Druggable Nucleolin Identifies Breast Tumours Associated with Poor Prognosis That Exhibit Different Biological Processes
Abstract
:1. Introduction
2. Results
2.1. Association of NCL mRNA Levels with Overall and Disease-Free Survival in Breast Cancer
2.2. Characterization of Breast Tumours Expressing Either Low or High NCL mRNA Levels
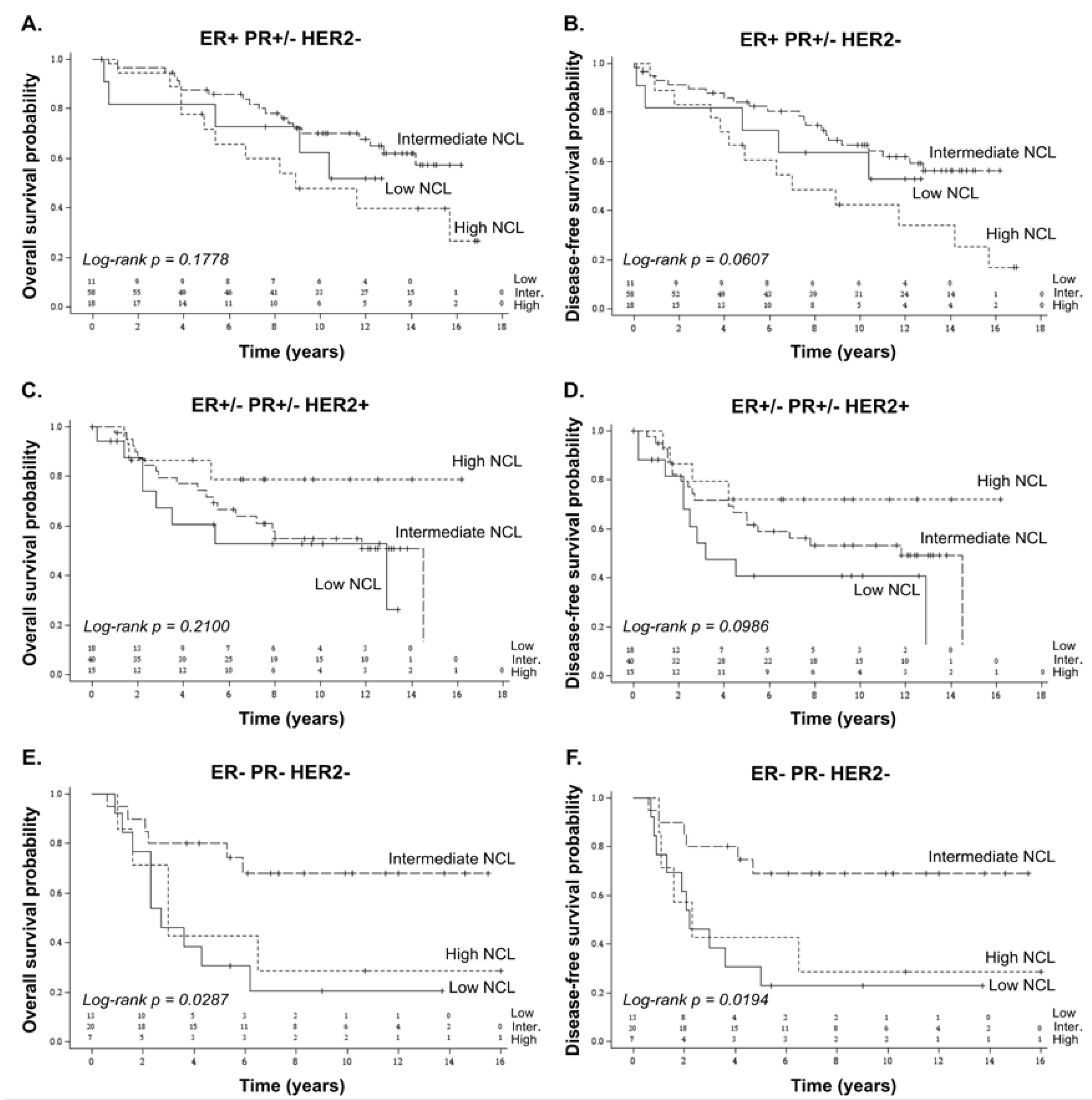
2.3. Prognostic Value of NCL in Breast Cancer Subtypes
3. Discussion
4. Materials and Methods
4.1. Human Breast Cancer and Healthy Donor Samples
4.2. RT-qPCR Analyses
4.3. Statistical Analyses
4.4. Transcriptomic Analyses
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Cardoso, F.; Harbeck, N.; Barrios, C.H.; Bergh, J.; Cortés, J.; El Saghir, N.; Francis, P.A.; Hudis, C.A.; Ohno, S.; Partridge, A.H.; et al. Research needs in breast cancer. Ann. Oncol. 2017, 28, 208–217. [Google Scholar] [CrossRef] [PubMed]
- Berger, C.M.; Gaume, X.; Bouvet, P. The roles of nucleolin subcellular localization in cancer. Biochimie 2015, 113, 78–85. [Google Scholar] [CrossRef] [PubMed]
- Gilles, M.-E.; Maione, F.; Cossutta, M.; Carpentier, G.; Caruana, L.; Di Maria, S.; Houppe, C.; Destouches, D.; Shchors, K.; Prochasson, C.; et al. Nucleolin Targeting Impairs the Progression of Pancreatic Cancer and Promotes the Normalization of Tumor Vasculature. Cancer Res. 2016, 76, 7181–7193. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ugrinova, I.; Petrova, M.; Chalabi-Dchar, M.; Bouvet, P. Multifaceted Nucleolin Protein and Its Molecular Partners in Oncogenesis. Adv. Protein Chem. Struct. Biol. 2018, 111, 133–164. [Google Scholar] [PubMed]
- Hovanessian, A.G.; Soundaramourty, C.; El Khoury, D.; Nondier, I.; Svab, J.; Krust, B. Surface expressed nucleolin is constantly induced in tumor cells to mediate calcium-dependent ligand internalization. PLoS ONE 2010, 5, e15787. [Google Scholar] [CrossRef] [PubMed]
- Gregório, A.C.; Lacerda, M.; Figueiredo, P.; Simões, S.; Dias, S.; Moreira, J.N. Meeting the needs of breast cancer: A nucleolin’s perspective. Crit. Rev. Oncol. Hematol. 2018, 125, 89–101. [Google Scholar] [CrossRef] [PubMed]
- Rosenberg, J.E.; Bambury, R.M.; Van Allen, E.M.; Drabkin, H.A.; Lara, P.N.; Harzstark, A.L.; Wagle, N.; Figlin, R.A.; Smith, G.W.; Garraway, L.A.; et al. A phase II trial of AS1411 (a novel nucleolin-targeted DNA aptamer) in metastatic renal cell carcinoma. Investig. New Drugs 2014, 32, 178–187. [Google Scholar] [CrossRef] [PubMed]
- Palmieri, D.; Richmond, T.; Piovan, C.; Sheetz, T.; Zanesi, N.; Troise, F.; James, C.; Wernicke, D.; Nyei, F.; Gordon, T.J.; et al. Human anti-nucleolin recombinant immunoagent for cancer therapy. Proc. Natl. Acad. Sci. USA 2015, 112, 9418–9423. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Christian, S.; Pilch, J.; Akerman, M.E.; Porkka, K.; Laakkonen, P.; Ruoslahti, E. Nucleolin expressed at the cell surface is a marker of endothelial cells in angiogenic blood vessels. J. Cell Biol. 2003, 163, 871–878. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Destouches, D.; El Khoury, D.; Hamma-Kourbali, Y.; Krust, B.; Albanese, P.; Katsoris, P.; Guichard, G.; Briand, J.P.; Courty, J.; Hovanessian, A.G. Suppression of tumor growth and angiogenesis by a specific antagonist of the cell-surface expressed nucleolin. PLoS ONE 2008, 3, e2518. [Google Scholar] [CrossRef] [PubMed]
- Guo, X.; Xiong, L.; Yu, L.; Li, R.; Wang, Z.; Ren, B.; Dong, J.; Li, B.; Wang, D. Increased level of nucleolin confers to aggressive tumor progression and poor prognosis in patients with hepatocellular carcinoma after hepatectomy. Diagn. Pathol. 2014, 9, 175. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Marcel, V.; Catez, F.; Berger, C.M.; Perrial, E.; Plesa, A.; Thomas, X.; Mattei, E.; Hayette, S.; Saintigny, P.; Bouvet, P.; et al. Expression Profiling of Ribosome Biogenesis Factors Reveals Nucleolin as a Novel Potential Marker to Predict Outcome in AML Patients. PLoS ONE 2017, 12, e0170160. [Google Scholar] [CrossRef] [PubMed]
- Qiu, W.; Zhou, F.; Zhang, Q.; Sun, X.; Shi, X.; Liang, Y.; Wang, X.; Yue, L. Overexpression of nucleolin and different expression sites both related to the prognosis of gastric cancer. APMIS 2013, 121, 919–925. [Google Scholar] [CrossRef] [PubMed]
- Wolfson, E.; Goldenberg, M.; Solomon, S.; Frishberg, A.; Pinkas-Kramarski, R. Nucleolin-binding by ErbB2 enhances tumorigenicity of ErbB2-positive breast cancer. Oncotarget 2016, 7, 65320–65334. [Google Scholar] [CrossRef] [PubMed]
- Pichiorri, F.; Palmieri, D.; De Luca, L.; Consiglio, J.; You, J.; Rocci, A.; Talabere, T.; Piovan, C.; Lagana, A.; Cascione, L.; et al. In vivo NCL targeting affects breast cancer aggressiveness through miRNA regulation. J. Exp. Med. 2013, 210, 951–968. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Peng, L.; Liang, J.; Wang, H.; Song, X.; Rashid, A.; Gomez, H.F.; Corley, L.J.; Abbruzzese, J.L.; Fleming, J.B.; Evans, D.B.; et al. High levels of nucleolar expression of nucleolin are associated with better prognosis in patients with stage II pancreatic ductal adenocarcinoma. Clin. Cancer Res. 2010, 16, 3734–3742. [Google Scholar] [CrossRef] [PubMed]
- Ridley, L.; Rahman, R.; Brundler, M.-A.; Ellison, D.; Lowe, J.; Robson, K.; Prebble, E.; Luckett, I.; Gilbertson, R.J.; Parkes, S.; et al. Children’s Cancer and Leukaemia Group Biological Studies Committee Multifactorial analysis of predictors of outcome in pediatric intracranial ependymoma. Neuro Oncol. 2008, 10, 675–689. [Google Scholar] [CrossRef] [PubMed]
- Zhao, H.; Huang, Y.; Xue, C.; Chen, Y.; Hou, X.; Guo, Y.; Zhao, L.; Hu, Z.H.; Huang, Y.; Luo, Y.; et al. Prognostic significance of the combined score of endothelial expression of nucleolin and CD31 in surgically resected non-small cell lung cancer. PLoS ONE 2013, 8, e54674. [Google Scholar] [CrossRef] [PubMed]
- Kumar, S.; Gomez, E.C.; Chalabi-Dchar, M.; Rong, C.; Das, S.; Ugrinova, I.; Gaume, X.; Monier, K.; Mongelard, F.; Bouvet, P. Integrated analysis of mRNA and miRNA expression in HeLa cells expressing low levels of Nucleolin. Sci. Rep. 2017, 7, 9017. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gaume, X.; Tassin, A.-M.; Ugrinova, I.; Mongelard, F.; Monier, K.; Bouvet, P. Centrosomal nucleolin is required for microtubule network organization. Cell Cycle 2015, 14, 902–919. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bourdon, J.-C.; Khoury, M.P.; Diot, A.; Baker, L.; Fernandes, K.; Aoubala, M.; Quinlan, P.; Purdie, C.A.; Jordan, L.B.; Prats, A.-C.; et al. p53 mutant breast cancer patients expressing p53γ have as good a prognosis as wild-type p53 breast cancer patients. Breast Cancer Res. 2011, 13, R7. [Google Scholar] [CrossRef] [PubMed]
- Ciriello, G.; Gatza, M.L.; Beck, A.H.; Wilkerson, M.D.; Rhie, S.K.; Pastore, A.; Zhang, H.; McLellan, M.; Yau, C.; Kandoth, C.; et al. Comprehensive Molecular Portraits of Invasive Lobular Breast Cancer. Cell 2015, 163, 506–519. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Maire, V.; Baldeyron, C.; Richardson, M.; Tesson, B.; Vincent-Salomon, A.; Gravier, E.; Marty-Prouvost, B.; De Koning, L.; Rigaill, G.; Dumont, A.; et al. TTK/hMPS1 is an attractive therapeutic target for triple-negative breast cancer. PLoS ONE 2013, 8, e63712. [Google Scholar] [CrossRef] [PubMed]
- Huang, D.W.; Sherman, B.T.; Lempicki, R.A. Systematic and integrative analysis of large gene lists using DAVID bioinformatics resources. Nat. Protoc. 2009, 4, 44–57. [Google Scholar] [CrossRef] [PubMed]
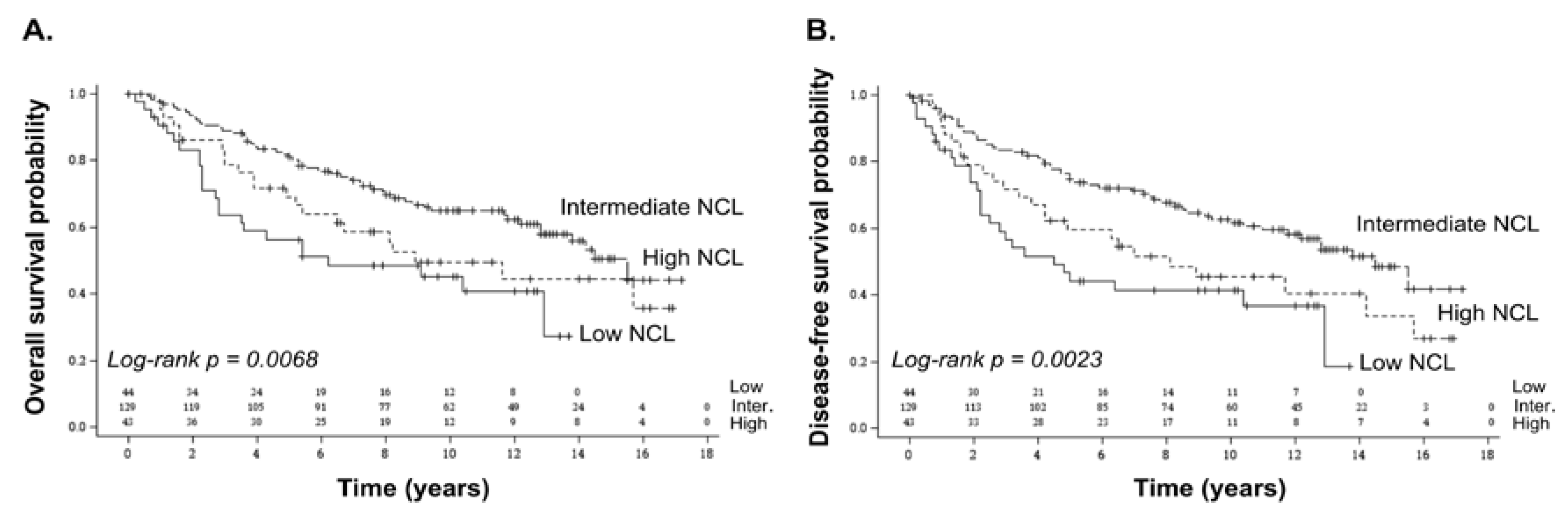
| Characteristics | All Samples (n = 216) | Low NCL (n = 44) | Intermediate NCL (n = 129) | High NCL (n = 43) | p-Value |
|---|---|---|---|---|---|
| Age | |||||
| Median | 59.0 | 58.5 | 59.0 | 60.0 | 0.557 # |
| (min–max) | (28–90) | (28–78) | (31–90) | (35–88) | |
| Invasive grade | |||||
| No data | 5 | 2 | 2 | 1 | |
| Grade 1 | 14 (6.6%) | 0 (0.0%) | 8 (6.3%) | 6 (14.3%) | 0.013 $ |
| Grade 2 | 59 (28.0%) | 7 (16.7%) | 42 (33.1%) | 10 (23.8%) | |
| Grade 3 | 138 (65.4%) | 35 (83.3%) | 77 (60.6%) | 26 (61.9%) | |
| Tumour size (mm) | |||||
| n | 204 | 42 | 120 | 42 | |
| Median | 28.0 | 28.5 | 28.0 | 26.0 | 0.641 # |
| (min–max) | (5–110) | (11–80) | (7–110) | (5–110) | |
| Invaded lymph node | |||||
| No (n = 0) | 98 (45.4%) | 21 (47.7%) | 58 (45.0%) | 19 (44.2%) | 0.936 † |
| Yes (n ≥ 1) | 118 (54.6%) | 23 (52.3%) | 71 (55.0%) | 24 (55.8%) | |
| ER status | |||||
| No data | 2 | 0 | 2 | 0 | |
| Negative | 70 (32.7%) | 25 (56.8%) | 34 (26.8%) | 11 (25.6%) | <0.001 † |
| Positive | 144 (67.3%) | 19 (43.2%) | 93 (73.2%) | 32 (74.4%) | |
| PR status | |||||
| No data | 2 | 0 | 2 | 0 | |
| Negative | 117 (54.7%) | 31 (70.5%) | 58 (45.7%) | 28 (65.1%) | 0.005 † |
| Positive | 97 (45.3%) | 13 (29.5%) | 69 (54.3%) | 15 (34.9%) | |
| HER2 status | |||||
| No data | 16 | 2 | 11 | 3 | |
| Negative | 127 (63.5%) | 24 (57.1%) | 78 (66.1%) | 25 (62.5%) | 0.579 † |
| Positive | 73 (36.5%) | 18 (42.9%) | 40 (33.9%) | 15 (37.5%) | |
| Intrinsic breast cancer subtype | |||||
| No data | 16 | 2 | 11 | 3 | |
| ER+ PR+/− HER2− | 87 (43.5%) | 11 (26.2%) | 58 (49.2%) | 18 (45.0%) | 0.105 † |
| ER+/− PR+/− HER2+ | 73 (36.5%) | 18 (42.9%) | 40 (33.9%) | 15 (37.5%) | |
| ER− PR− HER2− | 40 (20.0%) | 13 (31.0%) | 20 (16.9%) | 7 (17.5%) | |
| Variable | Overall Survival | Disease-Free Survival | |||||
|---|---|---|---|---|---|---|---|
| HR | 95% CI | p-Value | HR | 95% CI | p-Value | ||
| NCL | Intermediate | 1.00 | 1.00 | ||||
| Low | 2.31 | (1.39–3.85) | 2.32 | (1.43–3.78) | |||
| High | 1.55 | (0.93–2.58) | 0.0044 | 1.67 | (1.03–2.71) | 0.0019 | |
| Tumour size | <30 mm | 1.00 | 1.00 | ||||
| ≥30 mm | 2.53 | (1.66–3.85) | <0.0001 | 2.17 | (1.46–3.23) | 0.0001 | |
| Invaded lymph nodes | n = 0 | 1.00 | 1.00 | ||||
| n ≥ 1 | 1.60 | (1.04–2.46) | 0.0320 | 1.68 | (1.12–2.53) | 0.0130 | |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nguyen Van Long, F.; Lardy-Cleaud, A.; Bray, S.; Chabaud, S.; Dubois, T.; Diot, A.; Jordan, L.B.; Thompson, A.M.; Bourdon, J.-C.; Perol, D.; et al. Druggable Nucleolin Identifies Breast Tumours Associated with Poor Prognosis That Exhibit Different Biological Processes. Cancers 2018, 10, 390. https://doi.org/10.3390/cancers10100390
Nguyen Van Long F, Lardy-Cleaud A, Bray S, Chabaud S, Dubois T, Diot A, Jordan LB, Thompson AM, Bourdon J-C, Perol D, et al. Druggable Nucleolin Identifies Breast Tumours Associated with Poor Prognosis That Exhibit Different Biological Processes. Cancers. 2018; 10(10):390. https://doi.org/10.3390/cancers10100390
Chicago/Turabian StyleNguyen Van Long, Flora, Audrey Lardy-Cleaud, Susan Bray, Sylvie Chabaud, Thierry Dubois, Alexandra Diot, Lee B. Jordan, Alastair M. Thompson, Jean-Christophe Bourdon, David Perol, and et al. 2018. "Druggable Nucleolin Identifies Breast Tumours Associated with Poor Prognosis That Exhibit Different Biological Processes" Cancers 10, no. 10: 390. https://doi.org/10.3390/cancers10100390
APA StyleNguyen Van Long, F., Lardy-Cleaud, A., Bray, S., Chabaud, S., Dubois, T., Diot, A., Jordan, L. B., Thompson, A. M., Bourdon, J.-C., Perol, D., Bouvet, P., Diaz, J.-J., & Marcel, V. (2018). Druggable Nucleolin Identifies Breast Tumours Associated with Poor Prognosis That Exhibit Different Biological Processes. Cancers, 10(10), 390. https://doi.org/10.3390/cancers10100390









