Fused Deposition Modeling of Microfluidic Chips in Polymethylmethacrylate
Abstract
1. Introduction
2. Materials and Methods
2.1. Printing Materials
2.2. Fused Deposition Modeling
2.3. PMMA Precursor Preparation
2.4. Casting Process
2.5. Microfluidic Experiments
2.6. Contact Angle Measurements
2.7. UV-Vis Measurements
2.8. Autofluorescence Measurements
2.9. Biofunctionalization
3. Results and Discussion
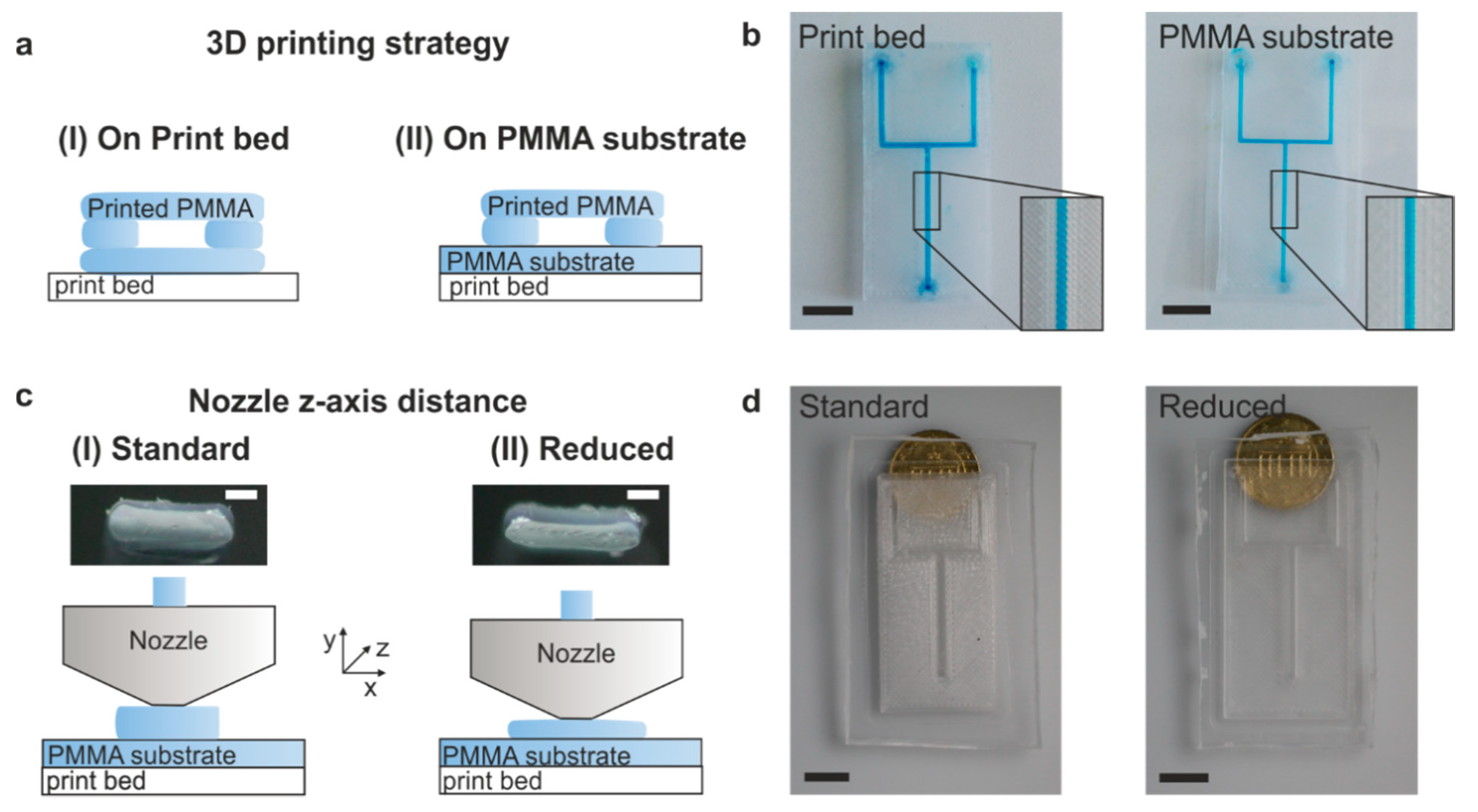
3.1. Optimum Printing Parameters and Transparency Optimization
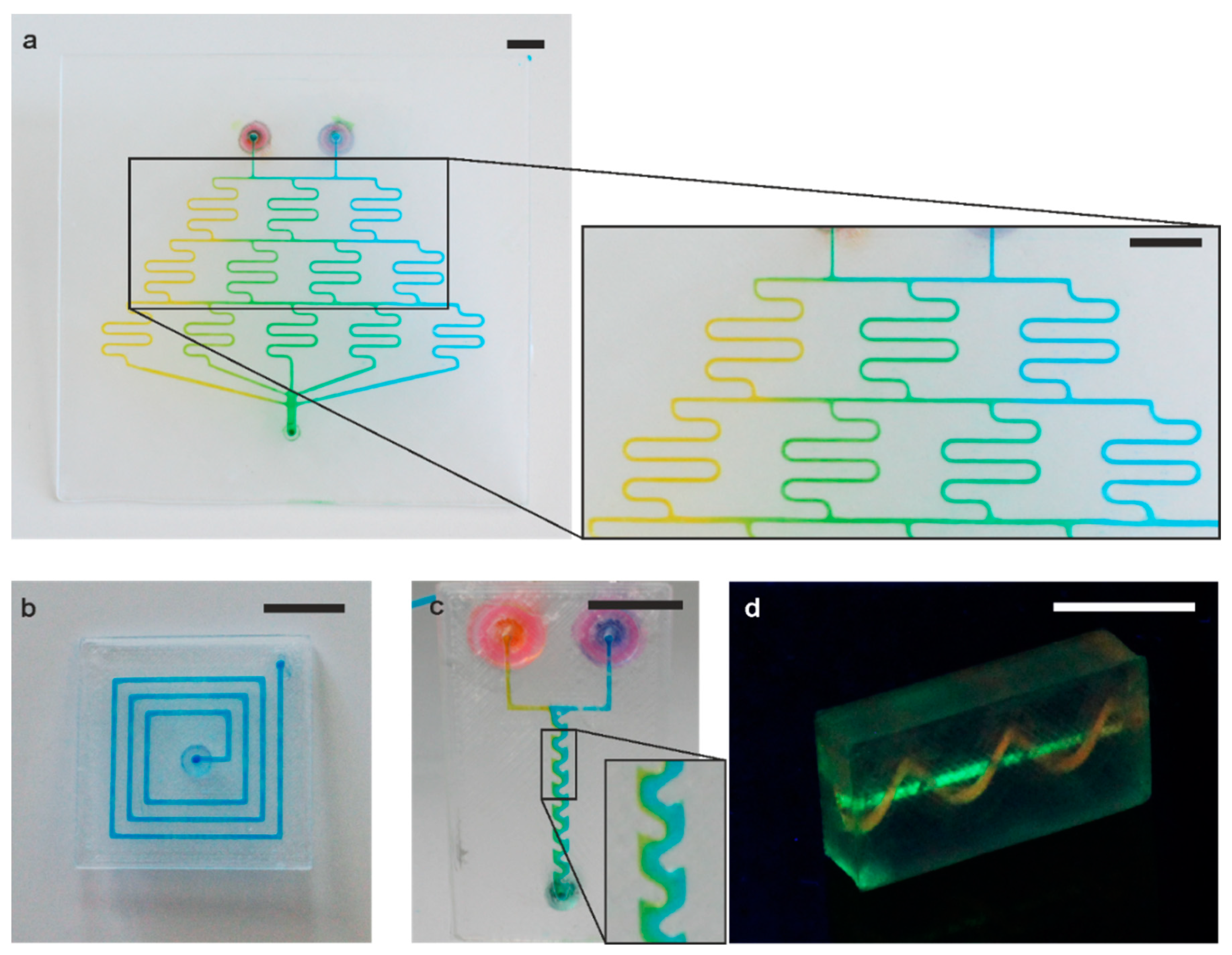
3.2. 3D Printed Microfluidic Devices
3.3. Integration of High-Resolution Microstructures
3.4. Characterization of Printed PMMA
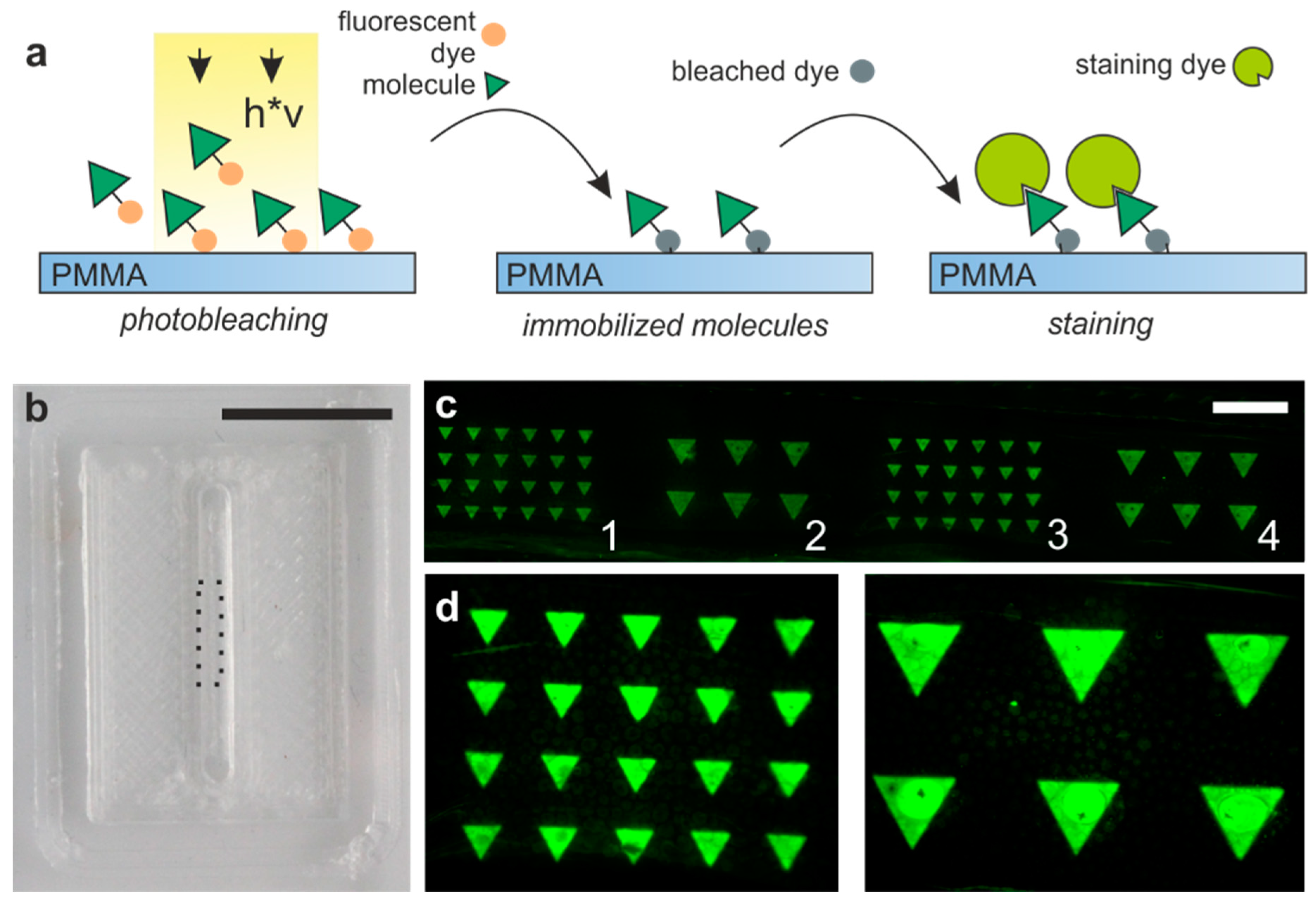
3.5. Biofunctionalization Inside the Printed Microfluidic PMMA Chips
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Waldbaur, A.; Rapp, H.; Lange, K.; Rapp, B.E. Let there be chip—Towards rapid prototyping of microfluidic devices: One-step manufacturing processes. Anal. Methods 2011, 3, 2681. [Google Scholar] [CrossRef]
- Waheed, S.; Cabot, J.M.; Macdonald, N.P.; Lewis, T.; Guijt, R.M.; Paull, B.; Breadmore, M.C. 3D printed microfluidic devices: Enablers and barriers. Lab Chip 2016, 16, 1993–2013. [Google Scholar] [CrossRef] [PubMed]
- Kotz, F.; Risch, P.; Helmer, D.; Rapp, B.E. High-Performance Materials for 3D Printing in Chemical Synthesis Applications. Adv. Mater. 2019, 31, 1805982. [Google Scholar] [CrossRef] [PubMed]
- Kotz, F.; Helmer, D.; Rapp, B.E. Emerging Technologies and Materials for High-Resolution 3D Printing of Microfluidic Chips. In Advances in Biochemical Engineering/Biotechnology; Springer: Berlin/Heidelberg, Germany, 2020. [Google Scholar] [CrossRef]
- Helmer, D.; Voigt, A.; Wagner, S.; Keller, N.; Sachsenheimer, K.; Kotz, F.; Nargang, T.M.; Rapp, B.E. Suspended Liquid Subtractive Lithography: One-step generation of 3D channel geometries in viscous curable polymer matrices. Sci. Rep. 2017, 7, 7387. [Google Scholar] [CrossRef] [PubMed]
- Romanov, V.; Samuel, R.; Chaharlang, M.; Jafek, A.R.; Frost, A.; Gale, B.K. FDM 3D printing of high-pressure, heat-resistant, transparent microfluidic devices. Anal. Chem. 2018, 90, 10450–10456. [Google Scholar] [CrossRef]
- Kotz, F.; Risch, P.; Helmer, D.; Rapp, B.E. Highly fluorinated methacrylates for optical 3D printing of microfluidic devices. Micromachines 2018, 9, 115. [Google Scholar] [CrossRef]
- Sochol, R.D.; Sweet, E.; Glick, C.C.; Venkatesh, S.; Avetisyan, A.; Ekman, K.F.; Raulinaitis, A.; Tsai, A.; Wienkers, A.; Korner, K.; et al. 3D printed microfluidic circuitry via multijet-based additive manufacturing. Lab Chip 2016, 16, 668. [Google Scholar] [CrossRef]
- Sachsenheimer, K.; Richter, C.; Helmer, D.; Kotz, F.; Rapp, B.E. A Nontoxic Battery with 3D-Printed Housing for On-Demand Operation of Microcontrollers in Microfluidic Sensors. Micromachines 2019, 10, 588. [Google Scholar] [CrossRef]
- Shallan, A.I.; Smejkal, P.; Corban, M.; Guijt, R.M.; Breadmore, M.C. Cost-effective three-dimensional printing of visibly transparent microchips within minutes. Anal. Chem. 2014, 86, 3124. [Google Scholar] [CrossRef]
- Rogers, C.I.; Qaderi, K.; Woolley, A.T.; Nordin, G.P. 3D printed microfluidic devices with integrated valves. Biomicrofluidics 2015, 9, 016501. [Google Scholar] [CrossRef]
- Lee, Y.-S.; Bhattacharjee, N.; Folch, A. 3D-printed Quake-style microvalves and micropumps. Lab Chip 2018, 18, 1207. [Google Scholar] [CrossRef] [PubMed]
- Becker, H. Mind the gap! Lab Chip 2010, 10, 271. [Google Scholar] [CrossRef] [PubMed]
- Li, F.; Macdonald, N.P.; Guijt, R.M.; Breadmore, M.C. Multimaterial 3D printed fluidic device for measuring pharmaceuticals in biological fluids. Lab Chip 2019, 19, 35. [Google Scholar] [CrossRef] [PubMed]
- Kitson, P.J.; Rosnes, M.H.; Sans, V.; Dragone, V.; Cronin, L. Configurable 3D-Printed millifluidic and microfluidic ‘lab on a chip’ reactionware devices. Lab Chip 2012, 12, 3267. [Google Scholar] [CrossRef] [PubMed]
- Pranzo, D.; Larizza, P.; Filippini, D.; Percoco, G. Extrusion-based 3D printing of microfluidic devices for chemical and biomedical applications: A topical review. Micromachines 2018, 9, 374. [Google Scholar] [CrossRef]
- Nelson, M.D.; Ramkumar, N.; Gale, B.K. Flexible, transparent, sub-100 µm microfluidic channels with fused deposition modeling 3D-printed thermoplastic polyurethane. J. Micromech. Microeng. 2019, 29, 095010. [Google Scholar] [CrossRef]
- Chen, Y.; Zhang, L.; Chen, G. Fabrication, modification, and application of poly (methyl methacrylate) microfluidic chips. Electrophoresis 2008, 29, 1801. [Google Scholar] [CrossRef]
- McCormick, R.M.; Nelson, R.J.; Alonso-Amigo, M.G.; Benvegnu, D.J.; Hooper, H.H. Microchannel electrophoretic separations of DNA in injection-molded plastic substrates. Anal. Chem. 1997, 69, 2626. [Google Scholar] [CrossRef]
- Martynova, L.; Locascio, L.E.; Gaitan, M.; Kramer, G.W.; Christensen, R.G.; MacCrehan, W.A. Fabrication of plastic microfluid channels by imprinting methods. Anal. Chem. 1997, 69, 4783. [Google Scholar] [CrossRef]
- Brister, P.C.; Weston, K.D. Patterned solvent delivery and etching for the fabrication of plastic microfluidic devices. Anal. Chem. 2005, 77, 7478. [Google Scholar] [CrossRef]
- Sun, X.; Peeni, B.A.; Yang, W.; Becerril, H.A.; Woolley, A.T. Rapid prototyping of poly (methyl methacrylate) microfluidic systems using solvent imprinting and bonding. J. Chromatogr. A 2007, 1162, 162. [Google Scholar] [CrossRef] [PubMed]
- Guckenberger, D.J.; de Groot, T.E.; Wan, A.M.; Beebe, D.J.; Young, E.W. Micromilling: A method for ultra-rapid prototyping of plastic microfluidic devices. Lab Chip 2015, 15, 2364. [Google Scholar] [CrossRef] [PubMed]
- Suriano, R.; Kuznetsov, A.; Eaton, S.M.; Kiyan, R.; Cerullo, G.; Osellame, R.; Chichkov, B.N.; Levi, M.; Turri, S. Femtosecond laser ablation of polymeric substrates for the fabrication of microfluidic channels. Appl. Surf. Sci. 2011, 257, 6243. [Google Scholar] [CrossRef]
- Qu, S.; Chen, X.; Chen, D.; Yang, P.; Chen, G. Poly (methyl methacrylate) CE microchips replicated from poly (dimethylsiloxane) templates for the determination of cations. Electrophoresis 2006, 27, 4910. [Google Scholar] [CrossRef] [PubMed]
- Chen, Z.; Gao, Y.; Su, R.; Li, C.; Lin, J. Fabrication and characterization of poly (methyl methacrylate) microchannels by in situ polymerization with a novel metal template. Electrophoresis 2003, 24, 3246. [Google Scholar] [CrossRef] [PubMed]
- Kotz, F.; Arnold, K.; Wagner, S.; Bauer, W.; Keller, N.; Nargang, T.M.; Helmer, D.; Rapp, B.E. Liquid PMMA: A high resolution polymethylmethacrylate negative photoresist as enabling material for direct printing of microfluidic chips. Advanced Eng. Mater. 2018, 20, 1700699. [Google Scholar] [CrossRef]
- Waldbaur, A.; Waterkotte, B.; Schmitz, K.; Rapp, B.E. Maskless projection lithography for the fast and flexible generation of grayscale protein patterns. Small 2012, 8, 1570. [Google Scholar] [CrossRef]
- Nargang, T.M.; Runck, M.; Helmer, D.; Rapp, B.E. Functionalization of paper using photobleaching: A fast and convenient method for creating paper-based assays with colorimetric and fluorescent readout. Eng. Life Sci. 2016, 16, 525. [Google Scholar] [CrossRef]
- Waterkotte, B.; Bally, F.; Nikolov, P.M.; Waldbaur, A.; Rapp, B.E.; Truckenmüller, R.; Lahann, J.; Schmitz, K.; Giselbrecht, S. Biofunctional micropatterning of thermoformed 3D substrates. Adv. Funct Mater. 2014, 24, 442. [Google Scholar] [CrossRef]
- Bressan, L.P.; Adamo, C.B.; Quero, R.F.; de Jesus, D.P.; da Silva, J.A.F. A simple procedure to produce FDM-based 3D-printed microfluidic devices with an integrated PMMA optical window. Anal. Methods 2019, 11, 1014. [Google Scholar] [CrossRef]
- Briggs, D.; Chan, H.; Hearn, M.J.; McBriar, D.I.; Munro, H.S. The contact angle of poly (methyl methacrylate) cast against glass. Langmuir 1990, 6, 420. [Google Scholar] [CrossRef]



© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kotz, F.; Mader, M.; Dellen, N.; Risch, P.; Kick, A.; Helmer, D.; Rapp, B.E. Fused Deposition Modeling of Microfluidic Chips in Polymethylmethacrylate. Micromachines 2020, 11, 873. https://doi.org/10.3390/mi11090873
Kotz F, Mader M, Dellen N, Risch P, Kick A, Helmer D, Rapp BE. Fused Deposition Modeling of Microfluidic Chips in Polymethylmethacrylate. Micromachines. 2020; 11(9):873. https://doi.org/10.3390/mi11090873
Chicago/Turabian StyleKotz, Frederik, Markus Mader, Nils Dellen, Patrick Risch, Andrea Kick, Dorothea Helmer, and Bastian E. Rapp. 2020. "Fused Deposition Modeling of Microfluidic Chips in Polymethylmethacrylate" Micromachines 11, no. 9: 873. https://doi.org/10.3390/mi11090873
APA StyleKotz, F., Mader, M., Dellen, N., Risch, P., Kick, A., Helmer, D., & Rapp, B. E. (2020). Fused Deposition Modeling of Microfluidic Chips in Polymethylmethacrylate. Micromachines, 11(9), 873. https://doi.org/10.3390/mi11090873




