Surface Modification of Electroosmotic Silicon Microchannel Using Thermal Dry Oxidation
Abstract
:1. Introduction
2. Microchannel Electroosmotic System Design
3. Fabrication Process of Electroosmotic Device (EO) Channel System
3.1. Fabrication of Silicon Microchannel Using Plasma Etching Procedure
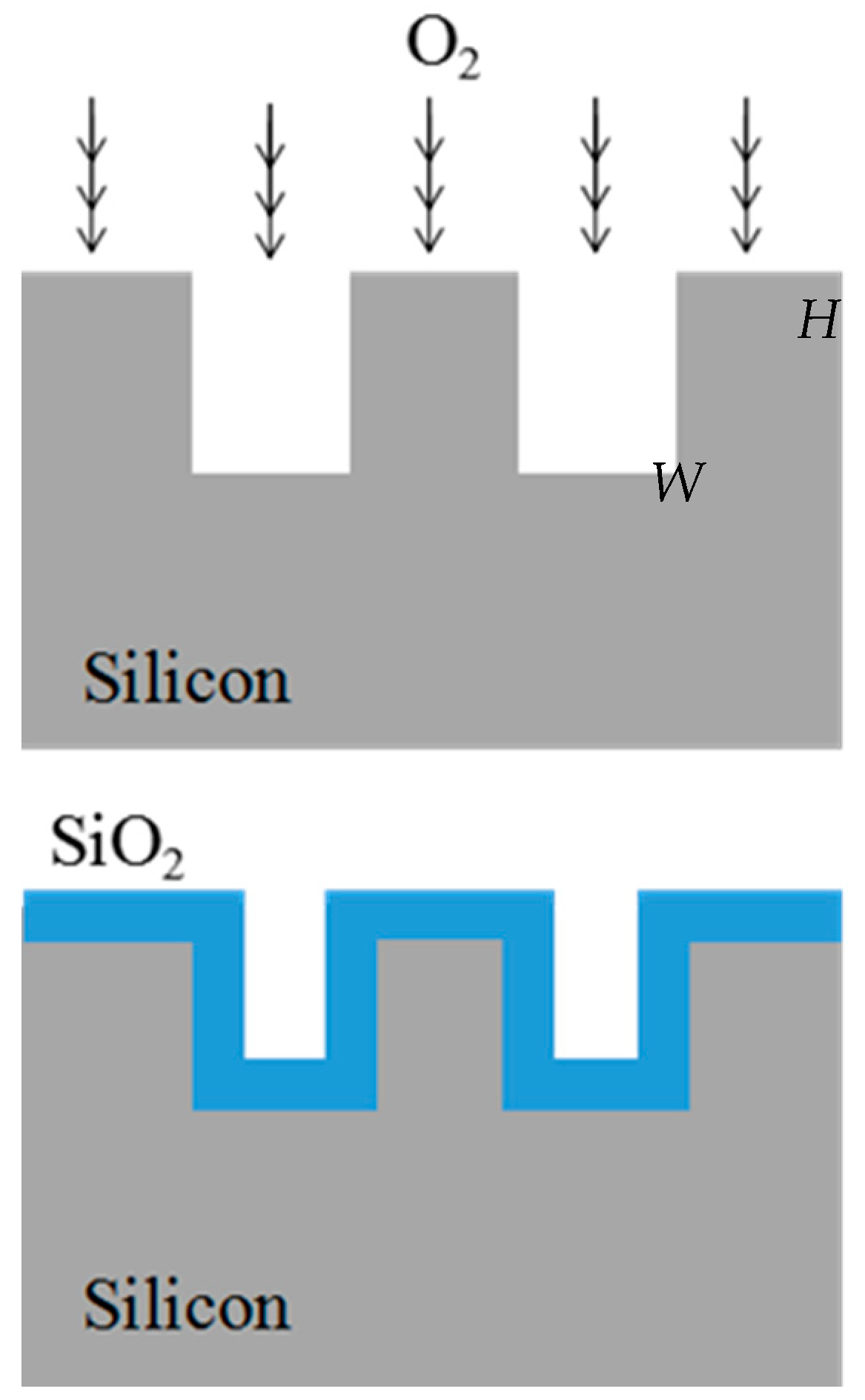
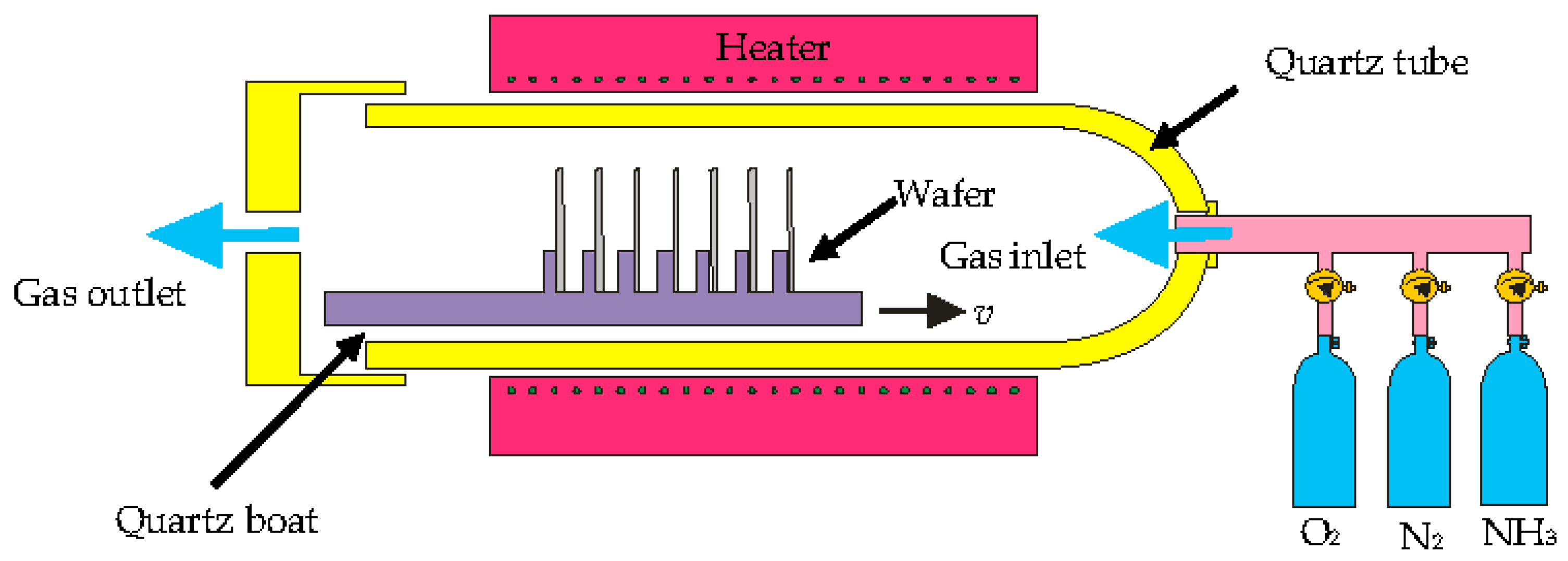
3.2. Surface Modification of Fabricated Silicon Microchannel Using Thermal Oxidation
4. Results and Discussion
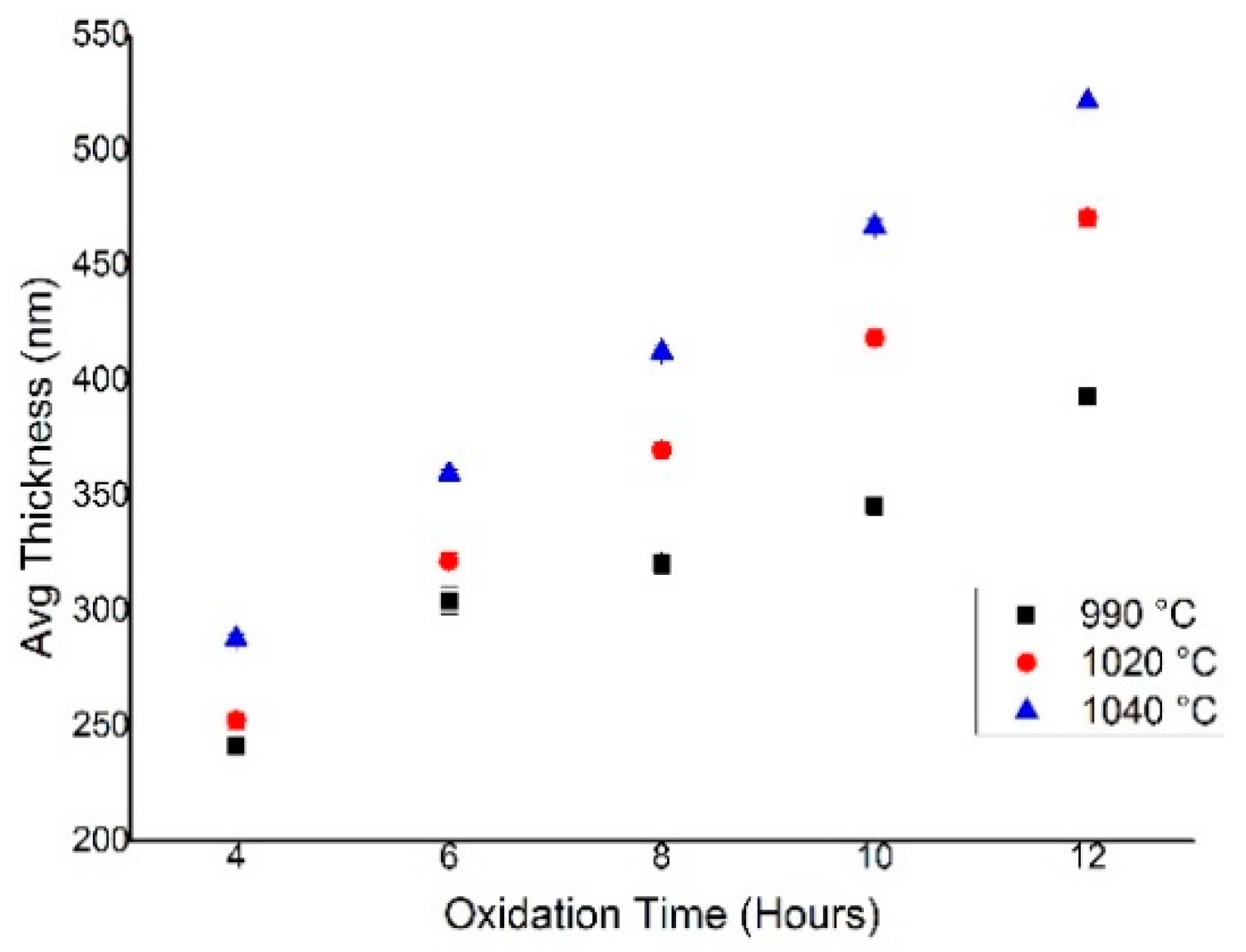
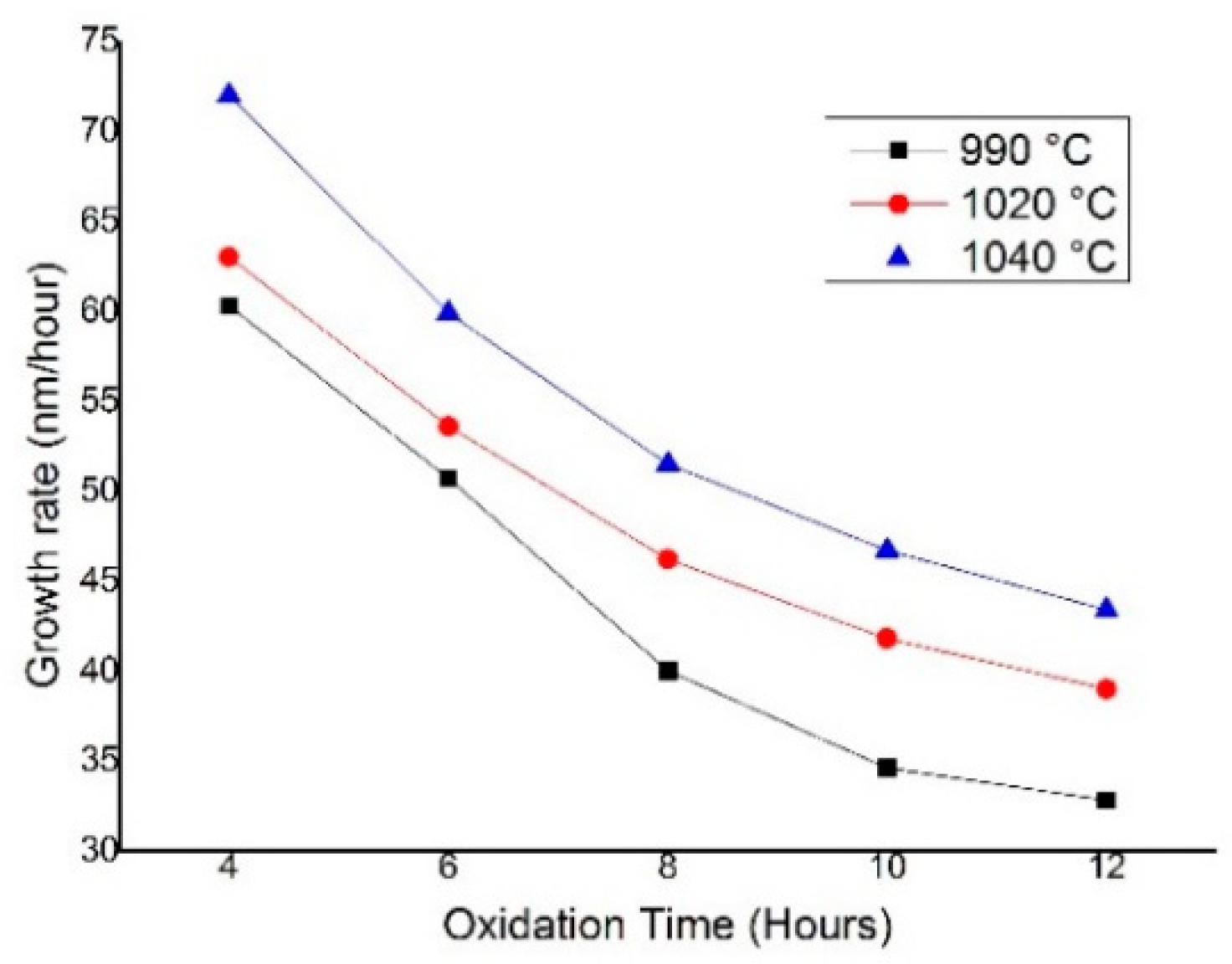
4.1. SiO2 Layer Thickness and Oxidation Growth Rate
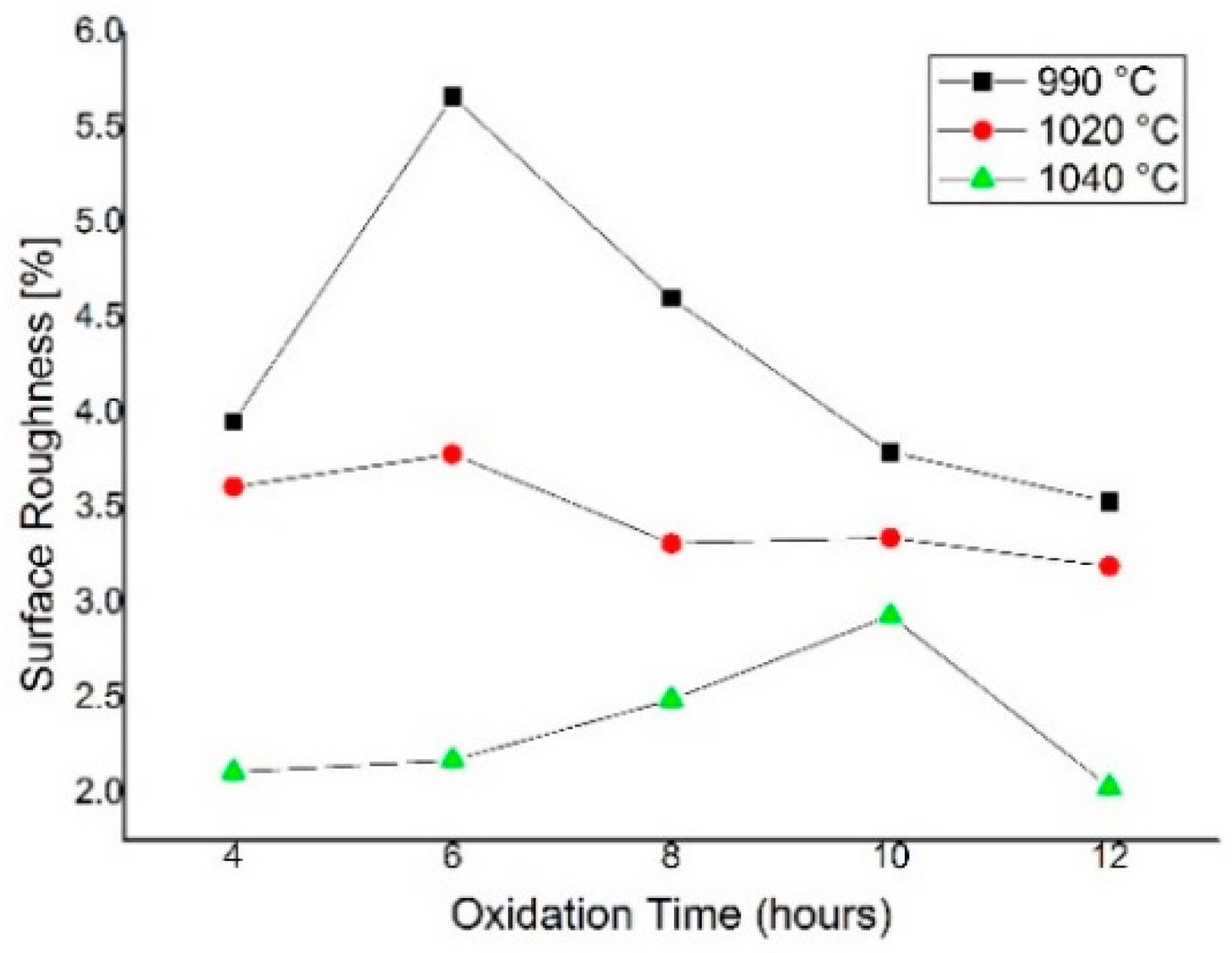
4.2. Surface Uniformity
4.3. Microchannel Structure Improvement after Thermal Oxidation
5. Conclusions
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Pennathur, S.; Santiago, J.G. Electrokinetic transport in nanochannels. 2. Experiments. Anal. Chem. 2005, 77, 6782–6789. [Google Scholar] [CrossRef] [PubMed]
- Buyong, M.R.; Larki, F.; Faiz, M.S.; Hamzah, A.A.; Yunas, J.; Majlis, B.Y. A tapered aluminium microelectrode array for improvement of dielectrophoresis-based particle manipulation. Sensors 2015, 15, 10973–10990. [Google Scholar] [CrossRef] [PubMed]
- Zeng, S.; Chen, C.H.; Mikkelsen, J.C.; Santiago, J.G. Fabrication and characterization of electroosmotic micropumps. Sens. Actuators B Chem. 2001, 79, 107–114. [Google Scholar] [CrossRef]
- Chen, C.H.; Santiago, J.G. A planar electroosmotic micropump. J. Microelectromech. Syst. 2002, 11, 672–683. [Google Scholar] [CrossRef]
- Garcia, A.L.; Ista, L.K.; Petsev, D.N.; O’Brien, M.J.; Bisong, P.; Mammoli, A.A.; Brueck, S.R.J.; López, G.P. Electrokinetic molecular separation in nanoscale fluidic channels. Lab Chip 2005, 5, 1271–1276. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez-Ruiz, I.; Babenko, V.; Martínez-Rodríguez, S.; Gavira, J.A. Protein separation under a microfluidic regime. Analyst 2018, 143, 606–619. [Google Scholar] [CrossRef] [PubMed]
- Masrie, M.; Majlis, B.Y.; Yunas, J. Fabrication of multilayer-PDMS based microfluidic device for bio-particles concentration detection. Bio-Med. Mater. Eng. 2014, 24, 1951–1958. [Google Scholar]
- Vourdas, N.; Tserepi, A.; Boudouvis, A.G.; Gogolides, E. Plasma processing for polymeric microfluidics fabrication and surface modification: Effect of super-hydrophobic walls on electroosmotic flow. Microelectron. Eng. 2008, 85, 1124–1127. [Google Scholar] [CrossRef]
- Thorslund, S.; Nikolajeff, S. Instant oxidation of closed microchannels. J. Micromech. Microeng. 2007, 17, 4. [Google Scholar] [CrossRef]
- Kirby, B.J.; Hasselbrink, E.F. Zeta potential of microfluidic substrates: 2. Data for polymers. Electrophoresis 2004, 25, 203–213. [Google Scholar] [CrossRef] [PubMed]
- Madou, M.J. Fundamentals of Microfabrication, 2nd ed.; CRC Press, Taylor & Francis Group: Boca Raton, FL, USA, 2002. [Google Scholar]
- Abdolvand, R.; Ayazi, F. An advanced reactive ion etching process for very high aspect-ratio sub-micron wide trenches in silicon. Sens. Actuators A Phys. 2008, 144, 109–116. [Google Scholar] [CrossRef]
- Miller, K.; Li, M.; Walsh, K.M.; Fu, X.A. The effects of DRIE operational parameters on vertically aligned micropillar arrays. J. Micromech. Microeng. 2013, 23, 035039. [Google Scholar] [CrossRef]
- Chang, C.; Wang, Y.F.; Kanamori, Y.; Shih, J.J.; Kawai, Y.; Lee, C.K.; Wu, K.C.; Esashi, M. Etching submicrometer trenches by using the Bosch process and its application to the fabrication of antireflection structures. J. Micromech. Microeng. 2005, 15, 580–585. [Google Scholar] [CrossRef]
- Barillaro, G.; Merlo, S.; Strambini, L.M. Bandgap tuning of silicon micromachined 1-D photonic crystals by thermal oxidation. IEEE J. Sel. Top. Quantum Electron. 2008, 14, 1074–1081. [Google Scholar] [CrossRef]
- Deal, B.E.; Grove, A.S. General relationship for the thermal oxidation of silicon. J. Appl. Phys. 1965, 36, 3770–3778. [Google Scholar] [CrossRef]
- Razeghi, M. Technology of Quantum Devices; Springer: Heidelberg, Germany, 2010; pp. 42–52. [Google Scholar]
- Gao, F.; Ylinen, S.; Kainlauri, M.; Kapulainen, M. Smooth silicon sidewall etching for waveguide structures using a modified Bosch process. J. Micro/Nanolithogr. MEMS MOEMS 2014, 13, 13010. [Google Scholar] [CrossRef]
- Qiao, R. Effects of molecular level surface roughness on electroosmotic flow. Microfluid. Nanofluid. 2007, 3, 33–38. [Google Scholar] [CrossRef]
- Masilamani, K.; Ganguly, S.; Feichtinger, C.; Bartuschat, D.; Rüde, U. Effects of surface roughness and electrokinetic heterogeneity on electroosmotic flow in microchannel. Fluid Dyn. Res. 2015, 47, 35505. [Google Scholar] [CrossRef]




© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tuan Yaakub, T.N.; Yunas, J.; Latif, R.; Hamzah, A.A.; Razip Wee, M.F.M.; Yeop Majlis, B. Surface Modification of Electroosmotic Silicon Microchannel Using Thermal Dry Oxidation. Micromachines 2018, 9, 222. https://doi.org/10.3390/mi9050222
Tuan Yaakub TN, Yunas J, Latif R, Hamzah AA, Razip Wee MFM, Yeop Majlis B. Surface Modification of Electroosmotic Silicon Microchannel Using Thermal Dry Oxidation. Micromachines. 2018; 9(5):222. https://doi.org/10.3390/mi9050222
Chicago/Turabian StyleTuan Yaakub, Tuan Norjihan, Jumril Yunas, Rhonira Latif, Azrul Azlan Hamzah, Mohd Farhanulhakim Mohd Razip Wee, and Burhanuddin Yeop Majlis. 2018. "Surface Modification of Electroosmotic Silicon Microchannel Using Thermal Dry Oxidation" Micromachines 9, no. 5: 222. https://doi.org/10.3390/mi9050222
APA StyleTuan Yaakub, T. N., Yunas, J., Latif, R., Hamzah, A. A., Razip Wee, M. F. M., & Yeop Majlis, B. (2018). Surface Modification of Electroosmotic Silicon Microchannel Using Thermal Dry Oxidation. Micromachines, 9(5), 222. https://doi.org/10.3390/mi9050222






