Microfluidic-Based Technique for Measuring RBC Aggregation and Blood Viscosity in a Continuous and Simultaneous Fashion
Abstract
1. Introduction
2. Materials and Methods
2.1. Blood Sample Preparation
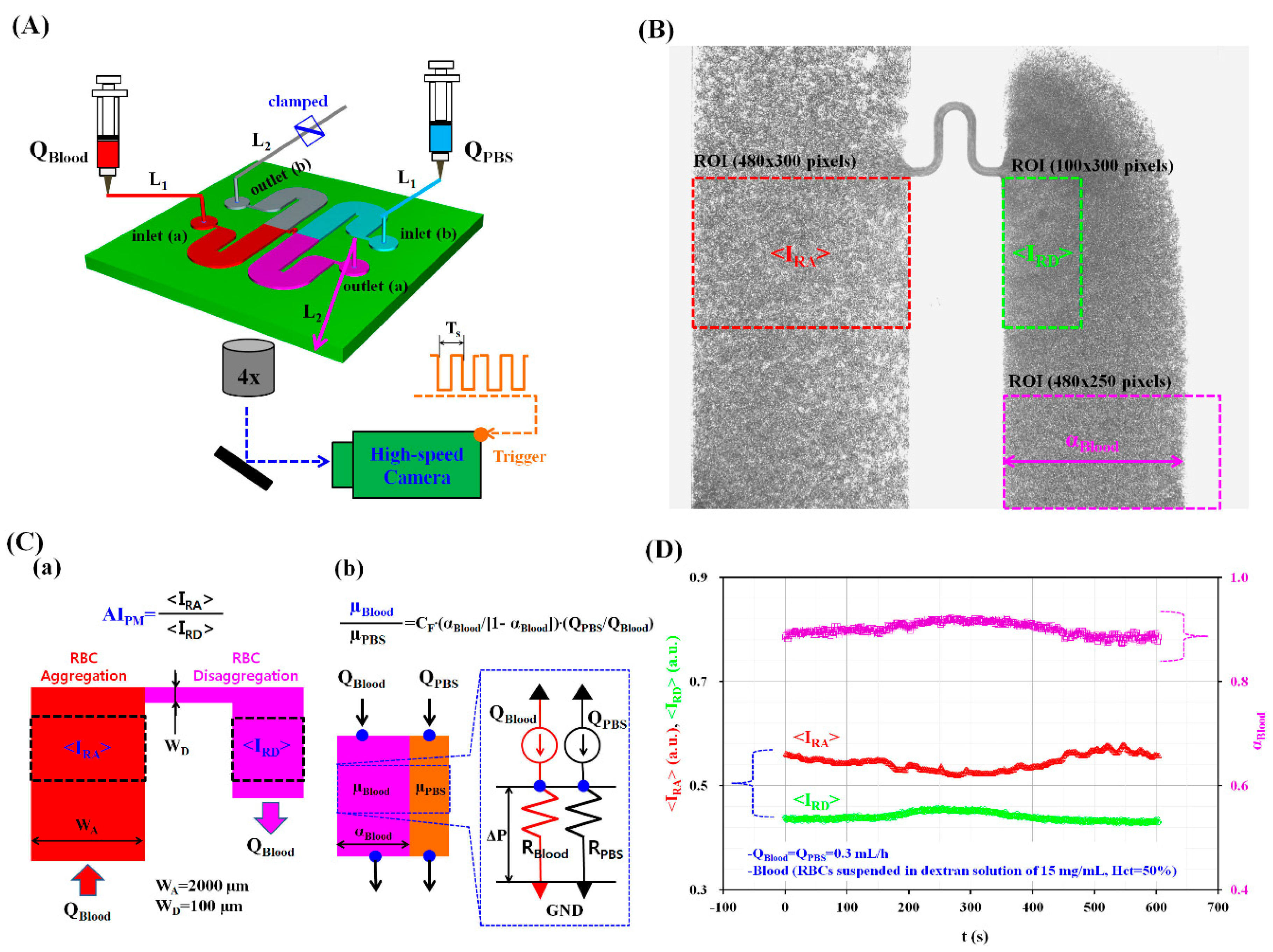
2.2. Fabrication of a Microfluidic Device and Experimental Procedure
2.3. Quantification of Image Intensity and Blood Flow-Rate
2.4. Simultaneous Measurement of RBC Aggregation and Blood Viscosity in Continuous Blood Flows
3. Results and Discussion
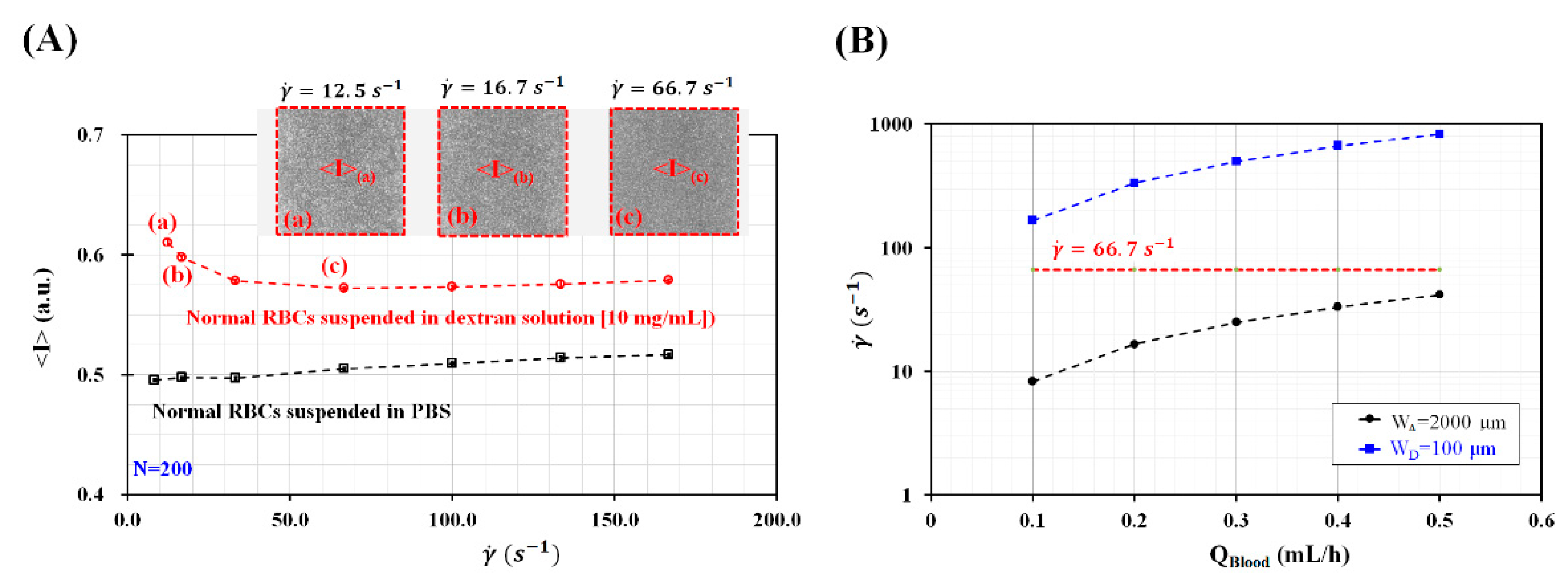
3.1. Determination of Channel Widths for Inducing and Disaggregating RBCs under Constant Value of Blood Flow Rates
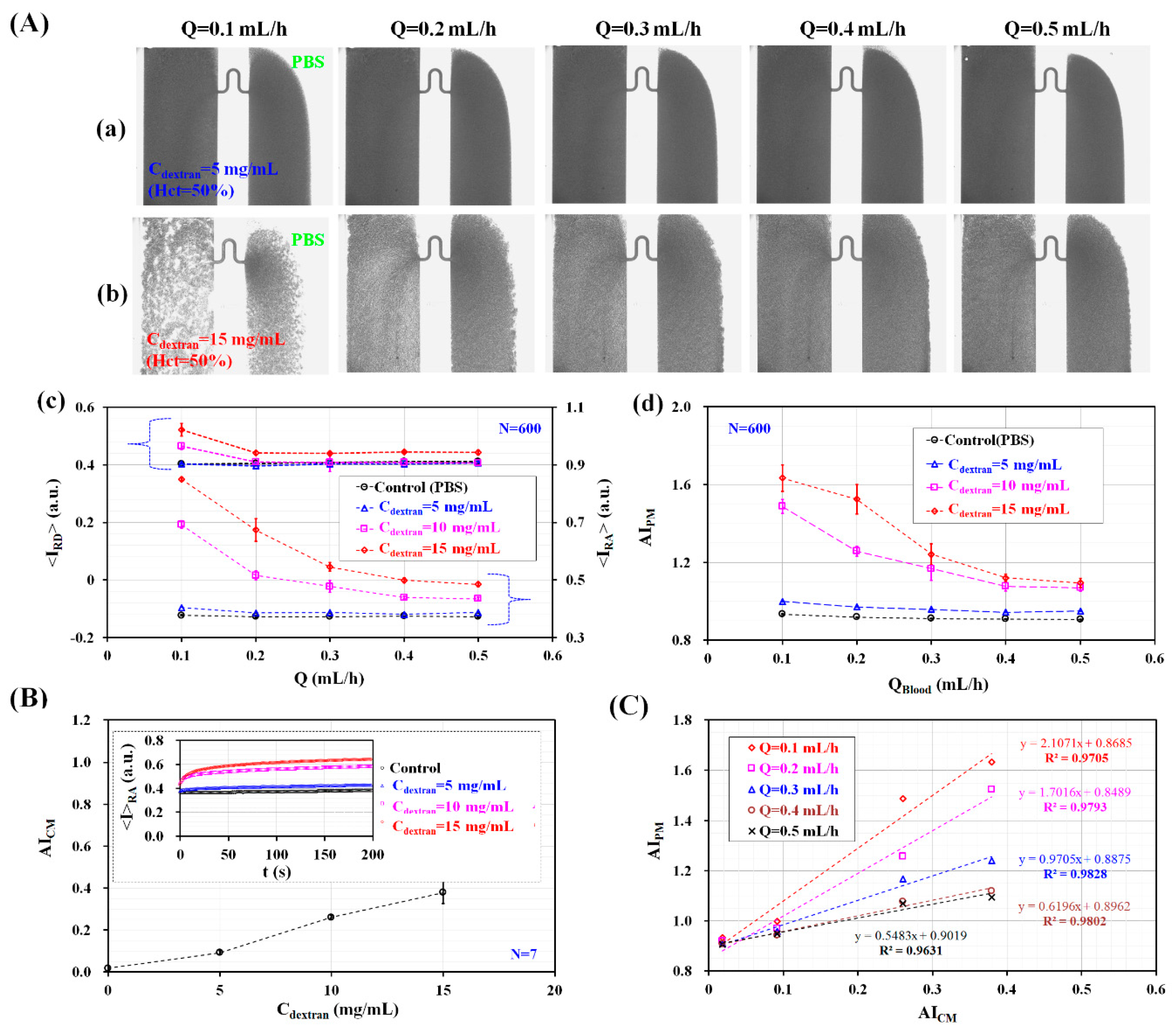
3.2. Quantitative Evaluation of RBC Aggregation Index and Blood Viscosity under Constant Blood Flows
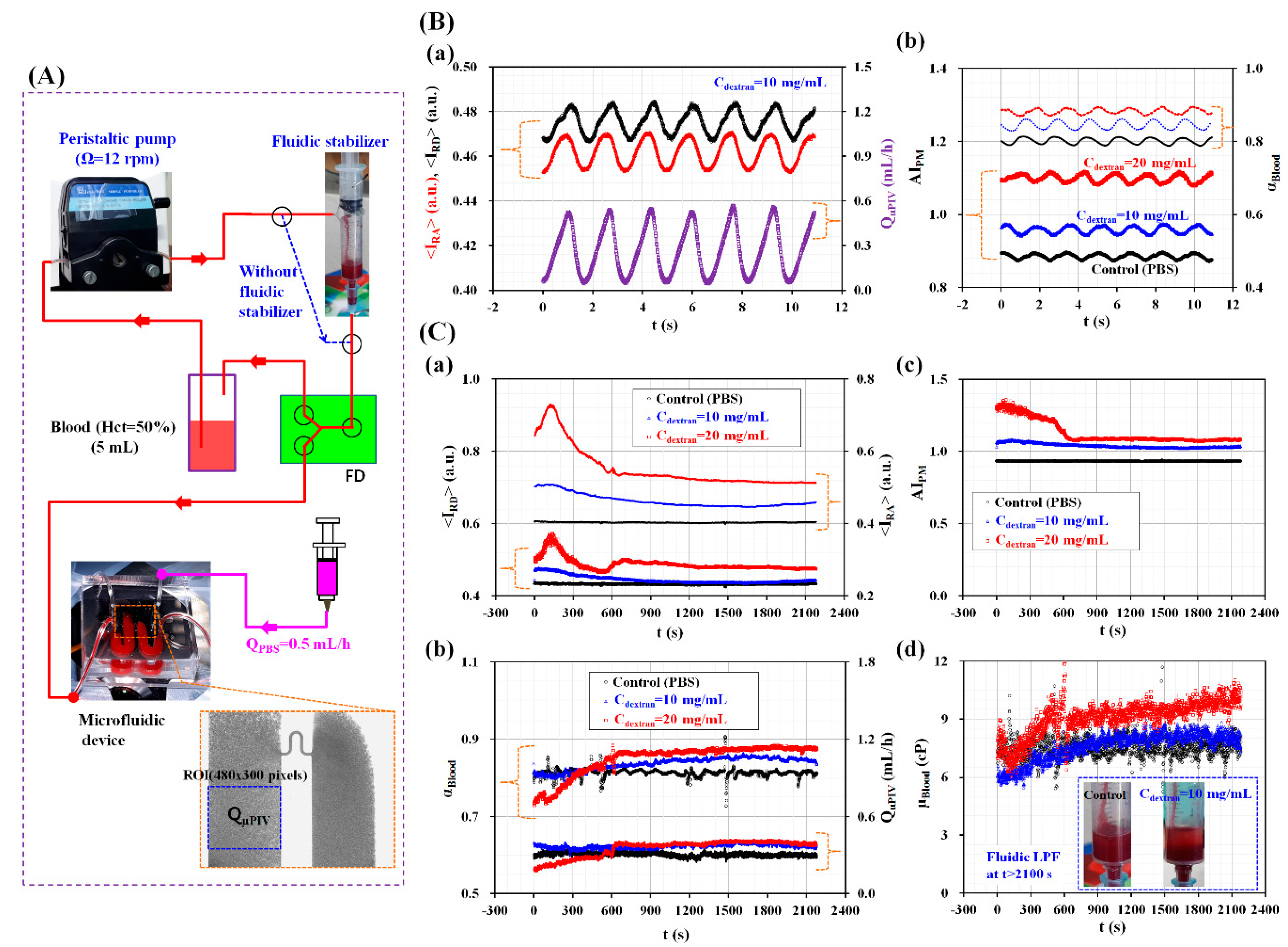
3.3. Quantitative Evaluation of RBC Aggregation Index and Blood Viscosity under Pulsatile Blood Flow
3.4. Quantitative Evaluation of RBC Aggregation and Blood Viscosity under an In-Vitro Fluidic Circuit
4. Conclusions
Supplementary Materials
Funding
Conflicts of Interest
References
- Benjamin, E.J.; Blaha, M.J.; Chiuve, S.E.; Cushman, M.; Das, S.R.; Deo, R.; Ferranti, S.D.D.; Floyd, J.; Fornage, M.; Gillespie, C.; et al. Heart disease and stroke statistics—2017 update a report from the American heart association. Circulation 2017, 135, e146–e603. [Google Scholar] [CrossRef] [PubMed]
- Upadhyay, R.K. Emerging risk biomarkers in cardiovascular diseases and disorders. J. Liqids 2015, 2015, 971453. [Google Scholar] [CrossRef] [PubMed]
- Wu, A.H.B. Markers for early detection of cardiac diseases. Scand. J. Clin. Lab. Investig. 2005, 240, 112–121. [Google Scholar] [CrossRef] [PubMed]
- Yayan, J. Erythrocyte sedimentation rate as a marker for coronary heart disease. Vasc. Health Risk Manag. 2012, 8, 219–223. [Google Scholar] [CrossRef] [PubMed]
- Danesh, J.; Collins, R.; Peto, R.; Lowe, G.D.O. Haematocrit, viscosity, erythrocyte sedimentation rate: Meta-analyses of prospective studies of coronary heart disease. Eur. Heart J. 2000, 21, 515–520. [Google Scholar] [CrossRef] [PubMed]
- Cho, Y.-I.; Cho, D.J. Hemorheology and Microvascular Disorders. Korean Circ. J. 2011, 41, 287–295. [Google Scholar] [CrossRef] [PubMed]
- Baskurt, O.K.; Meiselman, H.J. Blood rhelogy and hemodynamics. Semin. Thromb. Hemost. 2003, 29, 435–450. [Google Scholar] [PubMed]
- Kang, Y.J.; Lee, S.-J. In vitro and ex vivo measurement of the biophysical properties of blood using microfluidic platforms and animal models. Analyst 2018, 143, 2723–2749. [Google Scholar] [CrossRef] [PubMed]
- Kang, Y.J. Periodic and simultaneous quantification of blood viscosity and red blood cell aggregation using a microfluidic platform under in-vitro closed-loop circulation. Biomicrofluidics 2018, 12, 024116. [Google Scholar] [CrossRef] [PubMed]
- Kang, Y.J.; Yeom, E.; Lee, S.-J. Microfluidic biosensor for monitoring tmporal variations of hemorheological and hemodynamic properties using an extracorporeal rat bypass loop. Anal. Chem. 2013, 85, 10503–10511. [Google Scholar] [CrossRef] [PubMed]
- Yeom, E.; Kang, Y.J.; Lee, S.-J. Hybrid system for ex-vivo hemorheological and hemodynamic analysis: A feasibility study. Sci. Rep. 2015, 5, 11064. [Google Scholar] [CrossRef] [PubMed]
- Whittaker, S.R.F.; Winton, F.R. The apparent viscosity of blood flowing in the isolated hindlimb of the dog and its variation with corpuscular concentration. J. Physiol. 1933, 78, 339–369. [Google Scholar] [CrossRef] [PubMed]
- Ahn, C.B.; Kang, Y.J.; Kim, M.G.; Yang, S.; Lim, C.H.; Son, H.S.; Kim, J.S.; Lee, S.Y.; Son, K.H.; Sun, K. The effect of pulsatile versus nonpulsatile blood flow on viscoelasticity and red blood cell aggregation in extracorporeal circulation. Korean J. Thorac. Cardiovasc. Surg. 2016, 49, 145–150. [Google Scholar] [CrossRef] [PubMed]
- Kang, Y.J.; Kim, M.G.; Son, K.H.; Lim, C.H.; Son, H.S.; Yoon, S.Y.; Kwon, H.S.; Yang, S. Experimental investigation of pulsatility effect on the deformability and hemolysis of blood cells. Artif. Organs 2010, 34, E103–E109. [Google Scholar] [CrossRef] [PubMed]
- Isiksacan, Z.; Erel, O.; Elbuken, C. A portable microfluidic system for rapid measurement of the erythrocyte sedimentation rate. Lab Chip 2016, 16, 4682–4690. [Google Scholar] [CrossRef] [PubMed]
- Kang, Y.J. Microfluidic-based measurement of RBC aggregation and the ESR using a driving syringe system. Anal. Methods 2018, 10, 1805–1816. [Google Scholar] [CrossRef]
- Kang, Y.J.; Kim, B.J. Multiple and periodic measurement of RBC aggregation and ESR in parallel microfluidic channels under on-off blood flow control. Micromachines 2018, 9, 318. [Google Scholar] [CrossRef]
- Kang, Y.J. Microfluidic-based measurement method of red blood cell aggregation under hematocrit variations. Sensors 2017, 17, 2037. [Google Scholar] [CrossRef] [PubMed]
- Nam, J.-H.; Yang, Y.; Chung, S.; Shin, S. Comparison of light-transmission and -backscattering methods in the measurement of red blood cell aggregation. J. Biomed. Opt. 2010, 15, 027003. [Google Scholar] [CrossRef] [PubMed]
- Lim, H.-J.; Lee, Y.-J.; Nam, J.-H.; Chung, S.; Shin, S. Temperature-dependent threshold shear stress of red blood cell aggregation. J. Biomech. 2010, 43, 546–550. [Google Scholar] [CrossRef] [PubMed]
- Zhbanov, A.; Yang, S. Effects of aggregation on blood sedimentation and conductivity. PLoS ONE 2015, 10, e0129337. [Google Scholar] [CrossRef] [PubMed]
- Baskurt, O.K.; Uyuklu, M.; Meiselman, H.J. Time Course of Electrical Impedance During Red Blood Cell Aggregation in a Glass Tube: Comparison with Light Transmittance. IEEE Trans. Biomed. Eng. 2010, 57, 969–978. [Google Scholar] [CrossRef] [PubMed]
- Yeom, E.; Lee, S.J. Microfluidic-based speckle analysis for sensitive measurement of erythrocyte aggregation: A comparison of four methods for detection of elevated erythrocyte aggregation in diabetic rat blood. Biomicrofluidics 2015, 9, 024110. [Google Scholar] [CrossRef] [PubMed]
- Kang, Y.J. Continuous and simultaneous measurement of the biophysical properties of blood in a microfluidic environment. Analyst 2016, 141, 6583–6597. [Google Scholar] [CrossRef] [PubMed]
- Brust, M.; Aouane, O.; Thie´baud, M.; Flormann, D.; Verdier, C.; Kaestner, L.; Laschke, M.W.; Selmi, H.; Benyoussef, A.; Podgorski, T.; et al. The plasma protein fibrinogen stabilizes clusters of red blood cells in microcapillary flows. Sci. Rep. 2014, 4, 4348. [Google Scholar] [CrossRef] [PubMed]
- Kaliviotis, E.; Sherwood, J.M.; Balabani, S. Partitioning of red blood cell aggregates in bifurcating microscale flows. Sci. Rep. 2017, 7, 44563. [Google Scholar] [CrossRef] [PubMed]
- Kang, Y.J.; Ha, Y.-R.; Lee, S.-J. Microfluidic-based measurement of erythrocyte sedimentation rate for biophysical assessment of blood in an in vivo malaria-infected mouse. Biomicrofluidics 2014, 8, 044114. [Google Scholar] [CrossRef] [PubMed]
- Solomon, D.E.; Abdel-Raziq, A.; Vanapalli, S.A. A stress-controlled microfluidic shear viscometer based on smartphone imaging. Rheol. Acta 2016, 55, 727–738. [Google Scholar] [CrossRef]
- Srivastava, N.; Davenport, R.D.; Burns, M.A. Nanoliter Viscometer for Analyzing Blood Plasma and Other Liquid Samples. Anal. Chem. 2005, 77, 383–392. [Google Scholar] [CrossRef] [PubMed]
- Srivastava, N.; Burns, M.A. Analysis of non-Newtonian liquids using a microfluidic capillary viscometer. Anal. Chem. 2006, 78, 1690–1696. [Google Scholar] [CrossRef] [PubMed]
- Han, Z.; Tang, X.; Zheng, B. A PDMS viscometer for microliter Newtonian fluid. J. Micromech. Microeng. 2007, 17, 1828–1834. [Google Scholar] [CrossRef]
- Han, Z.; Zheng, B. A poly(dimethylsiloxane) viscometer for microliter power law fluid. J. Micromech. Microeng. 2009, 19, 115005. [Google Scholar] [CrossRef]
- Oh, S.; Kim, B.; Lee, J.K.; Choi, S. 3D-printed capillary circuits for rapid, low-cost, portable analysis ofblood viscosity. Sens. Actuator B Chem. 2018, 259, 106–113. [Google Scholar] [CrossRef]
- Choi, S.; Park, J.-K. Microfluidic rheometer for characterization of protein unfolding and aggregation in microflow. Small 2010, 6, 1306–1310. [Google Scholar] [CrossRef] [PubMed]
- Kim, B.J.; Lee, S.Y.; Jee, S.; Atajanov, A.; Yang, S. Micro-viscometer for measuring shear-varying blood viscosity over a wide-ranging shear rate. Sensors 2017, 17, 1442. [Google Scholar] [CrossRef] [PubMed]
- Kang, Y.J.; Yang, S. Integrated microfluidic viscometer equipped with fluid temperature controller for measurement of viscosity in complex fluid. Microfluid. Nanofluid. 2013, 14, 657–668. [Google Scholar] [CrossRef]
- Kang, Y.J.; Ryu, J.; Lee, S.-J. Label-free viscosity measurement of complex fluids using reversal flow switching manipulation in a microfluidic channel. Biomicrofluidics 2013, 7, 044106. [Google Scholar] [CrossRef] [PubMed]
- Kang, Y.J.; Yeom, E.; Lee, S.-J. A microfluidic device for simultaneous measurement of viscosity and flow rate of blood in a complex fluidic network. Biomicrofluidics 2013, 7, 054111. [Google Scholar] [CrossRef] [PubMed]
- Kang, Y.J.; Lee, S.-J. Blood viscoelasticity measurement using steady and transient flow controls of blood in a microfluidic analogue of Wheastone-bridge channel. Biomicrofluidics 2013, 7, 054122. [Google Scholar] [CrossRef] [PubMed]
- Pop, G.A.M.; Sisschops, L.L.A.; Iliev, B.; Struijk, P.C.; van der Heven, J.G.; Hoedemaekers, C.W.E. On-line blood viscosity monitoring in vivo with a central venous catheter using electrical impedance technique. Biosens. Bioelectron. 2013, 41, 595–601. [Google Scholar] [CrossRef] [PubMed]
- Zeng, H.; Zhao, Y. Rheological analysis of non-Newtonian blood flow using a microfluidic device. Sens. Actuator A Phys. 2011, 166, 207–213. [Google Scholar] [CrossRef]
- Otsu, N. A tlreshold selection method from gray-level histograms. IEEE Trans. Syst. Man. Cybern. 1979, 9, 62–66. [Google Scholar] [CrossRef]
- Kang, Y.J.; Ha, Y.-R.; Lee, S.-J. High-Throughput and Label-Free Blood-on-a-Chip for Malaria Diagnosis. Anal. Chem. 2016, 88, 2912–2922. [Google Scholar] [CrossRef] [PubMed]
- Kang, Y.J.; Yang, S. Fluidic low pass filter for hydrodynamic flow stabilization in microfluidic environments. Lab Chip 2012, 12, 1881–1889. [Google Scholar] [CrossRef] [PubMed]
- Kang, Y.J.; Yeom, E.; Seo, E.; Lee, S.-J. Bubble-free and pulse-free fluid delivery into microfluidic devices. Biomicrofluidics 2014, 8, 014102. [Google Scholar] [CrossRef] [PubMed]


| RBC Aggregation | Blood Viscosity | Simultaneous | Comments | Ref. | ||
|---|---|---|---|---|---|---|
| Methods | Continuous | Methods | Continuous | |||
| Photometric intensity | X | X | -Vacuum pump -Light backscattering or transmission | [19,20] | ||
| X | X | -Pinch valve -Light transmission | [15] | |||
| Electric impedance | X | X | -Pipette [21] or syringe pump [22] -Conductivity | [21,22] | ||
| Ultrasonic imaging | X | X | -Syringe pump -Speckle size | [23] | ||
| Microscopic imaging | X | X | -Pressure system -Aggregate size | [26] | ||
| X | X | -Syringe pump -Cluster size or occurrence | [25] | |||
| X | X | -Inverted syringe pump -intensity variations | [27] | |||
| Periodic (T = 4–5 min) | X | -Air-suction pump [18] or syringe pump [16,17] (on-off blood flows) -Intensity variations | [16,17,18] | |||
| Periodic (T = 4 min) | Modified parallel flows | O | O | -Two syringe pumps (on-off blood flows) -Intensity variations -Interfacial locations | [24] | |
| Periodic (T = 400 s) | Modified parallel flows | O | O | -Two syringe pumps (on-off blood flows) -Intensity variations -Interfacial locations | [9] | |
| Co-flowing streams | Periodic | X | -Two syringe pumps -Flow rate for relocating interface to the center | [34] | ||
| Microflow compartment | O | X | -Two syringe pumps -Numbers of channels filled with each fluid at the same flow rate | [35,36] | ||
| Reversal flow switching | Periodic | X | -Two syringe pumps -Flow rate for inducing reversal flow in the bridge channel | [10,11,37,38,39] | ||
| Advancing meniscus | X | X | -Surface tension-driven blood flow -Mean velocity | [29,30,31,32] | ||
| X | X | -Blood delivery (pressure [28] or pipette [33]) -Smartphone-based imaging | [28,33] | |||
| Electric impedance | O | X | -Syringe pump -Electric resistance | [40,41] | ||
© 2018 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kang, Y.J. Microfluidic-Based Technique for Measuring RBC Aggregation and Blood Viscosity in a Continuous and Simultaneous Fashion. Micromachines 2018, 9, 467. https://doi.org/10.3390/mi9090467
Kang YJ. Microfluidic-Based Technique for Measuring RBC Aggregation and Blood Viscosity in a Continuous and Simultaneous Fashion. Micromachines. 2018; 9(9):467. https://doi.org/10.3390/mi9090467
Chicago/Turabian StyleKang, Yang Jun. 2018. "Microfluidic-Based Technique for Measuring RBC Aggregation and Blood Viscosity in a Continuous and Simultaneous Fashion" Micromachines 9, no. 9: 467. https://doi.org/10.3390/mi9090467
APA StyleKang, Y. J. (2018). Microfluidic-Based Technique for Measuring RBC Aggregation and Blood Viscosity in a Continuous and Simultaneous Fashion. Micromachines, 9(9), 467. https://doi.org/10.3390/mi9090467





