Structural Stability of Optofluidic Nanostructures in Flow-Through Operation
Abstract
1. Introduction
2. Materials and Methods
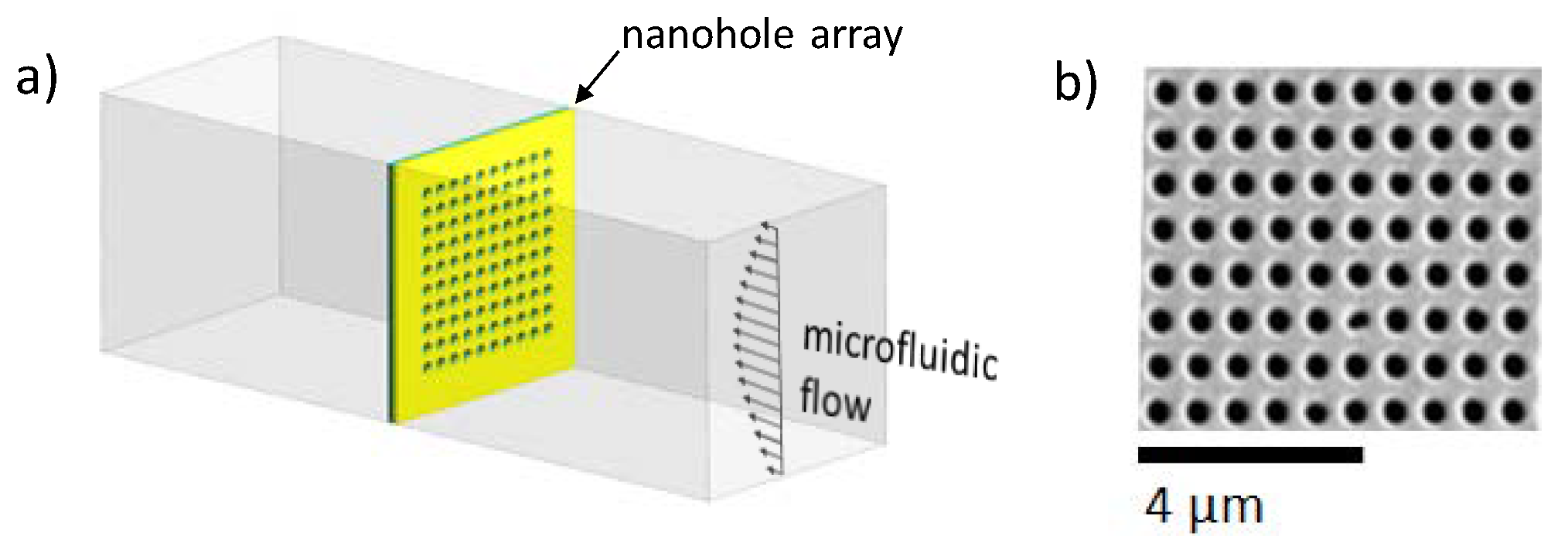
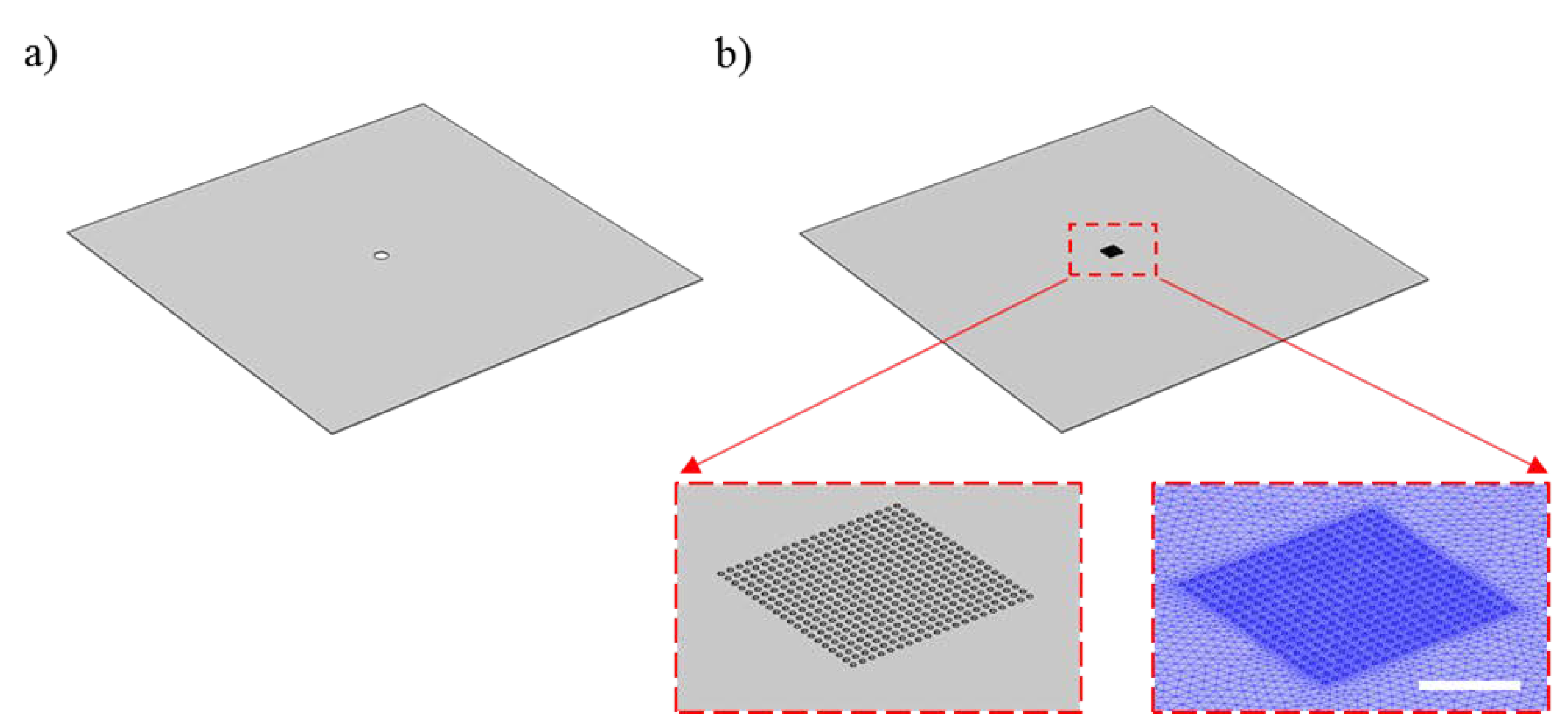
2.1. Fabrication of Periodic through Subwavelength Apertures
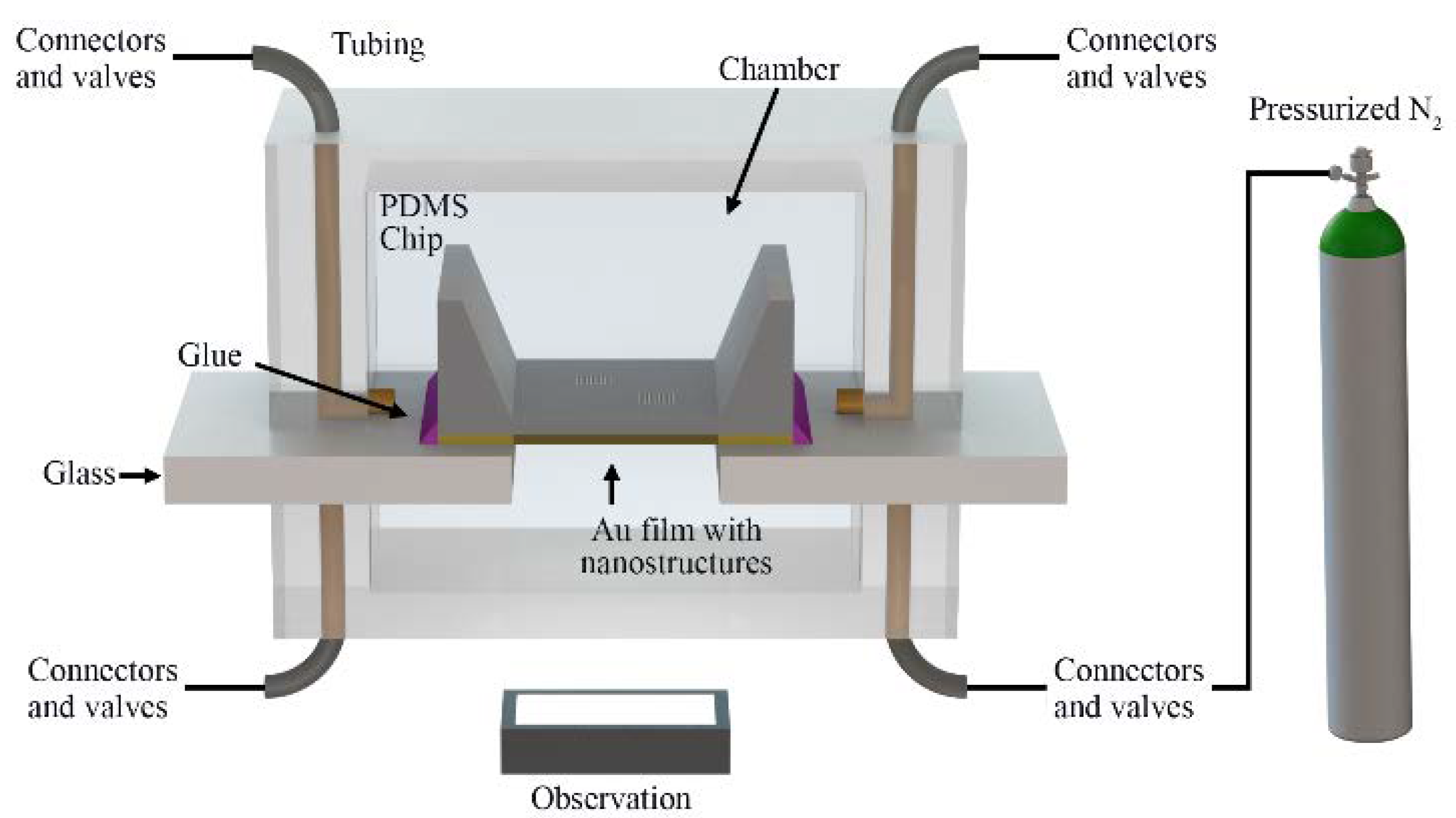
2.2. Fabrication of Microfluidic Chips
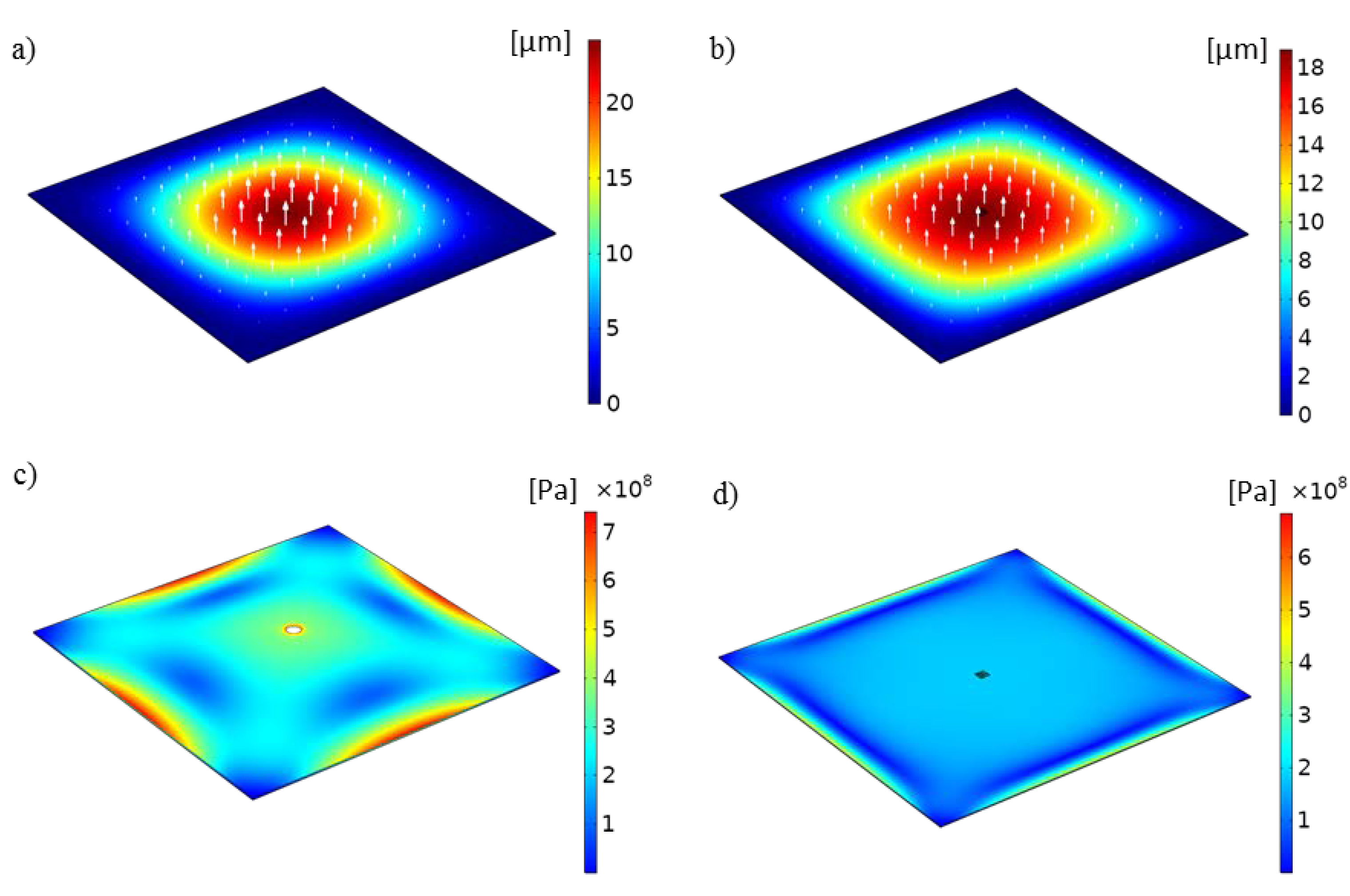
2.3. Optofluidic Structure Deflection Analysis
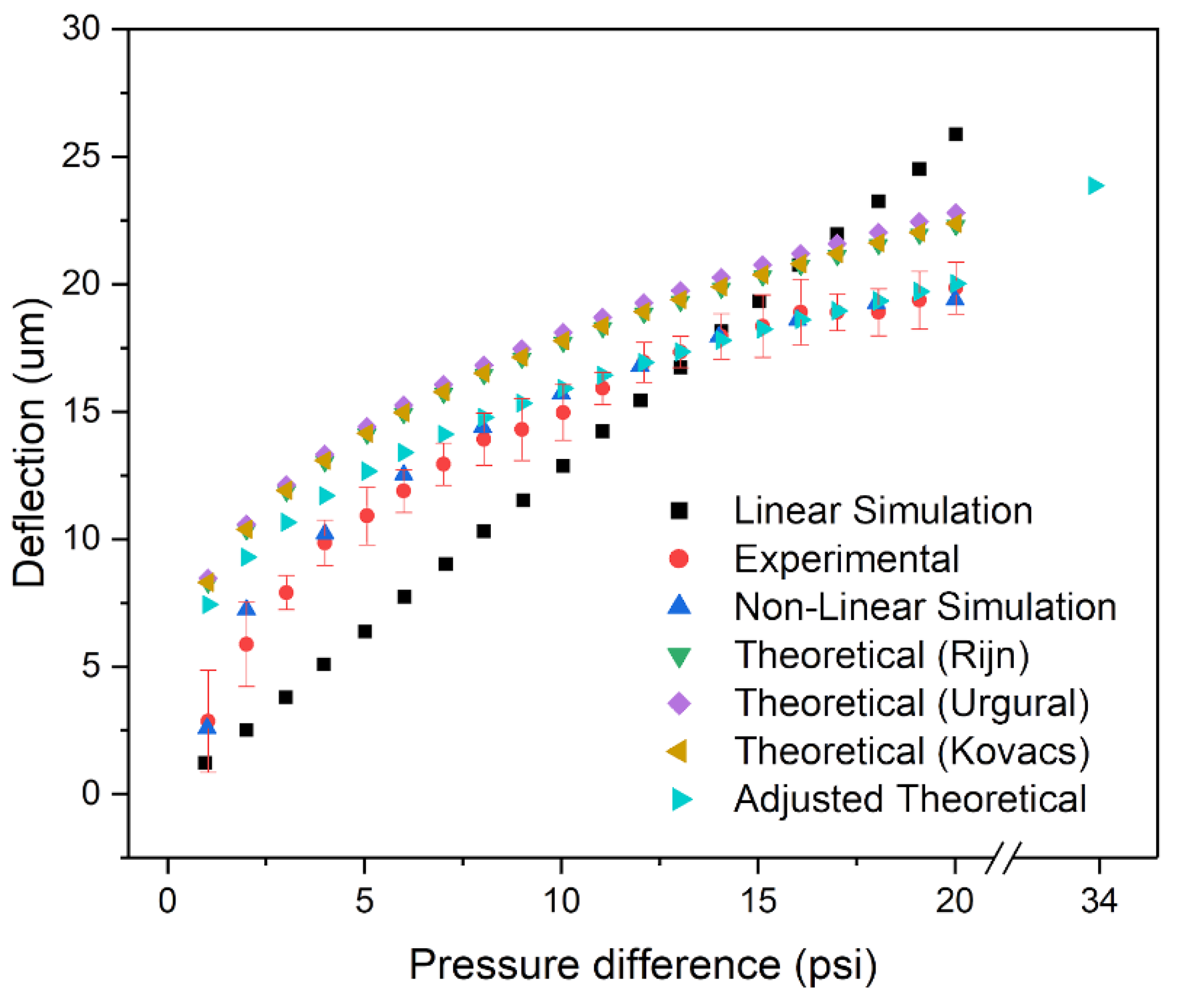
3. Results and Discussion
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Marcoux, R.M.; Vogenberg, F.R. Telehealth: Applications From a Legal and Regulatory Perspective. P T Peer-Rev. J. Formul. Manag. 2016, 41, 567–570. [Google Scholar]
- Preechaburana, P.; Gonzalez, M.C.; Suska, A.; Filippini, D. Surface Plasmon Resonance Chemical Sensing on Cell Phones. Angew. Chem. Int. Ed. 2012, 51, 11585–11588. [Google Scholar] [CrossRef]
- Comina, G.; Suska, A.; Filippini, D. Autonomous chemical sensing interface for universal cell phone readout. Angew. Chem. Int. Ed. 2015, 54, 8708–8712. [Google Scholar] [CrossRef]
- Srinivasan, B.; Tung, S. Development and applications of portable biosensors. J. Lab. Autom. 2015, 20, 365–389. [Google Scholar] [CrossRef]
- Li, G.; De Clercq, E. Therapeutic options for the 2019 novel coronavirus (2019-nCoV); Nature Publishing Group: New York, NY, USA, 2020. [Google Scholar]
- Boratto, P.V.; Oliveira, G.P.; Machado, T.B.; Andrade, A.C.S.; Baudoin, J.-P.; Klose, T.; Schulz, F.; Azza, S.; Decloquement, P.; Chabriere, E.; et al. A mysterious 80 nm amoeba virus with a near-complete” ORFan genome” challenges the classification of DNA viruses. bioRxiv 2020. [Google Scholar] [CrossRef]
- Brolo, A.G.; Gordon, R.; Leathem, B.; Kavanagh, K.L. Surface plasmon sensor based on the enhanced light transmission through arrays of nanoholes in gold films. Langmuir 2004, 20, 4813–4815. [Google Scholar] [CrossRef]
- Ebbesen, T.W.; Lezec, H.J.; Ghaemi, H.F.; Thio, T.; Wolff, P.A. Extraordinary optical transmission through sub-wavelength hole arrays. Nature 1998, 391, 667–669. [Google Scholar] [CrossRef]
- Craighead, H. Future lab-on-a-chip technologies for interrogating individual molecules. In Nanoscience And Technology: A Collection of Reviews from Nature Journals; World Scientific: Singapore, Singapore, 2010; pp. 330–336. [Google Scholar]
- Ahn, C.H.; Choi, J.-W.; Beaucage, G.; Nevin, J.H.; Lee, J.-B.; Puntambekar, A.; Lee, J.Y. Disposable smart lab on a chip for point-of-care clinical diagnostics. Proc. IEEE 2004, 92, 154–173. [Google Scholar] [CrossRef]
- Herrmann, M.; Veres, T.; Tabrizian, M. Enzymatically-generated fluorescent detection in micro-channels with internal magnetic mixing for the development of parallel microfluidic ELISA. Lab Chip 2006, 6, 555–560. [Google Scholar] [CrossRef]
- Chin, C.D.; Linder, V.; Sia, S.K. Lab-on-a-chip devices for global health: Past studies and future opportunities. Lab Chip 2007, 7, 41–57. [Google Scholar] [CrossRef]
- Gomez-Cruz, J.; Nair, S.; Ascanio, G.; Escobedo, C. Flow-through nanohole array based sensor implemented on analogue smartphone components. In Proceedings of the SPIE Nanoscience + Engineering, San Diego, CA, USA, 25 August 2017. [Google Scholar]
- Soler, M.; Belushkin, A.; Cavallini, A.; Kebbi-Beghdadi, C.; Greub, G.; Altug, H. Multiplexed nanoplasmonic biosensor for one-step simultaneous detection of Chlamydia trachomatis and Neisseria gonorrhoeae in urine. Biosens. Bioelectron. 2017, 94, 560–567. [Google Scholar] [CrossRef] [PubMed]
- Yanik, A.A.; Huang, M.; Kamohara, O.; Artar, A.; Geisbert, T.W.; Connor, J.H.; Altug, H. An Optofluidic Nanoplasmonic Biosensor for Direct Detection of Live Viruses from Biological Media. Nano Lett. 2010, 10, 4962–4969. [Google Scholar] [CrossRef]
- Escobedo, C.; Chou, Y.W.; Rahman, M.; Duan, X.B.; Gordon, R.; Sinton, D.; Brolo, A.G.; Ferreira, J. Quantification of ovarian cancer markers with integrated microfluidic concentration gradient and imaging nanohole surface plasmon resonance. Analyst 2013, 138, 1450–1458. [Google Scholar] [CrossRef]
- Gomez-Cruz, J.; Nair, S.; Manjarrez-Hernandez, A.; Gavilanes-Parra, S.; Ascanio, G.; Escobedo, C. Cost-effective flow-through nanohole array-based biosensing platform for the label-free detection of uropathogenic E. coli in real time. Biosens. Bioelectron. 2018, 106, 105–110. [Google Scholar] [CrossRef]
- Escobedo, C.; Brolo, A.G.; Gordon, R.; Sinton, D. Optofluidic concentration: Plasmonic nanostructure as concentrator and sensor. Nano Lett. 2012, 12, 1592–1596. [Google Scholar] [CrossRef] [PubMed]
- Cruz, S.; Hönig-d’Orville, A.; Müller, J. Fabrication and optimization of porous silicon substrates for diffusion membrane applications. J. Electrochem. Soc. 2005, 152, C418–C424. [Google Scholar] [CrossRef]
- Sinton, D.; Gordon, R.; Brolo, A.G. Nanohole arrays in metal films as optofluidic elements: Progress and potential. Microfluid. Nanofluidics 2008, 4, 107–116. [Google Scholar] [CrossRef]
- Wu, H.; Liu, G.; Wang, J. Atomistic and continuum simulation on extension behaviour of single crystal with nano-holes. Model. Simul. Mater. Sci. Eng. 2004, 12, 225. [Google Scholar] [CrossRef]
- Van Rijn, C.; van der Wekken, M.; Nijdam, W.; Elwenspoek, M. Deflection and maximum load of microfiltration membrane sieves made with silicon micromachining. J. Microelectromech. Syst. 1997, 6, 48–54. [Google Scholar] [CrossRef][Green Version]
- Escobedo, C.; Brolo, A.G.; Gordon, R.; Sinton, D. Nanoplasmonics as nanofluidics: Transport and sensing in flowthrough nanohole arrays. In Proceedings of the Microfluidics, BioMEMS, and Medical Microsystems IX, San Francisco, CA, USA, 14 February 2011. [Google Scholar]
- Escobedo, C. On-chip nanohole array based sensing: A review. Lab Chip 2013, 13, 2445–2463. [Google Scholar] [CrossRef]
- Duffy, D.C.; McDonald, J.C.; Schueller, O.J.A.; Whitesides, G.M. Rapid prototyping of microfluidic systems in poly(dimethylsiloxane). Anal. Chem. 1998, 70, 4974–4984. [Google Scholar] [CrossRef] [PubMed]
- Luecke, W.E.; Wiederhorn, S.M. A new model for tensile creep of silicon nitride. J. Am. Ceram. Soc. 1999, 82, 2769–2778. [Google Scholar] [CrossRef]
- Wunderlich, W. Ceramic Materials; InTech: Rijeka, Croatia, 2010. [Google Scholar]
- Ugural, A.C. Stresses in Beams, Plates, and Shells; CRC press: Boca Raton, FL, USA, 2009. [Google Scholar]
- Kovács, Á.; Vízváry, Z.; Kovács, A.; Mescheder, U. Large deflection analysis of perforated silicon nitride membranes. In Proceedings of the 6th ESMC, Budapest, Hungary, 28 August–1 September 2006. [Google Scholar]
- Kovacs, A.; Kovács, Á.; Pogány, M.; Mescheder, U. Mechanical investigation of perforated and porous membranes for micro-and nanofilter applications. Sens. Actuators B Chem. 2007, 127, 120–125. [Google Scholar] [CrossRef]
- Timoshenko, S.P.; Woinowsky-Krieger, S. Theory of Plates and Shells; McGraw-hill: New York, NY, USA, 1959. [Google Scholar]
- Boé, A.; Coulombier, M.; Safi, A.; Pardoen, T.; Raskin, J.-P. On-chip Testing Laboratory for Nanomechanical Characterization of Thin Films. In Proceedings of the 2009 SEM Annual Conference and Exposition on Experimental and Applied Mechanics, Albuquerque, NM, USA, 1–4 June 2009. [Google Scholar]

© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bdour, Y.; Gomez-Cruz, J.; Escobedo, C. Structural Stability of Optofluidic Nanostructures in Flow-Through Operation. Micromachines 2020, 11, 373. https://doi.org/10.3390/mi11040373
Bdour Y, Gomez-Cruz J, Escobedo C. Structural Stability of Optofluidic Nanostructures in Flow-Through Operation. Micromachines. 2020; 11(4):373. https://doi.org/10.3390/mi11040373
Chicago/Turabian StyleBdour, Yazan, Juan Gomez-Cruz, and Carlos Escobedo. 2020. "Structural Stability of Optofluidic Nanostructures in Flow-Through Operation" Micromachines 11, no. 4: 373. https://doi.org/10.3390/mi11040373
APA StyleBdour, Y., Gomez-Cruz, J., & Escobedo, C. (2020). Structural Stability of Optofluidic Nanostructures in Flow-Through Operation. Micromachines, 11(4), 373. https://doi.org/10.3390/mi11040373





