2019 Novel Coronavirus Disease (COVID-19): Paving the Road for Rapid Detection and Point-of-Care Diagnostics
Abstract
1. Introduction
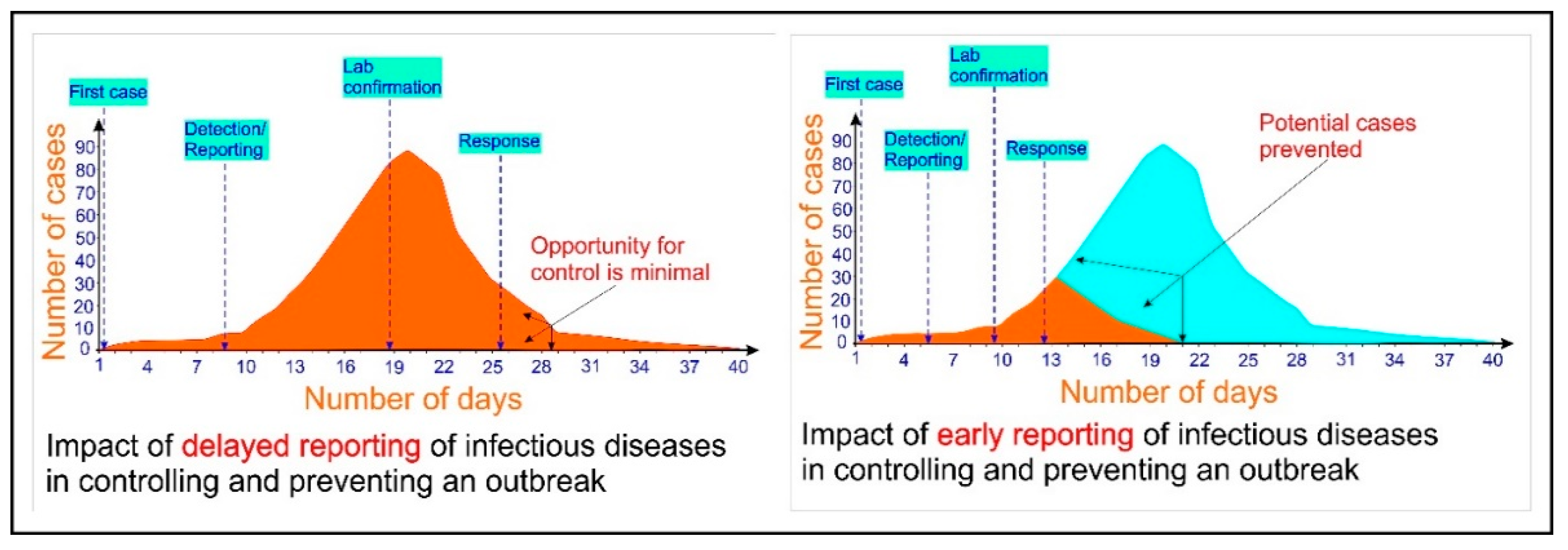
2. The Need for a Rapid Detection Method and Portable Detection Devices
3. A Potential Candidate for Rapid Detection of SARS-CoV-2: Loop-Mediated Isothermal Amplification (LAMP) Assays in PoC Devices
4. Other Important Factors in Fighting COVID-19
Author Contributions
Funding
Conflicts of Interest
References
- Novel Coronavirus (2019-nCoV), Situation Report 11. Available online: https://www.who.int/docs/default-source/coronaviruse/situation-reports/20200131-sitrep-11-ncov.pdf?sfvrsn=de7c0f7_4 (accessed on 13 March 2020).
- Novel Coronavirus ( 2019-nCoV ) Situation Report 48. Available online: https://www.who.int/docs/default-source/coronaviruse/situation-reports/20200308-sitrep-48-covid-19.pdf?sfvrsn=16f7ccef_4 (accessed on 13 March 2020).
- How 2019-nCoV Spreads. Available online: https://www.cdc.gov/coronavirus/2019-ncov/about/transmission.html (accessed on 13 March 2020).
- Holshue, M.L.; DeBolt, C.; Lindquist, S.; Lofy, K.H.; Wiesman, J.; Bruce, H.; Spitters, C.; Ericson, K.; Wilkerson, S.; Tural, A.; et al. First Case of 2019 Novel Coronavirus in the United States. N. Engl. J. Med. 2020, 382, 929–936. [Google Scholar] [CrossRef] [PubMed]
- Rothe, C.; Schunk, M.; Sothmann, P.; Bretzel, G.; Froeschl, G.; Wallrauch, C.; Zimmer, T.; Thiel, V.; Janke, C.; Guggemos, W.; et al. Transmission of 2019-nCoV Infection from an Asymptomatic Contact in Germany. N. Engl. J. Med. 2020, 382, 970–971. [Google Scholar] [CrossRef] [PubMed]
- Phan, L.T.; Nguyen, T.V.; Luong, Q.C.; Nguyen, T.V.; Nguyen, H.T.; Le, H.Q.; Nguyen, T.T.; Cao, T.M.; Pham, Q.D. Importation and Human-to-Human Transmission of a Novel Coronavirus in Vietnam. N. Engl. J. Med. 2020, 382, 872–874. [Google Scholar] [CrossRef] [PubMed]
- WHO, Novel Coronavirus, Situation Dashboard. Available online: https://www.who.int/emergencies/diseases/novel-coronavirus-2019/situation-reports/ (accessed on 9 March 2020).
- Cohen, J.; Kupferschmidt, K. Strategies shift as coronavirus pandemic looms. Science 2020, 367, 962–963. [Google Scholar] [CrossRef]
- Italy Announces Lockdown as Global Coronavirus Cases Surpass 105,000. Available online: https://edition.cnn.com/2020/03/08/asia/coronavirus-covid-19-update-intl-hnk/index.html (accessed on 8 March 2020).
- Wu, J.T.; Leung, K.; Leung, G.M. Nowcasting and forecasting the potential domestic and international spread of the 2019-nCoV outbreak originating in Wuhan, China: A modelling study. Lancet 2020, 395, 689–697. [Google Scholar] [CrossRef]
- Update: ‘A Bit Chaotic.’ Christening of New Coronavirus and Its Disease Name Create Confusion. Available online: https://www.sciencemag.org/news/2020/02/bit-chaotic-christening-new-coronavirus-and-its-disease-name-create-confusion (accessed on 13 March 2020).
- Ji, W.; Wang, W.; Zhao, X.; Zai, J.; Li, X. Homologous recombination within the spike glycoprotein of the newly identified coronavirus may boost cross-species transmission from snake to human. J. Med. Virol. 2020. [Google Scholar] [CrossRef]
- Cyranoski, D. Did pangolins spread the China coronavirus to people? Nature 2020. [Google Scholar] [CrossRef]
- WHO Recommendations to Reduce Risk of Transmission of Emerging Pathogens from Animals to Humans in Live Animal Markets. Available online: https://www.who.int/health-topics/coronavirus/who-recommendations-to-reduce-risk-of-transmission-of-emerging-pathogens-from-animals-to-humans-in-live-animal-markets (accessed on 13 March 2020).
- Wang, W.; Tang, J.; Wei, F. Updated understanding of the outbreak of 2019 novel coronavirus (2019-nCoV) in Wuhan, China. J. Med. Virol. 2020, 92, 441–447. [Google Scholar] [CrossRef]
- Tang, B.; Bragazzi, N.L.; Li, Q.; Tang, S.; Xiao, Y.; Wu, J. An updated estimation of the risk of transmission of the novel coronavirus (2019-nCov). Infect. Dis. Model. 2020, 5, 248–255. [Google Scholar] [CrossRef]
- Gallagher, T.M.; Buchmeier, M.J. Coronavirus Spike Proteins in Viral Entry and Pathogenesis. Virology 2001, 279, 371–374. [Google Scholar] [CrossRef]
- Schoeman, D.; Fielding, B.C. Coronavirus envelope protein: Current knowledge. Virol. J. 2019, 16, 69. [Google Scholar] [CrossRef] [PubMed]
- Gralinski, L.; Menachery, V.D. Return of the Coronavirus: 2019-nCoV. Viruses 2020, 12, 135. [Google Scholar] [CrossRef] [PubMed]
- Huang, C.; Wang, Y.; Li, X.; Ren, L.; Zhao, J.; Hu, Y.; Zhang, L.; Fan, G.; Xu, J.; Gu, X.; et al. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet 2020, 395, 497–506. [Google Scholar] [CrossRef]
- Nguyen, T.; Andreasen, S.Z.; Wolff, A.; Bang, D.D. From Lab on a Chip to Point of Care Devices: The Role of Open Source Microcontrollers. Micromachines 2018, 9, 403. [Google Scholar] [CrossRef] [PubMed]
- Ke, C.-M.; Tsai, H.-C.; Chen, Y.-S.; Huang, Y.-H.; Lai, Y.-Y.; Chen, Y.-J.; Ni, Y.-Y.; Li, C.-H.; Lee, S.S.-J. Outbreak investigation of pandemic influenza A H1N1 at the emergency department in a medical center in Southern Taiwan. J. Microbiol. Immunol. Infect. 2015, 48, S36. [Google Scholar] [CrossRef]
- Isere, E.E.; Fatiregun, A.; Ajayi, I.O. An overview of disease surveillance and notification system in Nigeria and the roles of clinicians in disease outbreak prevention and control. Niger. Med. J. 2015, 56, 161–168. [Google Scholar] [CrossRef] [PubMed]
- Jones, G.; Le Hello, S.; Silva, N.J.-D.; Vaillant, V.; De Valk, H.; Weill, F.-X.; Le Strat, Y. The French human Salmonella surveillance system: Evaluation of timeliness of laboratory reporting and factors associated with delays, 2007 to 2011. Eurosurveillance 2014, 19, 20664. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Coronavirus Infection Tally on Diamond Princess hits 135 as Tests for All Passengers Eyed. Available online: https://www.japantimes.co.jp/news/2020/02/10/national/japan-test-all-passengers-diamond-princess-cruise-ship-coronavirus/#.XkQLz2hKhaQ (accessed on 10 February 2020).
- Corman, V.M.; Landt, O.; Kaiser, M.; Molenkamp, R.; Meijer, A.; Chu, D.K.; Bleicker, T.; Brünink, S.; Schneider, J.; Schmidt, M.L.; et al. Detection of 2019 novel coronavirus (2019-nCoV) by real-time RT-PCR. Eurosurveillance 2020, 25, 2000045. [Google Scholar] [CrossRef] [PubMed]
- Chu, D.K.W.; Pan, Y.; Cheng, S.M.S.; Hui, K.P.Y.; Krishnan, P.; Liu, Y.; Ng, D.Y.M.; Wan, C.K.C.; Yang, P.; Wang, Q.; et al. Molecular Diagnosis of a Novel Coronavirus (2019-nCoV) Causing an Outbreak of Pneumonia. Clin. Chem. 2020, 7, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Bruning, A.H.; Aatola, H.; Toivola, H.; Ikonen, N.; Savolainen-Kopra, C.; Blomqvist, S.; Pajkrt, D.; Wolthers, K.C.; Koskinen, J.O. Rapid detection and monitoring of human coronavirus infections. New Microbes New Infect. 2018, 24, 52–55. [Google Scholar] [CrossRef]
- Gaunt, E.R.; Hardie, A.; Claas, E.C.J.; Simmonds, P.; Templeton, K.E. Epidemiology and Clinical Presentations of the Four Human Coronaviruses 229E, HKU1, NL63, and OC43 Detected over 3 Years Using a Novel Multiplex Real-Time PCR Method. J. Clin. Microbiol. 2010, 48, 2940–2947. [Google Scholar] [CrossRef] [PubMed]
- Cho, C.H.; Lee, C.K.; Nam, M.H.; Yoon, S.-Y.; Lim, C.S.; Cho, Y.; Kim, Y.K. Evaluation of the AdvanSure™ real-time RT-PCR compared with culture and Seeplex RV15 for simultaneous detection of respiratory viruses. Diagn. Microbiol. Infect. Dis. 2014, 79, 14–18. [Google Scholar] [CrossRef]
- There’s Only One Way to Know If You Have the Coronavirus, and It Involves Machines Full of Spit and Mucus. Available online: https://www.businessinsider.com/how-to-know-if-you-have-the-coronavirus-pcr-test-2020-1?r=US&IR=T (accessed on 13 March 2020).
- The US Fast-Tracked a Coronavirus Test to Speed Up Diagnoses. Available online: https://www.wired.com/story/the-us-fast-tracked-a-coronavirus-test/ (accessed on 13 March 2020).
- Assistance to Wuhan, High-level Mobile Biosafety Lab Departs. Available online: http://www.chinacdc.cn/yw_9324/202002/t20200204_212214.html (accessed on 13 March 2020).
- New Emergency Detection Laboratory Run by BGI Starts Trial Operation in Wuhan, Designed to Test 10,000 Samples Daily. Available online: https://www.bgi.com/global/company/news/new-emergency-detection-laboratory-run-by-bgi-starts-trial-operation-in-wuhan-designed-to-test-10000-samples-daily/ (accessed on 13 March 2020).
- Nagamine, K.; Hase, T.; Notomi, T. Accelerated reaction by loop-mediated isothermal amplification using loop primers. Mol. Cell. Probes 2002, 16, 223–229. [Google Scholar] [CrossRef] [PubMed]
- Francois, P.; Tangomo, M.; Hibbs, J.; Bonetti, E.-J.; Boehme, C.C.; Notomi, T.; Perkins, M.D.; Schrenzel, J. Robustness of a loop-mediated isothermal amplification reaction for diagnostic applications. FEMS Immunol. Med. Microbiol. 2011, 62, 41–48. [Google Scholar] [CrossRef] [PubMed]
- Galvez, L.C.; Barbosa, C.F.C.; Koh, R.B.L.; Aquino, V.M. Loop-mediated isothermal amplification (LAMP) assays for the detection of abaca bunchy top virus and banana bunchy top virus in abaca. Crop. Prot. 2020, 131, 105101. [Google Scholar] [CrossRef]
- Vivaldi Project, Technical University of Denmark. Available online: www.vivaldi-ia.eu (accessed on 13 March 2020).
- COVID-19 IgM/IgG Rapid Test. Available online: https://www.biomedomics.com/products/infectious-disease/covid-19-rt/ (accessed on 13 March 2020).
- Ahn, S.J.; Baek, Y.H.; Lloren, K.K.S.; Choi, W.-S.; Jeong, J.H.; Antigua, K.J.C.; Kwon, H.-I.; Park, S.-J.; Kim, E.-H.; Kim, Y.-I.; et al. Rapid and simple colorimetric detection of multiple influenza viruses infecting humans using a reverse transcriptional loop-mediated isothermal amplification (RT-LAMP) diagnostic platform. BMC Infect. Dis. 2019, 19, 676. [Google Scholar] [CrossRef] [PubMed]
- Jeon, S.B.; Seo, D.J.; Oh, H.; Kingsley, D.; Choi, C. Development of one-step reverse transcription loop-mediated isothermal amplification for norovirus detection in oysters. Food Control. 2017, 73, 1002–1009. [Google Scholar] [CrossRef]
- Wang, X.; Seo, D.J.; Lee, M.H.; Choi, C.; Onderdonk, A.B. Comparison of Conventional PCR, Multiplex PCR, and Loop-Mediated Isothermal Amplification Assays for Rapid Detection of Arcobacter Species. J. Clin. Microbiol. 2013, 52, 557–563. [Google Scholar] [CrossRef]
- US$675 Million Needed for New Coronavirus Preparedness and Response Global Plan. Available online: https://www.who.int/news-room/detail/05-02-2020-us-675-million-needed-for-new-coronavirus-preparedness-and-response-global-plan (accessed on 13 March 2020).
- Economic Vulnerabilities to the Coronavirus: Top Countries at Risk. Available online: https://www.odi.org/blogs/16639-economic-vulnerabilities-coronavirus-top-countries-risk (accessed on 13 March 2020).
- Wang, C.; Horby, P.W.; Hayden, F.G.; Gao, G.F. A novel coronavirus outbreak of global health concern. Lancet 2020, 395, 470–473. [Google Scholar] [CrossRef]
- Zhou, P.; Yang, X.-L.; Wang, X.-G.; Hu, B.; Zhang, L.; Zhang, W.; Si, H.-R.; Zhu, Y.; Li, B.; Huang, C.-L.; et al. A pneumonia outbreak associated with a new coronavirus of probable bat origin. Nature 2020, 1–4. [Google Scholar] [CrossRef]
- Zhang, L.; Liu, Y. Potential interventions for novel coronavirus in China: A systematic review. J. Med. Virol. 2020. [Google Scholar] [CrossRef] [PubMed]
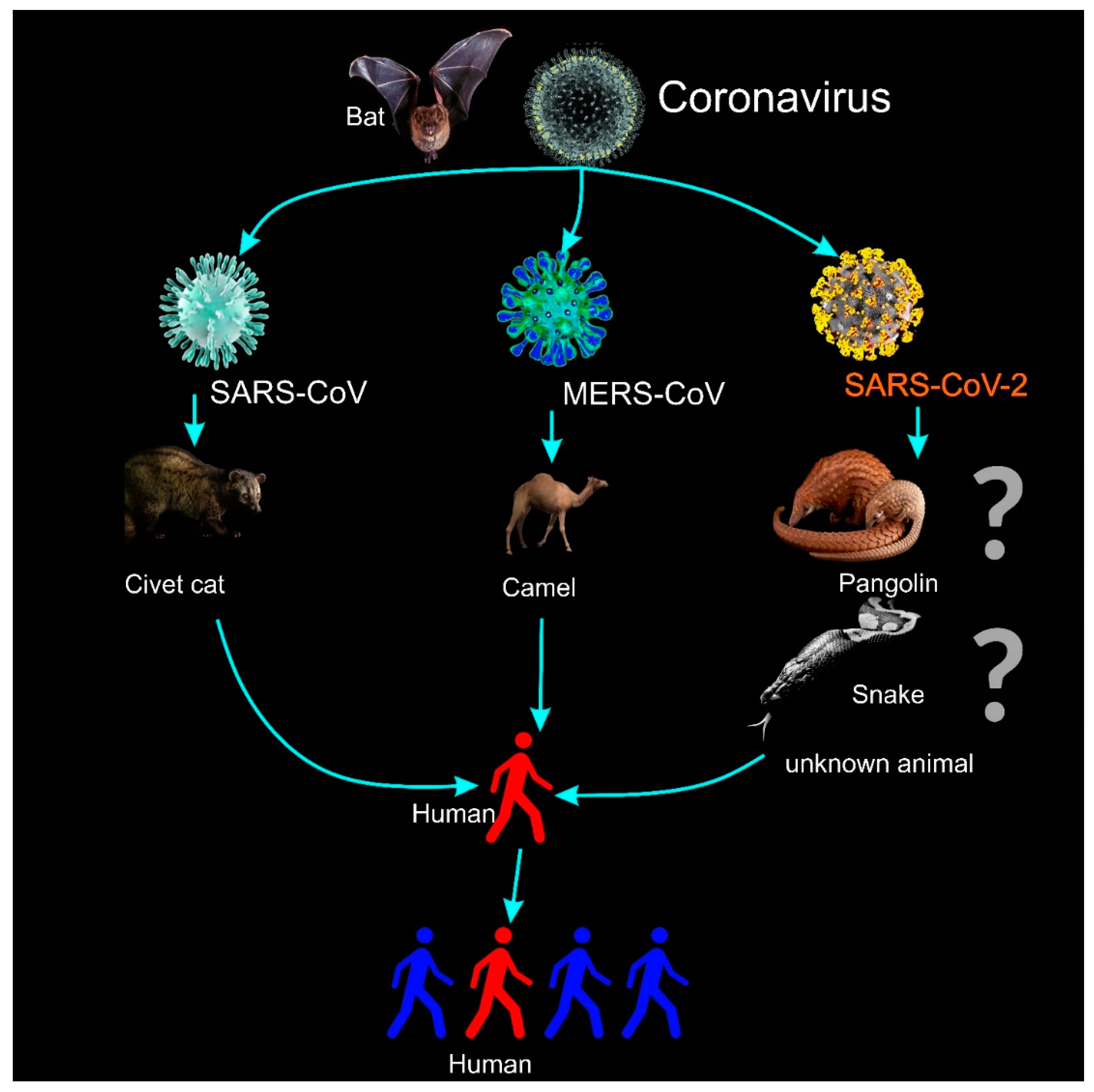
| Virus | Description |
|---|---|
| Coronaviruses (CoVs): | A large and diverse family of enveloped, positive-stranded RNA viruses, with a ~26–32 kilobase genome [16]. The Coronaviridae cover a broad host range, infecting many mammalian and avian species, and induce upper respiratory, gastrointestinal, hepatic, and central nervous system diseases [17]. In the last few decades, coronaviruses have been shown to be capable of also infecting humans. The outbreak of severe acute respiratory syndrome (SARS) in 2003, and, more recently, Middle-East respiratory syndrome (MERS) have proved the lethality of CoVs when they cross the species barrier and infect humans [18]. |
| 2019 novel coronavirus (SARS-CoV-2 [11]): | A new zoonotic human coronavirus, which was reported and announced by the Chinese Center for Disease Control and Prevention (CCDC) on 9 January 2020 [19]. In spite of the fact that the initial infected cases have been associated with the Huanan South China Seafood Market, the source of COVID-19 is still unknown (Figure 1). On 30 January 2020, the WHO declared a global public health emergency regarding the outbreak of COVID-19. On the 11 March 2020, the WHO declared the outbreak of COVID-19 a pandemic. |
| PCR | LAMP |
|---|---|
| Thermal cycling (Multiple heating and cooling cycle; hence, bulky and cumbersome). | Isothermal and continuous amplification (Smaller, simpler, hence portable). |
| Always requires sample concentration and preparation (Time-consuming). | For virus detection, for example, influenza [40] or human norovirus, LAMP assay offers one-step detection [41]. Sample preparation steps are simplified. |
| Multiple protocols (Complicated and requires a skilled technician). | Single protocol (Faster). |
| Inhibitors hinder the reaction. | Tolerate inhibitors and more stable. |
| Diagnostic sensitivity (95%) is currently reported lower than LAMP [33,41,42]. | Diagnostic sensitivity > 95%. |
| Established technique. | Applications using LAMP assays are being explored. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nguyen, T.; Duong Bang, D.; Wolff, A. 2019 Novel Coronavirus Disease (COVID-19): Paving the Road for Rapid Detection and Point-of-Care Diagnostics. Micromachines 2020, 11, 306. https://doi.org/10.3390/mi11030306
Nguyen T, Duong Bang D, Wolff A. 2019 Novel Coronavirus Disease (COVID-19): Paving the Road for Rapid Detection and Point-of-Care Diagnostics. Micromachines. 2020; 11(3):306. https://doi.org/10.3390/mi11030306
Chicago/Turabian StyleNguyen, Trieu, Dang Duong Bang, and Anders Wolff. 2020. "2019 Novel Coronavirus Disease (COVID-19): Paving the Road for Rapid Detection and Point-of-Care Diagnostics" Micromachines 11, no. 3: 306. https://doi.org/10.3390/mi11030306
APA StyleNguyen, T., Duong Bang, D., & Wolff, A. (2020). 2019 Novel Coronavirus Disease (COVID-19): Paving the Road for Rapid Detection and Point-of-Care Diagnostics. Micromachines, 11(3), 306. https://doi.org/10.3390/mi11030306






