protein p53 |
References
- Yeung, T.L.; Leung, C.S.; Yip, K.P.; Au Yeung, C.L.; Wong, S.T.; Mok, S.C. Cellular and molecular processes in ovarian cancer metastasis. A review in the theme: Cell and molecular processes in cancer metastasis. Am. J. Physiol. Cell Physiol. 2015, 309, C444–C456. [Google Scholar] [CrossRef] [PubMed]
- Pakish, J.B.; Jazaeri, A.A. Immunotherapy in gynecologic cancers: Are we there yet? Curr. Treat. Opt. Oncol. 2017, 18, 59. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez-Garcia, A.; Minutolo, N.G.; Robinson, J.M.; Powell, D.J. T-cell target antigens across major gynecologic cancers. Gynecol. Oncol. 2017, 145, 426–435. [Google Scholar] [CrossRef] [PubMed]
- Martin Lluesma, S.; Wolfer, A.; Harari, A.; Kandalaft, L.E. Cancer vaccines in ovarian cancer: How can we improve? Biomedicines 2016, 4, 10. [Google Scholar] [CrossRef] [PubMed]
- Pujol, J.L.; Vansteenkiste, J.F.; De Pas, T.M.; Atanackovic, D.; Reck, M.; Thomeer, M.; Douillard, J.Y.; Fasola, G.; Potter, V.; Taylor, P.; et al. Safety and immunogenicity of mage-a3 cancer immunotherapeutic with or without adjuvant chemotherapy in patients with resected stage Ib to III mage-a3-positive non-small-cell lung cancer. J. Thorac. Oncol. 2015, 10, 1458–1467. [Google Scholar] [CrossRef] [PubMed]
- Schlom, J. Therapeutic cancer vaccines: Current status and moving forward. J. Natl. Cancer Inst. 2012, 104, 599–613. [Google Scholar] [CrossRef] [PubMed]
- Hirayama, M.; Nishimura, Y. The present status and future prospects of peptide-based cancer vaccines. Int. Immunol. 2016, 28, 319–328. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Soto, A.E.; Schreiber, T.; Strbo, N.; Ganjei-Azar, P.; Miao, F.; Koru-Sengul, T.; Simpkins, F.; Nieves-Neira, W.; Lucci, J., 3rd; Podack, E.R. Cancer-testis antigen expression is shared between epithelial ovarian cancer tumors. Gynecol. Oncol. 2017, 145, 413–419. [Google Scholar] [CrossRef] [PubMed]
- Wen, Y.; Richardson, R.T.; Widgren, E.E.; O’Rand, M.G. Characterization of sp17: A ubiquitous three domain protein that binds heparin. Biochem. J. 2001, 357, 25–31. [Google Scholar] [CrossRef] [PubMed]
- Yamasaki, N.; Richardson, R.T.; O’Rand, M.G. Expression of the rabbit sperm protein sp17 in COS cells and interaction of recombinant sp17 with the rabbit zona pellucida. Mol. Reprod. Dev. 1995, 40, 48–55. [Google Scholar] [PubMed]
- Li, F.Q.; Han, Y.L.; Liu, Q.; Wu, B.; Huang, W.B.; Zeng, S.Y. Overexpression of human sperm protein 17 increases migration and decreases the chemosensitivity of human epithelial ovarian cancer cells. BMC Cancer 2009, 9, 323. [Google Scholar] [CrossRef] [PubMed]
- Nakazato, T.; Kanuma, T.; Tamura, T.; Faried, L.S.; Aoki, H.; Minegishi, T. Sperm protein 17 influences the tissue-specific malignancy of clear cell adenocarcinoma in human epithelial ovarian cancer. Int. J. Gynecol. Cancer 2007, 17, 426–432. [Google Scholar] [CrossRef] [PubMed]
- Grizzi, F.; Franceschini, B.; Hermonat, P.L.; Liu, Y.; Chiriva-Internati, M. Some remarks on the somatic expression of sperm protein 17. Int. J. Cancer 2004, 111, 972–973. [Google Scholar] [CrossRef] [PubMed]
- Arnaboldi, F.; Menon, A.; Menegola, E.; Di Renzo, F.; Mirandola, L.; Grizzi, F.; Figueroa, J.A.; Cobos, E.; Jenkins, M.; Barajon, I.; et al. Sperm protein 17 is an oncofetal antigen: A lesson from a murine model. Int. Rev. Immunol. 2014, 33, 367–374. [Google Scholar] [CrossRef] [PubMed]
- Straughn, J.M., Jr.; Shaw, D.R.; Guerrero, A.; Bhoola, S.M.; Racelis, A.; Wang, Z.; Chiriva-Internati, M.; Grizzle, W.E.; Alvarez, R.D.; Lim, S.H.; et al. Expression of sperm protein 17 (sp17) in ovarian cancer. Int. J. Cancer 2004, 108, 805–811. [Google Scholar] [CrossRef] [PubMed]
- Dadabayev, A.R.; Wang, Z.; Zhang, Y.; Zhang, J.; Robinson, W.R.; Lim, S.H. Cancer immunotherapy targeting sp17: When should the laboratory findings be translated to the clinics? Am. J. Hematol. 2005, 80, 6–11. [Google Scholar] [CrossRef] [PubMed]
- Chiriva-Internati, M.; Yu, Y.; Mirandola, L.; Jenkins, M.R.; Chapman, C.; Cannon, M.; Cobos, E.; Kast, W.M. Cancer testis antigen vaccination affords long-term protection in a murine model of ovarian cancer. PLoS ONE 2010, 5, e10471. [Google Scholar] [CrossRef] [PubMed]
- Zhu, X.; Lang, J. Programmed death-1 pathway blockade produces a synergistic antitumor effect: Combined application in ovarian cancer. J. Gynecol. Oncol. 2017, 28, e64. [Google Scholar] [CrossRef] [PubMed]
- Peng, J.; Hamanishi, J.; Matsumura, N.; Abiko, K.; Murat, K.; Baba, T.; Yamaguchi, K.; Horikawa, N.; Hosoe, Y.; Murphy, S.K.; et al. Chemotherapy induces programmed cell death-ligand 1 overexpression via the nuclear factor-kappaB to foster an immunosuppressive tumor microenvironment in ovarian cancer. Cancer Res. 2015, 75, 5034–5045. [Google Scholar] [CrossRef] [PubMed]
- Guo, Y.; Xu, F.; Lu, T.; Duan, Z.; Zhang, Z. Interleukin-6 signaling pathway in targeted therapy for cancer. Cancer Treat. Rev. 2012, 38, 904–910. [Google Scholar] [CrossRef] [PubMed]
- Siveen, K.S.; Sikka, S.; Surana, R.; Dai, X.; Zhang, J.; Kumar, A.P.; Tan, B.K.; Sethi, G.; Bishayee, A. Targeting the STAT3 signaling pathway in cancer: Role of synthetic and natural inhibitors. Biochim. Biophys. Acta 2014, 1845, 136–154. [Google Scholar] [CrossRef] [PubMed]
- Roby, K.F.; Taylor, C.C.; Sweetwood, J.P.; Cheng, Y.; Pace, J.L.; Tawfik, O.; Persons, D.L.; Smith, P.G.; Terranova, P.F. Development of a syngeneic mouse model for events related to ovarian cancer. Carcinogenesis 2000, 21, 585–591. [Google Scholar] [CrossRef] [PubMed]
- Gupta, G.; Sharma, R.; Chattopadhyay, T.K.; Gupta, S.D.; Ralhan, R. Clinical significance of sperm protein 17 expression and immunogenicity in esophageal cancer. Int. J. Cancer 2007, 120, 1739–1747. [Google Scholar] [CrossRef] [PubMed]
- Lacy, H.M.; Sanderson, R.D. Sperm protein 17 is expressed on normal and malignant lymphocytes and promotes heparan sulfate-mediated cell-cell adhesion. Blood 2001, 98, 2160–2165. [Google Scholar] [CrossRef] [PubMed]
- D’Almeida, S.M.; Kauffenstein, G.; Roy, C.; Basset, L.; Papargyris, L.; Henrion, D.; Catros, V.; Ifrah, N.; Descamps, P.; Croue, A.; et al. The ecto-ATPdase CD39 is involved in the acquisition of the immunoregulatory phenotype by m-csf-macrophages and ovarian cancer tumor-associated macrophages: Regulatory role of IL-27. Oncoimmunology 2016, 5, e1178025. [Google Scholar] [CrossRef] [PubMed]
- Montalban Del Barrio, I.; Penski, C.; Schlahsa, L.; Stein, R.G.; Diessner, J.; Wockel, A.; Dietl, J.; Lutz, M.B.; Mittelbronn, M.; Wischhusen, J.; et al. Adenosine-generating ovarian cancer cells attract myeloid cells which differentiate into adenosine-generating tumor associated macrophages—A self-amplifying, CD39- and CD73-dependent mechanism for tumor immune escape. J. Immunother. Cancer 2016, 4, 49. [Google Scholar] [CrossRef] [PubMed]
- Chen, M.W.; Yang, S.T.; Chien, M.H.; Hua, K.T.; Wu, C.J.; Hsiao, S.M.; Lin, H.; Hsiao, M.; Su, J.L.; Wei, L.H. The STAT3-miRNA-92-wnt signaling pathway regulates spheroid formation and malignant progression in ovarian cancer. Cancer Res. 2017, 77, 1955–1967. [Google Scholar] [CrossRef] [PubMed]
- Mirandola, L.; Figueroa, J.A.; Phan, T.T.; Grizzi, F.; Kim, M.; Rahman, R.L.; Jenkins, M.R.; Cobos, E.; Jumper, C.; Alalawi, R.; et al. Novel antigens in non-small cell lung cancer: Sp17, AKAP4, and PTTG1 are potential immunotherapeutic targets. Oncotarget 2015, 6, 2812–2826. [Google Scholar] [CrossRef] [PubMed]
- Xia, Q.Y.; Liu, S.; Li, F.Q.; Huang, W.B.; Shi, L.N.; Zhou, X.J. Sperm protein 17, mage-c1 and ny-eso-1 in hepatocellular carcinoma: Expression frequency and their correlation with clinical parameters. Int. J. Clin. Exp. Pathol. 2013, 6, 1610–1616. [Google Scholar] [CrossRef] [PubMed]
- Li, F.Q.; Liu, Q.; Han, Y.L.; Wu, B.; Yin, H.L. Sperm protein 17 is highly expressed in endometrial and cervical cancers. BMC Cancer 2010, 10, 429. [Google Scholar] [CrossRef] [PubMed]
- Xiang, S.D.; Gao, Q.; Wilson, K.L.; Heyerick, A.; Plebanski, M. A nanoparticle based sp17 peptide vaccine exposes new immuno-dominant and species cross-reactive b cell epitopes. Vaccines 2015, 3, 875–893. [Google Scholar] [CrossRef] [PubMed]
- Xiang, S.D.; Gao, Q.; Wilson, K.L.; Heyerick, A.; Plebanski, M. Mapping t and b cell epitopes in sperm protein 17 to support the development of an ovarian cancer vaccine. Vaccine 2015, 33, 5950–5959. [Google Scholar] [CrossRef] [PubMed]
- Alari-Pahissa, E.; Grandclement, C.; Jeevan-Raj, B.; Leclercq, G.; Veillette, A.; Held, W. Activation by slam family receptors contributes to nk cell mediated “missing-self” recognition. PLoS ONE 2016, 11, e0153236. [Google Scholar] [CrossRef] [PubMed]
- Chiriva-Internati, M.; Wang, Z.; Salati, E.; Bumm, K.; Barlogie, B.; Lim, S.H. Sperm protein 17 (sp17) is a suitable target for immunotherapy of multiple myeloma. Blood 2002, 100, 961–965. [Google Scholar] [CrossRef] [PubMed]
- Grizzi, F.; Chiriva-Internati, M.; Franceschini, B.; Bumm, K.; Colombo, P.; Ciccarelli, M.; Donetti, E.; Gagliano, N.; Hermonat, P.L.; Bright, R.K.; et al. Sperm protein 17 is expressed in human somatic ciliated epithelia. J. Histochem. Cytochem. 2004, 52, 549–554. [Google Scholar] [CrossRef] [PubMed]
- Classen, C.F.; Falk, C.S.; Friesen, C.; Fulda, S.; Herr, I.; Debatin, K.M. Natural killer resistance of a drug-resistant leukemia cell line, mediated by up-regulation of HLA class I expression. Haematologica 2003, 88, 509–521. [Google Scholar] [PubMed]
- Ljunggren, H.G.; Karre, K. Host resistance directed selectively against h-2-deficient lymphoma variants. Analysis of the mechanism. J. Exp. Med. 1985, 162, 1745–1759. [Google Scholar] [CrossRef] [PubMed]
- Vinay, D.S.; Ryan, E.P.; Pawelec, G.; Talib, W.H.; Stagg, J.; Elkord, E.; Lichtor, T.; Decker, W.K.; Whelan, R.L.; Kumara, H.; et al. Immune evasion in cancer: Mechanistic basis and therapeutic strategies. Semin. Cancer Biol. 2015, 35, S185–S198. [Google Scholar] [CrossRef] [PubMed]
- Andersson, E.; Villabona, L.; Bergfeldt, K.; Carlson, J.W.; Ferrone, S.; Kiessling, R.; Seliger, B.; Masucci, G.V. Correlation of HLA-a02* genotype and HLA class I antigen down-regulation with the prognosis of epithelial ovarian cancer. Cancer Immunol. Immunother. 2012, 61, 1243–1253. [Google Scholar] [CrossRef] [PubMed]
- Santoiemma, P.P.; Powell, D.J., Jr. Tumor infiltrating lymphocytes in ovarian cancer. Cancer Biol. Ther. 2015, 16, 807–820. [Google Scholar] [CrossRef] [PubMed]
- Kandalaft, L.E.; Motz, G.T.; Duraiswamy, J.; Coukos, G. Tumor immune surveillance and ovarian cancer: Lessons on immune mediated tumor rejection or tolerance. Cancer Metastasis Rev. 2011, 30, 141–151. [Google Scholar] [CrossRef] [PubMed]
- Yigit, R.; Massuger, L.F.; Figdor, C.G.; Torensma, R. Ovarian cancer creates a suppressive microenvironment to escape immune elimination. Gynecol. Oncol. 2010, 117, 366–372. [Google Scholar] [CrossRef] [PubMed]
- Vesely, M.D.; Kershaw, M.H.; Schreiber, R.D.; Smyth, M.J. Natural innate and adaptive immunity to cancer. Annu. Rev. Immunol. 2011, 29, 235–271. [Google Scholar] [CrossRef] [PubMed]
- Santoni, M.; Massari, F.; Del Re, M.; Ciccarese, C.; Piva, F.; Principato, G.; Montironi, R.; Santini, D.; Danesi, R.; Tortora, G.; et al. Investigational therapies targeting signal transducer and activator of transcription 3 for the treatment of cancer. Expert Opin. Investig. Drugs 2015, 24, 809–824. [Google Scholar] [CrossRef] [PubMed]
- Hamanishi, J.; Mandai, M.; Iwasaki, M.; Okazaki, T.; Tanaka, Y.; Yamaguchi, K.; Higuchi, T.; Yagi, H.; Takakura, K.; Minato, N.; et al. Programmed cell death 1 ligand 1 and tumor-infiltrating CD8+ T lymphocytes are prognostic factors of human ovarian cancer. Proc. Natl. Acad. Sci. USA 2007, 104, 3360–3365. [Google Scholar] [CrossRef] [PubMed]
- Webb, J.R.; Milne, K.; Kroeger, D.R.; Nelson, B.H. PD-L1 expression is associated with tumor-infiltrating T cells and favorable prognosis in high-grade serous ovarian cancer. Gynecol. Oncol. 2016, 141, 293–302. [Google Scholar] [CrossRef] [PubMed]
- Abiko, K.; Mandai, M.; Hamanishi, J.; Yoshioka, Y.; Matsumura, N.; Baba, T.; Yamaguchi, K.; Murakami, R.; Yamamoto, A.; Kharma, B.; et al. PD-L1 on tumor cells is induced in ascites and promotes peritoneal dissemination of ovarian cancer through CTL dysfunction. Clin. Cancer Res. 2013, 19, 1363–1374. [Google Scholar] [CrossRef] [PubMed]
- Atsaves, V.; Tsesmetzis, N.; Chioureas, D.; Kis, L.; Leventaki, V.; Drakos, E.; Panaretakis, T.; Grander, D.; Medeiros, L.J.; Young, K.H.; et al. PD-L1 is commonly expressed and transcriptionally regulated by STAT3 and myc in ALK-negative anaplastic large-cell lymphoma. Leukemia 2017, 31, 1633–1637. [Google Scholar] [CrossRef] [PubMed]
- Park, A.; Govindaraj, C.; Xiang, S.D.; Halo, J.; Quinn, M.; Scalzo-Inguanti, K.; Plebanski, M. Substantially modified ratios of effector to regulatory T cells during chemotherapy in ovarian cancer patients return to pre-treatment levels at completion: Implications for immunotherapy. Cancers 2012, 4, 581–600. [Google Scholar] [CrossRef] [PubMed]
- Madoux, F.; Koenig, M.; Sessions, H.; Nelson, E.; Mercer, B.A.; Cameron, M.; Roush, W.; Frank, D.; Hodder, P. Modulators of STAT transcription factors for the targeted therapy of cancer (STAT3 inhibitors). In Probe Reports from the Nih Molecular Libraries Program; National Center for Biotechnology Information: Bethesda, MD, USA, 2010. [Google Scholar]
- Yu, S.; Gong, L.S.; Li, N.F.; Pan, Y.F.; Zhang, L. Galangin (GG) combined with cisplatin (ddp) to suppress human lung cancer by inhibition of STAT3-regulated NF-kappaB and BCL-2/BAX signaling pathways. Biomed. Pharmacother. 2018, 97, 213–224. [Google Scholar] [CrossRef] [PubMed]
- Duan, Z.; Foster, R.; Bell, D.; Mahoney, J.; Wolak, K.; Valdya, A.; Hampel, C.; Lee, H.; Seiden, M. Signal transducers and activators of transcription 3 pathway activation in drug-resistant ovarian cancer. Clin. Cancer Res. 2006, 12, 5055–5063. [Google Scholar] [CrossRef] [PubMed]
- Walker, S.R.; Chaudhury, M.; Frank, D.A. STAT3 inhibition by microtubule-targeted drugs: Dual molecular effects of chemotherapeutic agents. Mol. Cell. Pharmacol. 2011, 3, 13–19. [Google Scholar] [PubMed]
- Su, W.P.; Lo, Y.C.; Yan, J.J.; Liao, I.C.; Tsai, P.J.; Wang, H.C.; Yeh, H.H.; Lin, C.C.; Chen, H.H.; Lai, W.W.; et al. Mitochondrial uncoupling protein 2 regulates the effects of paclitaxel on STAT3 activation and cellular survival in lung cancer cells. Carcinogenesis 2012, 33, 2065–2075. [Google Scholar] [CrossRef] [PubMed]
- Das, S.; Banerjee, S.; Majumder, S.; Chowdhury, B.P.; Goswami, A.; Halder, K.; Chakraborty, U.; Pal, N.K.; Majumdar, S. Immune subversion by mycobacterium tuberculosis through CCR5 mediated signaling: Involvement of IL-10. PLoS ONE 2014, 9, e92477. [Google Scholar] [CrossRef] [PubMed]
- Kozela, E.; Juknat, A.; Kaushansky, N.; Ben-Nun, A.; Coppola, G.; Vogel, Z. Cannabidiol, a non-psychoactive cannabinoid, leads to EGR2-dependent anergy in activated encephalitogenic T cells. J. Neuroinflamm. 2015, 12, 52. [Google Scholar] [CrossRef] [PubMed]
- Kitamura, H.; Kamon, H.; Sawa, S.; Park, S.J.; Katunuma, N.; Ishihara, K.; Murakami, M.; Hirano, T. IL-6-STAT3 controls intracellular MHC class II alphabeta dimer level through cathepsin s activity in dendritic cells. Immunity 2005, 23, 491–502. [Google Scholar] [CrossRef] [PubMed]
- Cancer Genome Atlas Research, N. Integrated genomic analyses of ovarian carcinoma. Nature 2011, 474, 609–615. [Google Scholar]
- Budczies, J.; Bockmayr, M.; Denkert, C.; Klauschen, F.; Groschel, S.; Darb-Esfahani, S.; Pfarr, N.; Leichsenring, J.; Onozato, M.L.; Lennerz, J.K.; et al. Pan-cancer analysis of copy number changes in programmed death-ligand 1 (PD-L1, CD274)—Associations with gene expression, mutational load and survival. Genes Chromosomes Cancer 2016, 55, 626–639. [Google Scholar] [CrossRef] [PubMed]
- Hamanishi, J.; Mandai, M.; Abiko, K.; Matsumura, N.; Baba, T.; Yoshioka, Y.; Kosaka, K.; Konishi, I. The comprehensive assessment of local immune status of ovarian cancer by the clustering of multiple immune factors. Clin. Immunol. 2011, 141, 338–347. [Google Scholar] [CrossRef] [PubMed]
- Wei, H.; Zhao, L.; Li, W.; Fan, K.; Qian, W.; Hou, S.; Wang, H.; Dai, M.; Hellstrom, I.; Hellstrom, K.E.; et al. Combinatorial PD-1 blockade and CD137 activation has therapeutic efficacy in murine cancer models and synergizes with cisplatin. PLoS ONE 2013, 8, e84927. [Google Scholar] [CrossRef] [PubMed]
- Dai, M.; Wei, H.; Yip, Y.Y.; Feng, Q.; He, K.; Popov, V.; Hellstrom, I.; Hellstrom, K.E. Long-lasting complete regression of established mouse tumors by counteracting th2 inflammation. J. Immunother. 2013, 36, 248–257. [Google Scholar] [CrossRef] [PubMed]
- Wei, H.; Zhao, L.; Hellstrom, I.; Hellstrom, K.E.; Guo, Y. Dual targeting of CD137 co-stimulatory and PD-1 co-inhibitory molecules for ovarian cancer immunotherapy. Oncoimmunology 2014, 3, e28248. [Google Scholar] [CrossRef] [PubMed]
- Disis, M.L.; Patel, M.R.; Pant, S.; Hamilton, E.P.; Lockhart, A.C.; Kelly, K.; Beck, J.T.; Gordon, M.S.; Weiss, G.J.; Taylor, M.H.; et al. Avelumab (msb0010718c; anti-PD-L1) in patients with recurrent/refractory ovarian cancer from the javelin solid tumor phase Ib trial: Safety and clinical activity. J. Clin. Oncol. 2016, 34, 5533. [Google Scholar]
- Filby, A.; Begum, J.; Jalal, M.; Day, W. Appraising the suitability of succinimidyl and lipophilic fluorescent dyes to track proliferation in non-quiescent cells by dye dilution. Methods 2015, 82, 29–37. [Google Scholar] [CrossRef] [PubMed]
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gao, Q.; Xiang, S.D.; Wilson, K.; Madondo, M.; Stephens, A.N.; Plebanski, M. Sperm Protein 17 Expression by Murine Epithelial Ovarian Cancer Cells and Its Impact on Tumor Progression. Cancers 2018, 10, 276. https://doi.org/10.3390/cancers10080276
Gao Q, Xiang SD, Wilson K, Madondo M, Stephens AN, Plebanski M. Sperm Protein 17 Expression by Murine Epithelial Ovarian Cancer Cells and Its Impact on Tumor Progression. Cancers. 2018; 10(8):276. https://doi.org/10.3390/cancers10080276
Chicago/Turabian StyleGao, Qian, Sue D. Xiang, Kirsty Wilson, Mutsa Madondo, Andrew N. Stephens, and Magdalena Plebanski. 2018. "Sperm Protein 17 Expression by Murine Epithelial Ovarian Cancer Cells and Its Impact on Tumor Progression" Cancers 10, no. 8: 276. https://doi.org/10.3390/cancers10080276
APA StyleGao, Q., Xiang, S. D., Wilson, K., Madondo, M., Stephens, A. N., & Plebanski, M. (2018). Sperm Protein 17 Expression by Murine Epithelial Ovarian Cancer Cells and Its Impact on Tumor Progression. Cancers, 10(8), 276. https://doi.org/10.3390/cancers10080276





