Protein Discovery: Combined Transcriptomic and Proteomic Analyses of Venom from the Endoparasitoid Cotesia chilonis (Hymenoptera: Braconidae)
Abstract
:1. Introduction
2. Results and Discussion
2.1. Transcriptome Analysis of Cotesia chilonis Venom Glands
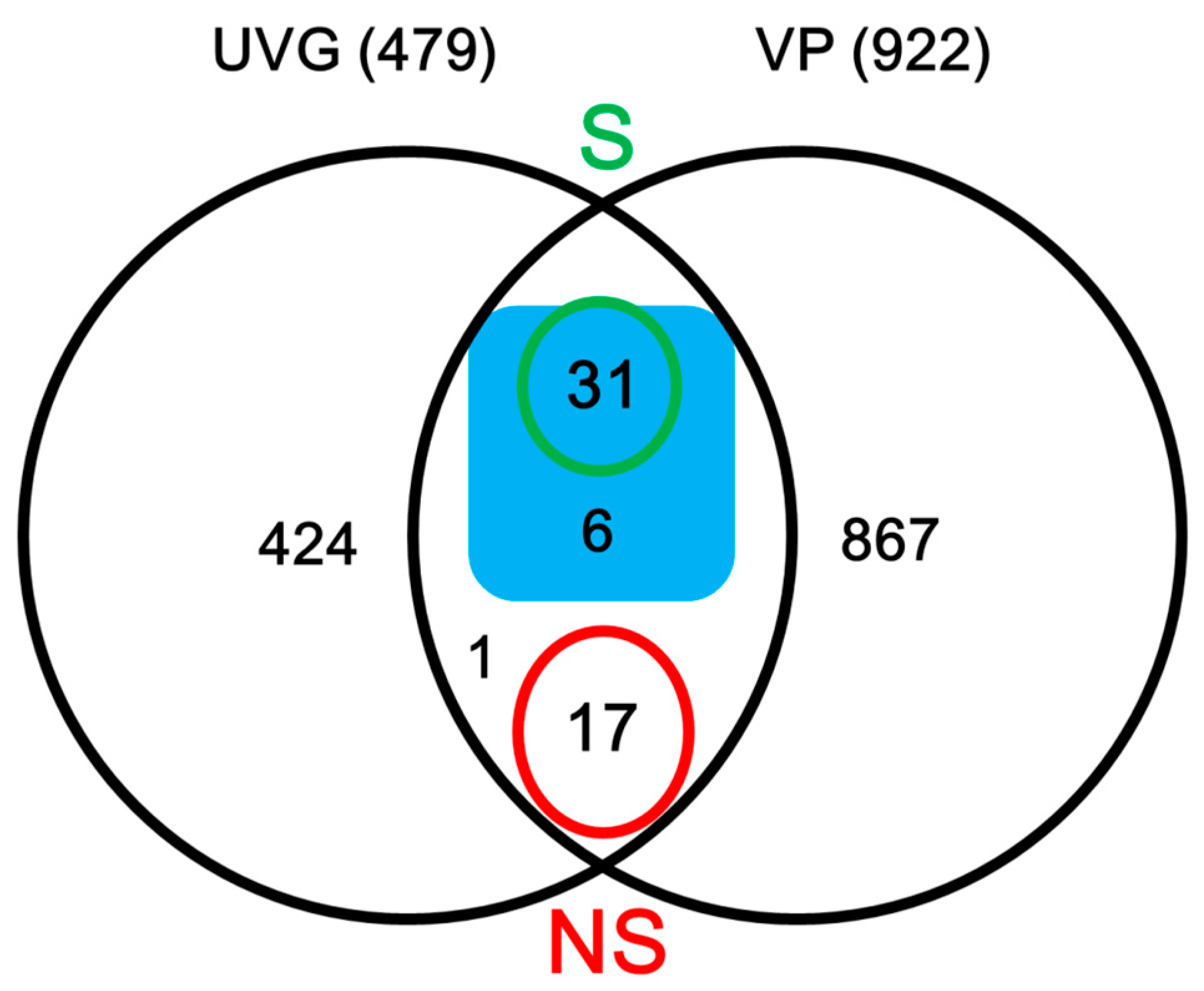
2.2. Identification of Secreted Proteins
2.2.1. Hydrolases
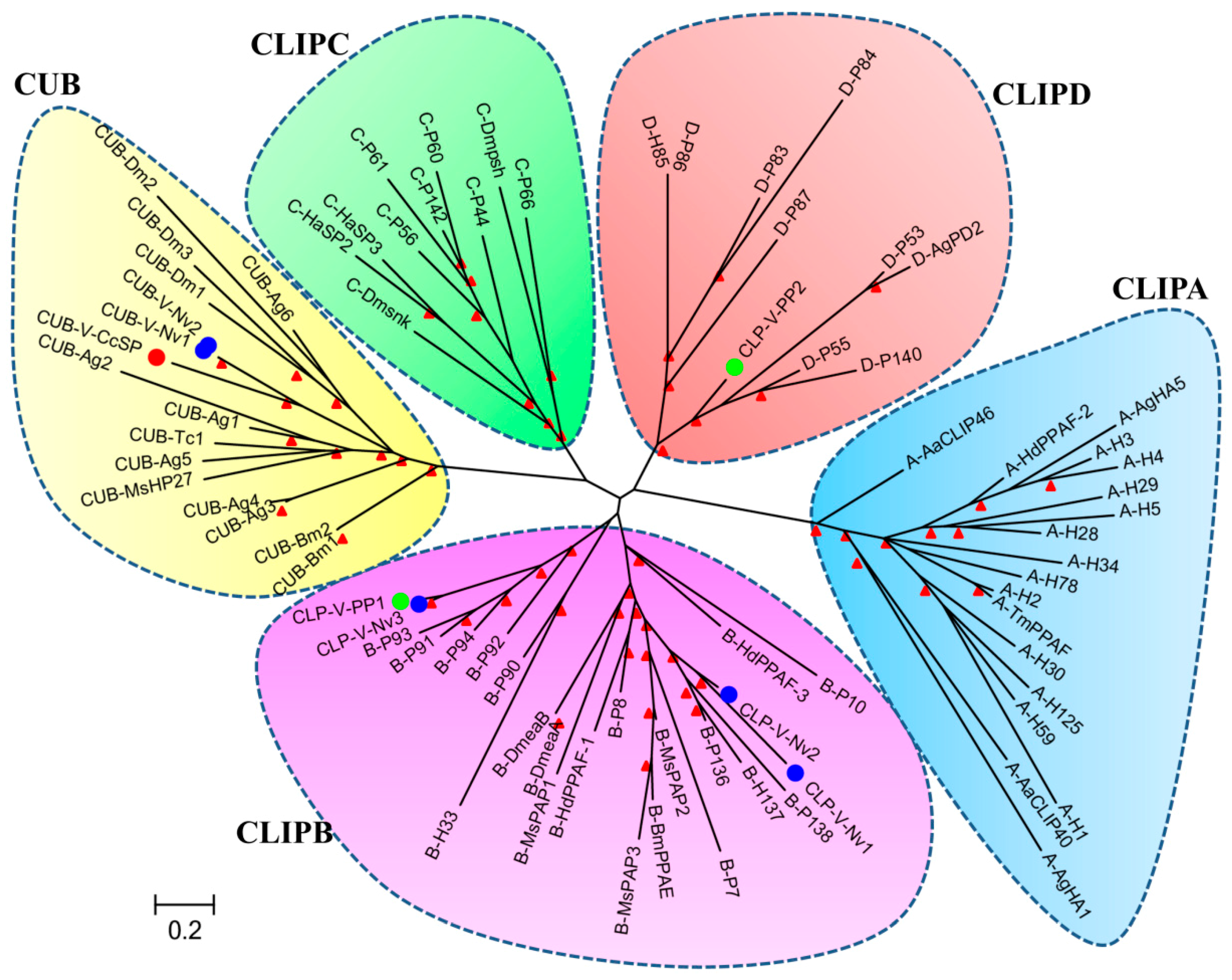
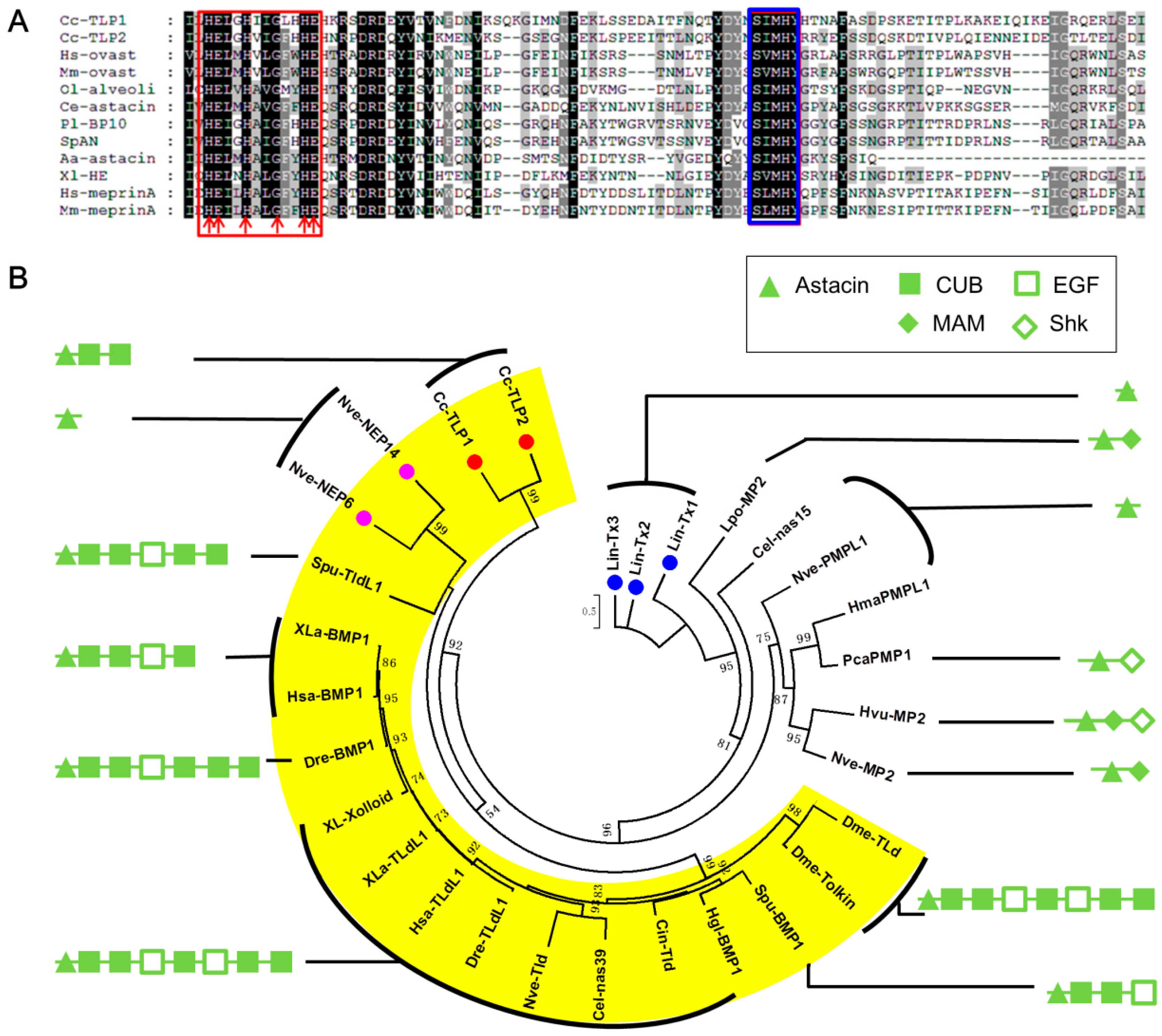
Proteases
Peptidases
Esterases
Glycosyl Hydrolase
Endonuclease
2.2.2. Isomerase
2.2.3. Extracellular Superoxide Dismutase 3
2.2.4. Serine Protease Inhibitors
2.2.5. Other Venom Proteins
Immunoevasive Protein-2
CRT
Vascular Endothelial Growth Factor Receptor 2
Proteins Involved in the Folding and Export of Secreted Venom Proteins
Other Unknown Proteins
3. Materials and Methods
3.1. Insect Rearing
3.2. Venom Gland Collection and Total RNA Isolation
3.3. Construction and Sequencing of cDNA Library
3.4. Data Analysis
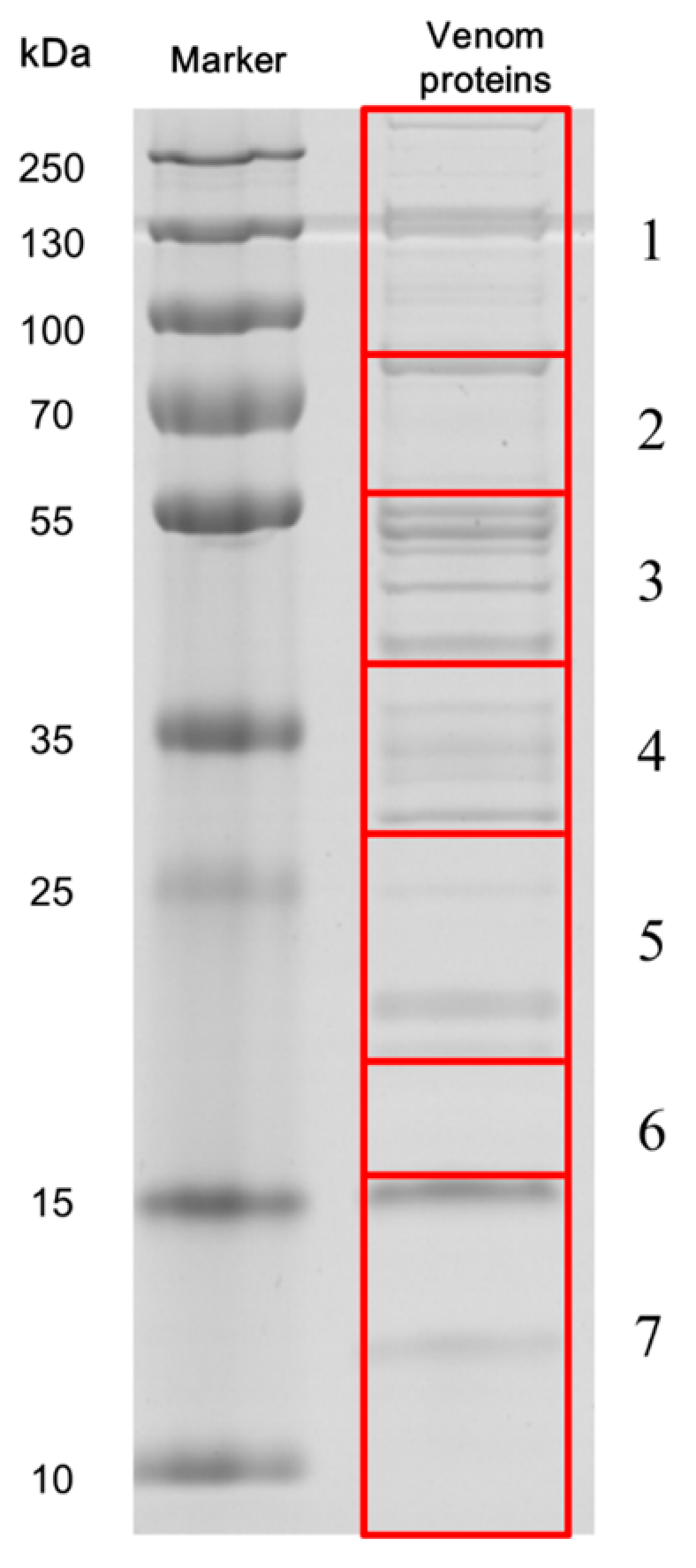
3.5. SDS-PAGE of Venom and Protein Identification
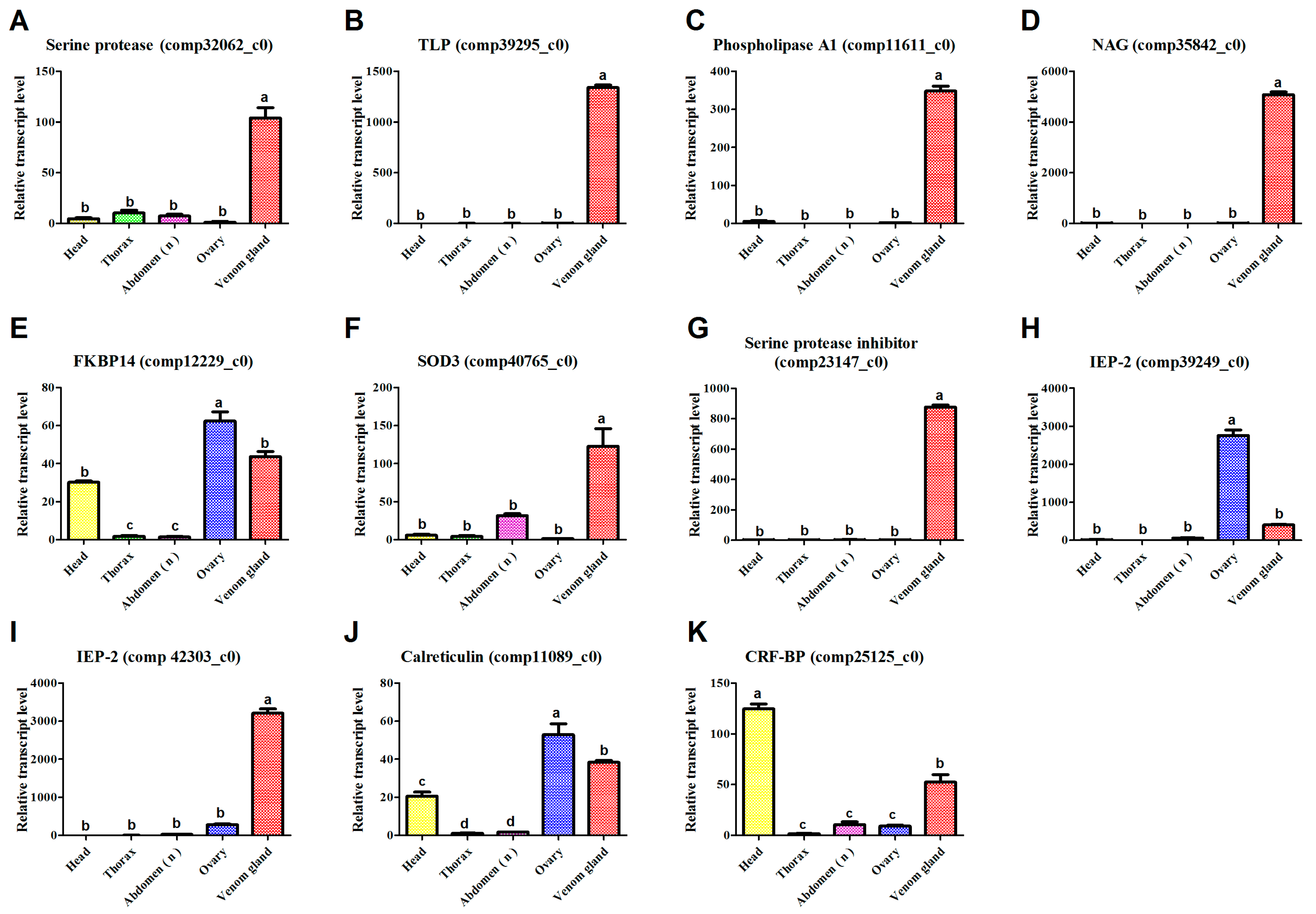
3.6. qPCR
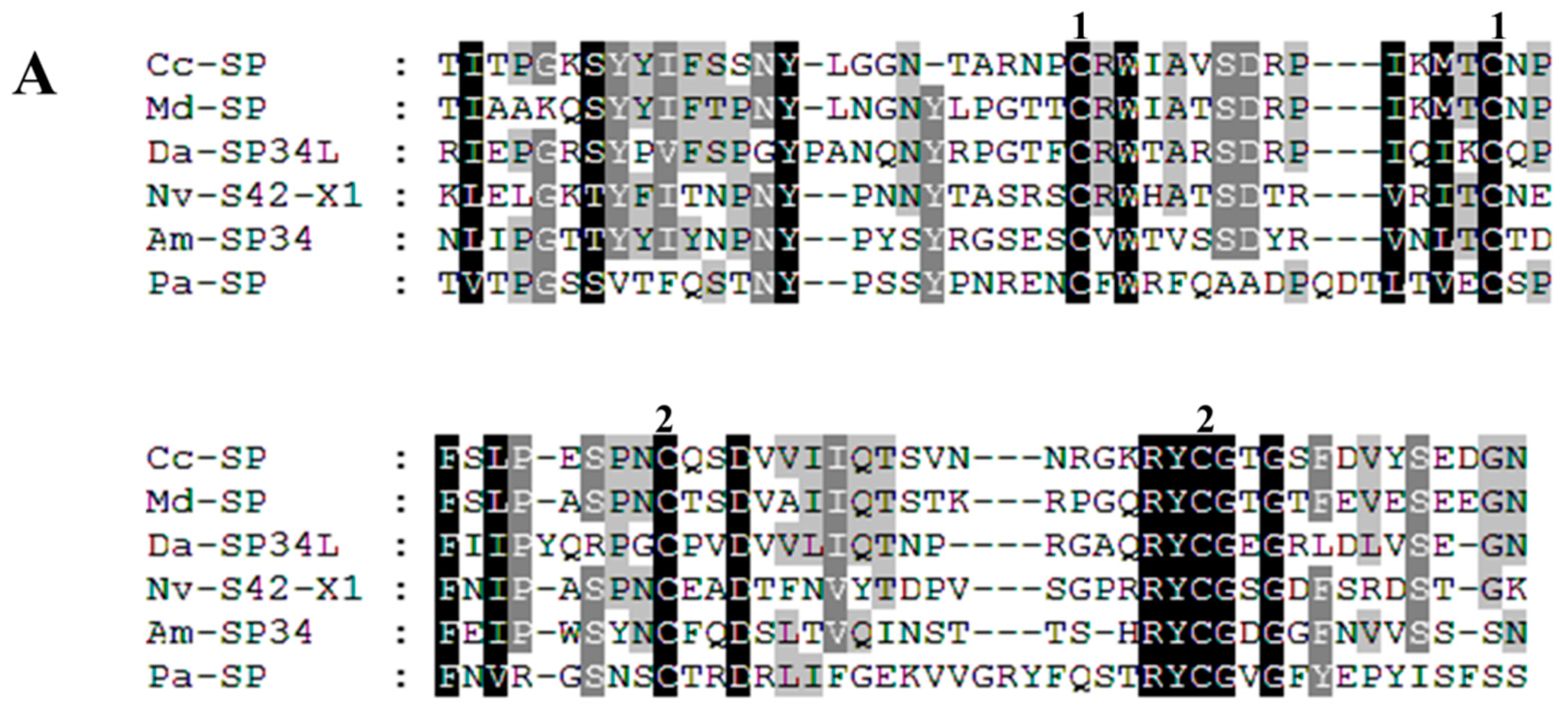
3.7. Sequence Alignment and Phylogenetic Analysis
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Asgari, S.; Rivers, D.B. Venom proteins from endoparasitoid wasps and their role in host-parasite interactions. Annu. Rev. Entomol. 2011, 56, 313–335. [Google Scholar] [CrossRef] [PubMed]
- Pennacchio, F.; Strand, M.R. Evolution of developmental strategies in parasitic Hymenoptera. Annu. Rev. Entomol. 2006, 51, 233–258. [Google Scholar] [CrossRef] [PubMed]
- Moreau, S.J.M.; Asgari, S. Venom proteins from parasitoid wasps and their biological functions. Toxins 2015, 7, 2385–2412. [Google Scholar] [CrossRef] [PubMed]
- Vincent, B.; Kaeslin, M.; Roth, T.; Heller, M.; Poulain, J.; Cousserans, F.; Schaller, J.; Poirie, M.; Lanzrein, B.; Drezen, J.M.; et al. The venom composition of the parasitic wasp Chelonus inanitus resolved by combined expressed sequence tags analysis and proteomic approach. BMC Genom. 2010, 11, 693. [Google Scholar] [CrossRef] [PubMed]
- Moore, E.L.; Haspel, G.; Libersat, F.; Adams, M.E. Parasitoid wasp sting: A cocktail of GABA, taurine, and β-alanine opens chloride channels for central synaptic block and transient paralysis of a cockroach host. J. Neurobiol. 2006, 66, 811–820. [Google Scholar] [CrossRef] [PubMed]
- Eldefrawi, A.T.; Eldefrawi, M.E.; Konno, K.; Mansour, N.A.; Nakanishi, K.; Oltz, E.; Usherwood, P.N.R. Structure and synthesis of a potent glutamate receptor antagonist in wasp venom. Proc. Natl. Acad. Sci. USA 1988, 85, 4910–4913. [Google Scholar] [CrossRef]
- Zhu, J.Y.; Wu, G.X.; Ze, S.Z.; Stanley, D.W.; Yang, B. Parasitization by Scleroderma guani influences protein expression in Tenebrio molitor pupae. J. Insect Physiol. 2014, 66, 37–44. [Google Scholar] [CrossRef]
- Danneels, E.L.; Formesyn, E.M.; Hahn, D.A.; Denlinger, D.L.; Cardoen, D.; Wenseleers, T.; Schoofs, L.; de Graaf, D.C. Early changes in the pupal transcriptome of the flesh fly Sarcophagha crassipalpis to parasitization by the ectoparasitic wasp, Nasonia vitripennis. Insect Biochem. Mol. Boil. 2013, 43, 1189–1200. [Google Scholar] [CrossRef] [PubMed]
- Parkinson, N.; Smith, I.; Audsley, N.; Edwards, J.P. Purification of pimplin, a paralytic heterodimeric polypeptide from venom of the parasitoid wasp Pimpla hypochondriaca, and cloning of the cDNA encoding one of the subunits. Insect Biochem. Mol. Boil. 2002, 32, 1769–1773. [Google Scholar] [CrossRef]
- Ergin, E.; Uckan, F.; Rivers, D.B.; Sak, O. In vivo and in vitro activity of venom from the endoparasitic wasp Pimpla turionellae (L.) (Hymenoptera: Ichneumonidae). Arch. Insect Biochem. Physiol. 2006, 61, 87–97. [Google Scholar] [CrossRef] [PubMed]
- Moreau, S.J.M.; Dingremont, A.; Doury, G.; Giordanengo, P. Effects of parasitism by Asobara tabida (Hymenoptera: Braconidae) on the development, survival and activity of Drosophila melanogaster larvae. J. Insect Physiol. 2002, 48, 337–347. [Google Scholar] [CrossRef]
- Desneux, N.; Barta, R.J.; Delebecque, C.J.; Heimpel, G.E. Transient host paralysis as a means of reducing self-superparasitism in koinobiont endoparasitoids. J. Insect Physiol. 2009, 55, 321–327. [Google Scholar] [CrossRef] [PubMed]
- Kaeslin, M.; Reinhard, M.; Buehler, D.; Roth, T.; Pfister-Wilhelm, R.; Lanzrein, B. Venom of the egg-larval parasitoid Chelonus inanitus is a complex mixture and has multiple biological effects. J. Insect Physiol. 2010, 56, 686–694. [Google Scholar] [CrossRef] [PubMed]
- Mabiala-Moundoungou, A.D.N.; Doury, G.; Eslin, P.; Cherqui, A.; Prevost, G. Deadly venom of Asobara japonica parasitoid needs ovarian antidote to regulate host physiology. J. Insect Physiol. 2010, 56, 35–41. [Google Scholar] [CrossRef] [PubMed]
- Asgari, S. Venom proteins from polydnavirus-producing endoparasitoids: Their role in host-parasite interactions. Arch. Insect Biochem. Physiol. 2006, 61, 146–156. [Google Scholar] [CrossRef] [PubMed]
- Labrosse, C.; Eslin, P.; Doury, G.; Drezen, J.M.; Poirie, M. Haemocyte changes in D. Melanogaster in response to long gland components of the parasitoid wasp Leptopilina boulardi: A Rho-GAP protein as an important factor. J. Insect Physiol. 2005, 51, 161–170. [Google Scholar] [CrossRef] [PubMed]
- Labrosse, C.; Staslak, K.; Lesobre, J.; Grangeia, A.; Huguet, E.; Drezen, J.M.; Poirie, M. A RhoGAP protein as a main immune suppressive factor in the Leptopilina boulardi (Hymenoptera, Figitidae)—Drosophila melanogaster interaction. Insect Biochem. Mol. Boil. 2005, 35, 93–103. [Google Scholar] [CrossRef] [PubMed]
- Zhang, G.; Schmidt, O.; Asgari, S. A calreticulin-like protein from endoparasitoid venom fluid is involved in host hemocyte inactivation. Dev. Comp. Immunol. 2006, 30, 756–764. [Google Scholar] [CrossRef] [PubMed]
- Richards, E.H.; Dani, M.P.; Bradish, H. Immunosuppressive properties of a protein (rVPr1) from the venom of the endoparasitic wasp, Pimpla hypochondriaca: Mechanism of action and potential use for improving biological control strategies. J. Insect Physiol. 2013, 59, 213–222. [Google Scholar] [CrossRef] [PubMed]
- Wu, M.L.; Ye, G.Y.; Zhu, J.Y.; Chen, X.X.; Hu, C. Isolation and characterization of an immunosuppressive protein from venom of the pupa-specific endoparasitoid Pteromalus puparum. J. Invertebr. Pathol. 2008, 99, 186–191. [Google Scholar] [CrossRef] [PubMed]
- Mortimer, N.T.; Goecks, J.; Kacsoh, B.Z.; Mobley, J.A.; Bowersock, G.J.; Taylor, J.; Schlenke, T.A. Parasitoid wasp venom SERCA regulates Drosophila calcium levels and inhibits cellular immunity. Proc. Natl. Acad. Sci. USA 2013, 110, 9427–9432. [Google Scholar] [CrossRef] [PubMed]
- Asgari, S.; Zareie, R.; Zhang, G.M.; Schmidt, O. Isolation and characterization of a novel venom protein from an endoparasitoid, Cotesia rubecula (Hym: Braconidae). Arch. Insect Biochem. Physiol. 2003, 53, 92–100. [Google Scholar] [CrossRef] [PubMed]
- Asgari, S.; Zhang, G.M.; Zareie, R.; Schmidt, O. A serine proteinase homolog venom protein from an endoparasitoid wasp inhibits melanization of the host hemolymph. Insect Biochem. Mol. Boil. 2003, 33, 1017–1024. [Google Scholar] [CrossRef]
- Colinet, D.; Dubuffet, A.; Cazes, D.; Moreau, S.; Drezen, J.-M.; Poirie, M. A serpin from the parasitoid wasp Leptopilina boulardi targets the Drosophila phenoloxidase cascade. Dev. Comp. Immunol. 2009, 33, 681–689. [Google Scholar] [CrossRef]
- Colinet, D.; Cazes, D.; Belghazi, M.; Gatti, J.-L.; Poirie, M. Extracellular superoxide dismutase in Insects characterization, function, and interspecific variation in parasitoid wasp venom. J. Biol. Chem. 2011, 286, 40110–40121. [Google Scholar] [CrossRef] [PubMed]
- Hoch, G.; Schopf, A. Effects of Glyptapanteles liparidis (Hym.: Braconidae) parasitism, polydnavirus, and venom on development of microsporidia-infected and uninfected Lymantria dispar (Lep.: Lymantriidae) larvae. J. Invertebr. Pathol. 2001, 77, 37–43. [Google Scholar] [CrossRef] [PubMed]
- Zhu, J.Y.; Ye, G.Y.; Dong, S.Z.; Fang, Q.; Hu, C. Venom of Pteromalus puparum (Hymenoptera: Pteromalidae) induced endocrine changes in the hemolymph of its host, Pieris rapae (Lepidoptera: Pieridae). Arch. Insect Biochem. Physiol. 2009, 71, 45–53. [Google Scholar] [CrossRef] [PubMed]
- Digilio, M.C.; Isidoro, N.; Tremblay, E.; Pennacchio, F. Host castration by Aphidius ervi venom proteins. J. Insect Physiol. 2000, 46, 1041–1050. [Google Scholar] [CrossRef]
- Falabella, P.; Riviello, L.; Caccialupi, P.; Rossodivita, T.; Valente, M.T.; De Stradis, M.L.; Tranfaglia, A.; Varricchio, P.; Gigliotti, S.; Graziani, F.; et al. A γ-glutamyl transpeptidase of Aphidius ervi venom induces apoptosis in the ovaries of host aphids. Insect Biochem. Mol. Boil. 2007, 37, 453–465. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, T. Effect of the venom of the endoparasitoid, Apanteles kariyai Watanabe, on the cellular defence reaction of the host, Pseudaletia separata Walker. J. Insect Physiol. 1987, 33, 413–420. [Google Scholar] [CrossRef]
- Stoltz, D.B.; Guzo, D.; Belland, E.R.; Lucarotti, C.J.; Mackinnon, E.A. Venom promotes uncoating in vitro and persistence in vivo of DNA from a braconid polydnavirus. J. Gen. Virol. 1988, 69, 903–907. [Google Scholar] [CrossRef]
- Gupta, P.; Ferkovich, S.M. Interaction of calyx fluid and venom from Microplitis croceipes (Braconidae) on developmental disruption of the natural host, Heliocoverpa zea, and two atypical hosts, Galleria mellonella and Spodoptera exigua. J. Insect Physiol. 1998, 44, 713–719. [Google Scholar] [CrossRef]
- Zhang, G.M.; Schmidt, O.; Asgari, S. A novel venom peptide from an endoparasitoid wasp is required for expression of polydnavirus genes in host hemocytes. J. Biol. Chem. 2004, 279, 41580–41585. [Google Scholar] [CrossRef] [PubMed]
- Dorémus, T.; Urbach, S.; Jouan, V.; Cousserans, F.; Ravallec, M.; Demettre, E.; Wajnberg, E.; Poulain, J.; Azema-Dossat, C.; Darboux, I.; et al. Venom gland extract is not required for successful parasitism in the polydnavirus-associated endoparasitoid Hyposoter didymator (Hym. Ichneumonidae) despite the presence of numerous novel and conserved venom proteins. Insect Biochem. Mol. Boil. 2013, 43, 292–307. [Google Scholar] [CrossRef] [PubMed]
- Burke, G.R.; Strand, M.R. Systematic analysis of a wasp parasitism arsenal. Mol. Ecol. 2014, 23, 890–901. [Google Scholar] [CrossRef] [PubMed]
- Goecks, J.; Mortimer, N.T.; Mobley, J.A.; Bowersock, G.J.; Taylor, J.; Schlenke, T.A. Integrative approach reveals composition of endoparasitoid wasp venoms. PLoS ONE 2013, 8, e64125. [Google Scholar] [CrossRef] [PubMed]
- Colinet, D.; Anselme, C.; Deleury, E.; Mancini, D.; Poulain, J.; Azema-Dossat, C.; Belghazi, M.; Tares, S.; Pennacchio, F.; Poirie, M.; et al. Identification of the main venom protein components of Aphidius ervi, a parasitoid wasp of the aphid model Acyrthosiphon pisum. BMC Genom. 2014, 15, 342. [Google Scholar] [CrossRef] [PubMed]
- Laurino, S.; Grossi, G.; Pucci, P.; Flagiello, A.; Bufo, S.A.; Bianco, G.; Salvia, R.; Vinson, S.B.; Vogel, H.; Falabella, P. Identification of major Toxoneuron nigriceps venom proteins using an integrated transcriptomic/proteomic approach. Insect Biochem. Mol. Boil. 2016, 76, 49–61. [Google Scholar] [CrossRef] [PubMed]
- Wu, S.F.; Sun, F.D.; Qi, Y.X.; Yao, Y.; Fang, Q.; Huang, J.; Stanley, D.; Ye, G.Y. Parasitization by Cotesia chilonis Influences gene expression in fatbody and hemocytes of Chilo suppressalis. PLoS ONE 2013, 8, e74309. [Google Scholar] [CrossRef] [PubMed]
- Lou, Y.G.; Zhang, G.R.; Zhang, W.Q.; Hu, Y.; Zhang, J. Reprint of: biological control of rice insect pests in China. Biol. Control 2014, 68, 103–116. [Google Scholar] [CrossRef]
- Qi, Y.X.; Teng, Z.W.; Gao, L.F.; Wu, S.F.; Huang, J.; Ye, G.Y.; Fang, Q. Transcriptome analysis of an endoparasitoid wasp Cotesia chilonis (Hymenoptera: Braconidae) reveals genes involved in successful parasitism. Arch. Insect Biochem. Physiol. 2014, 1, 1–19. [Google Scholar]
- Teng, Z.W.; Xu, G.; Gan, S.Y.; Chen, X.; Fang, Q.; Ye, G.Y. Effects of the endoparasitoid Cotesia chilonis (Hymenoptera: Braconidae) parasitism, venom, and calyx fluid on cellular and humoral immunity of its host Chilo suppressalis (Lepidoptera: Crambidae) larvae. J. Insect Physiol. 2016, 85, 46–56. [Google Scholar] [CrossRef] [PubMed]
- Grabherr, M.G.; Haas, B.J.; Yassour, M.; Levin, J.Z.; Thompson, D.A.; Amit, I.; Adiconis, X.; Fan, L.; Raychowdhury, R.; Zeng, Q.; et al. Full-length transcriptome assembly from RNA-Seq data without a reference genome. Nat. Biotechnol. 2011, 29, 644–652. [Google Scholar] [CrossRef] [PubMed]
- Colinet, D.; Deleury, E.; Anselme, C.; Cazes, D.; Poulain, J.; Azema-Dossat, C.; Belghazi, M.; Gatti, J.L.; Poirie, M. Extensive inter- and intraspecific venom variation in closely related parasites targeting the same host: The case of Leptopilina parasitoids of Drosophila. Insect Biochem. Mol. Boil. 2013, 43, 601–611. [Google Scholar] [CrossRef] [PubMed]
- Furihata, S.; Tanaka, K.; Ryuda, M.; Ochiai, M.; Matsumoto, H.; Csikos, G.; Hayakawa, Y. Immunoevasive protein (IEP)-containing surface layer covering polydnavirus particles is essential for viral infection. J. Invertebr. Pathol. 2014, 115, 26–32. [Google Scholar] [CrossRef] [PubMed]
- De Graaf, D.C.; Aerts, M.; Brunain, M.; Desjardins, C.A.; Jacobs, F.J.; Werren, J.H.; Devreese, B. Insights into the venom composition of the ectoparasitoid wasp Nasonia vitripennis from bioinformatic and proteomic studies. Insect Mol. Biol. 2010, 19, 11–26. [Google Scholar] [CrossRef] [PubMed]
- Choo, Y.M.; Lee, K.S.; Yoon, H.J.; Kim, B.Y.; Sohn, M.R.; Roh, J.Y.; Je, Y.H.; Kim, N.J.; Kim, I.; Woo, S.D.; et al. Dual function of a bee venom serine protease: prophenoloxidase-activating factor in arthropods and fibrin(ogen)olytic enzyme in mammals. PLoS ONE 2010, 5, e10393. [Google Scholar] [CrossRef] [PubMed]
- Yamada, K.; Takabatake, T.; Takeshima, K. Isolation and characterization of three novel serine protease genes from Xenopus laevis. Gene 2000, 252, 209–216. [Google Scholar] [CrossRef]
- Jiang, H.; Kanost, M.R. The clip-domain family of serine proteinases in arthropods. Insect Biochem. Mol. Boil. 2000, 30, 95–105. [Google Scholar] [CrossRef]
- Dos Santos, L.D.; Santos, K.S.; Aparecido Pinto, J.R.; Dias, N.B.; de Souza, B.M.; dos Santos, M.F.; Perales, J.; Domont, G.B.; Castro, F.M.; Kalil, J.E.; et al. Profiling the proteome of the venom from the social wasp Polybia paulista: A clue to understand the envenoming mechanism. J. Proteome Res. 2010, 9, 3867–3877. [Google Scholar] [CrossRef] [PubMed]
- Zou, Z.; Evans, J.D.; Lu, Z.; Zhao, P.; Williams, M.; Sumathipala, N.; Hetru, C.; Hultmark, D.; Jiang, H. Comparative genomic analysis of the Tribolium immune system. Genome Biol. 2007, 8, R177. [Google Scholar] [CrossRef] [PubMed]
- Xiong, G.-H.; Xing, L.-S.; Lin, Z.; Saha, T.T.; Wang, C.; Jiang, H.; Zou, Z. High throughput profiling of the cotton bollworm Helicoverpa armigera immunotranscriptome during the fungal and bacterial infections. BMC Genom. 2015, 16, 321. [Google Scholar] [CrossRef] [PubMed]
- Waterhouse, R.M.; Kriventseva, E.V.; Meister, S.; Xi, Z.; Alvarez, K.S.; Bartholomay, L.C.; Barillas-Mury, C.; Bian, G.; Blandin, S.; Christensen, B.M.; et al. Evolutionary dynamics of immune-related genes and pathways in disease-vector mosquitoes. Science 2007, 316, 1738–1743. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.-H.; Hu, Y.; Xing, L.-S.; Jiang, H.; Hu, S.-N.; Raikhel, A.S.; Zou, Z. A critical role for CLSP2 in the modulation of antifungal immune response in Mosquitoes. PLoS Pathog. 2015, 11. [Google Scholar] [CrossRef] [PubMed]
- Yan, Z.; Fang, Q.; Wang, L.; Liu, J.; Zhu, Y.; Wang, F.; Li, F.; Werren, J.H.; Ye, G. Insights into the venom composition and evolution of an endoparasitoid wasp by combining proteomic and transcriptomic analyses. Sci. Rep. 2016, 6, 19604. [Google Scholar] [CrossRef] [PubMed]
- Christophides, G.K.; Zdobnov, E.; Barillas-Mury, C.; Birney, E.; Blandin, S.; Blass, C.; Brey, P.T.; Collins, F.H.; Danielli, A.; Dimopoulos, G.; et al. Immunity-related genes and gene families in Anopheles gambiae. Science 2002, 298, 159–165. [Google Scholar] [CrossRef] [PubMed]
- Ross, J.; Jiang, H.; Kanost, M.R.; Wang, Y. Serine proteases and their homologs in the Drosophila melanogaster genome: An initial analysis of sequence conservation and phylogenetic relationships. Gene 2003, 304, 117–131. [Google Scholar] [CrossRef]
- PhylML with Smart Model Selection. Available online: http://www.atgc-montpellier.fr/phyml-sms/ (accessed on 17 February 2017).
- Kelwick, R.; Desanlis, I.; Wheeler, G.N.; Edwards, D.R. The ADAMTS (a disintegrin and metalloproteinase with thrombospondin motifs) family. Genome Biol. 2015, 16, 113. [Google Scholar] [CrossRef] [PubMed]
- Takeda, S.; Takeya, H.; Iwanaga, S. Snake venom metalloproteinases: structure, function and relevance to the mammalian ADAM/ADAMTS family proteins. Biochim. Biophys. Acta 2012, 1824, 164–176. [Google Scholar] [CrossRef] [PubMed]
- Smith, K.M.; Gaultier, A.; Cousin, H.; Alfandari, D.; White, J.M.; DeSimone, D.W. The cysteine-rich domain regulates ADAM protease function in vivo. J. Cell Biol. 2002, 159, 893–902. [Google Scholar] [CrossRef] [PubMed]
- Moran, Y.; Praher, D.; Schlesinger, A.; Ayalon, A.; Tal, Y.; Technau, U. Analysis of soluble protein contents from the nematocysts of a model sea anemone sheds light on venom evolution. Mar. Biotechnol. 2013, 15, 329–339. [Google Scholar] [CrossRef] [PubMed]
- Fry, B.G.; Roelants, K.; Champagne, D.E.; Scheib, H.; Tyndall, J.D.A.; King, G.F.; Nevalainen, T.J.; Norman, J.A.; Lewis, R.J.; Norton, R.S.; et al. The toxicogenomic multiverse: convergent recruitment of proteins into animal venoms. Annu. Rev. Genom. Hum. G 2009, 10, 483–511. [Google Scholar] [CrossRef] [PubMed]
- Bond, J.S.; Beynon, R.J. The astacin family of metalloendopeptidases. Protein Sci. 1995, 4, 1247–1261. [Google Scholar] [CrossRef] [PubMed]
- Xavier Gomis-Rueth, F.; Trillo-Muyo, S.; Stoecker, W. Functional and structural insights into astacin metallopeptidases. Biol. Chem. 2012, 393, 1027–1041. [Google Scholar]
- Trevisan-Silva, D.; Gremski, L.H.; Chaim, O.M.; da Silveira, R.B.; Meissner, G.O.; Mangili, O.C.; Barbaro, K.C.; Gremski, W.; Veiga, S.S.; Senff-Ribeiro, A. Astacin-like metalloproteases are a gene family of toxins present in the venom of different species of the brown spider (genus Loxosceles). Biochimie 2010, 92, 21–32. [Google Scholar] [CrossRef] [PubMed]
- Lee, V.S.Y.; Tu, W.C.; Jinn, T.R.; Peng, C.C.; Lin, L.J.; Tzen, J.T.C. Molecular cloning of the precursor polypeptide of mastoparan B and its putative processing enzyme, dipeptidyl peptidase IV, from the black-bellied hornet, Vespa basalis. Insect Mol. Biol. 2007, 16, 231–237. [Google Scholar] [CrossRef] [PubMed]
- Shao, C.; Shi, X.; Wehbi, H.; Zambonelli, C.; Head, J.F.; Seaton, B.A.; Roberts, M.F. Dimer structure of an interfacially impaired phosphatidylinositol-specific phospholipase C. J. Biol. Chem. 2007, 282, 9228–9235. [Google Scholar] [CrossRef] [PubMed]
- Chayakulkeeree, M.; Sorrel, T.C.; Siafakas, A.R.; Wilson, C.F.; Pantarat, N.; Gerik, K.J.; Boadle, R.; Djordievic, J.T. Role and mechanism of phosphatidylinositol-specific phospholipase C in survival and virulence of Cryptococcus neoformans. Mol. Microbiol. 2008, 69, 809–826. [Google Scholar] [PubMed]
- Lemieux, M.J.; Mark, B.L.; Cherney, M.M.; Withers, S.G.; Mahuran, D.J.; James, M.N.G. Crystallographic structure of human β-hexosaminidase A: Interpretation of Tay-Sachs mutations and loss of GM2 ganglioside hydrolysis. J. Mol. Biol. 2006, 359, 913–929. [Google Scholar] [CrossRef] [PubMed]
- Hogenkamp, D.G.; Arakane, Y.; Kramer, K.J.; Muthukrishnan, S.; Beeman, R.W. Characterization and expression of the β-N-acetylhexosaminidase gene family of Tribolium castaneum. Insect Biochem. Mol. Boil. 2008, 38, 478–489. [Google Scholar] [CrossRef] [PubMed]
- Jones, D.; Wache, S.; Chhokar, V. Toxins produced by arthropod parasites: salivary gland proteins of human body lice and venom proteins of chelonine wasps. Toxicon 1996, 34, 1421–1429. [Google Scholar] [CrossRef]
- Krishnan, A.; Nair, P.N.; Jones, D. Isolation, cloning, and characterization of new chitinase stored in active form in chitin-lined venom reservoir. J. Biol. Chem. 1994, 269, 20971–20976. [Google Scholar] [PubMed]
- Danpaiboon, W.; Reamtong, O.; Sookrung, N.; Seesuay, W.; Sakolvaree, Y.; Thanongsaksrikul, J.; Dong-din-on, F.; Srimanote, P.; Thueng-in, K.; Chaicumpa, W. Ophiophagus hannah venom: Proteome, components bound by Naja kaouthia antivenin and neutralization by N. kaouthia neurotoxin-specific human ScFv. Toxins 2014, 6, 1526–1558. [Google Scholar] [CrossRef] [PubMed]
- Neeman, I.; Calton, G.J.; Burnett, J.W. Purification and characterization of the endonuclease present in Physalia physalis venom. Comp. Biochem. Phys. B 1980, 67, 155–158. [Google Scholar] [CrossRef]
- Neeman, I.; Calton, G.J.; Burnett, J.W. Purification of an endonuclease present in Chrysaora quinquecirrha venom. Proc. Soc. Exp. Biol. Med. 1981, 166, 374–382. [Google Scholar] [CrossRef] [PubMed]
- Barik, S. Immunophilins: For the love of proteins. Cell. Mol. Life Sci. 2006, 63, 2889–2900. [Google Scholar] [CrossRef] [PubMed]
- Kang, C.B.; Ye, H.; Dhe-Paganon, S.; Yoon, H.S. FKBP family proteins: Immunophilins with versatile biological functions. Neurosignals 2008, 16, 318–325. [Google Scholar] [CrossRef] [PubMed]
- Alnemri, E.S.; Fernandesalnemri, T.; Pomerenke, K.; Robertson, N.M.; Dudley, K.; Dubois, G.C.; Litwack, G. FKBP46, a novel Sf9 insect cell nuclear immunophilin that forms a protein-kinase complex. J. Biol. Chem. 1994, 269, 30828–30834. [Google Scholar] [PubMed]
- Song, Q.S.; Alnemri, E.S.; Litwack, G.; Gilbert, L.I. An immunophilin is a component of the insect ecdysone receptor (EcR) complex. Insect Biochem. Mol. Boil. 1997, 27, 973–982. [Google Scholar] [CrossRef]
- Wilkinson, B.; Gilbert, H.F. Protein disulfide isomerase. BBA Proteins Proteom. 2004, 1699, 35–44. [Google Scholar] [CrossRef]
- Safavi-Hemami, H.; Gorasia, D.G.; Steiner, A.M.; Williamson, N.A.; Karas, J.A.; Gajewiak, J.; Olivera, B.M.; Bulaj, G.; Purcell, A.W. Modulation of conotoxin structure and function is achieved through a multienzyme complex in the venom glands of cone snails. J. Biol. Chem. 2012, 287, 34288–34303. [Google Scholar] [CrossRef] [PubMed]
- Di Luccio, E.; Azulay, D.O.; Regaya, I.; Fajloun, Z.; Sandoz, G.; Mansuelle, P.; Kharrat, R.; Fathallah, M.; Carrega, L.; Esteve, E.; et al. Parameters affecting in vitro oxidation/folding of maurotoxin, a four-disulphide-bridged scorpion toxin. Biochem. J. 2001, 358, 681–692. [Google Scholar] [CrossRef]
- Mathe-Hubert, H.; Colinet, D.; Deleury, E.; Belghazi, M.; Ravallec, M.; Poulain, J.; Dossat, C.; Poirie, M.; Gatti, J.-L. Comparative venomics of Psyttalia lounsburyi and P. concolor, two olive fruit fly parasitoids: A hypothetical role for a GH1 β-glucosidase. Sci. Rep. 2016, 6, 35873. [Google Scholar] [CrossRef]
- Parkinson, N.M.; Conyers, C.; Keen, J.; MacNicoll, A.; Smith, I.; Audsley, N.; Weaver, R. Towards a comprehensive view of the primary structure of venom proteins from the parasitoid wasp Pimpla hypochondriaca. Insect Biochem. Mol. Boil. 2004, 34, 565–571. [Google Scholar] [CrossRef] [PubMed]
- Hayakawa, Y.; Yazaki, K. Envelope protein of parasitic wasp symbiont virus, polydnavirus, protects the wasp eggs from cellular immune reactions by the host insect. Eur. J. Biochem. 1997, 246, 820–826. [Google Scholar] [CrossRef]
- Zhu, J.Y.; Fang, Q.; Wang, L.; Hu, C.; Ye, G.Y. Proteomic analysis of the venom from the endoparasitoid wasp Pteromalus puparum (Hymenoptera: Pteromalidae). Arch. Insect Biochem. Physiol. 2010, 75, 28–44. [Google Scholar] [CrossRef] [PubMed]
- Crawford, A.M.; Brauning, R.; Smolenski, G.; Ferguson, C.; Barton, D.; Wheeler, T.T.; Mcculloch, A. The constituents of Microctonus sp. parasitoid venoms. Insect Mol. Biol. 2008, 17, 313–324. [Google Scholar] [CrossRef] [PubMed]
- Ferreira, V.; Molina, M.C.; Valck, C.; Rojas, A.; Aguilar, L.; Ramirez, G.; Schwaeble, W.; Ferreira, A. Role of calreticulin from parasites in its interaction with vertebrate hosts. Mol. Immunol. 2004, 40, 1279–1291. [Google Scholar] [CrossRef] [PubMed]
- Siebert, A.L.; Wheeler, D.; Werren, J.H. A new approach for investigating venom function applied to venom calreticulin in a parasitoid wasp. Toxicon 2015, 107, 304–316. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.; Tang, X.; Gong, L. Mesencephalic astrocyte-derived neurotrophic factor and cerebral dopamine neurotrophic factor: New endoplasmic reticulum stress response proteins. Eur. J. Pharmacol. 2015, 750, 118–122. [Google Scholar] [CrossRef] [PubMed]
- Walsh, P.; Bursac, D.; Law, Y.C.; Cyr, D.; Lithgow, T. The J-protein family: Modulating protein assembly, disassembly and translocation. EMBO Rep. 2004, 5, 567–571. [Google Scholar] [CrossRef] [PubMed]
- Baryshev, M.; Sargsyan, E.; Mkrtchian, S. ERp29 is an essential endoplasmic reticulum factor regulating secretion of thyroglobulin. Biochem. Biophys. Res. Commun. 2006, 340, 617–624. [Google Scholar] [CrossRef] [PubMed]
- Sannino, S.; Anelli, T.; Cortini, M.; Masui, S.; Degano, M.; Fagioli, C.; Inaba, K.; Sitia, R. Progressive quality control of secretory proteins in the early secretory compartment by ERp44. J. Cell Sci. 2014, 127, 4260–4269. [Google Scholar] [CrossRef] [PubMed]
- Radons, J. The human HSP70 family of chaperones: Where do we stand? Cell Stress Chaperones 2016, 21, 379–404. [Google Scholar] [CrossRef] [PubMed]
- Bruneau, N.; Lombardo, D.; Levy, E.; Bendayan, M. Roles of molecular chaperones in pancreatic secretion and their involvement in intestinal absorption. Microsc. Res. Techniq. 2000, 49, 329–345. [Google Scholar] [CrossRef]
- Han, L.Z.; Li, S.B.; Liu, P.L.; Peng, Y.F.; Hou, M.L. New artificial diet for continuous rearing of Chilo suppressalis (Lepidoptera: Crambidae). Ann. Entomol. Soc. Am. 2012, 105, 253–258. [Google Scholar] [CrossRef]
- NCBI Short Read Archive Database. Available online: http://www.ncbi.nlm.nih.gov/Traces/sra/ (accessed on 12 October 2013).
- Robinson, M.D.; McCarthy, D.J.; Smyth, G.K. edgeR: A Bioconductor package for differential expression analysis of digital gene expression data. Bioinformatics 2010, 26, 139–140. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.K.; Feng, Z.X.; Wang, X.; Wang, X.W.; Zhang, X.G. DEGseq: An R package for identifying differentially expressed genes from RNA-seq data. Bioinformatics 2010, 26, 136–138. [Google Scholar] [CrossRef] [PubMed]
- Storey, J.D. The positive false discovery rate: A Bayesian interpretation and the q-value. Ann. Stat. 2003, 31, 2013–2035. [Google Scholar] [CrossRef]
- Young, M.D.; Wakefield, M.J.; Smyth, G.K.; Oshlack, A. Gene ontology analysis for RNA-seq: Accounting for selection bias. Genome Biol. 2010, 11, R14. [Google Scholar] [CrossRef] [PubMed]
- Eng, J.K.; McCormack, A.L.; Yates, J.R. An approach to correlate tandem mass-spectral data of peptides with amino-acid-sequences in a protein database. J. Am. Soc. Mass Spectrom. 1994, 5, 976–989. [Google Scholar] [CrossRef]
- Livak, K.J.; Schmittgen, T.D. Analysis of relative gene expression data using real-time quantitative PCR and the 2−ΔΔCT method. Methods 2001, 25, 402–408. [Google Scholar] [CrossRef] [PubMed]
- Bustin, S.A.; Benes, V.; Garson, J.A.; Hellemans, J.; Huggett, J.; Kubista, M.; Mueller, R.; Nolan, T.; Pfaffl, M.W.; Shipley, G.L.; et al. The MIQE guidelines: Minimum information for publication of quantitative real-time PCR experiments. Clin. Chem. 2009, 55, 611–622. [Google Scholar] [CrossRef] [PubMed]
- Tang, Q.Y.; Zhang, C.X. Data Processing System (DPS) software with experimental design, statistical analysis and data mining developed for use in entomological research. Insect Sci. 2013, 20, 254–260. [Google Scholar] [CrossRef] [PubMed]
- SignalP 4.1 Server. Available online: http://www.cbs.dtu.dk/services/SignalP/ (accessed on 7 December 2016).
- Larkin, M.A.; Blackshields, G.; Brown, N.P.; Chenna, R.; McGettigan, P.A.; McWilliam, H.; Valentin, F.; Wallace, I.M.; Wilm, A.; Lopez, R.; et al. Clustal W and clustal X version 2.0. Bioinformatics 2007, 23, 2947–2948. [Google Scholar] [CrossRef] [PubMed]
- BeetleBase. Available online: http://www.bioinformatics.ksu.edu/BeetleBase/ (accessed on 17 February 2017).
- Pfam HMM. Available online: http://www.ebi.ac.uk/Tools/pfa/pfamscan/ (accessed on 17 February 2017).
- Cao, X.; He, Y.; Hu, Y.; Zhang, X.; Wang, Y.; Zou, Z.; Chen, Y.; Blissard, G.W.; Kanost, M.R.; Jiang, H. Sequence conservation, phylogenetic relationships, and expression profiles of nondigestive serine proteases and serine protease homologs in Manduca sexta. Insect Biochem. Mol. Boil. 2015, 62, 51–63. [Google Scholar] [CrossRef] [PubMed]
- Gblocks Server. Available online: http://molevol.cmima.csic.es/castresana/Gblocks_server.html (accessed on 17 February 2017).
- Hu, J.; Xu, Q.Y.; Hu, S.F.; Yu, X.Q.; Liang, Z.K.; Zhang, W.Q. Hemomucin, an O-glycosylated protein on embryos of the wasp Macrocentrus cingulum that protects It against encapsulation by hemocytes of the host Ostrinia furnacalis. J. Innate Immun. 2014, 6, 663–675. [Google Scholar] [CrossRef] [PubMed]
- Asgari, S.; Theopold, U.; Wellby, C.; Schmidt, O. A protein with protective properties against the cellular defense reactions in insects. Proc. Natl. Acad. Sci. USA 1998, 95, 3690–3695. [Google Scholar] [CrossRef] [PubMed]
- Sanahuja, G.; Banakar, R.; Twyman, R.M.; Capell, T.; Christou, P. Bacillus thuringiensis: A century of research, development and commercial applications. Plant Biotechnol. J. 2011, 9, 283–300. [Google Scholar] [CrossRef] [PubMed]
- Irwin, M.E.; Kampmeier, G.E. Commercial products from insects. In Encyclopedia of Insects; Resh, V.H., Cardé, R.T., Eds.; Academic Press: San Diego, CA, USA, 2003. [Google Scholar]
- Zasloff, M. Antimicrobial peptides of multicellular organisms. Nature 2002, 415, 389–395. [Google Scholar] [CrossRef] [PubMed]

| Transcripts/Unigenes | Raw Reads | Clean Reads | No. of Transcripts/Unigenes | N50 | Meanlength | ||
|---|---|---|---|---|---|---|---|
| Venom Gland | Female Adult Carcass | Venom Gland | Female Adult Carcass | - | - | - | |
| Transcripts | 67,097,344 | 63,686,328 | 65,515,114 | 62,221,866 | 115,957 | 2393 | 1254 |
| Unigenes | 71,617 | 1309 | 759 | ||||
| GO_Accession | Description | # Unigenes | Percentages * |
|---|---|---|---|
| GO:0003735 | Structural constituent of ribosome | 37 | 13.3 |
| GO:0004553 | Hydrolase activity, hydrolyzing O-glycosyl compounds | 14 | 5.0 |
| GO:0004620 | Phospholipase activity | 7 | 2.5 |
| GO:0004629 | Phospholipase C activity | 6 | 2.2 |
| GO:0005198 | Structural molecule activity | 42 | 15.1 |
| GO:0008233 | Peptidase activity | 30 | 10.8 |
| GO:0008237 | Metallopeptidase activity | 15 | 5.4 |
| GO:0016798 | Hydrolase activity, acting on glycosyl bonds | 14 | 5.0 |
| GO:0046982 | Protein heterodimerization activity | 3 | 1.1 |
| Protein Name | RPKM/UniquePepCount | Putative Function | Sequence Length (Signal Peptide) | Blast Information (E-Value; Genbank No.; Species) | Gene ID | Accession No. |
|---|---|---|---|---|---|---|
| Hydrolases | ||||||
| Proteases | ||||||
| Cc-Ven1 | 106.52 (M)/2 | Serine protease | 390 (Y) | 2e−130; NP_001155078.1; Nasonia vitripennis | comp32062_c0 | KU663618 |
| Cc-Ven2 | 47.99 (L)/9 | Disintegrin and Metalloproteinase with thrombospondin motifs | 509 (Y) | 1e−58; EGI57486.1; Acromyrmex echinatior | comp35977_c0 | KU663619 |
| Cc-Ven3 | 212.13 (M)/11 | Tolloid-like protein | 478 (Y) | 1.00e−94; XP_003695260.2; Apis florea | comp35892_c0 | KU663620 |
| Cc-Ven4 | 219.32 (M)/30 | Tolloid-like protein | 477 (Y) | 7.00e−124; P_003695260.2; Apis florea | comp39295_c0 | KU663621 |
| Peptidases | ||||||
| Cc-Ven5 | 1665.96 (H)/18 | Retinoid-inducible serine carboxypeptidase-like | 415 (Y) | 5.00e−71; XP_001605442.1; Nasonia vitripennis | comp11050_c0 | KU663622 |
| Cc-Ven6 | 261.11 (M)/1 | Aminopeptidase N-like | 148 (NC) | 3.00e−41; EFN65598.1; Camponotus floridanus | comp43465_c0 | KU663623 |
| Esterases | ||||||
| Cc-Ven7 | 4876.97 (H)/9 | 1-phosphatidyl inositol phosphodiesterase precursor | 230 (NC) | 6.00e−10; XP 007809166.1; Metarhizium acridum | comp44319_c0 | KU663624 |
| Cc-Ven8 | 1010.49 (H)/30 | Phosphatidylinositol-specific phospholipase | 323 (Y) | 4.00e−52; EZA44899.1; Microplitis demolitor | comp43453_c0 | KU663625 |
| Cc-Ven9 | 7010.08 (H)/29 | Phosphatidylinositol-specific phospholipase | 323 (Y) | 2.00e−55; EZA44899.1; Microplitis demolitor | comp43453_c1 | KU663626 |
| Cc-Ven10 | 194.85 (M)/9 | Phospholipase A1 | 295 (Y) | 1.00e−54; EGI61859.1; Acromyrmex echinatior | comp11611_c0 | KU663627 |
| Cc-Ven11 | 427.00 (M)/14 | Phospholipase A1 | 313 (Y) | 6.00e−89; XP 011258702.1; Camponotus floridanus | comp35865_c0 | KU663628 |
| Glycosyl hydrolase | ||||||
| Cc-Ven12 | 437.68 (M)/35 | Chitooligosaccharidolytic β- N-acetylglucosaminidase | 603 (Y) | 0; XP_008213962.1; Nasonia vitripennis | comp35842_c0 | KU663629 |
| Endonuclease | ||||||
| Cc-Ven13 | 24.39 (L)/1 | Enzymatic polyprotein Endonuclease; Reverse transcriptase | 1660 (NC) | 1e−126; XP 002431503.1; Pediculus humanus corporis | comp43476_c1 | KU663630 |
| Isomerase | ||||||
| Cc-Ven14 | 202.99 (M)/12 | FK506-binding protein 14 | 239 (Y) | 6e−105; NP_476973.1; Drosophila melanogaster | comp12229_c0 | KU663631 |
| Cc-Ven15 | 203.89 (M)/14 | Protein disulfide-isomerase | 2002 (Y) | 0; XP_001602967.1; Nasonia vitripennis | comp32146_c0 | - |
| Cc-Ven16 | 412.07 (M)/1 | Protein disulfide-isomerase | 3554 (Y) | 8e−167; XP_011138070.1; Harpegnathos saltator | comp40525_c0 | - |
| Extracellular superoxide dismutase 3 | ||||||
| Cc-Ven17 | 10.97 (L)/1 | Extracellular superoxide dismutase 3 | 172 (Y) | 2.00e−56; AET83769.1/AET83767.1; Leptopilina boulardi) | comp40765_c0 | KU663632 |
| Serine protease inhibitors | ||||||
| Cc-Ven18 | 466.16 (M)/1 | Kazal-type proteinase inhibitor | 120 (Y) | 3.00e−22; BAO48212.1; Camponotus japonicus | comp23198_c0 | KU663633 |
| Cc-Ven19 | 4970.74 (H)/16 | Serine protease inhibitor (Serpin) | 406 (Y) | 3.00e−71; XP_008201843.1 Nasonia vitripennis | comp23147_c0 | KU663634 |
| Other venom proteins (Non enzymatic and inhibitor activities) | ||||||
| Cc-Ven20 | 89.09 (L)/10 | Immunoevasive protein-2 | 320 (NC) | 1.00e−62; BAB72015.1 Cotesia kariyai. | comp39249_c0 | KU663635 |
| Cc-Ven21 | 103.23 (M)/7 | Immunoevasive protein-2 | 180 (NC) | 5.00e−13; BAB72015.1 Cotesia kariyai. | comp42303_c0 | KU663636 |
| Cc-Ven22 | 377.40 (M)/21 | Calreticulin | 403 (Y) | 0; AAN73309.1; Cotesia rubecula | comp11089_c0 | KU663637 |
| Cc-Ven23 | 11187.79 (H)/34 | Venom protein Ci-48a | 384 (NC) | 7.00e−14; CBM69271.1; Chelonus inanitus | comp39158_c0 | KU663638 |
| Cc-Ven24 | 1862.61 (H)/5 | Icarapin-like precursor | 227 (Y) | 2.00e−20; NP 001012431.1; Apis mellifera | comp44327_c0 | KU663639 |
| Cc-Ven25 | 24.06 (L)/5 | Corticotropin-releasing factor-binding protein | 327 (Y) | 3.00e−95; XP_003692566.1; Apis florea | comp25125_c0 | KU663640 |
| Cc-Ven26 | 36.45 (L)/8 | Vascular endothelial growth factor receptor 2 | 231 (Y) | e−13; EFN76191.1; Harpegnathos saltator | comp23754_c0 | KU663641 |
| Cc-Ven27 | 36.81 (L)/11 | Mesencephalic astrocyte-derived neurotrophic factor | 842 (Y) | 3.82e−66; BAM18078.1; Papilio xuthus | comp10689_c0 | - |
| Cc-Ven28 | 59.91 (L)/1 | DnaJ homolog subfamily C member 10-like | 2650 (Y) | 0; XP_001606269.2; Nasonia vitripennis | comp32038_c0 | - |
| Cc-Ven29 | 64.12 (L)/3 | DnaJ homolog subfamily B member 11-like | 1665 (Y) | 0; XP_624603.2; Apis mellifera | comp45528_c0 | - |
| Cc-Ven30 | 67.36 (L)/6 | Endoplasmic reticulum protein ERp29 | 987 (Y) | 3.36e−103; EGI59372.1; Acromyrmex echinatior | comp32086_c0 | - |
| Cc-Ven31 | 92.63 (L)/1 | Endoplasmic reticulum resident protein 44 | 1350 (Y) | 0; XP_624571.2; Apis mellifera | comp11316_c0 | - |
| Cc-Ven32 | 101.69 (M)/52 | Heat shock 70 kDa protein | 2840 (Y) | 0; EFN61604.1; Camponotus floridanus | comp35875_c0 | - |
| Cc-Ven33 | 72.01 (L)/4 | Unknown | 357 (Y) | - | comp38220_c0 | KU663642 |
| Cc-Ven34 | 84.75 (L)/1 | Unknown | 132 (Y) | - | comp44992_c0 | KU663643 |
| Cc-Ven35 | 270.62 (M)/4 | Unknown | 516 (Y) | - | comp23465_c0 | KU663644 |
| Cc-Ven36 | 1465.76 (H)/20 | Unknown | 291 (Y) | - | comp39041_c2 | KU663645 |
| Cc-Ven37 | 1659.20 (H)/43 | Unknown | 1033 (Y) | comp39175_c0 | KU663646 | |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Teng, Z.-W.; Xiong, S.-J.; Xu, G.; Gan, S.-Y.; Chen, X.; Stanley, D.; Yan, Z.-C.; Ye, G.-Y.; Fang, Q. Protein Discovery: Combined Transcriptomic and Proteomic Analyses of Venom from the Endoparasitoid Cotesia chilonis (Hymenoptera: Braconidae). Toxins 2017, 9, 135. https://doi.org/10.3390/toxins9040135
Teng Z-W, Xiong S-J, Xu G, Gan S-Y, Chen X, Stanley D, Yan Z-C, Ye G-Y, Fang Q. Protein Discovery: Combined Transcriptomic and Proteomic Analyses of Venom from the Endoparasitoid Cotesia chilonis (Hymenoptera: Braconidae). Toxins. 2017; 9(4):135. https://doi.org/10.3390/toxins9040135
Chicago/Turabian StyleTeng, Zi-Wen, Shi-Jiao Xiong, Gang Xu, Shi-Yu Gan, Xuan Chen, David Stanley, Zhi-Chao Yan, Gong-Yin Ye, and Qi Fang. 2017. "Protein Discovery: Combined Transcriptomic and Proteomic Analyses of Venom from the Endoparasitoid Cotesia chilonis (Hymenoptera: Braconidae)" Toxins 9, no. 4: 135. https://doi.org/10.3390/toxins9040135
APA StyleTeng, Z.-W., Xiong, S.-J., Xu, G., Gan, S.-Y., Chen, X., Stanley, D., Yan, Z.-C., Ye, G.-Y., & Fang, Q. (2017). Protein Discovery: Combined Transcriptomic and Proteomic Analyses of Venom from the Endoparasitoid Cotesia chilonis (Hymenoptera: Braconidae). Toxins, 9(4), 135. https://doi.org/10.3390/toxins9040135






