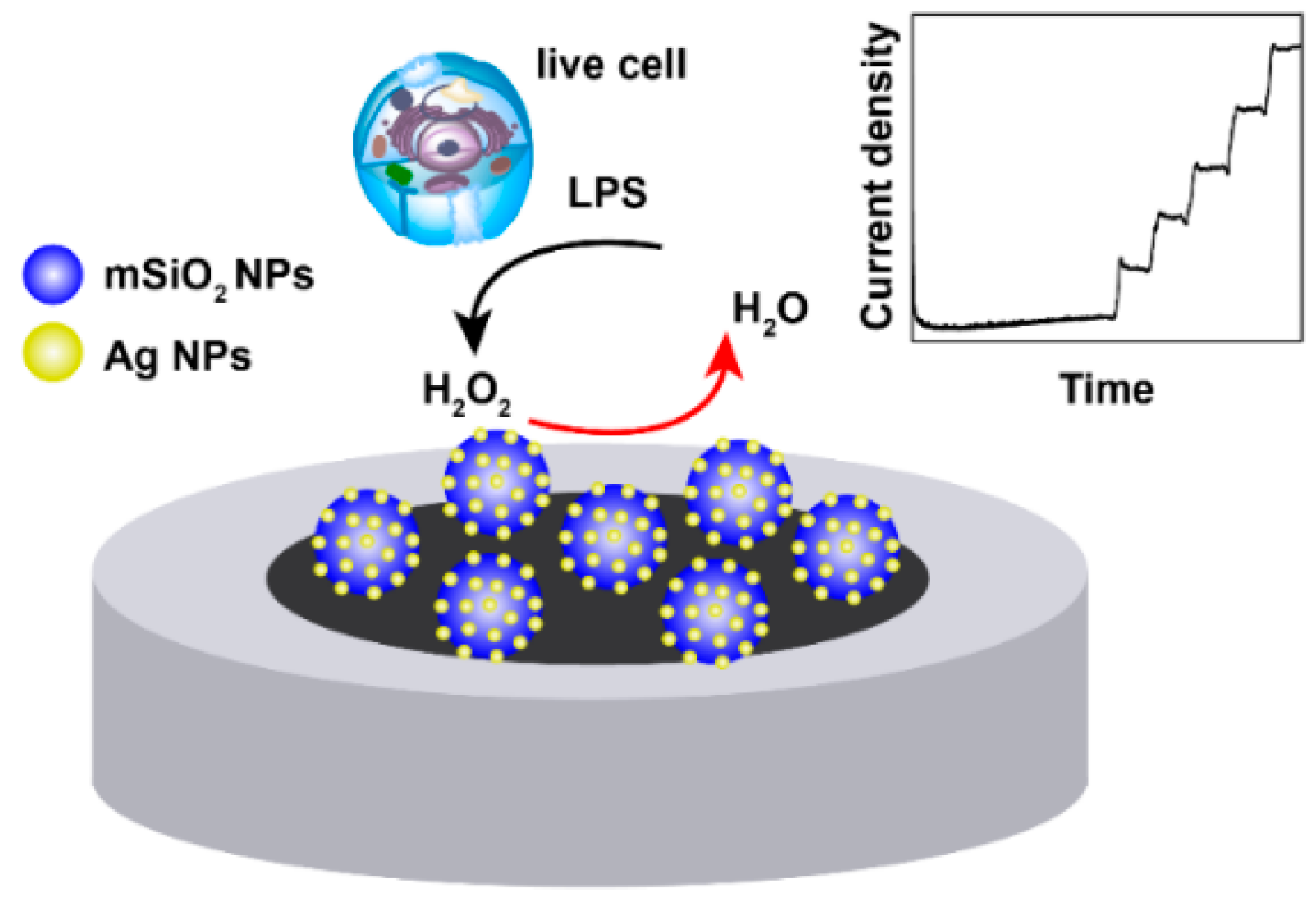
Silver Doped Mesoporous Silica Nanoparticles Based Electrochemical Enzyme-Less Sensor for Determination of H2O2 Released from Live Cells
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Preparation of Ag-mSiO2 Nanoparticles (NPs)
2.3. Preparation of H2O2 Biosensors
2.4. Determination of H2O2
2.4.1. Detection of Standard H2O2 Solution
2.4.2. Detection of H2O2 Released from Pheochromocytoma cells
2.5. Characterization
3. Results
3.1. Characterization of Ag-mSiO2 NPs
3.2. Electrocatalytic Reduction of H2O2
- H2O2 + e− ↔ OHads + OH−
- OHads + OH− ↔ OH−
- 2OH− + 2H+ ↔ 2H2O
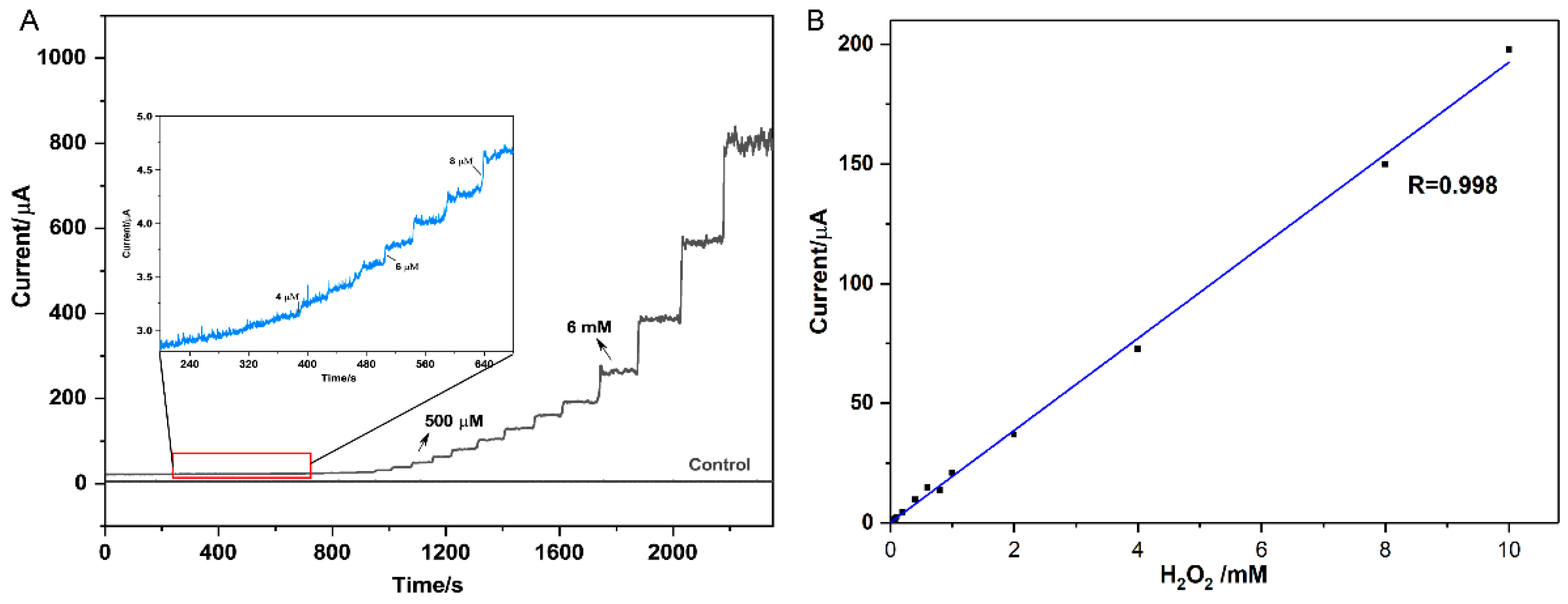
3.3. Amperometric Determination of H2O2
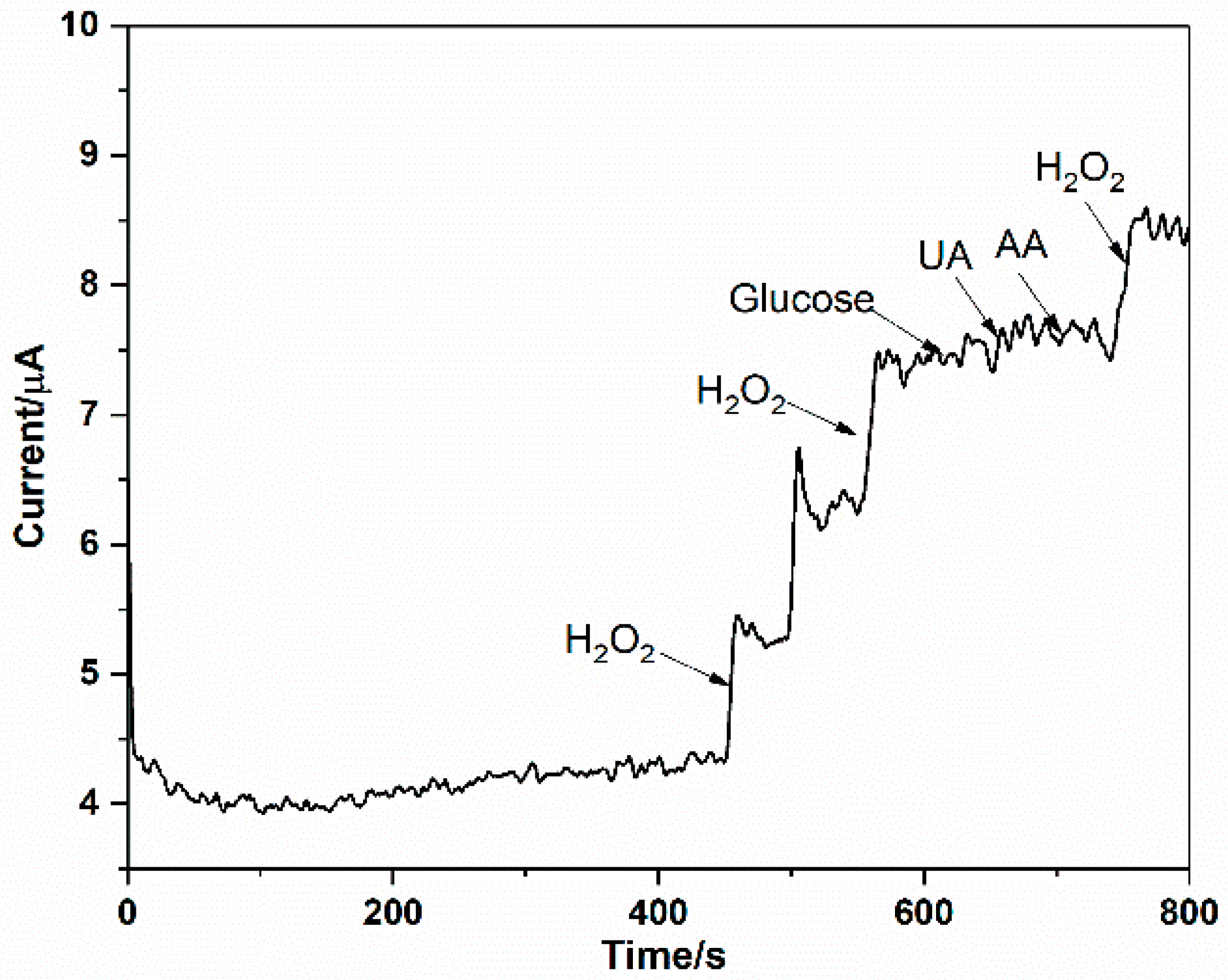
3.4. Interferences Study
3.5. Reproducibility and Stability
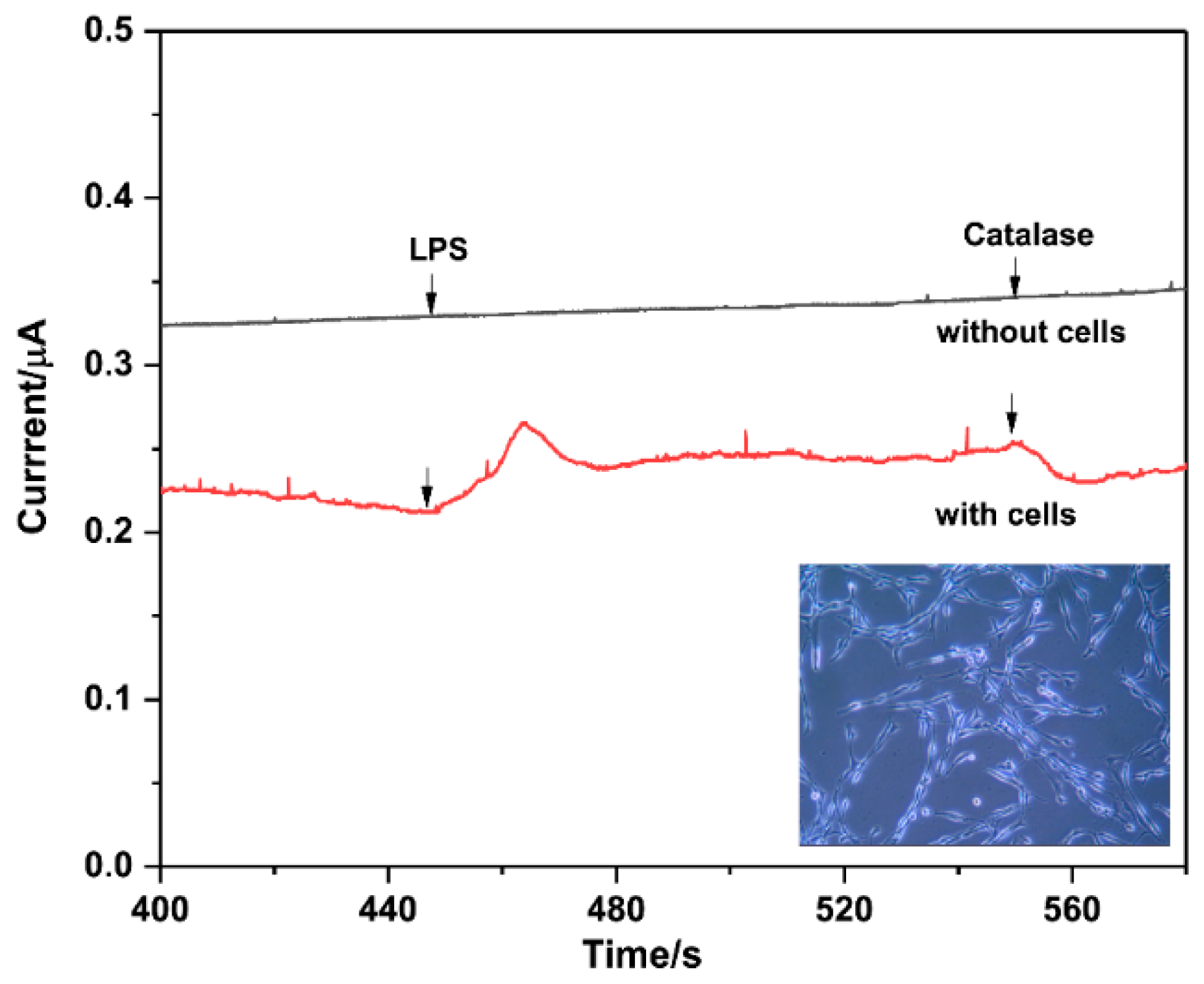
3.6. Determination of H2O2 Released from Pheochromocytoma Cells
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Luo, Y.; Liu, H.; Rui, Q.; Tian, Y. Detection of Extracellular H2O2 Released from Human Liver Cancer Cells Based on TiO2 Nanoneedles with Enhanced Electron Transfer of Cytochrome c. Anal. Chem. 2009, 81, 3035–3041. [Google Scholar] [CrossRef] [PubMed]
- Weinstain, R.; Savariar, E.N.; Felsen, C.N.; Tsien, R.Y. In vivo targeting of hydrogen peroxide by activatable cell-penetrating peptides. J. Am. Chem. Soc. 2014, 136, 874–877. [Google Scholar] [CrossRef] [PubMed]
- Min, Z.; Huang, X.; Liu, L.; Shen, H. Spectrophotometric determination of hydrogen peroxide by using the cleavage of Eriochrome black T in the presence of peroxidase. Talanta 1997, 44, 1407–1412. [Google Scholar] [CrossRef]
- Rapoport, R.; Hanukoglu, I.; Sklan, D. A fluorimetric assay for hydrogen peroxide, suitable for NAD(P)H-dependent superoxide generating redox systems. Anal. Biochem. 1994, 218, 309–313. [Google Scholar] [CrossRef] [PubMed]
- Klassen, N.V.; Marchington, D.; McGowan, H.C.E. H2O2 Determination by the I3- Method and by KMnO4 Titration. Anal. Chem. 1994, 66, 2921–2925. [Google Scholar] [CrossRef]
- Effkemann, S.; Pinkernell, U.; Karst, U. Peroxide analysis in laundry detergents using liquid chromatography. Anal. Chim. Acta 1998, 363, 97–103. [Google Scholar] [CrossRef]
- Yu, G.; Wu, W.; Pan, X.; Zhao, Q.; Wei, X.; Lu, Q. High sensitive and selective sensing of hydrogen peroxide released from pheochromocytoma cells based on Pt-Au bimetallic nanoparticles electrodeposited on reduced graphene sheets. Sensors 2015, 15, 2709–2722. [Google Scholar] [CrossRef]
- Wang, T.; Zhu, H.; Zhuo, J.; Zhu, Z.; Papakonstantinou, P.; Lubarsky, G.; Lin, J.; Li, M. Biosensor based on ultrasmall MoS2 nanoparticles for electrochemical detection of H2O2 released by cells at the nanomolar level. Anal. Chem. 2013, 85, 10289–10295. [Google Scholar] [CrossRef] [PubMed]
- Miao, P.; Wang, B.; Yin, J.; Chen, X.; Tang, Y. Electrochemical tracking hydrogen peroxide secretion in live cells based on autocatalytic oxidation reaction of silver nanoparticles. Electrochem. Commun. 2015, 53, 37–40. [Google Scholar] [CrossRef]
- Ensafi, A.A.; Rezaloo, F.; Rezaei, B. Electrochemical sensor based on porous silicon/silver nanocomposite for the determination of hydrogen peroxide. Sens. Actuat. B—Chem. 2016, 231, 239–244. [Google Scholar] [CrossRef]
- Katsuki, H.; Komarneni, S. Synthesis of Na-A and/or Na-X zeolite/porous carboncomposites from carbonized rice husk. J. Solid State Chem. 2009, 182, 1749–1753. [Google Scholar] [CrossRef]
- Pingarrón, J.M.; Yáñez-Sedeño, P.; González-Cortés, A. Gold nanoparticle-based electrochemical biosensors. Electrochim. Acta 2008, 53, 5848–5866. [Google Scholar] [CrossRef]
- Ju, J.; Chen, W. In situ growth of surfactant-free gold nanoparticles on nitrogen-doped graphene quantum dots for electrochemical detection of hydrogen peroxide in biological environments. Anal. Chem. 2015, 87, 1903–1910. [Google Scholar] [CrossRef] [PubMed]
- Yi, L.; Wei, W.; Zhao, C.; Yang, C.; Tian, L.; Liu, J.; Wang, X. Electrochemical oxidation of sodium borohydride on carbon supported Pt-Zn nanoparticle bimetallic catalyst and its implications to direct borohydride-hydrogen peroxide fuel cell. Electrochim. Acta 2015, 158, 209–218. [Google Scholar] [CrossRef]
- Huang, F.; Xue, L.; Zhang, H.; Guo, R.; Li, Y.; Liao, M.; Wang, M.; Lin, J. An enzyme-free biosensor for sensitive detection of Salmonella using curcumin as signal reporter and click chemistry for signal amplification. Theranostics 2018, 8, 6263–6273. [Google Scholar] [CrossRef]
- Maduraiveeran, G.; Kundu, M.; Sasidharan, M. Electrochemical detection of hydrogen peroxide based on silver nanoparticles via amplified electron transfer process. J. Mater. Sci. 2018, 53, 8328–8338. [Google Scholar] [CrossRef]
- Baghayeri, M.; Veisi, H.; Farhadi, S.; Beitollahi, H.; Maleki, B. Ag nanoparticles decorated Fe3O4/chitosan nanocomposite: Synthesis, characterization and application toward electrochemical sensing of hydrogen peroxide. J. Iran. Chem. Soc. 2018, 15, 1015–1022. [Google Scholar] [CrossRef]
- Kresge, C.T.; Leonowicz, M.E.; Roth, W.J.; Vartuli, J.C.; Beck, J.S. Ordered mesoporous molecular sieves synthesized by a liquid-crystal template mechanism. Nature 1992, 359, 710–712. [Google Scholar] [CrossRef]
- Tian, C.; Li, J.; Ma, C.; Wang, P.; Sun, X.; Fang, J. An ordered mesoporous Ag superstructure synthesized via a template strategy for surface-enhanced Raman spectroscopy. Nanoscale 2015, 7, 12318–12324. [Google Scholar] [CrossRef] [PubMed]
- Park, J.H.; Park, J.K.; Shin, H.Y. The preparation of Ag/mesoporous silica by direct silver reduction and Ag/functionalized mesoporous silica by in situ formation of adsorbed silver. Mater. Lett. 2007, 61, 156–159. [Google Scholar] [CrossRef]
- Han, J.; Fang, P.; Jiang, W.; Li, L.; Guo, R. Ag-nanoparticle-loaded mesoporous silica: Spontaneous formation of Ag nanoparticles and mesoporous silica SBA-15 by a one-pot strategy and their catalytic applications. Langmuir 2012, 28, 4768–4775. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.; Li, J.; Wang, J.; Qi, J.; Fan, W.; Shen, J.; Sun, X.; Han, W.; Wang, L. Synthesis of Ag@SiO2 yolk-shell nanoparticles for hydrogen peroxide detection. RSC Adv. 2015, 5, 17372–17378. [Google Scholar] [CrossRef]
- Khan, A.Y.; Bandyopadhyaya, R. Silver nanoparticle impregnated mesoporous silica as a non-enzymatic amperometric sensor for an aqueous solution of hydrogen peroxide. Electroanal. Chem. 2014, 727, 184–190. [Google Scholar] [CrossRef]
- Azizi, S.N.; Ghasemi, S.; Maybodi, A.S.; Azad, M.R. A new modified electrode based on Ag-doped mesoporous SBA-16 nanoparticles as non-enzymatic sensor for hydrogen peroxide. Sens. Actuat. B-Chem. 2015, 216, 271–278. [Google Scholar] [CrossRef]
- Viter, R.; Iatsunskyi, I. Nanomaterials Design for Sensing Applications; Zenkina, O.V., Ed.; Chapter 2: Metal Oxide Nanostructures in Sensing; Elsevier: Amsterdam, The Netherlands, 2019; pp. 41–91. [Google Scholar] [CrossRef]
- Brynildsen, M.P.; Winkler, J.A.; Spina, C.S.; MacDonald, I.C.; Collins, J.J. Potentiating antibacterial activity by predictably enhancing endogenous microbial ROS production. Nat. Biotech. 2013, 31, 160–165. [Google Scholar] [CrossRef] [PubMed]
- Sgibnev, A.; Kremleva, E. Influence of Hydrogen Peroxide, Lactic Acid, and Surfactants from Vaginal Lactobacilli on the Antibiotic Sensitivity of Opportunistic Bacteria. Probiot. Antimicrob. Proteins 2017, 9, 131. [Google Scholar] [CrossRef] [PubMed]
- Tian, Y.; Qi, J.; Zhang, W.; Cai, Q.; Jiang, X. Facile, one-pot synthesis, and antibacterial activity of mesoporous silica nanoparticles decorated with well-dispersed silver nanoparticles. ACS Appl. Mater. Interfaces 2014, 6, 12038–12045. [Google Scholar] [CrossRef] [PubMed]
- Kumari, M.; Jacob, J.; Philip, D. Green synthesis and applications of Au–Ag bimetallic nanoparticles. Spectrochim. Acta A 2015, 137, 185–192. [Google Scholar] [CrossRef] [PubMed]
- Zou, X.; He, Y.; Sun, P.; Zhao, J.; Cui, G. A novel dealloying strategy for fabricating nanoporous silver as an electrocatalyst for hydrogen peroxide detection. Appl. Surf. Sci. 2018, 447, 542–547. [Google Scholar] [CrossRef]


| Types of Electrode | Applied Potential (V) | Linear Range (µM) | Detection Limit (µM) | Samples | Reference |
|---|---|---|---|---|---|
| AgNP-NH2-SBA-15-GCE | −0.4 vs. SCE | 0.49–5.3 5.3–124.5 | N.A. | PBS buffer | [19] |
| Ag/SBA-16/CPE | −0.45 vs. Ag|AgCl | 20–8000 8000–20,000 | 2.95 | Hair dying cream | [20] |
| Ag@SiO2 YSNs | −0.5 vs. Ag|AgCl | 100–15,000 | 3.5 | PBS buffer | [18] |
| Ag NPs/porous silicon | −0.45 vs. Ag|AgCl | 1.65–500 | 0.45 | Hair color oxidant | [10] |
| Nanoporous silver/Ni wire | −0.35 vs. SCE | 10.0–8000 | 4.80 | PBS buffer | [30] |
| Ag-mSiO2 NPs/GCE | −0.5 vs. SCE | 4–10,000 | 3 | Live cell | This work |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yang, D.; Ni, N.; Cao, L.; Song, X.; Alhamoud, Y.; Yu, G.; Zhao, J.; Zhou, H. Silver Doped Mesoporous Silica Nanoparticles Based Electrochemical Enzyme-Less Sensor for Determination of H2O2 Released from Live Cells. Micromachines 2019, 10, 268. https://doi.org/10.3390/mi10040268
Yang D, Ni N, Cao L, Song X, Alhamoud Y, Yu G, Zhao J, Zhou H. Silver Doped Mesoporous Silica Nanoparticles Based Electrochemical Enzyme-Less Sensor for Determination of H2O2 Released from Live Cells. Micromachines. 2019; 10(4):268. https://doi.org/10.3390/mi10040268
Chicago/Turabian StyleYang, Danting, Ning Ni, Lu Cao, Xin Song, Yasmin Alhamoud, Guangxia Yu, Jinshun Zhao, and Haibo Zhou. 2019. "Silver Doped Mesoporous Silica Nanoparticles Based Electrochemical Enzyme-Less Sensor for Determination of H2O2 Released from Live Cells" Micromachines 10, no. 4: 268. https://doi.org/10.3390/mi10040268
APA StyleYang, D., Ni, N., Cao, L., Song, X., Alhamoud, Y., Yu, G., Zhao, J., & Zhou, H. (2019). Silver Doped Mesoporous Silica Nanoparticles Based Electrochemical Enzyme-Less Sensor for Determination of H2O2 Released from Live Cells. Micromachines, 10(4), 268. https://doi.org/10.3390/mi10040268





