4.1. Velocity Evaluation with Simultaneous Acquisition of the Two Images with Different Exposure Time
To acquire two pictures of different wavelengths simultaneously, we fabricated the following set-up of the system.
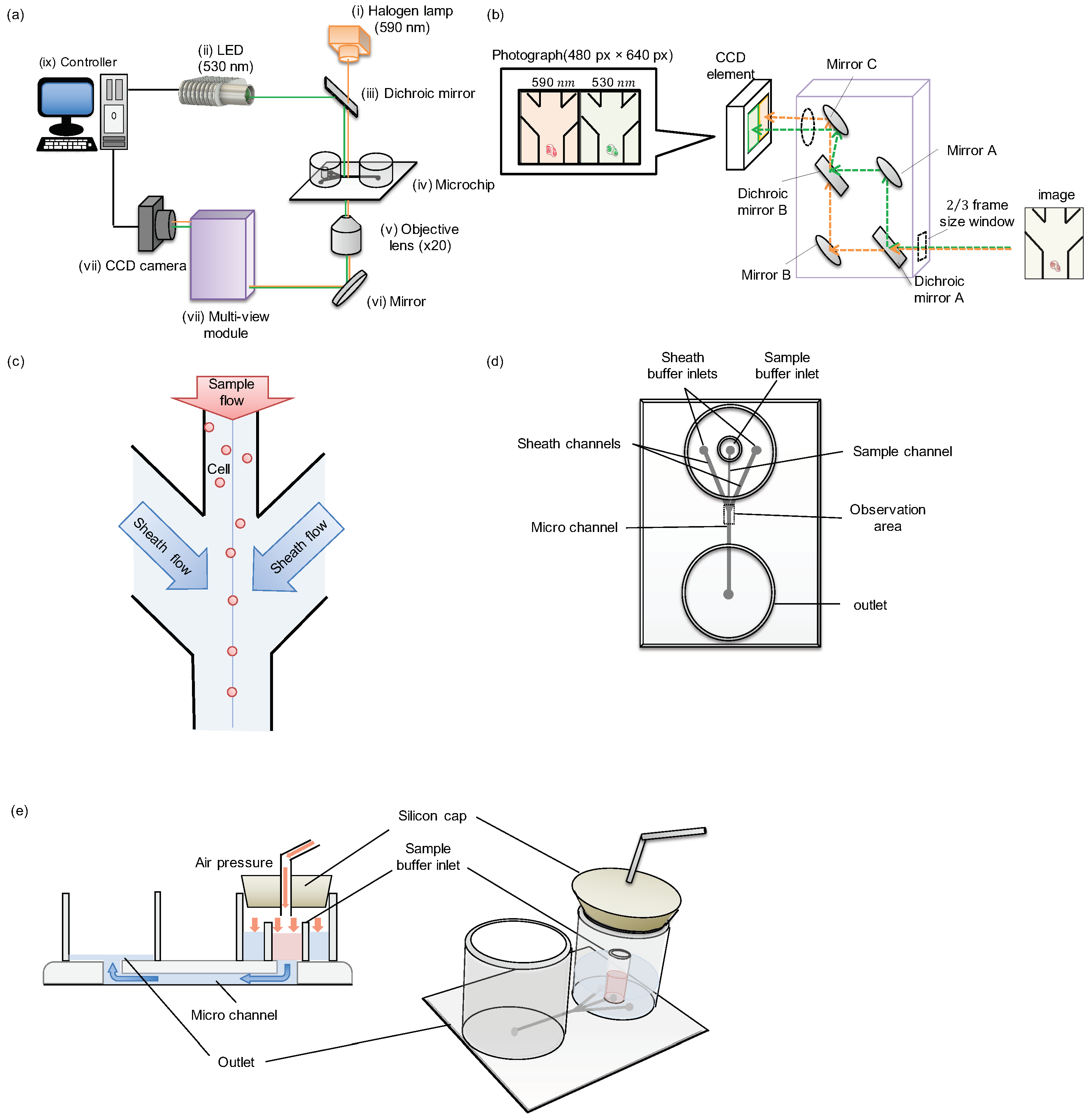
Figure 2a is a schematic illustration of the image acquisition flow cytometer with simultaneous two-wavelength differential image acquisition and analysis. The image acquisition interval was 5000
s, and the two light sources were irradiated in synchronization. The irradiation time of the halogen lamp was set to 4994
s by the camera’s electronic shutter, and the LED was set to 2500
s. These two lights were mixed at the dichroic mirror unit and followed the same optical path, and irradiated the sample in the microchip. The image of particles within the microchannel of the microchip was acquired by the 20× objective lens. After passing through the multi-view unit, the mixed image was separated into two pictures of different wavelengths of light and aligned to the CCD camera side-by-side after cutting each image to meet the 1/2 size of the CCD module area. By the above process, an image with two wavelengths of light can be acquired simultaneously in a single shot. The difference of their elongation can be compared side-by-side only within a picture of CCD image recording.
Figure 2b shows the optical path image diagram inside the multi-view unit. The light that was a mixture of two-wavelength was passed through the 2/3 frame window for cutting the acquired image to meet the size of 1/2 of the CCD module area, and was separated into two different wavelengths by the dichroic mirror A. These lights were controlled by the adjusted mirrors and were positioned to the left half or right half of the CCD camera’s detector to be aligned side by side.
Figure 2c is an image of cells flowing through a microchannel in a microchip. In this experiment, we adopted hydrodynamic focusing to compare the distribution of particles before and after focusing. The samples were applied to the upper center stream and focused at the center of the pathway by the two side sheath flows. Then the samples were gathered to the center in the downstream area.
A schematic diagram of the microchip is described in
Figure 2d. The sample and sheath buffer was placed in specified inlets, and the same air pressure was applied to these three inlets simultaneously to flow them into the microchannel equally.
Figure 2e shows the cross-sectional view of sample flow in the microchannel using air pressure. After the sample and the sheath buffers were inserted inside the specific inlets, a silicon cap lid was attached to seal the whole three inlets of the channels to apply the same air pressures for the balance of flow speeds. The samples and the sheath buffers were pushed by the applied air pressure and flow into the microchannels. The applied air pressure was controlled by the syringe pump and monitored. For example, when the applied pressure was 1.5 kPa, the max flow velocity at the center of the pathway was around 1 mm/s.
First, we examined the principle of velocity measurement with fixed 12 m polystyrene spheres under the dry condition on the mechanically controlled movement apparatus. A beam chopper (MC1000; THORLABS, Newton, NJ, USA) was inserted to the position of the microchip within the system and the movement of the particle fixed in the microchip on the beam chopper was observed.
For the velocity measurement, the images of samples were cut around the particle at 80 pixels (px) × 80 px from the full size of images of 530 and 590 nm wavelengths, and those images of the particles at two wavelengths with different irradiation times were saved as PNG files into the analysis computer. After the subtraction of the background with no prerecorded sample images, the 8-bit (256-step) grayscale values of obtained particle images were transformed into binary images using threshold values based on the average intensities of the acquired images of each wavelength. Using binarized images, the lengths of the X-axis, Y-axis, and the areas of particles were determined by counting the white pixels.
Figure 3a shows an example of a set of images of the single microparticle fixed in the channel. The upper two bright-field images are the micrographs of 590-nm with 4994
s (left) and 530-nm with 2500
s (left) (right) lights. These two images were acquired simultaneously as a single shot image in a CCD-camera. They were divided from the single-shot image of the particle with dichroic mirrors in the multi-view module. The lower two micrographs are their binarized images to measure their elongation automatically on a computer.
As shown in the images in
Figure 3a (i), the acquired images of fixed particle shapes with different wavelengths and exposure time showed the same spherical shape of the single 12
m polystyrene sphere. When we apply the 20× obj. lens with HXC13 CCD camera, the binarized data values of both wavelength images were the same and were 21 px in diameter for the
X-axis (perpendicular to the flow direction) and 21 px in diameter for
Y-axis (parallel to flow direction). The area of the particle was 360 px. As the size of one pixel was 0.704
m, the diameter of the particles can be measured as 14.7
m. The results confirmed no significant difference in image size and length caused by the difference in the wavelength of lights.
Although the beads were spherical during their fixed stationary position (stopping) (
Figure 3a (i)), elongated particle images were acquired due to the movement of the particles during the light exposure time (
Figure 3a (ii)). When the microbead moved to Y-direction (flow direction) with 1.4 mm/s mechanically by rotation of the beam chopper, the binarized images were changed to 19 (
X-axis), 28 (
Y-axis), and 472 px (area) for the left image (590 nm with 4994
s), and 19 (
X-axis), 23 (
Y-axis), and 359 px (area) for the right image (530 nm with 2500
s) (see
Figure 3a (ii)). The difference of the length of particles caused by the different irradiation time of lights was used for estimating the velocity of particle movement and was
[m/s], which was consistent with the rotation velocity. It should be noted that the
X-axis length of both particles was similarly shrunk by 2 px, which should not be changed in this experiment because this direction is perpendicular to the movement direction. It may be caused by the image blur of the total shape of the particle and might have changed the edge of binarized images of particles. In this experiment, we did not add any edge enhancement technology into the edge detection and applied same threshold value for moving particles as the stopped particles. When we applied more precise edge detection technology, more precise binarized data analysis can be adopted for this velocity measurement.
As shown in the images of
Figure 3a (ii), the difference of elongation ratio depended on the difference of irradiation time of two-wavelength lights. In other words, the exposure time changes the shapes of the sample in images. However, using the acquired velocity information, we can restore the original shape of the samples by exploiting the idea explained in
Figure 1b (iv). The principle-based reconstruction was shown in
Figure 3a (iii). The pixels of the lower half in the flow direction of the raw image of the sample was lifted to upstream direction by 10 px in one by one manner for the left image and by 5 px for the right image, where the values 10 px and 5 px were acquired from the calculation of the shift of the lower edge position pixels during the 4994
s and 2500
s exposure time with a flow velocity of particle, 2 px/ms.
Figure 3b demonstrates the result of the accuracy of the velocity measurement of this principle. The velocity of movement of the microparticle on the rotating beam chopper was observed at 0, 483.7, 725.4, 967.0, and 1208.3 mm/s, and the irradiation time of lights was set at 2500 and 4994
s for 530 and 590 nm lights, respectively. Exploiting the velocity measurement procedure described above, we plotted the relationship between the set rotation velocity of the beam chopper and the measured velocity of the 12
m polystyrene spheres. The results exhibited a linear correlation between the set velocities and measured velocities. Therefore, the result authenticated that the particle velocity can be measured with the single-shot of two-wavelength images using this method.
4.2. Flow Velocity Distribution in Microfluidic Pathway
Next, we examined the ability of flow velocity measurement of microparticles within the microchannel with shingle-shot image acquisition. In this experiment, 75
L of the sample was applied to the sample inlet of the microchip in the system. When the sample was microbeads, pure water was used as the buffer solution and sheath buffer, and when the sample was cells, the cell suspension was used as a sheath buffer. Air pressure was applied to both sample and sheath buffer inlets equally and simultaneously using a syringe pump to control the flow speed of samples (
Figure 2e). The microchip was illuminated by a halogen lamp that emits light continuously and an LED light with a 2.5 ms flushing every 5 ms intervals synchronized with the shutter opening timing of the CCD camera.
Figure 4 shows the measurement results of the velocity of 12
m microbeads flowing in the microchannel.
Figure 4a is an image of the PDMS microchannel, indicating the two data acquisition positions: one (data acquisition position 1) was the upper stream of sample inlet pathway before the junction of hydrodynamic focusing, and the other (data acquisition position 2) was the lower stream after hydrodynamic focusing where the sheath side buffers were expected to concentrate the samples into the center of the microchannel. Particles that have passed through these positions were observed and stored as captured images into the computer.
Figure 4b shows the spatial distribution of passed 142 particles at the upper stream (data acquisition position 1). The distribution demonstrates that the flow of the particles was dispersed all over the microfluidic pathway width.
Figure 4c shows the spatial distribution of the measured velocity of 142 particles in the microfluidic pathway (X position) at the upper stream (data acquisition position 1). The mean values and standard deviations of particles were plotted in the graph, and the dashed line was the curve fitting result. As described in the dashed line, the obtained results were consistent with the expected laminar flow in the microchannel.
Figure 4d is the velocity distribution of 142 particles at the upper stream (data acquisition position 1). As the histogram indicates, a wide variety of flow velocity exists in the microchannel.
Figure 4e shows the spatial distribution of passing 91 particles at the lower stream (data acquisition position 2). It was confirmed that the particles were centered by the hydrodynamic focusing of two side sheath buffer flows.
Figure 4f shows the spatial distribution of the measured velocity of 91 particles in the microfluidic pathway (X position) at the lower stream (data acquisition position 2). The mean values and standard deviations of particles were plotted in the graph, and the dashed line was the curve fitting result.
Figure 4g shows the velocity distribution of 91 particles at the lower stream (data acquisition position 2). Although the spatial distribution of the particles was focused on the center of microchannel by the hydrodynamic focusing, the flow velocity distribution still remained. This result suggests that the spatial focusing was not sufficient to reduce the distribution of flow velocities of particles.
From the above results, we can indicate our system was able to measure the velocity of particles in the microchannel with only one shot. In addition, we found that the maximum flow velocity of microparticles differs depending on the X position in the channel, and even though the X position was the same, the flow velocity still varied even after the hydrodynamic focusing. Based on these results, we can suggest that it is necessary for accurate flow velocity measurement of each particle for precise cell sorting downstream because we need to estimate the correct sorting time to shift a particular target particle at the sorting point with the accurate flow velocity.
4.4. Precise Size Measurement with Flow Velocity Correction of Particles in Microfluidic Flow
Imaging flow cytometry uses the images of particles to identify target samples instead of their indirect information, such as the intensity of diffraction or fluorescence. Hence, the precise measurement of the image-based index, including actual size or a particular shape, is essential. However, because of the movement of samples during the exposure time, the acquired images of samples were elongated to the flow direction as far as the exposure time exists. To overcome this problem, our single-shot velocity measurement method can also give us another advantage for improving the accuracy of sample sizes. When we can acquire the precise flow velocity of each particle, we can reconstruct the shape information of the particle based on this displacement information—as explained in
Figure 1b (iv).
Figure 6 shows the result of the actual shape restoration process of the same 142 samples of
Figure 4b–d.
Figure 6a shows the images of a stationary (stopping) particle in 590 and 530 nm wavelengths (upper) and their binarized images (lower). These binarized images of the 12
m microbead in 530 and 590 nm wavelengths were round-shaped.
Figure 6b shows images of a moving (elongated) particle and its binarized pictures in 530 and 590 nm wavelengths. The difference in elongation lengths was caused by the difference in the exposure time, 4994 and 2500
s. From the difference in particle’s elongation length and the difference in irradiation time of these two light sources, the velocity of this particle was determined as 1.69
m/s.
Figure 6c shows images obtained by the conventional restoration method, which just compressed the raw image of the flow direction (
Y-axis) to recover the amount of elongation that occurred during the exposure time. The irradiation time of the light having a wavelength of 590 nm is 4994
s. By using the irradiation time of the light and the calculated velocity (1.69
m/s), the particle’s elongation was identified to be 8.44
m. Since the length of the elongated particles was 14.8
m, As shown in
Figure 6c, the elongated shape of the particle was recovered to 12
m in the flow direction, which was obtained with the magnification of the elongated particle image at 8.44/14.8 in the flow direction. However, the shape of the particle was different from the spherical microbead. Hence, we can conclude that the conventional simple image reduction method fails to restore the original shape of the particles.
Figure 6d is the image obtained from the restoration method we proposed in
Figure 1b (iv), in which the lower parts of the elongated particle were lifted by the amount of elongation determined by multiplying the calculated velocity and irradiation time. As shown in the images, the original shape and size of the circular particle have been restored appropriately.
Figure 6e shows the relationship between the X position and the averaged area of 142 samples of
Figure 4b–d. In general, as shown in
Figure 6e (i), even though all the particle sizes were 12
m, the acquired raw area data of those samples were larger when the velocity of the particles was faster. When we applied the conventional method to all elongated particle images, the distribution of the difference in their areas was shown depending on the X position, and it can be seen that this method cannot restore the correct shape (
Figure 6e (ii)). On the other hand, when we applied our restoration method to the particles, the distribution of particle areas became flat (
Figure 6e (ii)). These results indicate that the importance of the correct restoration method to acquire accurate area information and also the importance of the acquisition of precise velocity information of each particle for its restoration.
4.5. Ability and Potential Limitation of This Method for Imaging Flow Cytometry Measurement
In this paper, we proposed the simple single-shot image-based velocity measurement method and its application for size correction. This method exploited the exposure time for image acquisition for velocity analysis. Usually, the exposure time is necessary to the image acquisition and the origin of quality reduction of images, namely image blur. However, focusing on the bright side of these characteristics enables us to acquire the precise measurement of velocity and size of samples as described above in this paper.
In principle, this method is simple and can be applied for all the measurements. However, the resolution and preciseness are limited to the ability and resolution of hardware. From this viewpoint, the ability of this method is strongly reliant on the progress of the technologies.
First, we focused on the limitations of camera resolution. If the moving distance of the particle is less than the minimum unit of resolution (1 px) during the light irradiation time, the image of the particle is obtained without stretching.
Figure 7a shows the relationship between particle velocity and maximum irradiation time. Capturing the particles without elongation is possible by setting the irradiation time below the time of the curve shown in the figure. On the other hand, since the velocity is calculated using the difference in the particle’s
Y-axis lengths, the difference between the irradiation times of the two wavelengths needs to be longer than the maximum irradiation time.
Next, we considered the influence on sorting accuracy. If multiple particles appear in the image clipped to 80 × 80 px, accurate particle recognition cannot be performed. Therefore, it is necessary to adjust the density and velocity of the sample particles.
Figure 7b shows the relationship between the speed and maximum density of 12
m beads at the data acquisition position 2. The minimum distance that prevents neighboring particles from being simultaneously captured was calculated and converted to density. For this purpose, the elongation of the particle, determined from the velocity and the irradiation time, was taken into account. Shutter time was set to 4994
s. A depth of 19.9
m, a mean X position of the flowing particles of 34.4
m, and a standard deviation of 5.31
m at data acquisition position 2 (
Figure 4f) were used in the calculation. In order to perform an accurate measurement, it is necessary to measure the sample at a density lower than the max density indicated by the straight line in the figure.