Multiple Stressors at the Land-Sea Interface: Cyanotoxins at the Land-Sea Interface in the Southern California Bight
Abstract
:1. Introduction
2. Results
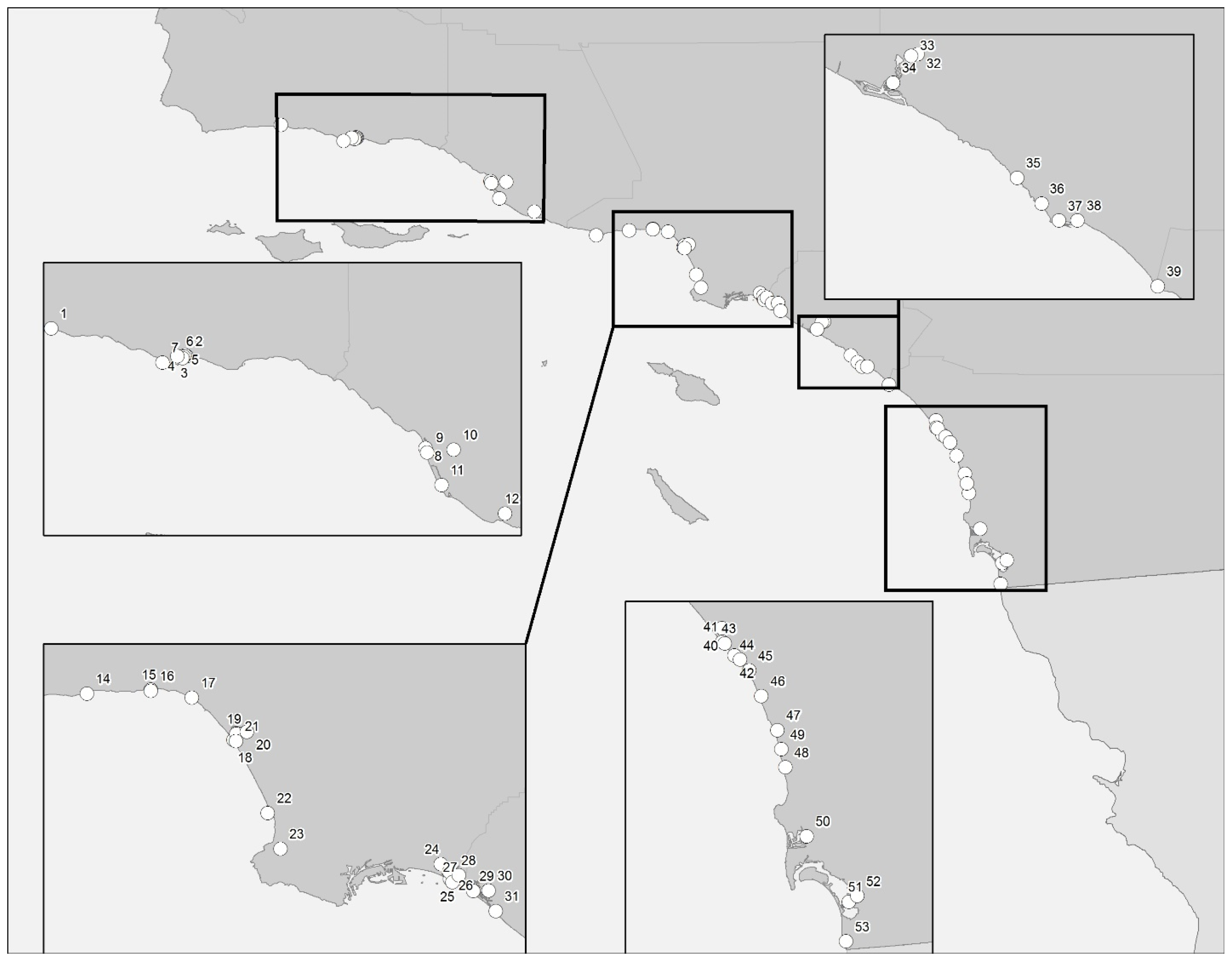
2.1. Santa Barbara/Ventura Counties
2.2. Los Angeles County
2.3. Orange County
2.4. San Diego County
3. Discussion
4. Methods
4.1. Sample Collection and Sites
4.2. Cyanobacterial Taxonomy
4.3. Cyanotoxin Screening/Assessment
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Carmichael, W.W. Cyanobacteria secondary metabolites—The cyanotoxins. J. Appl. Bact. 1992, 72, 445–459. [Google Scholar] [CrossRef]
- Harada, K. Laboratory Analysis of Cyanotoxins. In Toxic Cyanobacteria in Water: A Guide to Their Public Health Consequences, Monitoring, and Management; Chorus, I., Bartram, J., Eds.; E & FN Spon: London, UK, 1999; pp. 369–405. [Google Scholar]
- Brooks, B.W.; Lazorchak, J.M.; Howard, M.D.A.; Johnson, M.V.; Morton, S.L.; Perkins, D.A.; Reavie, E.D.; Scott, G.I.; Smith, S.A.; Steevens, J.A. Are harmful algal blooms becoming the greatest inland water quality threat to public health and aquatic ecosystems? Environ. Toxicol. Chem. 2016, 35, 6–13. [Google Scholar] [CrossRef] [PubMed]
- Paerl, H.W.; Huisman, J. Climate change: A catalyst for global expansion of harmful cyanobacterial blooms. Environ. Microbiol. Rep. 2009, 1, 27–37. [Google Scholar] [CrossRef] [PubMed]
- Kosten, S.; Huszar, V.L.M.; Bécares, E.; Costa, L.S.; van Donk, E.; Hansson, L.-A.; Jeppesen, E.; Kruk, C.; Lacerot, G.; Mazzeo, N.; et al. Warmer climates boost cyanobacterial dominance in shallow lakes. Glob. Chang. Biol. 2012, 18, 118–126. [Google Scholar] [CrossRef]
- Paerl, H.W.; Paul, V.J. Climate change: Links to global expansion of harmful cyanobacteria. Water Res. 2012, 46, 1349–1363. [Google Scholar] [CrossRef] [PubMed]
- Loftin, K.A.; Graham, J.L.; Hiborn, E.D.; Lehmann, S.C.; Meyer, M.T.; Dietze, J.E.; Griffith, C.B. Cyanotoxins of inland lakes of the United States—Occurrence and potential recreational health risks in the EPA National Lakes Assessment 2007. Harmful Algae 2016, 56. [Google Scholar] [CrossRef] [PubMed]
- Backer, L.; Carmichael, W.; Kirkpatrick, B.; Williams, C.; Irvin, M.; Zhou, Y.; Johnson, T.; Nierenberg, K.; Hill, V.; Kieszak, S.; Cheng, Y.-S. Recreational exposure to low concentrations of microcystins during an algal bloom in a small lake. Mar. Drugs 2008, 6, 389–406. [Google Scholar] [CrossRef] [PubMed]
- Ibelings, B.W.; Havens, K.E. Cyanobacterial toxins: A qualitative meta-analysis of concentrations, dosage and effects in freshwater, estuarine and marine biota. Adv. Exp. Med. Biol. 2008, 619, 675–732. [Google Scholar] [PubMed]
- WHO. Guidelines for Drinking-Water Quality, 2nd ed.; Health Criteria and Other Supporting Information; World Health Organization: Geneva, Switzerland, 1998; Volume 2. [Google Scholar]
- WHO. Recommendations for Public Water Systems to Manage Cyanotoxins in Drinking Water; World Health Organization: Geneva, Switzerland, 2015. [Google Scholar]
- U.S. EPA. Cyanobacteria and Cyanotoxins: Information for Drinking Water Systems Fact Sheet; EPA-810F11001; Office of Water: Washington, DC, USA, 2014.
- Codd, G.A.; Metcalf, J.S.; Beattie, K.A. Retention of Microcystis aeruginosa and microcystin by salad lettuce (Lactuca sativa) after spray irrigation with water containing cyanobacteria. Toxicon 1999, 37, 1181–1185. [Google Scholar] [CrossRef]
- Codd, G.A. Cyanobacterial toxins, the perception of water quality, and the priorisation of eutrophication control. Ecol. Eng. 2000, 16, 51–60. [Google Scholar] [CrossRef]
- Carmichael, W.W. Health effects of toxin producing cyanobacteria: “The CyanoHABS”. Hum. Ecol. Risk Assess. 2001, 7, 1393–1407. [Google Scholar] [CrossRef]
- Drobac, D.; Tokodi, N.; Lujic, J.; Marinovic, Z.; Subakov-Simic, G.; Dulic, T.; Vazic, T.; Nybom, S.; Meriluoto, J.; Codd, G.A.; et al. Cyanobacteria and cyanotoxins in fish ponds and their effects on fish tissue. Harmful Algae 2016, 55, 66–76. [Google Scholar] [CrossRef] [PubMed]
- Backer, L.C.; Miller, M. Sentinel animals in a one health approach to harmful cyanobacterial and algal blooms. Vet. Sci. 2016, 3, 8. [Google Scholar] [CrossRef] [PubMed]
- Rinehart, K.L.; Harada, K.; Namikoshi, M.; Chen, C.; Harvis, C.A. Nodularin, microcystin-LR and the configuration of Adda. J. Am. Chem. Soc. 1988, 110, 8557–8558. [Google Scholar] [CrossRef]
- Tanabe, Y.; Kaya, K.; Watanabe, M.M. Evidence for recombination in the microcystin synthetase (mcy) genes of toxic cyanobacteria Microcystis spp. Mol. Evol. 2004, 58, 633–641. [Google Scholar] [CrossRef] [PubMed]
- Dittmann, E.; Fewer, D.P.; Neilan, B.A. Cyanobacterial toxins: Biosynthetic routes and evolutionary roots. FEMS 2013, 37, 23–43. [Google Scholar] [CrossRef] [PubMed]
- Metcalf, J.S.; Banack, S.A.; Lindsay, J.; Morrison, L.F.; Cox, P.A.; Codd, G.A. Co-occurrence of β-N-methylamino-l-alanine, a neurotoxic amino acid with other cyanobacterial toxins in British waterbodies, 1990–2004. Environ. Microbiol. 2008, 10, 702–708. [Google Scholar] [CrossRef] [PubMed]
- Gorham, P.R. Toxic algae. In Algae and Man; Jackson, D., Ed.; Plenum Publishing Corp.: New York, NY, USA, 1964; pp. 307–336. [Google Scholar]
- Havens, K.E. Cyanobacteria blooms: Effects on aquatic ecosystems. Adv. Exp. Med. Biol. 2008, 619, 733–747. [Google Scholar] [PubMed]
- Zilius, M.; Bartoli, M.; Mariano Bresciani, M.; Katarzyte, M.; Ruginis, T.; Petkuviene, J.; Lubiene, I.; Giardino, C.; Bukaveckas, P.A.; de Wit, R.; et al. Feedback mechanisms between cyanobacterial blooms, transient hypoxia, and benthic phosphorus regeneration in shallow coastal environments. Estuaries Coasts 2014, 37, 680–694. [Google Scholar] [CrossRef]
- Horner, R.A.; Garrison, D.L.; Plumley, F.G. Harmful algal blooms and red tide problems on the U.S. west coast. Limnol. Oceanogr. 2004, 42, 1076–1088. [Google Scholar] [CrossRef]
- Trainer, V.L.; Bates, S.S.; Lundholm, N.; Thessen, A.E.; Cochlan, W.P.; Adams, N.G.; Trick, C.G. Pseudo-nitzschia physiological ecology, phylogeny, toxicity, monitoring and impacts on ecosystem health. Harmful Algae 2012, 14, 271–300. [Google Scholar] [CrossRef]
- Lehman, P.W.; Boyer, G.; Hall, C.; Waller, S.; Gehrts, K. Distribution and toxicity of a new colonial Microcystis aeruginosa bloom in the San Francisco Bay Estuary, California. Hydrobiologia 2005, 541, 87–99. [Google Scholar] [CrossRef]
- Drake, J.L.; Carpenter, E.J.; Cousins, M.; Nelson, K.L.; Guido-Zarate, A.; Loftin, K. Effects of light and nutrients on seasonal phytoplankton succession in a temperate eutrophic coastal lagoon. Hydrobiologia 2010, 654, 177–192. [Google Scholar] [CrossRef]
- Kurobe, T.; Baxa, D.V.; Mioni, C.E.; Kudela, R.M.; Smythe, T.R.; Waller, S.; Chapman, A.D.; Teh, S.J. Identification of harmful cyanobacteria in the Sacramento-San Joaquin Delta and Clear Lake, California by DNA barcoding. Springerplus 2013, 2, 491. [Google Scholar] [CrossRef] [PubMed]
- Miller, M.A.; Kudela, R.M.; Mekebri, A.; Crane, D.; Oates, S.C.; Tinker, M.T.; Staedler, M.; Miller, W.A.; Toy-Choutka, S.; Dominik, C.; et al. Evidence for a novel marine harmful algal bloom: Cyanotoxin (microcystin) transfer from Land to Sea Otters. PLoS ONE 2010, 5, e12576. [Google Scholar] [CrossRef] [PubMed]
- Preece, E.P.; Moore, B.C.; Hardy, F.J. Transfer of microcystin from freshwater lakes to Puget Sound, WA and toxin accumulation in marine mussels (Mytilus trossulus). Ecotoxicol. Environ. 2015, 122, 98–105. [Google Scholar] [CrossRef] [PubMed]
- Fetscher, A.E.; Howard, M.D.A.; Stancheva, R.; Kudela, R.M.; Stein, E.D.; Sutula, M.A.; Busse, L.B.; Sheath, R.C. Wadeable streams as widespread sources of benthic cyanotoxins in California, USA. Harmful Algae 2015, 49, 105–116. [Google Scholar] [CrossRef]
- Howard, M.D.A.; Nagoda, C.; Kudela, R.; Hayashi, K.; Tatters, A.O.; Caron, D.A.; Busse, L.; Brown, J.; Sutula, M.; Stein, E. Microcystin prevaleance throughout lentic water bodies in Southern California. Toxins. in preparation.
- Gibble, C.M.; Kudela, R.M. Detection of persistent microcystin toxins at the land-sea interface in Monterey Bay, California. Harmful Algae 2014, 39, 146–153. [Google Scholar] [CrossRef]
- Gibble, C.M.; Peacock, M.B.; Kudela, R.M. Evidence of freshwater algal toxins in marine shellfish: Implications for human and aquatic health. Harmful Algae 2016, 59, 59–66. [Google Scholar] [CrossRef] [PubMed]
- Takahashi, T.; Umehara, A.; Tsutsumi, H. Diffusion of microcystins (cyanobacteria hepatotoxins) from the reservoir of Isahaya Bay, Japan, into the marine and surrounding ecosystems as a result of large-scale drainage. Mar. Pollut. Bull. 2014, 89, 250–258. [Google Scholar] [CrossRef] [PubMed]
- Sivonen, K.; Kononen, K.; Carmichael, W.W.; Dahlem, A.M.; Rinehart, K.L.; Kiviranta, J.; Niemela, S.I. Occurrence of the hepatotoxic cyanobacterium Nodularia spumigena in the Baltic Sea and the structure of the toxin. Appl. Environ. Microbiol. 1989, 55, 1990–1995. [Google Scholar] [PubMed]
- Repka, S.; Koivula, M.; Harjunpa, V.; Rouhiainen, L.; Sivonen, K. Effects of phosphate and light on growth of and bioactive peptide production by the cyanobacterium Anabaena strain 90 and its anabaenopeptilide mutant. Appl. Environ. Microbiol. 2004, 70, 4551–4560. [Google Scholar] [CrossRef] [PubMed]
- Falconer, I.R.; Choice, A.; Hosja, W. Toxicity of edible mussels (Mytilus edulis) growing naturally in an estuary during a water bloom of the blue-green alga Nodularia spumigena. Environ. Toxicol. Water Qual. 1992, 7, 119–123. [Google Scholar] [CrossRef]
- Vareli, K.; Jaeger, W.; Touka, A.; Frillingos, S.; Briasoulis, E.; Sainis, I. Hepatotoxic seafood poisoning (HSP) due to microcystins: A threat from the ocean? Mar. Drugs 2013, 11, 2751–2768. [Google Scholar] [CrossRef] [PubMed]
- Kudela, R.M. The Year of Crazy. In Proceedings of the Conference Presentation, 8th Symposium on HABs in the U.S., Long Beach, CA, USA, November 2015.
- Mulvenna, V.; Dale, K.; Priestly, B.; Mueller, U.; Humpage, A.; Shaw, G.; Allinson, G.; Falconer, I. Health risk assessment for cyanobacterial toxins in seafood. J. Environ. Res. Public Health 2012, 9, 807–820. [Google Scholar] [CrossRef] [PubMed]
- Scholin, C.A.; Gulland, F.; Doucette, G.J.; Benson, S.; Busman, M.; Chavez, F.P.; Cordaro, J.; DeLong, R.; De Vogelaere, A.; Harvey, J.; et al. Mortality of sea lions along the central California coast linked to a toxic diatom bloom. Nature 2000, 403, 80–84. [Google Scholar] [CrossRef] [PubMed]
- Bargu, S.; Goldstein, T.; Roberts, K.; Li, C.; Gulland, F. Pseudo-nitzschia blooms, domoic acid, and related California sea lion strandings in Monterey Bay, California. Mar. Mamm. Sci. 2012, 28, 237–253. [Google Scholar] [CrossRef]
- MacKenzie, L.; Beuzenberg, V.; Holland, P.; McNabb, P.; Selwood, A. Solid phase adsorption toxin tracking (SPATT): A new monitoring tool that simulates the biotoxin contamination of filter feeding bivalves. Toxicon 2004, 44, 901–918. [Google Scholar] [CrossRef] [PubMed]
- Lane, J.Q.; Roddam, C.M.; Langlois, G.W.; Kudela, R.M. Application of Solid Phase Adsorption Toxin Tracking (SPATT) for field detection of the hydrophilic phycotoxins domoic acid and saxitoxin in coastal California. Limnol. Oceanogr. Methods 2010, 8, 645–660. [Google Scholar] [CrossRef]
- Rapala, J.; Sivonen, K.; Lyra, C.; Niemelae, S.I. Variation of microcystins, cyanobacterial hepatotoxins, in Anabaena spp. as a function of growth stimuli. Appl. Environ. Microbiol. 1997, 63, 2206–2212. [Google Scholar] [PubMed]
- Chorus, I.; Bartram, J. Toxic Cyanobacteria in Water; A Guide to Their Public Health Consequences, Monitoring and Management; E & FN Spon: London, UK, 1999. [Google Scholar]
- Davis, T.W.; Berry, D.L.; Boyer, G.L.; Gobler, C.J. The effects of temperature and nutrients on the growth and dynamics of toxic and non-toxic strains of Microcystis during cyanobacteria blooms. Harmful Algae 2009, 8, 715–725. [Google Scholar] [CrossRef]
- Paerl, H.W.; Otten, T.G. Duelling ‘CyanoHABs’: Unravelling the environmental drivers controlling dominance and succession among diazotrophic and non-N2-fixing harmful cyanobacteria. Environ. Microbiol. 2015, 18, 316–324. [Google Scholar] [CrossRef] [PubMed]
- Carmichael, W.W.; Gorham, P.R. The mosaic nature of toxicity in cyanobacteria blooms. In The Water Environment: Algal Toxins and Health; Carmichael, W.W., Ed.; Plenum Press: New York, NY, USA, 1981; pp. 161–172. [Google Scholar]
- Paerl, H.W. Microscale physiological and ecological studies of aquatic cyanobacteria: Macroscale implications. Microsc. Res. Tech. 1996, 33, 47–72. [Google Scholar] [CrossRef]
- Briand, E.; Escoffier, N.; Straub, C.; Sabart, M.; Quiblier, C.; Humbert, J. Spatiotemporal changes in the genetic diversity of a bloom-forming Microcystis aeruginosa (cyanobacteria) population. ISME J. 2009, 3, 419–429. [Google Scholar] [CrossRef] [PubMed]
- Cai, Y.; Kong, F.; Shi, L.; Yu, Y. Spatial heterogeneity of cyanobacterial communities and genetic variation of Microcystis populations within large, shallow eutrophic lakes (Lake Taihu and Lake Chaohu, China). J. Environ. Sci. 2012, 24, 1832–1842. [Google Scholar] [CrossRef]
- Anderson, D.M.; Burkholder, J.M.; Cochlan, W.P.; Glibert, P.M.; Gobler, C.J.; Heil, C.A.; Kudela, R.M.; Parsons, M.L.; Rensel, J.E.J.; et al. Harmful algal blooms and eutrophication: Examining linkages from selected coastal regions of the United States. Harmful Algae 2008, 8, 39–53. [Google Scholar] [CrossRef] [PubMed]
- Howard, M.D.A.; Sutula, M.; Caron, D.A.; Chao, Y.; Farrara, J.D.; Frenzel, H.; Jones, B.; Robertson, G.; McLaughlin, K.; Sengupta, A. Anthropogenic nutrient sources rival natural sources on small scales in the coastal waters of the Southern California Bight. Limnol. Oceanogr. 2014, 59, 285–297. [Google Scholar] [CrossRef]
- Pekar, H.; Westerberg, E.; Bruno, O.; Laane, A.; Persson, K.M.; Sundstrom, L.F.; Thim, A.M. Fast, rugged and sensitive ultra high pressure liquid chromatography tandem mass spectrometry method for analysis of cyanotoxins in raw water and drinking water-First findings of anatoxins, cylindrospermopsins and microcystin variants in Swedish source waters and infiltration ponds. J. Chromatogr. A 2016, 1429, 265–276. [Google Scholar]
- Graham, J.L.; Loftin, K.A.; Meyer, M.T.; Ziegler, A.C. Cyanotoxin mixtures and taste-and-odor compounds in cyanobacterial blooms from the Midwestern United States. Environ. Sci. Technol. 2010, 44, 7361–7368. [Google Scholar] [CrossRef] [PubMed]
- Glekis, S.; Zaoutsos, N. Cyanotoxin occurrence and potentially toxin producing cyanobacteria in freshwaters of Greece: A multi-disciplinary approach. Toxicon 2014, 78, 1–9. [Google Scholar]
- Sabart, M.; Crenn, K.; Perriere, F.; Abila, A.; Leremboure, M.; Colombet, J.; Jousse, C.; Latour, D. Co-occurrence of microcystin and anatoxin-a in the freshwater lake Aydat (France): Analytical and molecular approaches during a three-year survey. Harmful Algae 2015, 48, 12–20. [Google Scholar] [CrossRef]
- Jancula, D.; Strakova, L.; Sadilek, J.; Marsalek, B.; Babica, P. Survey of cyanobacterial toxins in Czech water reservoirs - the first observation of neurotoxic saxitoxins. Environ. Sci. Pollut. Res. 2014, 21, 8006–8015. [Google Scholar] [CrossRef] [PubMed]
- Bittencourt-Oliveira, M.C.; Hereman, T.C.; Cordeiro-Araújo, M.K.; Macedo-Silva, I.; Dias, C.T.; Sasaki, F.F.; Moura, A.N. Phytotoxicity associated to microcystins: A review. Braz. J. Biol. 2014, 74, 753–760. [Google Scholar] [CrossRef] [PubMed]
- Borges, H.L.F.; Branco, L.H.Z.; Martins, M.D.; Lima, C.S.; Barbosa, P.T.; Lira, G.A.S.T.; Bittencourt-Oliveria, M.C.; Molica, R.J.R. Cyanotoxin production and phylogeny of benthic cyanobacterial strains isolated from the northeast of Brazil. Harmful Algae 2015, 43, 46–57. [Google Scholar] [CrossRef]
- Rodriguez, I.; Rodriguez, C.; Alfonso, A.; Otero, P.; Meyer, T.; Breitenbach, U.; Botana, L.M. Toxin profile in samples collected in fresh and brackish water in Germany. Toxicon 2014, 91, 35–44. [Google Scholar] [CrossRef] [PubMed]
- Komárek, J.; Komárková, J. Review of the European Microcystis-morphospecies (Cyanoprokaryotes) from nature. Czech Phycol. 2002, 2, 1–24. [Google Scholar]
- Komárek, J.; Komárková, J. Phenotype diversity of the cyanoprokaryotic genus Cylindrospermopsis (Nostocales); review 2002. Czech Phycol. 2003, 3, 1–30. [Google Scholar]
- Komárek, J.; Komárková, J. Taxonomic review of the cyanoprokaryotic genera Planktothrix and Planktothricoides. Czech Phycol. 2004, 4, 1–18. [Google Scholar]
- Komárek, J.; Zapomĕlová, E. Planktic morphospecies of the cyanobacterial genus Anabaena = subg. Dolichospermum—1. Part: Coiled types. Fottea 2007, 7, 1–31. [Google Scholar]
- Komárek, J.; Jerzerová, J.; Komárek, O.; Zapomĕlová, E. Variability of Chroococcus (Cyanobacteria) morphospecies with regard to phylogenetic relationships. Hydrobiologia 2010, 639, 69–83. [Google Scholar] [CrossRef]
- Komárek, J. Diversita a Moderní Klasifikace Sinic (Cyanoprocaryota) [Diversity and Modern Classification of Cyanobacteria (Cyanoprokaryota). Dissertation, 1992. [Google Scholar]
- Rajaniemi, P.; Hrouzek, P.; Kaštovská, K.; Willame, R.; Rantala, A.; Hoffmann, L.; Komárek, J.; Sivonen, K. Phylogenetic and morphological evaluation of the genera Anabaena, Aphanizomenon, Trichormus and Nostoc (Nostocales, Cyanobacteria). Int. J. Syst. Evol. Microbiol. 2005, 55, 11–26. [Google Scholar] [CrossRef] [PubMed]
- Welschmeyer, N.A. Fluorometric analysis of chlorophyll a in the presence of chlorophyll b and pheopigments. Limnol. Oceanogr. 1994, 39, 1985–1992. [Google Scholar] [CrossRef]
- Harada, K.I.; Ohtani, I.; Iwamoto, K.; Suzuki, M.; Watanabe, M.F.; Watanabe, M.; Terao, K. Isolation of cylindrospermopsin from a cyanobacterium Umezakia natans and its screening method. Toxicon 1994, 32, 73–84. [Google Scholar] [CrossRef]
- Spoof, L.; Neffling, M.R.; Meriluoto, J. Fast separation of microcystins and nodularins on narrow-bore reversed-phase columns coupled to a conventional HPLC system. Toxicon 2010, 55, 954–964. [Google Scholar] [CrossRef] [PubMed]
- James, K.J.; Sherlock, I.R.; Stack, M.A. Anatoxin-a in Irish freshwater and cyanobacteria, determined using a new fluorimetric liquid chromatographic method. Toxicon 1997, 35, 963–971. [Google Scholar] [CrossRef]
- James, K.J.; Furey, A.; Sherlock, I.R.; Stack, M.A.; Twohig, M.; Caudwell, F.B.; Skulberg, O.M. Sensitive determination of anatoxin-a, homoanatoxin-a and their degradation products by liquid chromatography with fluorimetric detection. J. Chromatogr.A 1998, 798, 147–157. [Google Scholar] [CrossRef]
- Tatters, A.O.; Fu, F.-X.; Hutchins, D.A. High CO2 and silicate limitation synergistically increase the toxicity of Pseudo-nitzschia fraudulenta. PLoS ONE 2012, 7, e32116. [Google Scholar] [CrossRef] [PubMed]

| Locality | Aphanizomenon | Aphanocapsa | Aphanothece | Anabaena | Calothrix | Chroococcus | Cylindrospermum | Cylindrospermopsis | Dolichospermum | Geitlerinema | Gloeocapsa | Heteroleibleinia | Leibleinia | Lyngbya | Leptolyngbya | Merismopedia | Microcystis | Nodularia | Nostoc | Oscillatoria | Phormidium | Planktolyngbya | Topolythrix |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Arroyo Hondo | C | A | AC | A | |||||||||||||||||||
| Atascadero Creek | AC | C | P | ||||||||||||||||||||
| San Jose Creek | D | R | |||||||||||||||||||||
| San Pedro Creek | P | A | A | ||||||||||||||||||||
| Tecolotito Creek | D | ||||||||||||||||||||||
| Goleta Slough | A | C | R | R | P | P | C | R | P | ||||||||||||||
| Devereaux Slough | D | ||||||||||||||||||||||
| Ventura Harbor | P | A | A | ||||||||||||||||||||
| Santa Clara River Estuary | P | C | A | P | A | R | |||||||||||||||||
| Santa Clara River | PP | P | P | A | |||||||||||||||||||
| Channel Islands Harbor | D | ||||||||||||||||||||||
| Calleguas Creek | A | A | A | ||||||||||||||||||||
| Zuma Lagoon | P | D | A | ||||||||||||||||||||
| Malibu Lagoon | PPR | P | A | ||||||||||||||||||||
| Topanga Creek | P | P | P | P | P | P | P | CC | C | ||||||||||||||
| Topanga Lagoon | A | P | P | P | C | P | DP | ||||||||||||||||
| Rustic Creek | |||||||||||||||||||||||
| Ballona Lagoon | R | D | R | ||||||||||||||||||||
| Marina Del Rey | R | P | D | ||||||||||||||||||||
| Ballona Creek | C | P | D | ||||||||||||||||||||
| Del Rey Lagoon | P | D | P | P | R | ||||||||||||||||||
| King Harbor | |||||||||||||||||||||||
| Malaga Creek | P | D | A | ||||||||||||||||||||
| Colorado Lagoon | |||||||||||||||||||||||
| Alamitos Bay | |||||||||||||||||||||||
| Mother's Beach | D | A | |||||||||||||||||||||
| San Gabriel | |||||||||||||||||||||||
| San Gabriel up | |||||||||||||||||||||||
| Seal Beach | |||||||||||||||||||||||
| Huntington Harbour | D | ||||||||||||||||||||||
| Bolsa Chica Channel | D | ||||||||||||||||||||||
| Upper Newport | D | P | A | ||||||||||||||||||||
| Back Bay | D | C | A | P | |||||||||||||||||||
| Aliso Creek | P | D | P | P | |||||||||||||||||||
| Salt Creek | |||||||||||||||||||||||
| Dana Point Harbor | D | ||||||||||||||||||||||
| San Juan Creek | P | DAC | |||||||||||||||||||||
| San Mateo Creek | |||||||||||||||||||||||
| Santa Margarita | P | A | R | R | A | P | |||||||||||||||||
| Oceanside Harbor | |||||||||||||||||||||||
| San Luis Rey River | A | D | |||||||||||||||||||||
| Buena Vista Creek/Lagoon | D | A | CC | ||||||||||||||||||||
| Agua Hendionda | D | ||||||||||||||||||||||
| Batiquitos Lagoon | A | C | C | A | P | P | |||||||||||||||||
| San Elijo Lagoon | A | D | P | ||||||||||||||||||||
| Los Penasquitos Lagoon | P | D | |||||||||||||||||||||
| San Dieguito River | D | ||||||||||||||||||||||
| San Diego Creek | A | R | R | P | C | D | |||||||||||||||||
| Loma Alta Creek | R | P | P | A | P | A | |||||||||||||||||
| Mission Bay | P | C | D | P | |||||||||||||||||||
| San Diego Bay | C | D | |||||||||||||||||||||
| Tijuana River/Estuary | A | A | P | P | P | ||||||||||||||||||
| Sweetwater River | R | P | P | A | A |
| Locality | Anatoxins | Cylindro-Spermopsins | Microcystins | Saxitoxins | Nodularins |
|---|---|---|---|---|---|
| Arroyo Hondo | + | − | − | − | − |
| Atascadero Creek | + | − | + | − | − |
| San Jose Creek | − | − | − | − | − |
| San Pedro Creek | − | − | − | − | − |
| Tecolito Creek | + | − | − | − | − |
| Goleta Slough | + | − | + | − | − |
| Devereaux Slough | − | − | − | − | − |
| Ventura Harbor | − | − | − | − | − |
| Santa Clara River Est/Lagoon | + | − | − | + | − |
| Santa Clara River | − | + | + | − | − |
| Channel Islands Harbor | − | − | − | − | − |
| Calleguas Creek | − | + | − | + | − |
| Zuma Lagoon | − | − | − | − | − |
| Malibu Lagoon | + | − | − | − | − |
| Topanga Creek | + | − | + | − | − |
| Topanga Lagoon | − | − | + | − | − |
| Rustic Creek | − | − | − | − | − |
| Ballona Lagoon | − | − | − | + | − |
| Marina Del Rey | − | − | − | − | − |
| Ballona Creek | − | − | + | − | − |
| Del Rey Lagoon | − | − | − | − | − |
| King Harbor | − | − | − | − | − |
| Malaga Creek | − | − | − | − | − |
| Colorado Lagoon | − | − | − | − | − |
| Alamitos Bay | − | − | − | − | − |
| Mother's Beach | − | − | − | − | − |
| San Gabriel River | − | − | − | − | − |
| San Gabriel River upstream | − | − | − | − | − |
| Seal Beach | − | − | − | − | − |
| Huntington Harbour | − | − | − | − | − |
| Bolsa Chica Channel/Basin | − | − | − | − | − |
| Upper Newport | − | − | − | − | − |
| Back Bay | − | − | + | − | − |
| Aliso Creek | − | − | − | − | − |
| Salt Creek | − | − | − | − | − |
| Dana Point Harbor | − | − | − | − | − |
| San Juan Creek | + | − | − | + | − |
| San Mateo Creek | − | − | − | − | − |
| Santa Margarita | + | − | + | + | + |
| Oceanside Harbor | − | − | − | − | − |
| San Luis Rey River | − | − | − | − | − |
| Buena Vista Creek/Lagoon | + | + | + | − | − |
| Agua Hendionda | − | − | − | − | − |
| Batiquitos Lagoon | + | − | + | + | − |
| San Elijo Lagoon | − | − | + | + | − |
| Los Penasquitos | − | − | − | − | − |
| San Dieguito River | − | − | − | − | − |
| San Diego Creek | − | − | − | − | − |
| Loma Alta Creek | − | − | + | + | − |
| Mission Bay | − | − | − | − | − |
| San Diego Bay | − | − | − | − | − |
| Tijuana River/Estuary | − | − | + | − | − |
| Sweetwater River | − | − | + | + | − |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tatters, A.O.; Howard, M.D.A.; Nagoda, C.; Busse, L.; Gellene, A.G.; Caron, D.A. Multiple Stressors at the Land-Sea Interface: Cyanotoxins at the Land-Sea Interface in the Southern California Bight. Toxins 2017, 9, 95. https://doi.org/10.3390/toxins9030095
Tatters AO, Howard MDA, Nagoda C, Busse L, Gellene AG, Caron DA. Multiple Stressors at the Land-Sea Interface: Cyanotoxins at the Land-Sea Interface in the Southern California Bight. Toxins. 2017; 9(3):95. https://doi.org/10.3390/toxins9030095
Chicago/Turabian StyleTatters, Avery O., Meredith D.A. Howard, Carey Nagoda, Lilian Busse, Alyssa G. Gellene, and David A. Caron. 2017. "Multiple Stressors at the Land-Sea Interface: Cyanotoxins at the Land-Sea Interface in the Southern California Bight" Toxins 9, no. 3: 95. https://doi.org/10.3390/toxins9030095
APA StyleTatters, A. O., Howard, M. D. A., Nagoda, C., Busse, L., Gellene, A. G., & Caron, D. A. (2017). Multiple Stressors at the Land-Sea Interface: Cyanotoxins at the Land-Sea Interface in the Southern California Bight. Toxins, 9(3), 95. https://doi.org/10.3390/toxins9030095





