Marine Polysaccharide-Collagen Coatings on Ti6Al4V Alloy Formed by Self-Assembly
Abstract
1. Introduction
2. Materials and Methods
2.1. HE800 and GY785 Exopolysaccharides (EPS) Production
2.2. Preparation of HE800 and GY785 EPS Derivatives
2.3. Characterization of EPS Derivatives
2.3.1. Sugar Composition
2.3.2. Molecular Weight
2.3.3. Sulfate Content
2.4. Atomic Force Microscopy (AFM): Sample Preparation and Imaging
2.5. Production of Ti6Al4V Discs and Coating with Collagen Fibrils
2.6. Scanning Electron Microscopy (SEM) of Ti6Al4V Discs Coated with Collagen Fibrils and EPS Derivatives
2.7. Cell Biological Characterization of Ti6Al4V Discs Coated with Collagen Fibrils and EPS Derivatives
2.7.1. Cell Culture and Cell Seeding
2.7.2. Cell Viability
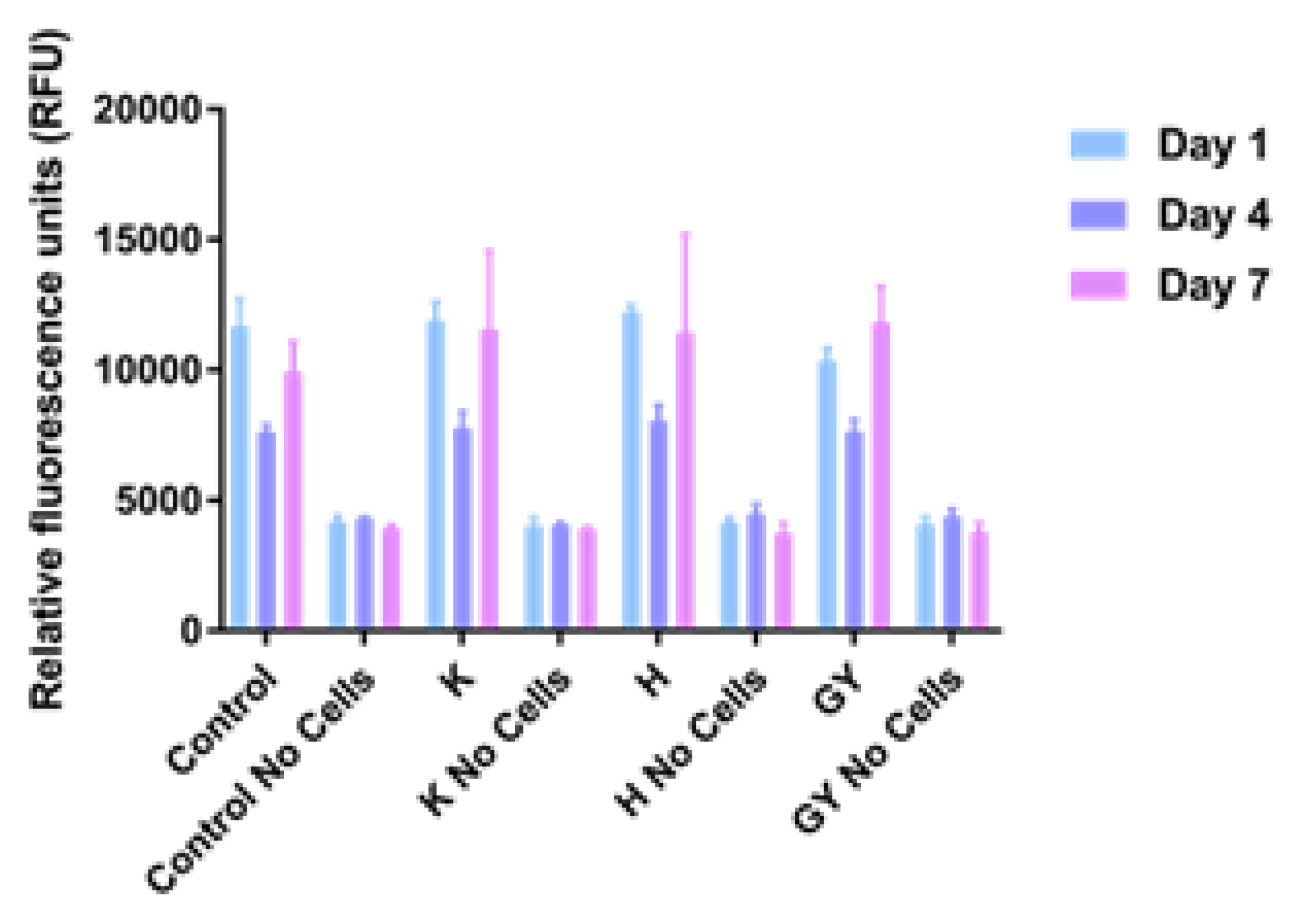
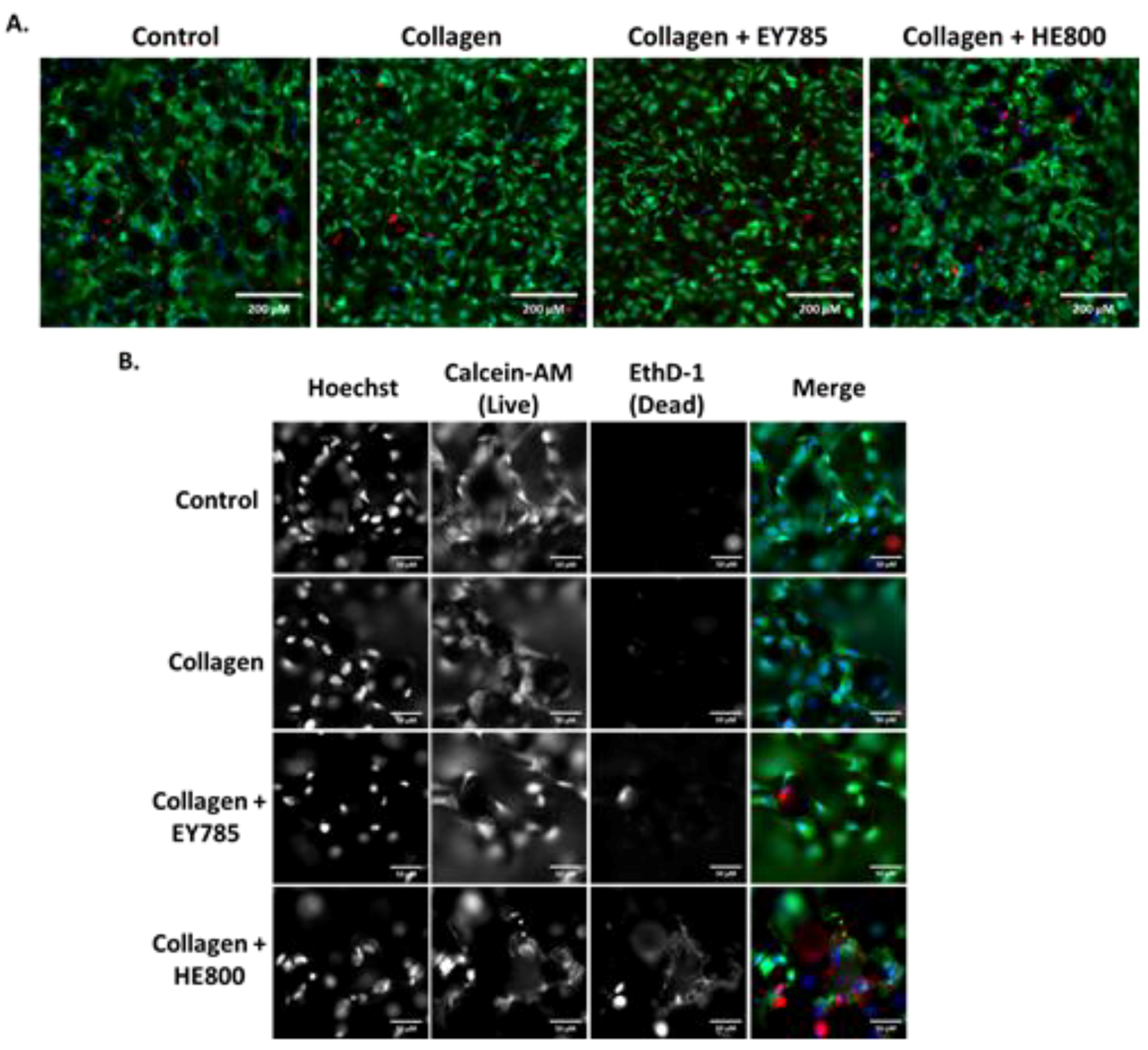
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Becker, D.; Geissler, U.; Hempel, U.; Bierbaum, S.; Scharnweber, D.; Worch, H.; Wenzel, K.W. Proliferation and differentiation of rat calvarial osteoblasts on type I collagen-coated titanium alloy. J. Biomed. Mater. Res. 2002, 59, 516–527. [Google Scholar] [CrossRef] [PubMed]
- Bierbaum, S.; Hempel, U.; Geissler, U.; Hanke, T.; Scharnweber, D.; Wenzel, K.W.; Worch, H. Modification of Ti6Al4V surfaces using collagen i, iii, and fibronectin. Ii. Influence on osteoblast responses. J. Biomed. Mater. Res. A 2003, 67, 431–438. [Google Scholar] [CrossRef] [PubMed]
- Geissler, U.; Hempel, U.; Wolf, C.; Scharnweber, D.; Worch, H.; Wenzel, K. Collagen type I-coating of Ti6Al4V promotes adhesion of osteoblasts. J. Biomed. Mater. Res. 2000, 51, 752–760. [Google Scholar] [CrossRef]
- Gomez-Guillen, M.C.; Gimenez, B.; Lopez-Caballero, M.E.; Montero, M.P. Functional and bioactive properties of collagen and gelatin from alternative sources: A review. J. Food Hydrocolloids 2011, 25, 1813–1827. [Google Scholar] [CrossRef]
- Bierbaum, S.; Douglas, T.; Hanke, T.; Scharnweber, D.; Tippelt, S.; Monsees, T.K.; Funk, R.H.; Worch, H. Collageneous matrix coatings on titanium implants modified with decorin and chondroitin sulfate: Characterization and influence on osteoblastic cells. J. Biomed. Mater. Res. A 2006, 77, 551–562. [Google Scholar] [CrossRef]
- Douglas, T.; Heinemann, S.; Mietrach, C.; Hempel, U.; Bierbaum, S.; Scharnweber, D.; Worch, H. Interactions of collagen types I and II with chondroitin sulfates a-c and their effect on osteoblast adhesion. Biomacromolecules 2007, 8, 1085–1092. [Google Scholar] [CrossRef]
- Raguénès, G.H.; Peres, A.; Ruimy, R.; Pignet, P.; Christen, R.; Loaëc, M.; Rougeaux, H.; Barbier, G.; Guezennec, J. Alteromonas infernus sp. Nov., a new polysaccharide-producing bacterium isolated from a deep-sea hydrothermal vent. J. Appl. Microbiol. 1997, 82, 422–430. [Google Scholar] [CrossRef]
- Rougeaux, H.; Kervarec, N.; Pichon, R.; Guezennec, J. Structure of the exopolysaccharide of vibrio diabolicus isolated from a deep-sea hydrothermal vent. Carbohydr. Res. 1999, 322, 40–45. [Google Scholar] [CrossRef]
- Zanchetta, P.; Lagarde, N.; Guezennec, J. A new bone-healing material: A hyaluronic acid-like bacterial exopolysaccharide. Calcif. Tissue Int. 2003, 72, 74–79. [Google Scholar] [CrossRef]
- Senni, K.; Gueniche, F.; Changotade, S.; Septier, D.; Sinquin, C.; Ratiskol, J.; Lutomski, D.; Godeau, G.; Guezennec, J.; Colliec-Jouault, S. Unusual glycosaminoglycans from a deep sea hydrothermal bacterium improve fibrillar collagen structuring and fibroblast activities in engineered connective tissues. Mar. Drugs 2013, 11, 1351–1369. [Google Scholar] [CrossRef]
- Roger, O.; Kervarec, N.; Ratiskol, J.; Colliec-Jouault, S.; Chevolot, L. Structural studies of the main exopolysaccharide produced by the deep-sea bacterium alteromonas infernus. Carbohydr. Res. 2004, 339, 2371–2380. [Google Scholar] [CrossRef] [PubMed]
- Colliec Jouault, S.; Chevolot, L.; Helley, D.; Ratiskol, J.; Bros, A.; Sinquin, C.; Roger, O.; Fischer, A.M. Characterization, chemical modifications and in vitro anticoagulant properties of an exopolysaccharide produced by alteromonas infernus. Biochim. Biophys. Acta 2001, 1528, 141–151. [Google Scholar] [CrossRef]
- Heymann, D.; Ruiz-Velasco, C.; Chesneau, J.; Ratiskol, J.; Sinquin, C.; Colliec-Jouault, S. Anti-metastatic properties of a marine bacterial exopolysaccharide-based derivative designed to mimic glycosaminoglycans. Molecules 2016, 21, 309. [Google Scholar] [CrossRef] [PubMed]
- Merceron, C.; Portron, S.; Vignes-Colombeix, C.; Rederstorff, E.; Masson, M.; Lesoeur, J.; Sourice, S.; Sinquin, C.; Colliec-Jouault, S.; Weiss, P.; et al. Pharmacological modulation of human mesenchymal stem cell chondrogenesis by a chemically oversulfated polysaccharide of marine origin: Potential application to cartilage regenerative medicine. Stem Cells 2012, 30, 471–480. [Google Scholar] [CrossRef] [PubMed]
- Rederstorff, E.; Rethore, G.; Weiss, P.; Sourice, S.; Beck-Cormier, S.; Mathieu, E.; Maillasson, M.; Jacques, Y.; Colliec-Jouault, S.; Fellah, B.H.; et al. Enriching a cellulose hydrogel with a biologically active marine exopolysaccharide for cell-based cartilage engineering. J. Tissue Eng. Regen. Med. 2017, 11, 1152–1164. [Google Scholar] [CrossRef] [PubMed]
- Smith, R.A.A.; Murali, S.; Rai, B.; Lu, X.; Lim, Z.X.H.; Lee, J.J.L.; Nurcombe, V.; Cool, S.M. Minimum structural requirements for bmp-2-binding of heparin oligosaccharides. Biomaterials 2018, 184, 41–55. [Google Scholar] [CrossRef] [PubMed]
- Ling, L.; Dombrowski, C.; Foong, K.M.; Haupt, L.M.; Stein, G.S.; Nurcombe, V.; van Wijnen, A.J.; Cool, S.M. Synergism between wnt3a and heparin enhances osteogenesis via a phosphoinositide 3-kinase/akt/runx2 pathway. J. Biol. Chem. 2010, 285, 26233–26244. [Google Scholar] [CrossRef]
- Ruiz-Velasco, C.; Baud’huin, M.; Sinquin, C.; Maillasson, M.; Heyman, D.; Colliec-Jouault, S.; Padrines, M. Effects of a sulfated exopolysaccharide produced by alteromonas infernus on bone biology. Glycobiology 2011, 21, 781–795. [Google Scholar] [CrossRef]
- Raguénès, G.; Christen, R.; Guezennec, J.; Pignet, P.; Barbier, G. Vibrio diabolicus sp. Nov., a new polysaccharide-secreting organism isolated from a deep-sea hydrothermal vent polychaete annelid, alvinella pompejana. Int. J. Syst. Bacteriol. 1997, 47, 989–995. [Google Scholar] [CrossRef]
- Chopin, N.; Sinquin, C.; Ratiskol, J.; Zykwinska, A.; Weiss, P.; Cerantola, S.; Le Bideau, J.; Colliec-Jouault, S. A direct sulfation process of a marine polysaccharide in ionic liquid. Biomed. Res. Int. 2015, 2015, 508656. [Google Scholar] [CrossRef]
- Senni, K.; Gueniche, F.; Yousfi, M.; Fioretti, F.; Godeau, G.; Colliec-Jouault, S. Sulfated Depolymerized Derivatives of Exopolysaccharides (eps) from Mesophilic Marine Bacteria, Method for Preparing Same, and Uses Thereof in Tissue Regeneration. U.S. Patent Application No. 11/629,579, 5 June 2008. [Google Scholar]
- Kamerling, J.P.; Gerwig, G.J.; Vliegenthart, J.F.; Clamp, J.R. Characterization by gas-liquid chromatography-mass spectrometry and proton-magnetic-resonance spectroscopy of pertrimethylsilyl methyl glycosides obtained in the methanolysis of glycoproteins and glycopeptides. Biochem. J. 1975, 151, 491–495. [Google Scholar] [CrossRef] [PubMed]
- Montreuil, J.; Bouquelet, S.; Debray, H.; Fournet, B.; Spik, G.; Strecker, G. A pratical approach. In Glycoptoteins in Carbohydrate Analysis; Chaplin, M.F., Kennedy, J.F., Eds.; IRL Press: Oxford, UK, 1986; pp. 143–204. [Google Scholar]
- Popov, V.; Muller-Kamskii, G.; Kovalevsky, A.; Dzhenzhera, G.; Strokin, E.; Kolomiets, A.; Ramon, J. Design and 3d-printing of titanium bone implants: Brief review of approach and clinical cases. J. Biomed. Eng. Lett. 2018, 8, 337–344. [Google Scholar] [CrossRef] [PubMed]
- Karamichos, D.; Brown, R.A.; Mudera, V. Complex dependence of substrate stiffness and serum concentration on cell-force generation. J. Biomed. Mater. Res. A 2006, 78, 407–415. [Google Scholar] [CrossRef] [PubMed]
- Vandrovcova, M.; Douglas, T.E.L.; Heinemann, S.; Scharnweber, D.; Dubruel, P.; Bacakova, L. Collagen-lactoferrin fibrillar coatings enhance osteoblast proliferation and differentiation. J. Biomed. Mater. Res. Part A 2015, 103, 525–533. [Google Scholar] [CrossRef] [PubMed]
- Douglas, T.E.L.; Hempel, U.; Zydek, J.; Vladescu, A.; Pietryga, K.; Kaeswurm, J.A.H.; Buchweitz, M.; Surmenev, R.A.; Surmeneva, M.A.; Cotrut, C.M.; et al. Pectin coatings on titanium alloy scaffolds produced by additive manufacturing: Promotion of human bone marrow stromal cell proliferation. Mater. Lett. 2018, 227, 225–228. [Google Scholar] [CrossRef]
- Rother, S.; Salbach-Hirsch, J.; Moeller, S.; Seemann, T.; Schnabelrauch, M.; Hofbauer, L.C.; Hintze, V.; Scharnweber, D. Bioinspired collagen/glycosaminoglycan-based cellular microenvironments for tuning osteoclastogenesis. ACS Appl. Mater. Interfaces 2015, 7, 23787–23797. [Google Scholar] [CrossRef]
- Salchert, K.; Oswald, J.; Streller, U.; Grimmer, M.; Herold, N.; Werner, C. Fibrillar collagen assembled in the presence of glycosaminoglycans to constitute bioartificial stem cell niches in vitro. J. Mater. Sci. Mater. Med. 2005, 16, 581–585. [Google Scholar] [CrossRef]
- Tsai, S.W.; Liu, R.L.; Hsu, F.Y.; Chen, C.C. A study of the influence of polysaccharides on collagen self-assembly: Nanostructure and kinetics. Biopolymers 2006, 83, 381–388. [Google Scholar] [CrossRef]
- Rederstorff, E.; Weiss, P.; Sourice, S.; Pilet, P.; Xie, F.; Sinquin, C.; Colliec-Jouault, S.; Guicheux, J.; Laib, S. An in vitro study of two gag-like marine polysaccharides incorporated into injectable hydrogels for bone and cartilage tissue engineering. Acta Biomater. 2011, 7, 2119–2130. [Google Scholar] [CrossRef]
- Zhao, N.; Wang, X.; Qin, L.; Guo, Z.; Li, D. Effect of molecular weight and concentration of hyaluronan on cell proliferation and osteogenic differentiation in vitro. Biochem. Biophys. Res. Commun. 2015, 465, 569–574. [Google Scholar] [CrossRef]


| Exopolysaccharides (EPS) Derivative | Osidic Composition (wt%) | S (wt%) | Mw (g/moL) | |||||
|---|---|---|---|---|---|---|---|---|
| Gal | Glc | GalA | GlcA | GalNAc | GlcNAc | |||
| HE800 derivative | 0 | 0 | 0 | 19.8 | 10.6 | 10.8 | 0 | 280 000 |
| GY785 derivative | 19.2 | 16.8 | 6.9 | 9.3 | 0 | 0 | 3 | 240 000 |
| Components | Volume, μL |
|---|---|
| Collagen Type I (4 mg/mL) | 280 |
| 10× MEM | 40 |
| HE800/GY785 derivative (5 mg/mL ddH2O) or ddH2O | 80 |
| 1 M NaOH solution | ~30 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Norris, K.; Mishukova, O.I.; Zykwinska, A.; Colliec-Jouault, S.; Sinquin, C.; Koptioug, A.; Cuenot, S.; Kerns, J.G.; Surmeneva, M.A.; Surmenev, R.A.; et al. Marine Polysaccharide-Collagen Coatings on Ti6Al4V Alloy Formed by Self-Assembly. Micromachines 2019, 10, 68. https://doi.org/10.3390/mi10010068
Norris K, Mishukova OI, Zykwinska A, Colliec-Jouault S, Sinquin C, Koptioug A, Cuenot S, Kerns JG, Surmeneva MA, Surmenev RA, et al. Marine Polysaccharide-Collagen Coatings on Ti6Al4V Alloy Formed by Self-Assembly. Micromachines. 2019; 10(1):68. https://doi.org/10.3390/mi10010068
Chicago/Turabian StyleNorris, Karl, Oksana I. Mishukova, Agata Zykwinska, Sylvia Colliec-Jouault, Corinne Sinquin, Andrei Koptioug, Stéphane Cuenot, Jemma G. Kerns, Maria A. Surmeneva, Roman A. Surmenev, and et al. 2019. "Marine Polysaccharide-Collagen Coatings on Ti6Al4V Alloy Formed by Self-Assembly" Micromachines 10, no. 1: 68. https://doi.org/10.3390/mi10010068
APA StyleNorris, K., Mishukova, O. I., Zykwinska, A., Colliec-Jouault, S., Sinquin, C., Koptioug, A., Cuenot, S., Kerns, J. G., Surmeneva, M. A., Surmenev, R. A., & Douglas, T. E. L. (2019). Marine Polysaccharide-Collagen Coatings on Ti6Al4V Alloy Formed by Self-Assembly. Micromachines, 10(1), 68. https://doi.org/10.3390/mi10010068










