A Nontoxic Battery with 3D-Printed Housing for On-Demand Operation of Microcontrollers in Microfluidic Sensors
Abstract
1. Introduction
2. Materials and Methods
3. Results
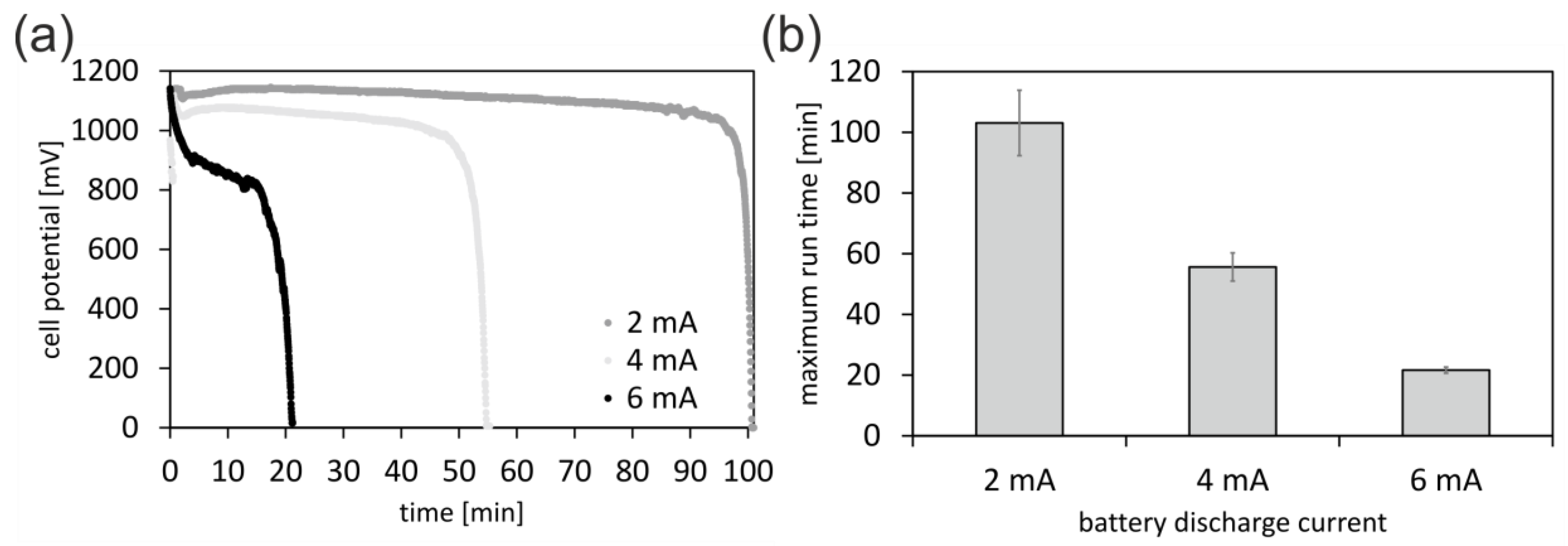
3.1. 3D-Printed Battery
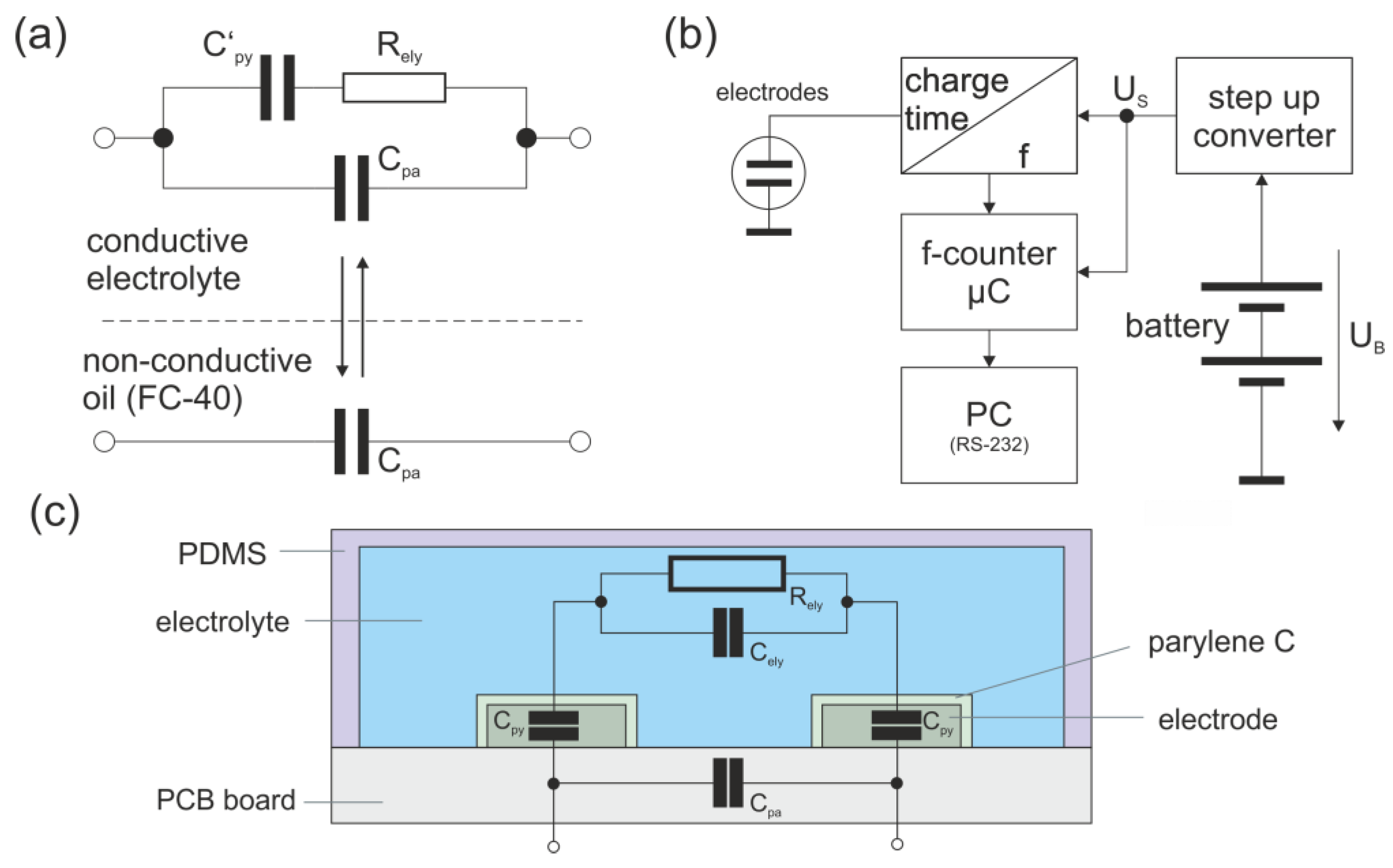
3.2. Portable Conductivity Sensor with Microcontroller
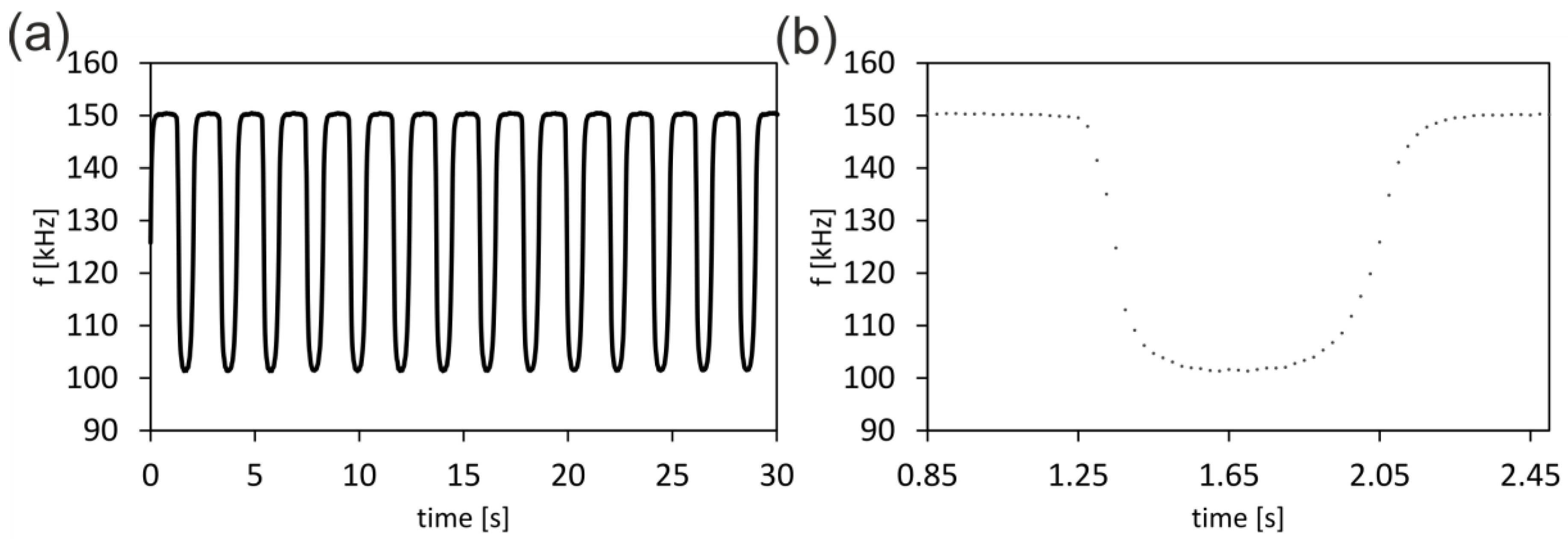
3.3. Conductivity Sensor Data Collection with Battery-Operated Microcontroller
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Martinez, A.W.; Phillips, S.T.; Carrilho, E.; Thomas, S.W.; Sindi, H.; Whitesides, G.M. Simple telemedicine for developing regions: Camera phones and paper-based microfluidic devices for real-time, off-site diagnosis. Anal. Chem. 2008, 80, 3699–3707. [Google Scholar] [CrossRef] [PubMed]
- Yu, H.; Tan, Y.; Cunningham, B.T. Smartphone fluorescence spectroscopy. Anal. Chem. 2014, 86, 8805–8813. [Google Scholar] [CrossRef] [PubMed]
- Park, T.S.; Li, W.; McCracken, K.E.; Yoon, J.Y. Smartphone quantifies salmonella from paper microfluidics. Lab Chip 2013, 13, 4832–4840. [Google Scholar] [CrossRef] [PubMed]
- You, D.J.; Park, T.S.; Yoon, J.Y. Cell-phone-based measurement of TSH using Mie scatter optimized lateral flow assays. Biosens. Bioelectron. 2013, 40, 180–185. [Google Scholar] [CrossRef] [PubMed]
- Priye, A.; Bird, S.W.; Light, Y.K.; Ball, C.S.; Negrete, O.A.; Meagher, R.J. A smartphone-based diagnostic platform for rapid detection of Zika, chikungunya, and dengue viruses. Sci. Rep. 2017, 7, 44778. [Google Scholar] [CrossRef] [PubMed]
- Huang, X.; Lin, X.; Urmann, K.; Li, L.; Xie, X.; Jiang, S.; Hoffmann, M.R. Smartphone-based in-gel loop-mediated isothermal amplification (gLAMP) system enables rapid coliphage MS2 quantification in environmental waters. Environ. Sci. Technol. 2018, 52, 6399–6407. [Google Scholar] [CrossRef] [PubMed]
- Luo, X.; Davis, J.J. Electrical biosensors and the label free detection of protein disease biomarkers. Chem. Soc. Rev. 2013, 42, 5944–5962. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, T.; Zoega Andreasen, S.; Wolff, A.; Duong Bang, D. From lab on a chip to point of care devices: The role of open source microcontrollers. Micromachines 2018, 9, 403. [Google Scholar] [CrossRef] [PubMed]
- Dlutowski, J.; Biver, C.J.; Wang, W.; Knighton, S.; Bumgarner, J.; Langebrake, L.; Moreno, W.; Cardenas-Valencia, A.M. The development of BCB-sealed galvanic cells. Case study: Aluminum-platinum cells activated with sodium hypochlorite electrolyte solution. J. Micromech. Microeng. 2007, 17, 1737–1745. [Google Scholar] [CrossRef]
- Lee, K.B.; Lin, L.W. Electrolyte-based on-demand and disposable microbattery. J. Microelectromech. S. 2003, 12, 840–847. [Google Scholar]
- Sammoura, F.; Lee, K.B.; Lin, L. Water-activated disposable and long shelf life microbatteries. Sens. Actuators A Phys. 2004, 111, 79–86. [Google Scholar] [CrossRef]
- Thom, N.K.; Yeung, K.; Pillion, M.B.; Phillips, S.T. “Fluidic batteries” as low-cost sources of power in paper-based microfluidic devices. Lab Chip 2012, 12, 1768–1770. [Google Scholar] [CrossRef] [PubMed]
- Mokhtar, M.; Talib, M.Z.M.; Majlan, E.H.; Tasirin, S.M.; Ramli, W.M.F.W.; Daud, W.R.W.; Sahari, J. Recent developments in materials for aluminum-air batteries: A review. J. Ind. Eng. Chem. 2015, 32, 1–20. [Google Scholar] [CrossRef]
- Abbasi, H.; Salehi, S.; Ghorbani, R.; Torabi, F.; Amidpour, M. Design and manufacturing of a micro zinc-air fuel cell for mobile applications. Iranica J. Energ. Environ. 2013, 4, 110–115. [Google Scholar] [CrossRef][Green Version]
- Liu, Y.; Qian, S.; Wenzhang, L.; Adair, K.R.; Li, J.; Sun, X. A comprehensive review on recent progress in aluminum-air batteries. Green Energ. Environ. 2017, 2, 246–277. [Google Scholar] [CrossRef]
- Cardenas-Valencia, A.M.; Challa, V.R.; Fries, D.; Langebrake, L.; Benson, R.F.; Bhansali, S. A micro-fluidic galvanic cell as an on-chip power source. Sens. Actuators B Chem. 2003, 95, 406–413. [Google Scholar] [CrossRef]
- Li, Q.; Bjerrum, N.J. Aluminum as anode for energy storage and conversion: A review. J. Power Sources 2002, 110, 1–10. [Google Scholar] [CrossRef]
- Chen, L.D.; Norskov, J.K.; Luntz, A.C. Al-air batteries: Fundamental thermodynamic limitations from first-principles theory. J. Phys. Chem. Lett. 2015, 6, 175–179. [Google Scholar] [CrossRef] [PubMed]


© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sachsenheimer, K.; Richter, C.; Helmer, D.; Kotz, F.; Rapp, B.E. A Nontoxic Battery with 3D-Printed Housing for On-Demand Operation of Microcontrollers in Microfluidic Sensors. Micromachines 2019, 10, 588. https://doi.org/10.3390/mi10090588
Sachsenheimer K, Richter C, Helmer D, Kotz F, Rapp BE. A Nontoxic Battery with 3D-Printed Housing for On-Demand Operation of Microcontrollers in Microfluidic Sensors. Micromachines. 2019; 10(9):588. https://doi.org/10.3390/mi10090588
Chicago/Turabian StyleSachsenheimer, Kai, Christiane Richter, Dorothea Helmer, Frederik Kotz, and Bastian Ernst Rapp. 2019. "A Nontoxic Battery with 3D-Printed Housing for On-Demand Operation of Microcontrollers in Microfluidic Sensors" Micromachines 10, no. 9: 588. https://doi.org/10.3390/mi10090588
APA StyleSachsenheimer, K., Richter, C., Helmer, D., Kotz, F., & Rapp, B. E. (2019). A Nontoxic Battery with 3D-Printed Housing for On-Demand Operation of Microcontrollers in Microfluidic Sensors. Micromachines, 10(9), 588. https://doi.org/10.3390/mi10090588





