A MEMS-Based Multi-Parameter Integrated Chip and Its Portable System for Water Quality Detection
Abstract
1. Introduction
2. Materials and Methods
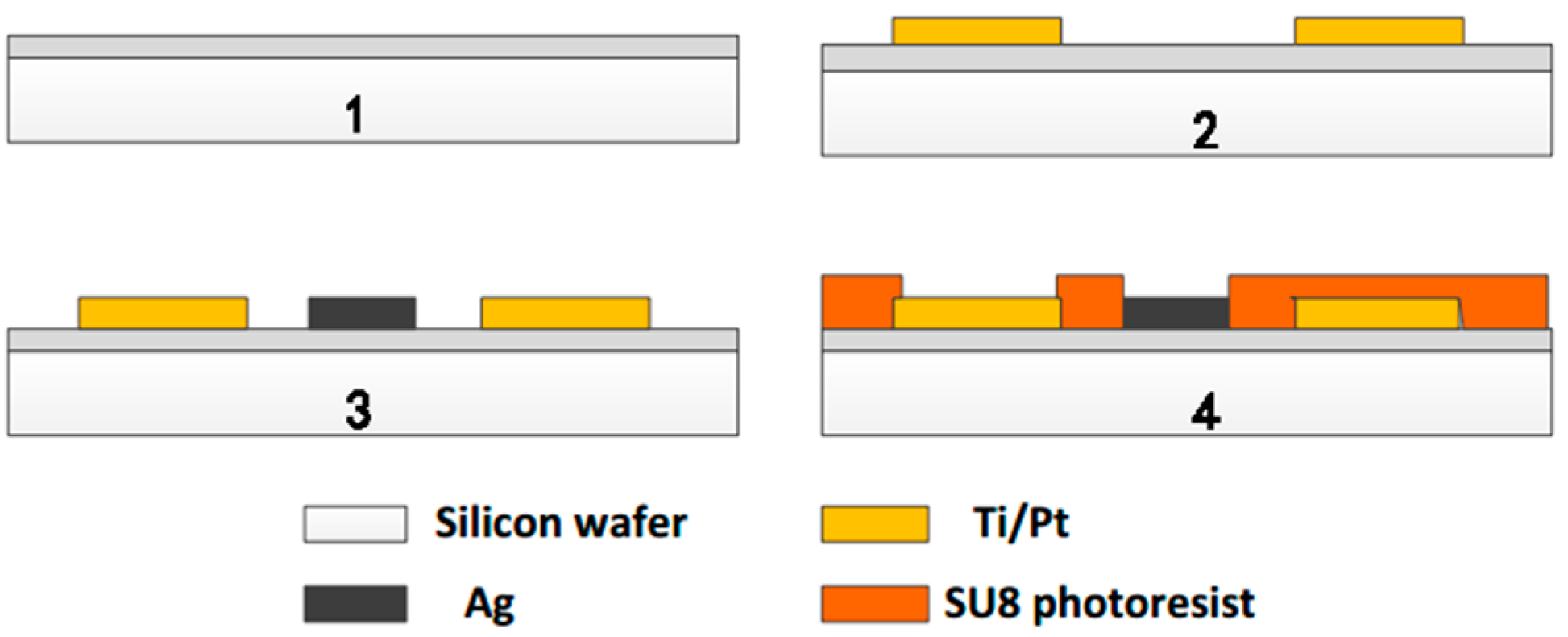
2.1. Multi-Parameter Integrated Sensor Chip
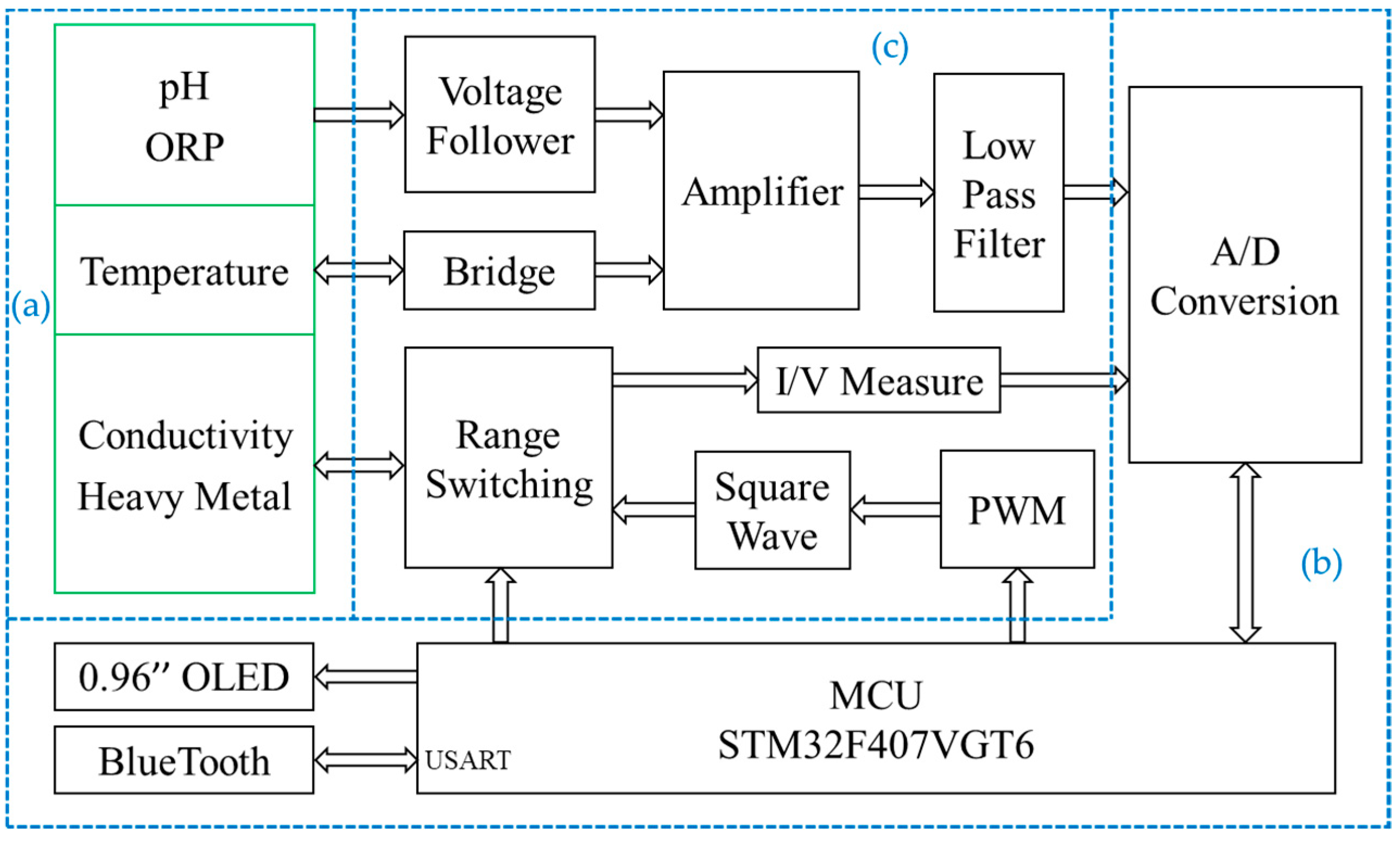
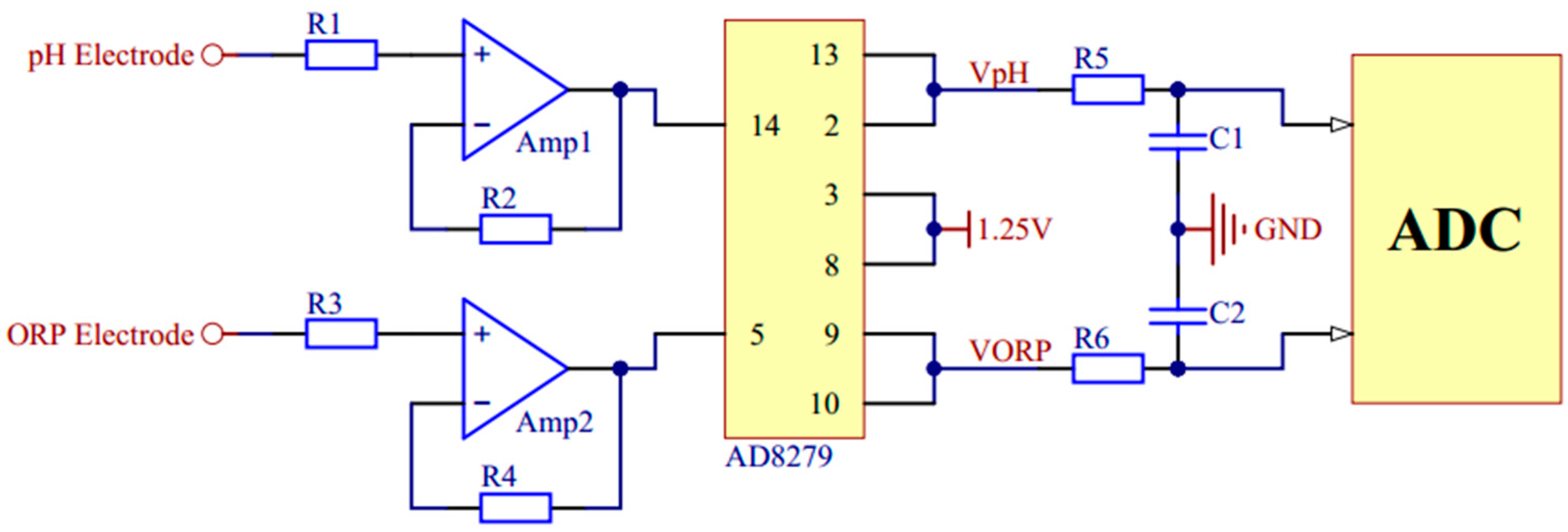
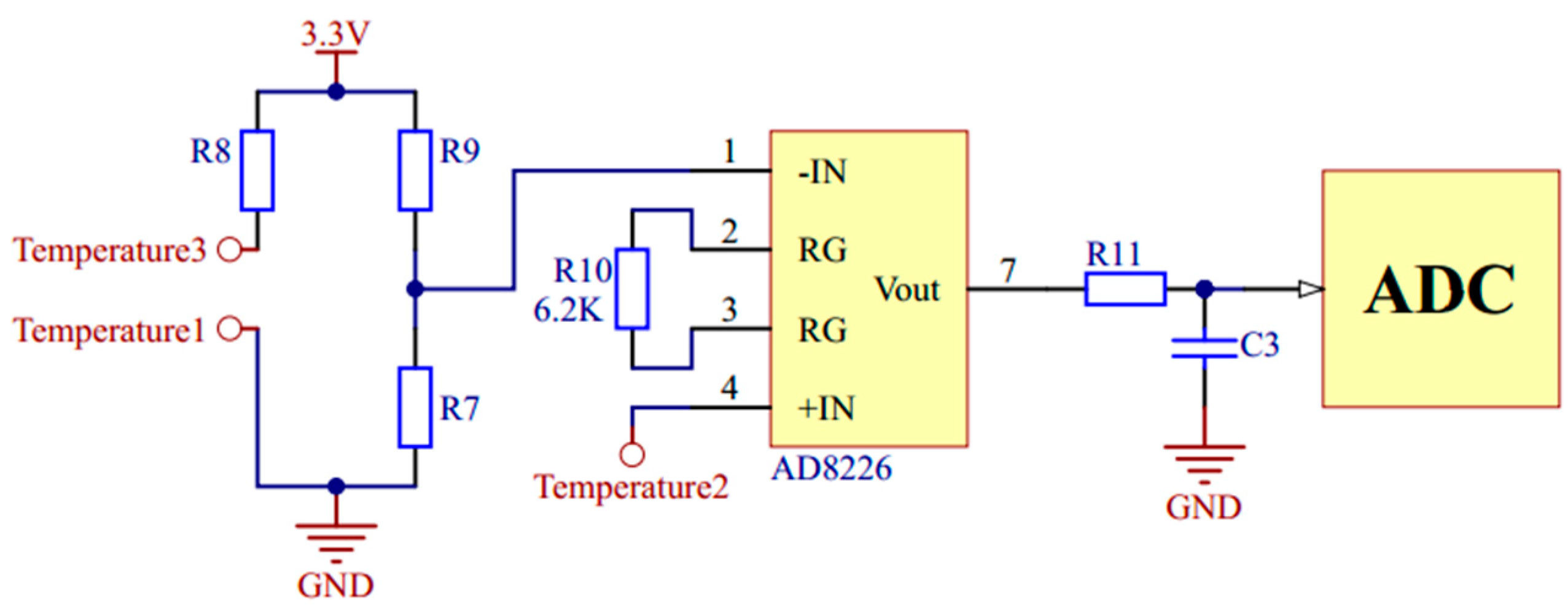
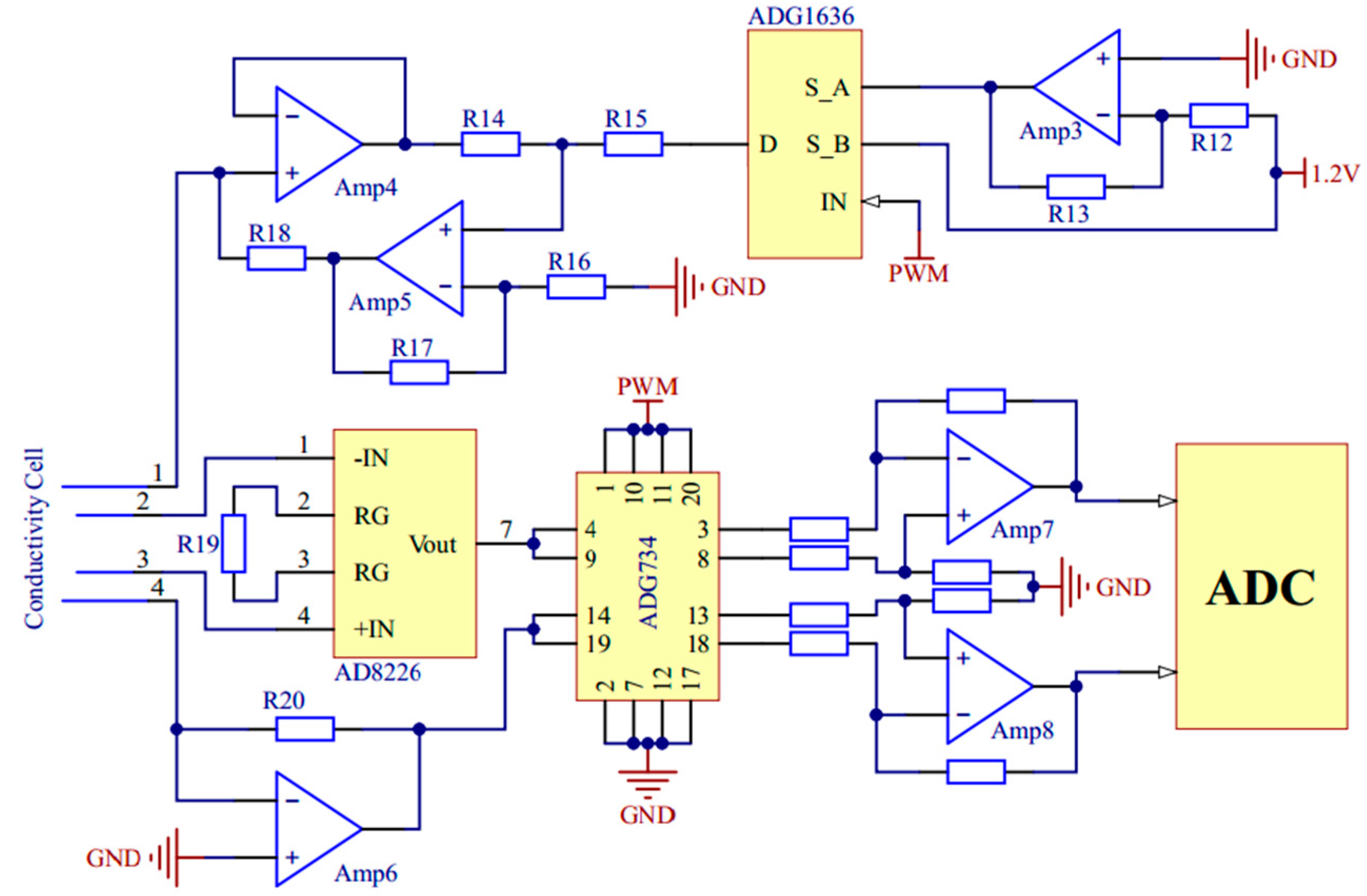
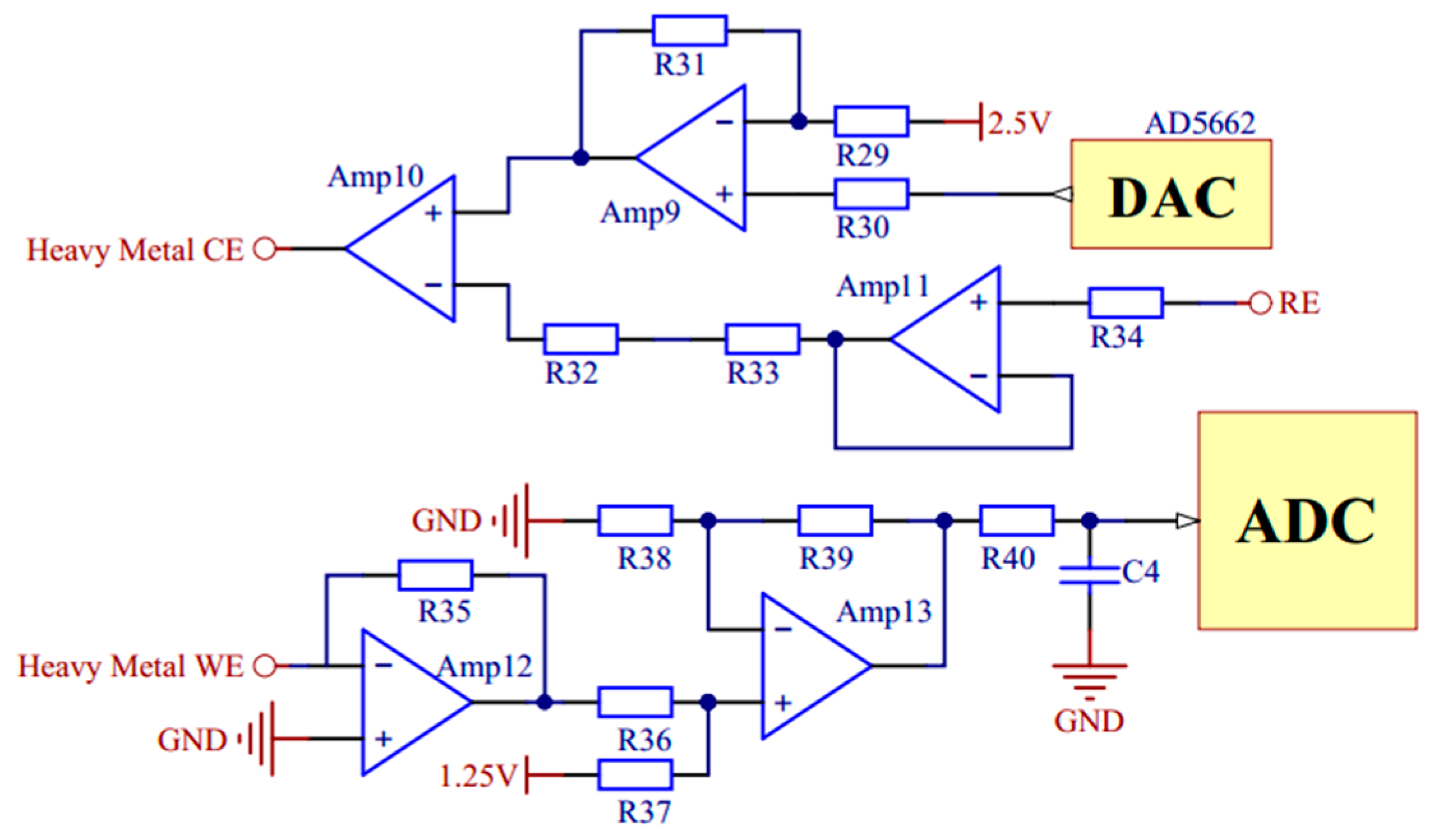
2.2. Hardware System
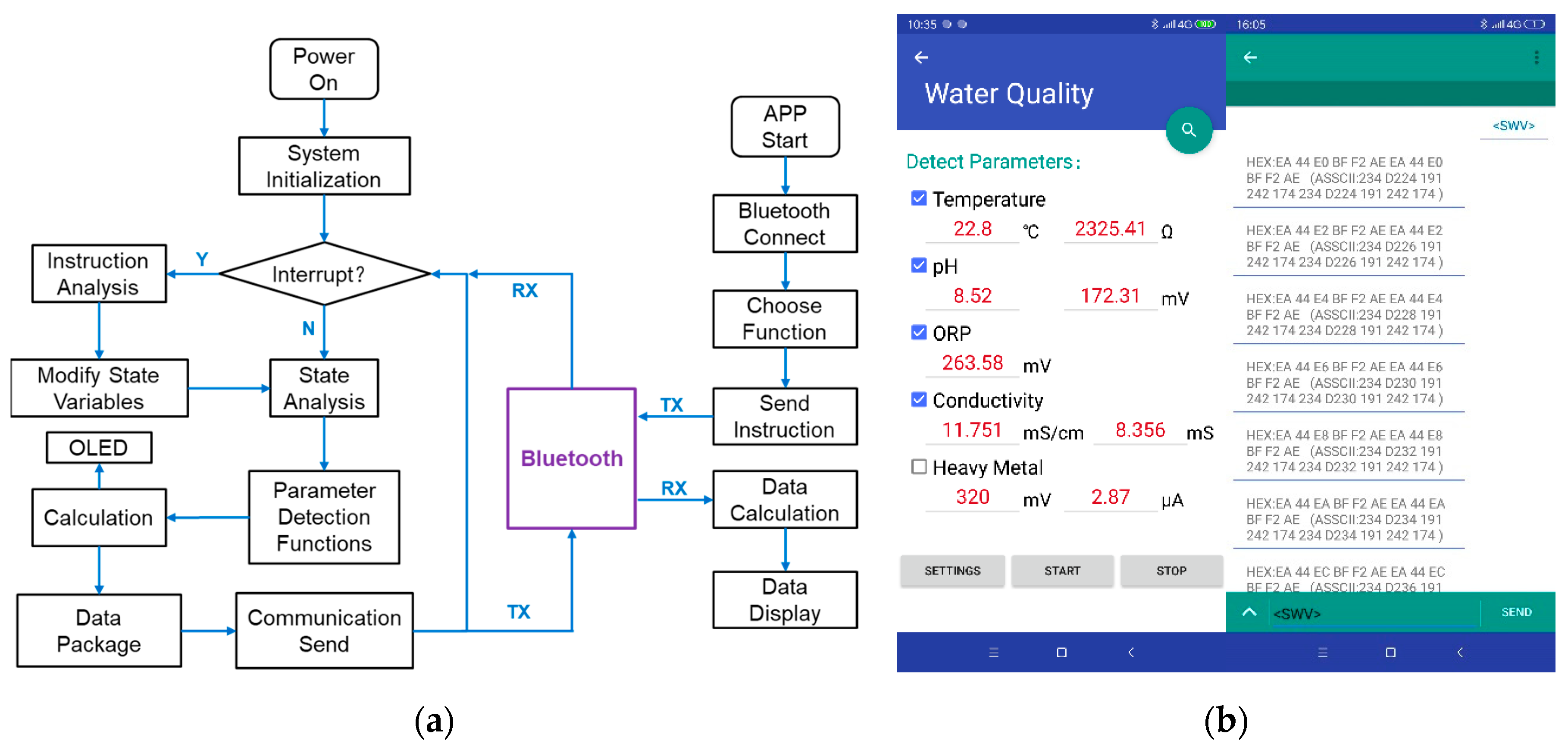
2.3. Software System
3. Results and Discussion
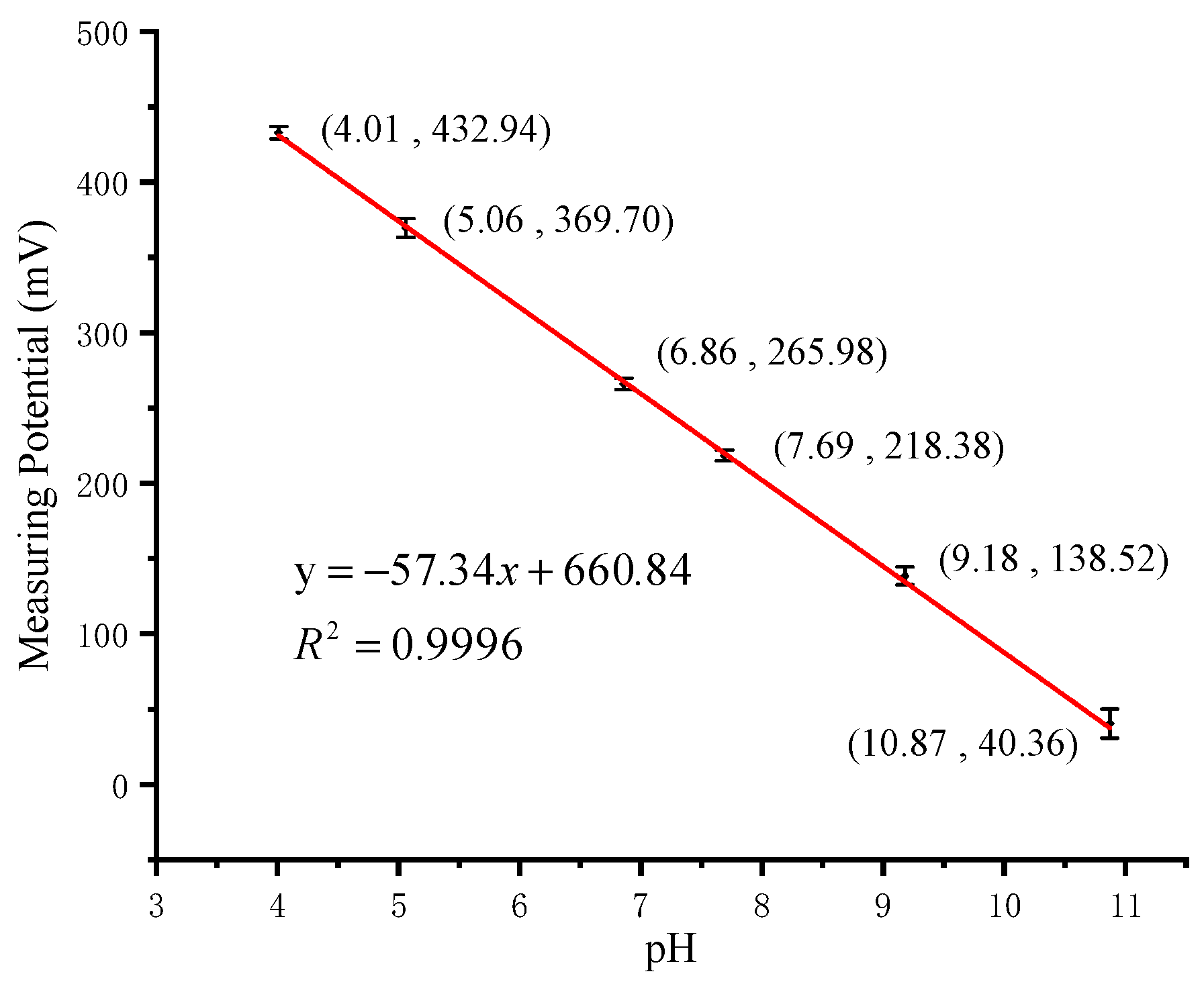
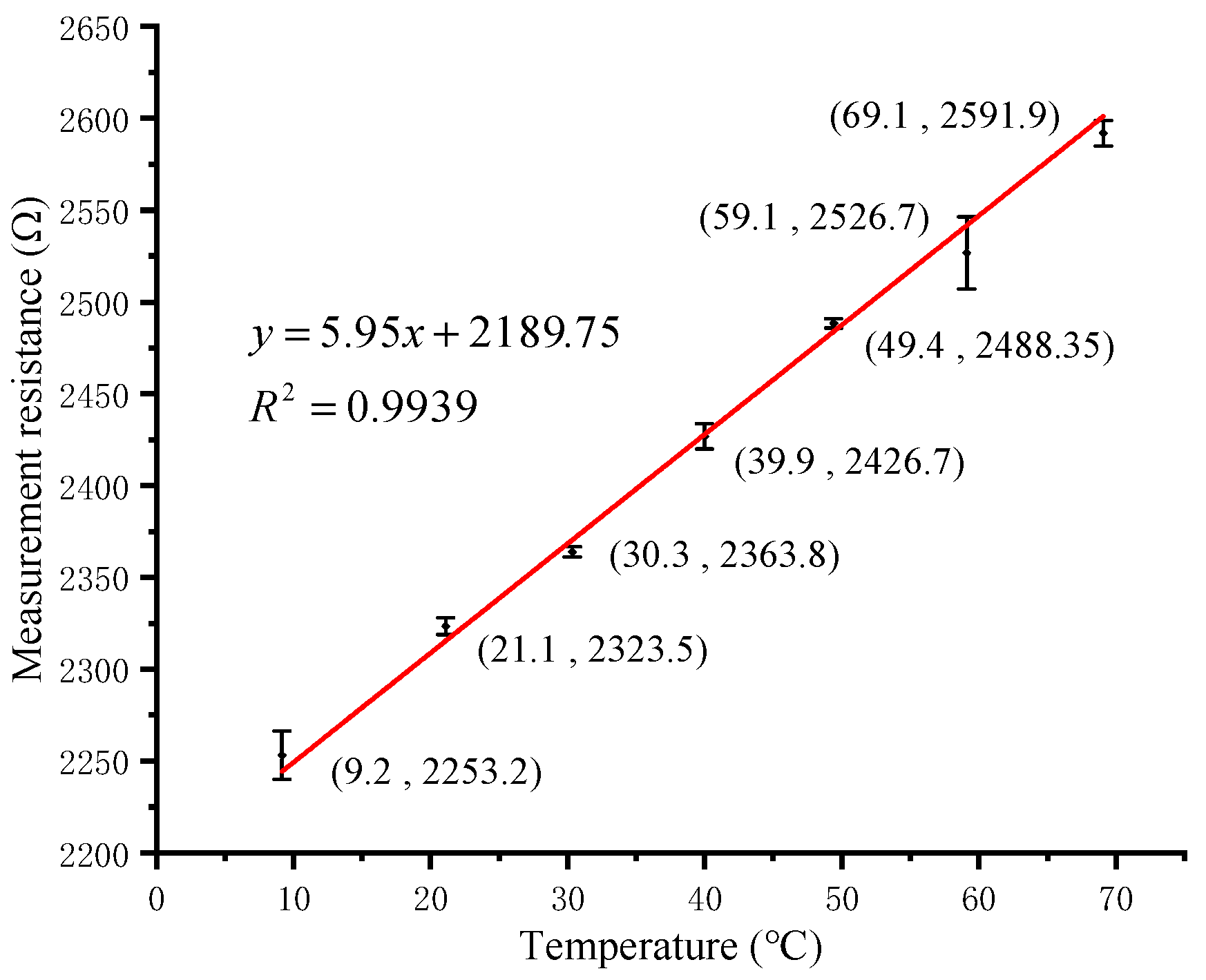
3.1. pH and Temperature Calibration Measurements
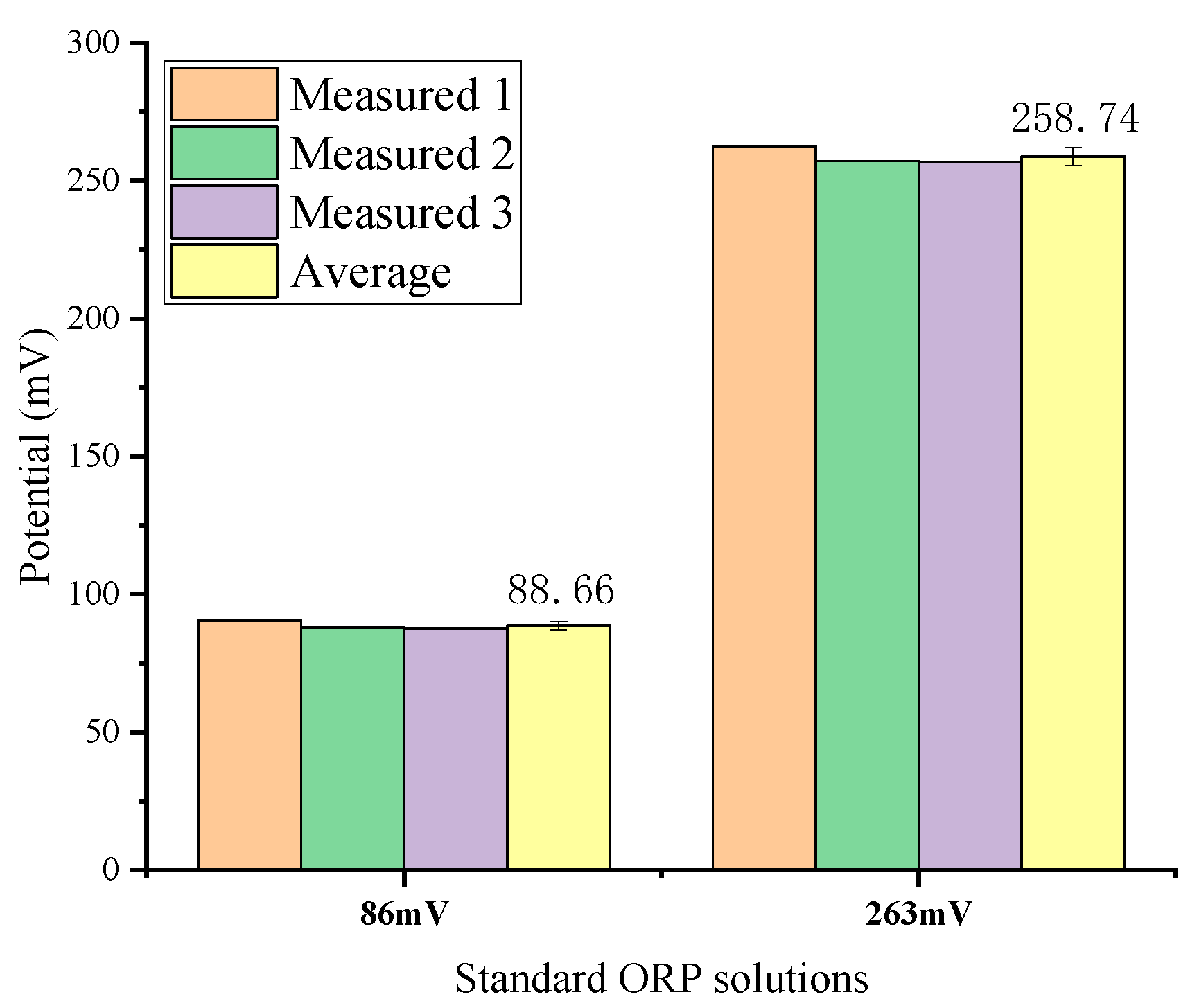
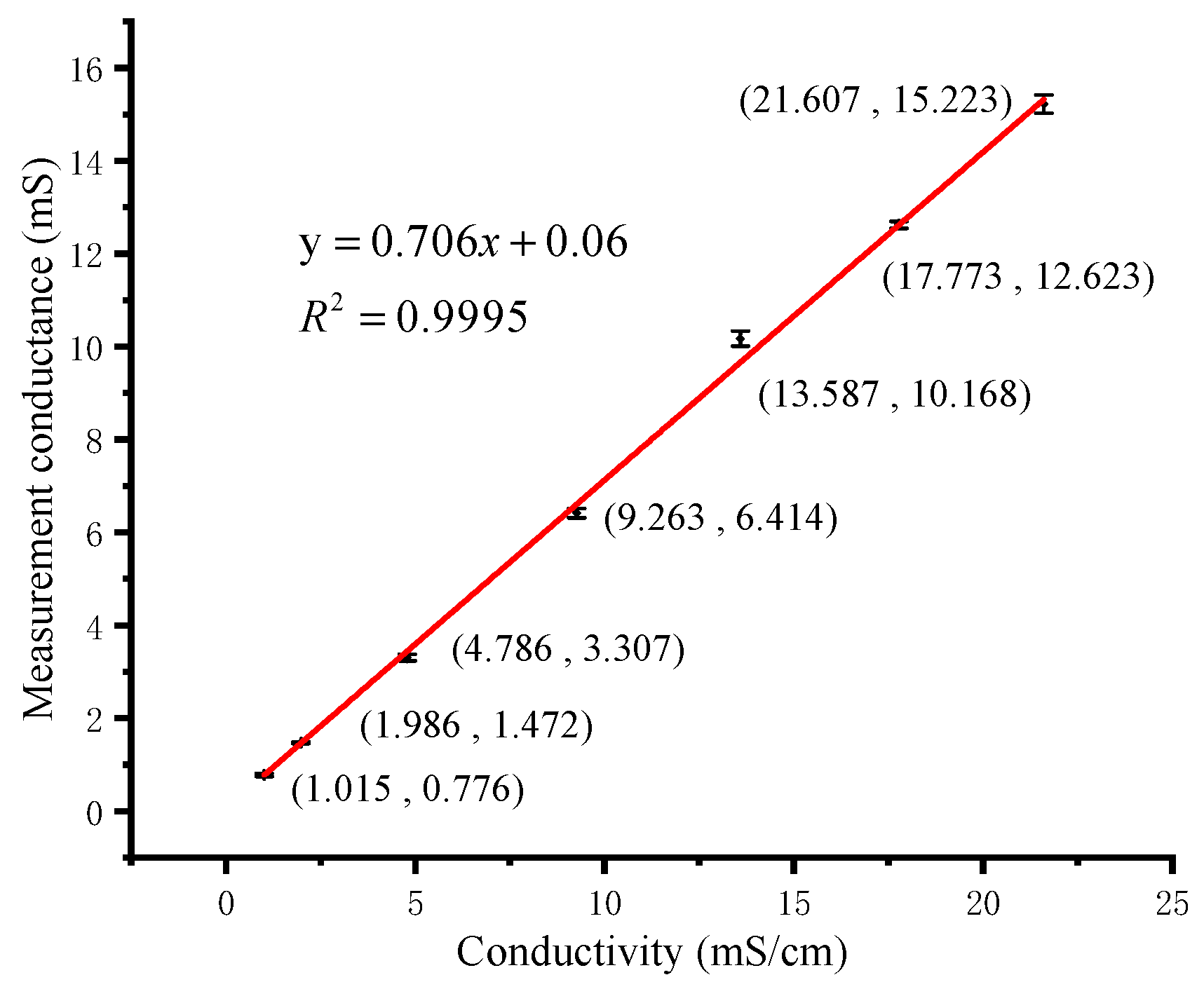
3.2. ORP and Conductivity Calibration Measurements
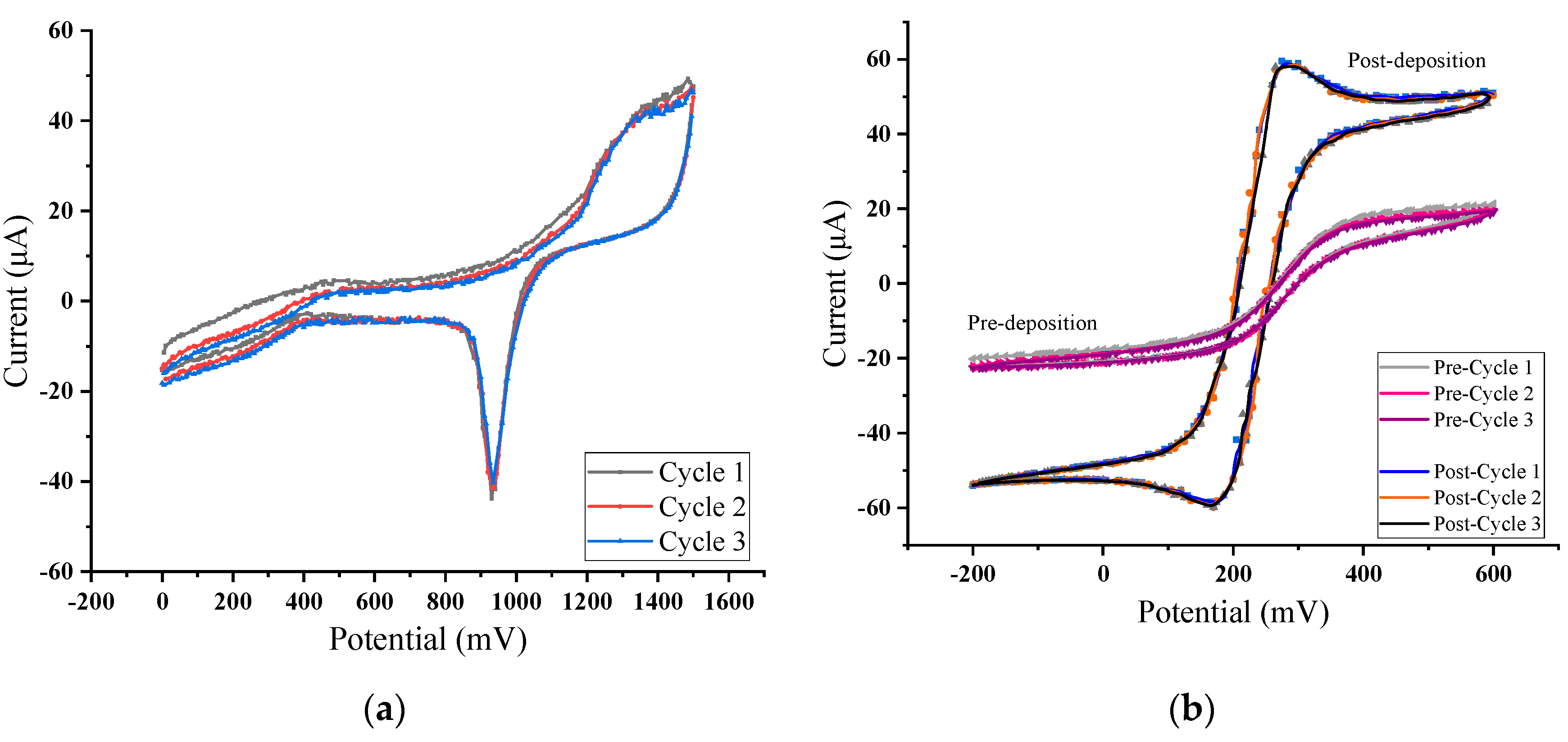
3.3. Copper Ions Measurement
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Banna, M.H.; Imran, S.; Francisque, A.; Najjaran, H.; Sadiq, R.; Rodriguez, M.; Hoorfar, M. Online drinking water quality monitoring: Review on available and emerging technologies. Crit. Rev. Environ. Sci. Technol. 2014, 44, 1370–1421. [Google Scholar] [CrossRef]
- Qin, Y.; Alam, A.U.; Pan, S.; Howlader, M.M.R.; Ghosh, R.; Hu, N.-X.; Jin, H.; Dong, S.; Chen, C.-H.; Deen, M.J. Integrated water quality monitoring system with pH, free chlorine, and temperature sensors. Sens. Actuators B Chem. 2018, 255, 781–790. [Google Scholar] [CrossRef]
- Chowdury, M.S.U.; Emran, T.B.; Ghosh, S.; Pathak, A.; Alam, M.M.; Absar, N.; Andersson, K.; Hossain, M.S. IoT based real-time river water quality monitoring system. Procedia Comput. Sci. 2019, 155, 161–168. [Google Scholar] [CrossRef]
- Zhou, B.; Bian, C.; Tong, J.; Xia, S. Fabrication of a miniature multi-parameter sensor chip for water quality assessment. Sensors 2017, 17, 157. [Google Scholar] [CrossRef] [PubMed]
- Martínez-Máñez, R.; Soto, J.; García-Breijo, E.; Gil, L.; Ibáñez, J.; Gadea, E. A multisensor in thick-film technology for water quality control. Sens. Actuators A Phys. 2005, 120, 589–595. [Google Scholar] [CrossRef]
- Qin, Y.; Alam, A.U.; Howlader, M.M.R.; Hu, N.-X.; Deen, M.J. Inkjet printing of a highly loaded palladium ink for integrated, low-cost pH sensors. Adv. Funct. Mater. 2016, 26, 4923–4933. [Google Scholar] [CrossRef]
- Banna, M.H.; Najjaran, H.; Sadiq, R.; Imran, S.A.; Rodriguez, M.J.; Hoorfar, M. Miniaturized water quality monitoring pH and conductivity sensors. Sens. Actuators B Chem. 2014, 193, 434–441. [Google Scholar] [CrossRef]
- Rotake, D.; Darji, A.D. Heavy metal ion detection in water using MEMS based sensor. Mater. Today Proc. 2018, 5, 1530–1536. [Google Scholar] [CrossRef]
- Jin, H.; Qin, Y.; Pan, S.; Alam, A.U.; Dong, S.; Ghosh, R.; Deen, M.J. Open-source low-cost wireless potentiometric instrument for pH determination experiments. J. Chem. Educ. 2018, 95, 326–330. [Google Scholar] [CrossRef]
- Giménez-Gómez, P.; Escudé-Pujol, R.; Jiménez-Jorquera, C.; Gutiérrez-Capitán, M. Multisensor portable meter for environmental applications. IEEE Sens. J. 2015, 15, 6517–6523. [Google Scholar] [CrossRef]
- Kruse, P. Review on water quality sensors. J. Phys. D Appl. Phys. 2018, 51, 203002. [Google Scholar] [CrossRef]
- Wang, J.; Bian, C.; Li, Y.; Sun, J.; Tong, J.; Xia, S. A multi-parameter integrated chip system for water quality detection. Int. J. Mod. Phys. B 2019, 33, 1950041. [Google Scholar] [CrossRef]
- Lin, W.-C.; Brondum, K.; Monroe, C.W.; Burns, M.A. Multifunctional water sensors for pH, ORP, and conductivity using only microfabricated platinum electrodes. Sensors 2017, 17, 1655. [Google Scholar] [CrossRef] [PubMed]
- Lee, W.H.; Lee, J.-H.; Choi, W.-H.; Hosni, A.A.; Papautsky, I.; Bishop, P.L. Needle-type environmental microsensors: Design, construction and uses of microelectrodes and multi-analyte MEMS sensor arrays. Meas. Sci. Technol. 2011, 22, 042001. [Google Scholar] [CrossRef]
- Qi, X.; Qian, J.; Chen, T.; Lu, D.; Chen, B. Electrochemical determination of Cu (II) ions based on Ag/Pd alloy for water quality early warning. Int. J. Electrochem. Sci. 2017, 12, 5511–5520. [Google Scholar] [CrossRef]
- Ghate, S.; Chaphekar, S.B. Plagiochasma appendiculatumas a biotest for water quality assessment. Environ. Pollut. 2000, 108, 173–181. [Google Scholar] [CrossRef]
- Campanella, L.; Cubadda, F.; Sammartino, M.P.; Saoncella, A. An algal biosensor for the monitoring of water toxicity in estuarine environments. Water Res. 2000, 35, 69–76. [Google Scholar] [CrossRef]
- Al-Yousuf, M.H.; El-Shahawi, M.S.; Al-Ghais, S.M. Trace metals in liver, skin and muscle of Lethrinus lentjan fish species in relation to body length and sex. Sci. Total Environ. 2000, 256, 87–94. [Google Scholar] [CrossRef]
- Lam, K.L.; Ko, P.W.; Wong, J.K.-Y.; Chan, K.M. Metal toxicity and metallothionein gene expression studies in common carp and tilapia. Mar. Environ. Res. 1998, 46, 563–566. [Google Scholar] [CrossRef]
- Iyengar, G.V.; Nair, P.P. Global outlook on nutrition and the environment: Meeting the challenges of the next millennium. Sci. Total Environ. 2000, 249, 331–346. [Google Scholar] [CrossRef]
- Gómez-Ariza, J.L.; Sánchez-Rodas, D.; Giráldez, I.; Morales, E. A comparison between ICP-MS and AFS detection for arsenic speciation in environmental samples. Talanta 2000, 51, 257–268. [Google Scholar] [CrossRef]
- Sales, A.; Alvarez, A.; Areal, M.R.; Maldonado, L.; Marchisio, P.; Rodríguez, M.; Bedascarrasbure, E. The effect of different propolis harvest methods on its lead contents determined by ET AAS and UV-visS. J. Hazard. Mater. 2006, A137, 1352–1356. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Bian, C.; Tong, J.; Sun, J.; Xia, S. Electrochemical measurement of copper, lead and zinc ions in tap water by using an environment-friendly microelectrode. Key Eng. Mater. 2013, 562–565, 1302–1307. [Google Scholar] [CrossRef]
- Environmental Quality Standards for Surface Water (GB 3838-2002). Available online: http://bz.mee.gov.cn/bzwb/shjbh/shjzlbz/200206/t20020601_66497.shtml (accessed on 19 December 2019).
- Standards for Drinking Water Quality (GB 5749-2006). Available online: http://openstd.samr.gov.cn/bzgk/gb/newGbInfo?hcno=73D81F4F3615DDB2C5B1DD6BFC9DEC86 (accessed on 19 December 2019).
- Standard Examination Methods for Drinking Water (GB/T 5750-2006). Available online: http://openstd.samr.gov.cn/bzgk/gb/newGbInfo?hcno=53FDB5BC0D19CE4941F07F5DB5635956 (accessed on 19 December 2019).
- Kurzweil, P. Metal oxides and ion-exchanging surfaces as pH sensors in liquids: State-of-the-art and outlook. Sensors 2009, 9, 4955–4985. [Google Scholar] [CrossRef]
- BLE103 Page. Available online: http://www.mokuai.cn/products/100.html (accessed on 19 December 2019).
- AD7790 Datasheet. Available online: https://www.analog.com/en/products/ad7790.html (accessed on 19 December 2019).
- Lisboa, T.P.; de Faria, L.V.; Matos, M.A.C.; Matos, R.C.; de Sousa, R.A. Simultaneous determination of cadmium, lead, and copper in the constituent parts of the illegal cigarettes by Square Wave Anodic Stripping Voltammetry. Microchem. J. 2019, 150, 104183. [Google Scholar] [CrossRef]
- Dai, X.; Nekrassova, O.; Hyde, M.E.; Compton, R.G. Anodic stripping voltammetry of Arsenic (III) using gold nanoparticle-modified electrodes. Anal. Chem. 2004, 76, 5924–5929. [Google Scholar] [CrossRef]
- Fundamentals of ORP Calibration. Available online: https://www.emerson.com/documents/automation/application-data-sheet-fundamentals-of-orp-calibration-rosemount-en-68452 (accessed on 19 December 2019).
- ORP Calibration Basics. Available online: https://www.hamiltoncompany.com/process-analytics/ph-and-orp-knowledge/orp-calibration-basics#top (accessed on 19 December 2019).
- Test Solutions of Electrolytic Conductivity Analyzer—Preparation Method of Sodium Chloride Solutions (GB/T 27503-2011). Available online: http://openstd.samr.gov.cn/bzgk/gb/newGbInfo?hcno=340174BB16B8FB30FE50504CD63B6820 (accessed on 19 December 2019).



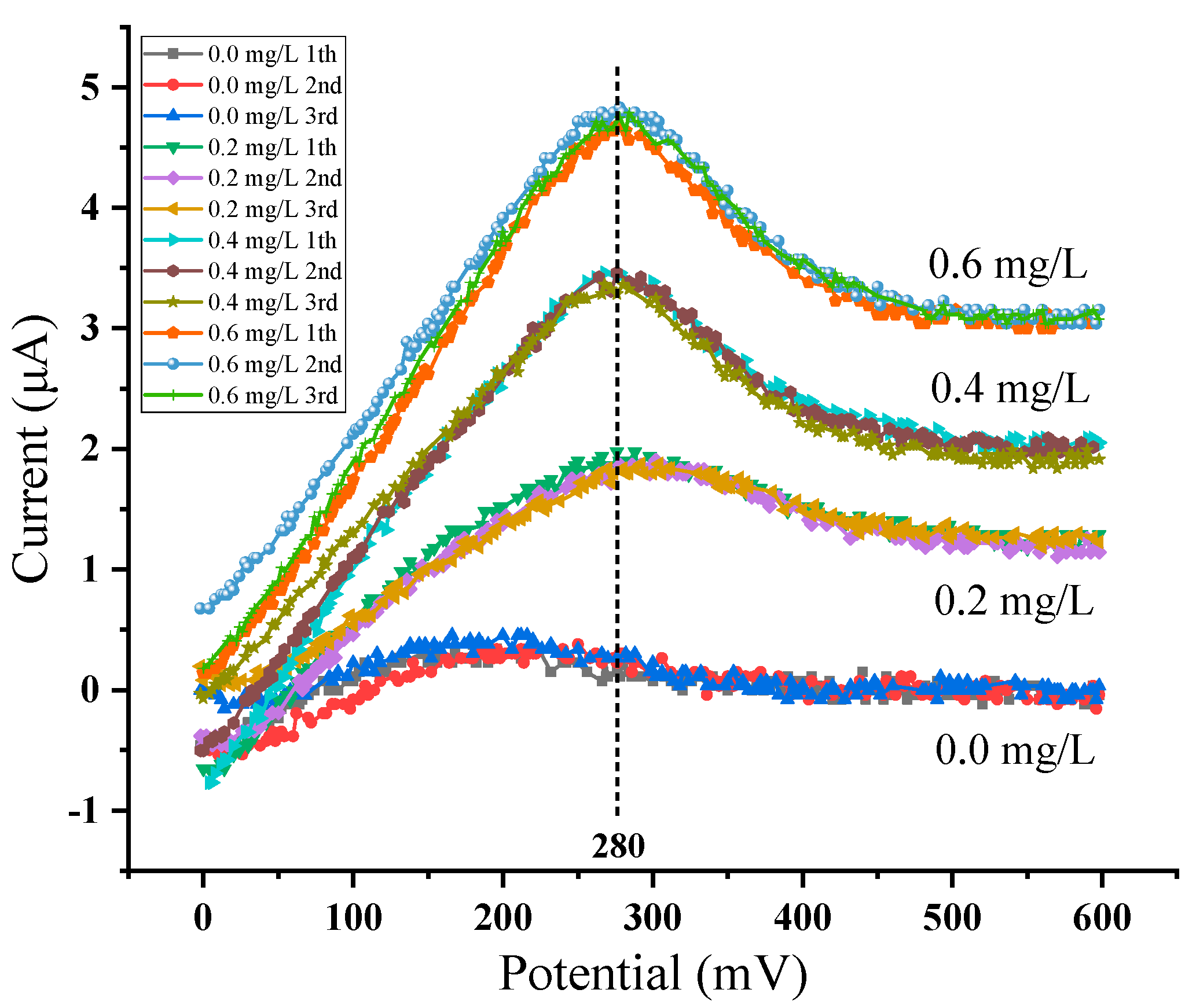
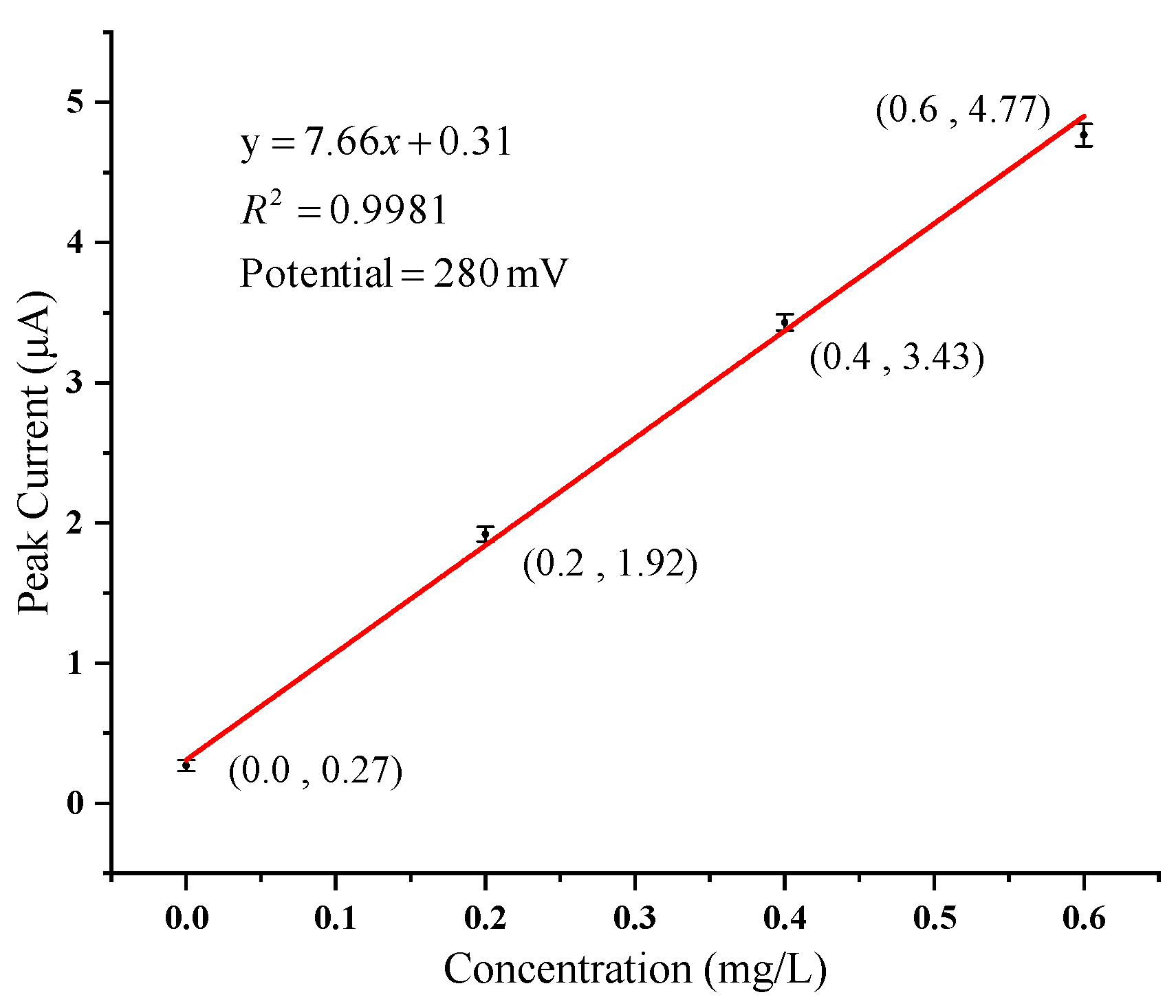
| Accumulation Time | Accumulation Potential | Step | Frequency | Initial Voltage | Final Voltage | Pulse Height |
|---|---|---|---|---|---|---|
| 300 s | −600 mV | 2 mV | 25 Hz | −100 mV | 800 mV | 25 mV |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wu, Z.; Wang, J.; Bian, C.; Tong, J.; Xia, S. A MEMS-Based Multi-Parameter Integrated Chip and Its Portable System for Water Quality Detection. Micromachines 2020, 11, 63. https://doi.org/10.3390/mi11010063
Wu Z, Wang J, Bian C, Tong J, Xia S. A MEMS-Based Multi-Parameter Integrated Chip and Its Portable System for Water Quality Detection. Micromachines. 2020; 11(1):63. https://doi.org/10.3390/mi11010063
Chicago/Turabian StyleWu, Ziyue, Jiaqi Wang, Chao Bian, Jianhua Tong, and Shanhong Xia. 2020. "A MEMS-Based Multi-Parameter Integrated Chip and Its Portable System for Water Quality Detection" Micromachines 11, no. 1: 63. https://doi.org/10.3390/mi11010063
APA StyleWu, Z., Wang, J., Bian, C., Tong, J., & Xia, S. (2020). A MEMS-Based Multi-Parameter Integrated Chip and Its Portable System for Water Quality Detection. Micromachines, 11(1), 63. https://doi.org/10.3390/mi11010063





