Allogeneic Hematopoietic Cell Transplantation for Older Adults with Acute Myeloid Leukemia
Abstract
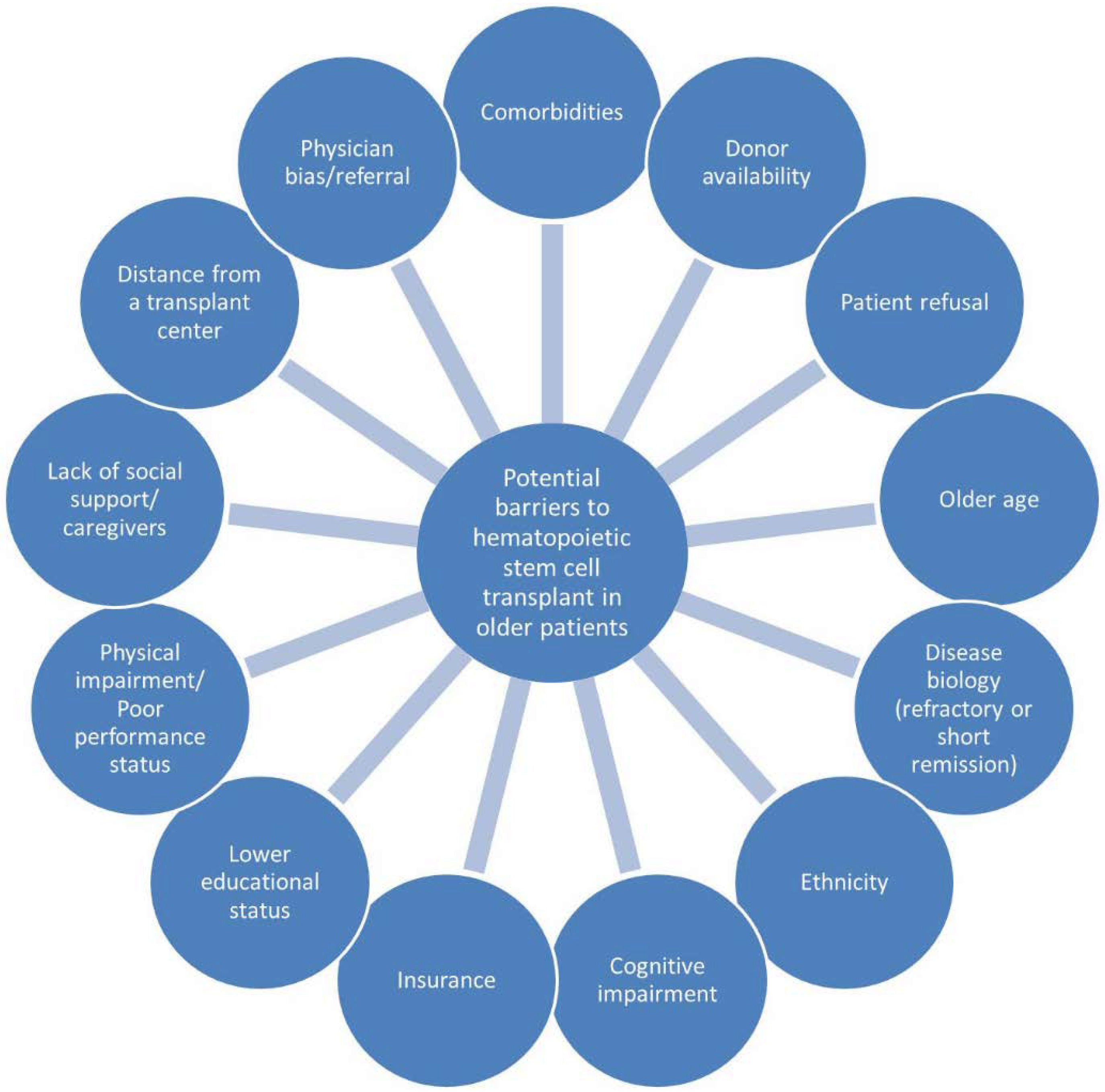
1. Introduction
2. Selection of Candidates for Hematopoietic Stem Cell Transplantation
3. Conditioning Regimens
4. Donor Sources for Older Transplant Recipients
5. Graft-Versus-Host Disease in the Older Transplant Patient
6. Post-Transplant Relapse
7. Research Gaps and Ongoing Studies
8. Conclusions
Funding
Conflicts of Interest
References
- Seer Cancer Statistics Factsheets: Acute Myeloid Leukemia; National Cancer Institute: Bethesda, MD, USA, 2018; Available online: http://seer.Cancer.Gov/statfacts/html/amyl.html (accessed on 1 June 2018).
- Menzin, J.; Lang, K.; Earle, C.C.; Kerney, D.; Mallick, R. The outcomes and costs of acute myeloid leukemia among the elderly. Arch. Intern. Med. 2002, 162, 1597–1603. [Google Scholar] [CrossRef] [PubMed]
- Leith, C.P.; Kopecky, K.J.; Chen, I.M.; Eijdems, L.; Slovak, M.L.; McConnell, T.S.; Head, D.R.; Weick, J.; Grever, M.R.; Appelbaum, F.R.; et al. Frequency and clinical significance of the expression of the multidrug resistance proteins MDR1/P-glycoprotein, MRP1, and LRP in acute myeloid leukemia: A southwest oncology group study. Blood 1999, 94, 1086–1099. [Google Scholar] [PubMed]
- Oran, B.; Weisdorf, D.J. Survival for older patients with acute myeloid leukemia: A population-based study. Haematologica 2012, 97, 1916–1924. [Google Scholar] [CrossRef] [PubMed]
- Koh, Y.; Kim, I.; Bae, J.Y.; Song, E.Y.; Kim, H.K.; Yoon, S.S.; Lee, D.S.; Park, S.S.; Park, M.H.; Park, S.; et al. Prognosis of secondary acute myeloid leukemia is affected by the type of the preceding hematologic disorders and the presence of trisomy 8. Jpn. J. Clin. Oncol. 2010, 40, 1037–1045. [Google Scholar] [CrossRef] [PubMed]
- Peccatori, J.; Ciceri, F. Allogeneic stem cell transplantation for acute myeloid leukemia. Haematologica 2010, 95, 857–859. [Google Scholar] [CrossRef] [PubMed]
- Podoltsev, N.A.; Stahl, M.; Zeidan, A.M.; Gore, S.D. Selecting initial treatment of acute myeloid leukaemia in older adults. Blood Rev. 2017, 31, 43–62. [Google Scholar] [CrossRef] [PubMed]
- Devine, S.M.; Owzar, K.; Blum, W.; Mulkey, F.; Stone, R.M.; Hsu, J.W.; Champlin, R.E.; Chen, Y.-B.; Vij, R.; Slack, J.; et al. Phase ii study of allogeneic transplantation for older patients with acute myeloid leukemia in first complete remission using a reduced-intensity conditioning regimen: Results from cancer and leukemia Group B 100103 (alliance for clinical trials in oncology)/blood and marrow transplant clinical trial network 0502. J. Clin. Oncol. 2015, 33, 4167–4175. [Google Scholar] [PubMed]
- Ustun, C.; Lazarus, H.M.; Weisdorf, D. To transplant or not: A dilemma for treatment of elderly AML patients in the twenty-first century. Bone Marrow Transplant. 2013, 48, 1497–1505. [Google Scholar] [CrossRef] [PubMed]
- Muffly, L.; Pasquini, M.C.; Martens, M.; Brazauskas, R.; Zhu, X.; Adekola, K.; Aljurf, M.; Ballen, K.K.; Bajel, A.; Baron, F.; et al. Increasing use of allogeneic hematopoietic cell transplantation in patients aged 70 years and older in the united states. Blood 2017, 130, 1156–1164. [Google Scholar] [CrossRef] [PubMed]
- Majhail, N.S.; Nayyar, S.; Santibanez, M.E.; Murphy, E.A.; Denzen, E.M. Racial disparities in hematopoietic cell transplantation in the united states. Bone Marrow Transplant. 2012, 47, 1385–1390. [Google Scholar] [CrossRef] [PubMed]
- Rashidi, A.; Ebadi, M.; Colditz, G.A.; DiPersio, J.F. Outcomes of allogeneic stem cell transplantation in elderly patients with acute myeloid leukemia: A systematic review and meta-analysis. Biol. Blood Marrow Transplant. 2016, 22, 651–657. [Google Scholar] [CrossRef] [PubMed]
- Lim, Z.; Brand, R.; Martino, R.; van Biezen, A.; Finke, J.; Bacigalupo, A.; Beelen, D.; Devergie, A.; Alessandrino, E.; Willemze, R.; et al. Allogeneic hematopoietic stem-cell transplantation for patients 50 years or older with myelodysplastic syndromes or secondary acute myeloid leukemia. J. Clin. Oncol. 2010, 28, 405–411. [Google Scholar] [CrossRef] [PubMed]
- Pohlen, M.; Groth, C.; Sauer, T.; Gorlich, D.; Mesters, R.; Schliemann, C.; Lenz, G.; Muller-Tidow, C.; Buchner, T.; Berdel, W.E.; et al. Outcome of allogeneic stem cell transplantation for AML and myelodysplastic syndrome in elderly patients (60 years). Bone Marrow Transplant. 2016, 51, 1441–1448. [Google Scholar] [CrossRef] [PubMed]
- Modi, D.; Deol, A.; Kim, S.; Ayash, L.; Alavi, A.; Ventimiglia, M.; Bhutani, D.; Ratanatharathorn, V.; Uberti, J.P. Age does not adversely influence outcomes among patients older than 60 years who undergo allogeneic hematopoietic stem cell transplant for AML and myelodysplastic syndrome. Bone Marrow Transplant. 2017, 52, 1530–1536. [Google Scholar] [CrossRef] [PubMed]
- Armand, P.; Kim, H.T.; Logan, B.R.; Wang, Z.; Alyea, E.P.; Kalaycio, M.E.; Maziarz, R.T.; Antin, J.H.; Soiffer, R.J.; Weisdorf, D.J.; et al. Validation and refinement of the disease risk index for allogeneic stem cell transplantation. Blood 2014, 123, 3664–3671. [Google Scholar] [CrossRef] [PubMed]
- Sorror, M.L.; Maris, M.B.; Storb, R.; Baron, F.; Sandmaier, B.M.; Maloney, D.G.; Storer, B. Hematopoietic cell transplantation (HCT)-specific comorbidity index: A new tool for risk assessment before allogeneic HCT. Blood 2005, 106, 2912–2919. [Google Scholar] [CrossRef] [PubMed]
- Muffly, L.S.; Kocherginsky, M.; Stock, W.; Chu, Q.; Bishop, M.R.; Godley, L.A.; Kline, J.; Liu, H.; Odenike, O.M.; Larson, R.A.; et al. Geriatric assessment to predict survival in older allogeneic hematopoietic cell transplantation recipients. Haematologica 2014, 99, 1373–1379. [Google Scholar] [CrossRef] [PubMed]
- Clough-Gorr, K.M.; Stuck, A.E.; Thwin, S.S.; Silliman, R.A. Older breast cancer survivors: Geriatric assessment domains are associated with poor tolerance of treatment adverse effects and predict mortality over 7 years of follow-up. J. Clin. Oncol. 2010, 28, 380–386. [Google Scholar] [CrossRef] [PubMed]
- Mohile, S.G.; Velarde, C.; Hurria, A.; Magnuson, A.; Lowenstein, L.; Pandya, C.; O’Donovan, A.; Gorawara-Bhat, R.; Dale, W. Geriatric assessment-guided care processes for older adults: A delphi consensus of geriatric oncology experts. J. Natl. Compr. Cancer Netw. 2015, 13, 1120–1130. [Google Scholar] [CrossRef]
- Extermann, M.; Boler, I.; Reich, R.R.; Lyman, G.H.; Brown, R.H.; DeFelice, J.; Levine, R.M.; Lubiner, E.T.; Reyes, P.; Schreiber, F.J.; et al. Predicting the risk of chemotherapy toxicity in older patients: The chemotherapy risk assessment scale for high-age patients (crash) score. Cancer 2012, 118, 3377–3386. [Google Scholar] [CrossRef] [PubMed]
- Artz, A.S. Biologic vs. physiologic age in the transplant candidate. In Hematology Am Soc Hematol Educ Program; ASH: Washington, DC, USA, 2016; Volume 2016, pp. 99–105. [Google Scholar]
- Rosko, A.; Artz, A. Aging: Treating the older patient. Biol. Blood Marrow Transplant. 2017, 23, 193–200. [Google Scholar] [CrossRef] [PubMed]
- Muffly, L.S.; Boulukos, M.; Swanson, K.; Kocherginsky, M.; Cerro, P.D.; Schroeder, L.; Pape, L.; Extermann, M.; Van Besien, K.; Artz, A.S. Pilot study of comprehensive geriatric assessment (CGA) in allogeneic transplant: CGA captures a high prevalence of vulnerabilities in older transplant recipients. Biol. Blood Marrow Transplant. 2013, 19, 429–434. [Google Scholar] [CrossRef] [PubMed]
- Jethava, Y.S.; Sica, S.; Savani, B.; Socola, F.; Jagasia, M.; Mohty, M.; Nagler, A.; Bacigalupo, A. Conditioning regimens for allogeneic hematopoietic stem cell transplants in acute myeloid leukemia. Bone Marrow Transplant. 2017, 52, 1504–1511. [Google Scholar] [CrossRef] [PubMed]
- Bacigalupo, A.; Ballen, K.; Rizzo, D.; Giralt, S.; Lazarus, H.; Ho, V.; Apperley, J.; Slavin, S.; Pasquini, M.; Sandmaier, B.M.; et al. Defining the intensity of conditioning regimens: Working definitions. Biol. Blood Marrow Transplant. 2009, 15, 1628–1633. [Google Scholar] [CrossRef] [PubMed]
- Cornelissen, J.J.; van Putten, W.L.; Verdonck, L.F.; Theobald, M.; Jacky, E.; Daenen, S.M.; van Marwijk Kooy, M.; Wijermans, P.; Schouten, H.; Huijgens, P.C.; et al. Results of a HOVON/SAKK donor versus no-donor analysis of myeloablative HLA-identical sibling stem cell transplantation in first remission acute myeloid leukemia in young and middle-aged adults: Benefits for whom? Blood 2007, 109, 3658–3666. [Google Scholar] [CrossRef] [PubMed]
- Aoudjhane, M.; Labopin, M.; Gorin, N.C.; Shimoni, A.; Ruutu, T.; Kolb, H.J.; Frassoni, F.; Boiron, J.M.; Yin, J.L.; Finke, J.; et al. Comparative outcome of reduced intensity and myeloablative conditioning regimen in hla identical sibling allogeneic haematopoietic stem cell transplantation for patients older than 50 years of age with acute myeloblastic leukaemia: A retrospective survey from the acute leukemia working party (ALWP) of the european group for blood and marrow transplantation (EBMT). Leukemia 2005, 19, 2304–2312. [Google Scholar] [PubMed]
- Luger, S.M.; Ringden, O.; Zhang, M.J.; Perez, W.S.; Bishop, M.R.; Bornhauser, M.; Bredeson, C.N.; Cairo, M.S.; Copelan, E.A.; Gale, R.P.; et al. Similar outcomes using myeloablative vs reduced-intensity allogeneic transplant preparative regimens for AML or MDS. Bone Marrow Transplant. 2012, 47, 203–211. [Google Scholar] [CrossRef] [PubMed]
- Shimoni, A.; Shem-Tov, N.; Volchek, Y.; Danylesko, I.; Yerushalmi, R.; Nagler, A. Allo-SCT for AML and MDS with treosulfan compared with BU-based regimens: Reduced toxicity vs. reduced intensity. Bone Marrow Transplant. 2012, 47, 1274–1282. [Google Scholar] [CrossRef] [PubMed]
- Scott, B.L.; Pasquini, M.C.; Logan, B.R.; Wu, J.; Devine, S.M.; Porter, D.L.; Maziarz, R.T.; Warlick, E.D.; Fernandez, H.F.; Alyea, E.P.; et al. Myeloablative versus reduced-intensity hematopoietic cell transplantation for acute myeloid leukemia and myelodysplastic syndromes. J. Clin. Oncol. 2017, 35, 1154–1161. [Google Scholar] [CrossRef] [PubMed]
- Sebert, M.; Porcher, R.; Robin, M.; Ades, L.; Boissel, N.; Raffoux, E.; Xhaard, A.; Dhedin, N.; Larghero, J.; Himberlin, C.; et al. Equivalent outcomes using reduced intensity or conventional myeloablative conditioning transplantation for patients aged 35 years and over with AML. Bone Marrow Transplant. 2015, 50, 74–81. [Google Scholar] [CrossRef] [PubMed]
- Martino, R.; de Wreede, L.; Fiocco, M.; van Biezen, A.; von dem Borne, P.A.; Hamladji, R.M.; Volin, L.; Bornhauser, M.; Robin, M.; Rocha, V.; et al. Comparison of conditioning regimens of various intensities for allogeneic hematopoietic SCT using HLA-identical sibling donors in AML and MDS with <10% BM blasts: A report from EBMT. Bone Marrow Transplant. 2013, 48, 761–770. [Google Scholar] [CrossRef] [PubMed]
- Ringden, O.; Erkers, T.; Aschan, J.; Garming-Legert, K.; Le Blanc, K.; Hagglund, H.; Omazic, B.; Svenberg, P.; Dahllof, G.; Mattsson, J.; et al. A prospective randomized toxicity study to compare reduced-intensity and myeloablative conditioning in patients with myeloid leukaemia undergoing allogeneic haematopoietic stem cell transplantation. J. Intern. Med. 2013, 274, 153–162. [Google Scholar] [CrossRef] [PubMed]
- Goyal, G.; Gundabolu, K.; Vallabhajosyula, S.; Silberstein, P.T.; Bhatt, V.R. Reduced-intensity conditioning allogeneic hematopoietic-cell transplantation for older patients with acute myeloid leukemia. Ther. Adv. Hematol. 2016, 7, 131–141. [Google Scholar] [CrossRef] [PubMed]
- Bornhauser, M.; Kienast, J.; Trenschel, R.; Burchert, A.; Hegenbart, U.; Stadler, M.; Baurmann, H.; Schafer-Eckart, K.; Holler, E.; Kroger, N.; et al. Reduced-intensity conditioning versus standard conditioning before allogeneic haemopoietic cell transplantation in patients with acute myeloid leukaemia in first complete remission: A prospective, open-label randomised phase 3 trial. Lancet Oncol. 2012, 13, 1035–1044. [Google Scholar] [CrossRef]
- McClune, B.L.; Weisdorf, D.J.; Pedersen, T.L.; Tunes da Silva, G.; Tallman, M.S.; Sierra, J.; Dipersio, J.; Keating, A.; Gale, R.P.; George, B.; et al. Effect of age on outcome of reduced-intensity hematopoietic cell transplantation for older patients with acute myeloid leukemia in first complete remission or with myelodysplastic syndrome. J. Clin. Oncol. 2010, 28, 1878–1887. [Google Scholar] [CrossRef] [PubMed]
- Zeng, W.; Huang, L.; Meng, F.; Liu, Z.; Zhou, J.; Sun, H. Reduced-intensity and myeloablative conditioning allogeneic hematopoietic stem cell transplantation in patients with acute myeloid leukemia and myelodysplastic syndrome: A meta-analysis and systematic review. Int. J. Clin. Exp. Med. 2014, 7, 4357–4368. [Google Scholar] [PubMed]
- Besse, K.; Maiers, M.; Confer, D.; Albrecht, M. On modeling human leukocyte antigen-identical sibling match probability for allogeneic hematopoietic cell transplantation: Estimating the need for an unrelated donor source. Biol. Blood Marrow Transplant. 2016, 22, 410–417. [Google Scholar] [CrossRef] [PubMed]
- Gragert, L.; Eapen, M.; Williams, E.; Freeman, J.; Spellman, S.; Baitty, R.; Hartzman, R.; Rizzo, J.D.; Horowitz, M.; Confer, D.; et al. HLA match likelihoods for hematopoietic stem-cell grafts in the U.S. Registry. N. Engl. J. Med. 2014, 371, 339–348. [Google Scholar] [CrossRef] [PubMed]
- Flowers, M.E.; Inamoto, Y.; Carpenter, P.A.; Lee, S.J.; Kiem, H.P.; Petersdorf, E.W.; Pereira, S.E.; Nash, R.A.; Mielcarek, M.; Fero, M.L.; et al. Comparative analysis of risk factors for acute graft-versus-host disease and for chronic graft-versus-host disease according to national institutes of health consensus criteria. Blood 2011, 117, 3214–3219. [Google Scholar] [CrossRef] [PubMed]
- Di Stasi, A.; Milton, D.R.; Poon, L.M.; Hamdi, A.; Rondon, G.; Chen, J.; Pingali, S.R.; Konopleva, M.; Kongtim, P.; Alousi, A.; et al. Similar transplantation outcomes for acute myeloid leukemia and myelodysplastic syndrome patients with haploidentical versus 10/10 human leukocyte antigen-matched unrelated and related donors. Biol. Blood Marrow Transplant. 2014, 20, 1975–1981. [Google Scholar] [CrossRef] [PubMed]
- Lee, C.J.; Savani, B.N.; Mohty, M.; Labopin, M.; Ruggeri, A.; Schmid, C.; Baron, F.; Esteve, J.; Gorin, N.C.; Giebel, S.; et al. Haploidentical hematopoietic cell transplantation for adult acute myeloid leukemia: A position statement from the acute leukemia working party of the European society for blood and marrow transplantation. Haematologica 2017, 102, 1810–1822. [Google Scholar] [CrossRef] [PubMed]
- Sandhu, K.S.; Brunstein, C.; DeFor, T.; Bejanyan, N.; Arora, M.; Warlick, E.; Weisdorf, D.; Ustun, C. Umbilical cord blood transplantation outcomes in acute myelogenous leukemia/myelodysplastic syndrome patients aged ≥ 70 years. Biol. Blood Marrow Transplant. 2016, 22, 390–393. [Google Scholar] [CrossRef] [PubMed]
- Mielcarek, M.; Storer, B.E.; Sandmaier, B.M.; Sorror, M.L.; Maloney, D.G.; Petersdorf, E.; Martin, P.J.; Storb, R. Comparable outcomes after nonmyeloablative hematopoietic cell transplantation with unrelated and related donors. Biol. Blood Marrow Transplant. 2007, 13, 1499–1507. [Google Scholar] [CrossRef] [PubMed]
- Schetelig, J.; Bornhauser, M.; Schmid, C.; Hertenstein, B.; Schwerdtfeger, R.; Martin, H.; Stelljes, M.; Hegenbart, U.; Schafer-Eckart, K.; Fussel, M.; et al. Matched unrelated or matched sibling donors result in comparable survival after allogeneic stem-cell transplantation in elderly patients with acute myeloid leukemia: A report from the cooperative German transplant study group. J. Clin. Oncol. 2008, 26, 5183–5191. [Google Scholar] [CrossRef] [PubMed]
- Ayuk, F.; Zabelina, T.; Wortmann, F.; Alchalby, H.; Wolschke, C.; Lellek, H.; Bacher, U.; Zander, A.; Kroger, N. Donor choice according to age for allo-SCT for AML in complete remission. Bone Marrow Transplant. 2013, 48, 1028–1032. [Google Scholar] [CrossRef] [PubMed]
- Servais, S.; Porcher, R.; Xhaard, A.; Robin, M.; Masson, E.; Larghero, J.; Ribaud, P.; Dhedin, N.; Abbes, S.; Sicre, F.; et al. Pre-transplant prognostic factors of long-term survival after allogeneic peripheral blood stem cell transplantation with matched related/unrelated donors. Haematologica 2014, 99, 519–526. [Google Scholar] [CrossRef] [PubMed]
- Friedman, J.S.; Alpdogan, O.; van den Brink, M.R.; Liu, C.; Hurwitz, D.; Boyd, A.; Kupper, T.S.; Burakoff, S.J. Increasing T-cell age reduces effector activity but preserves proliferative capacity in a murine allogeneic major histocompatibility complex-mismatched bone marrow transplant model. Biol. Blood Marrow Transplant. 2004, 10, 448–460. [Google Scholar] [CrossRef] [PubMed]
- Peffault de Latour, R.; Labopin, M.; Cornelissen, J.; Vigouroux, S.; Craddock, C.; Blaise, D.; Huyn, A.; Vindelov, L.; Maertens, J.; Chevallier, P.; et al. In patients older than 55 years with AML in first CR, should we search for a matched unrelated donor when an old sibling donor is available? Bone Marrow Transplant. 2015, 50, 1411–1415. [Google Scholar] [CrossRef] [PubMed]
- Howard, C.A.; Fernandez-Vina, M.A.; Appelbaum, F.R.; Confer, D.L.; Devine, S.M.; Horowitz, M.M.; Mendizabal, A.; Laport, G.G.; Pasquini, M.C.; Spellman, S.R. Recommendations for donor human leukocyte antigen assessment and matching for allogeneic stem cell transplantation: Consensus opinion of the blood and marrow transplant clinical trials network (BMT CTN). Biol. Blood Marrow Transplant. 2015, 21, 4–7. [Google Scholar] [CrossRef] [PubMed]
- Powles, R.L.; Morgenstern, G.R.; Kay, H.E.; McElwain, T.J.; Clink, H.M.; Dady, P.J.; Barrett, A.; Jameson, B.; Depledge, M.H.; Watson, J.G.; et al. Mismatched family donors for bone-marrow transplantation as treatment for acute leukaemia. Lancet 1983, 1, 612–615. [Google Scholar] [CrossRef]
- Beatty, P.G.; Clift, R.A.; Mickelson, E.M.; Nisperos, B.B.; Flournoy, N.; Martin, P.J.; Sanders, J.E.; Stewart, P.; Buckner, C.D.; Storb, R.; et al. Marrow transplantation from related donors other than HLA-identical siblings. N. Engl. J. Med. 1985, 313, 765–771. [Google Scholar] [CrossRef] [PubMed]
- Montoro, J.; Sanz, J.; Sanz, G.F.; Sanz, M.A. Advances in haploidentical stem cell transplantation for hematologic malignancies. Leuk. Lymphoma 2016, 57, 1766–1775. [Google Scholar] [CrossRef] [PubMed]
- Bashey, Z.A.; Zhang, X.; Brown, S.; Jackson, K.; Morris, L.E.; Holland, H.K.; Bashey, A.; Solomon, S.R.; Solh, M. Comparison of outcomes following transplantation with T-replete HLA-haploidentical donors using post-transplant cyclophosphamide to matched related and unrelated donors for patients with AML and MDS aged 60 years or older. Bone Marrow Transplant. 2018. [Google Scholar] [CrossRef] [PubMed]
- Ciurea, S.O.; Zhang, M.J.; Bacigalupo, A.A.; Bashey, A.; Appelbaum, F.R.; Aljitawi, O.S.; Armand, P.; Antin, J.H.; Chen, J.; Devine, S.M.; et al. Haploidentical transplant with posttransplant cyclophosphamide vs. matched unrelated donor transplant for acute myeloid leukemia. Blood 2015, 126, 1033–1040. [Google Scholar] [CrossRef] [PubMed]
- Rashidi, A.; DiPersio, J.F.; Westervelt, P.; Vij, R.; Schroeder, M.A.; Cashen, A.F.; Fehniger, T.A.; Romee, R. Comparison of outcomes after peripheral blood haploidentical versus matched unrelated donor allogeneic hematopoietic cell transplantation in patients with acute myeloid leukemia: A retrospective single-center review. Biol. Blood Marrow Transplant. 2016, 22, 1696–1701. [Google Scholar] [CrossRef] [PubMed]
- Rashidi, A.; Slade, M.; DiPersio, J.F.; Westervelt, P.; Vij, R.; Romee, R. Post-transplant high-dose cyclophosphamide after HLA-matched vs. haploidentical hematopoietic cell transplantation for AML. Bone Marrow Transplant. 2016, 51, 1561–1564. [Google Scholar] [CrossRef] [PubMed]
- Barker, J.N.; Krepski, T.P.; DeFor, T.E.; Davies, S.M.; Wagner, J.E.; Weisdorf, D.J. Searching for unrelated donor hematopoietic stem cells: Availability and speed of umbilical cord blood versus bone marrow. Biol. Blood Marrow Transplant. 2002, 8, 257–260. [Google Scholar] [CrossRef] [PubMed]
- Sanz, J.; Jaramillo, F.J.; Planelles, D.; Montesinos, P.; Lorenzo, I.; Moscardo, F.; Martin, G.; Lopez, F.; Martinez, J.; Jarque, I.; et al. Impact on outcomes of human leukocyte antigen matching by allele-level typing in adults with acute myeloid leukemia undergoing umbilical cord blood transplantation. Biol. Blood Marrow Transplant. 2014, 20, 106–110. [Google Scholar] [CrossRef] [PubMed]
- Weisdorf, D.; Eapen, M.; Ruggeri, A.; Zhang, M.J.; Zhong, X.; Brunstein, C.; Ustun, C.; Rocha, V.; Gluckman, E. Alternative donor transplantation for older patients with acute myeloid leukemia in first complete remission: A center for international blood and marrow transplant research-eurocord analysis. Biol. Blood Marrow Transplant. 2014, 20, 816–822. [Google Scholar] [CrossRef] [PubMed]
- Majhail, N.S.; Brunstein, C.G.; Shanley, R.; Sandhu, K.; McClune, B.; Oran, B.; Warlick, E.D.; Wagner, J.E.; Weisdorf, D.J. Reduced-intensity hematopoietic cell transplantation in older patients with AML/MDS: Umbilical cord blood is a feasible option for patients without HLA-matched sibling donors. Bone Marrow Transplant. 2012, 47, 494–498. [Google Scholar] [CrossRef] [PubMed]
- Nassereddine, S.; Rafei, H.; Elbahesh, E.; Tabbara, I. Acute graft versus host disease: A comprehensive review. Anticancer Res. 2017, 37, 1547–1555. [Google Scholar] [CrossRef] [PubMed]
- Jagasia, M.; Arora, M.; Flowers, M.E.; Chao, N.J.; McCarthy, P.L.; Cutler, C.S.; Urbano-Ispizua, A.; Pavletic, S.Z.; Haagenson, M.D.; Zhang, M.J.; et al. Risk factors for acute GVHD and survival after hematopoietic cell transplantation. Blood 2012, 119, 296–307. [Google Scholar] [CrossRef] [PubMed]
- Hahn, T.; McCarthy, P.L., Jr.; Zhang, M.J.; Wang, D.; Arora, M.; Frangoul, H.; Gale, R.P.; Hale, G.A.; Horan, J.; Isola, L.; et al. Risk factors for acute graft-versus-host disease after human leukocyte antigen-identical sibling transplants for adults with leukemia. J. Clin. Oncol. 2008, 26, 5728–5734. [Google Scholar] [CrossRef] [PubMed]
- Pidala, J.; Kurland, B.; Chai, X.; Majhail, N.; Weisdorf, D.J.; Pavletic, S.; Cutler, C.; Jacobsohn, D.; Palmer, J.; Arai, S.; et al. Patient-reported quality of life is associated with severity of chronic graft-versus-host disease as measured by NIH criteria: Report on baseline data from the chronic GVHD consortium. Blood 2011, 117, 4651–4657. [Google Scholar] [CrossRef] [PubMed]
- Sinding, C.; Wiernikowski, J.; Aronson, J. Cancer care from the perspectives of older women. Oncol. Nurs. Forum 2005, 32, 1169–1175. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.J.; Kim, H.T.; Ho, V.T.; Cutler, C.; Alyea, E.P.; Soiffer, R.J.; Antin, J.H. Quality of life associated with acute and chronic graft-versus-host disease. Bone Marrow Transplant. 2006, 38, 305–310. [Google Scholar] [CrossRef] [PubMed]
- Silvestri, G.; Pritchard, R.; Welch, H.G. Preferences for chemotherapy in patients with advanced non-small cell lung cancer: Descriptive study based on scripted interviews. BMJ 1998, 317, 771–775. [Google Scholar] [CrossRef] [PubMed]
- Wong, F.L.; Francisco, L.; Togawa, K.; Bosworth, A.; Gonzales, M.; Hanby, C.; Sabado, M.; Grant, M.; Forman, S.J.; Bhatia, S. Long-term recovery after hematopoietic cell transplantation: Predictors of quality-of-life concerns. Blood 2010, 115, 2508–2519. [Google Scholar] [CrossRef] [PubMed]
- Chiodi, S.; Spinelli, S.; Ravera, G.; Petti, A.R.; Van Lint, M.T.; Lamparelli, T.; Gualandi, F.; Occhini, D.; Mordini, N.; Berisso, G.; et al. Quality of life in 244 recipients of allogeneic bone marrow transplantation. Br. J. Haematol. 2000, 110, 614–619. [Google Scholar] [CrossRef] [PubMed]
- Bieri, S.; Roosnek, E.; Helg, C.; Verholen, F.; Robert, D.; Chapuis, B.; Passweg, J.; Miralbell, R.; Chalandon, Y. Quality of life and social integration after allogeneic hematopoietic SCT. Bone Marrow Transplant. 2008, 42, 819–827. [Google Scholar] [CrossRef] [PubMed]
- Hamilton, B.K.; Rybicki, L.; Dabney, J.; McLellan, L.; Haddad, H.; Foster, L.; Abounader, D.; Kalaycio, M.; Sobecks, R.; Dean, R.; et al. Quality of life and outcomes in patients 60 years of age after allogeneic hematopoietic cell transplantation. Bone Marrow Transplant. 2014, 49, 1426–1431. [Google Scholar] [CrossRef] [PubMed]
- Mielcarek, M.; Martin, P.J.; Leisenring, W.; Flowers, M.E.; Maloney, D.G.; Sandmaier, B.M.; Maris, M.B.; Storb, R. Graft-versus-host disease after nonmyeloablative versus conventional hematopoietic stem cell transplantation. Blood 2003, 102, 756–762. [Google Scholar] [CrossRef] [PubMed]
- Couriel, D.R.; Saliba, R.M.; Giralt, S.; Khouri, I.; Andersson, B.; de Lima, M.; Hosing, C.; Anderlini, P.; Donato, M.; Cleary, K.; et al. Acute and chronic graft-versus-host disease after ablative and nonmyeloablative conditioning for allogeneic hematopoietic transplantation. Biol. Blood Marrow Transplant. 2004, 10, 178–185. [Google Scholar] [CrossRef] [PubMed]
- Bashey, A.; Zhang, X.; Sizemore, C.A.; Manion, K.; Brown, S.; Holland, H.K.; Morris, L.E.; Solomon, S.R. T-cell-replete HLA-haploidentical hematopoietic transplantation for hematologic malignancies using post-transplantation cyclophosphamide results in outcomes equivalent to those of contemporaneous HLA-matched related and unrelated donor transplantation. J. Clin. Oncol. 2013, 31, 1310–1316. [Google Scholar] [CrossRef] [PubMed]
- Burroughs, L.M.; O’Donnell, P.V.; Sandmaier, B.M.; Storer, B.E.; Luznik, L.; Symons, H.J.; Jones, R.J.; Ambinder, R.F.; Maris, M.B.; Blume, K.G.; et al. Comparison of outcomes of HLA-matched related, unrelated, or HLA-haploidentical related hematopoietic cell transplantation following nonmyeloablative conditioning for relapsed or refractory Hodgkin lymphoma. Biol. Blood Marrow Transplant. 2008, 14, 1279–1287. [Google Scholar] [CrossRef] [PubMed]
- Raiola, A.M.; Dominietto, A.; di Grazia, C.; Lamparelli, T.; Gualandi, F.; Ibatici, A.; Bregante, S.; Van Lint, M.T.; Varaldo, R.; Ghiso, A.; et al. Unmanipulated haploidentical transplants compared with other alternative donors and matched sibling grafts. Biol. Blood Marrow Transplant. 2014, 20, 1573–1579. [Google Scholar] [CrossRef] [PubMed]
- Gutman, J.A.; Ross, K.; Smith, C.; Myint, H.; Lee, C.K.; Salit, R.; Milano, F.; Delaney, C.; Gao, D.; Pollyea, D.A. Chronic graft versus host disease burden and late transplant complications are lower following adult double cord blood versus matched unrelated donor peripheral blood transplantation. Bone Marrow Transplant. 2016, 51, 1588–1593. [Google Scholar] [CrossRef] [PubMed]
- Konuma, T.; Tsukada, N.; Kanda, J.; Uchida, N.; Ohno, Y.; Miyakoshi, S.; Kanamori, H.; Hidaka, M.; Sakura, T.; Onizuka, M.; et al. Comparison of transplant outcomes from matched sibling bone marrow or peripheral blood stem cell and unrelated cord blood in patients 50 years or older. Am. J. Hematol. 2016, 91, E284–E292. [Google Scholar] [CrossRef] [PubMed]
- Bensinger, W.I.; Martin, P.J.; Storer, B.; Clift, R.; Forman, S.J.; Negrin, R.; Kashyap, A.; Flowers, M.E.; Lilleby, K.; Chauncey, T.R.; et al. Transplantation of bone marrow as compared with peripheral-blood cells from hla-identical relatives in patients with hematologic cancers. N. Engl. J. Med. 2001, 344, 175–181. [Google Scholar] [CrossRef] [PubMed]
- Anasetti, C.; Logan, B.R.; Lee, S.J.; Waller, E.K.; Weisdorf, D.J.; Wingard, J.R.; Cutler, C.S.; Westervelt, P.; Woolfrey, A.; Couban, S.; et al. Peripheral-blood stem cells versus bone marrow from unrelated donors. N. Engl. J. Med. 2012, 367, 1487–1496. [Google Scholar] [CrossRef] [PubMed]
- Khoury, H.J.; Langston, A.A.; Kota, V.K.; Wilkinson, J.A.; Pusic, I.; Jillella, A.; Bauer, S.; Kim, A.S.; Roberts, D.; Al-Kadhimi, Z.; et al. Ruxolitinib: A steroid sparing agent in chronic graft-versus-host disease. Bone Marrow Transplant. 2018. [Google Scholar] [CrossRef] [PubMed]
- Jagasia, M.; Zeiser, R.; Arbushites, M.; Delaite, P.; Gadbaw, B.; Bubnoff, N.V. Ruxolitinib for the treatment of patients with steroid-refractory GVHD: An introduction to the reach trials. Immunotherapy 2018, 10, 391–402. [Google Scholar] [CrossRef] [PubMed]
- Miklos, D.; Cutler, C.S.; Arora, M.; Waller, E.K.; Jagasia, M.; Pusic, I.; Flowers, M.E.; Logan, A.C.; Nakamura, R.; Blazar, B.R.; et al. Ibrutinib for chronic graft-versus-host disease after failure of prior therapy. Blood 2017, 130, 2243–2250. [Google Scholar] [CrossRef] [PubMed]
- Schroeder, T.; Rachlis, E.; Bug, G.; Stelljes, M.; Klein, S.; Steckel, N.K.; Wolf, D.; Ringhoffer, M.; Czibere, A.; Nachtkamp, K.; et al. Treatment of acute myeloid leukemia or myelodysplastic syndrome relapse after allogeneic stem cell transplantation with azacitidine and donor lymphocyte infusions—A retrospective multicenter analysis from the German cooperative transplant study group. Biol. Blood Marrow Transplant. 2015, 21, 653–660. [Google Scholar] [CrossRef] [PubMed]
- Maury, S.; Lemoine, F.M.; Hicheri, Y.; Rosenzwajg, M.; Badoual, C.; Cherai, M.; Beaumont, J.L.; Azar, N.; Dhedin, N.; Sirvent, A.; et al. CD4+CD25+ regulatory t cell depletion improves the graft-versus-tumor effect of donor lymphocytes after allogeneic hematopoietic stem cell transplantation. Sci. Transl. Med. 2010, 2, 41ra52. [Google Scholar] [CrossRef] [PubMed]
- Bejanyan, N.; Weisdorf, D.J.; Logan, B.R.; Wang, H.L.; Devine, S.M.; de Lima, M.; Bunjes, D.W.; Zhang, M.J. Survival of patients with acute myeloid leukemia relapsing after allogeneic hematopoietic cell transplantation: A center for international blood and marrow transplant research study. Biol. Blood Marrow Transplant. 2015, 21, 454–459. [Google Scholar] [CrossRef] [PubMed]
- Shah, M.V.; Jorgensen, J.L.; Saliba, R.M.; Wang, S.A.; Alousi, A.M.; Andersson, B.S.; Bashir, Q.; Ciurea, S.O.; Kebriaei, P.; Marin, D.; et al. Early post-transplant minimal residual disease assessment improves risk stratification in acute myeloid leukemia. Biol. Blood Marrow Transplant. 2018, 23, S40–S41. [Google Scholar] [CrossRef]
- Platzbecker, U.; Middeke, J.M.; Sockel, K.; Mütherig, A.; Herbst, R.; Hänel, M.; Wolf, D.; Baldus, C.D.; Fransecky, L.; Noppeney, R.; et al. Minimal-residual disease guided treatment with azacitidine in MDS/AML patients at imminent risk of relapse: Results of the prospective relaza2 trial. Blood 2017, 130, 565. [Google Scholar]
- Kekre, N.; Kim, H.T.; Thanarajasingam, G.; Armand, P.; Antin, J.H.; Cutler, C.; Nikiforow, S.; Ho, V.T.; Koreth, J.; Alyea, E.P.; et al. Efficacy of immune suppression tapering in treating relapse after reduced intensity allogeneic stem cell transplantation. Haematologica 2015, 100, 1222–1227. [Google Scholar] [CrossRef] [PubMed]
- Bar, M.; Sandmaier, B.M.; Inamoto, Y.; Bruno, B.; Hari, P.; Chauncey, T.; Martin, P.J.; Storb, R.; Maloney, D.G.; Storer, B.; et al. Donor lymphocyte infusion for relapsed hematological malignancies after allogeneic hematopoietic cell transplantation: Prognostic relevance of the initial CD3+ T cell dose. Biol. Blood Marrow Transplant. 2013, 19, 949–957. [Google Scholar] [CrossRef] [PubMed]
- De Lima, M.; Giralt, S.; Thall, P.F.; de Padua Silva, L.; Jones, R.B.; Komanduri, K.; Braun, T.M.; Nguyen, H.Q.; Champlin, R.; Garcia-Manero, G. Maintenance therapy with low-dose azacitidine after allogeneic hematopoietic stem cell transplantation for recurrent acute myelogenous leukemia or myelodysplastic syndrome: A dose and schedule finding study. Cancer 2010, 116, 5420–5431. [Google Scholar] [CrossRef] [PubMed]
- Platzbecker, U.; Wermke, M.; Radke, J.; Oelschlaegel, U.; Seltmann, F.; Kiani, A.; Klut, I.M.; Knoth, H.; Rollig, C.; Schetelig, J.; et al. Azacitidine for treatment of imminent relapse in MDS or AML patients after allogeneic HSCT: Results of the RELAZA trial. Leukemia 2012, 26, 381–389. [Google Scholar] [CrossRef] [PubMed]
- Craddock, C.; Labopin, M.; Robin, M.; Finke, J.; Chevallier, P.; Yakoub-Agha, I.; Bourhis, J.H.; Sengelov, H.; Blaise, D.; Luft, T.; et al. Clinical activity of azacitidine in patients who relapse after allogeneic stem cell transplantation for acute myeloid leukemia. Haematologica 2016, 101, 879–883. [Google Scholar] [CrossRef] [PubMed]
- Pusic, I.; Choi, J.; Fiala, M.A.; Gao, F.; Holt, M.; Cashen, A.F.; Vij, R.; Abboud, C.N.; Stockerl-Goldstein, K.E.; Jacoby, M.A.; et al. Maintenance therapy with decitabine after allogeneic stem cell transplantation for acute myelogenous leukemia and myelodysplastic syndrome. Biol. Blood Marrow Transplant. 2015, 21, 1761–1769. [Google Scholar] [CrossRef] [PubMed]
- Schroeder, T.; Czibere, A.; Platzbecker, U.; Bug, G.; Uharek, L.; Luft, T.; Giagounidis, A.; Zohren, F.; Bruns, I.; Wolschke, C.; et al. Azacitidine and donor lymphocyte infusions as first salvage therapy for relapse of AML or MDS after allogeneic stem cell transplantation. Leukemia 2013, 27, 1229–1235. [Google Scholar] [CrossRef] [PubMed]
- Schroeder, T.; Rautenberg, C.; Kruger, W.; Platzbecker, U.; Bug, G.; Steinmann, J.; Klein, S.; Hopfer, O.; Nachtkamp, K.; Kondakci, M.; et al. Treatment of relapsed AML and MDS after allogeneic stem cell transplantation with decitabine and DLI-a retrospective multicenter analysis on behalf of the German cooperative transplant study group. Ann. Hematol. 2018, 97, 335–342. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.B.; Li, S.; Lane, A.A.; Connolly, C.; Del Rio, C.; Valles, B.; Curtis, M.; Ballen, K.; Cutler, C.; Dey, B.R.; et al. Phase I trial of maintenance sorafenib after allogeneic hematopoietic stem cell transplantation for FMS-like tyrosine kinase 3 internal tandem duplication acute myeloid leukemia. Biol. Blood Marrow Transplant. 2014, 20, 2042–2048. [Google Scholar] [CrossRef] [PubMed]
- Schlenk, R.; Döhner, K.; Salih, H.; Kündgen, A.; Fiedler, W.; Salwender, H.-J.; Westermann, J.; Götze, K.S.; Horst, H.-A.; Wulf, G.; et al. Midostaurin in combination with intensive induction and as single agent maintenance therapy after consolidation therapy with allogeneic hematopoietic stem cell transplantation or high-dose cytarabine. Blood 2015, 126, 322. [Google Scholar]
- DiNardo, C.D.; Rausch, C.R.; Benton, C.; Kadia, T.; Jain, N.; Pemmaraju, N.; Daver, N.; Covert, W.; Marx, K.R.; Mace, M.; et al. Clinical experience with the Bcl2-inhibitor venetoclax in combination therapy for relapsed and refractory acute myeloid leukemia and related myeloid malignancies. Am. J. Hematol. 2018, 93, 401–407. [Google Scholar] [CrossRef] [PubMed]
- Stein, E.M.; DiNardo, C.D.; Pollyea, D.A.; Fathi, A.T.; Roboz, G.J.; Altman, J.K.; Stone, R.M.; DeAngelo, D.J.; Levine, R.L.; Flinn, I.W.; et al. Enasidenib in mutant IDH2 relapsed or refractory acute myeloid leukemia. Blood 2017, 130, 722–731. [Google Scholar] [CrossRef] [PubMed]
- Davids, M.S.; Kim, H.T.; Bachireddy, P.; Costello, C.; Liguori, R.; Savell, A.; Lukez, A.P.; Avigan, D.; Chen, Y.B.; McSweeney, P.; et al. Ipilimumab for patients with relapse after allogeneic transplantation. N. Engl. J. Med. 2016, 375, 143–153. [Google Scholar] [CrossRef] [PubMed]
- Goodyear, O.C.; Dennis, M.; Jilani, N.Y.; Loke, J.; Siddique, S.; Ryan, G.; Nunnick, J.; Khanum, R.; Raghavan, M.; Cook, M.; et al. Azacitidine augments expansion of regulatory T cells after allogeneic stem cell transplantation in patients with acute myeloid leukemia (AML). Blood 2012, 119, 3361–3369. [Google Scholar] [CrossRef] [PubMed]
- El-Jawahri, A.; Pidala, J.; Inamoto, Y.; Chai, X.; Khera, N.; Wood, W.A.; Cutler, C.; Arora, M.; Carpenter, P.A.; Palmer, J.; et al. Impact of age on quality of life, functional status, and survival in patients with chronic graft-versus-host disease. Biol. Blood Marrow Transplant. 2014, 20, 1341–1348. [Google Scholar] [CrossRef] [PubMed]
- Grunwald, M.R.; Zimmerman, M.K.A.; Boselli, D.; Bohannon, L.M.; Robinson, M.M.; Peters, D.T.; Ai, J.; Knight, T.G.; Trivedi, J.; Plesca, D.; et al. Frontline azacitidine as a bridge to allogeneic transplantation in acute myeloid leukemia. Blood 2017, 130, 3864. [Google Scholar]
| Study | Study Design | N | Conditioning Regimens | Age, Median (Range) | OS | NRM/TRM, % | Relapse, % | Acute GVHD, % | Chronic GVHD, % |
|---|---|---|---|---|---|---|---|---|---|
| Aoudjhane 2005 [28] | Retrospective | 722 | MAC RIC | 54 (50–64) 57 (50–73) | 46 47 (2-year) | 32 18 (2-year) | 24 41 (2-year) | 31 22 (Grade II-IV, 100-day) | 56 48 (Any, 2-year) |
| Luger 2014 a [29] | Retrospective | 5179 (AML: 3834) | MAC RIC BM RIC PBSC NMA | 42 (18–68) 51 (19–69) 56 (18–70) 57 (18–70) | 34 33 33 26 (5-year) | 18 16 12 13 (3-month; 35% for all by year 5) | 32 42 39 43 (5-year) | 47 41 45 47 (Grade II-IV, 100-day) | 41 31 45 41 (Any, 5-year) |
| Shimoni 2012 a [30] | Prospective | 85 (AML: 50) | MAC RIC | 49 (18–66) 60 (29–75) | 50 44 (5-year) | 18 20 (5-year) | 40 47 (5-year) | 37 25 (Grade II-IV, 5-year) | 60 47 (Any, 5-year) |
| Scott 2017 a [31] | Randomized controlled trial | 272 (AML: 218) | MAC RIC | 55 (22–66) 55 (22–66) | 76 b 63 b (18-month) | 16 b 4 b (18-month) | 16 b 51 b (18-month) | 45 c 29 c (Grade II-IV, 100-day) | 64 c 48 c (Any, 18-month) |
| Sebert 2015 [32] | Prospective | 132 | MAC RIC | 44 (35–56) 54 (37–66) | 43 50 (4-year) | 28 21 (4-year) | 33 38 (4-year) | 61 35 (Grade II-IV, 120-day) | - - |
| Martino 2013 a [33] | Retrospective | 878 (AML: 864) | HyperMAC ConvMAC RIC NMA | 37 (18–60) 39 (21–63) 54 (19–70) 56 (41–72) | 51 d 56 d 53 d 29 d (7-year) | 27 e 22 e 18 e 29 e (7-year) | 22 f 24 f 34 f 46 f (7-year) | 29 27 25 30 (Any, 120-day) | 47 44 48 50 (Any, 7-year) |
| Donor Source | Benefits | Limitations |
|---|---|---|
| Matched-sibling | Low rates of graft rejection. Lower risk of GVHD compared to unrelated donor | Low availability of donors, especially in the older population [39] |
| Matched-unrelated | Increased availability of donors. | Donor search time can be prolonged [40] May be extremely difficult to identify donor in ethnic/racial minorities [11] Higher rates of GVHD when compared to matched sibling [41] |
| Haploidentical | Increased and rapid availability of donors. Lower cost than UCB and unrelated donors Availability of donor for future lymphocyte/stem cell donations. Comparable overall survival, relapse rates, and non-relapse mortality to other donor sources [42] | Increased risk of graft rejection Risk of severe GVHD without adequate post-transplant prophylaxis (this is somewhat mitigated by use of post-transplant cyclophosphamide) [43] |
| Umbilical cord blood | Lower rates of acute and chronic GVHD Donor search time is lower than matched unrelated donors [11] | Delayed engraftment Increased risk of graft failure Lower quantity of harvest, need for adequate cell dose. Higher rates of infections due to slower engraftment and immune reconstitution [44] No availability of future donor lymphocytes/stem cells |
| Intervention | Examples |
|---|---|
| Early tapering of immune suppression [91] | |
| Graded donor lymphocyte infusion (DLI) dosing [92] | |
| Hypomethylating agents | Azacitidine [93,94,95] |
| Decitabine [96] | |
| Hypomethylating agents and DLI [97,98] | |
| FMS-like tyrosine kinase (FLT) 3 inhibitors | Sorafenib [99] |
| Midostaurin [100] | |
| Possibly B-cell lymphoma 2 (Bcl-2) inhibitors | Venetoclax [101] |
| Possibly Isocitrate dehydrogenase (IDH) 2 inhibitors | Enasidenib [102] |
| Possibly checkpoint inhibitors or cytotoxic T-lymphocyte-associated protein (CTLA) 4 antagonists | Ipilimumab [103] |
| Regulatory T cells (Tregs) with azacitidine [104] |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lipof, J.J.; Loh, K.P.; O’Dwyer, K.; Liesveld, J.L. Allogeneic Hematopoietic Cell Transplantation for Older Adults with Acute Myeloid Leukemia. Cancers 2018, 10, 179. https://doi.org/10.3390/cancers10060179
Lipof JJ, Loh KP, O’Dwyer K, Liesveld JL. Allogeneic Hematopoietic Cell Transplantation for Older Adults with Acute Myeloid Leukemia. Cancers. 2018; 10(6):179. https://doi.org/10.3390/cancers10060179
Chicago/Turabian StyleLipof, Jodi J., Kah Poh Loh, Kristen O’Dwyer, and Jane L. Liesveld. 2018. "Allogeneic Hematopoietic Cell Transplantation for Older Adults with Acute Myeloid Leukemia" Cancers 10, no. 6: 179. https://doi.org/10.3390/cancers10060179
APA StyleLipof, J. J., Loh, K. P., O’Dwyer, K., & Liesveld, J. L. (2018). Allogeneic Hematopoietic Cell Transplantation for Older Adults with Acute Myeloid Leukemia. Cancers, 10(6), 179. https://doi.org/10.3390/cancers10060179




