1. Introduction
The incidence of head and neck squamous cell carcinoma (HNSCC) in Taiwan is different from that in Western countries. Betel nut chewing is endemic to Taiwan and is observed in >90% patients with HNSCC in Taiwan [
1,
2,
3,
4,
5]. Betel nut chewing results in a high risk of local recurrence and second primary HNSCC in patients with HNSCC in Taiwan [
1,
2,
3,
4,
5]. Due to betel nut chewing, the proportion of oral cavity and nonoral cavity cancers in patients with HNSCC in Taiwan is approximately 66 and 34%, respectively [
1,
2,
3,
4,
5,
6]. The proportion of oral cavity cancers in patients with HNSCC is higher in Taiwan than in other countries [
1,
2,
3,
4,
5,
6]. Treatments for Taiwanese patients with HNSCC might be complicated, and the frequency of reirradiation is higher in Taiwan than in areas where betel nut chewing is not endemic [
1,
2,
3,
4,
5]. Therefore, comprehensive curative surgery is the main treatment (accounting for 64.09% of all HNSCC treatments) for patients with HNSCC in Taiwan [
1,
2,
3,
4,
5,
6]. In addition, in Taiwan, at initial diagnosis, >50% of HNSCC cases are locoregionally advanced (Stages III and IV) HNSCC (LA-HNSCC) [
6]. Instead of RT or chemotherapy, the initial treatment for LA-HNSCC is surgical resection of the primary tumor and neck dissection, followed by postoperative radiotherapy (RT) or concurrent chemoradiotherapy (CCRT).
Curative head and neck surgery, including radical neck dissection, is associated with a mortality rate of 1.5–8.5% [
7,
8,
9,
10,
11]; however, the time interval between curative surgery and mortality has not been specified in the literature. Most studies on curative surgery for HNSCC were published in the 1970s to 1980s [
7,
8,
9,
10,
11]; however, surgical techniques have improved considerably in the past 20 years [
12,
13]. Definitive data on mortality rates after curative surgery for LA-HNSCC in the past 20 years, particularly in Asia, are not available. In this study, we estimated mortality rates after curative surgery in patients with LA-HNSCC between 2006 and 2015. A new comorbidity score to predict mortality rates in patients with LA-HNSCC receiving curative surgery was also proposed because modern RT techniques, chemotherapy regimens, induction chemotherapy, and immune therapy might be more suitable alternative curative-intent treatments than curative surgery for high-mortality risk patients with LA-HNSCC [
2,
3,
4,
14,
15,
16,
17,
18,
19,
20,
21].
The mean age of patients with HNSCC in Taiwan has been reported to be 55 years; consequently, the patients are generally individuals who provide the main economic support to their families [
1,
2,
3,
4,
5,
6]. We hope to reduce mortality rates after aggressive treatments in LA-HNSCC and propose a new comorbidity score to preoperatively predict 90-day mortality and overall survival in patients with LA-HNSCC who will receive curative surgery. The new comorbidity score can be used to determine whether curative surgery or other curative-intent aggressive treatments are the optimal treatment [
2,
4,
14,
15,
16,
17,
18,
19,
20,
21].
3. Results
Table 1 shows a comparison of demographic characteristics and mortality rates within 90 days after curative surgery between the mortality and survival groups. Significant differences were observed between the groups in the age (
p < 0.0001), sex (
p < 0.0001), and comorbidities, such as DM, HTN, and pneumonia.
Table 2 and
Table 3 present the relative risk for each variable estimated using univariate and multivariate Cox proportional hazard models. Fewer variables were significant in the multivariate model than in the univariate model, indicating strong collinearity between the variables. Therefore, we used a stepwise method in the multivariate model for variable selection (
Table 4). Among comorbidities, significant variables were HTN, pneumonia, COPD, sepsis, heart failure, DIC, ARDS, dementia, mild to severe liver disease, hemiplegia, moderate or severe renal disease, any tumor, and metastatic solid tumor.
The WCS was calculated using significant variables other than HTN because the HR of HTN was <1. Although HTN increased the risk of outcomes, which can be observed from univariate analysis (
Table 2, HR = 1.356, CI: 1.211–1.518), collinearity in multivariate model may have reduced the HR to <1 (
Table 4, HR = 0.803, CI: 0.708–0.91), which is a common statistical phenomenon. We calculated the WCS by adding points according to the HR of each risk factor. The points of each risk factor were assigned as the largest integer less than or equal to its HR (last column in
Table 4); for example, 2 points for pneumonia with an HR of 2.092 and 3 points for ARDS with an HR of 3.897. In the WSC, a high number of points were assigned to risk factors with high relative mortality risk within 90 days. In our study, the minimum and maximum values of the WCS were 0 and 18+, respectively. We collapsed the range of the WCS into 4 strata, namely the low-risk (WCS, 0–6; 90-day mortality rate, 0–1.57%), intermediate-risk (7–11; 2.71–9.99%), high-risk (12–16, 17.30–20.00%), and very-high-risk (17 to 18+; 46.15–50.00%) strata (
Table 4). We used the CCI for scoring to predict 90-day mortality compared with the current scoring system (
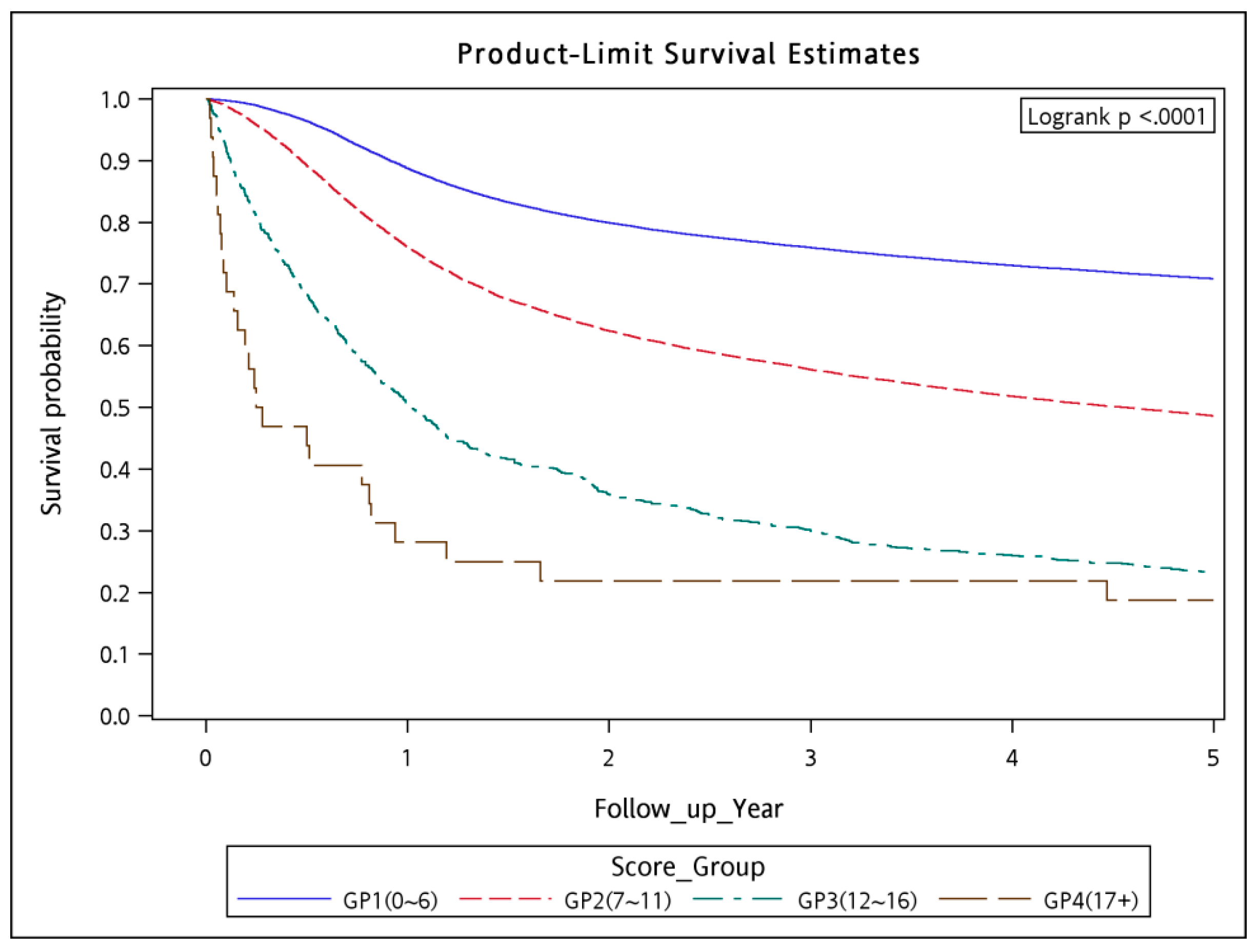
Figures S1 and S2), the risk groups of CCI were not feasible for predicting 90-day mortality in LA-HNSCC patients receiving curative surgery and could not reach statistical significance. In addition, there were scarcely LA-HNSCC patients with ASA classifications I and IV–V receiving curative surgery in our database. Therefore, we cannot use ASA classifications I–V to predict 90-day mortality in LA-HNSCC patients receiving curative surgery. The 90-day mortality rate tended to increase as the WCS increased, indicating the accuracy of the WCS. The 90-day mortality rate and five-year survival in the patients were estimated using the Kaplan–Meier method to analyze the risk of mortality associated with the 4 risk strata (
Figure 1 and
Figure 2). The 90-day survival rates were 98.97, 95.85, 81.20, and 53.13% in the low-, intermediate-, high-, and very-high-risk strata, respectively (log-rank test
p < 0.0001;
Figure 1). The five-year overall survival rates were 70.86, 48.62, 22.99, and 18.75% in the low-, intermediate-, high-, and very-high-risk strata, respectively (log-rank
p < 0.0001;
Figure 2).
4. Discussion
According to the Taiwan Cancer Registry report, 2017 edition [
6], >90% of curative surgery procedures for LA-HNSCC are conducted in top-ranking medical centers. The ranking is based on accreditation of hospitals in Taiwan into 4 levels since 1988 (medical center, regional hospitals, local hospital, clinics); the accreditation grade affects the service quality and specific patient volume of the hospital [
6]. Most curative surgery procedures were performed in hospitals with sufficient patient volume (>100 newly diagnosed patients with LA-HNSCC per year), thus leading to consistent patient outcomes for LA-HNSCC in Taiwan [
6,
27,
28,
29,
30]. Therefore, in Taiwan, the overall mortality rate within 90 days after curative surgery in the patients with LA-HNSCC was only 2.34% (
Table 5) after consultation with a professional head and neck surgeon and anesthesia consultation. In
Table 1, <3% of the patients with LA-HNSCC and heart valve dysfunctions and <5% of the patients with LA-HNSCC and moderate or severe liver disease received curative surgery. Surgeons were unwilling to perform curative surgery on the patients with LA-HNSCC and ESRD. However, the 90-day mortality remained 2.34%. Therefore, we wanted to develop a highly accurate predictor score to estimate mortality rates after curative surgery because CCRT or induction chemotherapy, followed by CCRT, might be an alternative treatment for patients with LA-HNSCC [
3,
4]. The proportion of the patients with LA-HNSCC with smoking habit (approximately 90%) in
Table 1 was consistent with that reported in previous studies in Taiwan [
1,
2,
3,
4,
5]. The 90-day mortality rate was proportional to age, particularly in the patients aged >70 years. This is the first study to show that age is a predictor of 90-day mortality in the patients with LA-HNSCC after curative surgery (
Table 1,
Table 2 and
Table 3).
Univariate and multivariate analyses revealed that age is an independent predictor of 90-day mortality after curative surgery in patients (
Table 2 and
Table 3). The male patients had higher 90-day mortality risk than did the female patients after curative surgery. These findings are consistent with those of previous studies, which reported endpoints different from those in our study [
31,
32]. From
Table 3, the patients with LA-HNSCC and pneumonia or COPD exhibited high mortality rates. These findings are consistent with those of previous studies [
33,
34]. However, this is the first study to demonstrate that preoperative pneumonia increases mortality rates in the patients with LA-HNSCC who received curative surgery. Notably, although heart valve dysfunction, MI, CVA, TIA, angina, or CADs were listed as risk factors in the ASA Physical Status Classification System before surgery [
24,
26], these factors were not risk factors for 90-day mortality in our study. This discrepancy can be explained by our inclusion of comorbidities observed >6 months before the index date and exclusion of the comorbidities observed within 6 months of the index date. The mortality rates of these acute vascular diseases might decrease considerably after 6 months of having these diseases [
35,
36,
37,
38]. This is the first study to show the absence of correlations between heart valve dysfunction, MI, CVA, TIA, angina, or CADs and 90-day mortality rates associated with curative surgery in the patients with LA-HNSCC. These findings are reliable references for head and neck surgeons in the future.
Notably, HF, DIC, and ARDS were independent risk factors for 90-day mortality, even when comorbidities observed 6 months before the index date were included. This is because HF, DIC, and ARDS are chronic diseases [
39,
40,
41,
42] and not acute vascular diseases. In addition, HF, DIC, and ARDS were also listed in the ASA Physical Status Classification System before surgery [
24,
26]. Dementia and hemiplegia might affect self-care by patients who receive surgery and might result in an increased mortality rate after surgery [
31,
43]. Our study is the first to demonstrate that dementia and hemiplegia were independent risk factors for mortality in the patients with LA-HNSCC who had received head and neck curative surgery. Head and neck surgeons should carefully consider curative surgery for patients with LA-HNSCC and dementia or hemiplegia. Furthermore, in our study, liver disease or renal disease were independent risk factors for 90-day mortality. Moreover, Cramer et al. showed that liver disease increases the risk of perioperative mortality in patients with HNSCC, and this risk should be carefully considered during surgical decision-making and postoperative care [
44]. ESRD was also an independent 90-day mortality risk factor in our study; this finding is consistent with the results of a previous study, which reported a slightly different endpoint from ours [
45]. However, leukemia and lymphoma were not risk factors for 90-day mortality in our study (
Table 3). Most patients with leukemia or lymphoma have long survival durations of >1 year [
46,
47]; this long survival duration might explain why leukemia and lymphoma did not affect 90-day mortality in our study. By contrast, non-HNSCC cancer with or without metastasis was an independent risk factor for 90-day mortality (
Table 3). This result may be attributable to the weakening of overall physical health, immunity, and the hematological system owing to previous cancer treatments, such as systemic chemotherapy, major surgical procedures, or RT, which increase 90-day mortality rates because of systemic infection complications, hospitalizations, and uncontrolled coagulation or hematological problems [
48,
49,
50]. For patients with LA-HNSCC and non-HNSCC cancer with or without metastasis, alternative curative-intent aggressive treatments might be considered [
4].
In our analysis, the WCS corresponded with not only the 90-day mortality rates but also with the overall survival rates (
Figure 2). Developing a new comorbidity score for predicting 90-day mortality in patients with LA-HNSCC who will receive curative surgery is currently valuable because of the evolution of contemporary chemotherapy, RT techniques, target therapy, or immunotherapy, particularly in the past 10 years [
14,
15,
16,
17,
18,
19,
20,
21]. An increasing number of alternative curative-intent aggressive treatments are available for patients with LA-HNSCC [
14,
15,
16,
17,
18,
19,
20,
21].
Because the development of new surgical procedures has minimized surgical morbidity and mortality, the contraindications to curative surgery for LA-HNSCC remain controversial [
12,
13]. However, patients with LA-HNSCC who have a high surgical risk because of comorbidities and whose condition cannot be optimized preoperatively should not be considered for new surgical procedures. Even after treatment by a professional head and neck surgeon and careful anesthesia consultation, the 90-day mortality in Taiwan remained at 2.34% from 2006 to 2015 (
Table 5). The WCS can serve as a valuable tool for preoperative prediction of the risk associated with curative surgery in patients with LA-HNSCC. After predicting the risk, other alternative curative-intent aggressive treatments can be considered in LA-HNSCC patients [
4,
14,
15,
16,
17,
18,
19,
20,
21].
To the best of our knowledge, our study is the first to use a comorbidity score to predict the 90-day mortality in the patients with LA-HNSCC who had received curative surgery. Figures revealed significant differences between low-, intermediate-, high-, and very-high-risk strata. These findings suggest that the WCS is a valid and specific tool for predicting 90-day and overall mortality in patients with LA-HNSCC who will receive curative surgery. Our literature review also revealed that our study also had the largest sample size among the studies that have proposed new comorbidity scores in the past 10 years.
This study has some limitations. First, the morbidity of curative surgery could not be determined because of differences in the levels of experience among surgeons and across hospitals; therefore, head and neck curative-surgery-related mortality estimates may have been biased. However, the Taiwan Cancer Registry report, 2017 edition, revealed that curative surgery for LA-HNSCC in Taiwan are mostly performed in hospitals with high patient volumes and large medical centers [
6]. Therefore, the outcomes of head and neck curative surgery would be consistent in Taiwan. Second, because all the patients with LA-HNSCC were enrolled from an Asian population and all the surgical procedures were performed by Taiwanese surgeons, the corresponding ethnic and regional susceptibility to this disease remain unclear; hence, our results should be cautiously extrapolated to non-Asian populations. Third, the diagnoses of all comorbid conditions were based on ICD-9-CM codes. However, the Taiwan Cancer Registry administration randomly reviews charts and interviews patients to verify the accuracy of the diagnoses. Hospitals with outlier chargers or practices may be audited and be subsequently heavily penalized if malpractice or discrepancies are identified. In addition, the quality and precision of ICD-9-CM codes in Taiwan have been verified and proven by previous studies [
51,
52]. Therefore, to obtain accurate information on population specificity and disease occurrence, large-scale randomized trials that compare carefully selected patients who had received suitable treatments are required. Fourth, we have scarcely very-high WCS patients (32/55,080 = 0.05%) and scarcely high WCS patients (<1000/55,080 = 1.60%). For remaining more than 98 percent of the patients only two risk strata groups are left and this is the same problem what we have with the ASA II and ASA III patients. Nevertheless, the individual 90-day mortality can be predicted upon the findings of this study because a big cancer registry supports this data. Finally, the TCRD does not contain information on dietary habits, socioeconomic status, or body mass index, which may all be risk factors for mortality. However, considering the magnitude and statistical significance of the effects observed in this study, these limitations are unlikely to have affected the conclusions.