Microfluidic Time-Delay Valve Mechanism on Paper-Based Devices for Automated Competitive ELISA
Abstract
1. Introduction
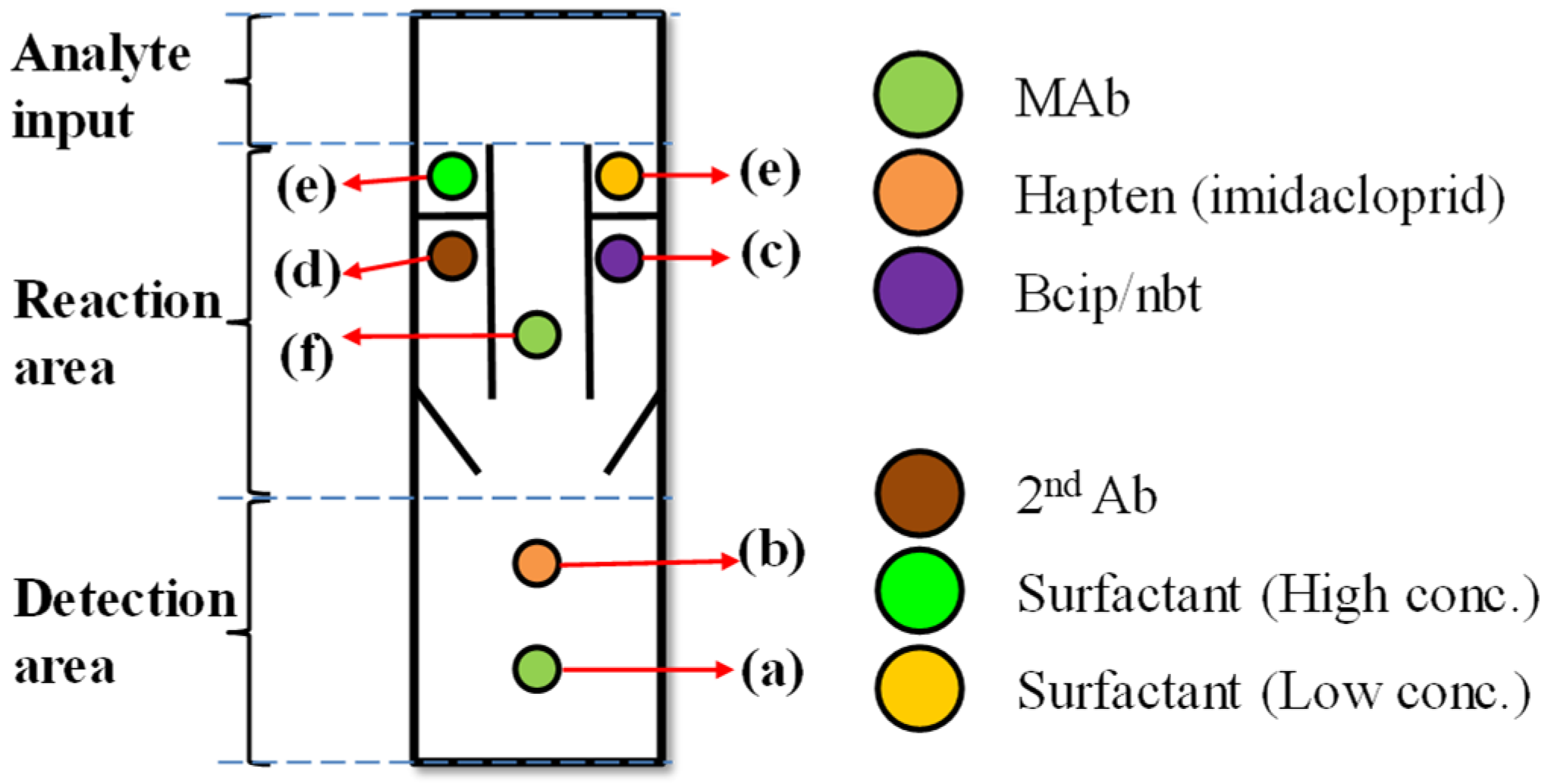
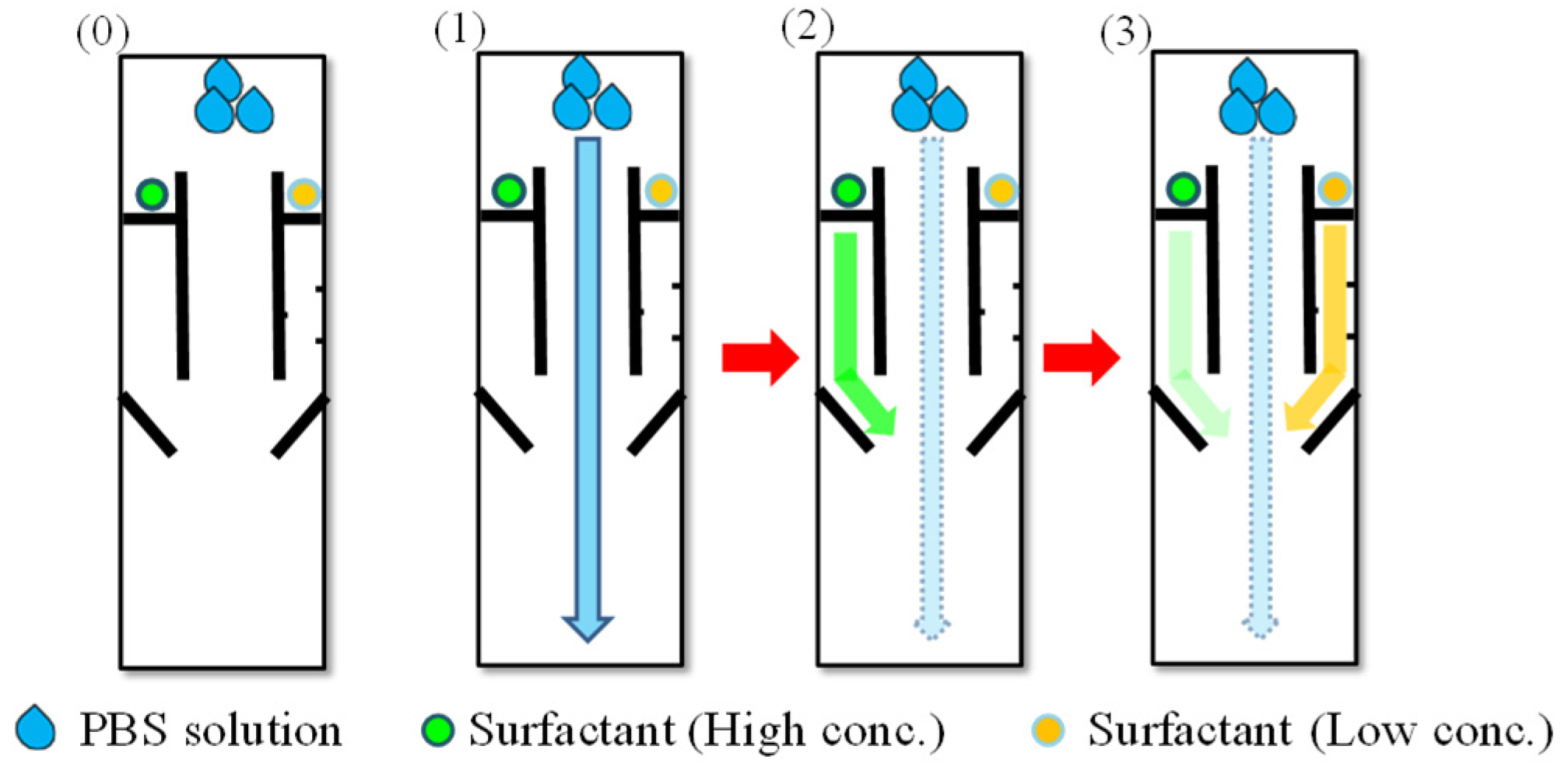
2. Mechanism and Methods
3. Device Fabrication
3.1. Device Fabrication
3.2. Preparation of the Paper-Based Enzyme-Linked Immunosorbent Assay (ELISA) Device for the Detection of Imidacloprid
3.3. Materials
3.4. Data Processing
4. Result and Discussion
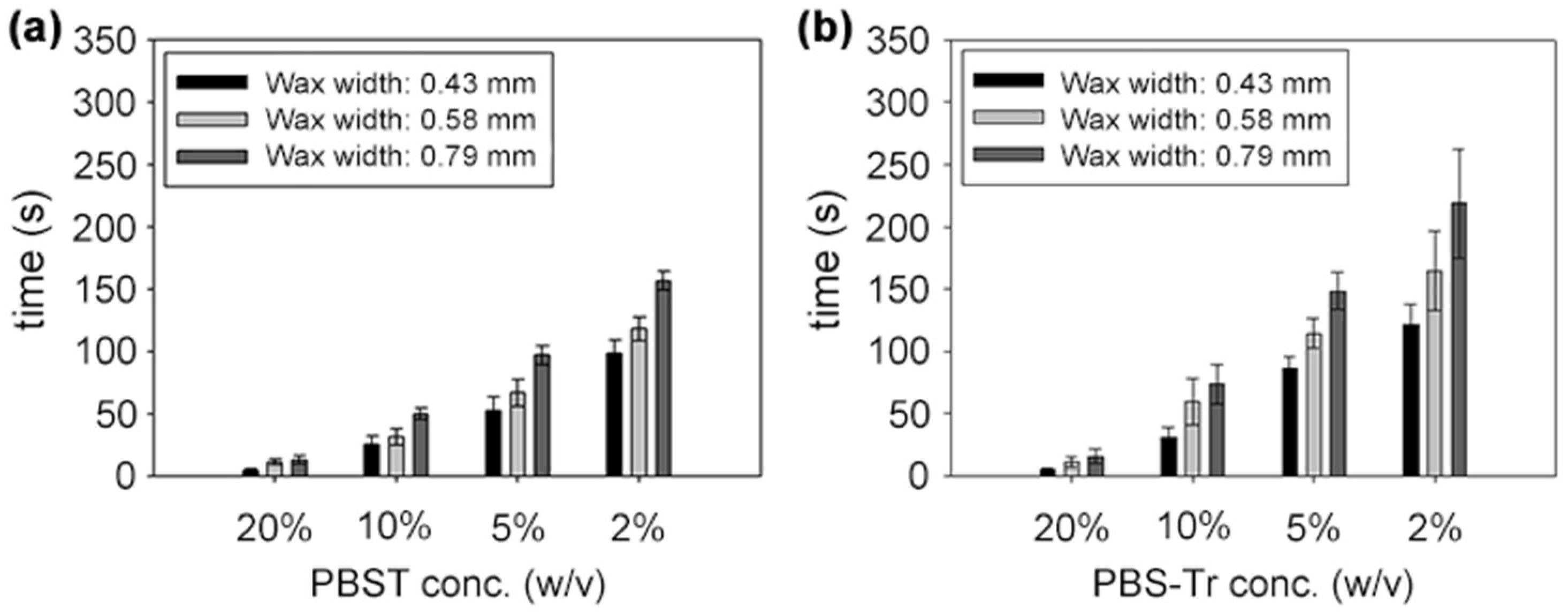
4.1. Time-Delay Valve
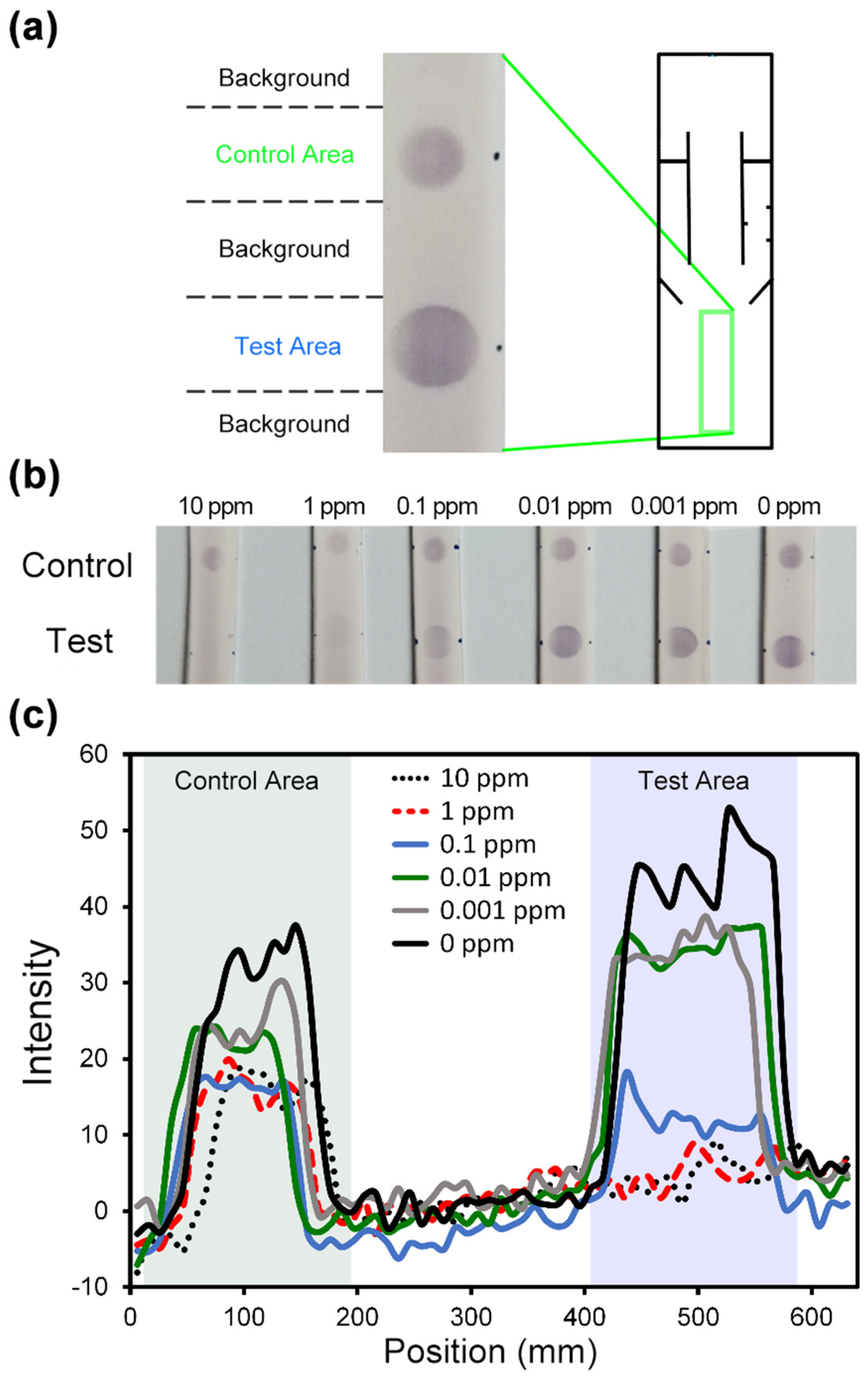
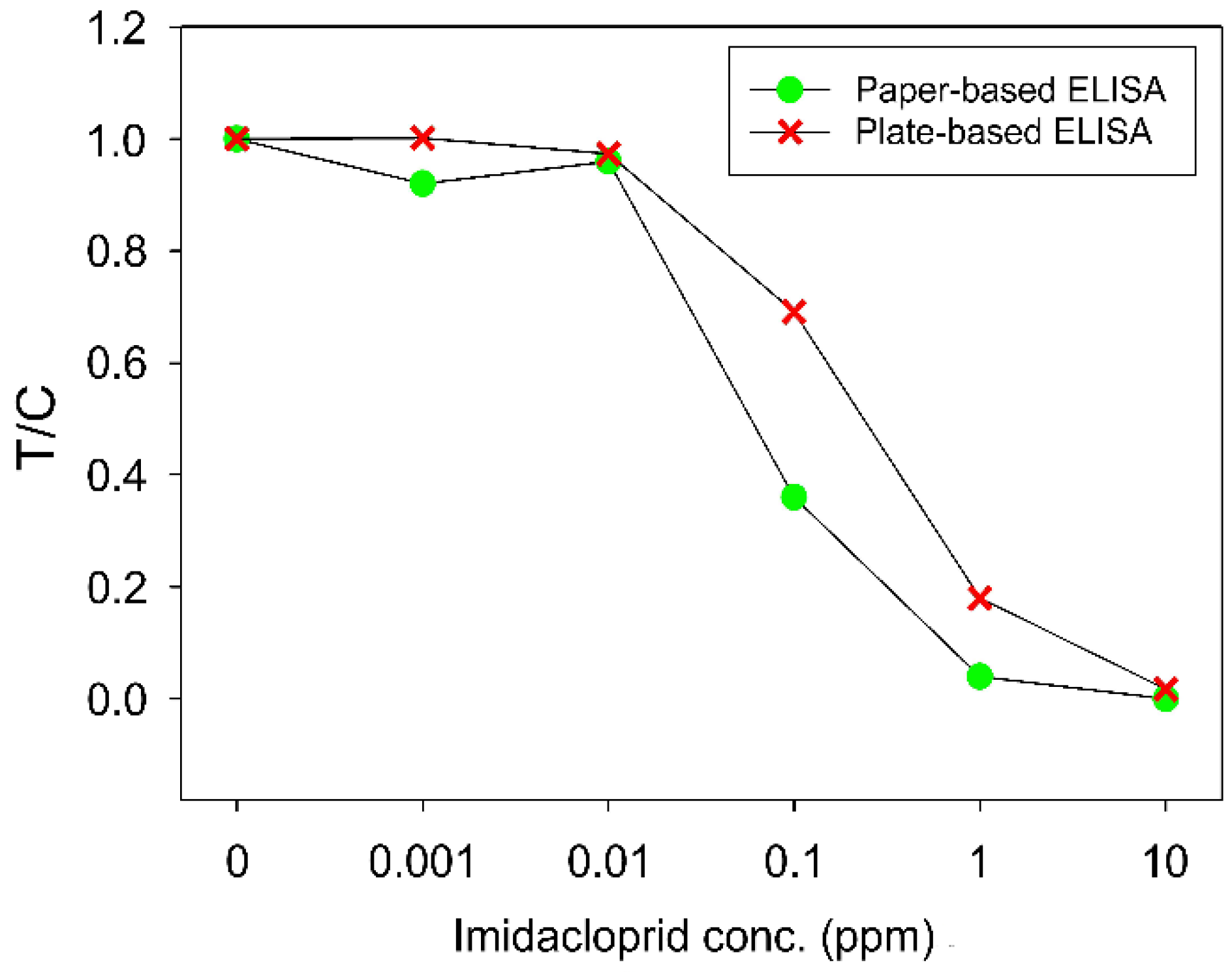
4.2. Performance of Autonomous Paper-Based Devices
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Alavanja, M.C.; Hoppin, J.A.; Kamel, F. Health effects of chronic pesticide exposure: Cancer and neurotoxicity. Annu. Rev. Public Health 2004, 25, 155–197. [Google Scholar] [CrossRef] [PubMed]
- Kamel, F.; Hoppin, J.A. Association of pesticide exposure with neurologic dysfunction and disease. Environ. Health Perspect. 2004, 112, 950–958. [Google Scholar] [CrossRef] [PubMed]
- Gilden, R.C.; Huffling, K.; Sattler, B. Pesticides and health risks. J. Obstet. Gynecol. Neonatal Nurs. 2010, 39, 103–110. [Google Scholar] [CrossRef] [PubMed]
- Navalon, A.; Gonzalez-Casado, A.; El-Khattabi, R.; Vilchez, J.L.; Fernández-Alba, A.R. Determination of imidacloprid in vegetable samples by gas chromatography–mass spectrometry. Analyst 1997, 122, 579–581. [Google Scholar] [CrossRef]
- MacDonald, L.M.; Meyer, T.R. Determination of imidacloprid and triadimefon in white pine by gas chromatography/mass spectrometry. J. Agric. Food Chem. 1998, 46, 3133–3138. [Google Scholar] [CrossRef]
- Baskaran, S.; Kookana, R.S.; Naidu, R. Determination of the insecticide imidacloprid in water and soil using high-performance liquid chromatography. J. Chromatogr. A 1997, 787, 271–275. [Google Scholar] [CrossRef]
- Lepowsky, E.; Ghaderinezhad, F.; Knowlton, S.; Tasoglu, S. Paper-based assays for urine analysis. Biomicrofluidics 2017, 11, 051501. [Google Scholar] [CrossRef]
- Li, C.Z.; Vandenberg, K.; Prabhulkar, S.; Zhu, X.; Schneper, L.; Methee, K.; Almeide, E. Paper based point-of-care testing disc for multiplex whole cell bacteria analysis. Biosens. Bioelectron. 2011, 26, 4342–4348. [Google Scholar] [CrossRef]
- Li, X.; Ballerini, D.R.; Shen, W. A perspective on paper-based microfluidics: Current status and future trends. Biomicrofluidics 2012, 6, 011301. [Google Scholar] [CrossRef]
- Xia, Y.; Si, J.; Li, Z. Fabrication techniques for microfluidic paper-based analytical devices and their applications for biological testing: A review. Biosens. Bioelectron. 2016, 77, 774–789. [Google Scholar] [CrossRef]
- Kung, C.T.; Hou, C.Y.; Wang, Y.N.; Fu, L.-M. Microfluidic paper-based analytical devices for environmental analysis of soil, air, ecology and river water. Sens. Actuators B Chem. 2019, 301, 126855. [Google Scholar] [CrossRef]
- Cheng, C.M.; Martinez, A.W.; Gong, J.; Mace, C.R.; Phillips, S.T.; Carrilho, E.; Whitesides, G.M. Paper-based ELISA. Angew. Chem. 2010, 122, 4881–4884. [Google Scholar] [CrossRef]
- Wang, S.; Ge, L.; Song, X.; Yu, J.; Ge, S.; Huang, J.; Zeng, F. Paper-based chemiluminescence ELISA: lab-on-paper based on chitosan modified paper device and wax-screen-printing. Biosens. Bioelectron. 2012, 31, 212–218. [Google Scholar] [CrossRef] [PubMed]
- Apilux, A.; Ukita, Y.; Chikae, M.; Chailapakul, O.; Takamura, Y. Development of automated paper-based devices for sequential multistep sandwich enzyme-linked immunosorbent assays using inkjet printing. Lab Chip 2013, 13, 126–135. [Google Scholar] [CrossRef] [PubMed]
- Carrilho, E.; Martinez, A.W.; Whitesides, G.M. Understanding wax printing: a simple micropatterning process for paper-based microfluidics. Anal. Chem. 2009, 81, 7091–7095. [Google Scholar] [CrossRef]
- Fu, E.; Lutz, B.; Kauffman, P.; Yager, P. Controlled reagent transport in disposable 2D paper networks. Lab Chip 2010, 10, 918–920. [Google Scholar] [CrossRef]
- Juncker, D.; Schmid, H.; Drechsler, U.; Wolf, H.; Wolf, M.; Michel, B.; Delamarche, E. Autonomous microfluidic capillary system. Anal. Chem. 2002, 74, 6139–6144. [Google Scholar] [CrossRef]
- Li, X.; Tian, J.; Nguyen, T.; Shen, W. Paper-based microfluidic devices by plasma treatment. Anal. Chem. 2008, 80, 9131–9134. [Google Scholar] [CrossRef]
- Martinez, A.W.; Phillips, S.T.; Nie, Z.; Cheng, C.M.; Carrilho, E.; Wiley, B.J.; Whitesides, G.M. Programmable diagnostic devices made from paper and tape. Lab Chip 2010, 10, 2499–2504. [Google Scholar] [CrossRef]
- Shin, J.H.; Park, J.; Kim, S.H.; Park, J.K. Programmed sample delivery on a pressurized paper. Biomicrofluidics 2014, 8, 054121. [Google Scholar] [CrossRef]
- Wang, X.; Hagen, J.A.; Papautsky, I. Paper pump for passive and programmable transport. Biomicrofluidics 2013, 7, 014107. [Google Scholar] [CrossRef] [PubMed]
- Chumo, B.; Muluneh, M.; Issadore, D. Laser micromachined hybrid open/paper microfluidic chips. Biomicrofluidics 2013, 7, 064109. [Google Scholar] [CrossRef] [PubMed]
- Gaspar, C.; Sikanen, T.; Franssila, S.; Jokinen, V. Inkjet printed silver electrodes on macroporous paper for a paper-based isoelectric focusing device. Biomicrofluidics 2016, 10, 064120. [Google Scholar] [CrossRef] [PubMed]
- Giokas, D.L.; Tsogas, G.Z.; Vlessidis, A.G. Programming fluid transport in paper-based microfluidic devices using razor-crafted open channels. Anal. Chem. 2014, 86, 6202–6207. [Google Scholar] [CrossRef]
- He, P.J.W.; Katis, I.N.; Eason, R.W.; Sones, C.L. Engineering fluidic delays in paper-based devices using laser direct-writing. Lab Chip 2015, 15, 4054–4061. [Google Scholar] [CrossRef]
- Oyola-Reynoso, S.; Frankiewicz, C.; Chang, B.; Chen, J.; Bloch, J.F.; Thuo, M.M. Paper-based microfluidic devices by asymmetric calendaring. Biomicrofluidics 2017, 11, 014104. [Google Scholar] [CrossRef]
- Yakoh, A.; Chaiyo, S.; Siangproh, W.; Chailapakul, O. 3D Capillary-driven paper-based sequential microfluidic device for electrochemical sensing applications. ACS Sens. 2019, 4, 1211–1221. [Google Scholar] [CrossRef]
- Lutz, B.; Liang, T.; Fu, E.; Ramachandran, S.; Kauffman, P.; Yager, P. Dissolvable fluidic time delays for programming multi-step assays in instrument-free paper diagnostics. Lab Chip 2013, 13, 2840–2847. [Google Scholar] [CrossRef]
- Chen, H.; Cogswell, J.; Anagnostopoulos, C.; Faghri, M. A fluidic diode, valves, and a sequential-loading circuit fabricated on layered paper. Lab Chip 2012, 12, 2909–2913. [Google Scholar] [CrossRef]
- Eddleston, M.; Bateman, D.N. Pesticides. Medicine 2012, 40, 147–150. [Google Scholar] [CrossRef]
- Elbert, A.; Becker, B.; Hartwig, J.; Erdelen, C. Imidacloprid-a new systemic insecticide. Pflanzenschutz Nachr. Bayer (Ger. FR) 1991, 44, 113–136. [Google Scholar]
- Lai, Y.T.; Tsai, J.S.; Hsu, J.C.; Lu, Y.W. Automated paper-based devices by microfluidic timing-valve for competitive ELISA. In Proceedings of the IEEE 30th International Conference on Micro Electro Mechanical Systems (MEMS), Las Vegas, NV, USA, 22–26 January 2017; pp. 1321–1324. [Google Scholar]
- Cai, L.; Xu, C.; Lin, S.; Luo, J.; Wu, M.; Yang, F. A simple paper-based sensor fabricated by selective wet etching of silanized filter paper using a paper mask. Biomicrofluidics 2014, 8, 056504. [Google Scholar] [CrossRef] [PubMed]
- Kumar, S.; Bhushan, P.; Krishna, V.; Bhattacharya, S. Tapered lateral flow immunoassay based point-of-care diagnostic device for ultrasensitive colorimetric detection of dengue NS1. Biomicrofluidics 2018, 12, 034104. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Han, D.; Pauletti, G.M.; Steckl, A.J. Engineering a simple lateral flow device for animal blood coagulation monitoring. Biomicrofluidics 2018, 12, 014110. [Google Scholar] [CrossRef] [PubMed]
- Syms, R. Rapid evaporation-driven chemical pre-concentration and separation on paper. Biomicrofluidics 2017, 11, 044116. [Google Scholar] [CrossRef]



© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lai, Y.-T.; Tsai, C.-H.; Hsu, J.-C.; Lu, Y.-W. Microfluidic Time-Delay Valve Mechanism on Paper-Based Devices for Automated Competitive ELISA. Micromachines 2019, 10, 837. https://doi.org/10.3390/mi10120837
Lai Y-T, Tsai C-H, Hsu J-C, Lu Y-W. Microfluidic Time-Delay Valve Mechanism on Paper-Based Devices for Automated Competitive ELISA. Micromachines. 2019; 10(12):837. https://doi.org/10.3390/mi10120837
Chicago/Turabian StyleLai, Yu-Ting, Chia-Hsin Tsai, Ju-Chun Hsu, and Yen-Wen Lu. 2019. "Microfluidic Time-Delay Valve Mechanism on Paper-Based Devices for Automated Competitive ELISA" Micromachines 10, no. 12: 837. https://doi.org/10.3390/mi10120837
APA StyleLai, Y.-T., Tsai, C.-H., Hsu, J.-C., & Lu, Y.-W. (2019). Microfluidic Time-Delay Valve Mechanism on Paper-Based Devices for Automated Competitive ELISA. Micromachines, 10(12), 837. https://doi.org/10.3390/mi10120837





