Recent Advances in Nanomaterials Used for Wearable Electronics
Abstract
1. Introduction
2. Nanomaterial-Enabled Wearable Electronics
2.1. Flexible and Biocompatible Substrates
2.2. Soft Nanoconductors for Wearables
2.3. Nanomaterial-Based Transducers
3. Applications of Nanomaterial-Enabled Wearable Electronics
4. Outlook
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Bandyopadhyay, D.; Sen, J. Internet of Things: Applications and Challenges in Technology and Standardization. Wirel. Pers. Commun. 2011, 58, 49–69. [Google Scholar] [CrossRef]
- Wortmann, F.; Flüchter, K. Internet of Things. Bus. Inf. Syst. Eng. 2015, 57, 221–224. [Google Scholar] [CrossRef]
- Stoppa, M.; Chiolerio, A. Wearable Electronics and Smart Textiles: A Critical Review. Sensors 2014, 14, 11957–11992. [Google Scholar] [CrossRef] [PubMed]
- Cima, M.J. Next-Generation Wearable Electronics. Nat. Biotechnol. 2014, 32, 642–643. [Google Scholar] [CrossRef]
- Shi, J.; Liu, S.; Zhang, L.; Yang, B.; Shu, L.; Yang, Y.; Ren, M.; Wang, Y.; Chen, J.; Chen, W. Smart Textile-integrated Microelectronic Systems for Wearable Applications. Adv. Mater. 2020, 32, 1901958. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.; Qing, H.; Li, Z.; Han, Y.L.; Lin, M.; Yang, H.; Li, A.; Lu, T.J.; Li, F.; Xu, F. A Promising Material for Human-Friendly Functional Wearable Electronics. Mater. Sci. Eng. R Rep. 2017, 112, 1–22. [Google Scholar] [CrossRef]
- Lou, Z.; Li, L.; Wang, L.; Shen, G. Recent Progress of Self-powered Sensing Systems for Wearable Electronics. Small 2017, 13, 1701791. [Google Scholar] [CrossRef]
- Jayathilaka, W.A.D.M.; Qi, K.; Qin, Y.; Chinnappan, A.; Serrano-García, W.; Baskar, C.; Wang, H.; He, J.; Cui, S.; Thomas, S.W.; et al. Significance of Nanomaterials in Wearables: A Review on Wearable Actuators and Sensors. Adv. Mater. 2019, 31, 1805921. [Google Scholar] [CrossRef]
- Reeder, B.; David, A. Health at Hand: A Systematic Review of Smart Watch Uses for Health and Wellness. J. Biomed. Inform. 2016, 63, 269–276. [Google Scholar] [CrossRef]
- Lu, T.-C.; Fu, C.-M.; Ma, M.H.-M.; Fang, C.-C.; Turner, A.M. Healthcare Applications of Smart Watches. Appl. Clin. Inform. 2016, 7, 850–869. [Google Scholar] [CrossRef]
- Xie, J.; Wen, D.; Liang, L.; Jia, Y.; Gao, L.; Lei, J. Evaluating the Validity of Current Mainstream Wearable Devices in Fitness Tracking under Various Physical Activities: Comparative Study. JMIR Mhealth Uhealth 2018, 6, e94. [Google Scholar] [CrossRef]
- Kaewkannate, K.; Kim, S. A Comparison of Wearable Fitness Devices. BMC Public Health 2016, 16, 433. [Google Scholar] [CrossRef]
- Rauschnabel, P.A.; Ro, Y.K. Augmented Reality Smart Glasses: An Investigation of Technology Acceptance Drivers. Int. J. Technol. Mark. 2016, 11, 123–148. [Google Scholar] [CrossRef]
- Kim, H.; Kwon, Y.; Lim, H.; Kim, J.; Kim, Y.; Yeo, W. Recent Advances in Wearable Sensors and Integrated Functional Devices for Virtual and Augmented Reality Applications. Adv. Funct. Mater. 2021, 31, 2005692. [Google Scholar] [CrossRef]
- Yu, X.; Xie, Z.; Yu, Y.; Lee, J.; Vazquez-Guardado, A.; Luan, H.; Ruban, J.; Ning, X.; Akhtar, A.; Li, D. Skin-Integrated Wireless Haptic Interfaces for Virtual and Augmented Reality. Nature 2019, 575, 473–479. [Google Scholar] [CrossRef] [PubMed]
- Trung, T.Q.; Le, H.S.; Dang, T.M.L.; Ju, S.; Park, S.Y.; Lee, N. Freestanding, Fiber-based, Wearable Temperature Sensor with Tunable Thermal Index for Healthcare Monitoring. Adv. Healthc. Mater. 2018, 7, 1800074. [Google Scholar] [CrossRef] [PubMed]
- Xu, K.; Lu, Y.; Takei, K. Multifunctional Skin-inspired Flexible Sensor Systems for Wearable Electronics. Adv. Mater. Technol. 2019, 4, 1800628. [Google Scholar] [CrossRef]
- Kim, J.; Khan, S.; Wu, P.; Park, S.; Park, H.; Yu, C.; Kim, W. Self-Charging Wearables for Continuous Health Monitoring. Nano Energy 2021, 79, 105419. [Google Scholar] [CrossRef]
- Sang, M.; Kang, K.; Zhang, Y.; Zhang, H.; Kim, K.; Cho, M.; Shin, J.; Hong, J.; Kim, T.; Lee, S.K. Ultrahigh Sensitive Au-Doped Silicon Nanomembrane Based Wearable Sensor Arrays for Continuous Skin Temperature Monitoring with High Precision. Adv. Mater. 2022, 34, 2105865. [Google Scholar] [CrossRef]
- Ma, Z.; Xiang, X.; Shao, L.; Zhang, Y.; Gu, J. Multifunctional Wearable Silver Nanowire Decorated Leather Nanocomposites for Joule Heating, Electromagnetic Interference Shielding and Piezoresistive Sensing. Angew. Chem. Int. Ed. 2022, 61, e202200705. [Google Scholar] [CrossRef]
- Karim, N.; Afroj, S.; Tan, S.; Novoselov, K.S.; Yeates, S.G. All Inkjet-Printed Graphene-Silver Composite Ink on Textiles for Highly Conductive Wearable Electronics Applications. Sci. Rep. 2019, 9, 8035. [Google Scholar] [CrossRef]
- Liang, X.; Li, H.; Dou, J.; Wang, Q.; He, W.; Wang, C.; Li, D.; Lin, J.; Zhang, Y. Stable and Biocompatible Carbon Nanotube Ink Mediated by Silk Protein for Printed Electronics. Adv. Mater. 2020, 32, 2000165. [Google Scholar] [CrossRef]
- Shajari, S.; Mahmoodi, M.; Rajabian, M.; Karan, K.; Sundararaj, U.; Sudak, L.J. Highly Sensitive and Stretchable Carbon Nanotube/Fluoroelastomer Nanocomposite with a Double-Percolated Network for Wearable Electronics. Adv. Electron. Mater. 2020, 6, 1901067. [Google Scholar] [CrossRef]
- Won, P.; Jeong, S.; Majidi, C.; Ko, S.H. Recent Advances in Liquid-Metal-Based Wearable Electronics and Materials. Iscience 2021, 24, 102698. [Google Scholar] [CrossRef]
- Yu, Y.; Guo, J.; Ma, B.; Zhang, D.; Zhao, Y. Liquid Metal-Integrated Ultra-Elastic Conductive Microfibers from Microfluidics for Wearable Electronics. Sci. Bull. 2020, 65, 1752–1759. [Google Scholar] [CrossRef] [PubMed]
- Chow, P.C.; Someya, T. Organic Photodetectors for Next-generation Wearable Electronics. Adv. Mater. 2020, 32, 1902045. [Google Scholar] [CrossRef]
- Promphet, N.; Ummartyotin, S.; Ngeontae, W.; Puthongkham, P.; Rodthongkum, N. Non-Invasive Wearable Chemical Sensors in Real-Life Applications. Anal. Chim. Acta 2021, 1179, 338643. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Fu, X.; He, J.; Shi, X.; Chen, T.; Chen, P.; Wang, B.; Peng, H. Application Challenges in Fiber and Textile Electronics. Adv. Mater. 2020, 32, 1901971. [Google Scholar] [CrossRef]
- Zhou, B.; Liu, Z.; Li, C.; Liu, M.; Jiang, L.; Zhou, Y.; Zhou, F.; Chen, S.; Jerrams, S.; Yu, J. A Highly Stretchable and Sensitive Strain Sensor Based on Dopamine Modified Electrospun SEBS Fibers and MWCNTs with Carboxylation. Adv. Electron. Mater. 2021, 7, 2100233. [Google Scholar] [CrossRef]
- Wang, L.; Chen, Y.; Lin, L.; Wang, H.; Huang, X.; Xue, H.; Gao, J. Highly Stretchable, Anti-Corrosive and Wearable Strain Sensors Based on the PDMS/CNTs Decorated Elastomer Nanofiber Composite. J. Chem. Eng. 2019, 362, 89–98. [Google Scholar] [CrossRef]
- Chen, X.; Liu, H.; Zheng, Y.; Zhai, Y.; Liu, X.; Liu, C.; Mi, L.; Guo, Z.; Shen, C. Highly Compressible and Robust Polyimide/Carbon Nanotube Composite Aerogel for High-Performance Wearable Pressure Sensor. ACS Appl. Mater. Interfaces 2019, 11, 42594–42606. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.; Chen, X.; Zheng, Y.; Zhang, D.; Zhao, Y.; Wang, C.; Pan, C.; Liu, C.; Shen, C. Lightweight, Superelastic, and Hydrophobic Polyimide Nanofiber /MXene Composite Aerogel for Wearable Piezoresistive Sensor and Oil/Water Separation Applications. Adv. Funct. Mater. 2021, 31, 2008006. [Google Scholar] [CrossRef]
- Hirsch, A.; Dejace, L.; Michaud, H.O.; Lacour, S.P. Harnessing the Rheological Properties of Liquid Metals to Shape Soft Electronic Conductors for Wearable Applications. Acc. Chem. Res. 2019, 52, 534–544. [Google Scholar] [CrossRef] [PubMed]
- Hu, Y.; Parida, K.; Zhang, H.; Wang, X.; Li, Y.; Zhou, X.; Morris, S.A.; Liew, W.H.; Wang, H.; Li, T. Bond Engineering of Molecular Ferroelectrics Renders Soft and High-Performance Piezoelectric Energy Harvesting Materials. Nat. Commun. 2022, 13, 5607. [Google Scholar] [CrossRef]
- Kuzubasoglu, B.A.; Sayar, E.; Bahadir, S.K. Inkjet-Printed CNT/PEDOT: PSS Temperature Sensor on a Textile Substrate for Wearable Intelligent Systems. IEEE Sens. J. 2021, 21, 13090–13097. [Google Scholar] [CrossRef]
- Shin, J.; Liu, Z.; Bai, W.; Liu, Y.; Yan, Y.; Xue, Y.; Kandela, I.; Pezhouh, M.; MacEwan, M.R.; Huang, Y.; et al. Bioresorbable Optical Sensor Systems for Monitoring of Intracranial Pressure and Temperature. Sci. Adv. 2019, 5, eaaw1899. [Google Scholar] [CrossRef] [PubMed]
- Liao, C.-H.; Cheng, C.-T.; Chen, C.-C.; Jow, U.-M.; Chen, C.-H.; Lai, Y.-L.; Chen, Y.-C.; Ho, D.-R. An Ingestible Electronics for Continuous and Real-Time Intraabdominal Pressure Monitoring. J. Pers. Med. 2020, 11, 12. [Google Scholar] [CrossRef]
- Kim, J.; Park, J.; Park, Y.-G.; Cha, E.; Ku, M.; An, H.S.; Lee, K.-P.; Huh, M.-I.; Kim, J.; Kim, T.-S.; et al. A Soft and Transparent Contact Lens for the Wireless Quantitative Monitoring of Intraocular Pressure. Nat. Biomed. Eng. 2021, 5, 772–782. [Google Scholar] [CrossRef]
- Kang, S.-K.; Murphy, R.K.J.; Hwang, S.-W.; Lee, S.M.; Harburg, D.V.; Krueger, N.A.; Shin, J.; Gamble, P.; Cheng, H.; Yu, S.; et al. Bioresorbable Silicon Electronic Sensors for the Brain. Nature 2016, 530, 71–76. [Google Scholar] [CrossRef]
- Koo, J.; MacEwan, M.R.; Kang, S.-K.; Won, S.M.; Stephen, M.; Gamble, P.; Xie, Z.; Yan, Y.; Chen, Y.-Y.; Shin, J. Wireless Bioresorbable Electronic System Enables Sustained Nonpharmacological Neuroregenerative Therapy. Nat. Med. 2018, 24, 1830–1836. [Google Scholar] [CrossRef]
- Song, Y.-N.; Ma, R.-J.; Xu, L.; Huang, H.-D.; Yan, D.-X.; Xu, J.-Z.; Zhong, G.-J.; Lei, J.; Li, Z.-M. Wearable Polyethylene/Polyamide Composite Fabric for Passive Human Body Cooling. ACS Appl. Mater. Interfaces 2018, 10, 41637–41644. [Google Scholar] [CrossRef]
- Luo, J.; Gao, S.; Luo, H.; Wang, L.; Huang, X.; Guo, Z.; Lai, X.; Lin, L.; Li, R.K.Y.; Gao, J. Superhydrophobic and Breathable Smart MXene-Based Textile for Multifunctional Wearable Sensing Electronics. J. Chem. Eng. 2021, 406, 126898. [Google Scholar] [CrossRef]
- Yang, Y.; Huang, Q.; Niu, L.; Wang, D.; Yan, C.; She, Y.; Zheng, Z. Waterproof, Ultrahigh Areal-Capacitance, Wearable Supercapacitor Fabrics. Adv. Mater. 2017, 29, 1606679. [Google Scholar] [CrossRef] [PubMed]
- Guo, Y.; Chen, S.; Sun, L.; Yang, L.; Zhang, L.; Lou, J.; You, Z. Degradable and Fully Recyclable Dynamic Thermoset Elastomer for 3D-Printed Wearable Electronics. Adv. Funct. Mater. 2021, 31, 2009799. [Google Scholar] [CrossRef]
- Li, Z.; Zhang, S.; Chen, Y.; Ling, H.; Zhao, L.; Luo, G.; Wang, X.; Hartel, M.C.; Liu, H.; Xue, Y.; et al. Gelatin Methacryloyl-Based Tactile Sensors for Medical Wearables. Adv. Funct. Mater. 2020, 30, 2003601. [Google Scholar] [CrossRef]
- Yan, Y.; Potts, M.; Jiang, Z.; Sencadas, V. Synthesis of Highly-Stretchable Graphene—Poly(Glycerol Sebacate) Elastomeric Nanocomposites Piezoresistive Sensors for Human Motion Detection Applications. Compos. Sci. Technol. 2018, 162, 14–22. [Google Scholar] [CrossRef]
- Xu, Y.; Sun, B.; Ling, Y.; Fei, Q.; Chen, Z.; Li, X.; Guo, P.; Jeon, N.; Goswami, S.; Liao, Y.; et al. Multiscale Porous Elastomer Substrates for Multifunctional On-Skin Electronics with Passive-Cooling Capabilities. Proc. Natl. Acad. Sci. USA 2020, 117, 205–213. [Google Scholar] [CrossRef]
- Dong, J.; Wang, D.; Peng, Y.; Zhang, C.; Lai, F.; He, G.; Ma, P.; Dong, W.; Huang, Y.; Parkin, I.P.; et al. Ultra-Stretchable and Superhydrophobic Textile-Based Bioelectrodes for Robust Self-Cleaning and Personal Health Monitoring. Nano Energy 2022, 97, 107160. [Google Scholar] [CrossRef]
- Hsu, P.-C.; Song, A.Y.; Catrysse, P.B.; Liu, C.; Peng, Y.; Xie, J.; Fan, S.; Cui, Y. Radiative Human Body Cooling by Nanoporous Polyethylene Textile. Science 2016, 353, 1019–1023. [Google Scholar] [CrossRef]
- Dan, L.; Shi, S.; Chung, H.-J.; Elias, A. Porous Polydimethylsiloxane–Silver Nanowire Devices for Wearable Pressure Sensors. ACS Appl. Nano Mater. 2019, 2, 4869–4878. [Google Scholar] [CrossRef]
- Zhai, Y.; Yu, Y.; Zhou, K.; Yun, Z.; Huang, W.; Liu, H.; Xia, Q.; Dai, K.; Zheng, G.; Liu, C.; et al. Flexible and Wearable Carbon Black/Thermoplastic Polyurethane Foam with a Pinnate-Veined Aligned Porous Structure for Multifunctional Piezoresistive Sensors. J. Chem. Eng. 2020, 382, 122985. [Google Scholar] [CrossRef]
- Liu, Q.; Huang, J.; Zhang, J.; Hong, Y.; Wan, Y.; Wang, Q.; Gong, M.; Wu, Z.; Guo, C.F. Thermal, Waterproof, Breathable, and Antibacterial Cloth with a Nanoporous Structure. ACS Appl. Mater. Interfaces 2018, 10, 2026–2032. [Google Scholar] [CrossRef]
- Chen, Z.; Wang, M.; Zhang, C.; Wei, Z.; Wang, Y.; Gao, C.; Zhu, J.; Gao, J.; Shen, M.; Gao, Q. A Breathable and Reliable Thermoplastic Polyurethane/Ag@K2Ti4O9 Composite Film with an Asymmetrical Porous Structure for Wearable Piezoresistive Sensors. J. Mater. Chem. C 2022, 10, 12986–12997. [Google Scholar] [CrossRef]
- Jeong, W.; Lee, S.; Choi, H.; Bae, J.; Lee, S.-H.; Ma, Y.; Yoo, S.; Ha, J.-H.; Hong, J.-I.; Park, S.; et al. Washable, Stretchable, and Reusable Core–Shell Metal Nanowire Network-Based Electronics on a Breathable Polymer Nanomesh Substrate. Mater. Today 2022, 61, 30–39. [Google Scholar] [CrossRef]
- Qiao, Y.; Li, X.; Wang, J.; Ji, S.; Hirtz, T.; Tian, H.; Jian, J.; Cui, T.; Dong, Y.; Xu, X.; et al. Intelligent and Multifunctional Graphene Nanomesh Electronic Skin with High Comfort. Small 2022, 18, 2104810. [Google Scholar] [CrossRef] [PubMed]
- Wu, C.; Kim, T.W.; Li, F.; Guo, T. Wearable Electricity Generators Fabricated Utilizing Transparent Electronic Textiles Based on Polyester/Ag Nanowires/Graphene Core–Shell Nanocomposites. ACS Nano 2016, 10, 6449–6457. [Google Scholar] [CrossRef]
- Kwon, J.; Suh, Y.D.; Lee, J.; Lee, P.; Han, S.; Hong, S.; Yeo, J.; Lee, H.; Ko, S.H. Recent Progress in Silver Nanowire Based Flexible/Wearable Optoelectronics. J. Mater. Chem. C 2018, 6, 7445–7461. [Google Scholar] [CrossRef]
- Jiang, Z.; Nayeem, M.O.G.; Fukuda, K.; Ding, S.; Jin, H.; Yokota, T.; Inoue, D.; Hashizume, D.; Someya, T. Highly Stretchable Metallic Nanowire Networks Reinforced by the Underlying Randomly Distributed Elastic Polymer Nanofibers via Interfacial Adhesion Improvement. Adv. Mater. 2019, 31, 1903446. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.; Kim, S.Y.; Chung, M.H.; Kim, J.; Kim, J.H. A One-Step Roll-to-Roll Process of Stable AgNW/PEDOT:PSS Solution Using Imidazole as a Mild Base for Highly Conductive and Transparent Films: Optimizations and Mechanisms. J. Mater. Chem. C 2015, 3, 5859–5868. [Google Scholar] [CrossRef]
- Sultana, N.; Chang, H.C.; Jefferson, S.; Daniels, D.E. Application of Conductive Poly(3,4-Ethylenedioxythiophene):Poly(Styrenesulfonate) (PEDOT:PSS) Polymers in Potential Biomedical Engineering. J. Pharm. Investig. 2020, 50, 437–444. [Google Scholar] [CrossRef]
- Lee, S.J.; Kim, Y.-H.; Kim, J.K.; Baik, H.; Park, J.H.; Lee, J.; Nam, J.; Park, J.H.; Lee, T.-W.; Yi, G.-R.; et al. A Roll-to-Roll Welding Process for Planarized Silver Nanowire Electrodes. Nanoscale 2014, 6, 11828–11834. [Google Scholar] [CrossRef]
- Hu, L.; Kim, H.S.; Lee, J.-Y.; Peumans, P.; Cui, Y. Scalable Coating and Properties of Transparent, Flexible, Silver Nanowire Electrodes. ACS Nano 2010, 4, 2955–2963. [Google Scholar] [CrossRef] [PubMed]
- Geim, A.K.; Novoselov, K.S. The Rise of Graphene. Nat. Mater. 2007, 6, 183–191. [Google Scholar] [CrossRef] [PubMed]
- Bunch, J.S.; van der Zande, A.M.; Verbridge, S.S.; Frank, I.W.; Tanenbaum, D.M.; Parpia, J.M.; Craighead, H.G.; McEuen, P.L. Electromechanical Resonators from Graphene Sheets. Science 2007, 315, 490–493. [Google Scholar] [CrossRef]
- Li, D.; Kaner, R.B. Graphene-Based Materials. Science 2008, 320, 1170–1171. [Google Scholar] [CrossRef]
- Huang, L.; Huang, Y.; Liang, J.; Wan, X.; Chen, Y. Graphene-Based Conducting Inks for Direct Inkjet Printing of Flexible Conductive Patterns and Their Applications in Electric Circuits and Chemical Sensors. Nano Res. 2011, 4, 675–684. [Google Scholar] [CrossRef]
- Cummins, G.; Desmulliez, M.P.Y. Inkjet Printing of Conductive Materials: A Review. Circuit World 2012, 38, 193–213. [Google Scholar] [CrossRef]
- Song, P.; Song, J.; Zhang, Y. Stretchable Conductor Based on Carbon Nanotube/Carbon Black Silicone Rubber Nanocomposites with Highly Mechanical, Electrical Properties and Strain Sensitivity. Compos. B Eng. 2020, 191, 107979. [Google Scholar] [CrossRef]
- Yu, Y.; Yan, C.; Zheng, Z. Polymer-Assisted Metal Deposition (PAMD): A Full-Solution Strategy for Flexible, Stretchable, Compressible, and Wearable Metal Conductors. Adv. Mater. 2014, 26, 5508–5516. [Google Scholar] [CrossRef] [PubMed]
- Yoon, S.; Kim, Y.J.; Lee, Y.R.; Lee, N.-E.; Won, Y.; Gandla, S.; Kim, S.; Kim, H.-K. Highly Stretchable Metal-Polymer Hybrid Conductors for Wearable and Self-Cleaning Sensors. NPG Asia Mater. 2021, 13, 4. [Google Scholar] [CrossRef]
- Miyamoto, A.; Lee, S.; Cooray, N.F.; Lee, S.; Mori, M.; Matsuhisa, N.; Jin, H.; Yoda, L.; Yokota, T.; Itoh, A.; et al. Inflammation-Free, Gas-Permeable, Lightweight, Stretchable on-Skin Electronics with Nanomeshes. Nat. Nanotechnol. 2017, 12, 907–913. [Google Scholar] [CrossRef]
- Malakooti, M.H.; Kazem, N.; Yan, J.; Pan, C.; Markvicka, E.J.; Matyjaszewski, K.; Majidi, C. Liquid Metal Supercooling for Low-Temperature Thermoelectric Wearables. Adv. Funct. Mater. 2019, 29, 1906098. [Google Scholar] [CrossRef]
- Xu, Y.; Su, Y.; Xu, X.; Arends, B.; Zhao, G.; Ackerman, D.N.; Huang, H.; Reid, S.P.; Santarpia, J.L.; Kim, C.; et al. Porous Liquid Metal–Elastomer Composites with High Leakage Resistance and Antimicrobial Property for Skin-Interfaced Bioelectronics. Sci. Adv. 2023, 9, eadf0575. [Google Scholar] [CrossRef]
- Chen, S.; Wang, H.-Z.; Zhao, R.-Q.; Rao, W.; Liu, J. Liquid Metal Composites. Matter 2020, 2, 1446–1480. [Google Scholar] [CrossRef]
- Yang, Y.; Sun, N.; Wen, Z.; Cheng, P.; Zheng, H.; Shao, H.; Xia, Y.; Chen, C.; Lan, H.; Xie, X.; et al. Liquid-Metal-Based Super-Stretchable and Structure-Designable Triboelectric Nanogenerator for Wearable Electronics. ACS Nano 2018, 12, 2027–2034. [Google Scholar] [CrossRef]
- Wang, B.; Thukral, A.; Xie, Z.; Liu, L.; Zhang, X.; Huang, W.; Yu, X.; Yu, C.; Marks, T.J.; Facchetti, A. Flexible and Stretchable Metal Oxide Nanofiber Networks for Multimodal and Monolithically Integrated Wearable Electronics. Nat. Commun. 2020, 11, 2405. [Google Scholar] [CrossRef] [PubMed]
- Fu, W.; Zhao, E.; Ren, X.; Magasinski, A.; Yushin, G. Hierarchical Fabric Decorated with Carbon Nanowire/Metal Oxide Nanocomposites for 1.6 V Wearable Aqueous Supercapacitors. Adv. Energy Mater. 2018, 8, 1703454. [Google Scholar] [CrossRef]
- Zhao, G.; Ling, Y.; Su, Y.; Chen, Z.; Mathai, C.J.; Emeje, O.; Brown, A.; Alla, D.R.; Huang, J.; Kim, C.; et al. Laser-Scribed Conductive, Photoactive Transition Metal Oxide on Soft Elastomers for Janus on-Skin Electronics and Soft Actuators. Sci. Adv. 2022, 8, eabp9734. [Google Scholar] [CrossRef] [PubMed]
- Gao, Y.; Li, Q.; Wu, R.; Sha, J.; Lu, Y.; Xuan, F. Laser Direct Writing of Ultrahigh Sensitive SiC-Based Strain Sensor Arrays on Elastomer toward Electronic Skins. Adv. Funct. Mater. 2019, 29, 1806786. [Google Scholar] [CrossRef]
- Nair, V.; Yi, J.; Isheim, D.; Rotenberg, M.; Meng, L.; Shi, F.; Chen, X.; Gao, X.; Prominski, A.; Jiang, Y.; et al. Laser Writing of Nitrogen-Doped Silicon Carbide for Biological Modulation. Sci. Adv. 2020, 6, eaaz2743. [Google Scholar] [CrossRef]
- Ates, H.C.; Nguyen, P.Q.; Gonzalez-Macia, L.; Morales-Narváez, E.; Güder, F.; Collins, J.J.; Dincer, C. End-to-End Design of Wearable Sensors. Nat. Rev. Mater. 2022, 7, 887–907. [Google Scholar] [CrossRef]
- Shin, J.; Jeong, B.; Kim, J.; Nam, V.B.; Yoon, Y.; Jung, J.; Hong, S.; Lee, H.; Eom, H.; Yeo, J.; et al. Sensitive Wearable Temperature Sensor with Seamless Monolithic Integration. Adv. Mater. 2020, 32, 1905527. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.; Wei, D.; Tang, L.; Song, X.; Luo, W.; Chu, J.; Gao, T.; Shi, H.; Du, C. Wearable Temperature Sensor Based on Graphene Nanowalls. RSC Adv. 2015, 5, 25609–25615. [Google Scholar] [CrossRef]
- Amjadi, M.; Kyung, K.-U.; Park, I.; Sitti, M. Stretchable, Skin-Mountable, and Wearable Strain Sensors and Their Potential Applications: A Review. Adv. Funct. Mater. 2016, 26, 1678–1698. [Google Scholar] [CrossRef]
- Ding, Y.; Xu, T.; Onyilagha, O.; Fong, H.; Zhu, Z. Recent Advances in Flexible and Wearable Pressure Sensors Based on Piezoresistive 3D Monolithic Conductive Sponges. ACS Appl. Mater. Interfaces 2019, 11, 6685–6704. [Google Scholar] [CrossRef]
- Zhang, Q.; Yang, G.; Xue, L.; Dong, G.; Su, W.; Cui, M.J.; Wang, Z.G.; Liu, M.; Zhou, Z.; Zhang, X. Ultrasoft and Biocompatible Magnetic-Hydrogel-Based Strain Sensors for Wireless Passive Biomechanical Monitoring. ACS Nano 2022, 16, 21555–21564. [Google Scholar] [CrossRef]
- Kim, J.-S.; So, Y.; Lee, S.; Pang, C.; Park, W.; Chun, S. Uniform Pressure Responses for Nanomaterials-Based Biological on-Skin Flexible Pressure Sensor Array. Carbon 2021, 181, 169–176. [Google Scholar] [CrossRef]
- Liu, Z.; Yin, Z.; Jiang, Y.; Zheng, Q. Dielectric Interface Passivation of Polyelectrolyte-Gated Organic Field-Effect Transistors for Ultrasensitive Low-Voltage Pressure Sensors in Wearable Applications. Mater. Today Electron. 2022, 1, 100001. [Google Scholar] [CrossRef]
- Sun, F.; Lu, Q.; Hao, M.; Wu, Y.; Li, Y.; Liu, L.; Li, L.; Wang, Y.; Zhang, T. An Artificial Neuromorphic Somatosensory System with Spatio-Temporal Tactile Perception and Feedback Functions. npj Flex. Electron. 2022, 6, 72. [Google Scholar] [CrossRef]
- Roy, S.; David-Pur, M.; Hanein, Y. Carbon Nanotube-Based Ion Selective Sensors for Wearable Applications. ACS Appl. Mater. Interfaces 2017, 9, 35169–35177. [Google Scholar] [CrossRef]
- Pirovano, P.; Dorrian, M.; Shinde, A.; Donohoe, A.; Brady, A.J.; Moyna, N.M.; Wallace, G.; Diamond, D.; McCaul, M. A Wearable Sensor for the Detection of Sodium and Potassium in Human Sweat during Exercise. Talanta 2020, 219, 121145. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.; Campbell, A.S.; Wang, J. Wearable Non-Invasive Epidermal Glucose Sensors: A Review. Talanta 2018, 177, 163–170. [Google Scholar] [CrossRef]
- Mondal, S.; Karuppuswami, S.; Steinhorst, R.; Chahal, P. A Wearable Passive PH Sensor for Health Monitoring. In Proceedings of the 2019 IEEE 69th Electronic Components and Technology Conference (ECTC), Las Vegas, NV, USA, 28–31 May 2019; IEEE: New York, NY, USA, 2019; pp. 1240–1245. [Google Scholar] [CrossRef]
- Nakata, S.; Shiomi, M.; Fujita, Y.; Arie, T.; Akita, S.; Takei, K. A Wearable PH Sensor with High Sensitivity Based on a Flexible Charge-Coupled Device. Nat. Electron. 2018, 1, 596–603. [Google Scholar] [CrossRef]
- Anastasova, S.; Crewther, B.; Bembnowicz, P.; Curto, V.; Ip, H.M.; Rosa, B.; Yang, G.-Z. A Wearable Multisensing Patch for Continuous Sweat Monitoring. Biosens. Bioelectron. 2017, 93, 139–145. [Google Scholar] [CrossRef]
- Jiang, C.; Li, X.; Yao, Y.; Ying, Y.; Ping, J. Fully Written Flexible Potentiometric Sensor Using Two-Dimensional Nanomaterial-Based Conductive Ink. Anal. Chem. 2018, 90, 13088–13095. [Google Scholar] [CrossRef]
- Frank, S.M.; Tran, K.M.; Fleisher, L.A.; Elrahmany, H.K. Clinical Importance of Body Temperature in the Surgical Patient. J. Therm. Biol. 2000, 25, 151–155. [Google Scholar] [CrossRef]
- Turkani, V.S.; Maddipatla, D.; Narakathu, B.B.; Bazuin, B.J.; Atashbar, M.Z. A Carbon Nanotube Based NTC Thermistor Using Additive Print Manufacturing Processes. Sens. Actuators A Phys. 2018, 279, 1–9. [Google Scholar] [CrossRef]
- Occhiuzzi, C.; Cippitelli, S.; Marrocco, G. Modeling, Design and Experimentation of Wearable RFID Sensor Tag. IEEE Trans. Antennas Propag. 2010, 58, 2490–2498. [Google Scholar] [CrossRef]
- Hong, H.; Hu, J.; Yan, X. UV Curable Conductive Ink for the Fabrication of Textile-Based Conductive Circuits and Wearable UHF RFID Tags. ACS Appl. Mater. Interfaces 2019, 11, 27318–27326. [Google Scholar] [CrossRef]
- Heo, J.S.; Eom, J.; Kim, Y.-H.; Park, S.K. Recent Progress of Textile-Based Wearable Electronics: A Comprehensive Review of Materials, Devices, and Applications. Small 2018, 14, 1703034. [Google Scholar] [CrossRef] [PubMed]
- Koo, I.M.; Jung, K.; Koo, J.C.; Nam, J.-D.; Lee, Y.K.; Choi, H.R. Development of Soft-Actuator-Based Wearable Tactile Display. IEEE Trans. Robot. 2008, 24, 549–558. [Google Scholar] [CrossRef]
- Yin, D.; Chen, Z.; Jiang, N.; Liu, Y.; Bi, Y.; Zhang, X.; Han, W.; Feng, J.; Sun, H. Highly Flexible Fabric-Based Organic Light-Emitting Devices for Conformal Wearable Displays. Adv. Mater. Technol. 2020, 5, 1900942. [Google Scholar] [CrossRef]
- Basar, R.; Ahmad, M.Y.; Cho, J.; Ibrahim, F. An Improved Wearable Resonant Wireless Power Transfer System for Biomedical Capsule Endoscope. IEEE Trans. Ind. Electron. 2018, 65, 7772–7781. [Google Scholar] [CrossRef]
- Heo, E.; Choi, K.-Y.; Kim, J.; Park, J.-H.; Lee, H. A Wearable Textile Antenna for Wireless Power Transfer by Magnetic Resonance. Text. Res. J. 2018, 88, 913–921. [Google Scholar] [CrossRef]
- Barron, E.J.; Peterson, R.S.; Lazarus, N.; Bartlett, M.D. Mechanically Cloaked Multiphase Magnetic Elastomer Soft Composites for Wearable Wireless Power Transfer. ACS Appl. Mater. Interfaces 2020, 12, 50909–50917. [Google Scholar] [CrossRef] [PubMed]
- Long, Y.; Wei, H.; Li, J.; Yao, G.; Yu, B.; Ni, D.; Gibson, A.L.; Lan, X.; Jiang, Y.; Cai, W.; et al. Effective Wound Healing Enabled by Discrete Alternative Electric Fields from Wearable Nanogenerators. ACS Nano 2018, 12, 12533–12540. [Google Scholar] [CrossRef]
- Ciui, B.; Martin, A.; Mishra, R.K.; Brunetti, B.; Nakagawa, T.; Dawkins, T.J.; Lyu, M.; Cristea, C.; Sandulescu, R.; Wang, J. Wearable Wireless Tyrosinase Bandage and Microneedle Sensors: Toward Melanoma Screening. Adv. Healthc. Mater. 2018, 7, 1701264. [Google Scholar] [CrossRef]
- Weinstein, R. RFID: A Technical Overview and Its Application to the Enterprise. IT Prof. 2005, 7, 27–33. [Google Scholar] [CrossRef]
- Sanchez-Romaguera, V.; Ziai, M.A.; Oyeka, D.; Barbosa, S.; Wheeler, J.S.R.; Batchelor, J.C.; Parker, E.A.; Yeates, S.G. Towards Inkjet-Printed Low Cost Passive UHF RFID Skin Mounted Tattoo Paper Tags Based on Silver Nanoparticle Inks. J. Mater. Chem. C 2013, 1, 6395–6402. [Google Scholar] [CrossRef]
- Kawamoto, H. The History of Liquid-Crystal Displays. Proc. IEEE 2002, 90, 460–500. [Google Scholar] [CrossRef]
- Anandan, M. Progress of LED Backlights for LCDs. J. Soc. Inf. Disp. 2008, 16, 287. [Google Scholar] [CrossRef]
- Jawad, A.M.; Nordin, R.; Gharghan, S.K.; Jawad, H.M.; Ismail, M. Opportunities and Challenges for Near-Field Wireless Power Transfer: A Review. Energies 2017, 10, 1022. [Google Scholar] [CrossRef]
- Bahk, J.-H.; Fang, H.; Yazawa, K.; Shakouri, A. Flexible Thermoelectric Materials and Device Optimization for Wearable Energy Harvesting. J. Mater. Chem. C 2015, 3, 10362–10374. [Google Scholar] [CrossRef]
- Lou, Z.; Wang, L.; Jiang, K.; Wei, Z.; Shen, G. Reviews of Wearable Healthcare Systems: Materials, Devices and System Integration. Mater. Sci. Eng. R Rep. 2020, 140, 100523. [Google Scholar] [CrossRef]
- Lee, H.; Song, C.; Hong, Y.S.; Kim, M.S.; Cho, H.R.; Kang, T.; Shin, K.; Choi, S.H.; Hyeon, T.; Kim, D.-H. Wearable/Disposable Sweat-Based Glucose Monitoring Device with Multistage Transdermal Drug Delivery Module. Sci. Adv. 2017, 3, e1601314. [Google Scholar] [CrossRef] [PubMed]
- Choi, Y.S.; Yin, R.T.; Pfenniger, A.; Koo, J.; Avila, R.; Benjamin Lee, K.; Chen, S.W.; Lee, G.; Li, G.; Qiao, Y.; et al. Fully Implantable and Bioresorbable Cardiac Pacemakers without Leads or Batteries. Nat. Biotechnol. 2021, 39, 1228–1238. [Google Scholar] [CrossRef] [PubMed]
- Dong, B.; Yang, Y.; Shi, Q.; Xu, S.; Sun, Z.; Zhu, S.; Zhang, Z.; Kwong, D.-L.; Zhou, G.; Ang, K.-W.; et al. Wearable Triboelectric–Human–Machine Interface (THMI) Using Robust Nanophotonic Readout. ACS Nano 2020, 14, 8915–8930. [Google Scholar] [CrossRef]
- Ha, M.; Lim, S.; Ko, H. Wearable and Flexible Sensors for User-Interactive Health-Monitoring Devices. J. Mater. Chem. B 2018, 6, 4043–4064. [Google Scholar] [CrossRef]
- Yang, M.; Ye, Z.; Alsaab, N.; Farhat, M.; Chen, P.-Y. In-Vitro Demonstration of Ultra-Reliable, Wireless and Batteryless Implanted Intracranial Sensors Operated on Loci of Exceptional Points. IEEE Trans. Biomed. Circuits Syst. 2022, 16, 287–295. [Google Scholar] [CrossRef]
- Ye, Z.; Ling, Y.; Yang, M.; Xu, Y.; Zhu, L.; Yan, Z.; Chen, P.-Y. A Breathable, Reusable, and Zero-Power Smart Face Mask for Wireless Cough and Mask-Wearing Monitoring. ACS Nano 2022, 16, 5874–5884. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.; Campbell, A.S.; de Ávila, B.E.-F.; Wang, J. Wearable Biosensors for Healthcare Monitoring. Nat. Biotechnol. 2019, 37, 389–406. [Google Scholar] [CrossRef] [PubMed]
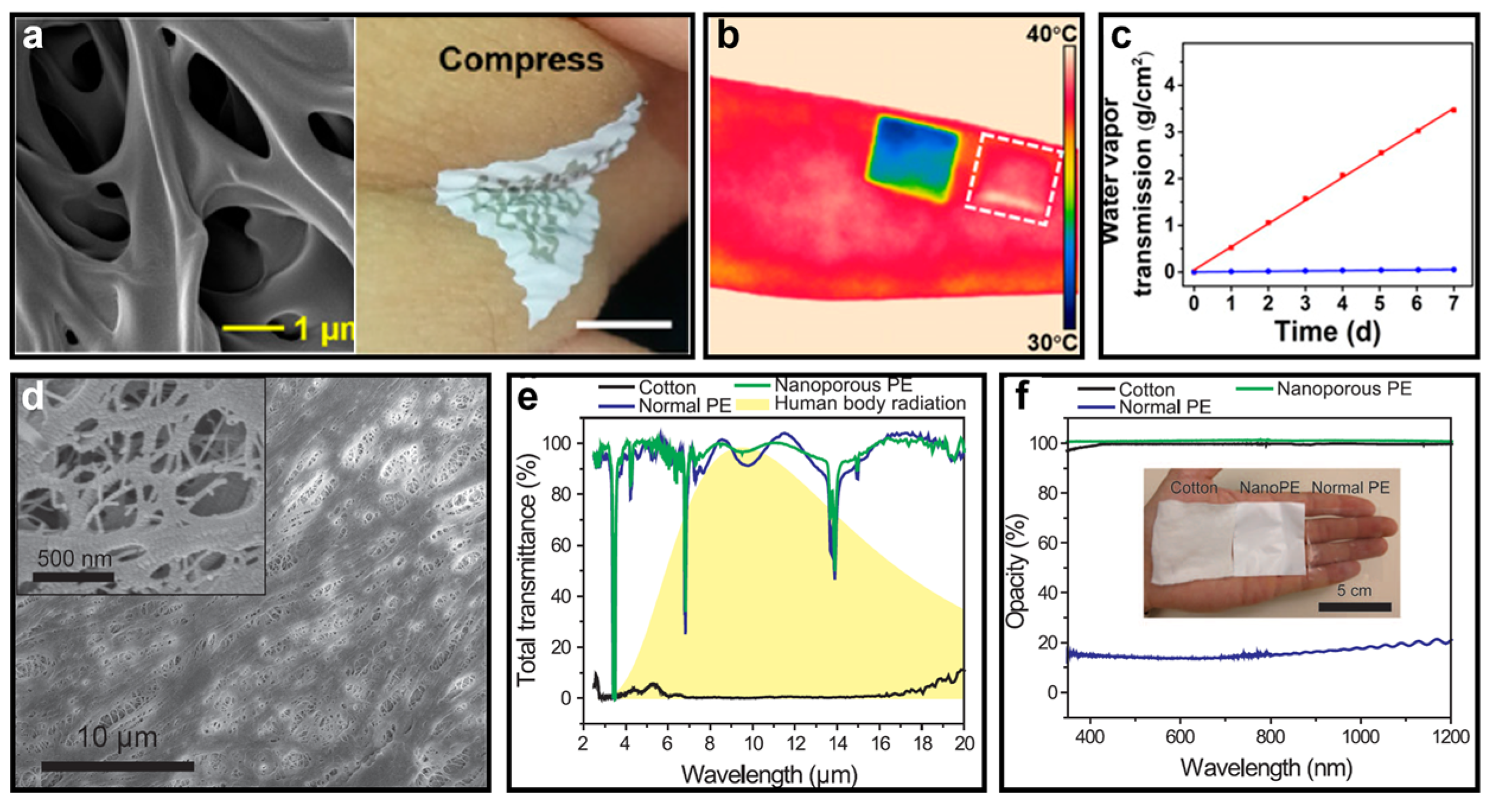

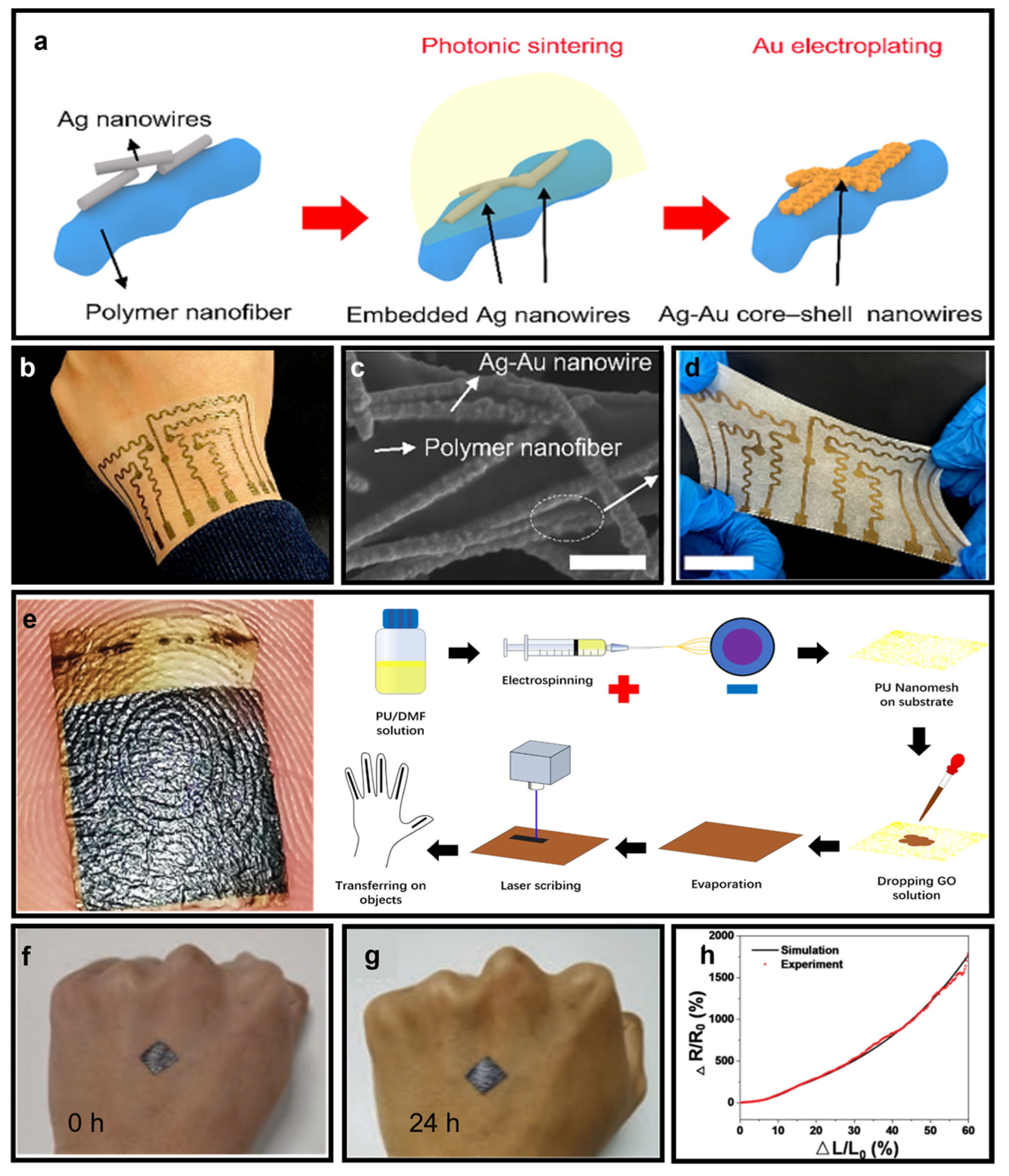
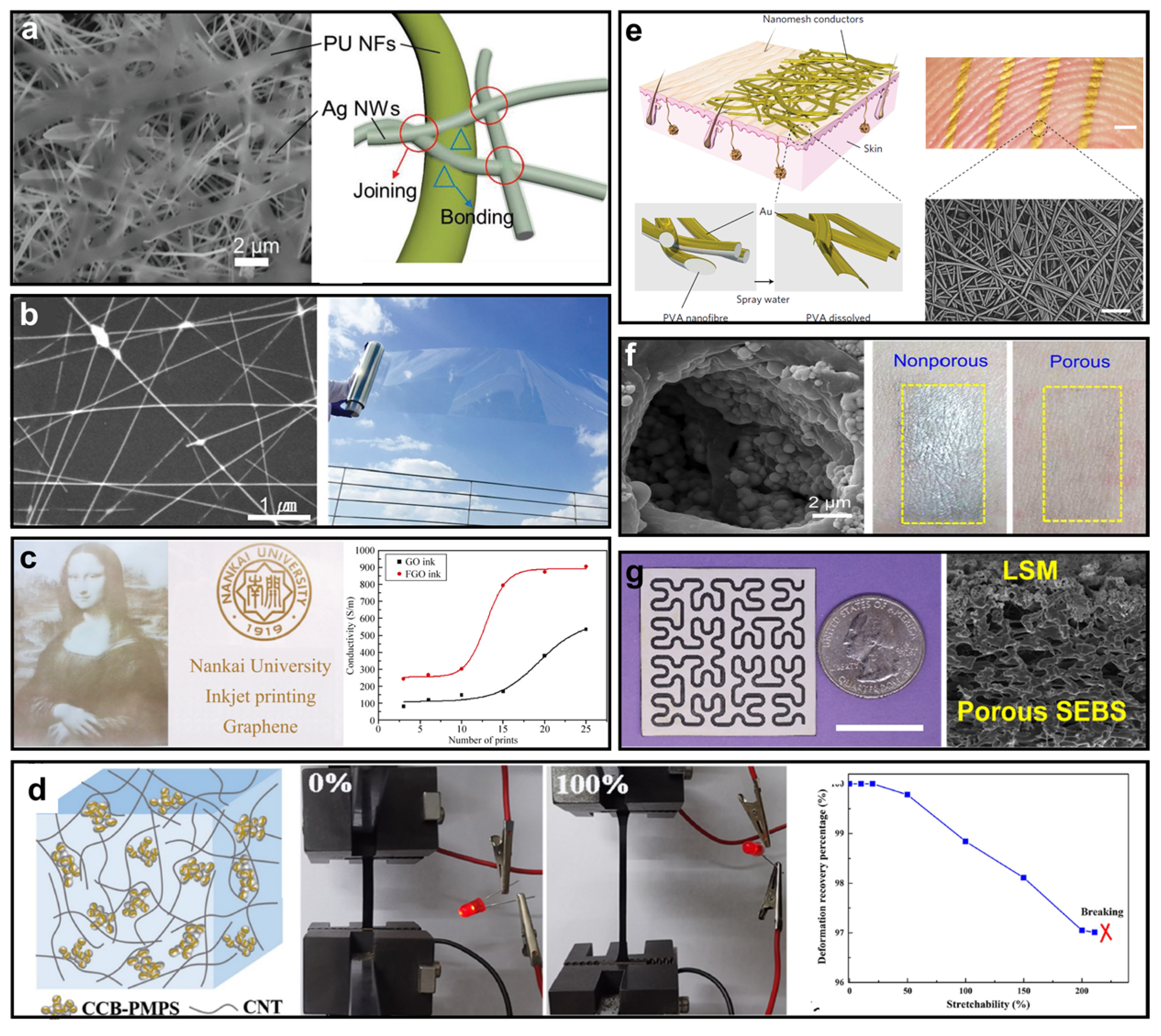

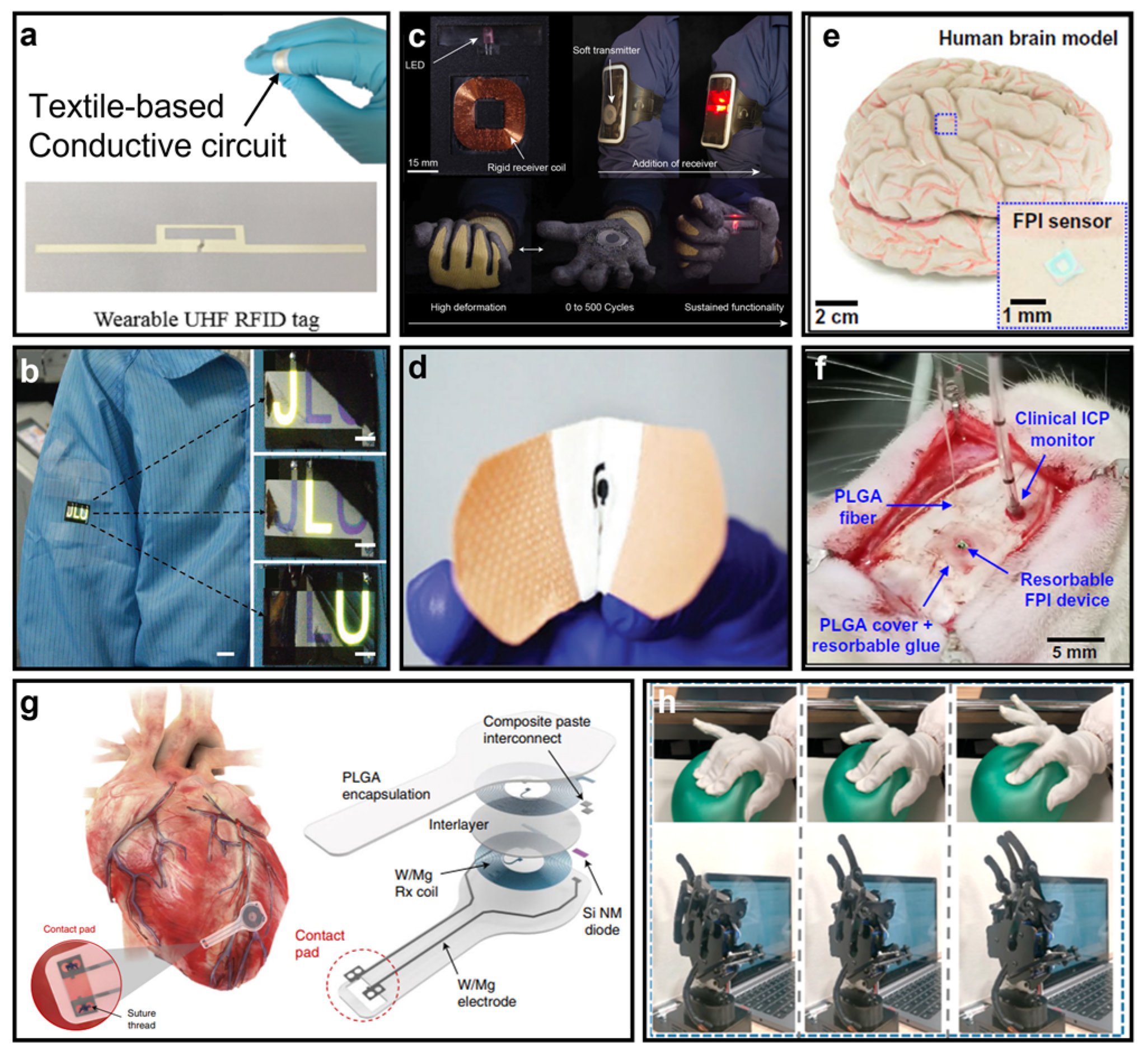

| Soft Substrate | Air Permeability | Waterproofness | Thermal Management | Stretchability | Pore Size |
|---|---|---|---|---|---|
| Porous SEBS [47] | High | Moderate | High | High | 200–800 nm |
| Porous PE [48] | No | No | Low | High | 50–1000 nm |
| Porous PDMS [52] | High | \ | Moderate | Moderate | ~500 nm |
| Porous PU [53] | High | Moderate | No | Moderate | \ |
| Soft Conductors | Flexibility | Stretchability | Toxicity | Fabrication Complexity/Cost | Conductivity |
|---|---|---|---|---|---|
| Ag NWs network [58] | High | High | \ | Moderate/high | 9190 S/cm |
| Ag NWs/PEDOT:PSS [59] | Moderate | Moderate | \ | Moderate/high | 1200 S/cm |
| Graphene oxide ink [60] | High | High | No | Easy/low | 900 S/cm |
| CNT-PMPS-CCB [61] | High | High | No | Moderate/Moderate | 248.8 S/m |
| Au nanomesh [62] | High | Moderate | No | Easy/high | 18,867 S/cm |
| Porous liquid metal [63] | High | High | No | Easy/low | 3.4 × 106 S/cm |
| Molybdenum dioxide [64] | Moderate | Moderate | No | Difficult/low | 11,363 S/cm |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yang, M.; Ye, Z.; Ren, Y.; Farhat, M.; Chen, P.-Y. Recent Advances in Nanomaterials Used for Wearable Electronics. Micromachines 2023, 14, 603. https://doi.org/10.3390/mi14030603
Yang M, Ye Z, Ren Y, Farhat M, Chen P-Y. Recent Advances in Nanomaterials Used for Wearable Electronics. Micromachines. 2023; 14(3):603. https://doi.org/10.3390/mi14030603
Chicago/Turabian StyleYang, Minye, Zhilu Ye, Yichong Ren, Mohamed Farhat, and Pai-Yen Chen. 2023. "Recent Advances in Nanomaterials Used for Wearable Electronics" Micromachines 14, no. 3: 603. https://doi.org/10.3390/mi14030603
APA StyleYang, M., Ye, Z., Ren, Y., Farhat, M., & Chen, P.-Y. (2023). Recent Advances in Nanomaterials Used for Wearable Electronics. Micromachines, 14(3), 603. https://doi.org/10.3390/mi14030603






