Microfluidic Device to Measure the Speed of C. elegans Using the Resistance Change of the Flexible Electrode
Abstract
:1. Introduction
2. Materials and Methods
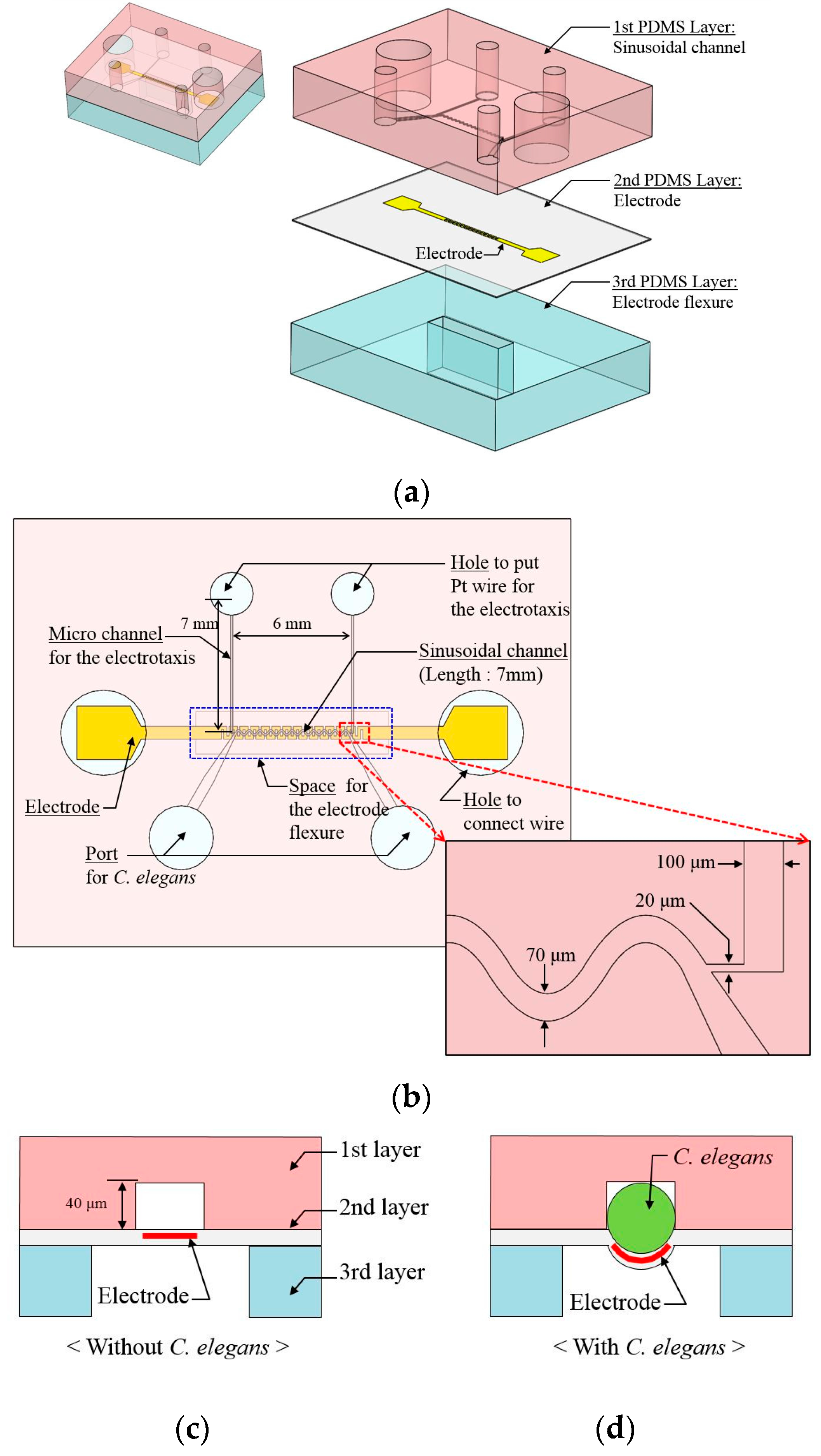
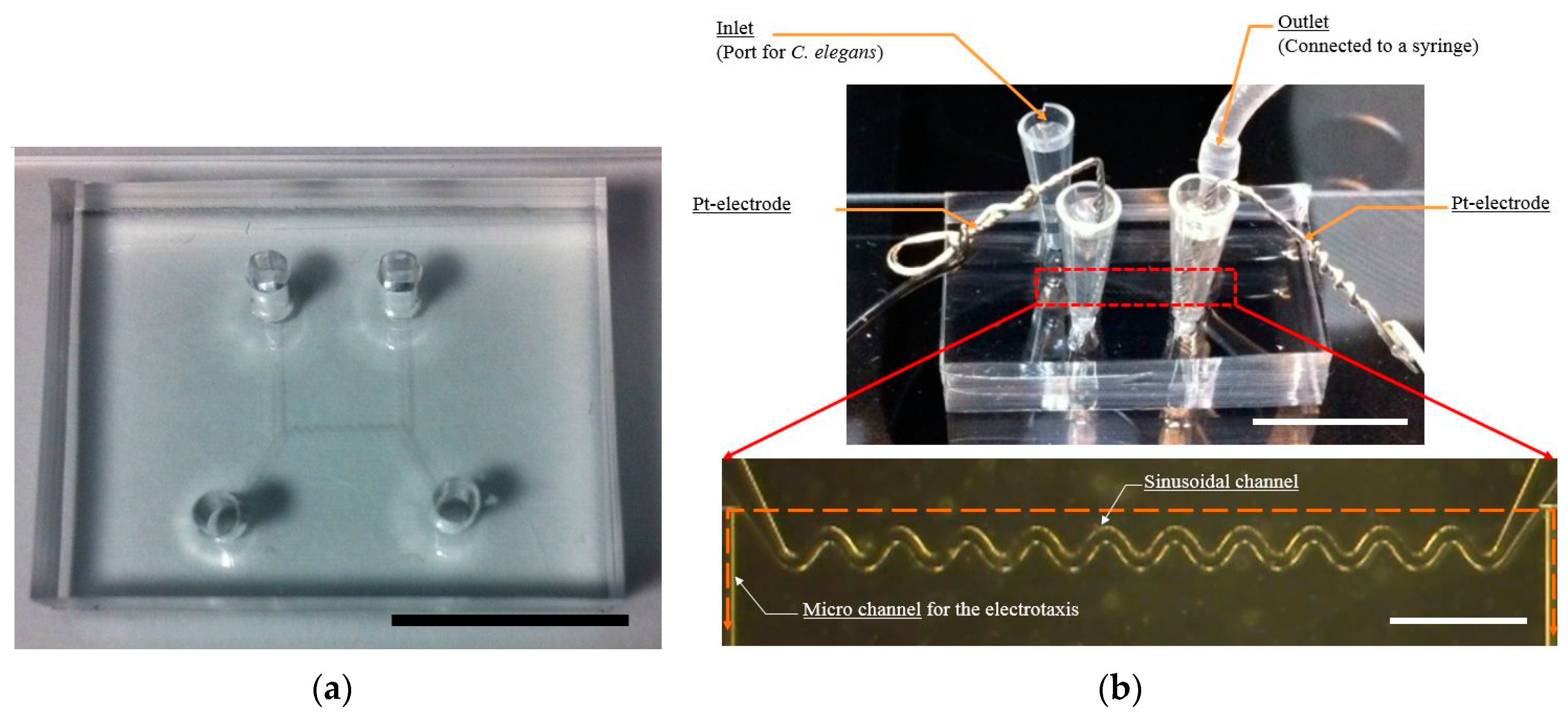
2.1. Microfluidic Device Design
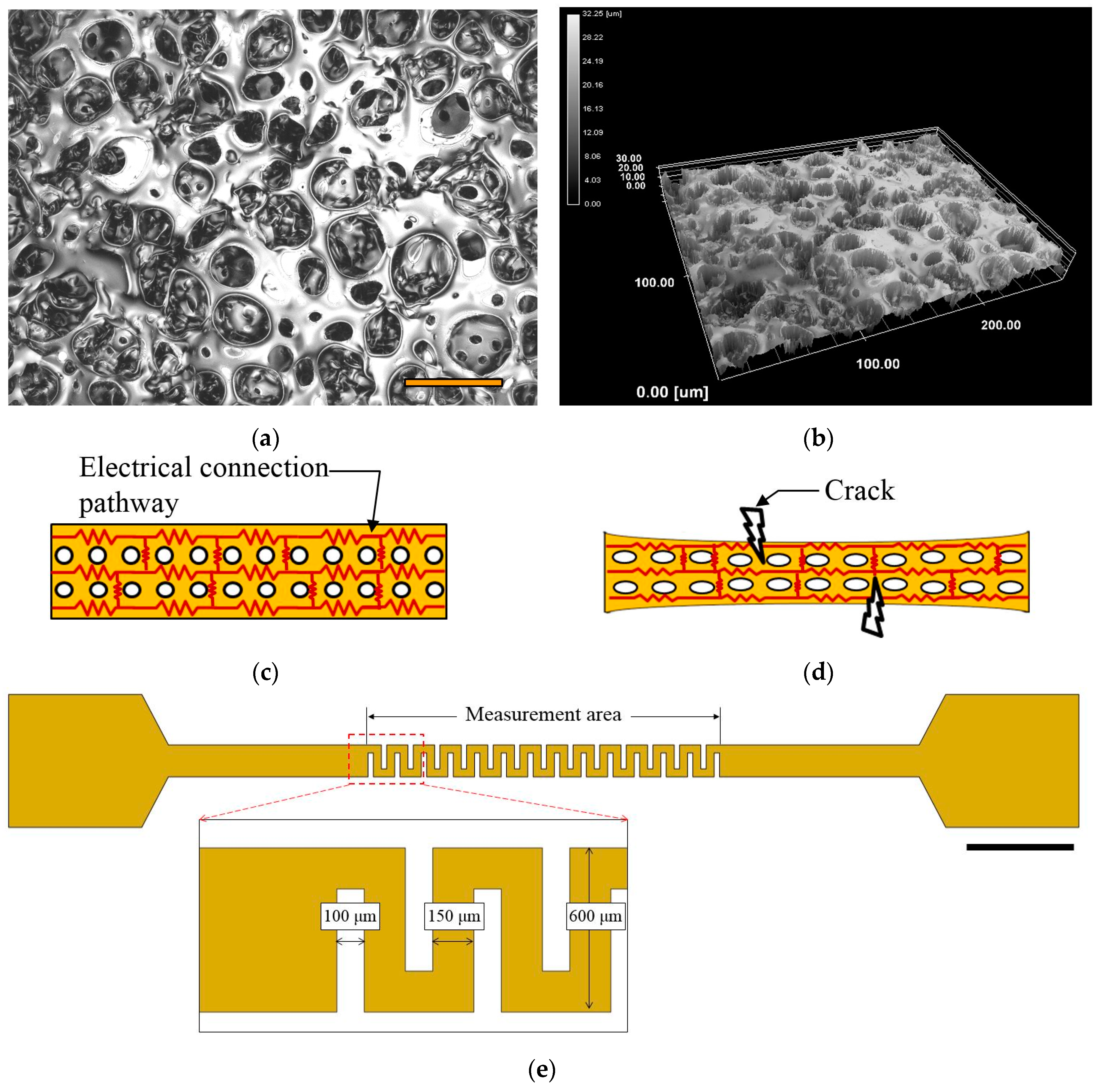
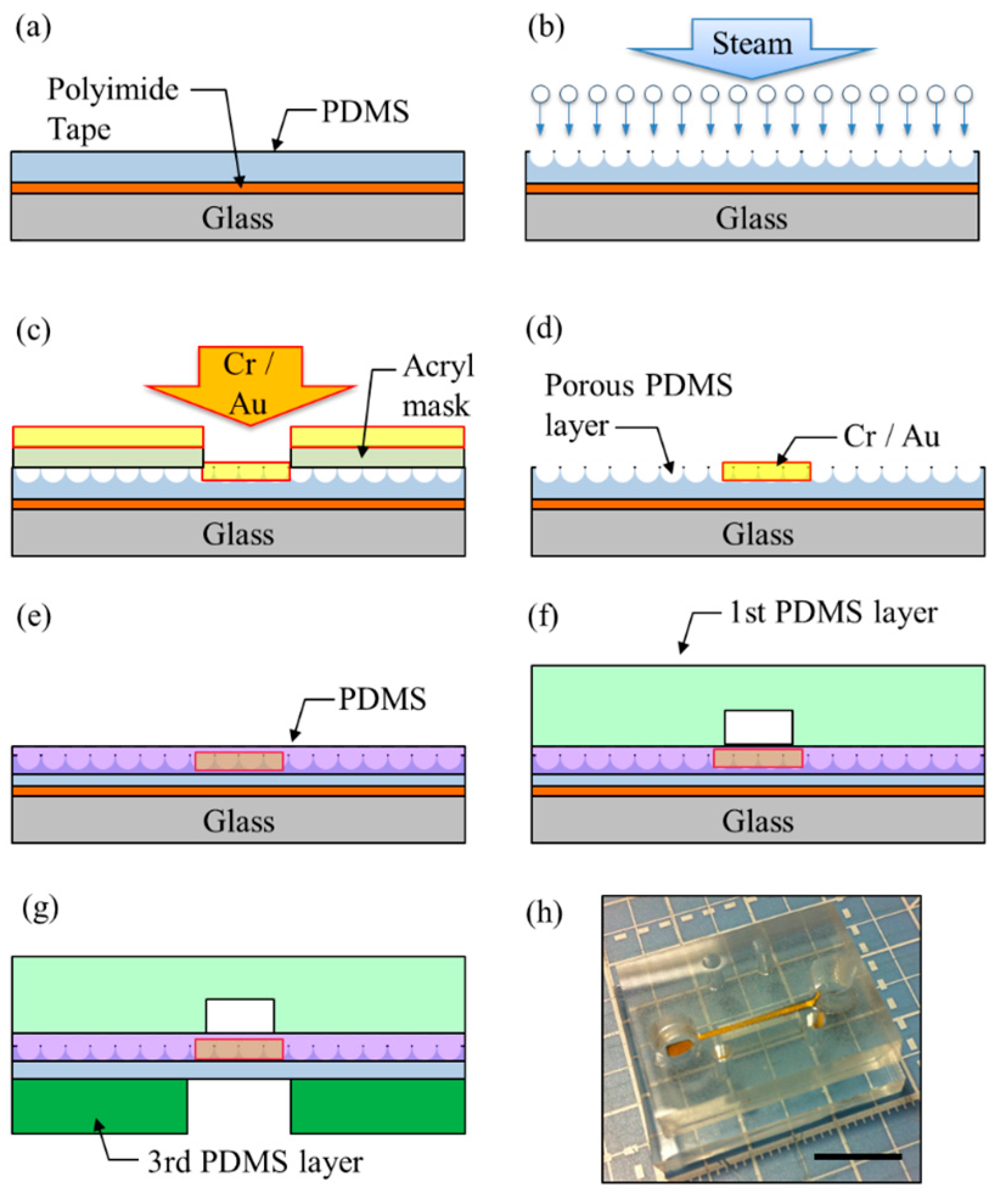
2.2. Fabrication of the Microfluidic Device
2.3. Preparation of C. elegans Strain
3. Results and Discussions
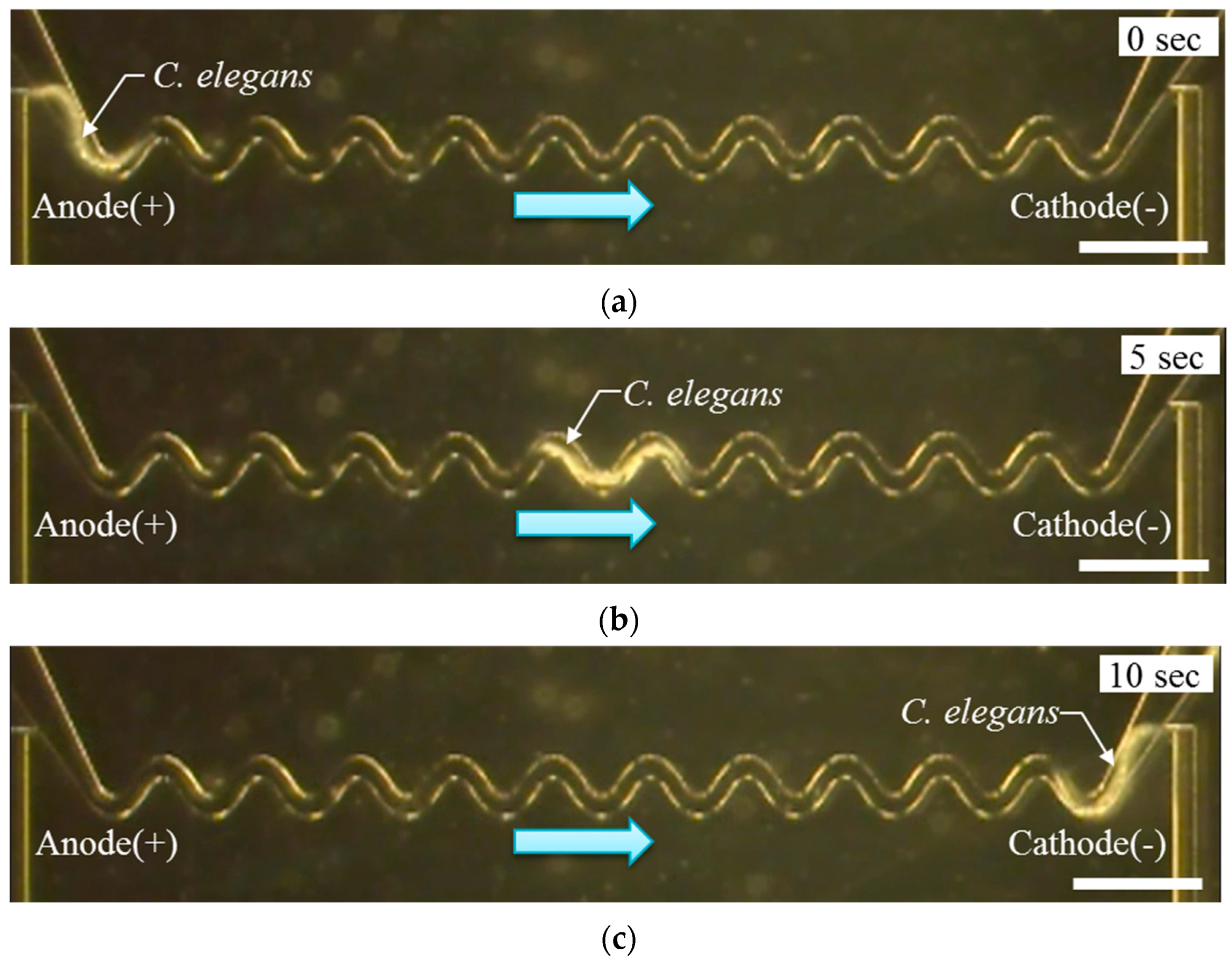
3.1. Electrotaxis Test Result
3.2. Speed Measurement Using Resistance Change
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Braungart, E.; Gerlach, M.; Riederer, P.; Baumeister, R.; Hoenerd, M.C. Caenorhabditis elegans MPP+ Model of Parkinson’s Disease for High-Throughput Drug Screenings. Neurodegener. Dis. 2004, 1, 175–183. [Google Scholar] [CrossRef] [PubMed]
- Ainscough, R.; Bardill, S.; Barlow, K. The C. elegans Sequencing Consortium. Genome sequence of the nematode C. elegans: A platform for investigating biology. Science 1998, 282, 2012–2018. [Google Scholar]
- Kaletta, T.; Hengartner, M.O. Finding function in novel targets: C. elegans as a model organism. Nat. Rev. Drug Discov. 2006, 5, 387–398. [Google Scholar] [CrossRef] [PubMed]
- Johnson, J.R.; Jenn, R.C.; Barclay, J.W.; Burgoyne, R.D.; Morgan, A. Caenorhabditis elegans: A useful tool to decipher neurodegenerative pathways. Biochem. Soc. Trans. 2010, 38, 559–563. [Google Scholar] [CrossRef] [PubMed]
- Voisine, C.; Hart, A.C. Caenorhabditis elegans as a model system for triplet repeat diseases. Methods Mol. Biol. 2004, 277, 141–160. [Google Scholar] [PubMed]
- Lakso, M.; Vartiainen, S.; Moilanen, A.; Sirviö, J.; Thomas, J.H.; Nass, R.; Blakely, R.D.; Wong, G. Dopaminergic neuronal loss and motor deficits in Caenorhabditis elegans overexpressing human alpha-synuclein. J. Neurochem. 2003, 86, 165–172. [Google Scholar] [CrossRef] [PubMed]
- Adam, J.H.; Shusei, H.; Guy, A.C.; Kim, A.C. C. elegans as a Model Organism to Investigate Molecular Pathways Involved with Parkinson’s Disease. Dev. Dyn. 2010, 239, 1282–1295. [Google Scholar]
- Rezai, P. Microfluidic Device for Nematode-Based Behavioral Assays Using Electrotaxis. Ph.D. Thesis, McMaster University, Hamilton, ON, Canada, 2010. [Google Scholar]
- Segalat, L. Invertebrate animal models of diseases as screening tools in drug discovery. ACS Chem. Biol. 2007, 2, 231–236. [Google Scholar] [CrossRef] [PubMed]
- Kirienko, N.V.; Mani, K.; Fay, D.S. Cancer models in Caenorhabditis elegans. Dev. Dyn. 2010, 239, 1413–1448. [Google Scholar] [PubMed]
- Hall, D.H.; Altun, Z.F. C. elegans Atlas; Cold Spring Harbor Laboratory Press: New York, NY, USA, 2008; pp. 1–15. [Google Scholar]
- Sulston, J.E.; Horvitz, H.R. Post-embryonic cell lineages of the nematode, Caenorhabditis elegans. Dev. Biol. 1977, 56, 110–156. [Google Scholar] [CrossRef]
- White, J.G.; Southgate, E.; Thomson, J.N.; Brenner, S. The structure of the nervous system of the nematode Caenorhabditis elegans. Philos. Trans. R. Soc. B 1986, 314, 1–340. [Google Scholar] [CrossRef]
- Kamath, R.S.; Fraser, A.G.; Dong, Y.; Poulin, G.; Durbin, R.; Gotta, M.; Kanapin, A.; Bot, N.L.; Moreno, S.; Sohrmann, M.; et al. Systematic functional analysis of the Caenorhabditis elegans genome using RNAi. Nature 2003, 421, 231–237. [Google Scholar] [CrossRef] [PubMed]
- Kamath, R.S.; Ahringer, J. Genorne-wide RNAi screening in Caenorhabditis elegans. Methods 2003, 30, 313–321. [Google Scholar] [CrossRef]
- Bargmann, C.I. Chemosensation in C. elegans. WormBook; The C. elegans Research Community: New York, NY, USA, 2006; pp. 1–29. [Google Scholar]
- Bergamasco, C.; Bazzicalupo, P. Chemical sensitivity in C. elegans. Cell. Mol. Life Sci. 2006, 63, 1510–1522. [Google Scholar] [CrossRef] [PubMed]
- Croll, N.A. Behavioural analysis of nematode movement. Adv. Parasitol. 1975, 13, 71–122. [Google Scholar] [PubMed]
- Lockery, S.R.; Lawton, K.J.; Doll, J.C.; Faumont, S.; Coulthard, S.M.; Thiele, T.R.; Chronis, N.; McCormick, K.E.; Goodman, M.B.; Pruitt, B.L.; et al. Artificial Dirt: Microfluidic Substrates for Nematode Neurobiology and Behavior. J. Neurophysiol. 2008, 99, 3136–3143. [Google Scholar] [CrossRef] [PubMed]
- Shingai, R. Durations and frequencies of free locomotion in wild type and GABAergic mutants of Caenorhabditis elegans. Neurosci. Res. 2000, 38, 71–83. [Google Scholar] [CrossRef]
- Dixon, S.J.; Roy, P.J. Muscle arm development in Caenorhabditis elegans. Development 2005, 132, 3079–3092. [Google Scholar] [CrossRef] [PubMed]
- Shen, X.N.; Sznitman, J.; Krajacic, P.; Lamitina, T.; Arratia, P.E. Undulatory Locomotion of Caenorhabditis elegans on Wet Surfaces. Biophys. J. 2012, 102, 2772–2781. [Google Scholar] [CrossRef] [PubMed]
- Boyd, W.A.; Smith, M.V.; Kissling, G.E.; Freedman, J.H. Medium- and high-throughput screening of neurotoxicants using C. elegans. Neurotoxicol. Teratol. 2010, 32, 68–73. [Google Scholar] [CrossRef] [PubMed]
- Ghanbari, A.; Nock, V.; Johari, S.; Blaikie, R.J.; Chen, X.; Wang, W. A micro-pillar-based on-chip system for continuous force measurement of C. elegans. J. Micromech. Microeng. 2012, 22, 095009. [Google Scholar] [CrossRef]
- Wang, D.Y.; Yang, P. Multi-biological defects caused by lead exposure exhibit transferable properties from exposed parents to their progeny in Caenorhabditis elegans. J. Environ. Sci. 2007, 19, 1367–1372. [Google Scholar] [CrossRef]
- Anderson, G.L.; Boyd, W.A.; Williams, P.L. Assessment of sublethal endpoints for toxicity testing with the nematode Caenorhabditis elegans. Environ. Toxicol. Chem. 2001, 20, 833–838. [Google Scholar] [CrossRef] [PubMed]
- Wang, D.; Xing, X. Assessment of locomotion behavioral defects induced by acute toxicity from heavy metal exposure in nematode Caenorhabditis elegans. J. Environ. Sci. 2008, 20, 1132–1137. [Google Scholar] [CrossRef]
- Doll, J.C.; Harjee, N.; Klejwa, N.; Kwon, R.; Coulthard, S.M.; Petzold, B.; Goodman, M.B.; Pruitt, P.L. SU-8 force sensing pillar arrays for biological measurements. Lab Chip 2009, 9, 1449–1454. [Google Scholar] [CrossRef] [PubMed]
- Liu, P.; Mao, D.; Martin, R.J.; Dong, L. An integrated fiber-optic microfluidic device for detection of muscular force generation of microscopic nematodes. Lab Chip 2012, 12, 3458–3466. [Google Scholar] [CrossRef] [PubMed]
- Parashar, A.; Lycke, R.; Carr, J.A.; Pandey, S. Amplitude-modulated sinusoidal microchannels for observing adaptability in C. elegans locomotion. Biomicrofluidics 2011, 5, 024112. [Google Scholar] [CrossRef] [PubMed]
- Sukul, N.C.; Croll, N.A. Influence of Potential Difference and Current on the Electrotaxis of Caenorhabditis elegans. J. Nematol. 1978, 10, 314–317. [Google Scholar] [PubMed]
- Gabel, C.V.; Gabel, H.; Pavlichin, D.; Kao, A.; Clark, D.A.; Samuel, A.D. Neural Circuits Mediate Electrosensory Behavior in Caenorhabditis elegans. J. Neurosci. 2007, 27, 7586–7596. [Google Scholar] [CrossRef] [PubMed]
- Viglierchio, D.R.; Yu, P.K. On nematode behavior in an electric field. Rev. Nematol. 1983, 6, 171–178. [Google Scholar]
- Rezai, P.; Siddiqui, A.; Ravi, P.; Selvaganapathy, P.R.; Gupta, B.P. Electrotaxis of Caenorhabditis elegans in a microfluidic environment. Lab Chip 2010, 10, 220–226. [Google Scholar] [CrossRef] [PubMed]
- Jeong, G.S.; Baek, D.H.; Jung, H.C.; Song, J.H.; Moon, J.H.; Hong, S.W.; Kim, I.Y.; Lee, S.H. Solderable and electroplatable flexible electronic circuit on a porous stretchable elastomer. Nat. Commun. 2012, 3, 977. [Google Scholar] [CrossRef] [PubMed]
- Whitesides, G.M.; Ostuni, E.; Takayama, S.; Jiang, X.; Ingber, D.E. Soft lithography in biology and biochemistry. Annu. Rev. Biomed. Eng. 2001, 3, 335–373. [Google Scholar] [CrossRef] [PubMed]
- Weibel, D.B.; Diluzio, W.R.; Whitesides, G.M. Microfabrication meets microbiology. Nat. Rev. Microbiol. 2007, 5, 209–218. [Google Scholar] [CrossRef] [PubMed]
- Brenner, S. The genetics of Caenorhabditis elegans. Genetics 1974, 77, 71–94. [Google Scholar] [PubMed]
- Byerly, L.; Cassada, R.; Russell, R. The life cycle of the nematode Caenorhabditis elegans. Dev. Biol. 1976, 51, 23–33. [Google Scholar] [CrossRef]
- Bischof, L.J.; Huffman, D.J.; Aroian, R.J. C. elegans: Methods and Applications; Humana Press: Totowa, NJ, USA, 2006; pp. 139–154. [Google Scholar]
- Williams, P.L.; Dusenbery, D.B. Aquatic toxicity testing using the nematode, Caenorhabditis elegans. Environ. Toxicol. Chem. 1990, 9, 1285–1290. [Google Scholar] [CrossRef]

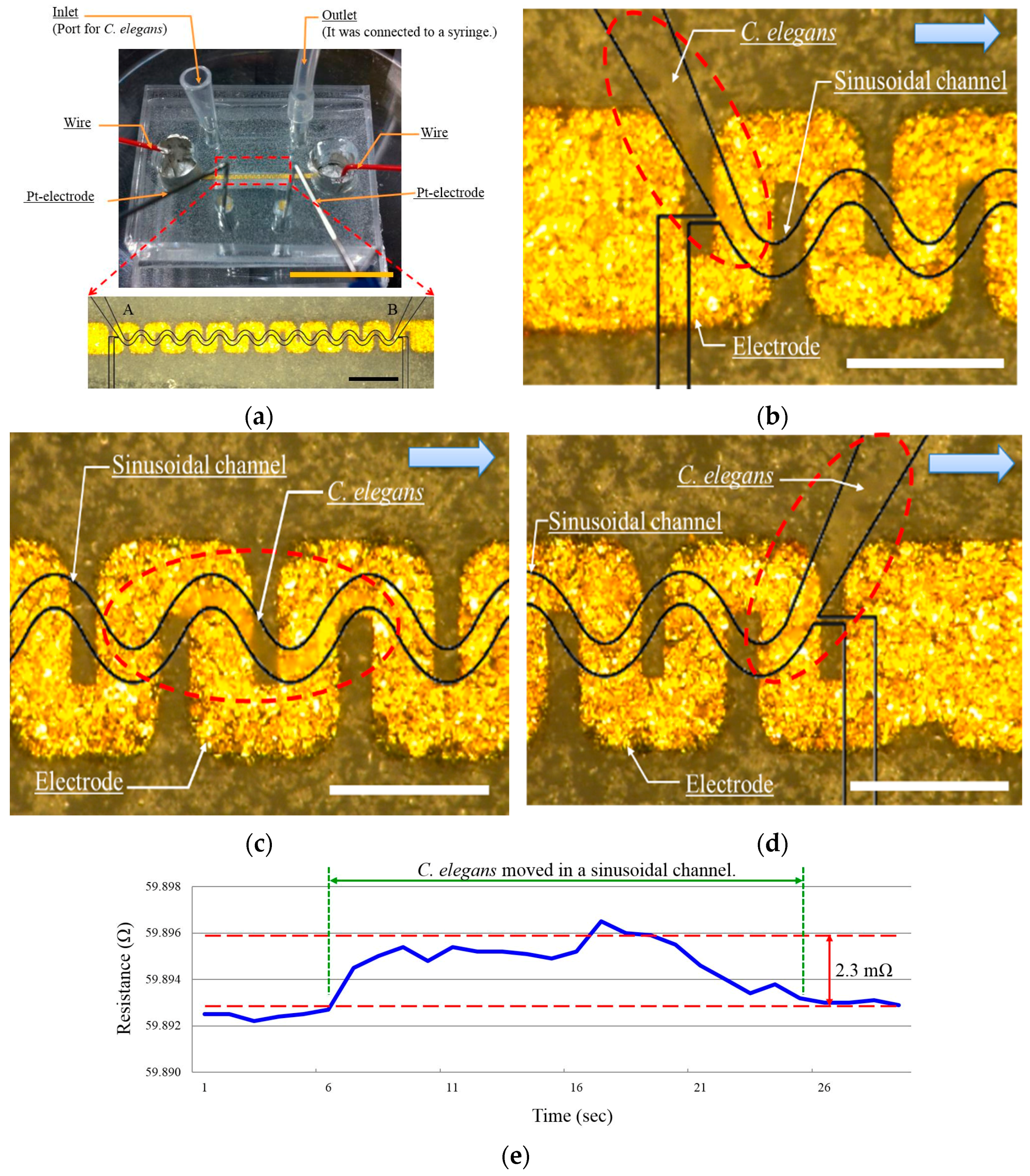
| No | Travel Time (s) | Resistance Change (mΩ) | Speed (mm/s) |
|---|---|---|---|
| 1 | 18.0 | 1.7 | 0.39 |
| 2 | 20.0 | 1.4 | 0.35 |
| 3 | 23.0 | 2.0 | 0.30 |
| 4 | 25.0 | 1.0 | 0.28 |
| 5 | 18.0 | 2.5 | 0.39 |
| 6 | 18.0 | 2.3 | 0.39 |
| Average (±SD) | 20.3 (±3.0) | 1.8 (±0.6) | 0.35 (±0.05) |
© 2016 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jung, J.; Nakajima, M.; Takeuchi, M.; Najdovski, Z.; Huang, Q.; Fukuda, T. Microfluidic Device to Measure the Speed of C. elegans Using the Resistance Change of the Flexible Electrode. Micromachines 2016, 7, 50. https://doi.org/10.3390/mi7030050
Jung J, Nakajima M, Takeuchi M, Najdovski Z, Huang Q, Fukuda T. Microfluidic Device to Measure the Speed of C. elegans Using the Resistance Change of the Flexible Electrode. Micromachines. 2016; 7(3):50. https://doi.org/10.3390/mi7030050
Chicago/Turabian StyleJung, Jaehoon, Masahiro Nakajima, Masaru Takeuchi, Zoran Najdovski, Qiang Huang, and Toshio Fukuda. 2016. "Microfluidic Device to Measure the Speed of C. elegans Using the Resistance Change of the Flexible Electrode" Micromachines 7, no. 3: 50. https://doi.org/10.3390/mi7030050
APA StyleJung, J., Nakajima, M., Takeuchi, M., Najdovski, Z., Huang, Q., & Fukuda, T. (2016). Microfluidic Device to Measure the Speed of C. elegans Using the Resistance Change of the Flexible Electrode. Micromachines, 7(3), 50. https://doi.org/10.3390/mi7030050






