Engineering Microneedles for Therapy and Diagnosis: A Survey
Abstract
1. Introduction
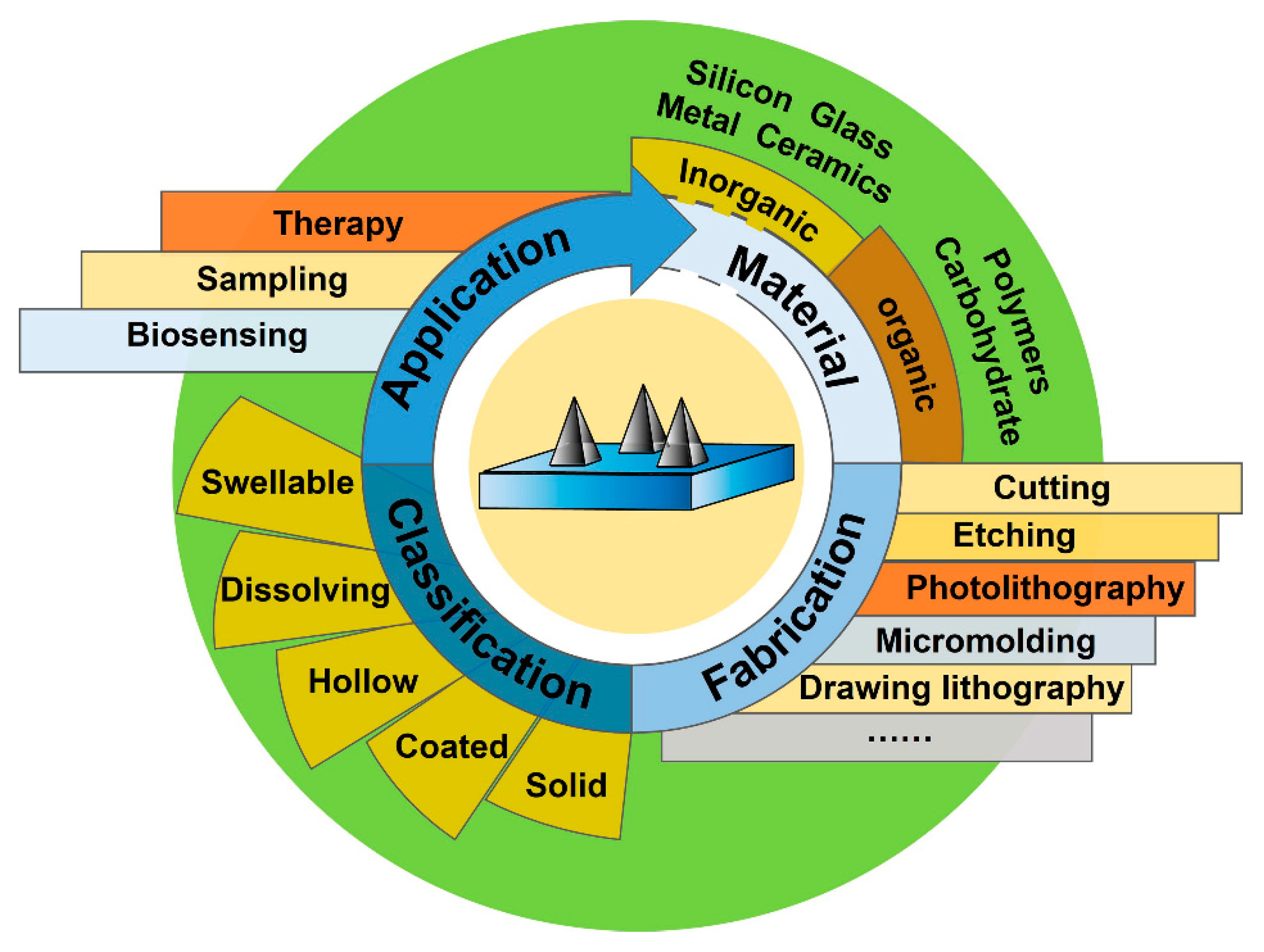
2. Materials and Fabrication Methods
2.1. Materials and Properties
2.1.1. Silicon
2.1.2. Glass
2.1.3. Ceramics
2.1.4. Metals
2.1.5. Polymers
2.1.6. Carbohydrate
2.2. Fabrication Methods of MNs
2.2.1. Cutting
2.2.2. Etching
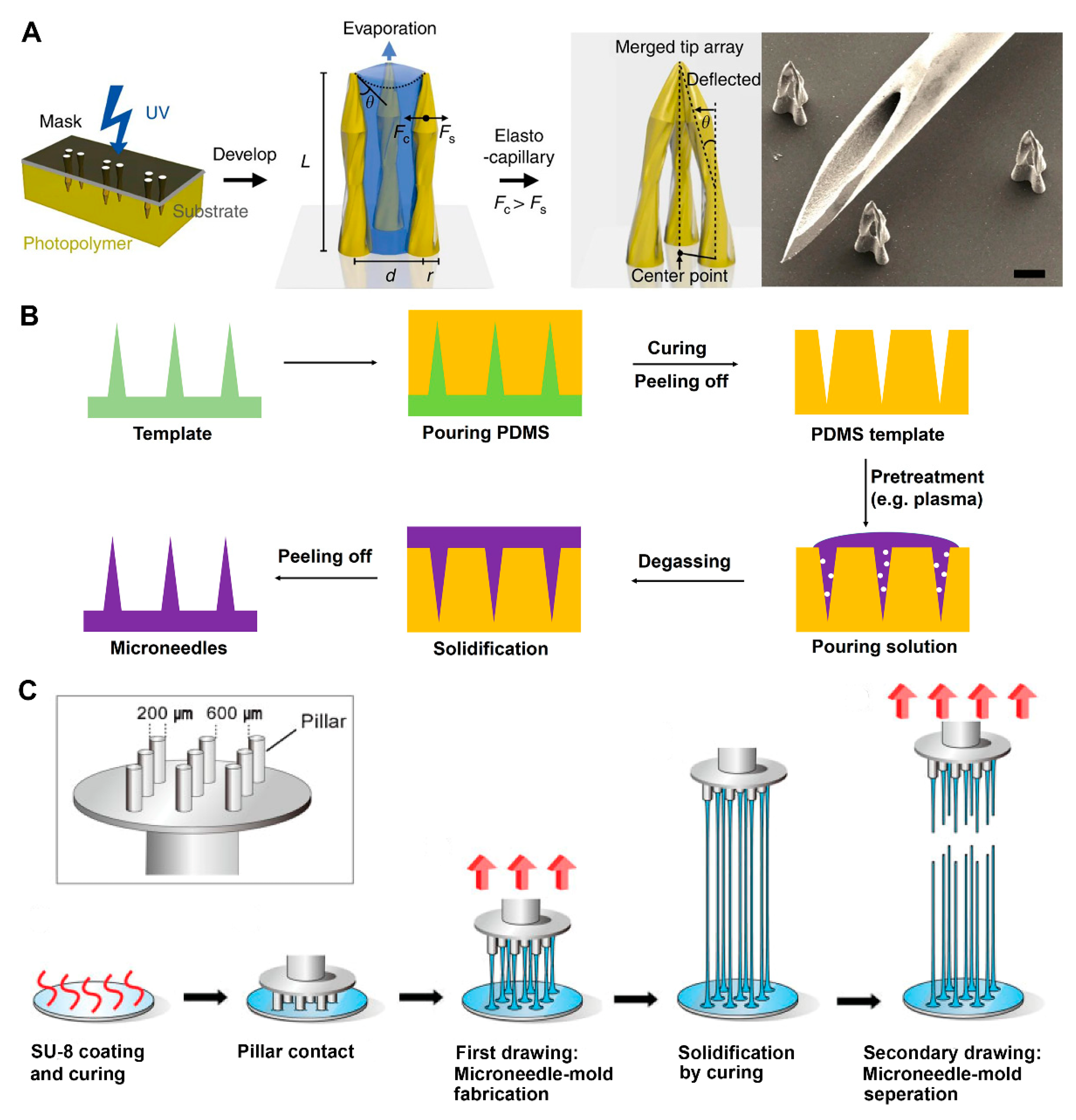
2.2.3. Photolithography
2.2.4. Micromolding
2.2.5. Drawing Lithography
3. Classification of MNs
3.1. Solid MNs
3.2. Coated MNs
3.3. Hollow MNs
3.4. Dissolving MNs
3.5. Swellable MNs
4. Application of Microneedles
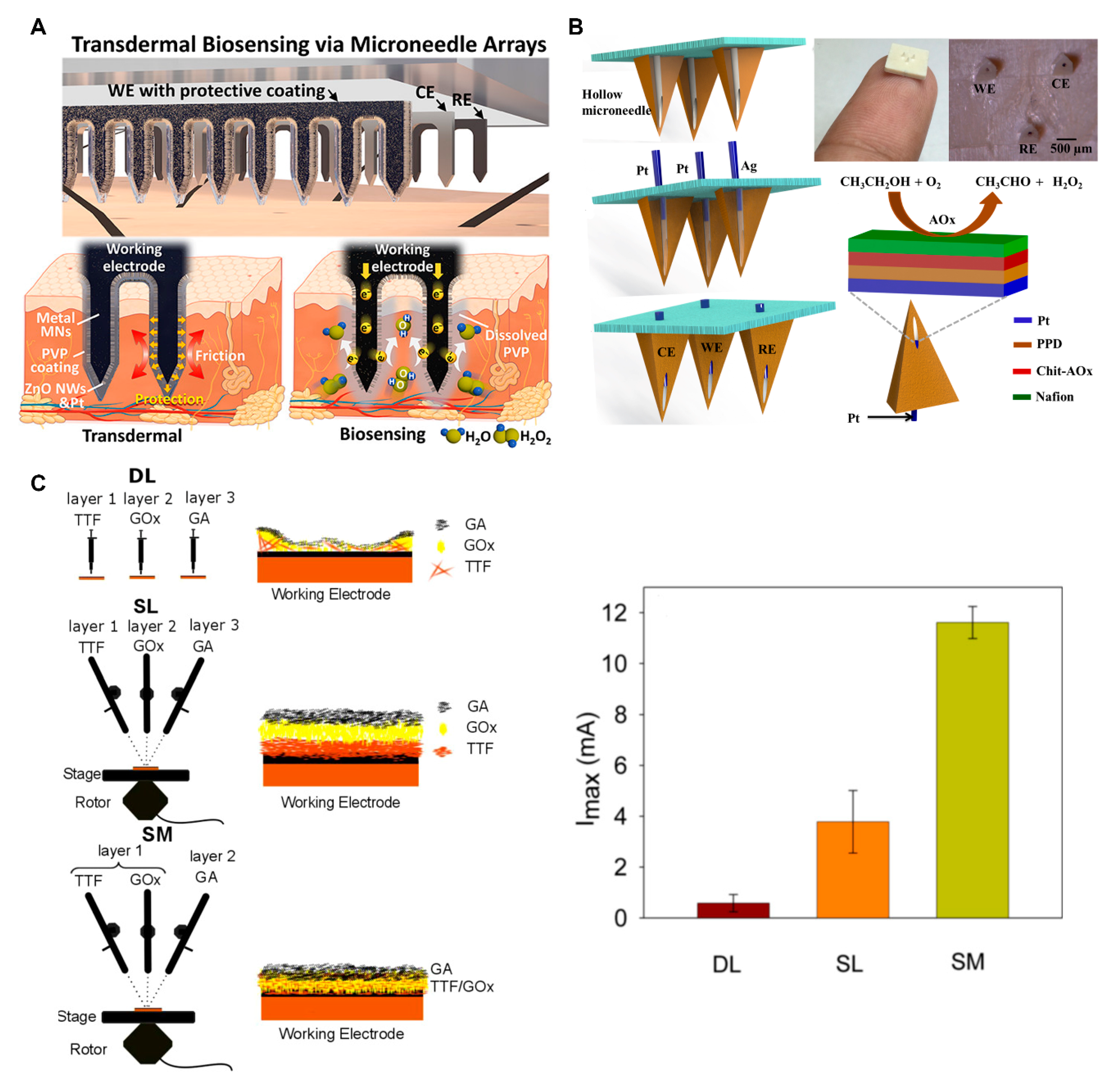
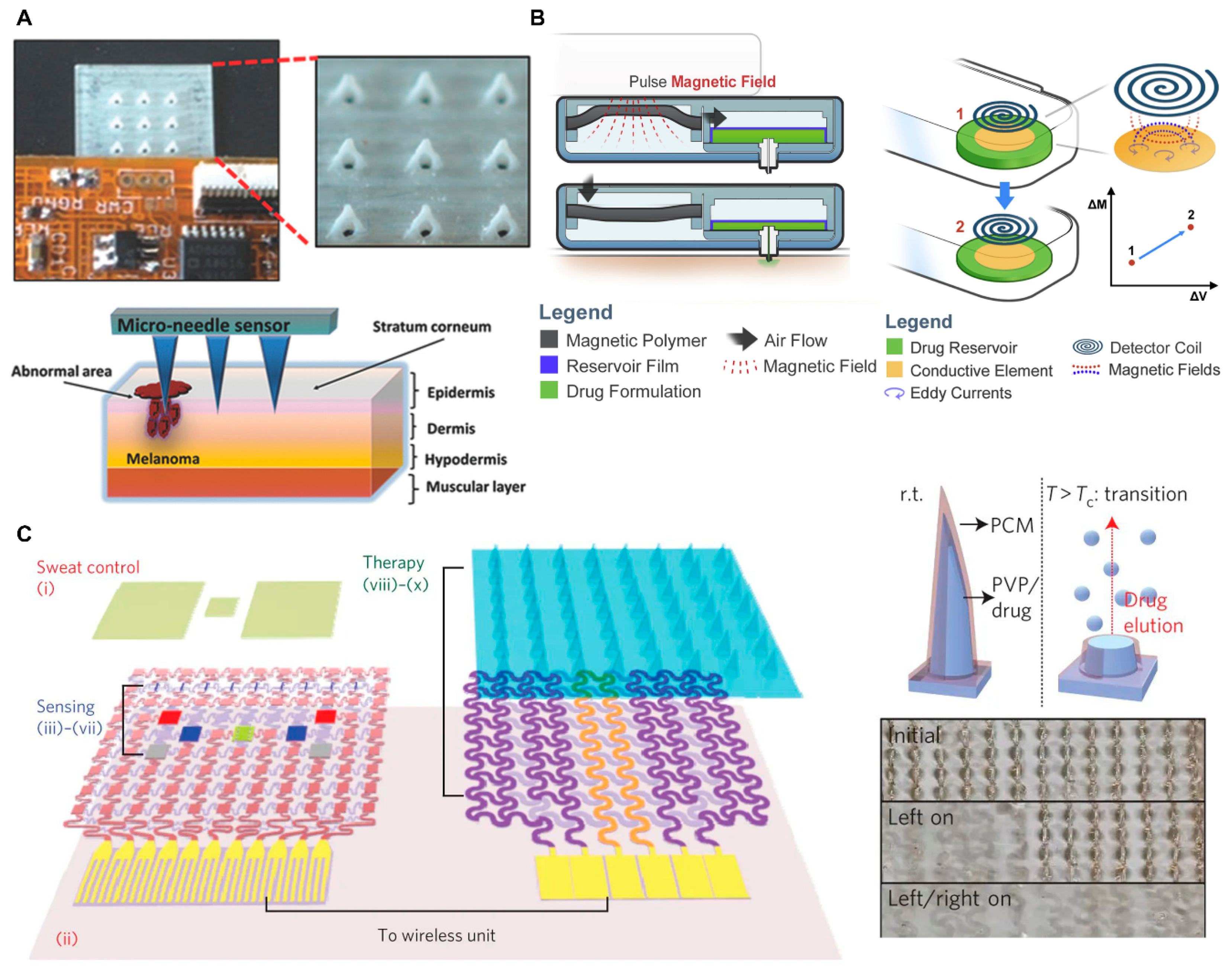
4.1. Transdermal Sensing
4.1.1. Electrochemistry
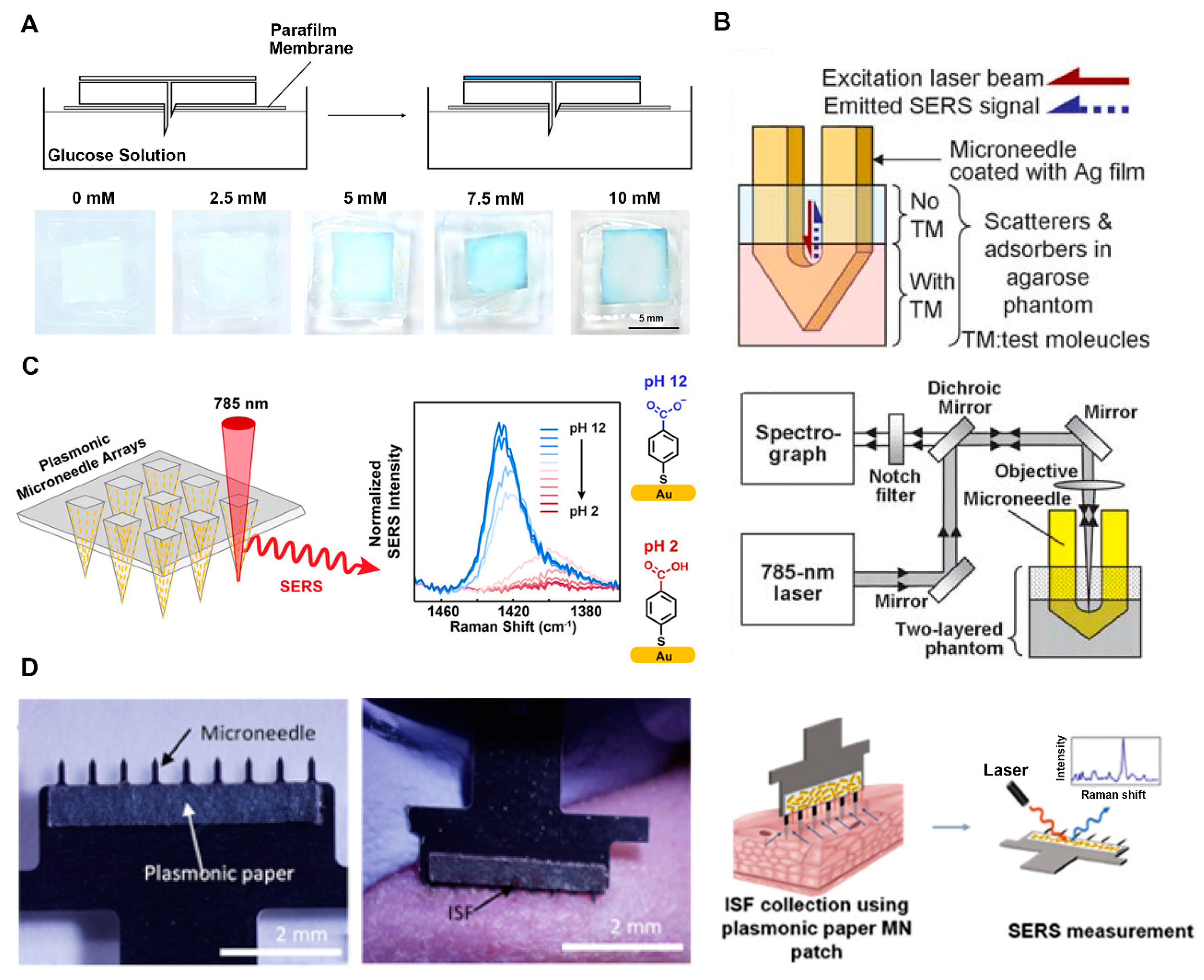
4.1.2. Colorimetric Determination
4.1.3. Surface-Enhanced Raman Scattering
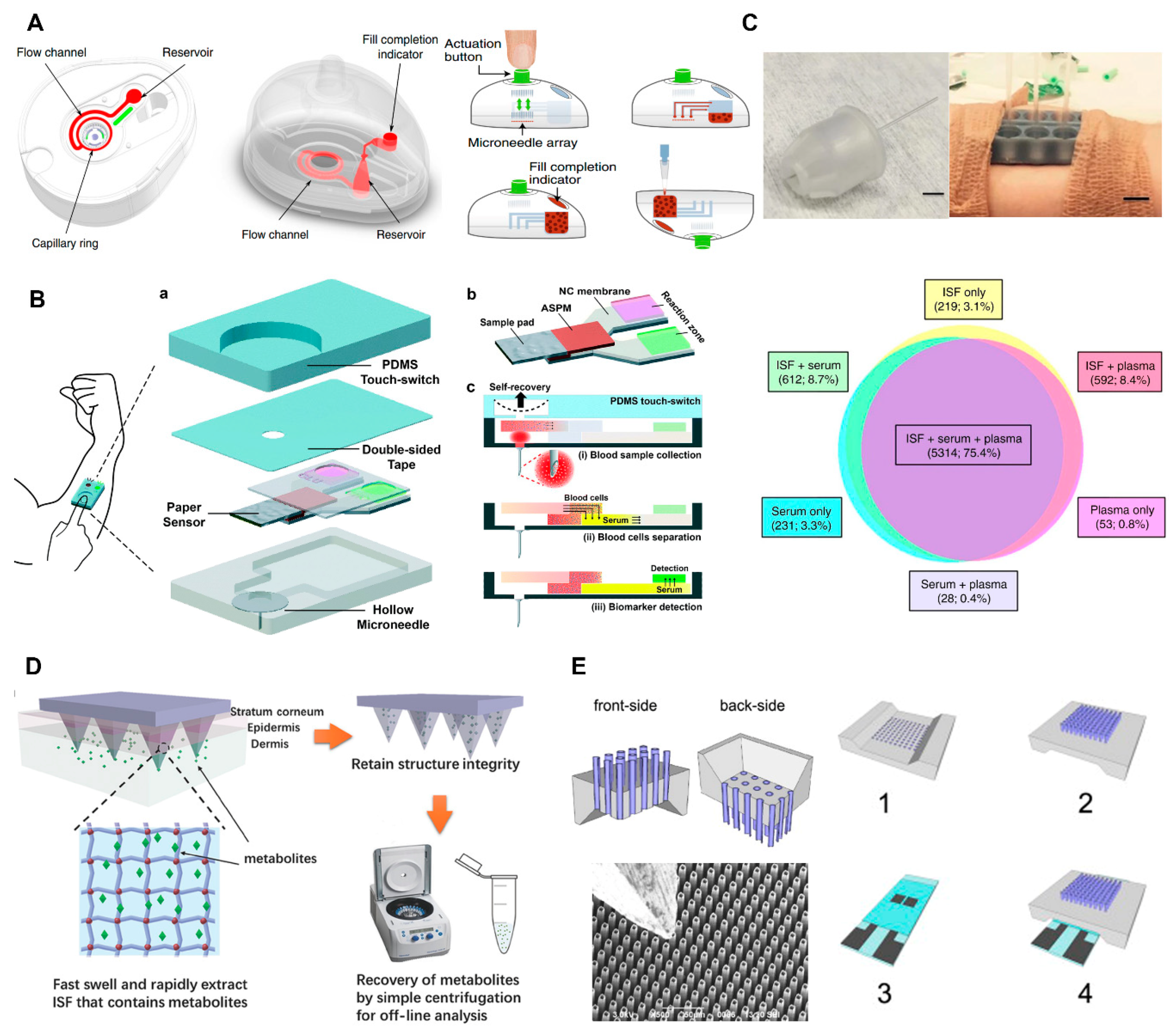
4.2. Biofluids Extraction
4.3. Disease Treatment
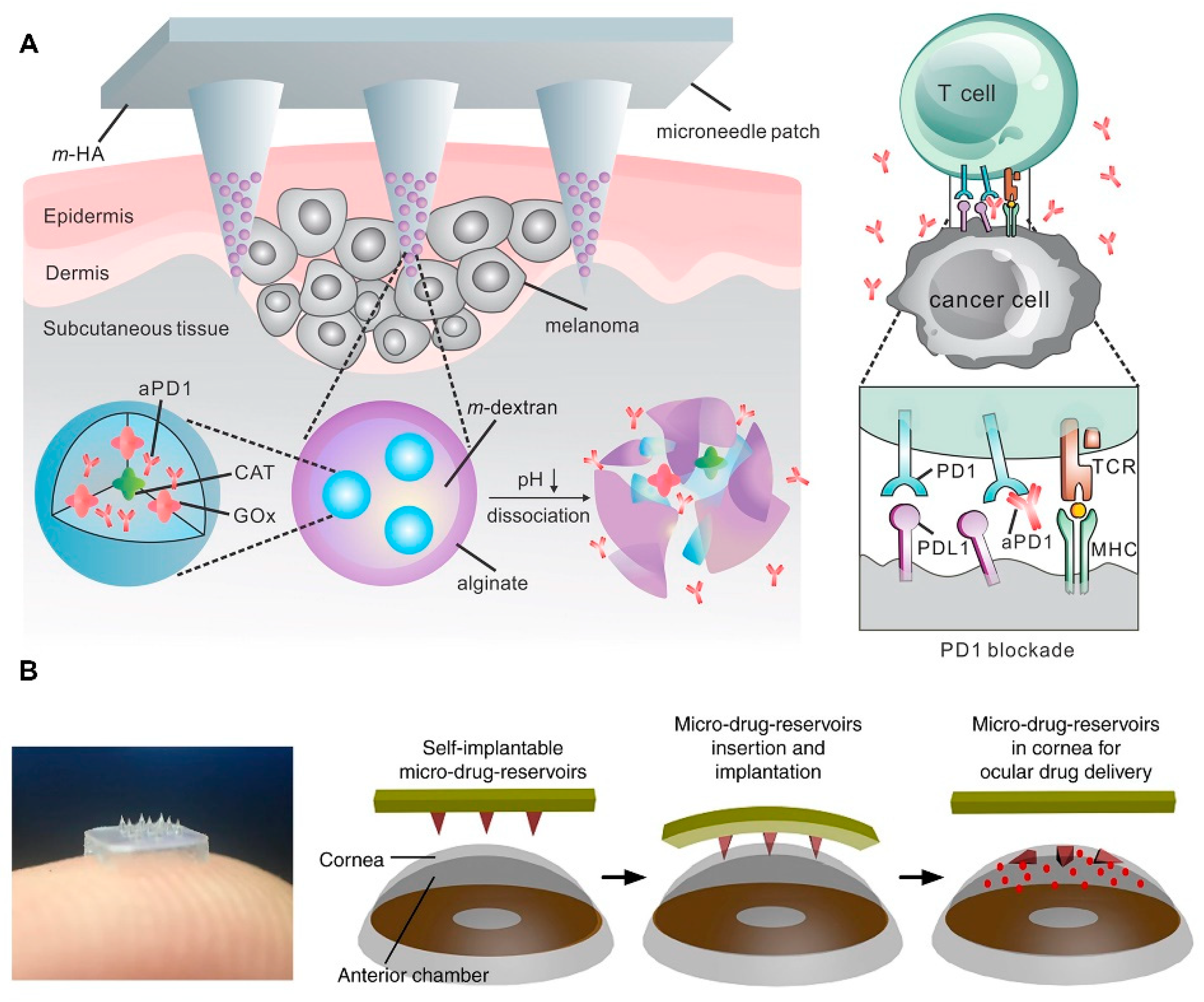
4.3.1. Drug Delivery
4.3.2. Vaccine Delivery
4.4. Microelectronic System Integrated MN Devices for Sensing and Therapy
5. Summary and Outlook
Author Contributions
Funding
Conflicts of Interest
References
- Razmi, N.; Hasanzadeh, M. Current advancement on diagnosis of ovarian cancer using biosensing of CA 125 biomarker: Analytical approaches. TrAC Trends Anal. Chem. 2018, 108, 1–12. [Google Scholar] [CrossRef]
- Shafiee, A.; Ghadiri, E.; Kassis, J.; Pourhabibi Zarandi, N.; Atala, A. Biosensing Technologies for Medical Applications, Manufacturing, and Regenerative Medicine. Curr. Stem Cell Rep. 2018, 4, 105–115. [Google Scholar] [CrossRef]
- Markets and Markets. Wearable Medical Devices Market by Device (Diagnostic (Heart, Pulse, BP, Sleep), Therapeutic (Pain, Insulin, Rehabilitation)), Application (Sport, Fitness, RPM), Type (Smartwatch, Patch), Distribution Channel (Pharmacy, Online) - Global Forecast to 2022. Available online: https://www.marketsandmarkets.com/Market-Reports/wearable-medical-device-market-81753973.html?gclid=EAIaIQobChMIseq6u-345wIVQaaWCh1Iigr2EAAYASAAEgKunPD_BwE (accessed on 1 March 2020).
- Quesada-González, D.; Merkoçi, A. Nanomaterial-based devices for point-of-care diagnostic applications. Chem. Soc. Rev. 2018, 47, 4697–4709. [Google Scholar] [CrossRef] [PubMed]
- Ma, G.; Wu, C. Microneedle, bio-microneedle and bio-inspired microneedle: A review. J. Control Release 2017, 251, 11–23. [Google Scholar] [CrossRef] [PubMed]
- Ekhlaspour, L.; Mondesir, D.; Lautsch, N.; Balliro, C.; Hillard, M.; Magyar, K.; Radocchia, L.G.; Esmaeili, A.; Sinha, M.; Russell, S.J. Comparative Accuracy of 17 Point-of-Care Glucose Meters. J. Diabetes Sci. Technol. 2016, 11, 558–566. [Google Scholar] [CrossRef] [PubMed]
- Moyo, S.; Mohammed, T.; Wirth, K.E.; Prague, M.; Bennett, K.; Holme, M.P.; Mupfumi, L.; Sebogodi, P.; Moraka, N.O.; Boleo, C.; et al. Point-of-Care Cepheid Xpert HIV-1 Viral Load Test in Rural African Communities Is Feasible and Reliable. J. Clin. Microbiol. 2016, 54, 3050. [Google Scholar] [CrossRef] [PubMed]
- Bantle, J.P.; Thomas, W. Glucose measurement in patients with diabetes mellitus with dermal interstitial fluid. J. Lab. Clin. Med. 1997, 130, 436–441. [Google Scholar] [CrossRef]
- Carneiro, G.; Aguiar, M.G.; Fernandes, A.P.; Ferreira, L.A.M. Drug delivery systems for the topical treatment of cutaneous leishmaniasis. Expert. Opin. Drug Del. 2012, 9, 1083–1097. [Google Scholar] [CrossRef]
- Scheuplein, R.J.; Blank, I.H. Permeability of the skin. Physiol. Rev. 1971, 51, 702–747. [Google Scholar] [CrossRef]
- Brull, R.; Hadzic, A.; Reina, M.A.; Barrington, M.J. Pathophysiology and Etiology of Nerve Injury Following Peripheral Nerve Blockade. Region Anesth. Pain. M. 2015, 40, 479–490. [Google Scholar] [CrossRef]
- Sokolowski, C.J., Jr.; Giovannitti, J.A.; Boynes, S.G. Needle Phobia: Etiology, Adverse Consequences, and Patient Management. Dent. Clin. N. Am. 2010, 54, 731–744. [Google Scholar] [CrossRef] [PubMed]
- Taddio, A.; McMurtry, C.M.; Shah, V.; Riddell, R.P.; Chambers, C.T.; Noel, M.; MacDonald, N.E.; Rogers, J.; Bucci, L.M.; Mousmanis, P.; et al. Reducing pain during vaccine injections: clinical practice guideline. CMAJ 2015, 187, 975–982. [Google Scholar] [CrossRef]
- Wang, M.; Hu, L.; Xu, C. Recent advances in the design of polymeric microneedles for transdermal drug delivery and biosensing. Lab Chip 2017, 17, 1373–1387. [Google Scholar] [CrossRef] [PubMed]
- Ventrelli, L.; Marsilio Strambini, L.; Barillaro, G. Microneedles for Transdermal Biosensing: Current Picture and Future Direction. Adv. Healthc. Mater. 2015, 4, 2606–2640. [Google Scholar] [CrossRef] [PubMed]
- Lutton, R.E.M.; Larrañeta, E.; Kearney, M.; Boyd, P.; Woolfson, A.D.; Donnelly, R.F. A novel scalable manufacturing process for the production of hydrogel-forming microneedle arrays. Int. J. Pharmaceut. 2015, 494, 417–429. [Google Scholar] [CrossRef] [PubMed]
- Chang, H.; Zheng, M.; Yu, X.; Than, A.; Seeni, R.Z.; Kang, R.; Tian, J.; Khanh, D.P.; Liu, L.; Chen, P.; et al. A Swellable Microneedle Patch to Rapidly Extract Skin Interstitial Fluid for Timely Metabolic Analysis. Adv. Mater. 2017, 29, 1702243. [Google Scholar] [CrossRef] [PubMed]
- Blicharz, T.M.; Gong, P.; Bunner, B.M.; Chu, L.L.; Leonard, K.M.; Wakefield, J.A.; Williams, R.E.; Dadgar, M.; Tagliabue, C.A.; El Khaja, R.; et al. Microneedle-based device for the one-step painless collection of capillary blood samples. Nat. Biomed.Eng. 2018, 2, 151–157. [Google Scholar] [CrossRef]
- Mani, G.K.; Miyakoda, K.; Saito, A.; Yasoda, Y.; Kajiwara, K.; Kimura, M.; Tsuchiya, K. Microneedle pH Sensor: Direct, Label-Free, Real-Time Detection of Cerebrospinal Fluid and Bladder pH. ACS Appl. Mater. Inter. 2017, 9, 21651–21659. [Google Scholar] [CrossRef]
- Tang, J.; Wang, J.; Huang, K.; Ye, Y.; Su, T.; Qiao, L.; Hensley, M.T.; Caranasos, T.G.; Zhang, J.; Gu, Z.; et al. Cardiac cell–integrated microneedle patch for treating myocardial infarction. Sci. Adv. 2018, 4, t9365. [Google Scholar] [CrossRef]
- Mohan, A.M.V.; Windmiller, J.R.; Mishra, R.K.; Wang, J. Continuous minimally-invasive alcohol monitoring using microneedle sensor arrays. Biosens. Bioelectron. 2017, 91, 574–579. [Google Scholar] [CrossRef]
- Li, J.; Liu, B.; Zhou, Y.; Chen, Z.; Jiang, L.; Yuan, W.; Liang, L. Fabrication of a Ti porous microneedle array by metal injection molding for transdermal drug delivery. PLoS ONE 2017, 12, e172043. [Google Scholar] [CrossRef] [PubMed]
- Petersen, K.E. Silicon as a mechanical material. Proc. IEEE 1982, 70, 420–457. [Google Scholar] [CrossRef]
- McConville, A.; Davis, J. Transdermal microneedle sensor arrays based on palladium: Polymer composites. Electrochem. Commun. 2016, 72, 162–165. [Google Scholar] [CrossRef]
- Chandrasekhar, S.; Iyer, L.K.; Panchal, J.P.; Topp, E.M.; Cannon, J.B.; Ranade, V.V. Microarrays and microneedle arrays for delivery of peptides, proteins, vaccines and other applications. Expert Opin. Drug Del. 2013, 10, 1155–1170. [Google Scholar] [CrossRef]
- Larrañeta, E.; Lutton, R.E.M.; Woolfson, A.D.; Donnelly, R.F. Microneedle arrays as transdermal and intradermal drug delivery systems: Materials science, manufacture and commercial development. Mat. Sci. Eng. R 2016, 104, 1–32. [Google Scholar] [CrossRef]
- Takeuchi, K.; Kim, B. Functionalized microneedles for continuous glucose monitoring. Nano Converg. 2018, 5, 28. [Google Scholar] [CrossRef]
- Liu, G.; Kong, Y.; Wang, Y.; Luo, Y.; Fan, X.; Xie, X.; Yang, B.; Wu, M.X. Microneedles for transdermal diagnostics: Recent advances and new horizons. Biomaterials 2020, 232, 119740. [Google Scholar] [CrossRef]
- Dardano, P.; Rea, I.; De Stefano, L. Microneedles-based electrochemical sensors: New tools for advanced biosensing. Curr. Opin. Electrochem. 2019, 17, 121–127. [Google Scholar] [CrossRef]
- Dharadhar, S.; Majumdar, A.; Dhoble, S.; Patravale, V. Microneedles for transdermal drug delivery: a systematic review. Drug. Dev. Ind. Pharm. 2019, 45, 188–201. [Google Scholar] [CrossRef]
- Sharma, S.; Hatware, K.; Bhadane, P.; Sindhikar, S.; Mishra, D.K. Recent advances in microneedle composites for biomedical applications: Advanced drug delivery technologies. Mat. Sci. Eng. C 2019, 103, 109717. [Google Scholar] [CrossRef]
- Babity, S.; Roohnikan, M.; Brambilla, D. Advances in the Design of Transdermal Microneedles for Diagnostic and Monitoring Applications. Small 2018, 14, 1803186. [Google Scholar] [CrossRef] [PubMed]
- Wang, R.; Wang, W.; Li, Z. An Improved Manufacturing Approach for Discrete Silicon Microneedle Arrays with Tunable Height-Pitch Ratio. Sensors 2016, 16, 1628. [Google Scholar] [CrossRef] [PubMed]
- Strambini, L.M.; Longo, A.; Scarano, S.; Prescimone, T.; Palchetti, I.; Minunni, M.; Giannessi, D.; Barillaro, G. Self-powered microneedle-based biosensors for pain-free high-accuracy measurement of glycaemia in interstitial fluid. Biosens. Bioelectron. 2015, 66, 162–168. [Google Scholar] [CrossRef]
- Das, A.; Singha, C.; Bhattacharyya, A. Development of silicon microneedle arrays with spontaneously generated micro-cavity ring for transdermal drug delivery. Microelectron. Eng. 2019, 210, 14–18. [Google Scholar] [CrossRef]
- Martanto, W.; Moore, J.S.; Kashlan, O.; Kamath, R.; Wang, P.M.; O’Neal, J.M.; Prausnitz, M.R. Microinfusion Using Hollow Microneedles. Pharm. Res. 2006, 23, 104–113. [Google Scholar] [CrossRef] [PubMed]
- Bhatnagar, S.; Gadeela, P.R.; Thathireddy, P.; Venuganti, V.V.K. Microneedle-based drug delivery: Materials of construction. J. Chem. Sci. 2019, 131, 90. [Google Scholar] [CrossRef]
- Larrañeta, E.; Singh, T.R.R. Microneedle Manufacturing and Testing. In Microneedles for Drug and Vaccine Delivery and Patient Monitoring; John Wiley & Sons: Hoboken, NJ, USA, 2018; pp. 21–70. [Google Scholar]
- Verhoeven, M.; Bystrova, S.; Winnubst, L.; Qureshi, H.; de Gruijl, T.D.; Scheper, R.J.; Luttge, R. Applying ceramic nanoporous microneedle arrays as a transport interface in egg plants and an ex-vivo human skin model. Microelectron. Eng. 2012, 98, 659–662. [Google Scholar] [CrossRef]
- van der Maaden, K.; Luttge, R.; Vos, P.J.; Bouwstra, J.; Kersten, G.; Ploemen, I. Microneedle-based drug and vaccine delivery via nanoporous microneedle arrays. Drug. Deliv. Transl. Res. 2015, 5, 397–406. [Google Scholar] [CrossRef]
- Schepens, B.; Vos, P.J.; Saelens, X.; van der Maaden, K. Vaccination with influenza hemagglutinin-loaded ceramic nanoporous microneedle arrays induces protective immune responses. Eur. J. Pharm. Biopharm. 2019, 136, 259–266. [Google Scholar] [CrossRef]
- Cahill, E.M.; Keaveney, S.; Stuettgen, V.; Eberts, P.; Ramos-Luna, P.; Zhang, N.; Dangol, M.; O’Cearbhaill, E.D. Metallic microneedles with interconnected porosity: A scalable platform for biosensing and drug delivery. Acta. Biomater. 2018, 80, 401–411. [Google Scholar] [CrossRef]
- Wang, R.; Jiang, X.; Wang, W.; Li, Z. A microneedle electrode array on flexible substrate for long-term EEG monitoring. Sens. Actuators B Chem. 2017, 244, 750–758. [Google Scholar] [CrossRef]
- Liu, F.; Lin, Z.; Jin, Q.; Wu, Q.; Yang, C.; Chen, H.; Cao, Z.; Lin, D.; Zhou, L.; Hang, T.; et al. Protection of Nanostructures-Integrated Microneedle Biosensor Using Dissolvable Polymer Coating. ACS Appl. Mater. Inter. 2019, 11, 4809–4819. [Google Scholar] [CrossRef] [PubMed]
- Andar, A.U.; Karan, R.; Pecher, W.T.; DasSarma, P.; Hedrich, W.D.; Stinchcomb, A.L.; DasSarma, S. Microneedle-Assisted Skin Permeation by Nontoxic Bioengineerable Gas Vesicle Nanoparticles. Mol. Pharm. 2017, 14, 953–958. [Google Scholar] [CrossRef] [PubMed]
- Vinayakumar, K.B.; Kulkarni, P.G.; Nayak, M.M.; Dinesh, N.S.; Hegde, G.M.; Ramachandra, S.G.; Rajanna, K. A hollow stainless steel microneedle array to deliver insulin to a diabetic rat. J. Micromech. Microeng. 2016, 26, 65013. [Google Scholar] [CrossRef]
- Sullivan, S.P.; Koutsonanos, D.G.; Del Pilar Martin, M.; Lee, J.W.; Zarnitsyn, V.; Choi, S.; Murthy, N.; Compans, R.W.; Skountzou, I.; Prausnitz, M.R. Dissolving polymer microneedle patches for influenza vaccination. Nat. Med. 2010, 16, 915–920. [Google Scholar] [CrossRef]
- Yun, S.; Lee, S.; Giri, S.S.; Kim, H.J.; Kim, S.G.; Kim, S.W.; Han, S.J.; Kwon, J.; Oh, W.T.; Chang Park, S. Vaccination of fish against Aeromonas hydrophila infections using the novel approach of transcutaneous immunization with dissolving microneedle patches in aquaculture. Fish Shellfish Immun. 2020, 97, 34–40. [Google Scholar] [CrossRef]
- Trautmann, A.; Roth, G.; Nujiqi, B.; Walther, T.; Hellmann, R. Towards a versatile point-of-care system combining femtosecond laser generated microfluidic channels and direct laser written microneedle arrays. Microsyst. Nanoeng. 2019, 5, 6. [Google Scholar] [CrossRef]
- Xiang, Z.; Wang, H.; Murugappan, S.K.; Yen, S.; Pastorin, G.; Lee, C. Dense vertical SU-8 microneedles drawn from a heated mold with precisely controlled volume. J. Micromech. Microeng. 2015, 25, 25013. [Google Scholar] [CrossRef]
- Lee, K.; Lee, C.Y.; Jung, H. Dissolving microneedles for transdermal drug administration prepared by stepwise controlled drawing of maltose. Biomaterials 2011, 32, 3134–3140. [Google Scholar] [CrossRef]
- Martin, C.J.; Allender, C.J.; Brain, K.R.; Morrissey, A.; Birchall, J.C. Low temperature fabrication of biodegradable sugar glass microneedles for transdermal drug delivery applications. J. Control. Release 2012, 158, 93–101. [Google Scholar] [CrossRef]
- Li, J.; Song, E.; Chiang, C.; Yu, K.J.; Koo, J.; Du, H.; Zhong, Y.; Hill, M.; Wang, C.; Zhang, J.; et al. Conductively coupled flexible silicon electronic systems for chronic neural electrophysiology. Proc. Natl. Acad. Sci. 2018, 115, E9542. [Google Scholar] [CrossRef] [PubMed]
- Ryhänen, T.; Pohjonen, H. Overview - Impact of Silicon MEMS—40 Years After. In Handbook of Silicon Based MEMS Materials and Technologies, 2nd ed.; Tilli, M., Motooka, T., Airaksinen, V., Franssila, S., Paulasto-Kröckel, M., Lindroos, V., Eds.; William Andrew Publishing: Boston, MA, USA, 2015; pp. xix–xxxvii. [Google Scholar]
- Kolli, C.S. Microneedles: bench to bedside. Ther. Deliv. 2015, 6, 1081–1088. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.; Park, J.; Prausnitz, M.R. Microneedles for drug and vaccine delivery. Adv. Drug Deliver. Rev. 2012, 64, 1547–1568. [Google Scholar] [CrossRef] [PubMed]
- Shaun, D.; Gittard, R.J.N. Applications of Microneedle Technology to Transdermal Drug Delivery. In Toxicology of the Skin; CRC Press: Boca Raton, FL, USA, 2010; pp. 301–317. [Google Scholar]
- Boks, M.A.; Unger, W.W.J.; Engels, S.; Ambrosini, M.; Kooyk, Y.V.; Luttge, R. Controlled release of a model vaccine by nanoporous ceramic microneedle arrays. Int. J. Pharm. 2015, 491, 375–383. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Zhou, Y.; Yang, J.; Ye, R.; Gao, J.; Ren, L.; Liu, B.; Liang, L.; Jiang, L. Fabrication of gradient porous microneedle array by modified hot embossing for transdermal drug delivery. Mater. Sci. Eng. C 2019, 96, 576–582. [Google Scholar] [CrossRef] [PubMed]
- Bollella, P.; Sharma, S.; Cass, A.E.G.; Tasca, F.; Antiochia, R. Minimally Invasive Glucose Monitoring Using a Highly Porous Gold Microneedles-Based Biosensor: Characterization and Application in Artificial Interstitial Fluid. Catalysts 2019, 9, 580. [Google Scholar] [CrossRef]
- Gholami, S.; Mohebi, M.; Hajizadeh-Saffar, E.; Ghanian, M.; Zarkesh, I.; Baharvand, H. Fabrication of microporous inorganic microneedles by centrifugal casting method for transdermal extraction and delivery. Int. J. Pharm. 2019, 558, 299–310. [Google Scholar] [CrossRef]
- Vallhov, H.; Xia, W.; Engqvist, H.; Scheynius, A. Bioceramic microneedle arrays are able to deliver OVA to dendritic cells in human skin. J. Mater. Chem. B 2018, 6, 6808–6816. [Google Scholar] [CrossRef]
- Cai, B.; Xia, W.; Bredenberg, S.; Li, H.; Engqvist, H. Bioceramic microneedles with flexible and self-swelling substrate. Eur. J. Pharm. Biopharm. 2015, 94, 404–410. [Google Scholar] [CrossRef]
- Ranamukhaarachchi, S.A.; Padeste, C.; Dübner, M.; Häfeli, U.O.; Stoeber, B.; Cadarso, V.J. Integrated hollow microneedle-optofluidic biosensor for therapeutic drug monitoring in sub-nanoliter volumes. Sci. Rep. 2016, 6, 29075. [Google Scholar] [CrossRef]
- Chen, M.; Huang, S.; Lai, K.; Ling, M. Fully embeddable chitosan microneedles as a sustained release depot for intradermal vaccination. Biomaterials 2013, 34, 3077–3086. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, M.; Goebeler, M. Immunology of metal allergies. JDDG J. Dtsch. Dermatol. Ges. 2015, 13, 653–659. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, E.M. Hydrogel: Preparation, characterization, and applications: A review. J. Adv. Res. 2015, 6, 105–121. [Google Scholar] [CrossRef] [PubMed]
- Yu, W.; Jiang, G.; Liu, D.; Li, L.; Tong, Z.; Yao, J.; Kong, X. Transdermal delivery of insulin with bioceramic composite microneedles fabricated by gelatin and hydroxyapatite. Mater. Sci. Eng. C 2017, 73, 425–428. [Google Scholar] [CrossRef] [PubMed]
- Donnelly, R.F.; Morrow, D.I.J.; Singh, T.R.R.; Migalska, K.; McCarron, P.A.; O’Mahony, C.; Woolfson, A.D. Processing difficulties and instability of carbohydrate microneedle arrays. Drug Deliv. Ind. Pharm. 2009, 35, 1242–1254. [Google Scholar] [CrossRef] [PubMed]
- Loizidou, E.Z.; Williams, N.A.; Barrow, D.A.; Eaton, M.J.; McCrory, J.; Evans, S.L.; Allender, C.J. Structural characterisation and transdermal delivery studies on sugar microneedles: Experimental and finite element modelling analyses. Eur. J. Pharm. Biopharm. 2015, 89, 224–231. [Google Scholar] [CrossRef]
- Waghule, T.; Singhvi, G.; Dubey, S.K.; Pandey, M.M.; Gupta, G.; Singh, M.; Dua, K. Microneedles: A smart approach and increasing potential for transdermal drug delivery system. Biomed. Pharmacother. 2019, 109, 1249–1258. [Google Scholar] [CrossRef]
- Vinayakumar, K.B.; Hegde, G.M.; Ramachandra, S.G.; Nayak, M.M.; Dinesh, N.S.; Rajanna, K. Development of cup shaped microneedle array for transdermal drug delivery. Biointerphases 2015, 10, 21008. [Google Scholar] [CrossRef]
- Bystrova, S.; Luttge, R. Micromolding for ceramic microneedle arrays. Microelectron. Eng. 2011, 88, 1681–1684. [Google Scholar] [CrossRef]
- Li, Z.; Yan, X.; Hu, M.; Tang, G.; Li, Z. The Study on Fabrication of Solid Metal Microneedles Based on Optimized Process of Electrochemical Etching and Cutting. In Proceedings of the 2018 IEEE International Conference of Intelligent Robotic and Control Engineering (IRCE), Lanzhou, China, 24–27 August 2018; pp. 56–59. [Google Scholar]
- García-López, E.; Siller, H.R.; Rodríguez, C.A. Study of the fabrication of AISI 316L microneedle arrays. Procedia Manuf. 2018, 26, 117–124. [Google Scholar] [CrossRef]
- Ahsanulhaq, Q.; Kim, J.H.; Hahn, Y. Etch-free selective area growth of well-aligned ZnO nanorod arrays by economical polymer mask for large-area solar cell applications. Sol. Energ. Mat. Sol. C 2012, 98, 476–481. [Google Scholar] [CrossRef]
- Kim, J.; Yoon, Y.; Allen, M.G. Computer numerical control (CNC) lithography: light-motion synchronized UV-LED lithography for 3D microfabrication. J. Micromech. Microeng. 2016, 26, 35003. [Google Scholar] [CrossRef]
- Williams, C.; Gordon, G.S.D.; Wilkinson, T.D.; Bohndiek, S.E. Grayscale-to-Color: Scalable Fabrication of Custom Multispectral Filter Arrays. ACS Photonics 2019, 6, 3132–3141. [Google Scholar] [CrossRef] [PubMed]
- Benoit, R.R.; Jordan, D.M.; Smith, G.L.; Polcawich, R.G.; Bedair, S.S.; Potrepka, D.M. Direct-Write Laser Grayscale Lithography for Multilayer Lead Zirconate Titanate Thin Films. IEEE Trans. Ultrason. Ferroelectr. Freq. Control 2018, 65, 889–894. [Google Scholar] [CrossRef] [PubMed]
- Kathuria, H.; Kochhar, J.S.; Fong, M.H.M.; Hashimoto, M.; Iliescu, C.; Yu, H.; Kang, L. Polymeric microneedle array fabrication by photolithography. JoVE 2015, e52914. [Google Scholar] [CrossRef]
- Lee, I.C.; He, J.; Tsai, M.; Lin, K. Fabrication of a novel partially dissolving polymer microneedle patch for transdermal drug delivery. J. Mater. Chem. B 2015, 3, 276–285. [Google Scholar] [CrossRef]
- González García, L.E.; MacGregor, M.N.; Visalakshan, R.M.; Ninan, N.; Cavallaro, A.A.; Trinidad, A.D.; Zhao, Y.; Hayball, A.J.D.; Vasilev, K. Self-sterilizing antibacterial silver-loaded microneedles. Chem. Commun. 2019, 55, 171–174. [Google Scholar] [CrossRef]
- Yang, S.; Feng, Y.; Zhang, L.; Chen, N.; Yuan, W.; Jin, T. A scalable fabrication process of polymer microneedles. Int. J. Nanomed. 2012, 7, 1415–1422. [Google Scholar] [CrossRef]
- Kathuria, H.; Kang, K.; Cai, J.; Kang, L. Rapid Microneedle Fabrication by Heating and Photolithography. Int. J. Pharm. 2019, 118992. [Google Scholar] [CrossRef]
- Zhou, J.; Ochoa, M.; Samaddar, S.; Rahimi, R.; Badwaik, V.D.; Thompson, D.H.; Ziaie, B. A rapid micro-molding process for fabricating polymeric biodegradable 3D structures using hydrophobic elastomeric molds. In Proceedings of the 2017 IEEE 30th International Conference on Micro Electro Mechanical Systems (MEMS), Las Vegas, NV, USA, 22–26 January 2017; pp. 422–425. [Google Scholar]
- Chen, H.; Wu, B.; Zhang, M.; Yang, P.; Yang, B.; Qin, W.; Wang, Q.; Wen, X.; Chen, M.; Quan, G.; et al. A novel scalable fabrication process for the production of dissolving microneedle arrays. Drug Deliv. Transl. Res. 2019, 9, 240–248. [Google Scholar] [CrossRef]
- McGrath, M.G.; Vucen, S.; Vrdoljak, A.; Kelly, A.; O Mahony, C.; Crean, A.M.; Moore, A. Production of dissolvable microneedles using an atomised spray process: Effect of microneedle composition on skin penetration. Eur. J. Pharm. Biopharm. 2014, 86, 200–211. [Google Scholar] [CrossRef]
- Lee, K.; Lee, H.C.; Lee, D.; Jung, H. Drawing Lithography: Three-Dimensional Fabrication of an Ultrahigh-Aspect-Ratio Microneedle. Adv. Mater. 2010, 22, 483–486. [Google Scholar] [CrossRef] [PubMed]
- Lee, K.; Jung, H. Drawing lithography for microneedles: A review of fundamentals and biomedical applications. Biomaterials 2012, 33, 7309–7326. [Google Scholar] [CrossRef] [PubMed]
- Chen, Z.; Ren, L.; Li, J.; Yao, L.; Chen, Y.; Liu, B.; Jiang, L. Rapid fabrication of microneedles using magnetorheological drawing lithography. Acta. Biomater. 2018, 65, 283–291. [Google Scholar] [CrossRef]
- Lim, J.; Tahk, D.; Yu, J.; Min, D.; Jeon, N.L. Design rules for a tunable merged-tip microneedle. Microsyst. Nanoeng. 2018, 4, 29. [Google Scholar] [CrossRef] [PubMed]
- Zhou, C.; Liu, Y.; Wang, H.; Zhang, P.; Zhang, J. Transdermal delivery of insulin using microneedle rollers in vivo. Int. J. Pharm. 2010, 392, 127–133. [Google Scholar] [CrossRef]
- Gupta, J.; Gill, H.S.; Andrews, S.N.; Prausnitz, M.R. Kinetics of skin resealing after insertion of microneedles in human subjects. J. Control. Release 2011, 154, 148–155. [Google Scholar] [CrossRef]
- Li, S.; Li, W.; Prausnitz, M. Individually coated microneedles for co-delivery of multiple compounds with different properties. Drug Deliv. Transl. Res. 2018, 8, 1043–1052. [Google Scholar] [CrossRef]
- Chinnadayyala, S.R.; Park, I.; Cho, S. Nonenzymatic determination of glucose at near neutral pH values based on the use of nafion and platinum black coated microneedle electrode array. Microchim. Acta. 2018, 185, 250. [Google Scholar] [CrossRef]
- Rawson, T.M.; Sharma, S.; Georgiou, P.; Holmes, A.; Cass, A.; O’Hare, D. Towards a minimally invasive device for beta-lactam monitoring in humans. Electrochem. Commun. 2017, 82, 1–5. [Google Scholar] [CrossRef]
- Zhao, X.; Birchall, J.C.; Coulman, S.A.; Tatovic, D.; Singh, R.K.; Wen, L.; Wong, F.S.; Dayan, C.M.; Hanna, S.J. Microneedle delivery of autoantigen for immunotherapy in type 1 diabetes. J. Control. Release 2016, 223, 178–187. [Google Scholar] [CrossRef] [PubMed]
- Pradeep Narayanan, S.; Raghavan, S. Fabrication and characterization of gold-coated solid silicon microneedles with improved biocompatibility. Int. J. Adv. Manuf. Technol. 2019, 104, 3327–3333. [Google Scholar] [CrossRef]
- van der Maaden, K.; Heuts, J.; Camps, M.; Pontier, M.; Terwisscha Van Scheltinga, A.; Jiskoot, W.; Ossendorp, F.; Bouwstra, J. Hollow microneedle-mediated micro-injections of a liposomal HPV E743–63 synthetic long peptide vaccine for efficient induction of cytotoxic and T-helper responses. J. Control. Release 2018, 269, 347–354. [Google Scholar] [CrossRef]
- Au, S.H. Dissolving microneedle vaccination—No magic needled. Sci. Transl. Med. 2018, 10, v343. [Google Scholar] [CrossRef]
- Zhan, H.; Ma, F.; Huang, Y.; Zhang, J.; Jiang, X.; Qian, Y. Application of composite dissolving microneedles with high drug loading ratio for rapid local anesthesia. Eur. J. Pharm. Sci. 2018, 121, 330–337. [Google Scholar] [CrossRef] [PubMed]
- Luo, Z.; Sun, W.; Fang, J.; Lee, K.; Li, S.; Gu, Z.; Dokmeci, M.R.; Khademhosseini, A. Biodegradable Gelatin Methacryloyl Microneedles for Transdermal Drug Delivery. Adv. Healthc. Mater. 2019, 8, 1801054. [Google Scholar] [CrossRef]
- Wang, Q.; Yao, G.; Dong, P.; Gong, Z.; Li, G.; Zhang, K.; Wu, C. Investigation on fabrication process of dissolving microneedle arrays to improve effective needle drug distribution. Eur. J. Pharm. Sci. 2015, 66, 148–156. [Google Scholar] [CrossRef]
- Lee, J.W.; Park, J.; Prausnitz, M.R. Dissolving microneedles for transdermal drug delivery. Biomaterials 2008, 29, 2113–2124. [Google Scholar] [CrossRef]
- Li, W.; Tang, J.; Terry, R.N.; Li, S.; Brunie, A.; Callahan, R.L.; Noel, R.K.; Rodríguez, C.A.; Schwendeman, S.P.; Prausnitz, M.R. Long-acting reversible contraception by effervescent microneedle patch. Sci. Adv. 2019, 5, w8145. [Google Scholar] [CrossRef]
- Cahill, E.M.; O Cearbhaill, E.D. Toward Biofunctional Microneedles for Stimulus Responsive Drug Delivery. Bioconjug. Chem. 2015, 26, 1289–1296. [Google Scholar] [CrossRef]
- Liu, S.; Yeo, D.C.; Wiraja, C.; Tey, H.L.; Mrksich, M.; Xu, C. Peptide delivery with poly(ethylene glycol) diacrylate microneedles through swelling effect. Bioeng. Transl. Med. 2017, 2, 258–267. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.; Jung, B.; Park, J. Hydrogel swelling as a trigger to release biodegradable polymer microneedles in skin. Biomaterials 2012, 33, 668–678. [Google Scholar] [CrossRef] [PubMed]
- Romanyuk, A.V.; Zvezdin, V.N.; Samant, P.; Grenader, M.I.; Zemlyanova, M.; Prausnitz, M.R. Collection of Analytes from Microneedle Patches. Anal. Chem. 2014, 86, 10520–10523. [Google Scholar] [CrossRef] [PubMed]
- Cobelli, C.; Schiavon, M.; Dalla Man, C.; Basu, A.; Basu, R. Interstitial Fluid Glucose Is Not Just a Shifted-in-Time but a Distorted Mirror of Blood Glucose: Insight from an In Silico Study. Diabetes Technol. Ther. 2016, 18, 505–511. [Google Scholar] [CrossRef]
- Reid, M.E.; Mallinson, G.; Sim, R.B.; Poole, J.; Pausch, V.; Merry, A.H.; Liew, Y.W.; Tanner, M.J. Biochemical studies on red blood cells from a patient with the Inab phenotype (decay-accelerating factor deficiency). Blood 1991, 78, 3291–3297. [Google Scholar] [CrossRef]
- Bollella, P.; Sharma, S.; Cass, A.E.G.; Antiochia, R. Microneedle-based biosensor for minimally-invasive lactate detection. Biosens. Bioelectron. 2019, 123, 152–159. [Google Scholar] [CrossRef]
- Yoon, H.S.; Lee, S.J.; Park, J.Y.; Paik, S.; Allen, M.G. A non-enzymatic micro-needle patch sensor for freecholesterol continuous monitoring. In Proceedings of the SENSORS, 2014 IEEE, Valencia, Spain, 2–5 November 2014; pp. 347–350. [Google Scholar]
- Lee, S.J.; Yoon, H.S.; Xuan, X.; Park, J.Y.; Paik, S.; Allen, M.G. A patch type non-enzymatic biosensor based on 3D SUS micro-needle electrode array for minimally invasive continuous glucose monitoring. Sens. Actuators B Chem. 2016, 222, 1144–1151. [Google Scholar] [CrossRef]
- Hegarty, C.; McKillop, S.; Dooher, T.; Dixon, D.; Davis, J. Composite Microneedle Arrays Modified With Palladium Nanoclusters for Electrocatalytic Detection of Peroxide. IEEE Sens. Lett. 2019, 3, 1–4. [Google Scholar] [CrossRef]
- Windmiller, J.R.; Valdés-Ramírez, G.; Zhou, N.; Zhou, M.; Miller, P.R.; Jin, C.; Brozik, S.M.; Polsky, R.; Katz, E.; Narayan, R.; et al. Bicomponent Microneedle Array Biosensor for Minimally-Invasive Glutamate Monitoring. Electroanal 2011, 23, 2302–2309. [Google Scholar] [CrossRef]
- Windmiller, J.R.; Zhou, N.; Chuang, M.; Valdés-Ramírez, G.; Santhosh, P.; Miller, P.R.; Narayan, R.; Wang, J. Microneedle array-based carbon paste amperometric sensors and biosensors. Analyst 2011, 136, 1846–1851. [Google Scholar] [CrossRef]
- Yoon, Y.; Lee, G.S.; Yoo, K.; Lee, J. Fabrication of a Microneedle/CNT Hierarchical Micro/Nano Surface Electrochemical Sensor and Its In-Vitro Glucose Sensing Characterization. Sensors 2013, 13, 16672–16681. [Google Scholar] [CrossRef] [PubMed]
- Samavat, S.; Lloyd, J.; O Dea, L.; Zhang, W.; Preedy, E.; Luzio, S.; Teng, K.S. Uniform sensing layer of immiscible enzyme-mediator compounds developed via a spray aerosol mixing technique towards low cost minimally invasive microneedle continuous glucose monitoring devices. Biosens. Bioelectron. 2018, 118, 224–230. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.; Zhang, C.; Xiao, J.; You, J.; Zhang, W.; Liu, Y.; Xu, L.; Liu, A.; Xin, H.; Wang, X. Local extraction and detection of early stage breast cancers through a microneedle and nano-Ag/MBL film based painless and blood-free strategy. Mater. Sci. Eng. C 2020, 109, 110402. [Google Scholar] [CrossRef]
- Mukerjee, E.V.; Collins, S.D.; Isseroff, R.R.; Smith, R.L. Microneedle array for transdermal biological fluid extraction and in situ analysis. Sens. Actuators A Phys. 2004, 114, 267–275. [Google Scholar] [CrossRef]
- Nicholas, D.; Logan, K.A.; Sheng, Y.; Gao, J.; Farrell, S.; Dixon, D.; Callan, B.; McHale, A.P.; Callan, J.F. Rapid paper based colorimetric detection of glucose using a hollow microneedle device. Int. J. Pharm. 2018, 547, 244–249. [Google Scholar] [CrossRef]
- Wang, Y.; Kneipp, H.; Perelman, L.T.; Itzkan, I.; Dasari, R.R.; Feld, M.S.; Kneipp, K. Single Molecule Detection Using Surface-Enhanced Raman Scattering (SERS). Phys. Rev. Lett. 1997, 78, 1667–1670. [Google Scholar] [CrossRef]
- Xu, K.; Zhou, R.; Takei, K.; Hong, M. Toward Flexible Surface-Enhanced Raman Scattering (SERS) Sensors for Point-of-Care Diagnostics. Adv. Sci. 2019, 6, 1900925. [Google Scholar] [CrossRef] [PubMed]
- Krafft, C.; Dietzek, B.; Popp, J. Raman and CARS microspectroscopy of cells and tissues. Analyst 2009, 134, 1046–1057. [Google Scholar] [CrossRef]
- Yuen, C.; Liu, Q. Towards in vivo intradermal surface enhanced Raman scattering (SERS) measurements: silver coated microneedle based SERS probe. J. Biophotonics 2014, 7, 683–689. [Google Scholar] [CrossRef]
- Park, J.E.; Yonet-Tanyeri, N.; Vander, E.E.; Henry, A.I.; Perez, W.B.; Mrksich, M.; Van Duyne, R.P. Plasmonic Microneedle Arrays for in Situ Sensing with Surface-Enhanced Raman Spectroscopy (SERS). Nano Lett. 2019, 19, 6862–6868. [Google Scholar] [CrossRef]
- Kolluru, C.; Gupta, R.; Jiang, Q.; Williams, M.; Gholami, D.H.; Cao, S.; Noel, R.K.; Singamaneni, S.; Prausnitz, M.R. Plasmonic Paper Microneedle Patch for On-Patch Detection of Molecules in Dermal Interstitial Fluid. ACS Sens. 2019, 4, 1569–1576. [Google Scholar] [CrossRef] [PubMed]
- Yao, W.; Tao, C.; Zou, J.; Zheng, H.; Zhu, J.; Zhu, Z.; Zhu, J.; Liu, L.; Li, F.; Song, X. Flexible two-layer dissolving and safing microneedle transdermal of neurotoxin: A biocomfortable attempt to treat Rheumatoid Arthritis. Int. J. Pharm. 2019, 563, 91–100. [Google Scholar] [CrossRef] [PubMed]
- Li, C.G.; Dangol, M.; Lee, C.Y.; Jang, M.; Jung, H. A self-powered one-touch blood extraction system: a novel polymer-capped hollow microneedle integrated with a pre-vacuum actuator. Lab Chip 2015, 15, 382–390. [Google Scholar] [CrossRef] [PubMed]
- Li, C.G.; Joung, H.; Noh, H.; Song, M.; Kim, M.; Jung, H. One-touch-activated blood multidiagnostic system using a minimally invasive hollow microneedle integrated with a paper-based sensor. Lab Chip 2015, 15, 3286–3292. [Google Scholar] [CrossRef] [PubMed]
- Waltz, E. After Theranos. Nat. Biotechnol. 2017, 35, 11–15. [Google Scholar] [CrossRef] [PubMed]
- Miller, P.R.; Taylor, R.M.; Tran, B.Q.; Boyd, G.; Glaros, T.; Chavez, V.H.; Krishnakumar, R.; Sinha, A.; Poorey, K.; Williams, K.P.; et al. Extraction and biomolecular analysis of dermal interstitial fluid collected with hollow microneedles. Commun. Biol. 2018, 1, 173. [Google Scholar] [CrossRef] [PubMed]
- Takeuchi, K.; Takama, N.; Kim, B.; Sharma, K.; Paul, O.; Ruther, P. Microfluidic chip to interface porous microneedles for ISF collection. Biomed. Microdevices 2019, 21, 28. [Google Scholar] [CrossRef]
- Henry, S.; McAllister, D.V.; Allen, M.G.; Prausnitz, M.R. Microfabricated Microneedles: A Novel Approach to Transdermal Drug Delivery. J. Pharm. Sci. 1998, 87, 922–925. [Google Scholar] [CrossRef]
- Gualeni, B.; Coulman, S.A.; Shah, D.; Eng, P.F.; Ashraf, H.; Vescovo, P.; Blayney, G.J.; Piveteau, L.D.; Guy, O.J.; Birchall, J.C. Minimally invasive and targeted therapeutic cell delivery to the skin using microneedle devices. Brit. J. Dermatol. 2018, 178, 731–739. [Google Scholar] [CrossRef]
- Wang, S.; Zhu, M.; Zhao, L.; Kuang, D.; Kundu, S.C.; Lu, S. Insulin-Loaded Silk Fibroin Microneedles as Sustained Release System. ACS Biomater. Sci. Eng. 2019, 5, 1887–1894. [Google Scholar] [CrossRef]
- Thakur, R.R.S.; Tekko, I.A.; Al-Shammari, F.; Ali, A.A.; McCarthy, H.; Donnelly, R.F. Rapidly dissolving polymeric microneedles for minimally invasive intraocular drug delivery. Drug Deliv. Transl. Res. 2016, 6, 800–815. [Google Scholar] [CrossRef] [PubMed]
- Lau, S.; Fei, J.; Liu, H.; Chen, W.; Liu, R. Multilayered pyramidal dissolving microneedle patches with flexible pedestals for improving effective drug delivery. J. Control. Release 2017, 265, 113–119. [Google Scholar] [CrossRef]
- Seong, K.; Seo, M.; Hwang, D.Y.; O’Cearbhaill, E.D.; Sreenan, S.; Karp, J.M.; Yang, S.Y. A self-adherent, bullet-shaped microneedle patch for controlled transdermal delivery of insulin. J. Control. Release 2017, 265, 48–56. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Ye, Y.; Hochu, G.M.; Sadeghifar, H.; Gu, Z. Enhanced Cancer Immunotherapy by Microneedle Patch-Assisted Delivery of Anti-PD1 Antibody. Nano Lett. 2016, 16, 2334–2340. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Liu, Q.; Yu, J.; Yu, S.; Wang, J.; Qiang, L.; Gu, Z. Locally Induced Adipose Tissue Browning by Microneedle Patch for Obesity Treatment. ACS Nano 2017, 11, 9223–9230. [Google Scholar] [CrossRef] [PubMed]
- Lee, K.; Goudie, M.J.; Tebon, P.; Sun, W.; Luo, Z.; Lee, J.; Zhang, S.; Fetah, K.; Kim, H.; Xue, Y.; et al. Non-transdermal microneedles for advanced drug delivery. Adv. Drug Deliv. Rev. 2019, in press. [Google Scholar] [CrossRef] [PubMed]
- Than, A.; Liu, C.; Chang, H.; Duong, P.K.; Cheung, C.M.G.; Xu, C.; Wang, X.; Chen, P. Self-implantable double-layered micro-drug-reservoirs for efficient and controlled ocular drug delivery. Nat. Commun. 2018, 9, 4433. [Google Scholar] [CrossRef]
- Chen, S.; Matsumoto, H.; Moro-oka, Y.; Tanaka, M.; Miyahara, Y.; Suganami, T.; Matsumoto, A. Smart Microneedle Fabricated with Silk Fibroin Combined Semi-interpenetrating Network Hydrogel for Glucose-Responsive Insulin Delivery. ACS Biomater. Sci. Eng. 2019, 5, 5781–5789. [Google Scholar] [CrossRef]
- Yu, J.; Zhang, Y.; Ye, Y.; DiSanto, R.; Sun, W.; Ranson, D.; Ligler, F.S.; Buse, J.B.; Gu, Z. Microneedle-array patches loaded with hypoxia-sensitive vesicles provide fast glucose-responsive insulin delivery. Proc. Natl. Acad. Sci. USA 2015, 112, 8260. [Google Scholar] [CrossRef]
- Raphael, A.P.; Prow, T.W.; Crichton, M.L.; Chen, X.; Fernando, G.J.P.; Kendall, M.A.F. Targeted, Needle-Free Vaccinations in Skin using Multilayered, Densely-Packed Dissolving Microprojection Arrays. Small 2010, 6, 1785–1793. [Google Scholar] [CrossRef]
- Chong, S.Z.; Evrard, M.; Ng, L.G. Lights, Camera, and Action: Vertebrate Skin Sets the Stage for Immune Cell Interaction with Arthropod-Vectored Pathogens. Front. Immunol. 2013, 4, 286. [Google Scholar] [CrossRef] [PubMed]
- Combadière, B.; Vogt, A.; Mahé, B.; Costagliola, D.; Hadam, S.; Bonduelle, O.; Sterry, W.; Staszewski, S.; Schaefer, H.; van der Werf, S.; et al. Preferential amplification of CD8 effector-T cells after transcutaneous application of an inactivated influenza vaccine: a randomized phase I trial. PLoS ONE 2010, 5, e10818. [Google Scholar] [CrossRef]
- Boopathy, A.V.; Mandal, A.; Kulp, D.W.; Menis, S.; Bennett, N.R.; Watkins, H.C.; Wang, W.; Martin, J.T.; Thai, N.T.; He, Y.; et al. Enhancing humoral immunity via sustained-release implantable microneedle patch vaccination. Proc. Natl. Acad. Sci. USA 2019, 116, 16473. [Google Scholar] [CrossRef] [PubMed]
- Duong, H.T.T.; Yin, Y.; Thambi, T.; Kim, B.S.; Jeong, J.H.; Lee, D.S. Highly potent intradermal vaccination by an array of dissolving microneedle polypeptide cocktails for cancer immunotherapy. J. Mater. Chem. B 2020, 8, 1171–1181. [Google Scholar] [CrossRef]
- Li, Z.; He, Y.; Deng, L.; Zhang, Z.; Lin, Y. A fast-dissolving microneedle array loaded with chitosan nanoparticles to evoke systemic immune responses in mice. J. Mater. Chem. B 2020, 8, 216–225. [Google Scholar] [CrossRef] [PubMed]
- Uppu, D.S.S.M.; Turvey, M.E.; Sharif, A.R.M.; Bidet, K.; He, Y.; Ho, V.; Tambe, A.D.; Lescar, J.; Tan, E.Y.; Fink, K.; et al. Temporal release of a three-component protein subunit vaccine from polymer multilayers. J. Control. Release 2020, 317, 130–141. [Google Scholar] [CrossRef] [PubMed]
- Rouphael, N.G.; Paine, M.; Mosley, R.; Henry, S.; McAllister, D.V.; Kalluri, H.; Pewin, W.; Frew, P.M.; Yu, T.; Thornburg, N.J.; et al. The safety, immunogenicity, and acceptability of inactivated influenza vaccine delivered by microneedle patch (TIV-MNP 2015): a randomised, partly blinded, placebo-controlled, phase 1 trial. Lancet 2017, 390, 649–658. [Google Scholar] [CrossRef]
- Höschler, K.; Zambon, M.C. Influenza vaccine: going through a sticky patch. Lancet 2017, 390, 627–628. [Google Scholar] [CrossRef]
- Levin, Y.; Kochba, E.; Shukarev, G.; Rusch, S.; Herrera-Taracena, G.; van Damme, P. A phase 1, open-label, randomized study to compare the immunogenicity and safety of different administration routes and doses of virosomal influenza vaccine in elderly. Vaccine 2016, 34, 5262–5272. [Google Scholar] [CrossRef]
- Troy, S.B.; Kouiavskaia, D.; Siik, J.; Kochba, E.; Beydoun, H.; Mirochnitchenko, O.; Levin, Y.; Khardori, N.; Chumakov, K.; Maldonado, Y. Comparison of the Immunogenicity of Various Booster Doses of Inactivated Polio Vaccine Delivered Intradermally Versus Intramuscularly to HIV-Infected Adults. J. Infect. Dis. 2015, 211, 1969–1976. [Google Scholar] [CrossRef]
- DeMuth, P.C.; Li, A.V.; Abbink, P.; Liu, J.; Li, H.; Stanley, K.A.; Smith, K.M.; Lavine, C.L.; Seaman, M.S.; Kramer, J.A.; et al. Vaccine delivery with microneedle skin patches in nonhuman primates. Nat. Biotechnol. 2013, 31, 1082–1085. [Google Scholar] [CrossRef] [PubMed]
- Meyer, B.K.; Kendall, M.A.F.; Williams, D.M.; Bett, A.J.; Dubey, S.; Gentzel, R.C.; Casimiro, D.; Forster, A.; Corbett, H.; Crichton, M.; et al. Immune response and reactogenicity of an unadjuvanted intradermally delivered human papillomavirus vaccine using a first generation Nanopatch™ in rhesus macaques: An exploratory, pre-clinical feasibility assessment. Vaccine X 2019, 2, 100030. [Google Scholar] [CrossRef] [PubMed]
- Ansaldi, F.; de Florentiis, D.; Durando, P.; Icardi, G. Fluzone® Intradermal vaccine: a promising new chance to increase the acceptability of influenza vaccination in adults. Expert Rev. Vaccines 2012, 11, 17–25. [Google Scholar] [CrossRef] [PubMed]
- Atmar, R.L.; Patel, S.M.; Keitel, W.A. Intanza®: a new intradermal vaccine for seasonal influenza. Expert Rev. Vaccines 2010, 9, 1399–1409. [Google Scholar] [CrossRef] [PubMed]
- Chabanon, A.L.; Bricout, H.; Ballandras, C.; Souverain, A.; Caroe, T.D.; Butler, K.M. Report from enhanced safety surveillance of two influenza vaccines (Vaxigrip and Intanza 15 μg) in two European countries during influenza season 2016/17 and comparison with 2015/16 season. Hum. Vaccines Immunother. 2018, 14, 378–385. [Google Scholar] [CrossRef] [PubMed]
- Bricout, H.; Chabanon, A.L.; Souverain, A.; Sadorge, C.; Vesikari, T.; Caroe, T.D. Passive enhanced safety surveillance for Vaxigrip and Intanza 15 µg in the United Kingdom and Finland during the northern hemisphere influenza season 2015/16. Euro. Surveill. 2017, 22, 30527. [Google Scholar] [CrossRef]
- Ciui, B.; Martin, A.; Mishra, R.K.; Brunetti, B.; Nakagawa, T.; Dawkins, T.J.; Lyu, M.; Cristea, C.; Sandulescu, R.; Wang, J. Wearable Wireless Tyrosinase Bandage and Microneedle Sensors: Toward Melanoma Screening. Adv. Healthc. Mater. 2018, 7, 1701264. [Google Scholar] [CrossRef]
- Jayaneththi, V.R.; Aw, K.; Sharma, M.; Wen, J.; Svirskis, D.; McDaid, A.J. Controlled transdermal drug delivery using a wireless magnetic microneedle patch: Preclinical device development. Sens. Actuators B Chem. 2019, 297, 126708. [Google Scholar] [CrossRef]
- Lee, H.; Choi, T.K.; Lee, Y.B.; Cho, H.R.; Ghaffari, R.; Wang, L.; Choi, H.J.; Chung, T.D.; Lu, N.; Hyeon, T.; et al. A graphene-based electrochemical device with thermoresponsive microneedles for diabetes monitoring and therapy. Nat. Nanotechnol. 2016, 11, 566–572. [Google Scholar] [CrossRef]
- Lee, H.; Song, C.; Hong, Y.S.; Kim, M.S.; Cho, H.R.; Kang, T.; Shin, K.; Choi, S.H.; Hyeon, T.; Kim, D. Wearable/disposable sweat-based glucose monitoring device with multistage transdermal drug delivery module. Sci. Adv. 2017, 3, e1601314. [Google Scholar] [CrossRef]
- Van Damme, P.; Oosterhuis-Kafeja, F.; Van der Wielen, M.; Almagor, Y.; Sharon, O.; Levin, Y. Safety and efficacy of a novel microneedle device for dose sparing intradermal influenza vaccination in healthy adults. Vaccine 2009, 27, 454–459. [Google Scholar] [CrossRef] [PubMed]
- Sharma, S.; Zapatero-Rodríguez, J.; Estrela, P.O.; Kennedy, R. Point-of-Care Diagnostics in Low Resource Settings: Present Status and Future Role of Microfluidics. Biosensors 2015, 5, 577–601. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.; Yang, H.; Eum, J.; Ma, Y.; Fakhraei Lahiji, S.; Jung, H. Implantable powder-carrying microneedles for transdermal delivery of high-dose insulin with enhanced activity. Biomaterials 2020, 232, 119733. [Google Scholar] [CrossRef] [PubMed]

| Materials | Advantages | Disadvantages | Application | Reference |
|---|---|---|---|---|
| Silicon | Hard, Mature fabrication techniques, Biocompatible | Brittle, Easy to produce sharp waste | Solid, Coated, Hollow MNs | [33,34,35] |
| Glass | Transparent, Chemical inert, and Low-cost | Brittle, Cumbersome fabrication | Hollow MNs | [36,37,38] |
| Ceramics | Natural porous | Significant brittle fracture, Long fabrication time | Hollow, Dissolving MNs | [39,40,41] |
| Metal | High conductivity, Biocompatibility, and Some nanostructured metals have catalytic activity | Allergenic risk, High cost for noble metals | Solid, Coated, Hollow MNs | [42,43,44,45,46] |
| Polymers | Easy fabrication, Some are biodegradable or swellable | Low mechanical strength | Solid, Hollow, Coated, Dissolving, Swellable MNs | [17,47,48,49,50] |
| Carbohydrate | Biodegradable, Biocompatible | Low mechanical strength, High processing temperatures and hygroscopicity | Dissolving MNs | [26,51,52] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Xie, L.; Zeng, H.; Sun, J.; Qian, W. Engineering Microneedles for Therapy and Diagnosis: A Survey. Micromachines 2020, 11, 271. https://doi.org/10.3390/mi11030271
Xie L, Zeng H, Sun J, Qian W. Engineering Microneedles for Therapy and Diagnosis: A Survey. Micromachines. 2020; 11(3):271. https://doi.org/10.3390/mi11030271
Chicago/Turabian StyleXie, Liping, Hedele Zeng, Jianjun Sun, and Wei Qian. 2020. "Engineering Microneedles for Therapy and Diagnosis: A Survey" Micromachines 11, no. 3: 271. https://doi.org/10.3390/mi11030271
APA StyleXie, L., Zeng, H., Sun, J., & Qian, W. (2020). Engineering Microneedles for Therapy and Diagnosis: A Survey. Micromachines, 11(3), 271. https://doi.org/10.3390/mi11030271




