Benthic Archives Reveal Recurrence and Dominance of Toxigenic Cyanobacteria in a Eutrophic Lake over the Last 220 Years
Abstract
1. Introduction
2. Results
2.1. Sedimentary Cored Chronology
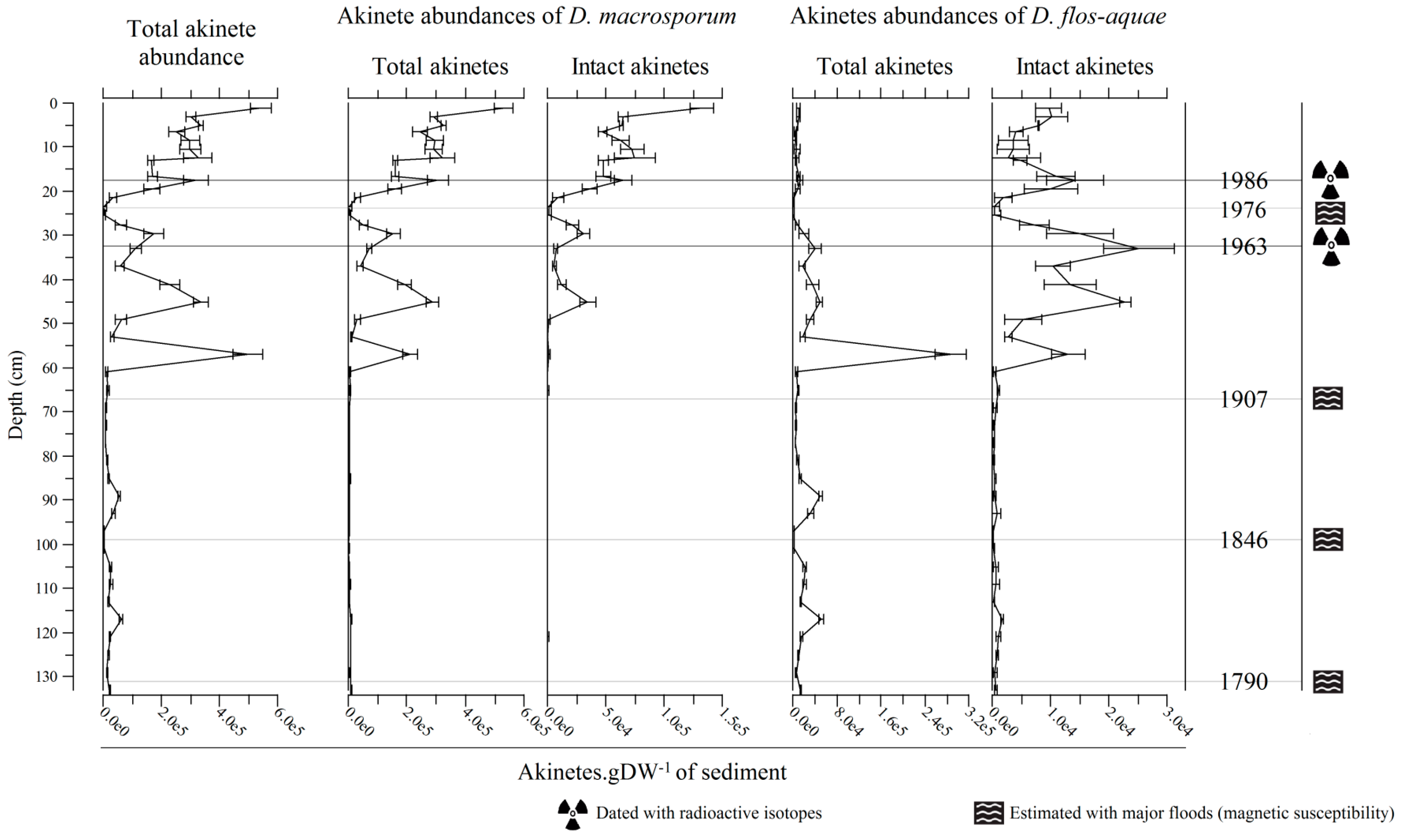
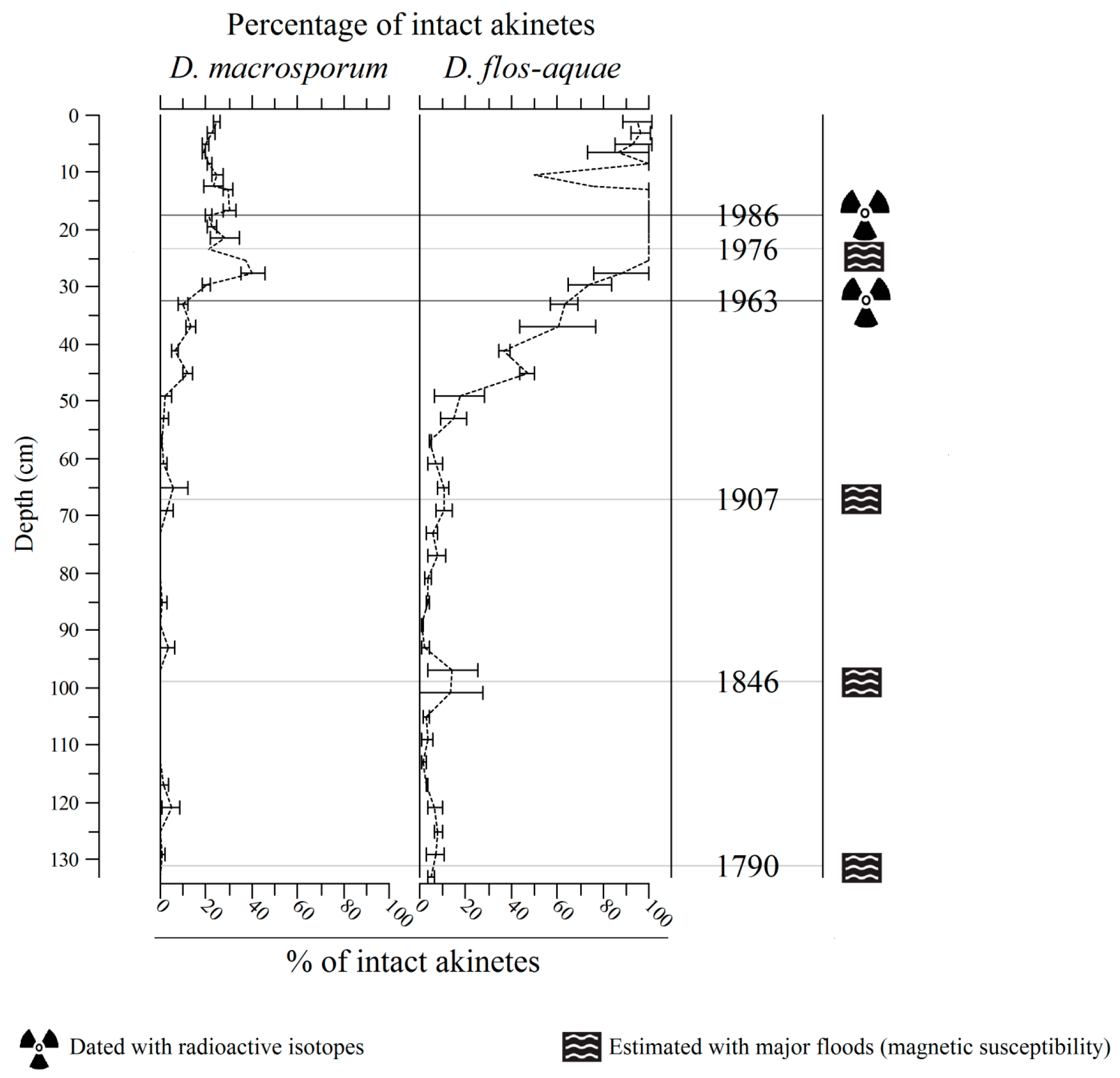
2.2. Vertical Akinete Distribution in Sediment
2.2.1. Akinete Abundance along the Core
2.2.2. Akinete Integrity along the Core
2.3. Target Gene Detection in Total Sediment
2.4. Target Gene Detection in Extracted Intact Akinetes
2.5. Multiple Factor Analysis
3. Discussion
4. Conclusions
5. Materials and Methods
5.1. Study Site and Sampling
5.2. Core Characterization, Sampling and Dating
5.3. Akinete Extraction from Sediment and Microscopic Counts
5.4. Akinete Purifications
5.5. DNA Extractions from Total Sediment and Purified Akinetes and Cyanobacterial 16S RNAr Gene Amplification
5.6. Cyanotoxin Gene Amplification
5.7. Statistical Analysis
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Merel, S.; Walker, D.; Chicana, R.; Snyder, S.; Baurès, E.; Thomas, O. State of knowledge and concerns on cyanobacterial blooms and cyanotoxins. Environ. Int. 2013, 59, 303–327. [Google Scholar] [CrossRef] [PubMed]
- Schopf, J.W. Cyanobacteria: Pioneers of the early Earth. Nova Hedwig. Beih. 1996, 112, 13–32. [Google Scholar]
- De Candolle, A.P. Notice sur la matière qui a coloré le lac de Morat. Mém. Soc. Phys. Hist. Nat. 1985, 3, 129–146. [Google Scholar]
- Domaizon, I.; Savichtcheva, O.; Debroas, D.; Arnaud, F.; Villar, C.; Pignol, C.; Alric, B.; Perga, M.E. DNA from lake sediments reveals the long-term dynamics and diversity of Synechococcus assemblages. Biogeosci. Discuss. 2013, 10, 2515–2564. [Google Scholar] [CrossRef]
- Martinez De La Escalera, G.; Antoniades, D.; Bonilla, S.; Piccini, C. Application of ancient DNA to the reconstruction of past microbial assemblages and for the detection of toxic cyanobacteria in subtropical freshwater ecosystems. Mol. Ecol. 2014, 23, 5791–5802. [Google Scholar] [CrossRef] [PubMed]
- Kaplan-levy, R.N.; Hadas, O.; Summers, M.L.; Rücker, J.; Sukenik, A. Akinetes: Dormant Cells of Cyanobacteria. In Dormancy and Resistance in Harsh Environments; Springer: Berlin/Heidelberg, Germany, 2010; pp. 5–27. [Google Scholar]
- Capo, E.; Debroas, D.; Arnaud, F.; Domaizon, I. Is Planktonic Diversity Well Recorded in Sedimentary DNA? Toward the Reconstruction of Past Protistan Diversity. Microb. Ecol. 2015, 70, 865–875. [Google Scholar] [CrossRef] [PubMed]
- Van Geel, B.; Mur, L.R.; Ralska-jasiewiczowa, M.; Goslar, T. Fossil akinetes of Aphanizomenon and Anabaena as indicators for medieval phosphate-eutrophication of Lake Gosciaz (Central Poland). Rev. Paleobot. Palynol. 1994, 83, 97–105. [Google Scholar] [CrossRef]
- Eilers, J.M.; Kann, J.; Cornett, J.; Moser, K.; Amand, A.S. Paleolimnological evidence of change in a shallow, hypereutrophic lake: Upper Klamath Lake, Oregon, USA. Hydrobiologia 2004, 520, 7–18. [Google Scholar] [CrossRef]
- Wood, S.; Jentzsch, K.; Rueckert, A.; Hamilton, D.P.; Cary, S.C. Hindcasting cyanobacterial communities in Lake Okaro with germination experiments and genetic analyses. FEMS Microbiol. Ecol. 2009, 67, 252–260. [Google Scholar] [CrossRef] [PubMed]
- Pal, S.; Pick, F.R. Temporal trends in cyanobacteria revealed through DNA and pigment analyses of temperate lake sediment cores. J. Paleolimnol. 2015, 54, 87–101. [Google Scholar] [CrossRef]
- Monchamp, M.; Walser, J.; Pomati, F. Sedimentary DNA Reveals Cyanobacterial Community Diversity over 200 Years in Two Perialpine Lakes. Appl. Environ. Microbiol. 2016, 82, 6472–6482. [Google Scholar] [PubMed]
- Misson, B.; Donnadieu-bernard, F.; Godon, J.; Latour, D. Short- and long-term dynamics of the toxic potential and genotypic structure in benthic populations of Microcystis. Water Res. 2012, 42, 1438–1446. [Google Scholar] [CrossRef] [PubMed]
- Delvin, J.; Edwards, O.; Gorham, P.; Hunter, N.; Pike, R.K.; Stavric, B. Anatoxin-a, a toxic alkaloid from AnabaenaJEos-aquae NRC-44h1. Can. J. Chem. 1977, 55, 1367–1371. [Google Scholar]
- Cadel-Six, S.; Peyraud-Thomas, C.; Brient, L.; De Marsac, N.T.; Rippka, R.; Méjean, A. Different genotypes of anatoxin-producing cyanobacteria coexist in the Tarn River, France. Appl. Environ. Microbiol. 2007, 73, 7605–7614. [Google Scholar] [CrossRef] [PubMed]
- Ballot, A.; Fastner, J.; Wiedner, C. Paralytic shellfish poisoning toxin-producing cyanobacterium Aphanizomenon gracile in Northeast Germany. Appl. Environ. Microbiol. 2010, 76, 1173–1180. [Google Scholar] [CrossRef] [PubMed]
- Rantala-Ylinen, A.; Kana, S.; Wang, H.; Rouhiainen, L.; Wahlsten, M.; Rizzi, E.; Berg, K.; Gugger, M.; Sivonen, K. Anatoxin-a Synthetase Gene Cluster of the Cyanobacterium Anabaena sp. Strain 37 and Molecular Methods To Detect Potential Producers. Appl. Environ. Microbiol. 2011, 77, 7271–7278. [Google Scholar] [CrossRef] [PubMed]
- Sabart, M.; Crenn, K.; Perrière, F.; Abila, A.; Leremboure, M.; Colombet, J.; Jousse, C.; Latour, D. Co-occurrence of microcystin and anatoxin-a in the freshwater lake Aydat (France): Analytical and molecular approaches during a three-year survey. Harmful Algae 2015, 48, 12–20. [Google Scholar] [CrossRef]
- Legrand, B.; Lesobre, J.; Colombet, J.; Latour, D.; Sabart, M. Molecular tools to detect anatoxin-a genes in aquatic ecosystems: Toward a new nested PCR-based method. Harmful Algae 2016, 58, 16–22. [Google Scholar] [CrossRef] [PubMed]
- Lavrieux, M.; Disnar, J.-R.; Chapron, E.; Bréheret, J.-G.; Jacob, J.; Miras, Y.; Reyss, J.-L.; Andrieu-Ponel, V.; Fabien, A. 6700-year sedimentary record of climatic and anthropic signals in Lake Aydat ( French Massif Central) signals in Lake Aydat (French Massif Central). Holocene 2013, 23, 1317–1328. [Google Scholar] [CrossRef]
- Legrand, B.; Lamarque, A.; Sabart, M.; Latour, D. Characterization of akinetes from cyanobacterial strains and lake sediment: A study of their resistance and toxic potential. Harmful Algae 2016, 59, 42–50. [Google Scholar] [CrossRef] [PubMed]
- Michelin, Y. Les Jardins de Vulcain. Paysages D’hier, D’aujourd’hui et de Demain Dans la Chaîne Des Puys du Massif Central Français; de la Maison des Sciences de l’Homme: Paris, France, 1996. [Google Scholar]
- Paerl, H.W.; Hall, N.S.; Calandrino, E.S. Controlling harmful cyanobacterial blooms in a world experiencing anthropogenic and climatic-induced change. Sci. Total Environ. 2011, 409, 1739–1745. [Google Scholar] [CrossRef] [PubMed]
- Paerl, H.W.; Otten, T.G. Harmful cyanobacterial blooms: Causes, consequences, and controls. Microb. Ecol. 2013, 65, 995–1010. [Google Scholar] [CrossRef] [PubMed]
- Mitrovic, S.M.; Oliver, R.L.; Rees, C.; Bowling, L.C.; Buckney, R.T. Critical flow velocities for the growth and dominance of Anabaena circinalis in some turbid freshwater rivers. Freshw. Biol. 2003, 48, 164–174. [Google Scholar] [CrossRef]
- Livingstone, D.; Jaworski, G.H.M. The viability of akinetes of blue-green algae recovered from the sediments of Rostherne Mere. Br. Phycol. J. 1980, 15, 357–364. [Google Scholar] [CrossRef]
- Legrand, B.; Le Jeune, A.H.; Colombet, J.; Thouvenot, A.; Latour, D. Are akinetes representative of past nostocalean blooms? A case study of their benthic spatio-temporal distribution and potential for germination in a eutrophic lake. Appl. Environ. Microbiol. Submitted.
- Gerphagnon, M.; Latour, D.; Colombet, J.; Sime-Ngando, T. Fungal parasitism: Life cycle, dynamics and impact on cyanobacterial blooms. PLoS ONE 2013, 8, e60894. [Google Scholar] [CrossRef] [PubMed]
- Gerphagnon, M.; Colombet, J.; Latour, D.; Sime-Ngando, T. Spatial and temporal changes of parasitic chytrids of cyanobacteria. Sci. Rep. 2017, 7. [Google Scholar] [CrossRef] [PubMed]
- Nichols, J.M.; Adams, D.G.; Carr, N.G.; Whitton, B.A. (Eds.) The Biology of Cyanobacteria; Blackwell: Oxford, UK, 1982; pp. 387–412. [Google Scholar]
- Herdman, M. Akinetes: Structure and function. In The cyanobacteria; Fay, P., van, B.C., Eds.; Elsevier: Amsterdam, The Netherlands, 1987; pp. 227–250. [Google Scholar]
- Herdman, M. Cellular differentiation: Akinetes. Methods Enzymol. 1987, 167, 222–232. [Google Scholar]
- Cardemil, L.; Wolk, C.P. The polysaccharides from heterocyst and spore envelopes of a blue-green alga. Methylation analysis and structure of the backbones. J. Biol. Chem. 1976, 251, 2967–2975. [Google Scholar] [PubMed]
- Cardemil, L.; Wolk, C.P. The polysaccharides from heterocyst and spore envelopes of a blue-green alga. Structure of the basic repeating unit. J. Biol. Chem. 1979, 254, 736–741. [Google Scholar] [PubMed]
- Le lac d’Aydat (Puy de Dôme): Proposotion de Restauration de la qualité des eaux. In Proceedings of the French Water Monitoring Policies, Orcet, France, 1992.
- Lafforgue, M.; Szeligiewicz, W.; Devaux, J.; Poulint, M. Selective mechanisms controlling algal succession in Aydat Lake. Water Sci. Technol. 1995, 32, 117–127. [Google Scholar]
- Syndicat Mixte de la Vallée de la Veyre et de l’Auzon. 2017. Available online: http://www.smvva.fr/actions/lacs-et-zones-humides (accessed on 22 August 2017).
- Aleya, L. Dyanmique des Populations Phytoplanctoniques du Lac D’aydat (Puy-De-Dôme, France): Bomasse et Activité métabolique de Diverses Fractions de Taille. Ph.D. Thesis, Université Blaise Pascal de Clermont-Ferrand, Aubière, France, 1987. [Google Scholar]
- Lafforgue, M. Modélisation du Fonctionnement D’un Écosystéme Lacustre: Le Lac D’aydat. Ph.D. Thesis, Ecole Nationale Supérieure des Mines de Paris, Paris, France, 1990. [Google Scholar]
- French Database from: L’Ecole Des Hautes Etudes en Sciences Sociales (Ehess). 2016. Available online: http://cassini.ehess.fr/cassini/fr/html/fiche.php?select_resultat=2247# (accessed on 6 July 2017).
- Mantzouki, E.; Visser, P.M.; Bormans, M.; Ibelings, B.W. Understanding the key ecological traits of cyanobacteria as a basis for their management and control in changing lakes. Aquat. Ecol. 2016, 50, 333–350. [Google Scholar] [CrossRef]
- Hansen, J.; Ruedy, R.; Sato, M.; Lo, K. Global surface temperature change. Rev. Geophys. 2010, 48, 1–29. [Google Scholar] [CrossRef]
- Briand, E.; Escoffier, N.; Straub, C.; Sabart, M.; Quiblier, C.; Humbert, J.-F. Spatiotemporal changes in the genetic diversity of a bloom-forming Microcystis aeruginosa (cyanobacteria) population. ISME J. 2009, 3, 419–429. [Google Scholar] [CrossRef] [PubMed]
- Graham, J.L.; Loftin, K.A.; Meyer, M.T.; Ziegler, A.C. Co-occurrence of Toxins and Taste-and-Odor Compounds in Cyanobacterial Blooms from the Midwestern United States. Environ. Sci. Technol. 2006, 44, 7361–7368. [Google Scholar] [CrossRef] [PubMed]
- Rapala, J.; Sivonen, K.; Lyra, C.; Niemelä, S.I. Variation of microcystins in anabaena spp. as a function of growth stimuli. Appl. Environ. Microbiol. 1997, 63, 2206–2212. [Google Scholar] [PubMed]
- Park, H.; Watanabe, M.F.; Harada, K.; Nagai, H.; Suzuki, M. Hepatotoxin (Microcystin) and Neurotoxin (Anatoxin-a) Contained in Natural Blooms and Strains of Cyanobacteria From Japanese Freshwaters. Nat. Toxins 1993, 1, 353–360. [Google Scholar] [CrossRef] [PubMed]
- Park, H.; Kim, B.; Kim, E.; Okino, T. Hepatotoxic Microcystins and Neurotoxic Anatoxin-a in Cyanobacterial Blooms from Korean Lakes. Environ. Toxicol. Water Qual. 1998, 13, 225–234. [Google Scholar] [CrossRef]
- Ballot, A.; Pflugmacher, S.; Wiegand, C.; Kotut, K.; Krienitz, L. Cyanobacterial toxins in Lake Baringo, Kenya. Limnologica 2003, 33, 2–9. [Google Scholar] [CrossRef]
- Pirszel, J.; Adamczyk, A. Relationship between cyanobacterial bloom composition and anatoxin-a and microcystin occurrence in the eutrophic dam reservoir (SE Poland). Pol. J. Ecol. 2004, 52, 479–490. [Google Scholar]
- Dadheech, P.K.; Krienitz, L.; Kotut, K.; Ballot, A.; Casper, P. Molecular detection of uncultured cyanobacteria and aminotransferase domains for cyanotoxin production in sediments of different Kenyan lakes. FEMS Microbiol. Ecol. 2009, 68, 340–350. [Google Scholar] [CrossRef] [PubMed]
- Kirchner, G. 210Pb as a tool for establishing sediment chronologies: Examples of potentials and limitations of conventional dating models. J. Environ. Radioact. 2011, 102, 490–494. [Google Scholar] [CrossRef] [PubMed]
- Bonnett, P.J.P.; Appleby, P.G. Deposition and transport of radionuclides within an upland drainage basin in mid-Wales. In Environmental History and Palaeolimnology; Springer: Dordrecht, The Netherlands, 1991; pp. 71–76. [Google Scholar]
- Reyss, J.L.; Schmidt, S.; Legeleux, F.; Bonté, P. Large, low background well-type detectors for measurements of environmental radioactivity. Nucl. Instrum. Methods Phys. Res. Sect. A Accel. Spectrom. Detect. Assoc. Equip. 1995, 357, 391–397. [Google Scholar] [CrossRef]
- Cirés, S.; Lars, W.; Agha, R.; Quesada, A. Overwintering populations of Anabaena, Aphanizomenon and Microcystis as potential inocula for summer blooms. J. Plankton Res. 2013, 35, 1254–1266. [Google Scholar] [CrossRef]
- Ramm, J.; Rücker, J.; Knie, M.; Nixdorf, B. Lost in the dark: Estimation of the akinete pool for the recruitment of Nostocales populations (cyanobacteria) in a temperate deep lake. J. Plankton Res. 2017, 39, 392–403. [Google Scholar] [CrossRef]
- Bourrelly, P. Les Algues D’eau Douce, Tome I: Algues Vertes, Editions N ed; Boubée et Cie.: Paris, France, 1966. [Google Scholar]
- Geitler, L. Cyanophyceae; Johnson: New York, NY, USA, 1932; pp. 1196–1200. [Google Scholar]
- Komárek, J. Süsswasserflora von Mitteleuropa: Bd. 19/3. Cyanoprokaryota 3. Teil; Springer Spektrum: Berlin, Germany, 2013. [Google Scholar]
- Gerphagnon, M.; Latour, D.; Colombet, J.; Sime-Ngando, T. A Double Staining Method Using SYTOX Green and Calcofluor White for Studying Fungal Parasites of Phytoplankton. Appl. Environ. Microbiol. 2013, 79, 3943–3951. [Google Scholar] [CrossRef] [PubMed]
- Nubel, U.; Muyzer, G.; Garcia-pichel, F.; Muyzer, G. PCR primers to amplify 16S rRNA genes from cyanobacteria PCR Primers to Amplify 16S rRNA Genes from Cyanobacteria. Microbiology 1997, 63, 3327–3332. [Google Scholar]
- Hisbergues, M.; Christiansen, G.; Rouhiainen, L.; Sivonen, K.; Börner, T. PCR-based identification of microcystin-producing genotypes of different cyanobacterial genera. Arch. Microbiol. 2003, 180, 402–410. [Google Scholar] [CrossRef] [PubMed]





| Type of PCR | Primer Name | Sequence (5′-3′) | Target Gene | Gene Amplicon Size (bp) | References |
|---|---|---|---|---|---|
| Classic | cya359F | GGGGAATYTTCCGCAATGGG | Cyanobacterial | 403 | [60] |
| PCR | cya781R | GACTACTGGGGTATCTAATCCCATT | 16S rRNA gene | ||
| Nested | anxgen-F2 | ATGGTCAGAGGTTTTACAAG | anaC | 861 | [19] modified |
| PCR 1 | anxgen-R | CGACTCTTAATCATGCGATC | from [17] | ||
| Nested | anaCgen-F2 | TCTGGTATTCAGTMCCCTCYAT | anaC | 519 | [19] modified |
| PCR 2 | anxgen-R | CGACTCTTAATCATGCGATC | from [17] | ||
| Classic | mcyA-Cd 1F | AAAATTAAAAGCCGTATCAAA | mcyA | 291 | [61] |
| PCR | mcyA-Cd 1R | AAAAGTGTTTTATTAGCGGCTCAT |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Legrand, B.; Lamarque, A.; Sabart, M.; Latour, D. Benthic Archives Reveal Recurrence and Dominance of Toxigenic Cyanobacteria in a Eutrophic Lake over the Last 220 Years. Toxins 2017, 9, 271. https://doi.org/10.3390/toxins9090271
Legrand B, Lamarque A, Sabart M, Latour D. Benthic Archives Reveal Recurrence and Dominance of Toxigenic Cyanobacteria in a Eutrophic Lake over the Last 220 Years. Toxins. 2017; 9(9):271. https://doi.org/10.3390/toxins9090271
Chicago/Turabian StyleLegrand, Benjamin, Amélie Lamarque, Marion Sabart, and Delphine Latour. 2017. "Benthic Archives Reveal Recurrence and Dominance of Toxigenic Cyanobacteria in a Eutrophic Lake over the Last 220 Years" Toxins 9, no. 9: 271. https://doi.org/10.3390/toxins9090271
APA StyleLegrand, B., Lamarque, A., Sabart, M., & Latour, D. (2017). Benthic Archives Reveal Recurrence and Dominance of Toxigenic Cyanobacteria in a Eutrophic Lake over the Last 220 Years. Toxins, 9(9), 271. https://doi.org/10.3390/toxins9090271





