A Perspective on the Rise of Optofluidics and the Future
Abstract
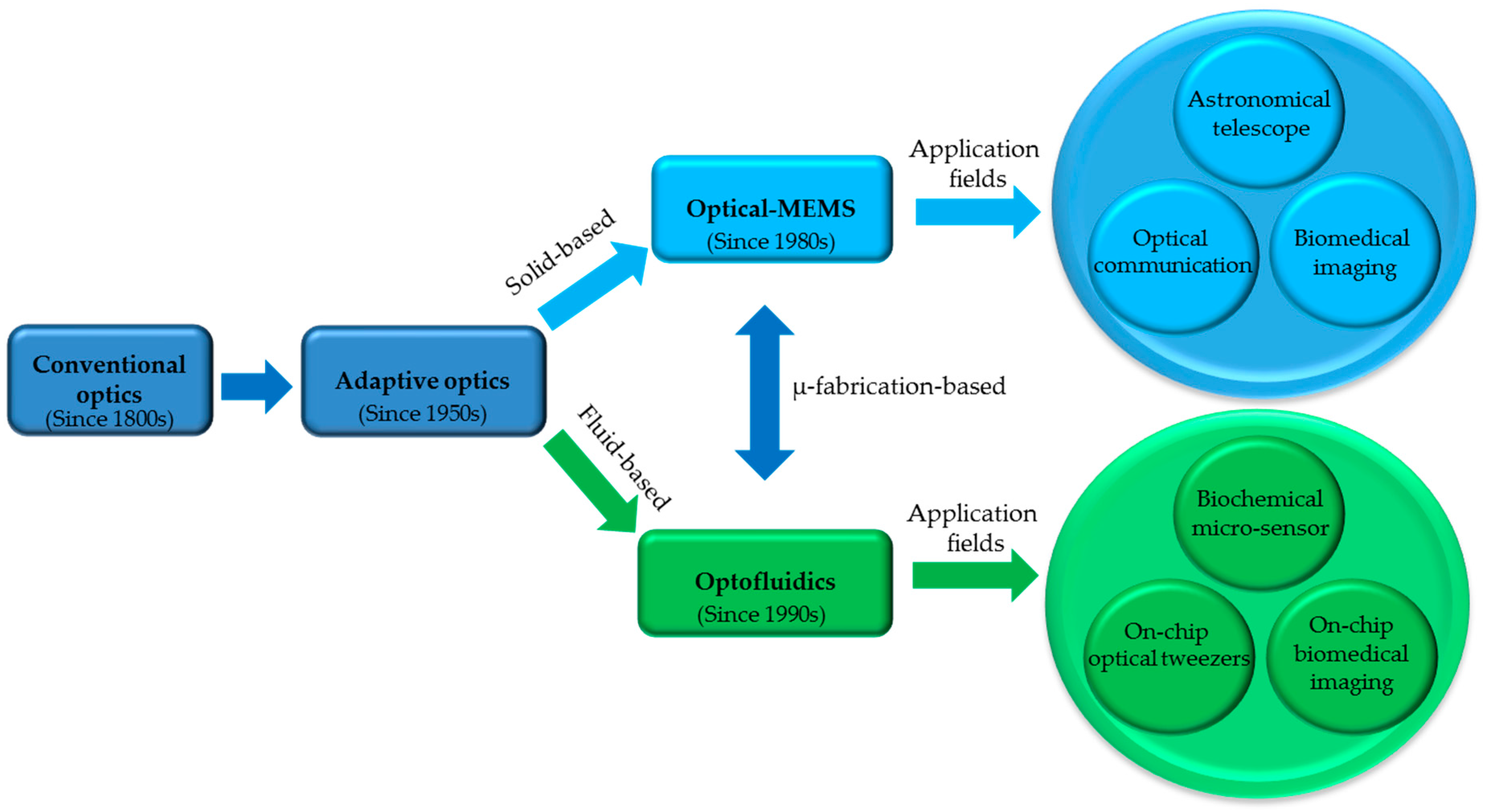
:1. The Origin of Optofluidics and Its Motivation
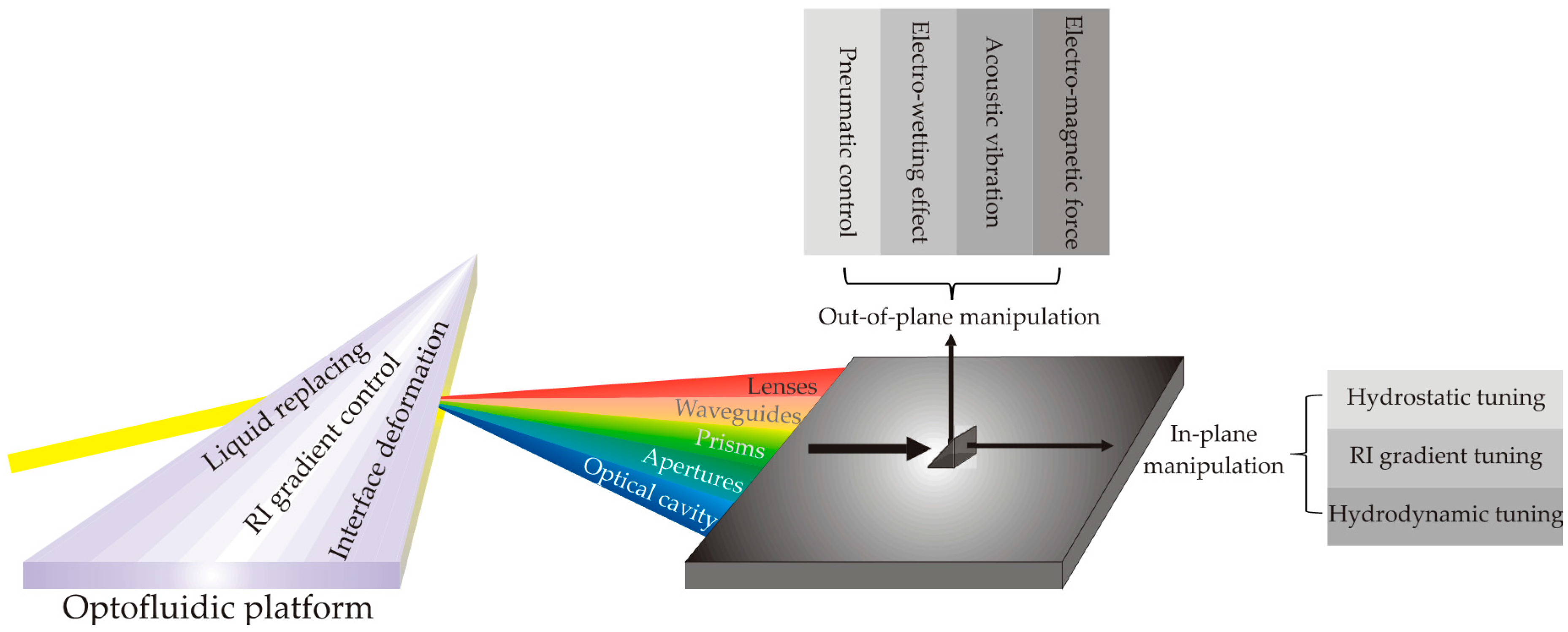
2. The Inventions of Optofluidic Components
2.1. Liquid Replacing
2.2. Liquid RI Gradient Control
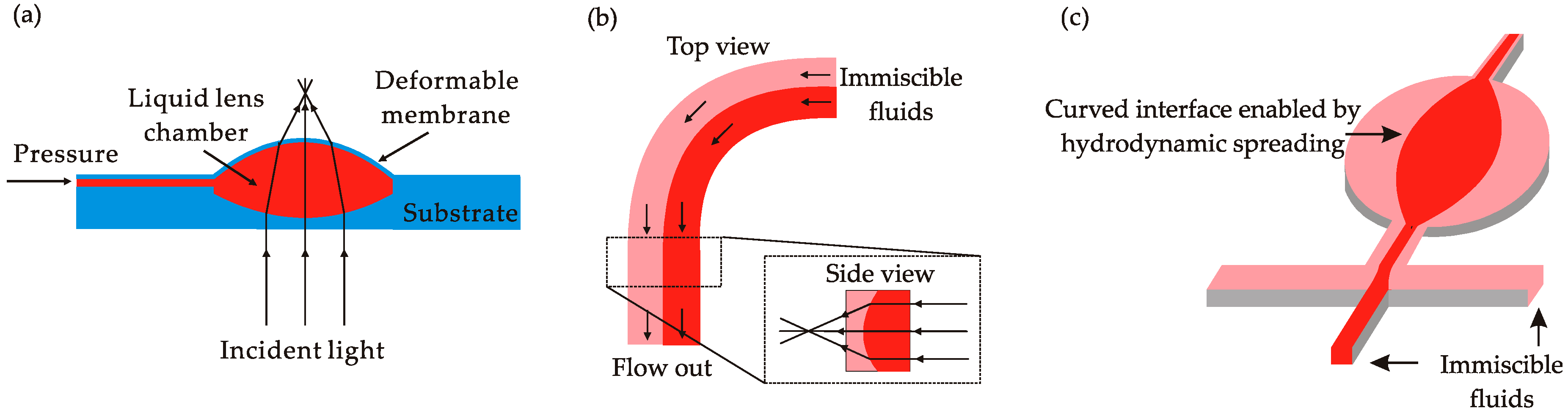
2.3. Fluid Interface Deformation
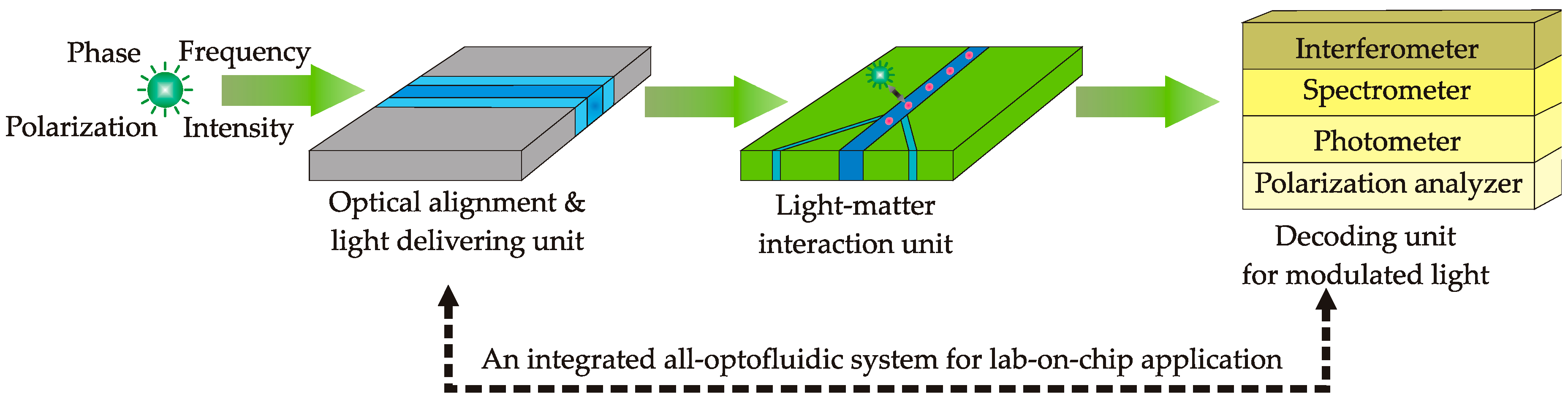
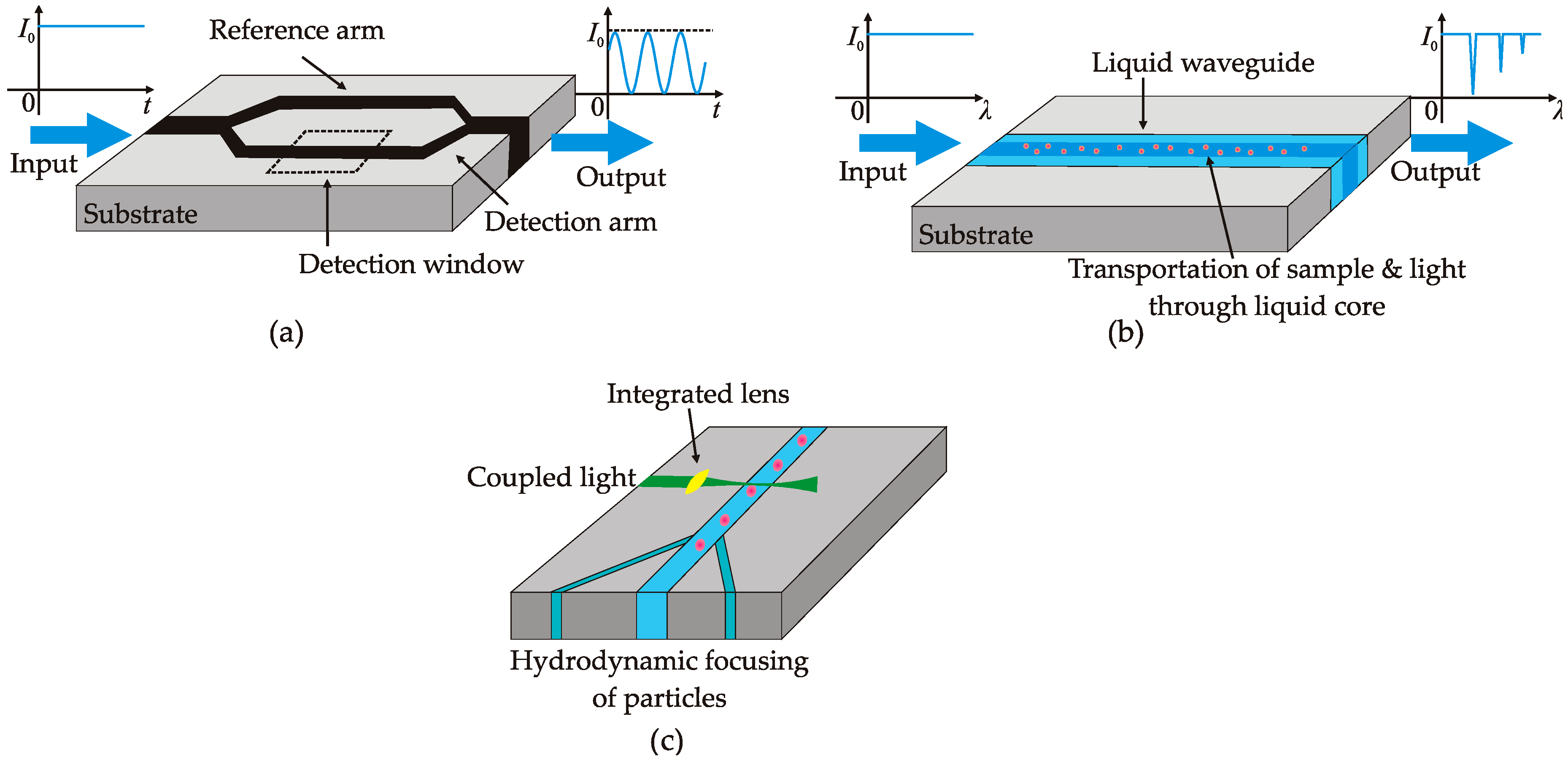
3. Instrumentation of Optofluidics
3.1. Optofluidic Interferometry
3.2. Optofluidic Spectroscopy
3.3. Optofluidic Cytometry
3.4. On-Chip Manipulation of Micro-Object
4. Commercialization of Optofluidics
5. Discussions and Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Schmidt, H.; Hawkins, A.R. The photonic integration of non-solid media using optofluidics. Nat. Photonics 2011, 5, 598–604. [Google Scholar] [CrossRef]
- Wood, R.W. The mercury paraboloid as a reflecting telescope. Astrophys. J. 1909, 29, 164–176. [Google Scholar] [CrossRef]
- Monat, C.; Domachuk, P.; Eggleton, B.J. Integrated optofluidics: A new river of light. Nat. Photonics 2007, 1, 106–114. [Google Scholar] [CrossRef]
- Petersen, K.E. Silicon torsional scanning mirror. IBM J. Res. Dep. 1980, 24, 631–637. [Google Scholar] [CrossRef]
- Psaltis, D.; Quake, S.R.; Yang, C. Developing optofluidic technology through the fusion of microfluidics and optics. Nature 2006, 442, 381–386. [Google Scholar] [CrossRef] [PubMed]
- Kim, E.; Baaske, M.D.; Vollmer, F. Towards next-generation label-free biosensors: Recent advances in whispering gallery mode sensors. Lab Chip 2017, 17, 1190–1205. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Li, H.; Zhao, L.; Wu, B.; Liu, S.; Liu, Y.; Yang, J. A review of droplet resonators: Operation method and application. Opt. Laser Technol. 2016, 86, 61–68. [Google Scholar] [CrossRef]
- Fan, X.; White, I.M.; Shopova, S.I.; Zhu, H.; Suter, J.D.; Sun, Y. Sensitive optical biosensors for unlabeled targets: A review. Anal. Chim. Acta 2008, 620, 8–26. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Watts, B.R.; Guo, T.; Zhang, Z.; Xu, C.; Fang, Q. Optofluidic device based microflow cytometers for particle/cell detection: A review. Micromachines 2016, 7, 70. [Google Scholar] [CrossRef]
- Erickson, D.; Sinton, D.; Psaltis, D. Optofluidics for energy applications. Nat. Photonics 2011, 5, 583–590. [Google Scholar] [CrossRef]
- Monat, C.; Domachuk, P.; Grillet, C.; Collins, M.; Eggleton, B.J.; Cronin-Golomb, M.; Mutzenich, S.; Mahmud, T.; Rosengarten, G.; Mitchell, A. Optofluidics: A novel generation of reconfigurable and adaptive compact architectures. Microfluid. Nanofluid. 2008, 4, 81–95. [Google Scholar] [CrossRef]
- Huang, N.-T.; Zhang, H.; Chung, M.-T.; Seo, J.H.; Kurabayashi, K. Recent advancements in optofluidics-based single-cell analysis: Optical on-chip cellular manipulation, treatment, and property detection. Lab Chip 2014, 14, 1230–1245. [Google Scholar] [CrossRef] [PubMed]
- Yang, T.; Bragheri, F.; Minzioni, P. A comprehensive review of optical stretcher for cell mechanical characterization at single-cell level. Micromachines 2016, 7, 90. [Google Scholar] [CrossRef]
- Brennan, D.; Justice, J.; Corbett, B.; McCarthy, T.; Galvin, P. Emerging optofluidic technologies for point-of-care genetic analysis systems: A review. Anal. Bioanal. Chem. 2009, 395, 621–636. [Google Scholar] [CrossRef] [PubMed]
- Mak, J.S.W.; Rutledge, S.A.; Abu-Ghazalah, R.M.; Eftekhari, F.; Irizar, J.; Tam, N.C.M.; Zheng, G.; Helmy, A.S. Recent developments in optofluidic-assisted raman spectroscopy. Prog. Quantum Electron. 2013, 37, 1–50. [Google Scholar] [CrossRef]
- Schmidt, H.; Hawkins, A.R. Optofluidic waveguides: I. Concepts and implementations. Microfluid. Nanofluid. 2008, 4, 3–16. [Google Scholar] [CrossRef] [PubMed]
- Testa, G.; Persichetti, G.; Bernini, R. Liquid core arrow waveguides: A promising photonic structure for integrated optofluidic microsensors. Micromachines 2016, 7, 47. [Google Scholar] [CrossRef]
- Mishra, K.; van den Ende, D.; Mugele, F. Recent developments in optofluidic lens technology. Micromachines 2016, 7, 102. [Google Scholar] [CrossRef]
- Nguyen, N.-T. Micro-optofluidic lenses: A review. Biomicrofluidics 2010, 4, 031501. [Google Scholar] [CrossRef] [PubMed]
- Pan, M.; Kim, M.; Kuiper, S.; Tang, S.K.Y. Actuating fluid-fluid interfaces for the reconfiguration of light. IEEE J. Sel. Top. Quantum Electron. 2015, 21, 444–455. [Google Scholar] [CrossRef]
- Domachuk, P.; Nguyen, H.C.; Eggleton, B.J. Transverse probed microfluidic switchable photonic crystal fiber devices. IEEE Photonics Technol. Lett. 2004, 16, 1900–1902. [Google Scholar] [CrossRef]
- Intonti, F.; Vignolini, S.; Türck, V.; Colocci, M.; Bettotti, P.; Pavesi, L.; Schweizer, S.L.; Wehrspohn, R.; Wiersma, D. Rewritable photonic circuits. Appl. Phys. Lett. 2006, 89, 211117. [Google Scholar] [CrossRef]
- Speijcken, N.W.L.; Dündar, M.A.; Bedoya, A.C.; Monat, C.; Grillet, C.; Domachuk, P.; Nötzel, R.; Eggleton, B.J.; van der Heijden, R.W. In situ optofluidic control of reconfigurable photonic crystal cavities. Appl. Phys. Lett. 2012, 100, 261107. [Google Scholar] [CrossRef]
- Erickson, D.; Rockwood, T.; Emery, T.; Scherer, A.; Psaltis, D. Nanofluidic tuning of photonic crystal circuits. Opt. Lett. 2006, 31, 59–61. [Google Scholar] [CrossRef] [PubMed]
- Arango, F.B.; Christiansen, M.B.; Gersborg-Hansen, M.; Kristensen, A. Optofluidic tuning of photonic crystal band edge lasers. Appl. Phys. Lett. 2007, 91, 223503. [Google Scholar] [CrossRef]
- Shi, J.; Juluri, B.K.; Lin, S.C.S.; Lu, M.; Gao, T.; Huang, T.J. Photonic crystal composites-based wide-band optical collimator. J. Appl. Phys. 2010, 108, 043514. [Google Scholar] [CrossRef]
- Mortensen, N.A.; Xiao, S. Slow-light enhancement of beer-lambert-bouguer absorption. Appl. Phys. Lett. 2007, 90, 141108. [Google Scholar] [CrossRef]
- Smith, C.L.C.; Wu, D.K.C.; Lee, M.W.; Monat, C.; Tomljenovic-Hanic, S.; Grillet, C.; Eggleton, B.J.; Freeman, D.; Ruan, Y.; Madden, S.; et al. Microfluidic photonic crystal double heterostructures. Appl. Phys. Lett. 2007, 91, 121103. [Google Scholar] [CrossRef]
- Domachuk, P.; Nguyen, H.C.; Eggleton, B.J.; Straub, M.; Gu, M. Microfluidic tunable photonic band-gap device. Appl. Phys. Lett. 2004, 84, 1838–1840. [Google Scholar] [CrossRef]
- Campbell, K.; Groisman, A.; Levy, U.; Pang, L.; Mookherjea, S.; Psaltis, D.; Fainman, Y. A microfluidic 2*2 optical switch. Appl. Phys. Lett. 2004, 85, 6119–6121. [Google Scholar] [CrossRef]
- Groisman, A.; Zamek, S.; Campbell, K.; Pang, L.; Levy, U.; Fainman, Y. Optofluidic 1*4 switch. Opt. Express 2008, 16, 13499–13508. [Google Scholar] [CrossRef] [PubMed]
- Li, X.C.; Wu, J.; Liu, A.Q.; Li, Z.G.; Soew, Y.C.; Huang, H.J.; Xu, K.; Lin, J.T. A liquid waveguide based evanescent wave sensor integrated onto a microfluidic chip. Appl. Phys. Lett. 2008, 93, 193901. [Google Scholar] [CrossRef]
- Schelle, B.; Dreb, P.; Franke, H.; Klein, K.F.; Slupek, J. Physical characterization of lightguide capillary cells. J. Phys. D Appl. Phys. 1999, 32, 3157–3163. [Google Scholar] [CrossRef]
- Datta, A.; Eom, I.Y.; Dhar, A.; Kuban, P.; Manor, R.; Ahmad, I.; Gangopadhyay, S.; Dallas, T.; Holtz, M.; Temkin, H.; et al. Microfabrication and characterization of teflon af-coated liquid core waveguide channels in silicon. IEEE Sens. J. 2003, 3, 788–795. [Google Scholar] [CrossRef]
- Llobera, A.; Wilke, R.; Buttgenbach, S. Poly(dimethylsiloxane) hollow abbe prism with microlenses for detection based on absorption and refractive index shift. Lab Chip 2004, 4, 24–27. [Google Scholar] [CrossRef] [PubMed]
- Llobera, A.; Wilke, R.; Buttgenbach, S. Optimization of poly(dimethylsiloxane) hollow prisms for optical sensing. Lab Chip 2005, 5, 506–511. [Google Scholar] [CrossRef] [PubMed]
- Erickson, D.; Heng, X.; Li, Z.; Rockwood, T.; Emery, T.; Zhang, Z.; Scherer, A.; Yang, C.; Psaltis, D. Optofluidics. Proc. SPIE 2005, 5908, 1–12. [Google Scholar]
- Olshanky, R.; Keck, D.B. Pulse broadening in graded-index optical fibers. Appl. Opt. 1976, 15, 483–491. [Google Scholar] [CrossRef] [PubMed]
- Koike, Y.; Ishigure, T.; Nihei, E. High-bandwidth graded-index polymer optical fiber. J. Lightwave Technol. 1995, 13, 1475–1489. [Google Scholar] [CrossRef]
- Ishigure, T.; Koike, Y.; Fleming, J.W. Optimum index profile of the perfluorinated polymer-based GI polymer optical fiber and its dispersion properties. J. Lightwave Technol. 2000, 18, 178–184. [Google Scholar] [CrossRef]
- Van Buren, M.; Riza, N.A. Foundations for low-loss fiber gradient-index lens pair coupling with the self-imaging mechanism. Appl. Opt. 2003, 42, 550–565. [Google Scholar] [CrossRef] [PubMed]
- Pierscionek, B.K.; Chan, D.Y.C. Refractive index gradient of human lenses. Optom. Vis. Sci. 1989, 66, 822–829. [Google Scholar] [CrossRef] [PubMed]
- Ren, H.; Fan, Y.H.; Gauza, S.; Wu, S.T. Tunable-focus flat liquid crystal spherical lens. Appl. Phys. Lett. 2004, 84, 4789–4791. [Google Scholar] [CrossRef]
- Ren, H.; Wu, S.T. Tunable electronic lens using a gradient polymer network liquid crystal. Appl. Phys. Lett. 2003, 82, 22–24. [Google Scholar] [CrossRef]
- Jones, C.E.; Atchison, D.A.; Meder, R.; Pope, J.M. Refractive index distribution and optical properties of the isolated human lens measured using magnetic resonance imaging (MRI). Vis. Res. 2005, 45, 2352–2366. [Google Scholar] [CrossRef] [PubMed]
- Mao, X.; Lin, S.C.S.; Lapsley, M.I.; Shi, J.; Juluri, B.K.; Huang, T.J. Tunable liquid gradient refractive index (L-GRIN) lens with two degrees of freedom. Lab Chip 2009, 9, 2050–2058. [Google Scholar] [CrossRef] [PubMed]
- Huang, H.; Mao, X.; Lin, S.C.S.; Kiraly, B.; Huang, Y.; Huang, T.J. Tunable two-dimensional liquid gradient refractive index (L-GRIN) lens for variable light focusing. Lab Chip 2010, 10, 2387–2393. [Google Scholar] [CrossRef] [PubMed]
- Le, Z.; Sun, Y.; Du, Y. Liquid gradient refractive index microlens for dynamically adjusting the beam focusing. Micromachines 2015, 6, 1984–1995. [Google Scholar] [CrossRef]
- Zhao, H.T.; Yang, Y.; Chin, L.K.; Chen, H.F.; Zhu, W.M.; Zhang, J.B.; Yap, P.H.; Liedberg, B.; Wang, K.; Wang, G.; et al. Optofluidic lens with low spherical and low field curvature aberrations. Lab Chip 2016, 16, 1617–1624. [Google Scholar] [CrossRef] [PubMed]
- Tang, S.K.Y.; Mayers, B.T.; Vezenov, D.V.; Whitesides, G.M. Optical waveguiding using thermal gradients across homogeneous liquids in microfluidic channels. Appl. Phys. Lett. 2006, 88, 061112. [Google Scholar] [CrossRef]
- Chen, Q.; Jian, A.; Li, Z.; Zhang, X. Optofluidic tunable lenses using laser-induced thermal gradient. Lab Chip 2016, 16, 104–111. [Google Scholar] [CrossRef] [PubMed]
- Higginson, K.A.; Costolo, M.A.; Rietman, E.A. Adaptive geometric optics derived from nonlinear acoustic effects. Appl. Phys. Lett. 2004, 84, 843–845. [Google Scholar] [CrossRef]
- Higginson, K.A.; Costolo, M.A.; Rietman, E.A.; Ritter, J.M.; Lipkens, B. Tunable optics derived from nonlinear acoustic effects. J. Appl. Phys. 2004, 95, 5896–5904. [Google Scholar] [CrossRef]
- McLeod, E.; Arnold, C.B. Mechanics and refractive power optimization of tunable acoustic gradient lenses. J. Appl. Phys. 2007, 102, 033104. [Google Scholar] [CrossRef]
- McLeod, E.; Hopkins, A.B.; Arnold, C.B. Multiscale bessel beams generated by a tunable acoustic gradient index of refraction lens. Opt. Lett. 2006, 31, 3155–3157. [Google Scholar] [CrossRef] [PubMed]
- Mermillod-Blondin, A.; McLeod, E.; Arnold, C.B. High-speed varifocal imaging with a tunable acoustic gradient index of refraction lens. Opt. Lett. 2008, 33, 2146–2148. [Google Scholar] [CrossRef] [PubMed]
- Olivier, N.; Mermillod-Blondin, A.; Arnold, C.B.; Beaurepaire, E. Two-photon microscopy with simultaneous standard and extended depth of field using a tunable acoustic gradient-index lens. Opt. Lett. 2009, 34, 1684–1686. [Google Scholar] [CrossRef] [PubMed]
- Duocastella, M.; Sun, B.; Arnold, C.B. Simultaneous imaging of multiple focal planes for three-dimensional microscopy using ultra-high-speed adaptive optics. J. Biomed. Opt. 2012, 17, 050505. [Google Scholar] [CrossRef] [PubMed]
- Ahn, S.H.; Kim, Y.K. Proposal of human eye's crystalline lens-like variable focusing lens. Sens. Actuators A Phys. 1999, 78, 48–53. [Google Scholar] [CrossRef]
- Werber, A.; Zappe, H. Tunable microfluidic microlenses. Appl. Opt. 2005, 44, 3238–3245. [Google Scholar] [CrossRef] [PubMed]
- Zhang, D.Y.; Justis, N.; Lo, Y.H. Fluidic adaptive lens of transformable lens type. Appl. Phys. Lett. 2004, 84, 4194–4196. [Google Scholar] [CrossRef]
- Song, C.; Xi, L.; Jiang, H. Liquid acoustic lens for photoacoustic tomography. Opt. Lett. 2013, 38, 2930–2933. [Google Scholar] [CrossRef] [PubMed]
- López, C.A.; Hirsa, A.H. Fast focusing using a pinned-contact oscillating liquid lens. Nat. Photonics 2008, 2, 610–613. [Google Scholar] [CrossRef]
- Lee, S.W.; Lee, S.S. Focal tunable liquid lens integrated with an electromagnetic actuator. Appl. Phys. Lett. 2007, 90, 121129. [Google Scholar] [CrossRef]
- Malouin, B.A., Jr.; Vogel, M.J.; Olles, J.D.; Cheng, L.; Hirsa, A.H. Electromagnetic liquid pistons for capillarity-based pumping. Lab Chip 2010, 11, 393–397. [Google Scholar] [CrossRef] [PubMed]
- Gorman, C.B.; Biebuyck, H.A.; Whitesides, G.M. Control of the shape of liquid lenses on a modified gold surface using an applied electrical potential across a self-assembled monolayer. Langmuir 1995, 11, 2242–2246. [Google Scholar] [CrossRef]
- López, C.A.; Lee, C.C.; Hirsa, A.H. Electrochemically activated adaptive liquid lens. Appl. Phys. Lett. 2005, 87, 1–3. [Google Scholar] [CrossRef]
- Mishra, K.; Murade, C.; Carreel, B.; Roghair, I.; Oh, J.M.; Manukyan, G.; van den Ende, D.; Mugele, F. Optofluidic lens with tunable focal length and asphericity. Sci. Rep. 2014, 4, 6378. [Google Scholar] [CrossRef] [PubMed]
- Lien, V.; Berdichevsky, Y.; Lo, Y.H. Microspherical surfaces with predefined focal lengths fabricated using microfluidic capillaries. Appl. Phys. Lett. 2003, 83, 5563–5565. [Google Scholar] [CrossRef]
- Shi, J.; Stratton, Z.; Lin, S.C.S.; Huang, H.; Huang, T.J. Tunable optofluidic microlens through active pressure control of an air-liquid interface. Microfluid. Nanofluid. 2010, 9, 313–318. [Google Scholar] [CrossRef]
- Dong, L.; Jiang, H. Tunable and movable liquid microlens in situ fabricated within microfluidic channels. Appl. Phys. Lett. 2007, 91, 041109. [Google Scholar] [CrossRef]
- Mao, X.; Stratton, Z.I.; Nawaz, A.A.; Lin, S.-C.S.; Huang, T.J. Optofluidic tunable microlens by manipulating the liquid meniscus using a flared microfluidic structure. Biomicrofluidics 2010, 4, 043007. [Google Scholar] [CrossRef] [PubMed]
- Wolfe, D.B.; Conroy, R.S.; Garstecki, P.; Mayers, B.T.; Fischbach, M.A.; Paul, K.E.; Prentiss, M.; Whitesides, G.M. Dynamic control of liquid-core/liquid-cladding optical waveguides. Proc. Natl. Acad. Sci. USA 2004, 101, 12434–12438. [Google Scholar] [CrossRef] [PubMed]
- Mao, X.; Waldeisen, J.R.; Juluri, B.K.; Huang, T.J. Hydrodynamically tunable optofluidic cylindrical microlens. Lab Chip 2007, 7, 1303–1308. [Google Scholar] [CrossRef] [PubMed]
- Song, C.; Nguyen, N.T.; Asundi, A.K.; Tan, S.H. Tunable micro-optofluidic prism based on liquid-core liquid-cladding configuration. Opt. Lett. 2010, 35, 327–329. [Google Scholar] [CrossRef] [PubMed]
- Xiong, S.; Liu, A.Q.; Chin, L.K.; Yang, Y. An optofluidic prism tuned by two laminar flows. Lab Chip 2011, 11, 1864–1869. [Google Scholar] [CrossRef] [PubMed]
- Tang, S.K.Y.; Stan, C.A.; Whitesides, G.M. Dynamically reconfigurable liquid-core liquid-cladding lens in a microfluidic channel. Lab Chip 2008, 8, 395–401. [Google Scholar] [CrossRef] [PubMed]
- Rosenauer, M.; Vellekoop, M.J. Characterization of a microflow cytometer with an integrated three-dimensional optofluidic lens system. Biomicrofluidics 2010, 4, 043005. [Google Scholar] [CrossRef] [PubMed]
- Song, C.; Nguyen, N.-T.; Asundi, A.K.; Low, C.L.-N. Tunable optofluidic aperture configured by a liquid-core/liquid-cladding structure. Opt. Lett. 2011, 36, 1767–1769. [Google Scholar] [CrossRef] [PubMed]
- Seow, Y.C.; Liu, A.Q.; Chin, L.K.; Li, X.C.; Huang, H.J.; Cheng, T.H.; Zhou, X.Q. Different curvatures of tunable liquid microlens via the control of laminar flow rate. Appl. Phys. Lett. 2008, 93, 084101. [Google Scholar] [CrossRef]
- Song, C.; Nguyen, N.T.; Asundi, A.K.; Low, C.L.N. Biconcave micro-optofluidic lens with low-refractive-index liquids. Opt. Lett. 2009, 34, 3622–3624. [Google Scholar] [CrossRef] [PubMed]
- Song, C.; Nguyen, N.T.; Tan, S.H.; Asundi, A.K. Modelling and optimization of micro optofluidic lenses. Lab Chip 2009, 9, 1178–1184. [Google Scholar] [CrossRef] [PubMed]
- Song, C.; Nguyen, N.T.; Tan, S.H.; Asundi, A.K. A tuneable micro-optofluidic biconvex lens with mathematically predictable focal length. Microfluid. Nanofluid. 2010, 9, 889–896. [Google Scholar] [CrossRef]
- Song, C.; Nguyen, N.T.; Tan, S.H.; Asundi, A.K. A micro optofluidic lens with short focal length. J. Micromech. Microeng. 2009, 19, 085012. [Google Scholar] [CrossRef]
- Song, C.; Nguyen, N.T.; Yap, Y.F.; Luong, T.D.; Asundi, A.K. Multi-functional, optofluidic, in-plane, bi-concave lens: Tuning light beam from focused to divergent. Microfluid. Nanofluid. 2010, 10, 671–678. [Google Scholar] [CrossRef]
- Kopp, D.; Lehmann, L.; Zappe, H. Optofluidic laser scanner based on a rotating liquid prism. Appl. Opt. 2016, 55, 2136–2142. [Google Scholar] [CrossRef] [PubMed]
- Heideman, R.G.; Kooyman, R.P.H.; Greve, J. Development of an optical waveguide interferometric immunosensor. Sens. Actuators B Chem. 1991, 4, 297–299. [Google Scholar] [CrossRef]
- Heideman, R.G.; Kooyman, R.P.H.; Greve, J. Performance of a highly sensitive optical waveguide mach-zehnder interferometer immunosensor. Sens. Actuators B Chem. 1993, 10, 209–217. [Google Scholar] [CrossRef]
- Ingenhoff, J.; Drapp, B.; Gauglitz, G. Biosensors using integrated optical devices. Fresenius J. Anal. Chem. 1993, 346, 580–583. [Google Scholar] [CrossRef]
- Gauglitz, G.; Ingenhoff, J. Design of new integrated optical substrates for immuno-analytical applications. Fresenius J. Anal. Chem. 1994, 349, 355–359. [Google Scholar] [CrossRef]
- Schipper, E.F.; Brugman, A.M.; Dominguez, C.; Lechuga, L.M.; Kooyman, R.P.H.; Greve, J. The realization of an integrated mach-zehnder waveguide immunosensor in silicon technology. Sens. Actuators B Chem. 1997, 40, 147–153. [Google Scholar] [CrossRef]
- Heideman, R.G.; Lambeck, P.V. Remote opto-chemical sensing with extreme sensitivity: Design, fabrication and performance of a pigtailed integrated optical phase-modulated Mach–Zehnder interferometer system. Sens. Actuators B Chem. 1999, 61, 100–127. [Google Scholar] [CrossRef]
- Crespi, A.; Gu, Y.; Ngamsom, B.; Hoekstra, H.J.W.M.; Dongre, C.; Pollnau, M.; Ramponi, R.; van den Vlekkert, H.H.; Watts, P.; Cerullo, G.; et al. Three-dimensional mach-zehnder interferometer in a microfluidic chip for spatially-resolved label-free detection. Lab Chip 2010, 10, 1167–1173. [Google Scholar] [CrossRef] [PubMed]
- Bedoya, A.C.; Monat, C.; Domachuk, P.; Grillet, C.; Eggleton, B.J. Measuring the dispersive properties of liquids using a microinterferometer. Appl. Opt. 2011, 50, 2408–2412. [Google Scholar] [CrossRef] [PubMed]
- Lapsley, M.I.; Chiang, I.K.; Zheng, Y.B.; Ding, X.; Mao, X.; Huang, T.J. A single-layer, planar, optofluidic mach-zehnder interferometer for label-free detection. Lab Chip 2011, 11, 1795–1800. [Google Scholar] [CrossRef] [PubMed]
- Dumais, P.; Callender, C.L.; Noad, J.P.; Ledderhof, C.J. Integrated optical sensor using a liquid-core waveguide in a mach-zehnder interferometer. Opt. Express 2008, 16, 18164–18172. [Google Scholar] [CrossRef] [PubMed]
- Bernini, R.; Testa, G.; Zeni, L.; Sarro, P.M. Integrated optofluidic mach–zehnder interferometer based on liquid core waveguides. Appl. Phys. Lett. 2008, 93, 011106. [Google Scholar] [CrossRef]
- Testa, G.; Huang, Y.; Sarro, P.M.; Zeni, L.; Bernini, R. High-visibility optofluidic Mach–Zehnder interferometer. Opt. Lett. 2010, 35, 1584–1586. [Google Scholar] [CrossRef] [PubMed]
- Yin, D.; Barber, J.P.; Hawkins, A.R.; Schmidt, H. Highly efficient fluorescence detection in picoliter volume liquid-core waveguides. Appl. Phys. Lett. 2005, 87, 211111. [Google Scholar] [CrossRef]
- Duguay, M.A.; Kokubun, Y.; Koch, T.L.; Pfeiffer, L. Antiresonant reflecting optical waveguides in SiO2-Si multilayer structures. Appl. Phys. Lett. 1986, 49, 13–15. [Google Scholar] [CrossRef]
- Yin, D.; Deamer, D.W.; Schmidt, H.; Barber, J.P.; Hawkins, A.R. Single-molecule detection sensitivity using planar integrated optics on a chip. Opt. Lett. 2006, 31, 2136–2138. [Google Scholar] [CrossRef] [PubMed]
- Testa, G.; Persichetti, G.; Sarro, P.M.; Bernini, R. A hybrid silicon-PDMS optofluidic platform for sensing applications. Biomed. Opt. Express 2014, 5, 417–426. [Google Scholar] [CrossRef] [PubMed]
- Campopiano, S.; Bernini, R.; Zeni, L.; Sarro, P.M. Microfluidic sensor based on integrated optical hollow waveguides. Opt. Lett. 2004, 29, 1894–1896. [Google Scholar] [CrossRef] [PubMed]
- Dongre, C.; van Weerd, J.; Bellini, N.; Osellame, R.; Cerullo, G.; van Weeghel, R.; Hoekstra, H.J.W.M.; Pollnau, M. Dual-point dual-wavelength fluorescence monitoring of DNA separation in a lab on a chip. Biomed. Opt. Express 2010, 1, 729–735. [Google Scholar] [CrossRef] [PubMed]
- Bernini, R.; De Nuccio, E.; Minardo, A.; Zeni, L.; Sarro, P.M. Integrated silicon optical sensors based on hollow core waveguide. Proc. SPIE 2007, 6477, 647714. [Google Scholar]
- Yu, J.Q.; Yang, Y.; Liu, A.Q.; Chin, L.K.; Zhang, X.M. Microfluidic droplet grating for reconfigurable optical diffraction. Opt. Lett. 2010, 35, 1890–1892. [Google Scholar] [CrossRef] [PubMed]
- Mao, X.; Lin, S.C.S.; Dong, C.; Huang, T.J. Single-layer planar on-chip flow cytometer using microfluidic drifting based three-dimensional (3D) hydrodynamic focusing. Lab Chip 2009, 9, 1583–1589. [Google Scholar] [CrossRef] [PubMed]
- Tung, Y.C.; Zhang, M.; Lin, C.T.; Kurabayashi, K.; Skerlos, S.J. PDMS-based opto-fluidic micro flow cytometer with two-color, multi-angle fluorescence detection capability using pin photodiodes. Sens. Actuators B Chem. 2004, 98, 356–367. [Google Scholar] [CrossRef]
- Pamme, N.; Koyama, R.; Manz, A. Counting and sizing of particles and particle agglomerates in a microfluidic device using laser light scattering: Application to a particle-enhanced immunoassay. Lab Chip 2003, 3, 187–192. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; El-Ali, J.; Engelund, M.; Gotsæd, T.; Perch-Nielsen, I.R.; Mogensen, K.B.; Snakenborg, D.; Kutter, J.P.; Wolff, A. Measurements of scattered light on a microchip flow cytometer with integrated polymer based optical elements. Lab Chip 2004, 4, 372–377. [Google Scholar] [CrossRef] [PubMed]
- Godin, J.; Lien, V.; Lo, Y.-H. Demonstration of two-dimensional fluidic lens for integration into microfluidic flow cytometers. Appl. Phys. Lett. 2006, 89, 061106. [Google Scholar] [CrossRef]
- Barat, D.; Benazzi, G.; Mowlem, M.C.; Ruano, J.M.; Morgan, H. Design, simulation and characterisation of integrated optics for a microfabricated flow cytometer. Opt. Commun. 2010, 283, 1987–1992. [Google Scholar] [CrossRef]
- Song, C.; Luong, T.-D.; Kong, T.F.; Nguyen, N.-T.; Asundi, A.K. Disposable flow cytometer with high efficiency in particle counting and sizing using an optofluidic lens. Opt. Lett. 2011, 36, 657–659. [Google Scholar] [CrossRef] [PubMed]
- Ashkin, A. Acceleration and trapping of particles by radiation pressure. Phys. Rev. Lett. 1970, 24, 156–159. [Google Scholar] [CrossRef]
- Ashkin, A.; Dziedzic, J.M.; Bjorkholm, J.E.; Chu, S. Observation of a single-beam gradient force optical trap for dielectric particles. Opt. Lett. 1986, 11, 288–290. [Google Scholar] [CrossRef] [PubMed]
- Lin, H.-C.; Hsu, L. Dynamic and Programmable Cell-Sorting by Using Microfluidics and Holographic Optical Tweezers. Proc. SPIE 2005, 5930, 59301–59308. [Google Scholar]
- Wang, M.M.; Tu, E.; Raymond, D.E.; Yang, J.M.; Zhang, H.; Hagen, N.; Dees, B.; Mercer, E.M.; Forster, A.H.; Kariv, I.; et al. Microfluidic sorting of mammalian cells by optical force switching. Nat. Biotechnol. 2005, 23, 83–87. [Google Scholar] [CrossRef] [PubMed]
- Applegate, R.W.; Squier, J.; Vestad, T.; Oakey, J.; Marr, D.W.M. Optical trapping, manipulation, and sorting of cells and colloids in microfluidic systems with diode laser bars. Opt. Express 2004, 12, 4390–4398. [Google Scholar] [CrossRef] [PubMed]
- Lee, K.S.; Lee, K.H.; Kim, S.B.; Jung, J.H.; Ha, B.H.; Sung, H.J.; Kim, S.S. Refractive-index-based optofluidic particle manipulation. Appl. Phys. Lett. 2013, 103, 073701. [Google Scholar]
- Lee, K.H.; Lee, K.S.; Jung, J.H.; Chang, C.B.; Sung, H.J. Optical mobility of blood cells for label-free cell separation applications. Appl. Phys. Lett. 2013, 102, 141911. [Google Scholar]
- Kim, S.B.; Yoon, S.Y.; Sung, H.J.; Kim, S.S. Cross-type optical particle separation in a microchannel. Anal. Chem. 2008, 80, 2628–2630. [Google Scholar] [CrossRef] [PubMed]
- Tag Optics. Available online: Http://www.tag-optics.com/TL2ProductLine.php (accessed on 1 April 2017).
- Varioptic. Available online: Http://www.Varioptic.Com/products/variable-focus/ (accessed on 1 April 2017).
- Optotune. Available online: Http://www.Optotune.Com/ (accessed on 1 April 2017).
- World Precision Instruments. Available online: Https://www.Wpiinc.Com/ (accessed on 1 April 2017).
- Nanotweezer Developed by Optofluidics. Available online: Http://opfluid.Com/products/nanotweezer/ (accessed on 1 April 2017).
- Yit-Fatt, Y.; Say-Hwa, T.; Nam-Trung, N.; Murshed, S.M.S.; Teck-Neng, W.; Levent, Y. Thermally mediated control of liquid microdroplets at a bifurcation. J. Phys. D Appl. Phys. 2009, 42, 065503. [Google Scholar]
- Say-Hwa, T.; Murshed, S.M.S.; Nam-Trung, N.; Teck Neng, W.; Levent, Y. Thermally controlled droplet formation in flow focusing geometry: Formation regimes and effect of nanoparticle suspension. J. Phys. D Appl. Phys. 2008, 41, 165501. [Google Scholar]
- Xi, H.-D.; Guo, W.; Leniart, M.; Chong, Z.Z.; Tan, S.H. AC electric field induced droplet deformation in a microfluidic T-junction. Lab Chip 2016, 16, 2982–2986. [Google Scholar] [CrossRef] [PubMed]
- Tan, S.H.; Semin, B.; Baret, J.-C. Microfluidic flow-focusing in ac electric fields. Lab Chip 2014, 14, 1099–1106. [Google Scholar] [CrossRef] [PubMed]
- Ma, Z.; Teo, A.; Tan, S.; Ai, Y.; Nguyen, N.-T. Self-aligned interdigitated transducers for acoustofluidics. Micromachines 2016, 7, 216. [Google Scholar] [CrossRef]

© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Song, C.; Tan, S.H. A Perspective on the Rise of Optofluidics and the Future. Micromachines 2017, 8, 152. https://doi.org/10.3390/mi8050152
Song C, Tan SH. A Perspective on the Rise of Optofluidics and the Future. Micromachines. 2017; 8(5):152. https://doi.org/10.3390/mi8050152
Chicago/Turabian StyleSong, Chaolong, and Say Hwa Tan. 2017. "A Perspective on the Rise of Optofluidics and the Future" Micromachines 8, no. 5: 152. https://doi.org/10.3390/mi8050152
APA StyleSong, C., & Tan, S. H. (2017). A Perspective on the Rise of Optofluidics and the Future. Micromachines, 8(5), 152. https://doi.org/10.3390/mi8050152






