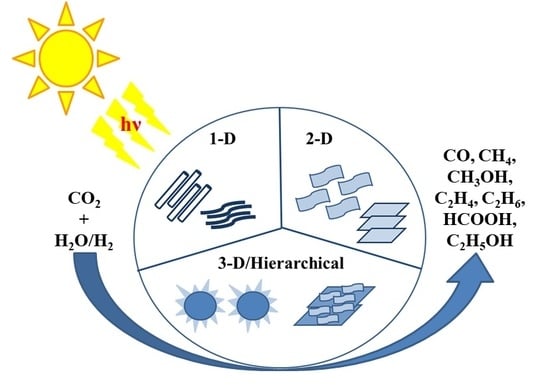
TiO2 Based Nanostructures for Photocatalytic CO2 Conversion to Valuable Chemicals
Abstract
1. Introduction
2. Photocatalytic CO2 Conversion: Fundamentals and Mechanism
3. One-Dimensional (1-D) Nanostructured Photocatalysts
4. Two Dimensional (2-D) Nanostructured Photocatalysts
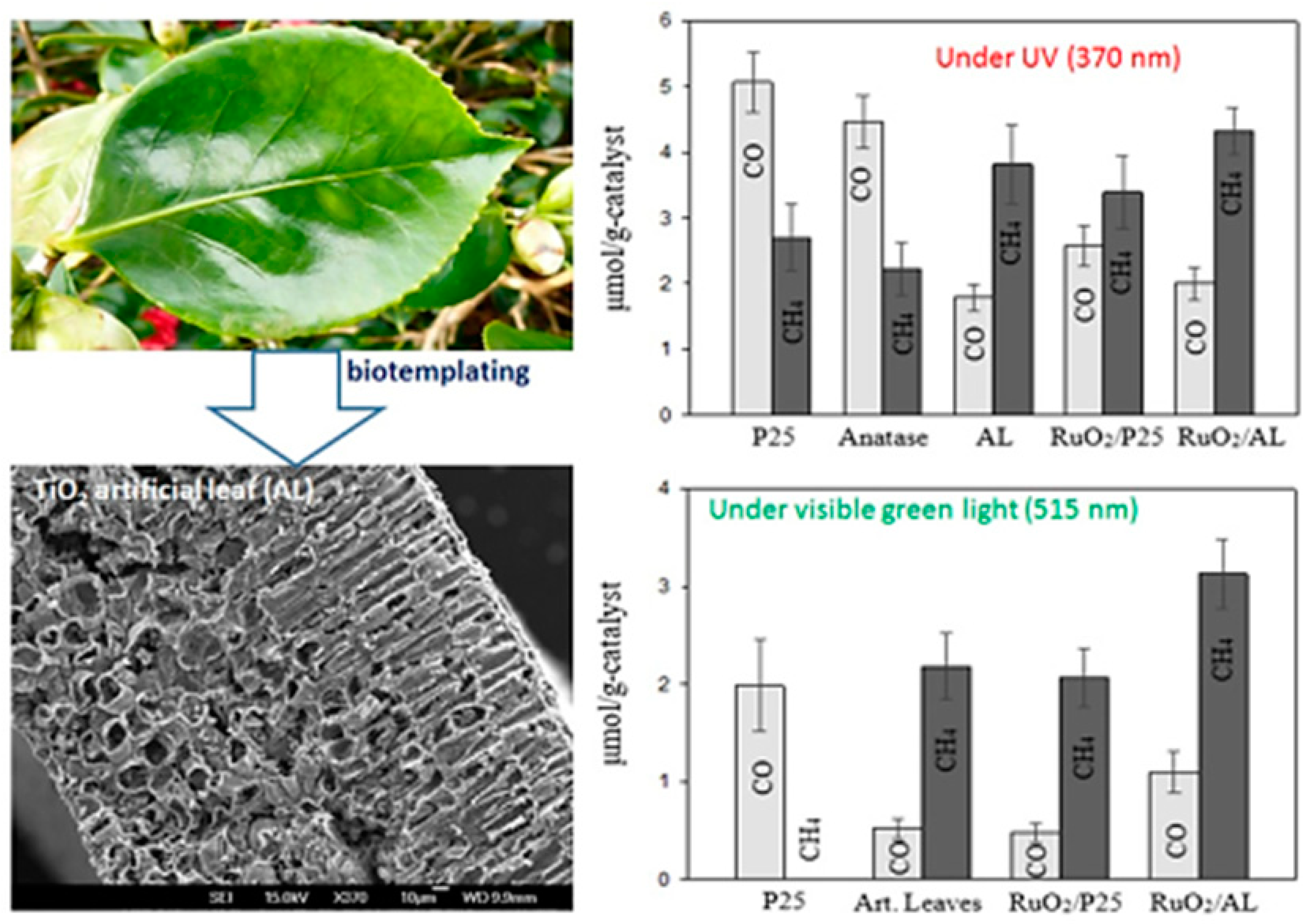
5. Hierarchical Nanostructures: A Dynamic and Potent Approach
6. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Szulejko, J.E.; Kumar, P.; Deep, A.; Kim, K.H. Global warming projections to 2100 using simple CO2 greenhouse gas modeling and comments on CO2 climate sensitivity factor. Atmos. Pollut. Res. 2017, 8, 136–140. [Google Scholar] [CrossRef]
- Kim, D.; Sakimoto, K.K.; Hong, D.; Yang, P. Artificial photosynthesis for sustainable fuel and chemical production. Angew. Chem. Int. Ed. 2015, 54, 3259–3266. [Google Scholar] [CrossRef] [PubMed]
- Barber, J. Photosynthetic energy conversion: Natural and artificial. Chem. Soc. Rev. 2009, 38, 185–196. [Google Scholar] [CrossRef] [PubMed]
- Neaţu, Ş.; Maciá-Agulló, J.A.; Garcia, H. Solar light photocatalytic CO2 reduction: General considerations and selected bench-mark photocatalysts. Int. J. Mol. Sci. 2014, 15, 5246–5262. [Google Scholar] [CrossRef] [PubMed]
- Ali, S.; Razzaq, A.; In, S.-I. Development of graphene based photocatalysts for CO2 reduction to C1 chemicals: A brief overview. Catal. Today 2018. [Google Scholar] [CrossRef]
- Fujishima, A.; Honda, K. Electrochemical Photolysis of Water One and Two-dimensional Structure of Poly (L-Alanine) shown by Specific Heat Measurements at Low. Nature 1972, 238, 37–38. [Google Scholar] [CrossRef] [PubMed]
- Liu, G.; Hoivik, N.; Wang, K.; Jakobsen, H. Engineering TiO2 nanomaterials for CO2 conversion/solar fuels. Sol. Energy Mater. Sol. Cells 2012, 105, 53–68. [Google Scholar] [CrossRef]
- Anpo, M. Photocatalytic reduction of CO2 with H2O on highly dispersed Ti-oxide catalysts as a model of artificial photosynthesis. J. CO2 Util. 2013, 1, 8–17. [Google Scholar] [CrossRef]
- Zhang, B.; Cao, S.; Du, M.; Ye, X.; Wang, Y.; Ye, J. Titanium Dioxide (TiO2) Mesocrystals: Synthesis, Growth Mechanisms and Photocatalytic Properties. Catalysts 2019, 9, 91. [Google Scholar] [CrossRef]
- Dalrymple, O.K.; Stefanakos, E.; Trotz, M.A.; Goswami, D.Y. A review of the mechanisms and modeling of photocatalytic disinfection. Appl. Catal. B Environ. 2010, 98, 27–38. [Google Scholar] [CrossRef]
- Kozak, M.; Mazierski, P.; Żebrowska, J.; Kobylański, M.; Klimczuk, T.; Lisowski, W.; Trykowski, G.; Nowaczyk, G.; Zaleska-Medynska, A. Electrochemically Obtained TiO2/CuxOy Nanotube Arrays Presenting a Photocatalytic Response in Processes of Pollutants Degradation and Bacteria Inactivation in Aqueous Phase. Catalysts 2018, 8, 237. [Google Scholar] [CrossRef]
- In, S.-I.; Nielsen, M.G.; Vesborg, P.C.K.; Hou, Y.; Abrams, B.L.; Henriksen, T.R.; Hansen, O.; Chorkendorff, I. Photocatalytic methane decomposition over vertically aligned transparent TiO2 nanotube arrays. Chem. Commun. Camb. 2011, 47, 2613–2615. [Google Scholar] [CrossRef]
- In, S.-I.; Hou, Y.; Abrams, B.L.; Vesborg, P.C.K.; Chorkendorff, I. Controlled Directional Growth of TiO2 Nanotubes. J. Electrochem. Soc. 2010, 157, E69. [Google Scholar] [CrossRef]
- Ren, Y.; Dong, Y.; Feng, Y.; Xu, J. Compositing Two-Dimensional Materials with TiO2 for Photocatalysis. Catalysts 2018, 8, 590. [Google Scholar] [CrossRef]
- Qu, J.; Sha, L.; Wu, C.; Zhang, Q. Applications of Mechanochemically Prepared Layered Double Hydroxides as Adsorbents and Catalysts: A Mini-Review. Nanomaterials 2019, 9, 80. [Google Scholar] [CrossRef] [PubMed]
- Liao, T.-W.; Verbruggen, S.; Claes, N.; Yadav, A.; Grandjean, D.; Bals, S.; Lievens, P. TiO2 Films Modified with Au Nanoclusters as Self-Cleaning Surfaces under Visible Light. Nanomaterials 2018, 8, 30. [Google Scholar] [CrossRef] [PubMed]
- Xue, Y.; Wu, Z.; He, X.; Yang, X.; Chen, X.; Gao, Z. Constructing a Z-scheme Heterojunction of Egg-Like Core@shell CdS@TiO2 Photocatalyst via a Facile Reflux Method for Enhanced Photocatalytic Performance. Nanomaterials 2019, 9, 222. [Google Scholar] [CrossRef] [PubMed]
- Foura, G.; Chouchou, N.; Soualah, A.; Kouachi, K.; Guidotti, M.; Robert, D. Fe-Doped TiO2 Supported on HY Zeolite for Solar Photocatalytic Treatment of Dye Pollutants. Catalysts 2017, 7, 344. [Google Scholar] [CrossRef]
- Pathakoti, K.; Manubolu, M.; Hwang, H.-M. Chapter 48—Nanotechnology applications for environmental industry. In Handbook of Nanomaterials for Industrial Applications; Mustansar Hussain, C., Ed.; Elsevier: Amsterdam, The Netherlands, 2018; pp. 894–907. [Google Scholar]
- Ge, M.; Cao, C.; Huang, J.; Li, S.; Chen, Z.; Zhang, K.Q.; Al-Deyab, S.S.; Lai, Y. A review of one-dimensional TiO2 nanostructured materials for environmental and energy applications. J. Mater. Chem. A 2016, 4, 6772–6801. [Google Scholar] [CrossRef]
- Li, X.; Yu, J.; Jaroniec, M. Hierarchical photocatalysts. Chem. Soc. Rev. 2016, 45, 2603–2636. [Google Scholar] [CrossRef]
- Parayil, S.K.; Razzaq, A.; Park, S.M.; Kim, H.R.; Grimes, C.A.; In, S.-I. Photocatalytic conversion of CO2 to hydrocarbon fuel using carbon and nitrogen co-doped sodium titanate nanotubes. Appl. Catal. A General 2015, 498, 205–213. [Google Scholar] [CrossRef]
- Razzaq, A.; Sinhamahapatra, A.; Kang, T.H.; Grimes, C.A.; Yu, J.S.; In, S.-I. Efficient solar light photoreduction of CO2 to hydrocarbon fuels via magnesiothermally reduced TiO2 photocatalyst. Appl. Catal. B Environ. 2017, 215, 28–35. [Google Scholar] [CrossRef]
- Sorcar, S.; Hwang, Y.; Grimes, C.A.; In, S.I. Highly enhanced and stable activity of defect-induced titania nanoparticles for solar light-driven CO2 reduction into CH4. Mater. Today 2017, 20, 507–515. [Google Scholar] [CrossRef]
- In, S.; Orlov, A.; Berg, R.; García, F.; Pedrosa-Jimenez, S.; Tikhov, M.S.; Wright, D.S.; Lambert, R.M. Effective visible light-activated B-doped and B,N-codoped TiO2 photocatalysts. J. Am. Chem. Soc. 2007, 129, 13790–13791. [Google Scholar] [CrossRef]
- Kim, M.; Kim, Y.K.; Lim, S.K.; Kim, S.; In, S.I. Efficient visible light-induced H2 production by Au@CdS/TiO2 nanofibers: Synergistic effect of core-shell structured Au@CdS and densely packed TiO2 nanoparticles. Appl. Catal. B Environ. 2015, 166–167, 423–431. [Google Scholar] [CrossRef]
- Lee, H.; In, S.; Horn, M.W. Plasmonic enhancement of CO2 conversion to methane using sculptured copper thin films grown directly on TiO2. Thin Solid Films 2014, 565, 105–110. [Google Scholar] [CrossRef]
- Kim, M.; Razzaq, A.; Kim, Y.K.; Kim, S.; In, S.I. Synthesis and characterization of platinum modified TiO2-embedded carbon nanofibers for solar hydrogen generation. RSC Adv. 2014, 4, 51286–51293. [Google Scholar] [CrossRef]
- Jedsukontorn, T.; Saito, N.; Hunsom, M. Photoinduced Glycerol Oxidation over Plasmonic Au and AuM (M = Pt, Pd and Bi) Nanoparticle-Decorated TiO2 Photocatalysts. Nanomaterials 2018, 8, 269. [Google Scholar] [CrossRef]
- Razzaq, A.; Grimes, C.A.; In, S.I. Facile fabrication of a noble metal-free photocatalyst: TiO2 nanotube arrays covered with reduced graphene oxide. Carbon 2016, 98, 537–544. [Google Scholar] [CrossRef]
- Zubair, M.; Kim, H.; Razzaq, A.; Grimes, C.A.; In, S.I. Solar spectrum photocatalytic conversion of CO2 to CH4 utilizing TiO2 nanotube arrays embedded with graphene quantum dots. J. CO2 Util. 2018, 26, 70–79. [Google Scholar] [CrossRef]
- Tang, B.; Chen, H.; Peng, H.; Wang, Z.; Huang, W. Graphene Modified TiO2 Composite Photocatalysts: Mechanism, Progress and Perspective. Nanomaterials 2018, 8, 105. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.-C.; Gopalan, A.-I.; Sai-Anand, G.; Lee, K.-P.; Kim, W.-J. Preparation of Visible Light Photocatalytic Graphene Embedded Rutile Titanium(IV) Oxide Composite Nanowires and Enhanced NOx Removal. Catalysts 2019, 9, 170. [Google Scholar] [CrossRef]
- Park, S.M.; Razzaq, A.; Park, Y.H.; Sorcar, S.; Park, Y.; Grimes, C.A.; In, S.-I. Hybrid CuxO-TiO2 Heterostructured Composites for Photocatalytic CO2 Reduction into Methane Using Solar Irradiation: Sunlight into Fuel. ACS Omega 2016, 1, 868–875. [Google Scholar] [CrossRef]
- Zubair, M.; Razzaq, A.; Grimes, C.A.; In, S.-I. Cu2ZnSnS4 (CZTS)-ZnO: A noble metal-free hybrid Z-scheme photocatalyst for enhanced solar-spectrum photocatalytic conversion of CO2 to CH4. J. CO2 Util. 2017, 20, 301–311. [Google Scholar] [CrossRef]
- In, S.-I.; Vaughn, D.D.; Schaak, R.E. Hybrid CuO-TiO2−xNx hollow nanocubes for photocatalytic conversion of CO2 into methane under solar irradiation. Angew. Chem. Int. Ed. 2012, 51, 3915–3918. [Google Scholar] [CrossRef] [PubMed]
- Parayil, S.K.; Razzaq, A.; In, S.-I. Formation of Titania-Silica Mixed Oxides in Solvent Mixtures and Their Influences for the Photocatalytic CO2 Conversion to Hydrocarbon. J. Nanosci. Nanotechnol. 2015, 15, 7285–7292. [Google Scholar] [CrossRef]
- White, J.L.; Baruch, M.F.; Pander, J.E.; Hu, Y.; Fortmeyer, I.C.; Park, J.E.; Zhang, T.; Liao, K.; Gu, J.; Yan, Y.; et al. Light-Driven Heterogeneous Reduction of Carbon Dioxide: Photocatalysts and Photoelectrodes. Chem. Rev. 2015, 115, 12888–12935. [Google Scholar] [CrossRef] [PubMed]
- Markewitz, P.; Kuckshinrichs, W.; Leitner, W.; Linssen, J.; Zapp, P.; Bongartz, R.; Schreiber, A.; Müller, T.E. Worldwide innovations in the development of carbon capture technologies and the utilization of CO2. Energy Environ. Sci. 2012, 5, 7281. [Google Scholar] [CrossRef]
- Habisreutinger, S.N.; Schmidt-Mende, L.; Stolarczyk, J.K. Photocatalytic reduction of CO2 on TiO2 and other semiconductors. Angew. Chem. Int. Ed. 2013, 52, 7372–7408. [Google Scholar] [CrossRef] [PubMed]
- Roy, S.C.; Varghese, O.K.; Paulose, M.; Grimes, C.A. Toward Solar Fuels: Photocatalytic Hydrocarbons. ACS Nano 2010, 4, 1259–1278. [Google Scholar] [CrossRef]
- Anpo, M.; Yamashita, H.; Ichihashi, Y.; Ehara, S. Photocatalytic reduction of CO2 with H2O on various titanium oxide catalysts. J. Electroanal. Chem. 1995, 396, 21–26. [Google Scholar] [CrossRef]
- Shkrob, I.A.; Dimitrijevic, N.M.; Marin, T.W.; He, H.; Zapol, P. Heteroatom-transfer coupled photoreduction and carbon dioxide fixation on metal oxides. J. Phys. Chem. C 2012, 116, 9461–9471. [Google Scholar] [CrossRef]
- Tan, S.S.; Zou, L.; Hu, E. Kinetic modelling for photosynthesis of hydrogen and methane through catalytic reduction of carbon dioxide with water vapour. Catal. Today 2008, 131, 125–129. [Google Scholar] [CrossRef]
- He, H.; Zapol, P.; Curtiss, L.A. Computational screening of dopants for photocatalytic two-electron reduction of CO2 on anatase (101) surfaces. Energy Environ. Sci. 2012, 5, 6196. [Google Scholar] [CrossRef]
- Cheng, M.; Yang, S.; Chen, R.; Zhu, X.; Liao, Q.; Huang, Y. Copper-decorated TiO2 nanorod thin films in optofluidic planar reactors for efficient photocatalytic reduction of CO2. Int. J. Hydrogen Energy 2017, 42, 9722–9732. [Google Scholar] [CrossRef]
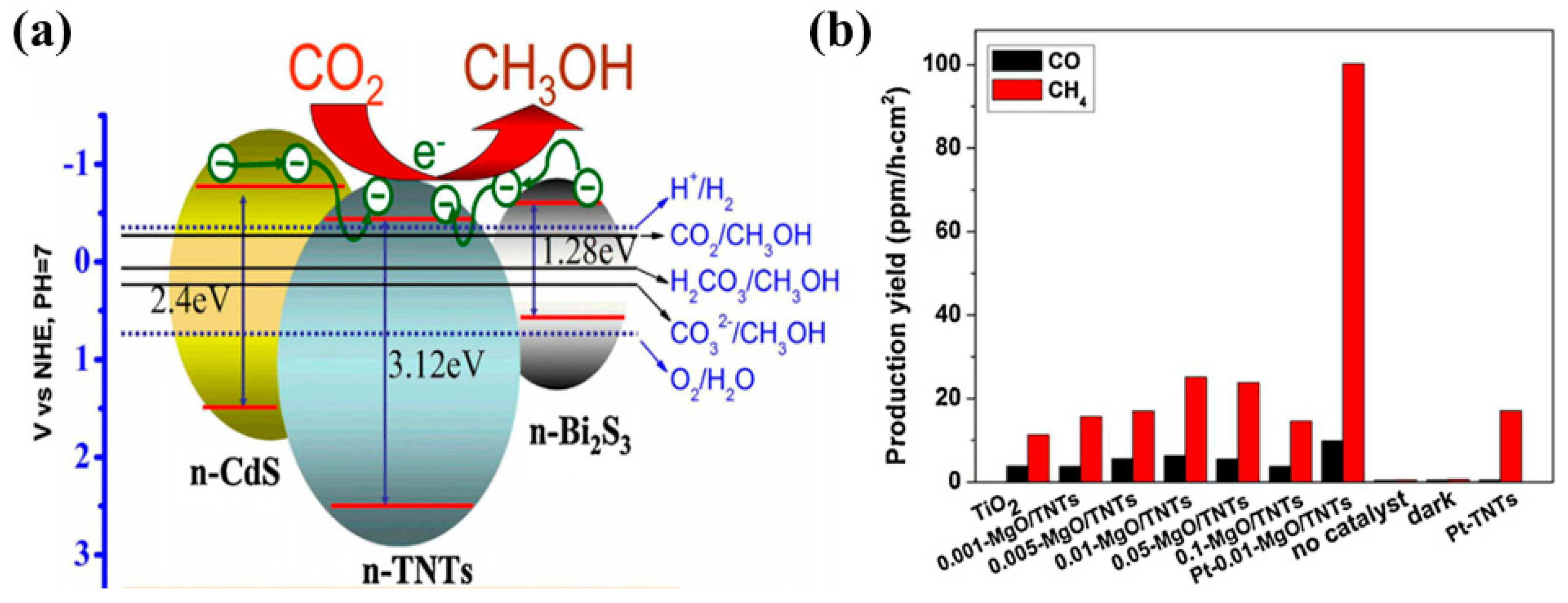
- Li, X.; Liu, H.; Luo, D.; Li, J.; Huang, Y.; Li, H.; Fang, Y.; Xu, Y.; Zhu, L. Adsorption of CO2 on heterostructure CdS(Bi2S3)/TiO2 nanotube photocatalysts and their photocatalytic activities in the reduction of CO2 to methanol under visible light irradiation. Chem. Eng. J. 2012, 180, 151–158. [Google Scholar] [CrossRef]
- Li, Q.; Zong, L.; Li, C.; Yang, J. Reprint of “Photocatalytic reduction of CO2 on MgO/TiO2 nanotube films”. Appl. Surf. Sci. 2014, 319, 16–20. [Google Scholar] [CrossRef]
- Qingli, W.; Zhaoguo, Z.; Xudong, C.; Zhengfeng, H.; Peimei, D.; Yi, C.; Xiwen, Z. Photoreduction of CO2 using black TiO2 films under solar light. J. CO2 Util. 2015, 12, 7–11. [Google Scholar] [CrossRef]
- Li, Y.; Zhang, W.; Shen, X.; Peng, P.; Xiong, L.; Yu, Y. Octahedral Cu2O-modified TiO2 nanotube arrays for efficient photocatalytic reduction of CO2. Cuihua Xuebao Chin. J. Catal. 2015, 36, 2229–2236. [Google Scholar] [CrossRef]
- Liu, E.; Qi, L.; Bian, J.; Chen, Y.; Hu, X.; Fan, J.; Liu, H.; Zhu, C.; Wang, Q. A facile strategy to fabricate plasmonic Cu modified TiO2 nano-flower films for photocatalytic reduction of CO2 to methanol. Mater. Res. Bull. 2015, 68, 203–209. [Google Scholar] [CrossRef]
- Cheng, M.; Yang, S.; Chen, R.; Zhu, X.; Liao, Q.; Huang, Y. Visible light responsive CdS sensitized TiO2 nanorod array films for efficient photocatalytic reduction of gas phase CO2. Mol. Catal. 2018, 448, 185–194. [Google Scholar] [CrossRef]
- Li, Y.; Wang, C.; Song, M.; Li, D.; Zhang, X.; Liu, Y. TiO2−x/CoOx photocatalyst sparkles in photothermocatalytic reduction of CO2 with H2O steam. Appl. Catal. B Environ. 2018, 243, 760–770. [Google Scholar] [CrossRef]
- Tahir, M.; Tahir, B.; Amin, N.A.S. Gold-nanoparticle-modified TiO2 nanowires for plasmon-enhanced photocatalytic CO2 reduction with H2 under visible light irradiation. Appl. Surf. Sci. 2015, 356, 1289–1299. [Google Scholar] [CrossRef]
- Tahir, M.; Tahir, B.; Amin, N.A.S.; Zakaria, Z.Y. Photo-induced reduction of CO2 to CO with hydrogen over plasmonic Ag-NPs/TiO2 NWs core/shell hetero-junction under UV and visible light. J. CO2 Util. 2017, 18, 250–260. [Google Scholar] [CrossRef]
- Tahir, M.; Tahir, B.; Amin, N.A.S. Synergistic effect in plasmonic Au/Ag alloy NPs co-coated TiO2 NWs toward visible-light enhanced CO2 photoreduction to fuels. Appl. Catal. B Environ. 2017, 204, 548–560. [Google Scholar] [CrossRef]
- Low, J.; Qiu, S.; Xu, D.; Jiang, C.; Cheng, B. Direct evidence and enhancement of surface plasmon resonance effect on Ag-loaded TiO2 nanotube arrays for photocatalytic CO2 reduction. Appl. Surf. Sci. 2018, 434, 423–432. [Google Scholar] [CrossRef]
- Su, K.Y.; Chen, C.Y.; Wu, R.J. Preparation of Pd/TiO2 nanowires for the photoreduction of CO2 into renewable hydrocarbon fuels. J. Taiwan Inst. Chem. Eng. 2019, 96, 409–418. [Google Scholar] [CrossRef]
- Song, G.; Xin, F.; Yin, X. Photocatalytic reduction of carbon dioxide over ZnFe2O4/TiO2 nanobelts heterostructure in cyclohexanol. J. Colloid Interface Sci. 2015, 442, 60–66. [Google Scholar] [CrossRef]
- Yang, J.; Wen, Z.; Shen, X.; Dai, J.; Li, Y.; Li, Y. A comparative study on the photocatalytic behavior of graphene-TiO2 nanostructures: Effect of TiO2 dimensionality on interfacial charge transfer. Chem. Eng. J. 2018, 334, 907–921. [Google Scholar] [CrossRef]
- Kar, P.; Zeng, S.; Zhang, Y.; Vahidzadeh, E.; Manuel, A.; Kisslinger, R.; Alam, K.M.; Thakur, U.K.; Mahdi, N.; Kumar, P. High rate CO2 photoreduction using flame annealed TiO2 nanotubes. Appl. Catal. B Environ. 2019, 243, 522–536. [Google Scholar] [CrossRef]
- Zhou, S.; Liu, Y.; Li, J.; Wang, Y.; Jiang, G.; Zhao, Z.; Wang, D.; Duan, A.; Liu, J.; Wei, Y. Facile in situ synthesis of graphitic carbon nitride (g-C3N4)-N-TiO2 heterojunction as an efficient photocatalyst for the selective photoreduction of CO2 to CO. Appl. Catal. B Environ. 2014, 158–159, 20–29. [Google Scholar] [CrossRef]
- Crake, A.; Christoforidis, K.C.; Godin, R.; Moss, B.; Kafizas, A.; Zafeiratos, S.; Durrant, J.R.; Petit, C. Titanium dioxide/carbon nitride nanosheet nanocomposites for gas phase CO2 photoreduction under UV-visible irradiation. Appl. Catal. B Environ. 2019, 242, 369–378. [Google Scholar] [CrossRef]
- Shi, H.; Long, S.; Hu, S.; Hou, J.; Ni, W.; Song, C.; Li, K.; Gurzadyan, G.G.; Guo, X. Interfacial charge transfer in 0D/2D defect-rich heterostructures for efficient solar-driven CO2 reduction. Appl. Catal. B Environ. 2019, 245, 760–769. [Google Scholar] [CrossRef]
- Qamar, S.; Lei, F.; Liang, L.; Gao, S.; Liu, K.; Sun, Y.; Ni, W.; Xie, Y. Ultrathin TiO2 flakes optimizing solar light driven CO2 reduction. Nano Energy 2016, 26, 692–698. [Google Scholar] [CrossRef]
- Liu, Y.; Miao, C.; Yang, P.; He, Y.; Feng, J.; Li, D. Synergetic promotional effect of oxygen vacancy-rich ultrathin TiO2 and photochemical induced highly dispersed Pt for photoreduction of CO2 with H2O. Appl. Catal. B Environ. 2019, 244, 919–930. [Google Scholar] [CrossRef]
- He, Z.; Tang, J.; Shen, J.; Chen, J.; Song, S. Enhancement of photocatalytic reduction of CO2 to CH4 over TiO2 nanosheets by modifying with sulfuric acid. Appl. Surf. Sci. 2016, 364, 416–427. [Google Scholar] [CrossRef]
- Alves Melo Júnior, M.; Morais, A.; Nogueira, A.F. Boosting the solar-light-driven methanol production through CO2 photoreduction by loading Cu2O on TiO2-pillared K2Ti4O9. Microporous Mesoporous Mater. 2016, 234, 1–11. [Google Scholar] [CrossRef]
- Low, J.; Zhang, L.; Tong, T.; Shen, B.; Yu, J. TiO2/MXene Ti3C2 composite with excellent photocatalytic CO2 reduction activity. J. Catal. 2018, 361, 255–266. [Google Scholar] [CrossRef]
- Yuan, L.; Lu, K.Q.; Zhang, F.; Fu, X.; Xu, Y.J. Unveiling the interplay between light-driven CO2 photocatalytic reduction and carbonaceous residues decomposition: A case study of Bi2WO6-TiO2 binanosheets. Appl. Catal. B Environ. 2018, 237, 424–431. [Google Scholar] [CrossRef]
- Adekoya, D.O.; Tahir, M.; Amin, N.A.S. g-C3N4/(Cu/TiO2) nanocomposite for enhanced photoreduction of CO2 to CH3OH and HCOOH under UV/visible light. J. CO2 Util. 2017, 18, 261–274. [Google Scholar] [CrossRef]
- Linares, N.; Silvestre-Albero, A.M.; Serrano, E.; Silvestre-Albero, J.; García-Martínez, J. Mesoporous materials for clean energy technologies. Chem. Soc. Rev. 2014, 43, 7681–7717. [Google Scholar] [CrossRef] [PubMed]
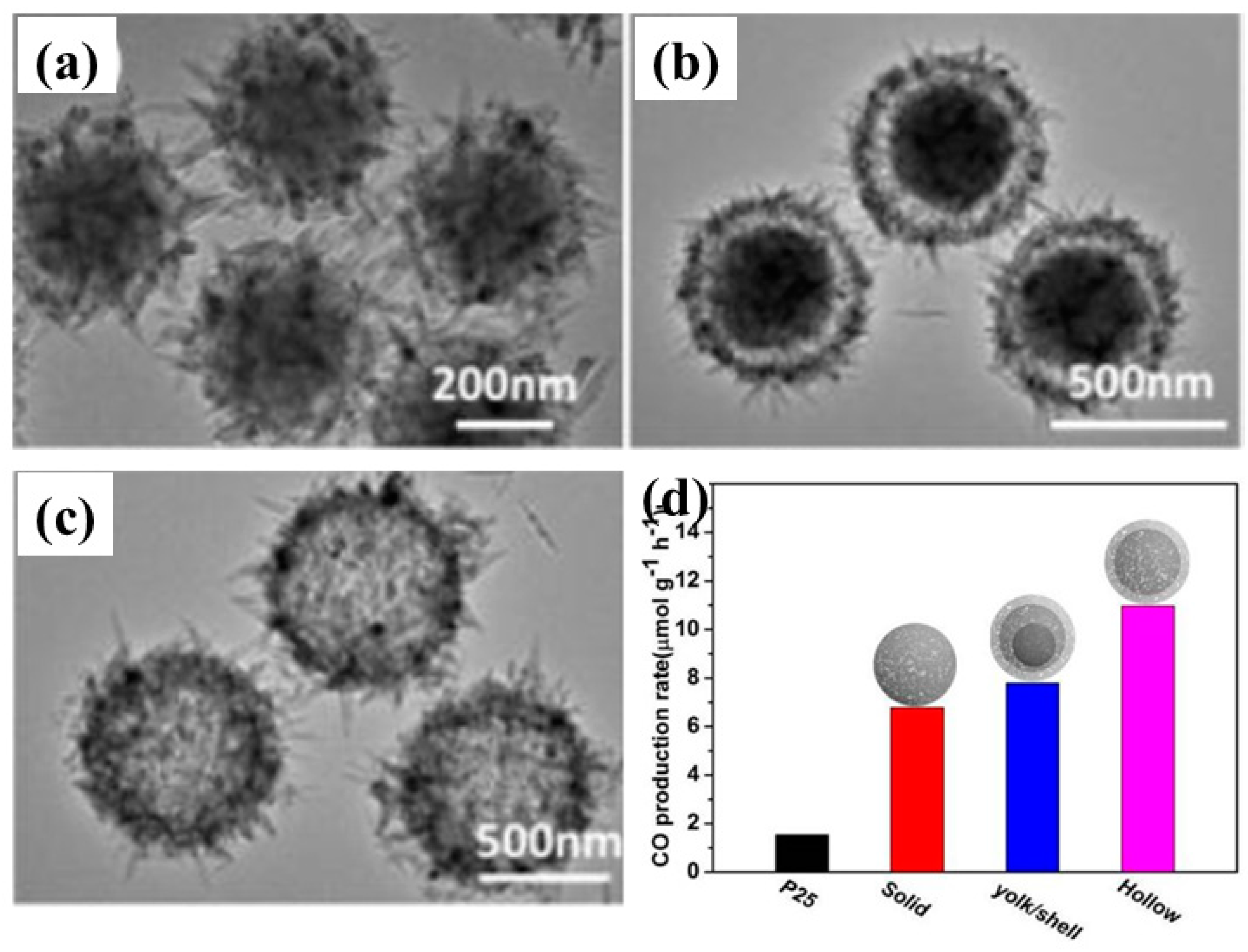
- Wang, H.; Wu, D.; Wu, W.; Wang, D.; Gao, Z.; Xu, F.; Cao, K.; Jiang, K. Preparation of TiO2 microspheres with tunable pore and chamber size for fast gaseous diffusion in photoreduction of CO2 under simulated sunlight. J. Colloid Interface Sci. 2019, 539, 194–202. [Google Scholar] [CrossRef] [PubMed]
- Hashemizadeh, I.; Golovko, V.B.; Choi, J.; Tsang, D.C.W.; Yip, A.C.K. Photocatalytic reduction of CO2 to hydrocarbons using bio-templated porous TiO2 architectures under UV and visible light. Chem. Eng. J. 2018, 347, 64–73. [Google Scholar] [CrossRef]
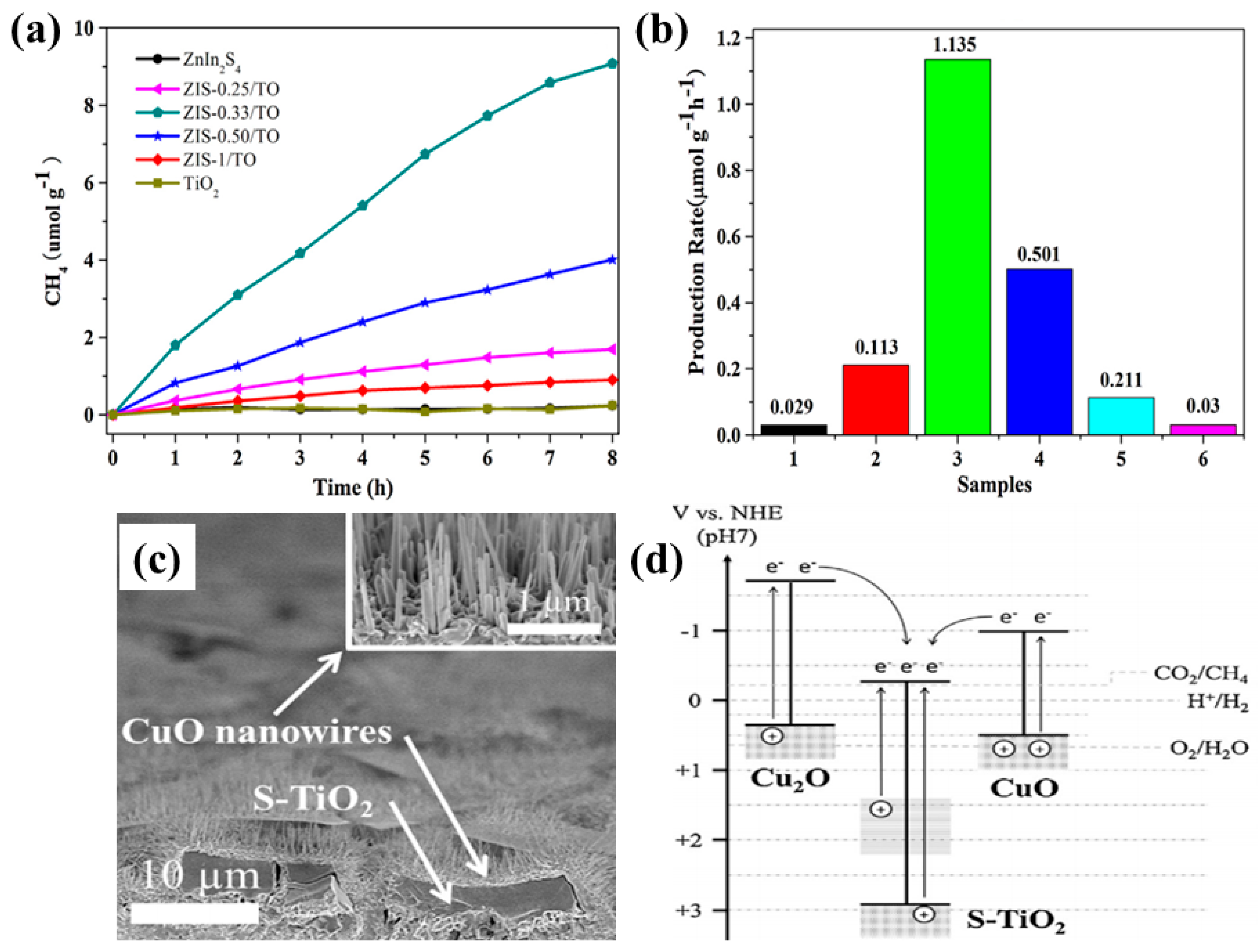
- Yang, G.; Chen, D.; Ding, H.; Feng, J.; Zhang, J.Z.; Zhu, Y.; Hamid, S.; Bahnemann, D.W. Well-designed 3D ZnIn2S4 nanosheets/TiO2 nanobelts as direct Z-scheme photocatalysts for CO2 photoreduction into renewable hydrocarbon fuel with high efficiency. Appl. Catal. B Environ. 2017, 219, 611–618. [Google Scholar] [CrossRef]
- Kim, H.R.; Razzaq, A.; Grimes, C.A.; In, S.I. Heterojunction p-n-p Cu2O/S-TiO2/CuO: Synthesis and application to photocatalytic conversion of CO2 to methane. J. CO2 Util. 2017, 20, 91–96. [Google Scholar] [CrossRef]
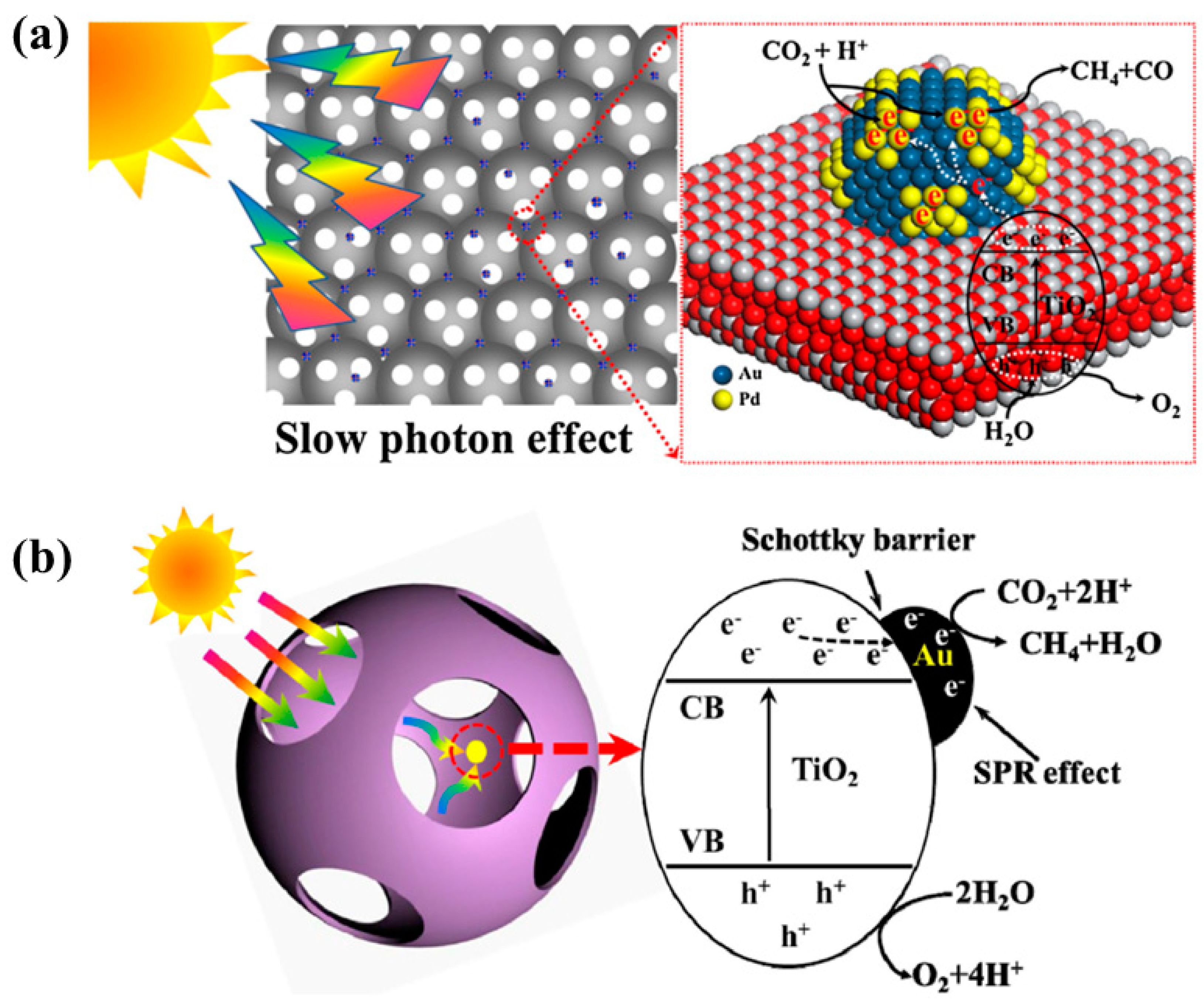
- Jiao, J.; Wei, Y.; Zhao, Y.; Zhao, Z.; Duan, A.; Liu, J.; Pang, Y.; Li, J.; Jiang, G.; Wang, Y. AuPd/3DOM-TiO2 catalysts for photocatalytic reduction of CO2: High efficient separation of photogenerated charge carriers. Appl. Catal. B Environ. 2017, 209, 228–239. [Google Scholar] [CrossRef]
- Jiao, J.; Wei, Y.; Zhao, Z.; Zhong, W.; Liu, J.; Li, J.; Duan, A.; Jiang, G. Synthesis of 3D ordered macroporous TiO2-supported Au nanoparticle photocatalysts and their photocatalytic performances for the reduction of CO2 to methane. Catal. Today 2015, 258, 319–326. [Google Scholar] [CrossRef]
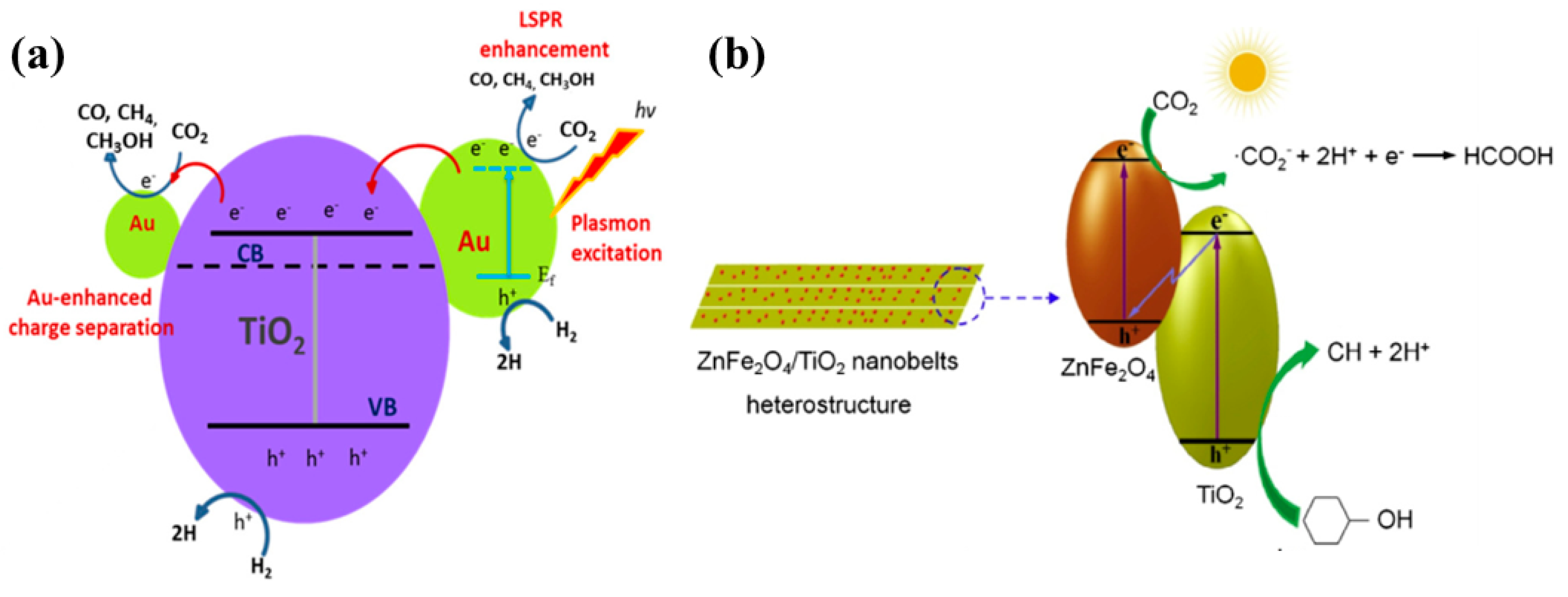
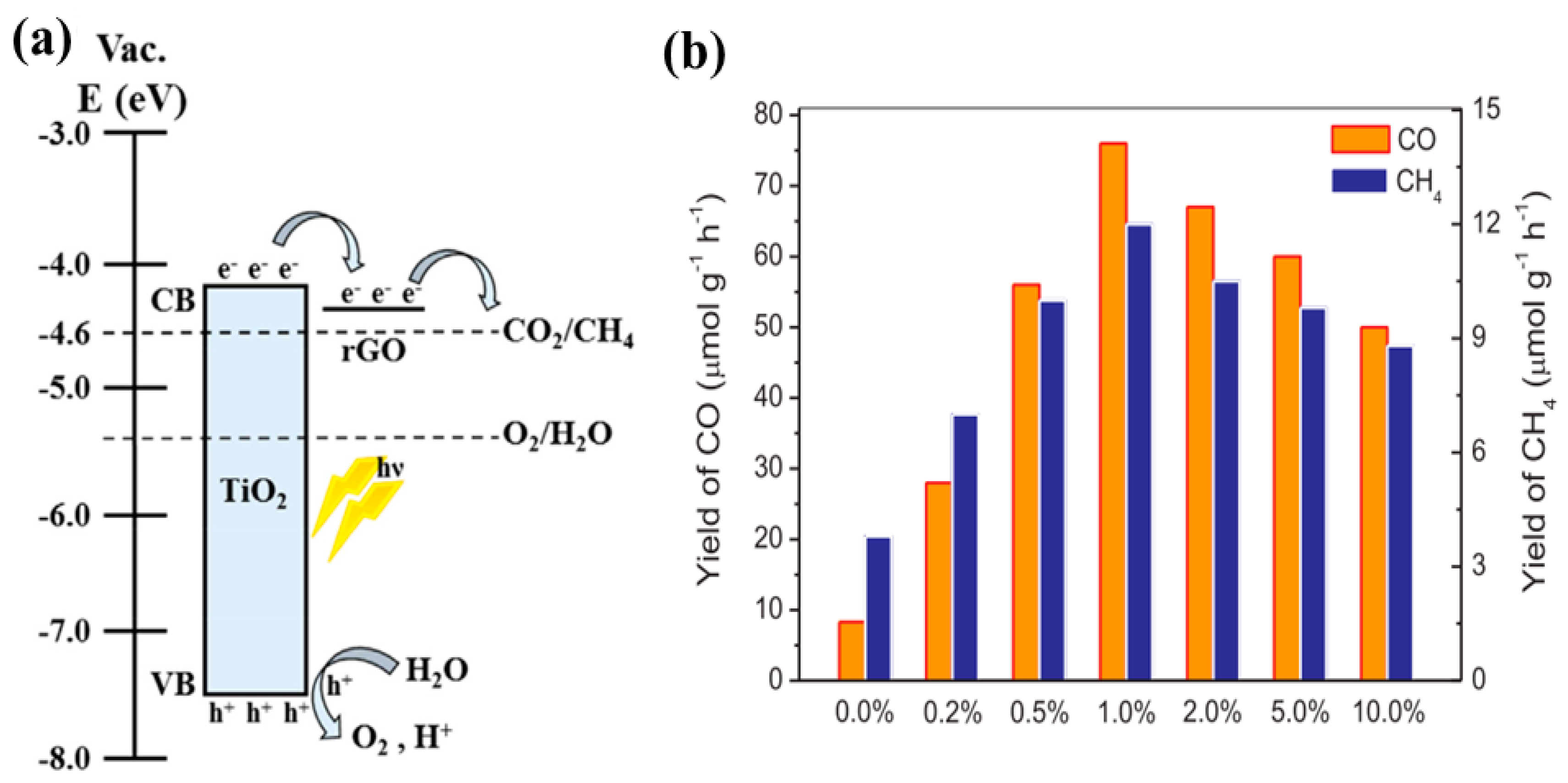
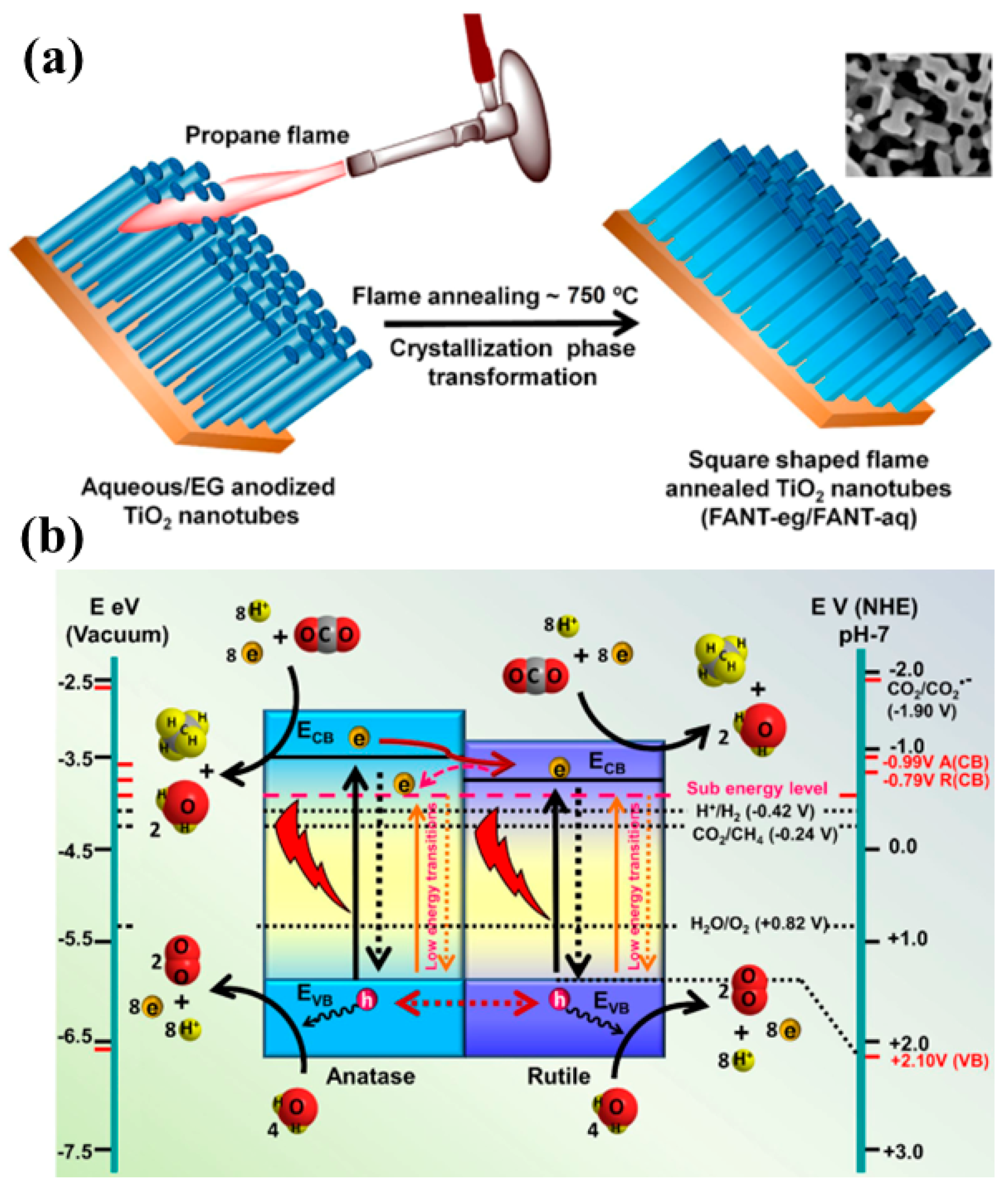
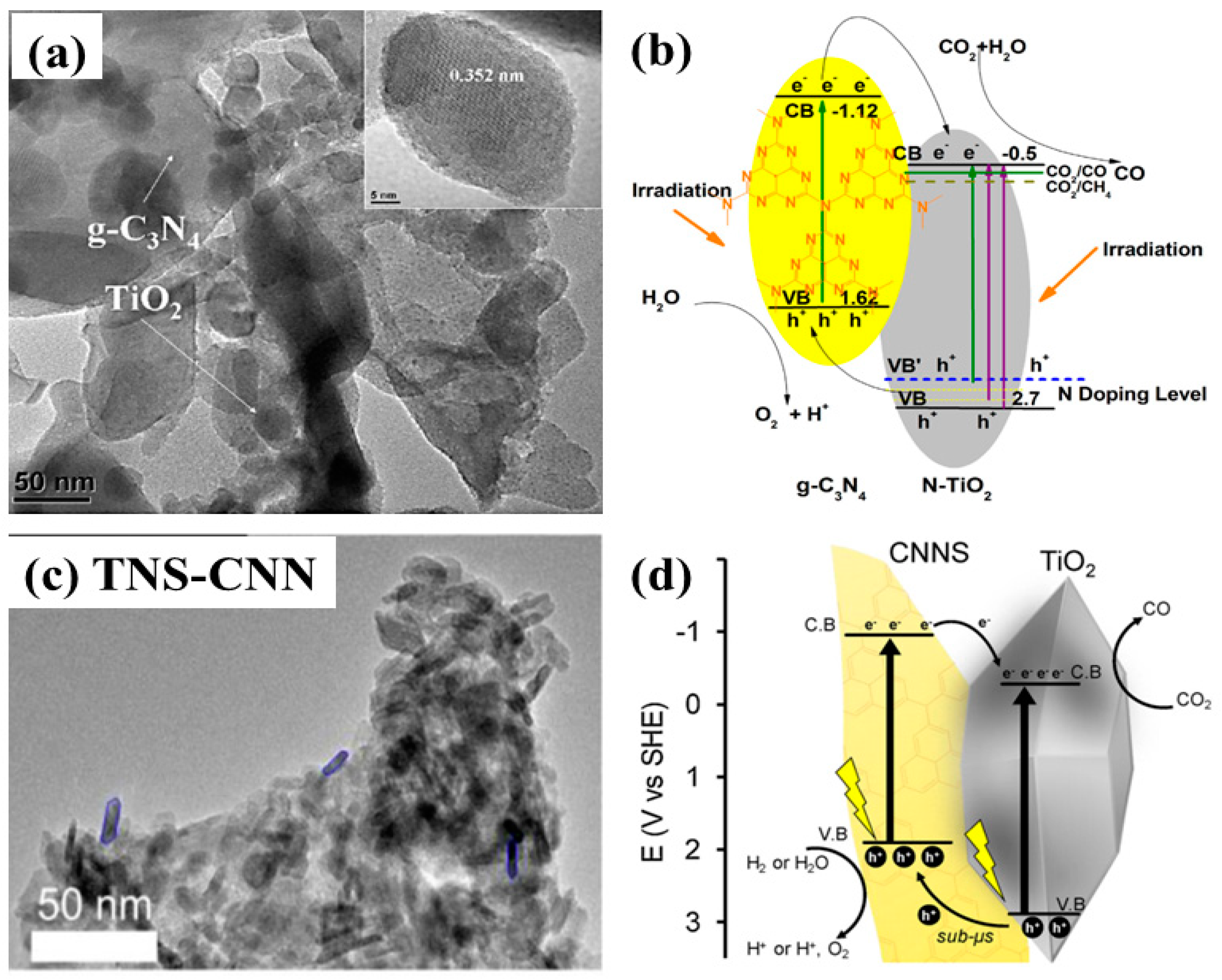
| No. | Reactions | E° vs. NHE (V) |
|---|---|---|
| 1 | CO2 + 2H+ + 2e− → HCOOH | −0.61 V |
| 2 | CO2 + 2H+ + 2e− → CO + H2O | −0.53 V |
| 3 | CO2 + 4H+ + 4e− → HCHO + H2O | −0.48 V |
| 4 | CO2 + 6H+ + 6e− → CH3OH + H2O | −0.38 V |
| 5 | CO2 + 8H+ + 8e− → CH4 + 2H2O | −0.24 V |
| 6 | 2H+ + 2e− → H2 | −0.41 V |
| Water Oxidation Reactio | ||
| 7 | 2H2O → O2 + 4H+ + 4e− | +0.81V |
| 1-D Nanostructure | Light Source and Reactants | Production Rate of Value-Added Chemicals | Key Parameters for Improved Performance | Ref. |
|---|---|---|---|---|
| CdS/TiO2 nanotubes and Bi2S3/TiO2 nanotubes | 500 W Xenon lamp CO2 bubbled through a solution of Sodium hydroxide and sodium nitrite | After 5 h of irradiation CH3OH: 102.5 µmol/L for TNT CH3OH: 159.5 µmol/L for CdS/TNT CH3OH and 224.6 µmol/L for Bi2S3/TNT | Improved surface area Light absorption enhancement Improved CO2 adsorption Efficient electron–hole separation | [47] |
| MgO amorphous layers on Pt loaded TiO2 nanotubes networks | 300 W Hg lamp CO2 bubbled through water | CO: 10.4 ppm/h cm−2, and CH4: 100.2 ppm/h cm−2 for Pt loaded on 0.01 M MgO coated TiO2 Nanotube Networks | Improved CO2 adsorption Enhanced electron–hole separation | [48] |
| Black TiO2 films with grid-like structures | 300 W Xenon lamp CO2 bubbled through water | CO: 115 µmol/g h, and CH4: 12 µmol/g h | Improved light absorption Improved charge separation due to extended charge lifetime | [49] |
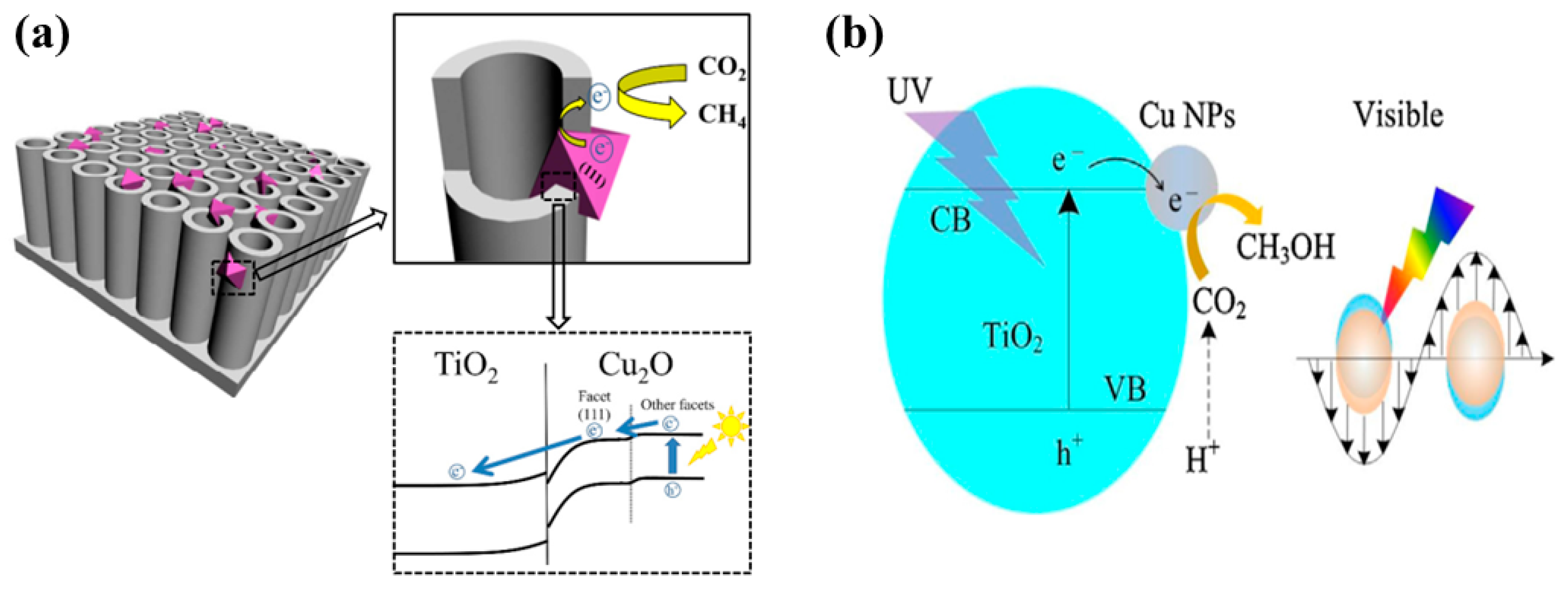
| Cu2O nanoparticles modified TiO2 nanotube arrays | 350 W Xenon lamp with and without UV cutoff filter CO2 bubbled through water | After 4 h of irradiation using simulated solar light CH4: 400 ppm/g for sample with 15 min of Cu2O electrodeposition After 4 h of visible light irradiation CH4: 8 ppm/g for sample with 30 min of Cu2O electrodeposition | Visible light absorption by Cu2O TNT providing a pathway for efficient electron–hole charge separation | [50] |
| Cu deposited on TiO2 nanoflowers films | 500 W Xenon lamp with UV cutoff filter CO2 bubbled through water | CH3OH: 1.8 µmol/cm2 h under UV and Visible light irradiation For 0.5 Cu/TiO2 film | LSPR effect due to Cu nanoparticles and efficient charge transfer property | [51] |
| Au nanoparticles deposited on TiO2 nanowires | HID 35 W Car lamp CO2 and H2 gaseous mixture | CO: 1237 µmol/g h, CH4: 13 µmol/g h, and CH3OH:12.65 µmol/g h For 0.5 Au TiO2 NW | LSPR effect Efficient photogenerated charge extraction Improved Surface area | [54] |
| ZnFe2O4 nanoparticles on TiO2 nanobelts | 250 W high pressure Hg lamp CO2 and cyclohexanol | After 8 h of UV illumination Cyclohexanone (CH):170.2 µmol/g and Cyclohexyl formate (CF): 178.1 µmol/g With sample containing 9.78 wt. % loading of ZnFe2O4 | Improved Charge separation by a Z-scheme mechanism Enhanced Surface area | [59] |
| TNT arrays covered with rGO TiO2 nanoparticles | 100 W Xenon solar simulator CO2 and Water vapors | CH4: 5.67 ppm/cm2 h | Efficient Charge separation Light absorption enhancement | [30] |
| Ag NPs TiO2 nanowires | 35 W HID car lamp as visible light source 200 W Hg Reflector lamp as a UV light source CO2 and H2 at feed ratio of 1.0, temperature 100 °C and Pressure 1 atm. | CO: 983 µmol/g h CH4: 9.73 µmol/g h CH3OH: 13 µmol/g h From 3% Ag deposited TiO2 NWs | Moderate surface areas LSPR effects and efficient charge separation | [55] |
| Au-Ag NPs TiO2 nanowires | 35 W HID car lamp as visible light source 200 W Hg Reflector lamp as a UV light source CO2 and H2 at feed ratio of 1.0, temperature 100 °C and Pressure 1 atm. | CO: 1813 µmol/g h CH4: 35 µmol/g h C2H4: 0.95 µmol/g h C2H6: 2.52 µmol/g h C3H6: 3.94 µmol/g h C3H8: 3.52 µmol/g h CH3OH: 18.76 µmol/g h From, 2% Ag-0.5% Au deposited TiO2 NWs | Improved surface area LSPR effects and efficient charge separation | [56] |
| Ag loaded TiO2 nanotube arrays (TNT) | 300 W Xenon arc lamp with a 400 nm cutoff filter CO2 and water vapors | CH4: 48 mmol/h m2 Using the sample with electrodeposited Ag NPs at 3 V and 1 min | Schottkey Junction formation and SPR effect leading to injection of hot electrons to TiO2 CB | [57] |
| CdS QDs-Cu2+-TiO2 nanorods (NR) | 300 W solar simulated Xenon lamp CO2 and water vapors mixture Temperature of 80 °C | C2H5OH: 109.12 µmol/g h Using sample with 2 SILAR cycles CdS QDs-0.02 M Cu2+ ion onto TiO2 NRs | Improved surface area Extended light absorption of the photocatalysts | [52] |
| Graphene QDs deposited TiO2 nanotube arrays (TNT) | 100 W Xenon Solar simulator CO2 and water vapors | CH4: 1.98 ppm/h cm2 Employing G-TNT 3 sample prepared with 3 sec electrophoretic deposition time of GQDs | Enhanced light absorption with efficient extraction of photogenerated electron–hole pairs | [31] |
| Graphene TiO2 nanostructures including nanoparticles, nanotubes and nanosheets | 300 W Xenon lamp CO2 and water vapors | CO: 75.8 µmol/g h CH4: 12.3 µmol/g h By 1% graphene TiO2 nanotubes | Increased surface area Increased interaction between the photogenerated electrons Improved CO2 adsorption | [60] |
| Flame annealed TiO2 nanotubes (FANT) | 100 W Xenon solar simulator Also 50 W LED lamps employed as a visible light source CO2 and water vapors | CH4: 156 µmol/g h From FANT-aq, synthesized using water as an electrolyte medium | Visible light activity due to defect mediated performance enhancement Higher density of electromagnetic hotspots for visible light and stronger absorption of UV light | [61] |
| Pd-TiO2 nanowires (NW) | 400 W Hg Lamp CO2 and Water vapors | After 8 h of irradiation CO: 50.4 µmol/g CH4:26.7 µmol/g Using 0.5% Pd-TiO2 NW | Pd NPs acts as an electron transfer mediator leading to improved charge transfer | [58] |
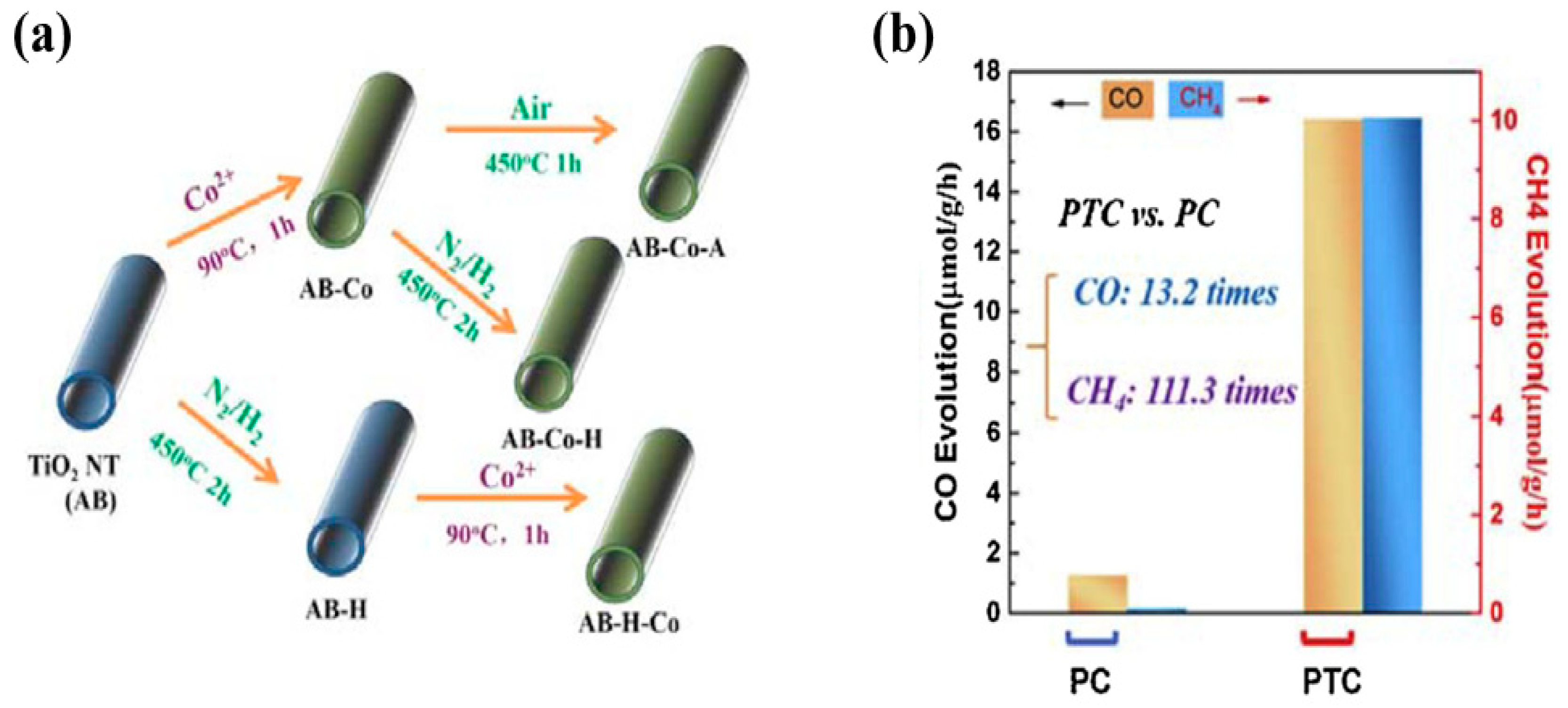
| TiO2 nanotubes (NT) coated with CoOx nanoclusters | 150 W UV lamp with temperature increased to 393 K CO2 and water vapors | After 8 h of irradiation CO: 16.403 µmol/g CH4: 10.051 µmol/g-s | Improved surface areas Efficient photogenerated electrons-holes separation Oxygen vacancies improved CO2 adsorption | [53] |
| 2-D Nanostructure | Light Source and Reactants | Production Rate of Value-Added Chemicals | Key Parameters for Improved Performance | Ref. |
|---|---|---|---|---|
| g-C3N4/N-TiO2 nanosheets | 300 W Xenon arc lamp CO2 bubbled through Deionized water | After 12 h of irradiation CO: 14.73 µmole/g, employing CT7 sample CO: 5.71 µmole/g CH4: 3.94 µmole/g, employing sample CT5 | Moderate surface area Extended light absorption Efficient charge separation at the heterojunction Product selectivity due to band regulation | [62] |
| TiO2 nanosheets modified with sulfuric acid | 500 W Xe Arc lamp CO2 and water vapors | After 4 h of irradiation CH4: 7.63 µmole/g | Acidification facilitates oxidation of water by Ti-OH Ti3+ active sites i.e., oxygen vacancies enhanced adsorption of CO2 Efficient charge separation | [67] |
| Ultrathin TiO2 nanosheets | 300 W Hg Lamp CO2 bubbled through Solution of photocatalyst powder in water | Formate formation 1.9 µmole/g h 450 times higher than counterpart 9 time higher than commercially available anatase TiO2 | Surface area increased Promoted life time of electron Efficient charge separation across the 2-D path | [65] |
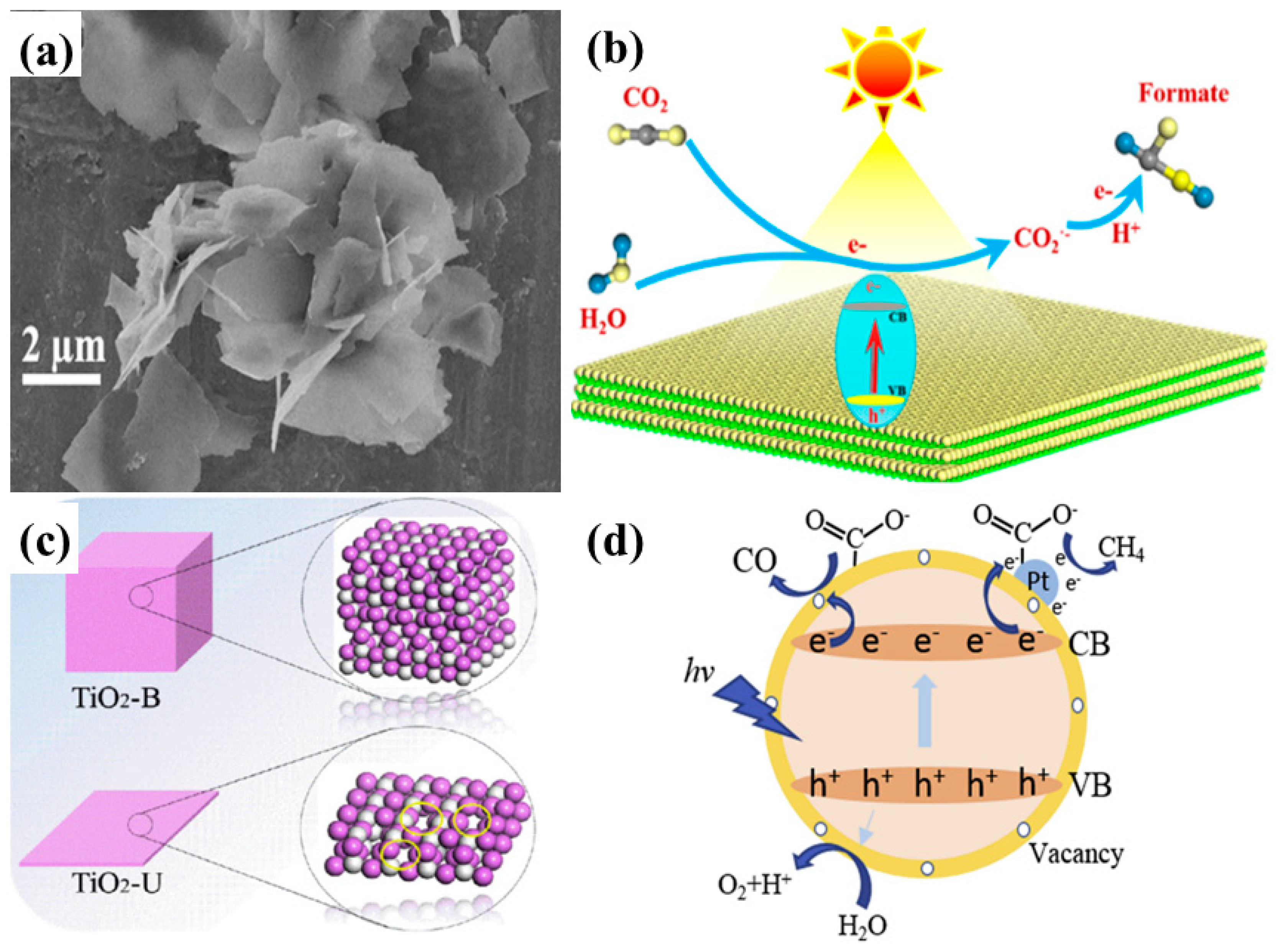
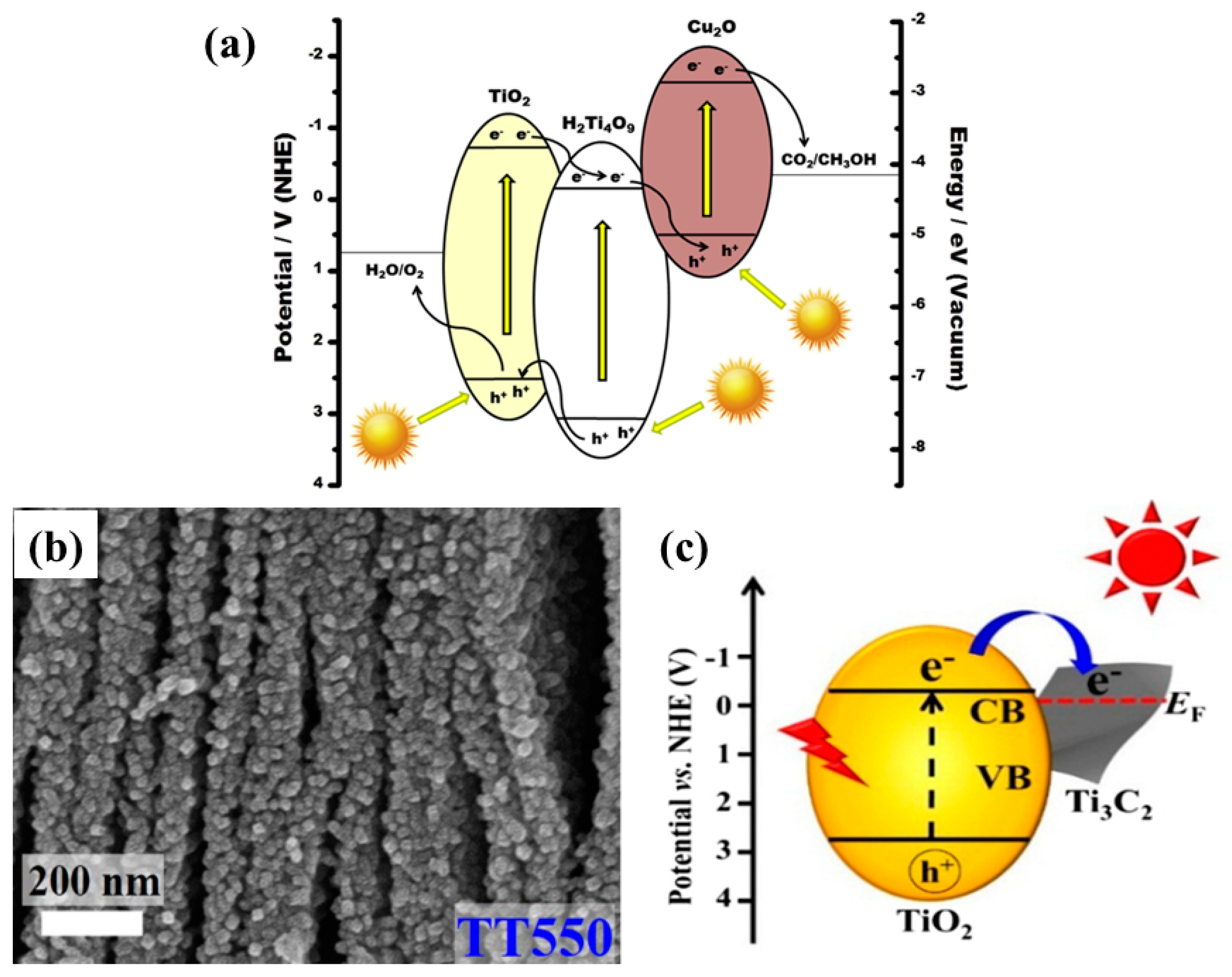
| Cu2O nanoparticles loaded on TiO2 pillared K2Ti4O7 layers | Polychromatic light AM 1.5 from solar simulator CO2 and water vapors | After 5 h of irradiation CH3OH: 2.93 µmole/g 2 times more as compared to pristine sample | Increased surface area Visible light absorption Efficient charge separation | [68] |
| Cu modified g-C3N4 sheets with TiO2 nanoparticles | 254 nm UV Lamp as a UV light source 500 W Xe arc lamp as a visible light source CO2 bubbled through the water solution containing photocatalyst sample | After 8 h of irradiation under UV light CH3OH: 2574 µmol/g, HCOOH: 5069 µmol/g Under Visible light CH3OH: 614 µmol/g, HCOOH:6709 µmol/g Optimum sample: 3 wt.% Cu and 30:70 ratio of g-C3N4 and TiO2 | Extended light absorption and efficient charge separation by copper doping Band edges alignment reflects the selectivity for CH3OH and HCOOH | [71] |
| TiO2 nanoparticles on Ti3C2 nanosheets | UV LED 3 W 365 nm CO2 and water vapor generated in situ by reaction of NaHCO3 and HCl | CH4: 0.22 µmole/ h for 50 mg sample with small amounts of CH3OH and C2H5OH | Improved surface area Nanosheets providing active sites Efficient electron hole separation | [69] |
| Pt nanoparticles loaded ultrathin TiO2 nanosheets | 300 W Xenon lamp CO2 and Water vapors | CH4: 66.4 µmole/h g CO: 54.2 µmole/h | 26 times higher surface area Efficient electron hole separation Improved CO2 adsorption due to defective surface | [66] |
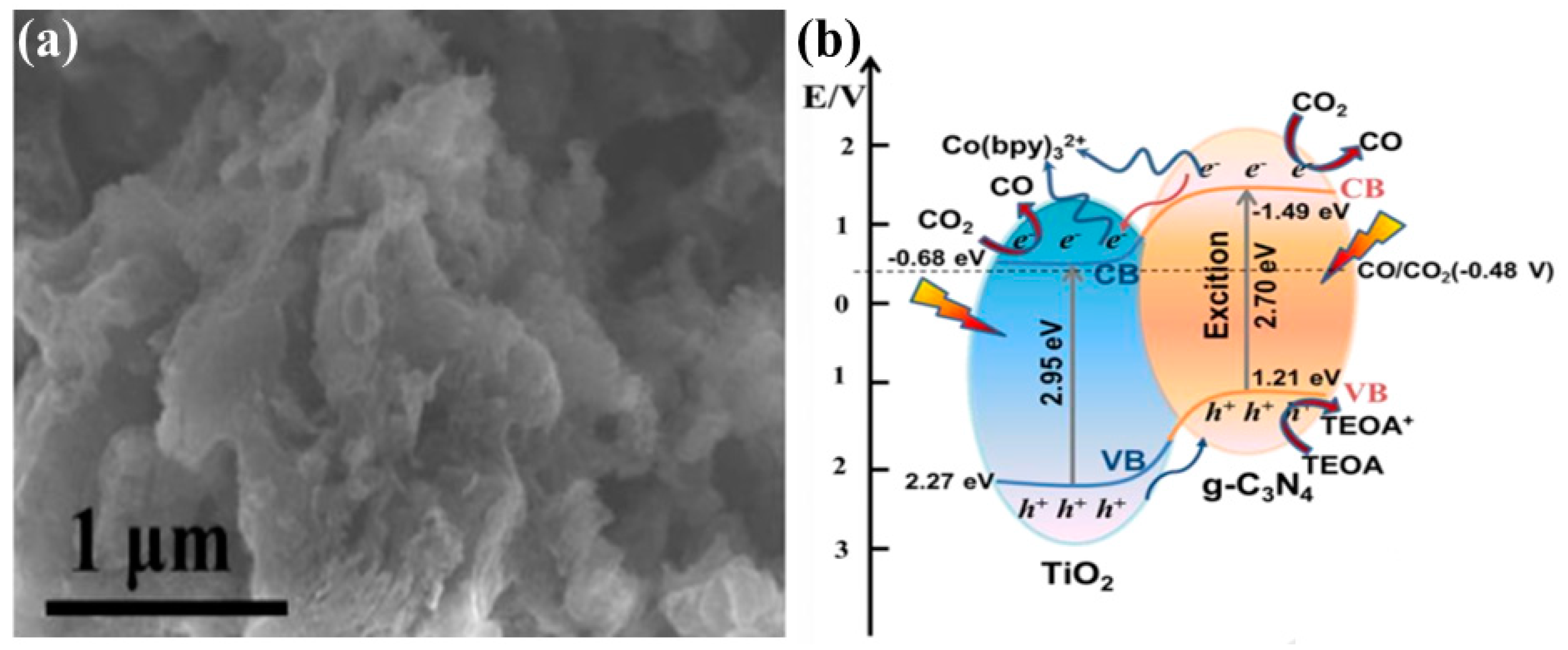
| 2-D g-C3N4 with 0-D TiO2−x nanoparticles | 300 W Xenon lamp CO2 bubbled through solution containing 5 mg photocatalyst dispersed in 5 mL of solution of MeCN/TEOA with cocatalyst of Co(bpy)32+ | After 5 h of irradiation CO: 388.9 μmol/g 5 times higher than pristine g-C3N4 | Promoted charge transfer due to electron channel formed between g-C3N4 and TiO2 | [64] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Razzaq, A.; In, S.-I. TiO2 Based Nanostructures for Photocatalytic CO2 Conversion to Valuable Chemicals. Micromachines 2019, 10, 326. https://doi.org/10.3390/mi10050326
Razzaq A, In S-I. TiO2 Based Nanostructures for Photocatalytic CO2 Conversion to Valuable Chemicals. Micromachines. 2019; 10(5):326. https://doi.org/10.3390/mi10050326
Chicago/Turabian StyleRazzaq, Abdul, and Su-Il In. 2019. "TiO2 Based Nanostructures for Photocatalytic CO2 Conversion to Valuable Chemicals" Micromachines 10, no. 5: 326. https://doi.org/10.3390/mi10050326
APA StyleRazzaq, A., & In, S.-I. (2019). TiO2 Based Nanostructures for Photocatalytic CO2 Conversion to Valuable Chemicals. Micromachines, 10(5), 326. https://doi.org/10.3390/mi10050326