In Vivo Characterization of Intracortical Probes with Focused Ion Beam-Etched Nanopatterned Topographies
Abstract
1. Introduction
2. Materials and Methods
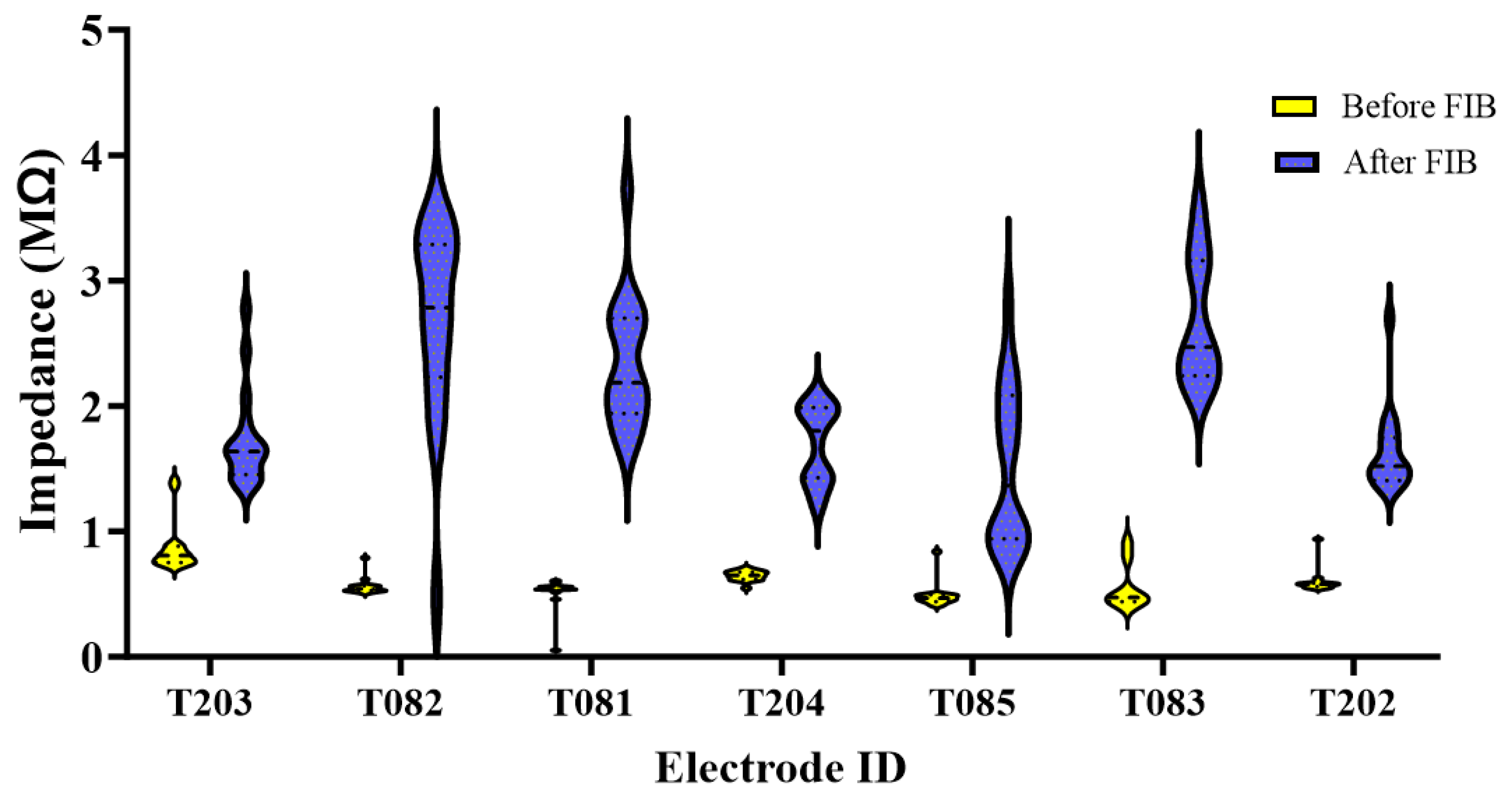
2.1. Neural Probe Manufacturing
2.2. Animals Surgeries
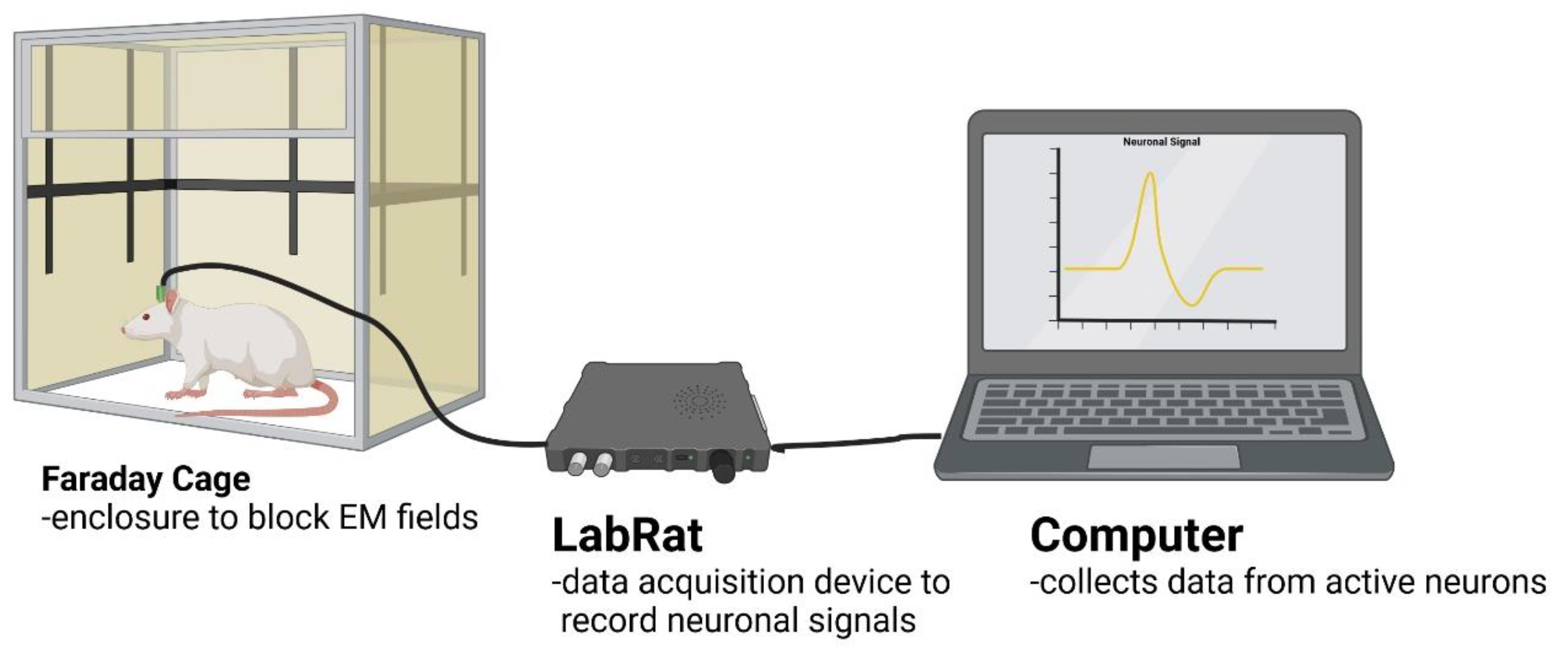
2.3. Neural Recordings
2.4. Recording Analysis
2.5. Cardiac Perfusions
2.6. RNA Isolation
2.7. Gene Expression Assay
2.8. Data Visualization and Statistical Analysis
3. Results
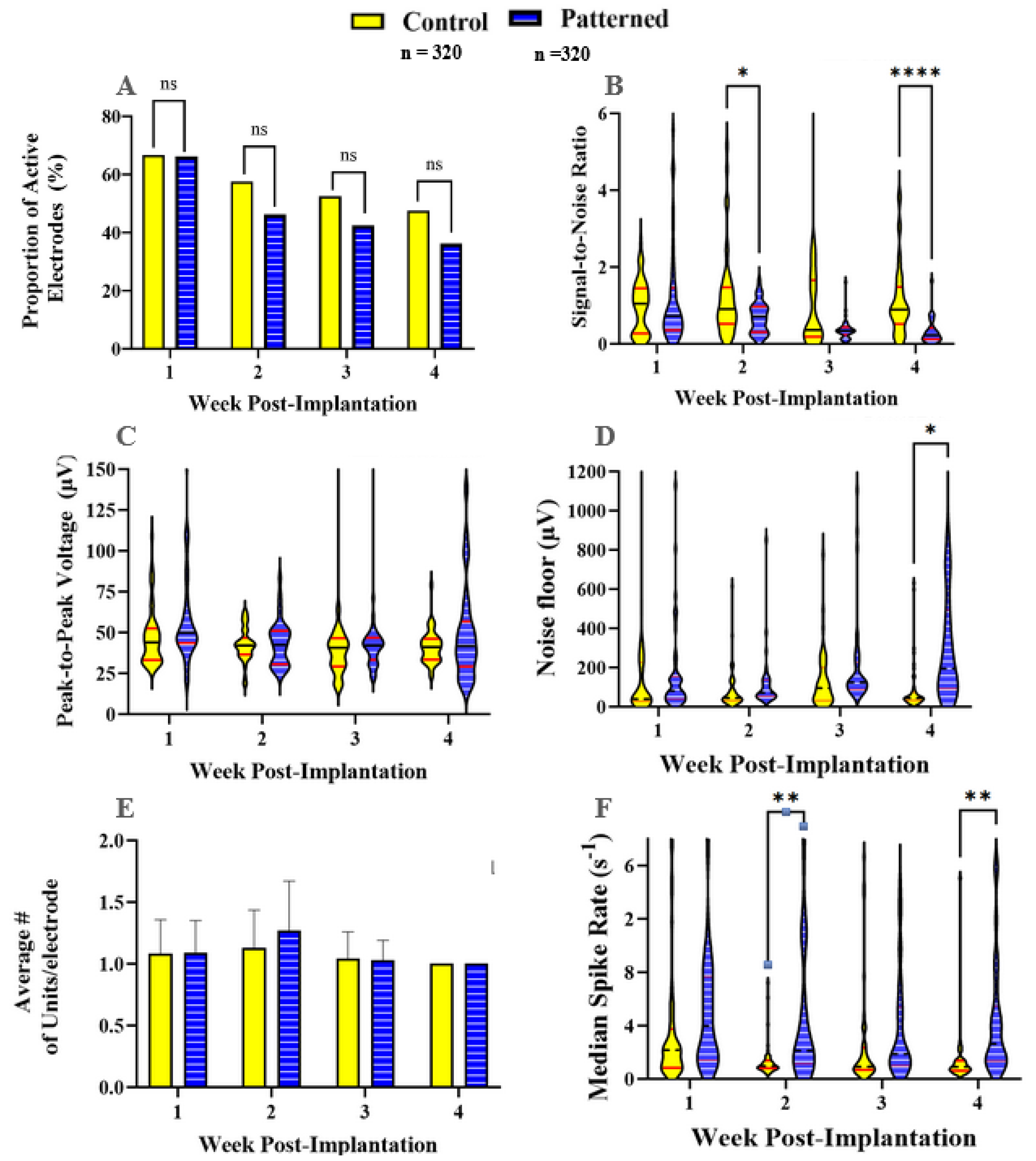
3.1. Recording Metrics
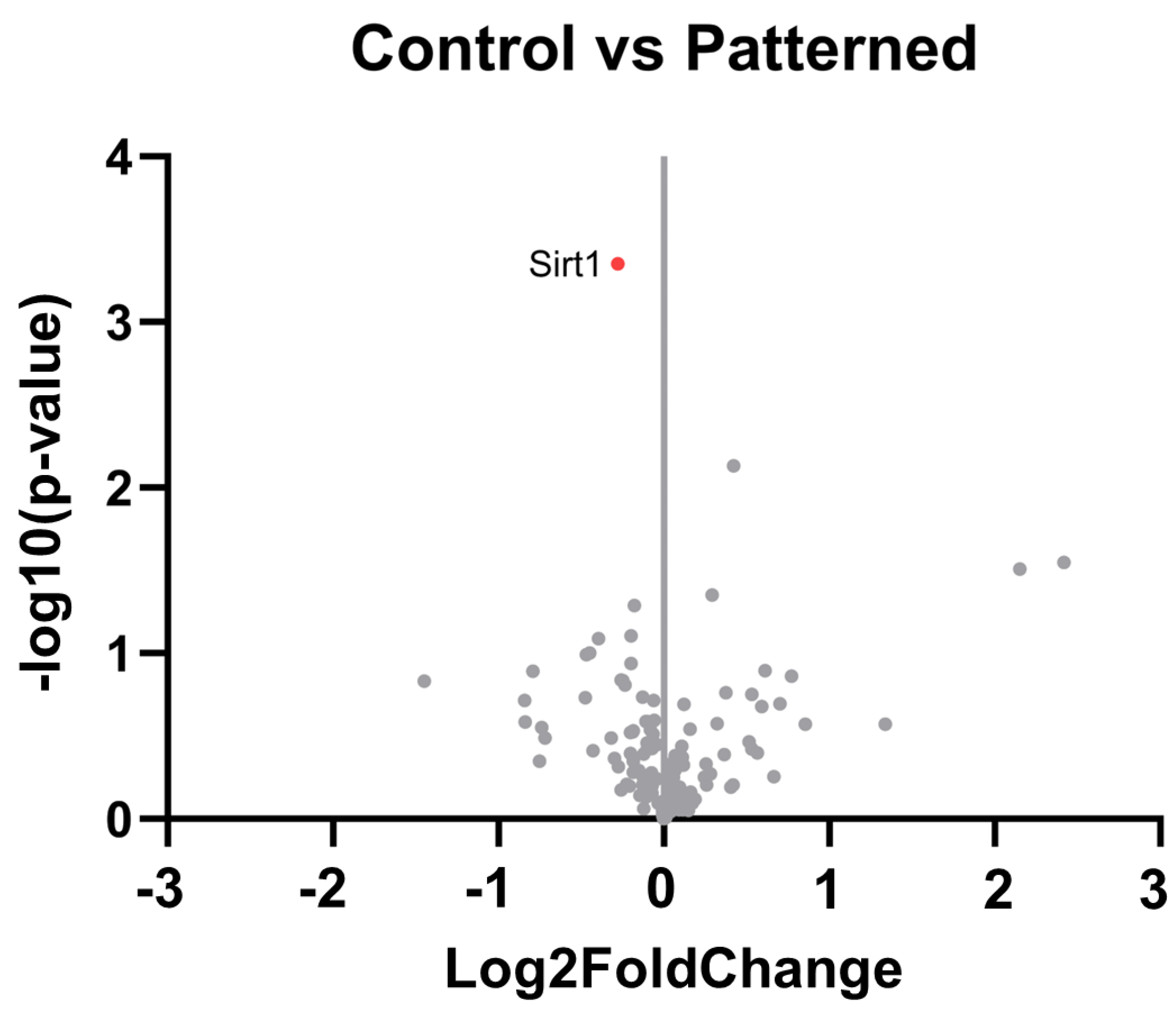
3.2. Gene Expression Analysis
4. Discussion
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Bullard, A.J.; Hutchison, B.C.; Lee, J.; Chestek, C.A.; Patil, P.G. Estimating Risk for Future Intracranial, Fully Implanted, Modular Neuroprosthetic Systems: A Systematic Review of Hardware Complications in Clinical Deep Brain Stimulation and Experimental Human Intracortical Arrays. Neuromodulation 2020, 23, 411–426. [Google Scholar] [CrossRef]
- Collinger, J.L.; Wodlinger, B.; Downey, J.E.; Wang, W.; Tyler-Kabara, E.C.; Weber, D.J.; McMorland, A.J.; Velliste, M.; Boninger, M.L.; Schwartz, A.B. High-performance neuroprosthetic control by an individual with tetraplegia. Lancet 2013, 381, 557–564. [Google Scholar] [CrossRef]
- Ajiboye, A.B.; Willett, F.R.; Young, D.R.; Memberg, W.D.; Murphy, B.A.; Miller, J.P.; Walter, B.L.; Sweet, J.A.; Hoyen, H.A.; Keith, M.W.; et al. Restoration of reaching and grasping movements through brain-controlled muscle stimulation in a person with tetraplegia: A proof-of-concept demonstration. Lancet 2017, 398, 1821–1830. [Google Scholar] [CrossRef]
- Willett, F.R.; Avansino, D.T.; Hochberg, L.R.; Henderson, J.M.; Shenoy, K.V. High-performance brain-to-text communication via handwriting. Nature 2021, 593, 249–254. [Google Scholar] [CrossRef] [PubMed]
- Kim, R.; Sejnowski, T.J. Strong inhibitory signaling underlies stable temporal dynamics and working memory in spiking neural networks. Nat. Neurosci. 2021, 24, 129–139. [Google Scholar] [CrossRef] [PubMed]
- Liew, Y.J.; Pala, A.; Whitmire, C.J.; Stoy, W.A.; Forest, C.R.; Stanley, G.B. Inferring thalamocortical monosynaptic connectivity in vivo. J. Neurophysiol. 2021, 125, 2408–2431. [Google Scholar] [CrossRef] [PubMed]
- Moffitt, M.A.; McIntyre, C.C. Model-based analysis of cortical recording with silicon microelectrodes. Clin. Neurophysiol. 2005, 116, 2240–2250. [Google Scholar] [CrossRef] [PubMed]
- Dunlap, C.F.; IV, S.C.C.; Meyers, E.C.; Bockbrader, M.A.; Friedenberg, D.A. Classifying intracortical brain-machine interface signal disruptions based on system performance and applicable compensatory strategies: A review. Front. Neurorobot. 2020, 14, 76. [Google Scholar] [CrossRef]
- Herwik, S.; Kisban, S.; Aarts, A.A.A.; Seidl, K.; Girardeau, G.; Benchenane, K.; Zugaro, M.B.; Wiener, S.I.; Paul, O.; Neves, H.P.; et al. Fabrication technology for silicon-based microprobe arrays used in acute and sub-chronic neural recording. J. Micromech. Microeng. 2009, 19, 074008. [Google Scholar] [CrossRef]
- Wise, K.D. Silicon microsystems for neuroscience and neural prostheses. IEEE Eng. Med. Biol. Mag. 2005, 24, 22–29. Available online: http://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Retrieve&db=PubMed&dopt=Citation&list_uids=16248114 (accessed on 1 February 2024). [CrossRef] [PubMed]
- Wellman, S.M.; Li, L.; Yaxiaer, Y.; McNamara, I.; Kozai, T.D.Y. Revealing Spatial and Temporal Patterns of Cell Death, Glial Proliferation, and Blood-Brain Barrier Dysfunction Around Implanted Intracortical Neural Interfaces. Front. Neurosci. 2019, 13, 493. [Google Scholar] [CrossRef]
- Bennett, C.; Mohammed, F.; Alvarez-Ciara, A.; Nguyen, M.A.; Dietrich, W.D.; Rajguru, S.M.; Streit, W.J.; Prasad, A. Neuroinflammation, oxidative stress, and blood-brain barrier (BBB) disruption in acute Utah electrode array implants and the effect of deferoxamine as an iron chelator on acute foreign body response. Biomaterials 2019, 188, 144–159. [Google Scholar] [CrossRef]
- Usoro, J.; Sturgill, B.; Musselman, K.; Capadona, J.R.; Pancrazio, J.J. On the definition of ‘chronic’ for intracortical microelectrode array applications. Micromachines 2021, 12, 972. [Google Scholar] [CrossRef]
- Usoro, J.O.; Dogra, K.; Abbott, J.R.; Radhakrishna, R.; Cogan, S.F.; Pancrazio, J.J.; Patnaik, S.S. Influence of Implantation Depth on the Performance of Intracortical Probe Recording Sites. Micromachines 2021, 12, 1158. [Google Scholar] [CrossRef]
- Jorfi, M.; Skousen, J.L.; Weder, C.; Capadona, J.R. Progress towards biocompatible intracortical microelectrodes for neural interfacing applications. J. Neural Eng. 2015, 12, 011001. [Google Scholar] [CrossRef]
- Ravikumar, M.; Sunil, S.; Black, J.; Barkauskas, D.; Haung, A.Y.; Miller, R.H.; Selkirk, S.M.; Capadona, J.R. The Roles of Blood-derived Macrophages and Resident Microglia in the Neuroinflammatory Response to Implanted Intracortical Microelectrodes. Biomaterials 2014, 35, 8049–8064. [Google Scholar] [CrossRef] [PubMed]
- Kozai, T.D.Y.; Marzullo, T.C.; Hooi, F.; Langhals, N.B.; Majewska, A.K.; Brown, E.B.; Kipke, D.R. Reduction of neurovascular damage resulting from microelectrode insertion into the cerebral cortex using in vivo two-photon mapping. J. Neural Eng. 2010, 7, 046011. [Google Scholar] [CrossRef] [PubMed]
- Readnower, R.D.; Chavko, M.; Adeeb, S.; Conroy, M.D.; Pauly, J.R.; McCarron, R.M.; Sullivan, P.G. Increase in blood-brain barrier permeability, oxidative stress, and activated microglia in a rat model of blast-induced traumatic brain injury. J. Neurosci. Res. 2010, 88, 3530–3539. [Google Scholar] [CrossRef] [PubMed]
- Alam, A.; Thelin, E.P.; Tajsic, T.; Khan, D.Z.; Khellaf, A.; Patani, R.; Helmy, A. Cellular infiltration in traumatic brain injury. J. Neuroinflammation 2020, 17, 328. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Qin, C.; Huang, J.; Tang, X.; Liu, C.; Huang, K.; Xu, J.; Guo, G.; Tong, A.; Zhou, L. The role of astrocytes in oxidative stress of central nervous system: A mixed blessing. Cell Prolif. 2020, 53, e12781. [Google Scholar] [CrossRef] [PubMed]
- Biswas, K.; Alexander, K.; Francis, M.M. Reactive Oxygen Species: Angels and Demons in the Life of a Neuron. NeuroSci 2022, 3, 11. [Google Scholar] [CrossRef]
- Guttenplan, K.A.; Stafford, B.K.; El-Danaf, R.N.; Adler, D.I.; Münch, A.E.; Weigel, M.K.; Huberman, A.D.; Liddelow, S.A. Neurotoxic Reactive Astrocytes Drive Neuronal Death after Retinal Injury. Cell Rep. 2020, 31, 107776. [Google Scholar] [CrossRef]
- Ereifej, E.S.; Rial, G.; Hermann, J.K.; Smith, C.S.; Meade, S.; Rayyan, J.; Chen, K.; Feng, H.; Capadona, J.R. Implantation of Neural Probes in the Brain Elicits Oxidative Stress. Front. Bioeng. Biotechnol. 2018, 6. under revision. [Google Scholar] [CrossRef]
- Harris, J.P.; Capadona, J.R.; Miller, R.H.; Healy, B.C.; Shanmuganathan, K.; Rowan, S.J.; Weder, C.; Tyler, D.J. Mechanically adaptive intracortical implants improve the proximity of neuronal cell bodies. J. Neural Eng. 2011, 8, 066011. [Google Scholar] [CrossRef]
- Nguyen, J.K.; Park, D.J.; Skousen, J.L.; Hess-Dunning, A.; Tyler, D.J.; Rowan, S.J.; Weder, C.; Capadona, J.R. Mechanically-compliant intracortical implants reduce the neuroinflammatory response. J. Neural Eng. 2014, 11, 056014. [Google Scholar] [CrossRef]
- Ware, T.; Simon, D.; Rennaker, R.L.; Voit, W. Smart Polymers for Neural Interfaces. Polym. Rev. 2013, 53, 108–129. [Google Scholar] [CrossRef]
- Hernandez-Reynoso, A.G.; Sturgill, B.; Hoeferlin, G.F.; Druschel, L.; Krebs, O.K.; Menendez, D.M.; Thai, T.T.D.; Smith, T.J.; Duncan, J.; Zhang, J.; et al. The Effect of MnTBAP Coatings on the Acute and Sub-Chronic Recording Performance of Planar Silicon Intracortical Microelectrode Arrays. Biomaterials 2023, 303, 122351. [Google Scholar] [CrossRef] [PubMed]
- Potter-Baker, K.A.; Nguyen, J.K.; Kovach, K.M.; Gitomer, M.M.; Srail, T.W.; Stewart, W.G.; Skousen, J.L.; Capadona, J.R. Development of Superoxide Dismutase Mimetic Surfaces to Reduce Accumulation of Reactive Oxygen Species Surrounding Intracortical Microelectrodes. J. Mater. Chem. B 2014, 2, 2248–2258. [Google Scholar] [CrossRef] [PubMed]
- Zheng, X.S.; Snyder, N.R.; Woeppel, K.; Barengo, J.H.; Li, X.; Eles, J.; Kolarcik, C.L.; Cui, X.T. A superoxide scavenging coating for improving tissue response to neural implants. Acta Biomater. 2019, 99, 72–83. [Google Scholar] [CrossRef] [PubMed]
- Chen, R.; Funnell, J.L.; Quinones, G.B.; Bentley, M.; Capadona, J.R.; Gilbert, R.J.; Palermo, E.F. Poly(pro-curcumin) Materials Exhibit Dual Release Rates and Prolonged Antioxidant Activity as Thin Films and Self-Assembled Particles. Biomacromolecules 2023, 24, 294–307. [Google Scholar] [CrossRef] [PubMed]
- Ziemba, A.M.; Woodson, M.C.C.; Funnell, J.L.; Wich, D.; Balouch, B.; Rende, D.; Amato, D.N.; Bao, J.; Oprea, I.; Cao, D.; et al. Development of a Slow-Degrading Polymerized Curcumin Coating for Intracortical Microelectrodes. ACS Appl. Bio. Mater. 2023, 6, 806–818. [Google Scholar] [CrossRef] [PubMed]
- Kim, N.M.Y.; Schwartzman, W.; Sarno, D.; Wynder, R.; Hoeferlin, G.F.; Laela, G.; Capadona, J.R.; Hess-Dunning, A. Fabrication Methods and Chronic In Vivo Validation of Mechanically Adaptive Microfluidic Intracortical Devices. Micromachines 2023, 14, 1015. [Google Scholar] [CrossRef] [PubMed]
- Capadona, J.R.; Shoffstall, A.J.; Pancrazio, J.J. Neuron-like neural probes. Nat. Mater. 2019, 1, 429–431. [Google Scholar] [CrossRef]
- Xie, C.; Liu, J.; Fu, T.-M.; Dai, X.; Zhou, W.; Lieber, C.M. Three-dimensional macroporous nanoelectronic networks as minimally invasive brain probes. Nat. Mater. 2015, 14, 1286–1292. [Google Scholar] [CrossRef]
- Seymour, J.P.; Kipke, D.R. Neural probe design for reduced tissue encapsulation in CNS. Biomaterials 2007, 28, 3594–3607. [Google Scholar] [CrossRef]
- Hong, G.; Lieber, C.M. Novel electrode technologies for neural recordings. Nat. Rev. 2019, 20, 330–345. [Google Scholar] [CrossRef] [PubMed]
- Shoffstall, A.; Capadona, J.R. Prospects for a robust cortical recording interface. In Neuromodulation; Academic Press: Cambridge, MA, USA, 2018; pp. 393–413. [Google Scholar]
- Lam, D.; Enright, H.A.; Cadena, J.; Peters, S.K.G.; Sales, A.P.; Osburn, J.J.; Soscia, D.A.; Kulp, K.S.; Wheeler, E.K.; Fischer, N.O. Tissue-specific extracellular matrix accelerates the formation of neural networks and communities in a neuron-glia co-culture on a multi-electrode array. Sci. Rep. 2019, 9, 4159. [Google Scholar] [CrossRef]
- Kriparamanan, R.; Aswath, P.; Zhou, A.; Tang, L.; Nguyen, K.T. Nanotopography: Cellular responses to nanostructured materials. J. Nanosci. Nanotechnol. 2006, 6, 1905–1919. [Google Scholar] [CrossRef]
- Ereifej, E.S.; Matthew, H.W.; Newaz, G.; Mukhopadhyay, A.; Auner, G.; Salakhutdinov, I.; VandeVord, P.J. Nanopatterning effects on astrocyte reactivity. J. Biomed. Mater. Res. A 2013, 101, 1743–1757. [Google Scholar] [CrossRef]
- Ereifej, E.S.; Smith, C.S.; Meade, S.M.; Chen, K.; Feng, H.; Capadona, J.R. The neuroinflammatory response to nanopatterning parallel grooves into the surface structure of intracortical microelectrodes. Adv. Funct. Mater. 2018, 28, 1704420. [Google Scholar] [CrossRef]
- Kim, Y.; Meade, S.M.; Chen, K.; Feng, H.; Rayyan, J.; Hess-Dunning, A.; Ereifej, E.S. Nano-Architectural Approaches for Improved Intracortical Interface Technologies. Front. Neurosci. 2018, 12, 456. [Google Scholar] [CrossRef]
- Selvakumaran, J.; Keddie, J.L.; Ewins, D.J.; Hughes, M.P. Protein adsorption on materials for recording sites on implantable microelectrodes. J. Mater. Sci. Mater. Med. 2008, 19, 143–151. [Google Scholar] [CrossRef]
- Ereifej, E.S.; Cheng, M.M.; Mao, G.; Van de Vord, P.J. Examining the inflammatory response to nanopatterned polydimethylsiloxane using organotypic brain slice methods. J. Neurosci. Methods 2013, 217, 17–25. [Google Scholar] [CrossRef]
- Bedell, H.W.; Hermann, J.K.; Ravikumar, M.; Lin, S.; Rein, A.; Li, X.; Molinich, E.; Smith, P.; Sidik, S.; Taylor, D.M.; et al. Targeting CD14 on blood derived cells improves intracortical microelectrode performance. Biomaterials 2018, 163, 163–173. [Google Scholar] [CrossRef] [PubMed]
- Bedell, H.W.; Song, S.; Li, X.; Molinich, E.; Lin, S.; Stiller, A.; Danda, V.; Ecker, M.; Shoffstall, A.J.; Voit, W.E.; et al. Understanding the Effects of Both CD14-Mediated Innate Immunity and Device/Tissue Mechanical Mismatch in the Neuroinflammatory Response to Intracortical Microelectrodes. Front. Neurosci. 2018, 12, 772. [Google Scholar] [CrossRef] [PubMed]
- Hermann, J.K.; Ravikumar, M.; Shoffstall, A.; Ereifej, E.; Kovach, K.; Chang, J.; Soffer, A.; Wong, C.; Srivastava, V.; Smith, P.; et al. Inhibition of the cluster of differentiation 14 innate immunity pathway with IAXO-101 improves chronic microelectrode performance. J. Neural Eng. 2017, 15, 025002. [Google Scholar] [CrossRef] [PubMed]
- Hermann, J.K.; Capadona, J.R. Understanding the Role of Innate Immunity in the Response to Intracortical Microelectrodes. Crit. Rev. Biomed. Eng. 2018, 46, 341–367. [Google Scholar] [CrossRef] [PubMed]
- Hermann, J.K.; Lin, S.; Soffer, A.; Wong, C.; Srivastava, V.; Chang, J.; Sunil, S.; Sudhakar, S.; Tomaszewski, W.H.; Protasiewicz, G.; et al. The Role of Toll-Like Receptor 2 and 4 Innate Immunity Pathways in Intracortical Microelectrode-Induced Neuroinflammation. Front. Bioeng. Biotechnol. 2018, 6, 113. [Google Scholar] [CrossRef]
- Hoeferlin, G.F.; Bajwa, T.; Olivares, H.; Zhang, J.; Druschel, L.N.; Sturgill, B.; Sobota, M.; Boucher, P.; Duncan, J.; Hernandez-Reynoso, A.G.; et al. Antioxidant Dimethyl Fumarate Temporarily but not Chronically Improves Microelectrode Performance. Micromachines 2023, 14, 1902. [Google Scholar] [CrossRef]
- Song, S.; Druschel, L.N.; Chan, R.; Capadona, J.R. Differential expression of genes involved in the chronic response to intracortical microelectrodes. Acta Biomater. 2023, 1, 348–362. [Google Scholar] [CrossRef]
- Song, S.; Druschel, L.N.; Conard, J.; Wang, J.; Kasthuri, N.M.; Chan, E.R.; Capadona, J.R. Differential expression of genes involved in the chronic response to intracortical microelectrodes implanted in complement factor 3 depleted mice. Brain Behav. Immun. 2024. under review. [Google Scholar]
- Sturgill, B.S.; Hernandez-Reynoso, A.G.; Druschel, L.; Smith, T.J.; Boucher, P.E.; Hoeferlin, G.F.; Thai, T.T.D.; Jiang, M.S.; Hess, J.; Alam, N.N.; et al. Reactive Amine Functionalized Microelectrode Arrays Provide Short-Term Benefit but Long-Term Detriment to In Vivo Recording Performance. ACS Appl. Bio Mater. 2024, accepted—in press. [Google Scholar] [CrossRef]
- Kim, L.D.Y.; Mueller, N.; Sarno, D.; Gisser, K.; Hess-Dunning, A.; Capadona, J.R. In Vivo Validation of a Mechanically Adaptive Microfluidic Intracortical Device as a Platform for Sustain Local Drug Delivery. Front. Biomater. Sci. 2023, 2, 1279367. [Google Scholar] [CrossRef]
- Song, S.; Regan, B.; Ereifej, E.S.; Chan, E.R.; Capadona, J.R. Neuroinflammatory Gene Expression Analysis Reveals Pathways of Interest as Potential Targets to Improve the Recording Performance of Intracortical Microelectrodes. Cells 2022, 11, 2348. [Google Scholar] [CrossRef] [PubMed]
- Bedell, H.W.; Schaub, N.J.; Capadona, J.R.; Ereifej, E.S. Differential expression of genes involved in the acute innate immune response to intracortical microelectrodes. Acta Biomater. 2020, 102, 205–219. [Google Scholar] [CrossRef] [PubMed]
- NIH. Sirt1 Sirtuin 1 [Rattus norvegicus (Norway Rat)]. Available online: https://www.ncbi.nlm.nih.gov/gene/309757/, https://www.frontiersin.org/articles/10.3389/fimmu.2022.831168/full; (accessed on 1 November 2023).
- Lim, J.H.; Lee, Y.M.; Chun, Y.S.; Chen, J.; Kim, J.E.; Park, J.W. Sirtuin 1 modulates cellular responses to hypoxia by deacetylating hypoxia-inducible factor 1alpha. Mol Cell 2010, 38, 864–878. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Liu, Y.; Wang, Y.; Chao, Y.; Zhang, J.; Jia, Y.; Tie, J.; Hu, D. Regulation of SIRT1 and Its Roles in Inflammation. Front. Immunol. 2022, 13, 831168. [Google Scholar] [CrossRef] [PubMed]
- Usoro, J.O.; Sturgill, B.S.; Musselman, K.C.; Capadona, J.R.; Pancrazio, J.J. Intracortical Microelectrode Array Unit Yield under Chronic Conditions: A Comparative Evaluation. Micromachines 2021, 12, 972. [Google Scholar] [CrossRef] [PubMed]
- Cogan, S.F. Neural stimulation and recording electrodes. Annu. Rev. Biomed. Eng. 2008, 10, 275–309. [Google Scholar] [CrossRef] [PubMed]
- Stiller, A.M.; Usoro, J.O.; Lawson, J.; Araya, B.; González-González, M.A.; Danda, V.R.; Voit, W.E.; Black, B.J.; Pancrazio, J.J. Mechanically Robust, Softening Shape Memory Polymer Probes for Intracortical Recording. Micromachines 2020, 11, 619. [Google Scholar] [CrossRef]
- Chapman, C.A.R.; Chen, H.; Stamou, M.; Biener, J.; Biener, M.M.; Lein, P.J.; Seker, E. Nanoporous Gold as a Neural Interface Coating: Effects of Topography, Surface Chemistry, and Feature Size. ACS Appl. Mater. Inter. 2015, 7, 7093–7100. [Google Scholar] [CrossRef] [PubMed]
- Simitzi, C.; Ranella, A.; Stratakis, E. Controlling the Morphology and Outgrowth of Nerve and Neuroglial Cells: The Effect of Surface Topography. Acta Biomater. 2017, 51, 21–52. [Google Scholar] [CrossRef] [PubMed]


| Custom Gene Panel | NanoString Preset Panel | Housekeeping Genes | |||||
|---|---|---|---|---|---|---|---|
| Aim2 | Gsta1 | Scd1 | Abl1 | Fas | Lpo | Sirt1 | Hprt |
| Arc | Gsta2 | Serpina3n | Ager | Fn1 | Lrrk2 | Sirt2 | Rpl13a |
| Bdnf | Gstm2 | Sod3 | Aif1 | Fos | Mapt | Slc8a1 | Rps18 |
| Blnk | Hmox1 | Spp1 | Akt1 | Fxn | Mgmt | Snca | Sdha |
| C3 | Il1b | Srxn1 | Apoe | Gnao1 | Mmp14 | Sod1 | Tbp |
| C3ar1 | Il2rg | Tnfrsf1a | App | Gpr37 | Mutyh | Sod2 | Ubc |
| C4a | Irak4 | Tnfrsf25 | Atf4 | Gsk3b | Ncf1 | Src | |
| C5ar1 | Irf7 | Txnrd1 | Atp13a2 | Gsr | Nefh | Stx2 | |
| Casp8 | Itgam | Tyrobp | Atp7a | Gss | Ngfg | Tnf | |
| Ccl1 | Keap1 | Vegfa | Atrn | Gstp1 | Ngfr | Tor1a | |
| Cd14 | Lilrb4a | Bad | Gucy1b3 | Nme5 | Tpm1 | ||
| Cd36 | Mmp12 | Bcl2 | H2-t23 | Nol3 | Trp53 | ||
| Cd45 | Mpeg1 | Bnip3 | Hdac2 | Nos1 | Trpm2 | ||
| Cd68 | Nfe2l2 | Casp3 | Hdac6 | Nos3 | Txnl1 | ||
| Cd74 | Noxa1 | Ccl5 | Hgf | Nr4a2 | Ubqln1 | ||
| Cd84 | Nqo1 | Ccs | Hif1a | Oxr1 | Xbp1 | ||
| Clec7a | Nr2f6 | Cdk2 | Hspb1 | Park7 | |||
| Ctss | Osgin1 | Cln8 | Htra2 | Parp1 | |||
| Dock2 | Osmr | Cybb | Idh1 | Pdgfrb | |||
| Ehd2 | Prnp | Cycs | Il1r1 | Pink1 | |||
| Ercc6 | Psmb8 | Ddit3 | Il6 | Pla2g4a | |||
| Fcer1g | Ptgs2 | Dnm2 | Ins2 | Ppargc1a | |||
| Fcgr2b | Ptpn6 | Ep300 | Ipcef1 | Psen1 | |||
| Gfap | Ptx3 | Erlec1 | Jun | Rela | |||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Duncan, J.L.; Wang, J.J.; Glusauskas, G.; Weagraff, G.R.; Gao, Y.; Hoeferlin, G.F.; Hunter, A.H.; Hess-Dunning, A.; Ereifej, E.S.; Capadona, J.R. In Vivo Characterization of Intracortical Probes with Focused Ion Beam-Etched Nanopatterned Topographies. Micromachines 2024, 15, 286. https://doi.org/10.3390/mi15020286
Duncan JL, Wang JJ, Glusauskas G, Weagraff GR, Gao Y, Hoeferlin GF, Hunter AH, Hess-Dunning A, Ereifej ES, Capadona JR. In Vivo Characterization of Intracortical Probes with Focused Ion Beam-Etched Nanopatterned Topographies. Micromachines. 2024; 15(2):286. https://doi.org/10.3390/mi15020286
Chicago/Turabian StyleDuncan, Jonathan L., Jaime J. Wang, Gabriele Glusauskas, Gwendolyn R. Weagraff, Yue Gao, George F. Hoeferlin, Allen H. Hunter, Allison Hess-Dunning, Evon S. Ereifej, and Jeffrey R. Capadona. 2024. "In Vivo Characterization of Intracortical Probes with Focused Ion Beam-Etched Nanopatterned Topographies" Micromachines 15, no. 2: 286. https://doi.org/10.3390/mi15020286
APA StyleDuncan, J. L., Wang, J. J., Glusauskas, G., Weagraff, G. R., Gao, Y., Hoeferlin, G. F., Hunter, A. H., Hess-Dunning, A., Ereifej, E. S., & Capadona, J. R. (2024). In Vivo Characterization of Intracortical Probes with Focused Ion Beam-Etched Nanopatterned Topographies. Micromachines, 15(2), 286. https://doi.org/10.3390/mi15020286






