Pump-Free Microfluidic Hemofiltration Device
Abstract
1. Introduction
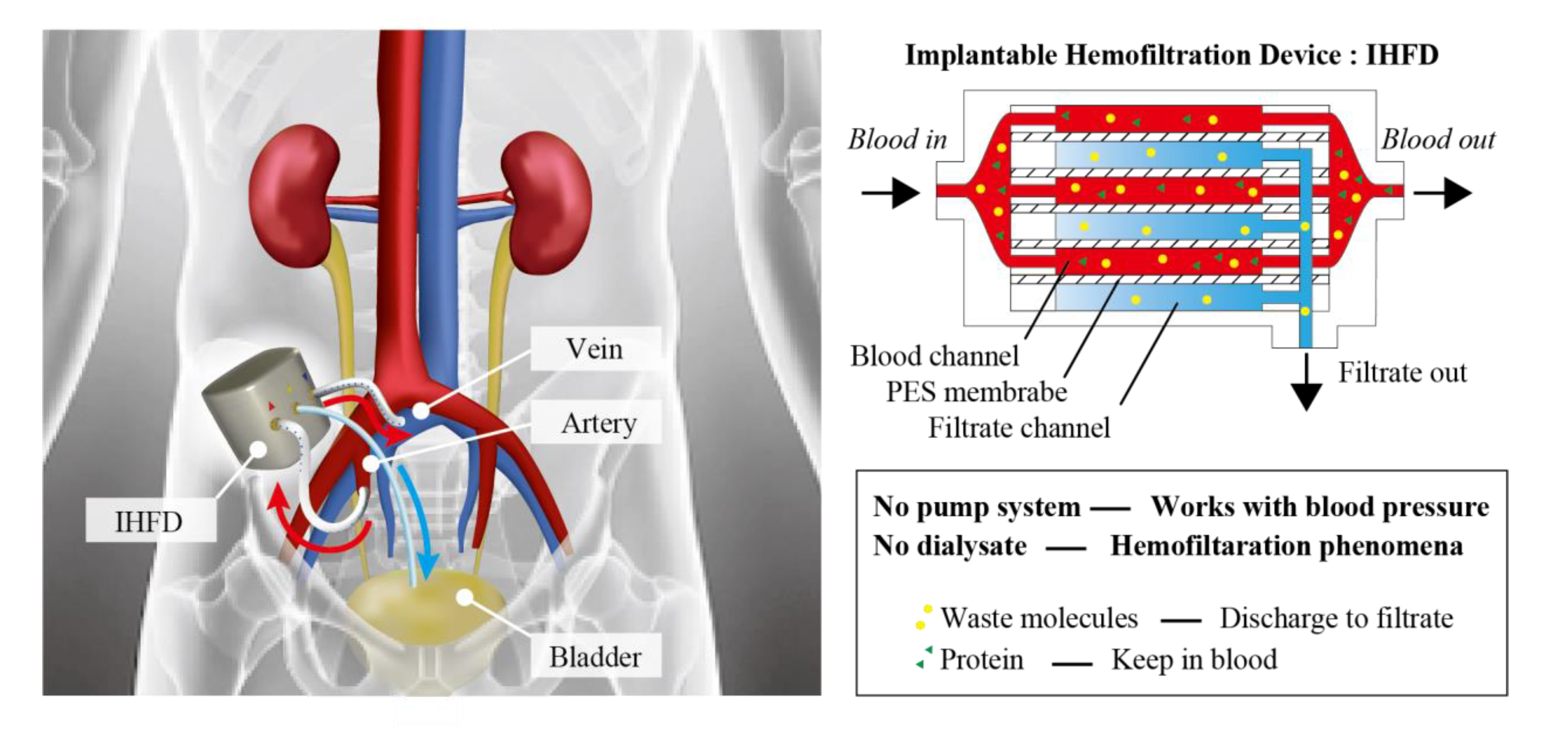
2. Device Design and Experimental Method
2.1. Device Design
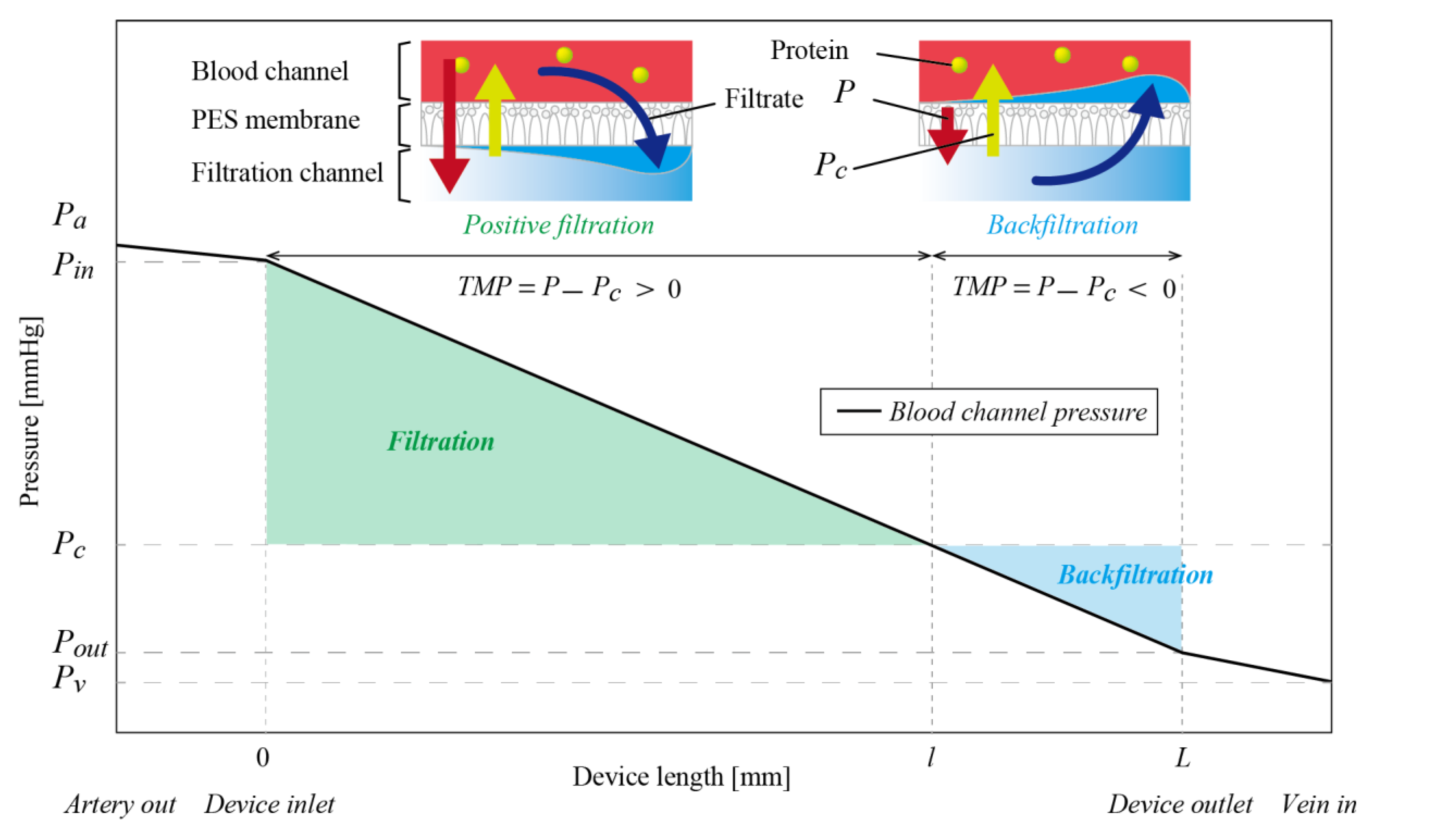
2.2. Amount of Filtrate
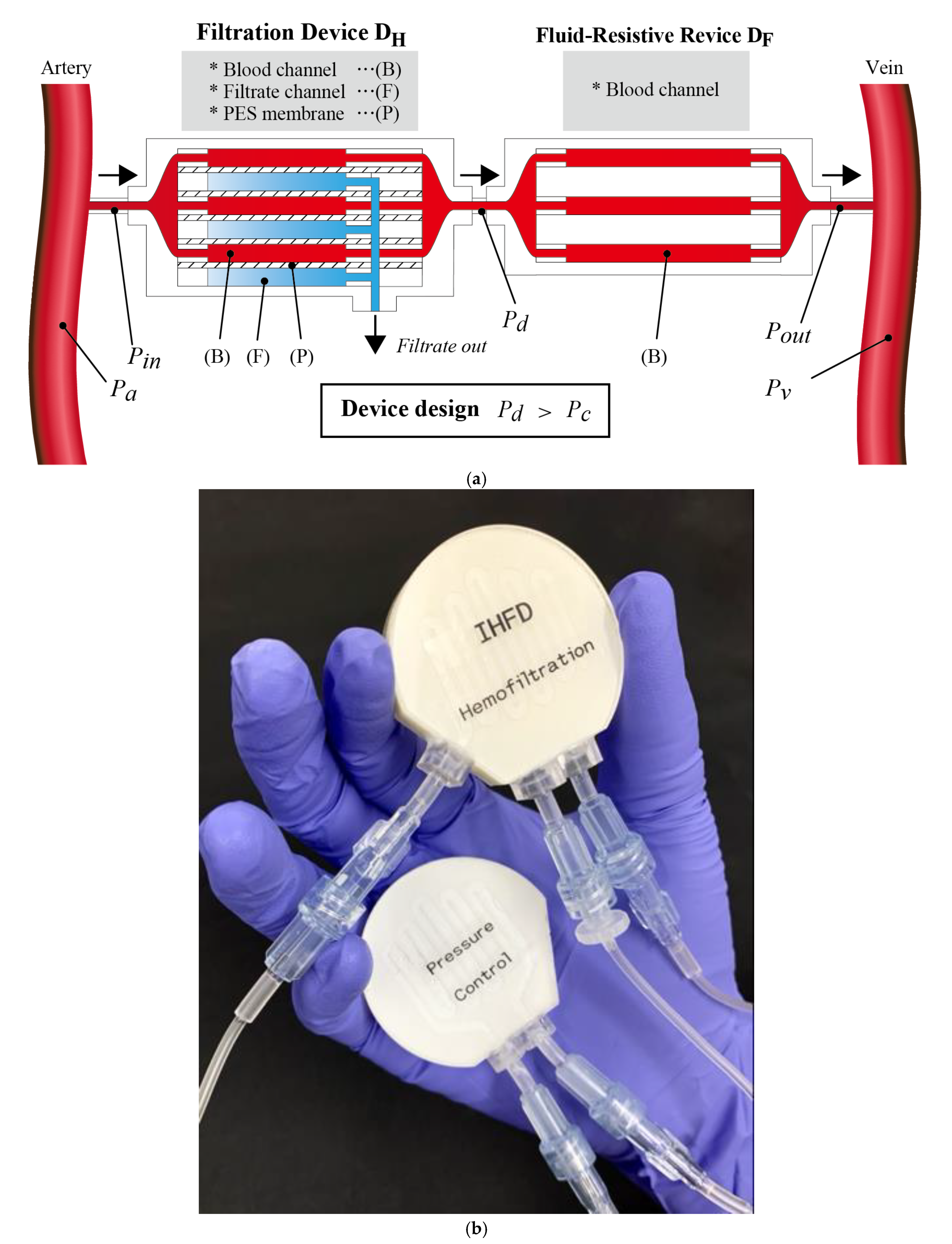
2.3. Hemofiltration Device DH and Fluid-Resistive Device DF
2.4. Experimental Method
2.4.1. Device Fabrication
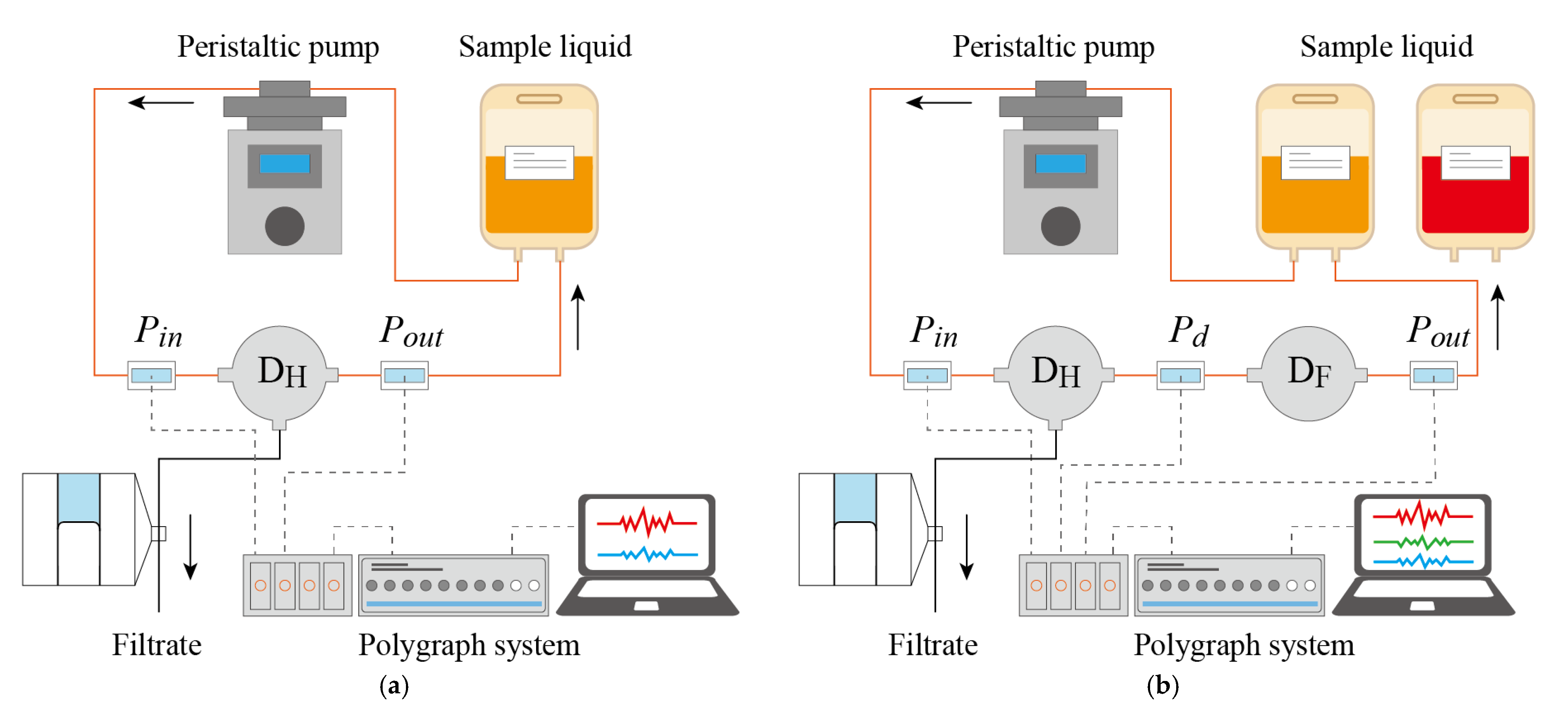
2.4.2. Perfusion System for In Vitro Experiments
2.4.3. Filtration Coefficient
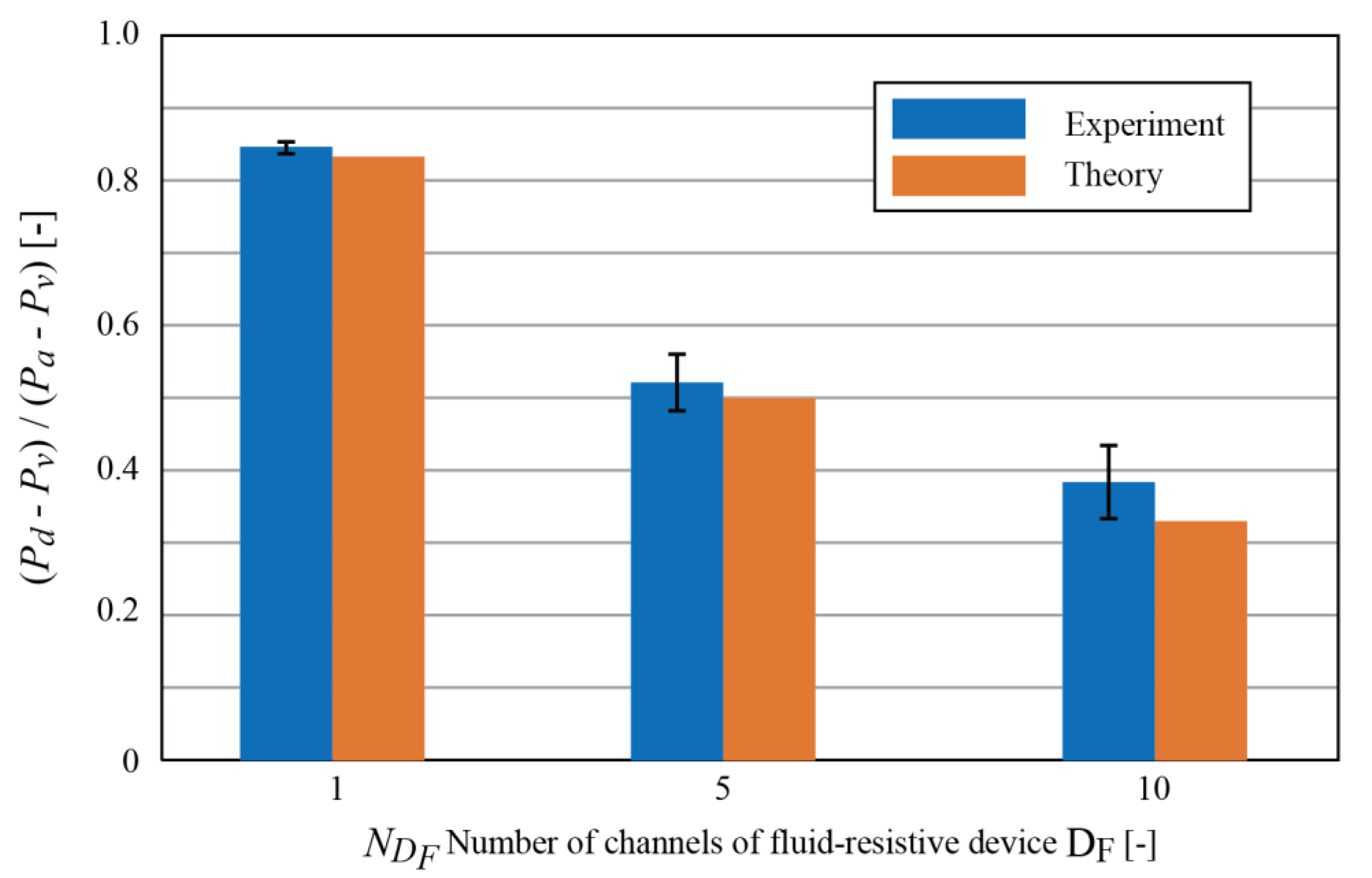
2.4.4. Effect of Fluid-Resistive Device DF
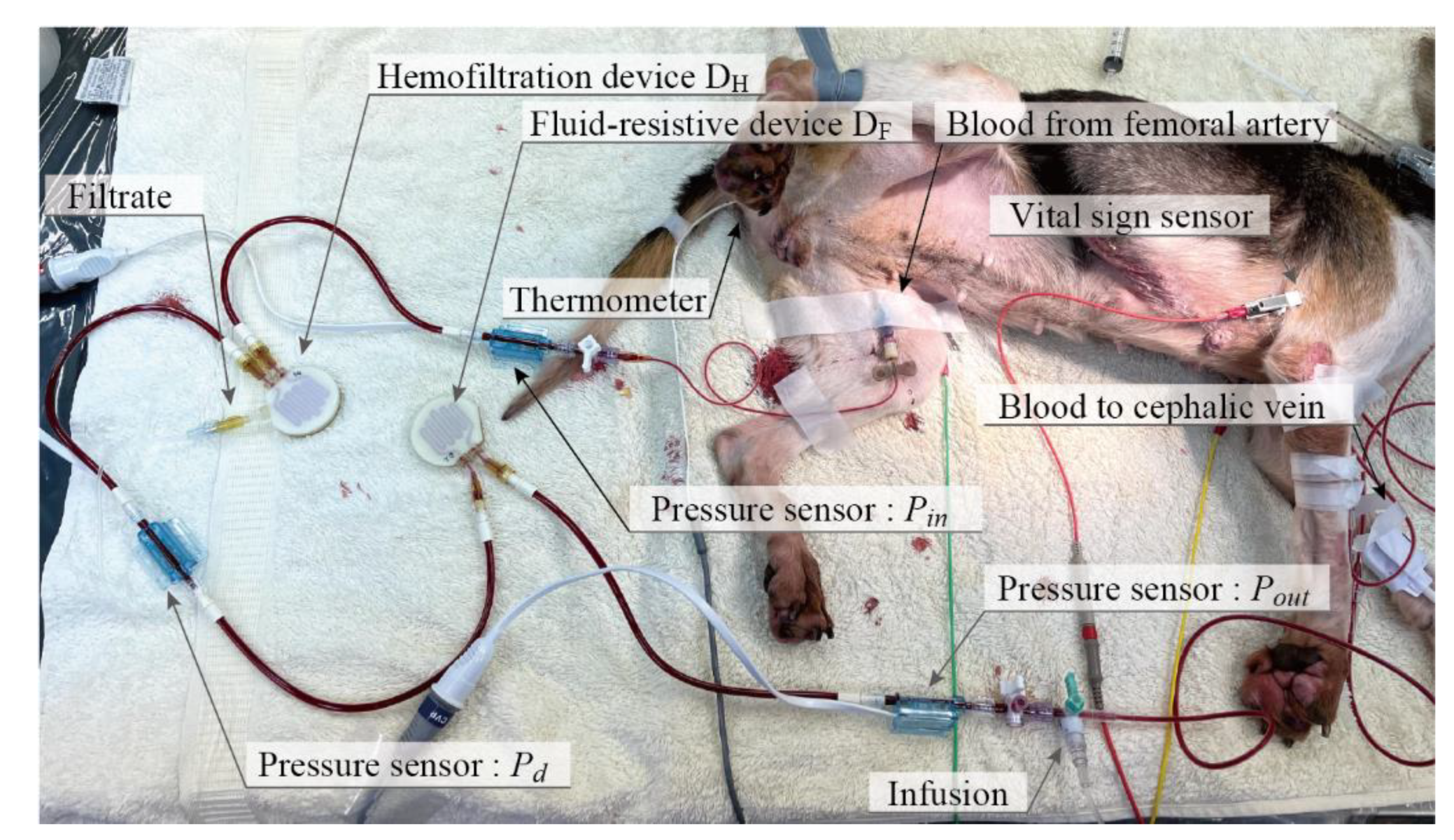
2.4.5. Ex Vivo Experiments
3. Results
3.1. In Vitro Experiment
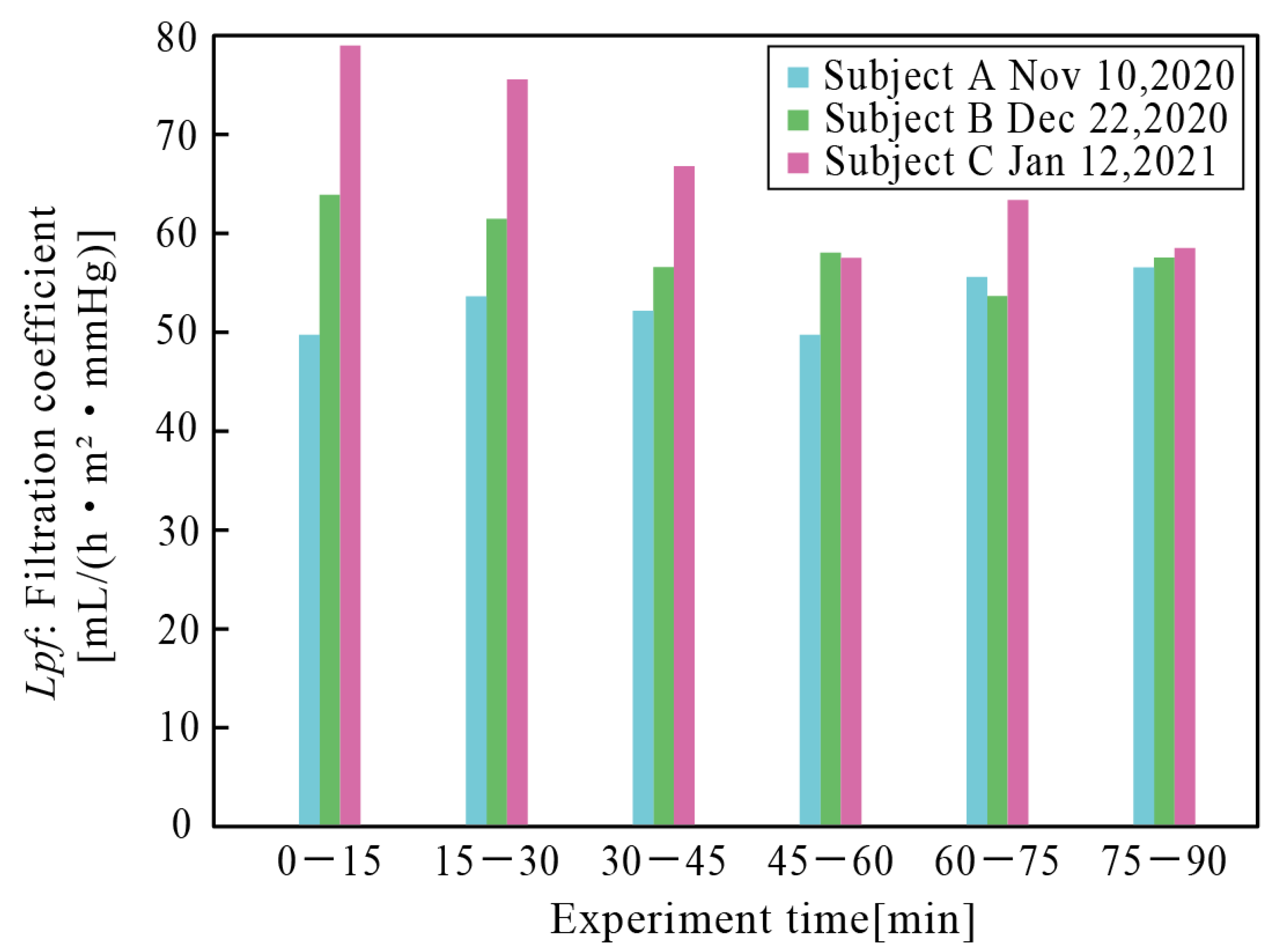
3.1.1. Filtration Coefficient
3.1.2. Effect of Fluid-Resistive Device DF
3.2. Ex Vivo Experiment
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Jaber, B.L.; Zimmerman, D.L.; Teehan, G.S.; Swedko, P.; Burns, K.; Meyer, K.B.; Leypoldt, J.K. Daily Hemofiltration for End-Stage Renal Disease: A Feasibility and Efficacy Trial. Blood Purif. 2004, 22, 481–489. [Google Scholar] [CrossRef]
- Clark, W.R.; Hamburger, R.J.; Lysaght, M.J. Effect of membrane composition and structure on solute removal and biocompatibility in hemodialysis. Kidney Int. 1999, 56, 2005–2015. [Google Scholar] [CrossRef]
- Hüfler, M.; von Herrath, D.; Asmus, G.; Schaefer, K. The Hemofiltration System: Variations of the Negative Pressure. Ren. Fail. 1977, 1, 567–574. [Google Scholar] [CrossRef] [PubMed]
- Morikami, Y.; Fujimori, A.; Kumei, M.; Haibara, H.; Okada, S.; Mizobuchi, N.; Sakai, M.; Nakanishi, T. Influence of blood flow rate on transmembrane pressure in hemodialysis and pre-dilution on-line hemodiafiltration. Nihon Toseki Igakkai Zasshi 2016, 49, 187–190. [Google Scholar] [CrossRef]
- To, N.; Sanada, I.; Ito, H.; Prihandana, G.S.; Morita, S.; Kanno, Y.; Miki, N. Water-Permeable Dialysis Membranes for Multi-Layered Microdialysis System. Front. Bioeng. Biotechnol. 2015, 3, 70. [Google Scholar] [CrossRef]
- Fissell, W.H.; Dubnisheva, A.; Eldridge, A.N.; Fleischman, A.J.; Zydney, A.L.; Roy, S. High-performance silicon nanopore hemofiltration membranes. J. Membr. Sci. 2009, 326, 58–63. [Google Scholar] [CrossRef] [PubMed]
- Chui, B.W.; Wright, N.J.; Ly, J.; Maginnis, D.A.; Haniff, T.M.; Blaha, C.; Fissell, W.H.; Roy, S. A Scalable, Hierarchical Rib Design for Larger-Area, Higher-Porosity Nanoporous Membranes for the Implantable Bio-Artificial Kidney. J. Microelectromech. Syst. 2020, 29, 762–768. [Google Scholar] [CrossRef]
- Nicolaides, A.N.; Hussein, M.K.; Szendro, G.; Christopoulos, D.; Vasdekis, S.; Clarke, H. The relation of venous ulceration with ambulatory venous pressure measurements. J. Vasc. Surg. 1993, 17, 414–419. [Google Scholar] [CrossRef]
- Yamamoto, K.I.; Hiwatari, M.; Kohori, F.; Sakai, K.; Fukuda, M.; Hiyoshi, T. Membrane fouling and dialysate flow pattern in an internal filtration-enhancing dialyzer. J. Artif. Organs Off. J. Jpn. Soc. Artif. Organs 2005, 8, 198–205. [Google Scholar] [CrossRef]
- Ronco, C. Backfiltration in Clinical Dialysis: Nature of the Phenomenon, Mechanisms and Possible Solutions. Int. J. Artif. Organs 2018, 13, 11–21. [Google Scholar] [CrossRef]
- Nakagawa, Y.; Nakamura, R.; Matsuda, K.; Yasumura, Y. A new vascular graft technique for hemodialysis Composite graft using polyurethane and expanded-polytetrafluoroethylene. Nihon Toseki Igakkai Zasshi 2000, 33, 1087–1092. [Google Scholar] [CrossRef][Green Version]
- Ikeda, Y.; Kohjiya, S.; Yamashita, S.; Hayashi, H.; Okuno, T. Separated microphase structure and mechanical properties of blood--compatible polyurethaneureas. J. Rheol. 1998, 35, 692. [Google Scholar] [CrossRef]
- Grasel, T.G.; Cooper, S.L. Surface properties and blood compatibility of polyurethaneureas. Biomaterials 1986, 7, 315–328. [Google Scholar] [CrossRef]
- Canaan-Kuhl, S.; Venkatraman, E.S.; Ernst, S.I.B.; Olshen, R.A.; Myers, B.D. Relationships among protein and albumin concentrations and oncotic pressure in nephrotic plasma. Am. J. Physiol. Ren. Fluid Electrolyte Physiol. 1993, 264, F1052–F1059. [Google Scholar] [CrossRef] [PubMed]
- Weisberg, H.F. Osmotic pressure of the serum proteins. Ann. Clin. Lab. Sci. 1987, 8, 155–164. [Google Scholar]
- Feher, J. Osmosis and Osmotic Pressure. Quant. Hum. Physiol. 2017, 182–198. [Google Scholar] [CrossRef]
- Nitta, S.; Ohnuki, T.; Ohkuda, K.; Nakada, T.; Staub, N.C. The corrected protein equation to estimate plasma colloid osmotic pressure and its development on a nomogram. Tohoku J. Exp. Med. 1981, 135, 43–49. [Google Scholar] [CrossRef]
- Ahlqvist, J.; Staub, N.C. Plasma protein osmotic pressure equations for humans. J. Appl. Physiol. 2003, 94, 1288–1289. [Google Scholar] [CrossRef]
- Zhao, C.; Zhou, X.; Yue, Y. Determination of pore size and pore size distribution on the surface of hollow-fiber filtration membranes: A review of methods. Desalination 2000, 129, 107–123. [Google Scholar] [CrossRef]
- Vilakati, G.D.; Hoek, E.M.V.; Mamba, B.B. Investigating the structure and water permeation of membranes modified with natural and synthetic additives using tensile, porosity, and glass transition temperature studies. J. Appl. Polym. Sci. 2014, 131, 40616. [Google Scholar] [CrossRef]
- Navar, P.D.; Navar, L.G. Relationship between colloid osmotic pressure and plasma protein concentration in the dog. Am. J. Physiol. 1977, 233, 2241–2244. [Google Scholar] [CrossRef]
- Yamada, S.; Grady, M.K.; Licko, V.; Staub, N.C. Plasma protein osmotic pressure equations and nomogram for sheep. J. Appl. Physiol. 1991, 71, 481–487. [Google Scholar] [CrossRef]
- Thomas, L.A.; Brown, S.A. Relationship between colloid osmotic pressure and plasma protein concentration in cattle, horses, dogs, and cats. Am. J. Vet. Res. 1992, 53, 2241–2244. [Google Scholar]
- Stoquart, C.; Vázquez-Rodríguez, G.A.; Servais, P.; Barbeau, B. Gamma irradiation: A method to produce an abiotic control for biological activated carbon. Environ. Technol. 2013, 34, 3079–3085. [Google Scholar] [CrossRef] [PubMed]
- Prihandana, G.S.; Sanada, I.; Ito, H.; Noborisaka, M.; Kanno, Y.; Suzuki, T.; Miki, N. Antithrombogenicity of Fluorinated Diamond-Like Carbon Films Coated Nano Porous Polyethersulfone (PES) Membrane. Materials 2013, 6, 4309–4323. [Google Scholar] [CrossRef]
- Nakamura, M.; Ukita, T. Steady Separation Flow of a Non-Newtonian Fluid through an Axisymmetric Conical Diffuser. NAGARE 1992, 11, 245–253. [Google Scholar] [CrossRef]
- Tomizawa, M.; Hori, S.; Nishimura, N.; Omori, C.; Nakai, Y.; Miyake, M.; Yoneda, T.; Fujimoto, K. Arterial reconstruction using the donor’s gonadal vein in living renal transplantation with multiple renal arteries: A case report and a literature review. BMC Nephrol. 2020, 21, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Mohamed, I.H.; Bagul, A.; Doughman, T.; Nicholson, M.L. Use of internal iliac artery as a side-to-end anastomosis in renal transplantation. Ann. R. Coll. Surg. Engl. 2015, 94, e36–e37. [Google Scholar] [CrossRef]
- Grimm, K.A.; Lamont, L.A.; Tranquilli, W.J.; Greene, S.A.; Robertson, S.A. Veterinary Anesthesia and Analgesia. In The Fifth Edition of Lumb and Jones; John Wiley & Sons: Hoboken, NJ, USA, 2017; pp. 130–146. ISBN 978-1-119-42137-5. [Google Scholar]



| Subject A | Subject B | Subject C | ||
|---|---|---|---|---|
| Date of experiment | 10 November 2020 | 22 December 2020 | 12 January 2021 | |
| Sex | Male | Female | Male | |
| Age (years) | 4.0 | 16.8 | 4.3 | |
| Body weight (kg) | 13.2 | 5.6 | 11.2 | |
| Blood pressure (mmHg) | sBP | 125 | 81 | 121 |
| dBP | 51 | 57 | 55 | |
| MAP | 86 | 67 | 82 | |
| Subject A | ||||
| Time (min) | Filtrate (mL) | (mmHg) | (mmHg) | (mmHg) |
| 0–15 | 1.89 | 58.04 | 54.08 | 46.85 |
| 15–30 | 1.70 | 51.66 | 47.60 | 40.50 |
| 30–45 | 1.63 | 51.35 | 47.20 | 40.01 |
| 45–60 | 1.56 | 51.71 | 47.49 | 40.16 |
| 60–75 | 1.59 | 48.62 | 44.63 | 37.78 |
| 75–90 | 1.34 | 43.46 | 39.81 | 33.75 |
| Subject B | ||||
| Time (min) | Filtrate (mL) | (mmHg) | (mmHg) | (mmHg) |
| 0–15 | 1.53 | 36.38 | 33.33 | 25.35 |
| 15–30 | 1.41 | 35.32 | 32.49 | 24.47 |
| 30–45 | 1.39 | 36.93 | 34.00 | 25.62 |
| 45–60 | 1.43 | 36.92 | 33.94 | 25.66 |
| 60–75 | 1.35 | 37.37 | 34.39 | 26.04 |
| 75–90 | 1.44 | 37.20 | 34.21 | 26.11 |
| Subject C | ||||
| Time (min) | Filtrate (mL) | (mmHg) | (mmHg) | (mmHg) |
| 0–15 | 1.29 | 33.28 | 27.55 | 19.59 |
| 15–30 | 1.32 | 34.35 | 28.44 | 20.12 |
| 30–45 | 1.32 | 36.67 | 30.21 | 21.18 |
| 45–60 | 1.27 | 38.86 | 31.95 | 22.16 |
| 60–75 | 1.40 | 38.87 | 31.94 | 22.07 |
| 75–90 | 1.23 | 37.75 | 31.13 | 21.49 |
| Subject A | ||||||
| Sample | Alb (g/dL) | BUN (mg/dL) | Cre (mg/dL) | Na (mmol/L) | K (mmol/L) | Cl (mmol/L) |
| Blood (before experiment) | 1.4 | 13 | 0.6 | 149 | 3.6 | 109 |
| Blood (after experiment) | 1.5 | 13 | 0.4 | 148 | 3.6 | 109 |
| Filtrate (0–15) | - * | 15 | 0.5 | 146 | 3.5 | 110 |
| 15–30 | - | 16 | 0.6 | 147 | 3.7 | 110 |
| 30–45 | - | 16 | 0.6 | 147 | 3.7 | 108 |
| 45–60 | - | 16 | 0.6 | 145 | 3.6 | 109 |
| 60–75 | - | 16 | 0.5 | 146 | 3.6 | 108 |
| 75–90 | - | 15 | 0.5 | 145 | 3.6 | 108 |
| Subject B | ||||||
| Sample | Alb (g/dL) | BUN (mg/dL) | Cre (mg/dL) | Na (mmol/L) | K (mmol/L) | Cl (mmol/L) |
| Blood (before experiment) | 1.2 | 11 | - ** | 133 | 3.5 | 112 |
| Blood (after experiment) | - | 13 | - | 138 | 3.4 | 112 |
| Filtrate (0–15) | - | 14 | - | 143 | 3.5 | 104 |
| 15–30 | - | 14 | - | 141 | 3.8 | 104 |
| 30–45 | - | 15 | - | 142 | 3.7 | 103 |
| 45–60 | - | 15 | - | 142 | 3.7 | 103 |
| 60–75 | - | 16 | - | 141 | 3.6 | 103 |
| 75–90 | - | 16 | - | 143 | 3.6 | 104 |
| Subject C | ||||||
| Sample | Alb (g/dL) | BUN (mg/dL) | Cre (mg/dL) | Na (mmol/L) | K (mmol/L) | Cl (mmol/L) |
| Blood (before experiment) | 2.2 | 17 | 0.4 | 148 | 4.4 | 102 |
| Blood (after experiment) | 2.2 | 19 | 0.4 | 147 | 5.2 | 102 |
| Filtrate (0–15) | - | 13 | - | 144 | 3.9 | 106 |
| 15–30 | - | 24 | 0.5 | 146 | 3.9 | 106 |
| 30–45 | - | 23 | 0.5 | 144 | 4.2 | 107 |
| 45–60 | - | 25 | 0.5 | 145 | 4.4 | 107 |
| 60–75 | - | 24 | 0.5 | 146 | 4.5 | 106 |
| 75–90 | - | 25 | 0.5 | 144 | 4.6 | 103 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ito, T.; Ota, T.; Kono, R.; Miyaoka, Y.; Ishibashi, H.; Komori, M.; Yasukawa, A.; Kanno, Y.; Miki, N. Pump-Free Microfluidic Hemofiltration Device. Micromachines 2021, 12, 992. https://doi.org/10.3390/mi12080992
Ito T, Ota T, Kono R, Miyaoka Y, Ishibashi H, Komori M, Yasukawa A, Kanno Y, Miki N. Pump-Free Microfluidic Hemofiltration Device. Micromachines. 2021; 12(8):992. https://doi.org/10.3390/mi12080992
Chicago/Turabian StyleIto, Takahiro, Takashi Ota, Rei Kono, Yoshitaka Miyaoka, Hidetoshi Ishibashi, Masaki Komori, Akio Yasukawa, Yoshihiko Kanno, and Norihisa Miki. 2021. "Pump-Free Microfluidic Hemofiltration Device" Micromachines 12, no. 8: 992. https://doi.org/10.3390/mi12080992
APA StyleIto, T., Ota, T., Kono, R., Miyaoka, Y., Ishibashi, H., Komori, M., Yasukawa, A., Kanno, Y., & Miki, N. (2021). Pump-Free Microfluidic Hemofiltration Device. Micromachines, 12(8), 992. https://doi.org/10.3390/mi12080992







